Abstract
Background. Understanding potential sex differences in stroke recovery is important for prognosis, ensuring appropriate allocation of health care resources, and for stratification in research studies. Previously, functional measures have shown poorer outcomes for females, however, little is known about sex differences that may exist in specific motor and sensory impairments. Objective. The aim of this study was to utilize robotic assessments of motor and sensory impairments to determine if there are sex differences at the impairment level in stroke recovery over the first 6 months poststroke. Methods. We used robotic and clinical assessments of motor and sensory impairments at 1, 6, 12, and 26 weeks poststroke in 108 males and 52 females. Linear mixed models were used to examine the effect of sex on recovery poststroke, controlling for age and lesion volume. Results. In general, we did not find significant sex differences across a range of assessments. The exception to this was a sex × age interaction for the Purdue Pegboard Assessment, where we found that females had better performance than males at younger ages (<62 years), but males had better performance at older ages. Conclusions. While recruitment biases need to be acknowledged when generalizing our results to stroke recovery at-large, our results suggest that sex differences do not exist at the impairment level poststroke.
Introduction
There has been a recent drive to address issues of sex and gender in health research. Sex refers to the biological factors that differentiate males and females, while gender encompasses socially constructed roles and behaviors. Funding agencies, including the National Institutes of Health and Canadian Institutes of Health Research, are pushing for sex and gender to be accounted for in the formulation of research questions, experimental design, data analysis, and reporting of results. These initiatives stem from the fact that women have been historically underrepresented in clinical trials,1-3 which is problematic when considering the role sex and gender can have on disease presentation and treatment outcomes.4,5 As addressing sex and gender in study design and analysis can significantly increase the complexity and expense of a research study, it is imperative for researchers to know how sex and/or gender may affect stroke recovery in order to plan accordingly.
Sex differences have been previously documented in stroke incidence and outcomes. Females have a higher lifetime risk of stroke than males, largely due to an increased risk period from a longer life expectancy.6,7 In general, studies have found that females have worse functional outcomes and more deaths due to stroke compared to males 8 and are less likely to be discharged home. 9 Poorer outcomes in females versus males have been attributed to older age at stroke onset, greater stroke severity, and lower baseline function10-12; however, sex differences in outcomes still exist after statistically adjusting for these factors. 9 In terms of treatment effects, females have been found to benefit more from intravenous thrombolysis than their male counterparts,13-15 yet there is no differential treatment effect by sex for endovascular thrombectomy. 16 The sex differences (or lack thereof) in treatment responses to acute medical therapies illustrates both the complexity and importance of understanding how treatments may affect males and females differently.
Previous research on sex differences in stroke recovery has focused on functional outcomes measured at 90 or 180 days poststroke, using such scales as the Modified Rankin Scale or Barthel Index.11,12,17-19 While functional outcomes are typically related to impairment-based measures, functional outcomes can be affected by compensations (eg, performing an activity with the opposite hand). Impairment-based measures can give insight into the underlying causes of functional limitations, differentiating between factors such as strength, coordination, and sensory impairments. If sex differences at the impairment level do exist, this would present a clear explanation of differing functional outcomes and provide an intermediary treatment target to alleviate the poorer outcomes in females. Furthermore, as many research studies are interested in understanding and treating specific poststroke impairments, determining if there are sex differences at the impairment level is crucial for the development of future studies.
The objective of this study was to utilize robotic assessments of motor and sensory impairments to determine if there are sex differences at the impairment level in stroke recovery over the first 6 months poststroke. Commonly used clinical measures of impairment and function were also examined. We hypothesized that sex differences would not exist at the impairment level, but that based on prior findings, 9 males would have better functional recovery as measured by the Functional Independence Measure compared with females. Our collective clinical experience in treating stroke patients suggested sex differences would not be present at the impairment level, but this needed to be empirically tested. Results of this study can be used to guide researchers on how to account for potential sex differences in designing future studies.
Methods
Participants
Participants were selected from the larger RESTART study, which is a prospective longitudinal study using clinical and robotic measures to track recovery at 1, 6, 12, and 26 weeks poststroke. From the larger study, participants were included if they met the following inclusion criteria: had a first-time unilateral clinical stroke; completed at least 3 of the 4 possible timepoints; had magnetic resonance imaging (MRI) or computed tomography (CT) imaging with a defined acute lesion; and were older than 18 years. Participants were excluded if they had bilateral, brainstem, or cerebellar stroke; preexisting neurologic disease; upper extremity orthopedic conditions; evidence of apraxia based on screening with the TULIA (Test of Upper Limb Apraxia) 20 ; or were unable to follow task instructions or provide informed consent. Bilateral, brainstem, or cerebellar strokes were excluded due to the nature of the robotic assessments requiring a clear “affected” and “unaffected” side, as well as difficulties with registering brainstem and cerebellar strokes to a common Montreal Neurological Institute (MNI) brain. 21
Robotic Assessments
All participants underwent robotic assessments at each timepoint using the KINARM exoskeleton robot (Kinarm, Kingston, Ontario, Canada). Participants were seated in a wheelchair base with their arms supported by troughs in the horizontal plane. Four standardized tasks were used to assess motor, sensory, and cognitive impairment. All tasks have been validated against commonly used clinical measures of impairment and function and found to correlate significantly.22-25 Visually guided reaching is a visuomotor task consisting of center-out reaches to 8 peripheral targets with a cursor displayed at the fingertip. 24 Position matching is a test of position sense, in which the stroke-affected limb is moved by the robot and the participant is instructed to mirror match the position with the other limb. 22 Object hit is a test of bimanual sensorimotor control, where participants use virtual paddles displayed at their fingertips to hit away balls that fall from the top of the screen with increasing speed and frequency over the course of the task. 23 Object hit and avoid is a variant of this task with additional cognitive demands, where participants are instructed to only hit 2 specific shapes of targets and avoid 6 shapes that serve as distractors. 25 Vision of the participants’ actual arms and hands were occluded for all tasks. The tasks are each illustrated in Figure 1.
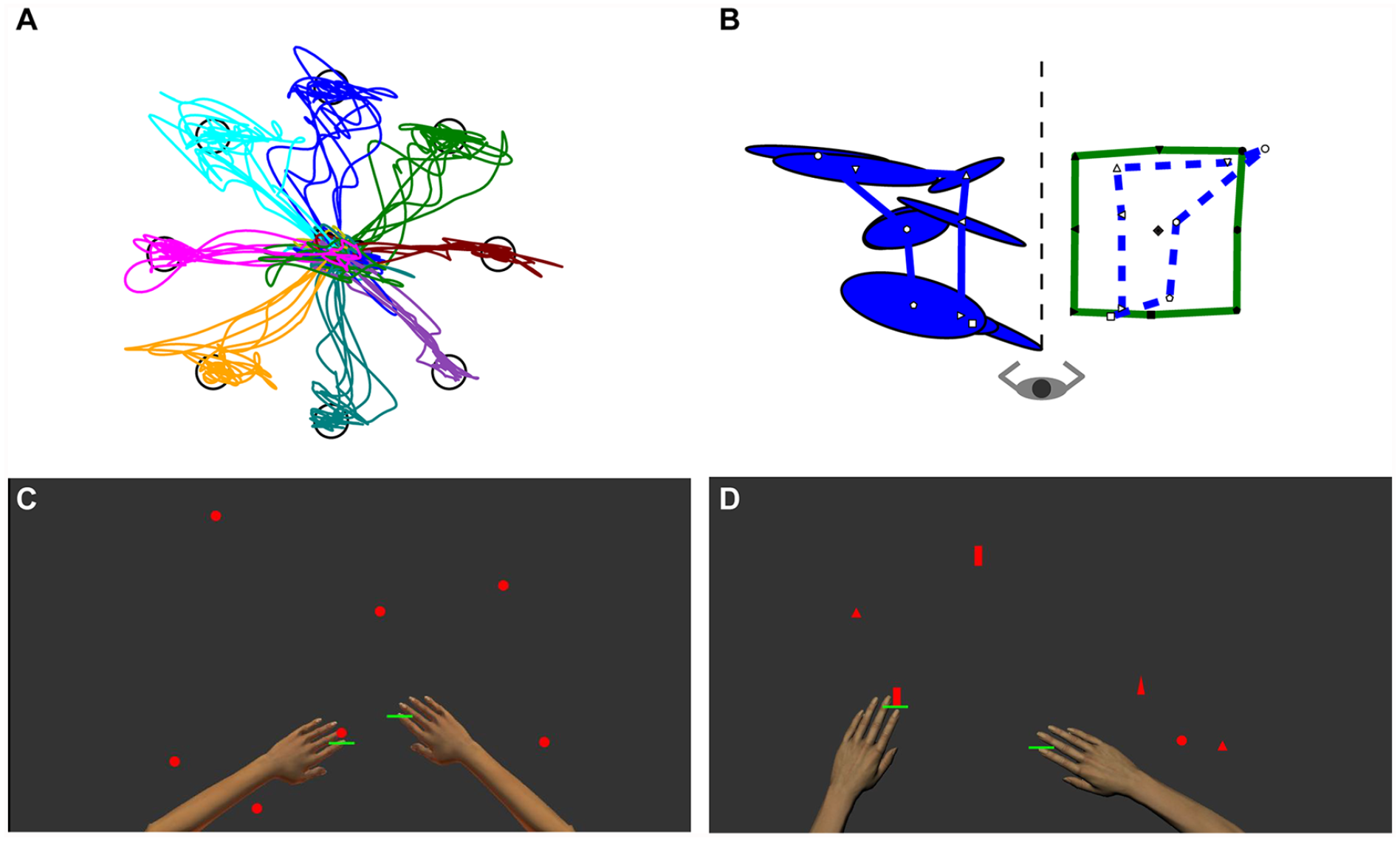
Robotic tasks in an individual with stroke. (A) Visually guided reaching: The hand paths for centre-out reaches with the stroke-affected arm to each of the 8 peripheral targets are illustrated. (B) Position matching: The robot moves the stroke-affected (in the example shown, right) limb to one of 9 targets, which are shown connected by the green rectangle. The participant then mirror matches the positions with the left arm, with the ellipses representing variability. The average matched positions are reflected onto the positions of the right arm, illustrated by the blue dotted line. (C) Object hit: Green paddles are displayed at the participant’s fingertips and the participant attempts to hit away the red balls that fall from the top of the screen. (D) Object hit and avoid: Similar to object hit, but the participant is instructed to only hit 2 specific shapes while avoiding 6 other distractor shapes. During the tasks, the arms and hands are not visible, only the green paddles.
Performance on each task is quantified using parameters that have been previously detailed,22-26 such as reaction time and initial direction angle for visually guided reaching, variability for position matching, targets hit for object hit, and distractor hits for object hit and avoid. 25 Each parameter is transformed into a z-score based on a large cohort of controls (visually guided reaching, N = 307; position matching, N = 799; object hit, N = 598; object hit and avoid, N = 509), taking into account age, sex, and handedness. Based on performance on each parameter, a single composite task score is calculated to reflect overall performance using the root-mean square distance of each parameter z-score. The composite score is then normalized based on control participants to create the task score. Task scores can be interpreted as standard deviations from the expected performance for control participants of the same age, sex, and handedness, with scores greater than 1.96 being considered abnormal. 26 Thereby, higher scores reflect worse performance. By accounting for age and sex in the task scores, we are able to examine the role of age and sex on recovery from stroke, controlling for baseline differences in behavior that might exist. For instance, full recovery for an 80-year-old female would entail them returning to the normal performance expected for an 80-year-old female, rather than what would be considered normal for a 20-year-old male.
Clinical Assessments
Participants completed a battery of clinical assessments at each timepoint that were administered and scored by a trained therapist. The Chedoke McMaster Stroke Assessment (CMSA) is an assessment of arm and hand motor impairment, with individual scores for the arm and hand components ranging from 1 (greatest impairment) to 7 (least impairment). 27 For our analysis, CMSA arm and hand scores were summed for a maximum score of 14. 27 While summing arm and hand scores has been previously done27,28 and found to be highly correlated with the Fugl-Meyer Assessment, 27 we also examined the hand and arm scores individually, as reported in Supplemental Table S3. The Purdue Pegboard Assessment is a test of manual dexterity that measures how many pegs a participant can place into holes in 30 seconds. 29 Strength testing of upper extremity muscles was also conducted, and Medical Research Council (MRC) strength scores for shoulder abductors and finger extensors were summed to produce a SAFE (Shoulder Abduction–Finger Extensor) score. The SAFE score has been previously used as a prognostic indicator for upper extremity recovery.30,31 We must note that previous use of the SAFE score is often within 72 hours poststroke, whereas our assessments were completed at approximately 1 week post-stroke. The Functional Independence Measure (FIM) is a measure of functional ability that scores the level of assistance needed to complete activities of daily living. FIM scores range from 18 (dependent in all activities of daily living) to 126 (independent in all activities). 32
Imaging
Clinical imaging was acquired based on the acute stroke protocol at the Foothills Medical Centre in Calgary, Alberta. Brain CT or MRI scans were acquired as soon as possible after stroke onset. Because these were clinical scans, variability in scan parameters existed. MRI scans were either acquired on a 1.5-T Siemens scanner or 3-T General Electric (GE) Medical Systems scanner. CTs were acquired on Siemens or GE scanners. For MRIs, echo time (TE) ranged from 98.4 to 146.9 ms and repetition time (TR) ranged from 8000 to 10 500 ms. In-plane resolution was 1 mm × 1 mm for all scans. Slice thickness ranged from 3 to 5 mm across scans with 0-mm interslice gaps.
Lesions were hand-drawn on the CT images or FLAIR (fluid-attenuated inversion recovery) MRI images using diffusion weighted images as guides. All lesions were verified by a stroke neurologist. Lesions were then normalized to the MNI template using the clinical toolbox in SPM8 (http://www.nitrc.org/projects/clinicaltbx) 33 for CT scans and linear and nonlinear toolboxes in FSL for MRI images.34,35 Normalized lesions were used to calculate lesion volume.
Prior work has suggested that measures of corticospinal tract lesion load may be more indicative of motor recovery than lesion volume alone.36-38 We therefore calculated a weighted lesion load for each participant. Using a mask of the corticospinal tract from the JHU White Matter Atlas, 39 the overlap between the lesion and the corticospinal tract was calculated for each slice in MNI space. The amount of overlap on each slice was then weighted by multiplying it by the maximum cross-sectional area of the entire tract divided by the cross-sectional area of the current slice. This weighting accounts for the denser fiber tracts through the narrower regions of the tract by weighting damage to these areas higher.40,41
Statistical Analysis
Age and lesion volumes were compared between males and females using t test and Mann-Whitney U test, respectively. Linear mixed effects models were used to assess the effects of sex, age, and lesion volume on longitudinal recovery. While the primary interest of this study was sex differences, age and lesion volume were also included as fixed effects since prior work has shown these factors may explain differences in males and females.10,11 Random slope and intercept models were used with fixed main effects of time poststroke (in weeks), sex (0 for males, 1 for females), age (in years), and lesion volume (in cm3). Based on visual inspection of recovery curves, time was modeled as a quadratic function (time 2 + time). The interaction of sex × time was examined to see if the recovery curves were different between males and females. Additionally, the interaction of sex × age was tested to determine if potential sex differences were age dependent. If the sex × age interaction was not significant, the model was repeated with the interaction removed to facilitate interpretation of the main effects. An interaction of sex × lesion volume (or lesion load) was tested but removed from the final model if not significant. We also tested the models with affected arm included as a fixed effect but removed from the final model if not significant. Models were repeated with lesion load instead of lesion volume to examine if there was an effect of damage specifically to the corticospinal tracts. All statistical analyses were conducted using MATLAB 2015a (Mathworks). A P value of .05 was used to indicate significance. Fixed effects coefficients are reported as the estimate and 95% confidence interval. The assumptions of linear mixed-effects models were checked. In the case that residuals were not normal, transformations were attempted and noted in the results.
Results
Participant Demographics
In total, 160 participants were included, consisting of 108 males and 52 females. Participant demographics are shown in Table 1. Of our participants, 73 males and 33 females completed all 4 time points. There were no significant differences in age (P = .44) or lesion volume (Mann-Whitney U = 2674.5, P = .627) between males and females. A heat map of the lesion locations can be found in Supplemental Figure S1.
Demographic Information and Clinical Scores. a
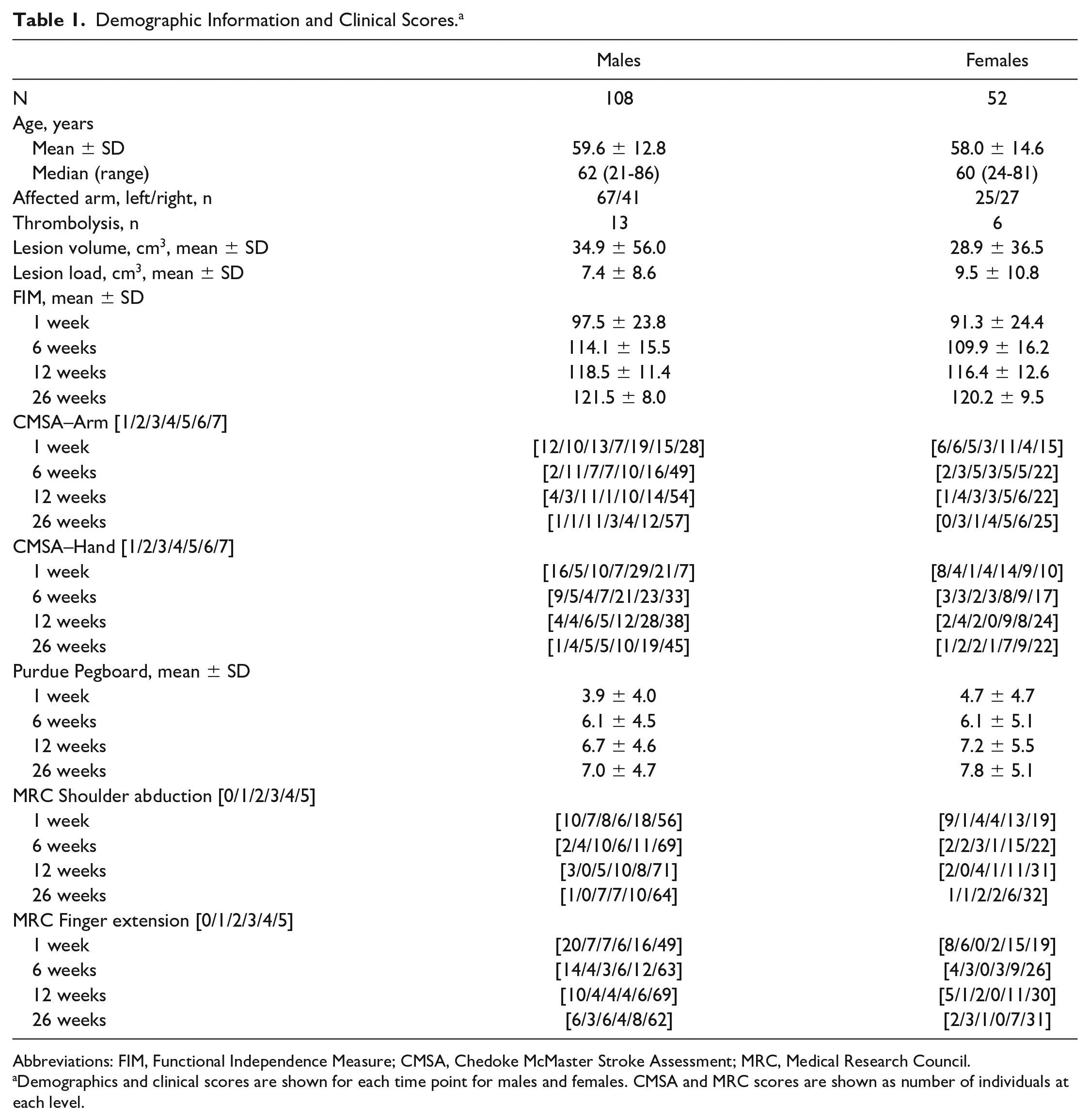
Abbreviations: FIM, Functional Independence Measure; CMSA, Chedoke McMaster Stroke Assessment; MRC, Medical Research Council.
Demographics and clinical scores are shown for each time point for males and females. CMSA and MRC scores are shown as number of individuals at each level.
Robotic Assessments
Recovery curves of individual male and female participants and group averages for males and females are shown in Figure 2. Considerable variability can be observed in the recovery curves. Simple visual inspection of the figure does not reveal any obvious systematic effect of sex. Results of the finalized linear mixed models are shown in Table 2. Results of the linear mixed models using corticospinal tract lesion load are shown in Supplemental Table S1. The sex × age interaction was not significant for any robotic task, and therefore removed from the final models. The sex × lesion volume or sex × lesion load interactions were also found to not be significant and removed from the final models. The sex × time interaction, which would indicate different recovery trajectories for males and females, was not significant for any robotic task, nor were the main effects of sex and age.
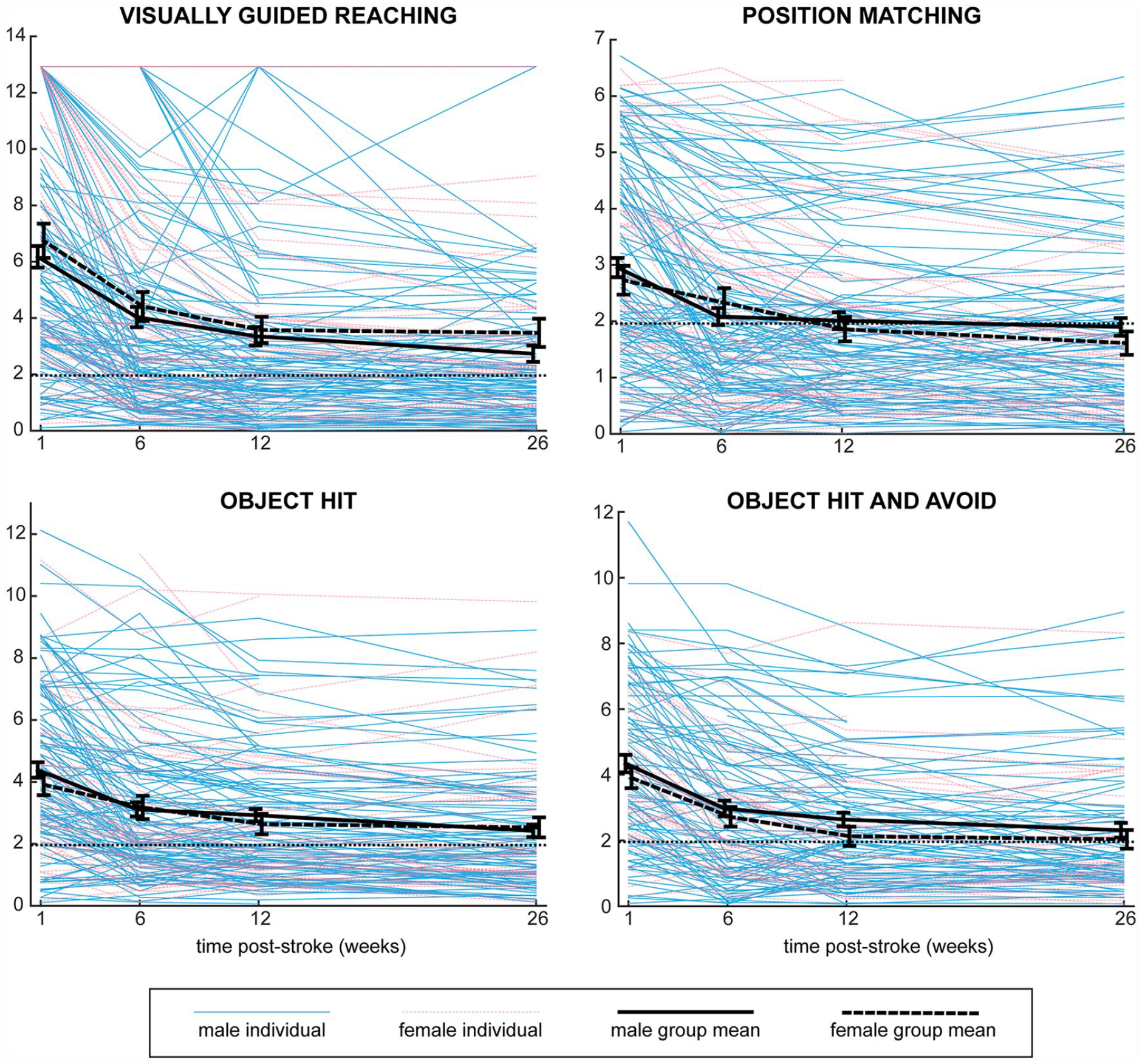
Recovery curves for males and females on robotic tasks. Task scores across the time points for each individual are shown, coded for sex. Mean scores with error bars representing standard error are superimposed for males and females. Task scores represent standard deviations from normal, with higher scores representing worse performance, and scores below the dotted line at 1.96 representing performance in the normal range.
Results of Linear Mixed Effects Models. a
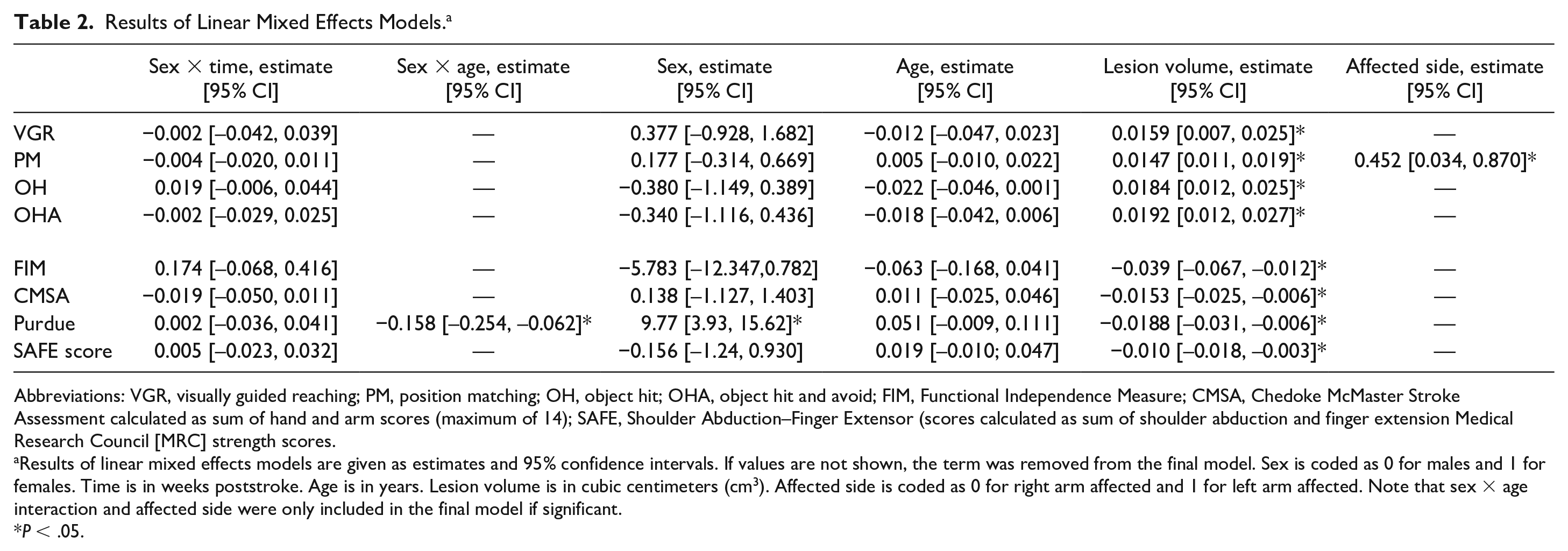
Abbreviations: VGR, visually guided reaching; PM, position matching; OH, object hit; OHA, object hit and avoid; FIM, Functional Independence Measure; CMSA, Chedoke McMaster Stroke Assessment calculated as sum of hand and arm scores (maximum of 14); SAFE, Shoulder Abduction–Finger Extensor (scores calculated as sum of shoulder abduction and finger extension Medical Research Council [MRC] strength scores.
Results of linear mixed effects models are given as estimates and 95% confidence intervals. If values are not shown, the term was removed from the final model. Sex is coded as 0 for males and 1 for females. Time is in weeks poststroke. Age is in years. Lesion volume is in cubic centimeters (cm3). Affected side is coded as 0 for right arm affected and 1 for left arm affected. Note that sex × age interaction and affected side were only included in the final model if significant.
P < .05.
In addition to examining how factors may interact with sex, we also explored the role of potential confounding variables for stroke recovery including lesion volume and affected side (see Table 2). Lesion volume was found to be significant, with increases in lesion volumes associated with increased task scores (poorer performance). When we examined for the impact of affected side, it was only significant for the position matching task. This means that individuals whose left arm was affected had higher task scores, or worse performance on the position matching task. On the remainder of the robotic assessments, there was no impact of affected side.
Supplemental Table S2 shows the results for the analysis including only participants who completed all four time points. The only difference between this subanalysis and the full group was that there was a significant main effect for age for the object hit and avoid task.
Clinical Assessments
Recovery curves for each clinical measure are shown in Figure 3, with results of the linear mixed models shown in Table 2. Results for the CMSA arm and hand assessments are shown separately in Supplemental Table S3. As with robotic assessments, simple visual inspection reveals no clear difference between males and females in the figure. For the FIM, CMSA, and SAFE scores, the sex × age interaction was not significant, and therefore not included in final models. For these three clinical measures, the sex × time interaction as well as main effects of sex and age were also non-significant. Lesion volume did have a significant impact on performance of clinical measures, with larger lesions associated with poorer performance. The Purdue Pegboard was the only assessment for which there was a significant sex × age interaction, as well as a main effect of sex. Based on the coefficients, these results can be interpreted as younger females having better performance (more pegs) than younger males, but after 62 years of age, the males outperformed females. This interaction is illustrated in Figure 4. The subgroups based on age (younger or older than 62) are used only for visualization purposes, as age was treated as a continuous variable in all analyses. As with the other clinical measures, lesion volume was also significant for the Purdue Pegboard Assessment, with larger lesions associated with fewer pegs. It is important to note that the residuals of the linear mixed models were not normal for the FIM and Purdue Pegboard Assessment. Transformations were attempted, however, since these assessments are not normal due to ceiling effects (FIM) and floor effects (Purdue), normality could not be achieved. While fixed effects in linear mixed models are robust to non-normality, caution in interpreting the results is warranted.
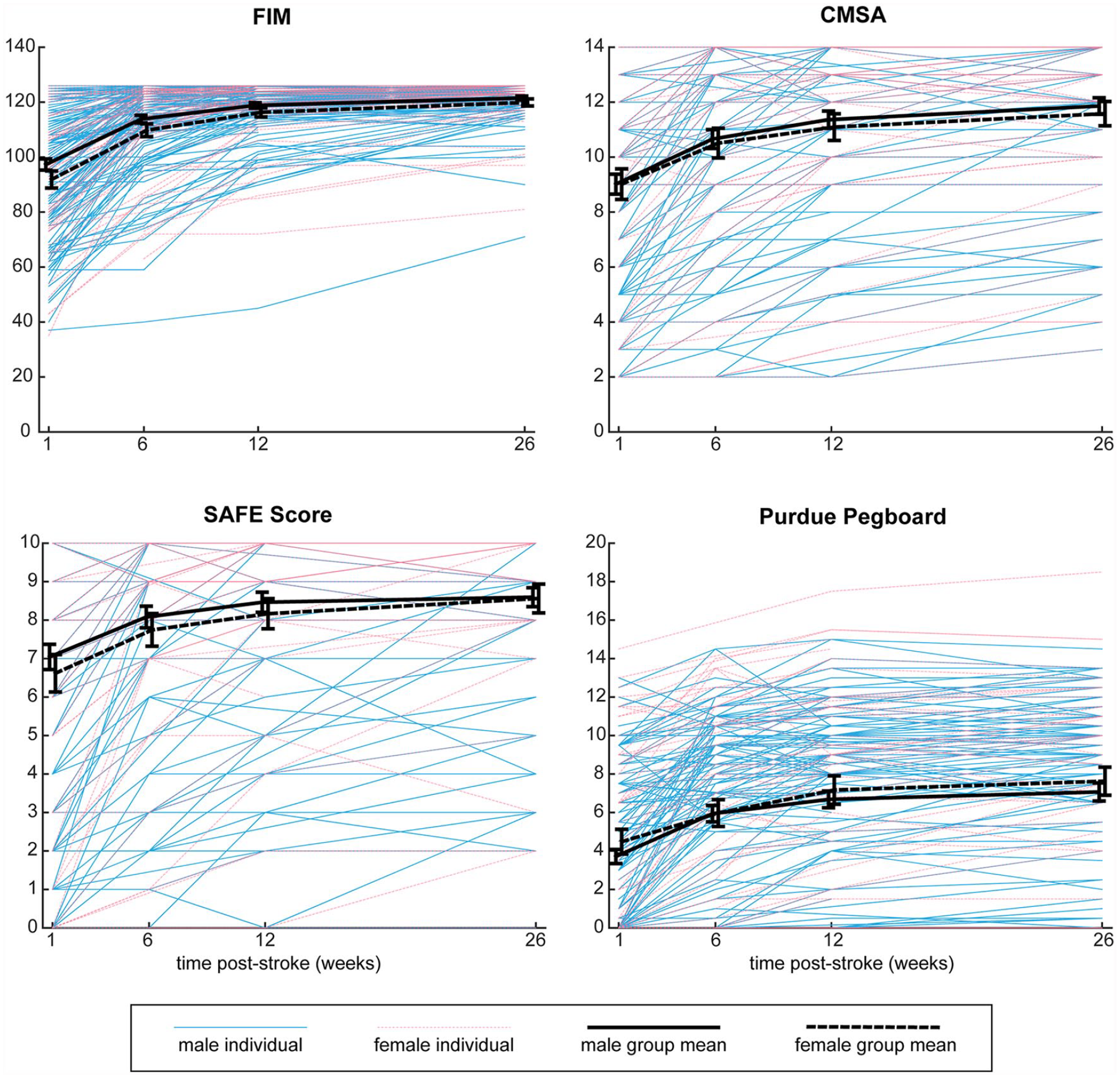
Recovery curves for males and females on clinical assessments. Scores on clinical assessments across the timepoints are shown for each individual, coded for sex. Mean scores with error bars representing standard error are superimposed for males and female. FIM, Functional Independence Measure with maximum score of 126; CMSA, Chedoke McMaster Stroke Assessment, sum of arm and hand scores, with maximum score of 14. SAFE score, sum of MRC muscle strength grades for shoulder abductors and finger extensors, maximum score of 10. Purdue Pegboard Assessment, number of pegs participant could place in the pegboard in 30 seconds using the stroke affected hand.
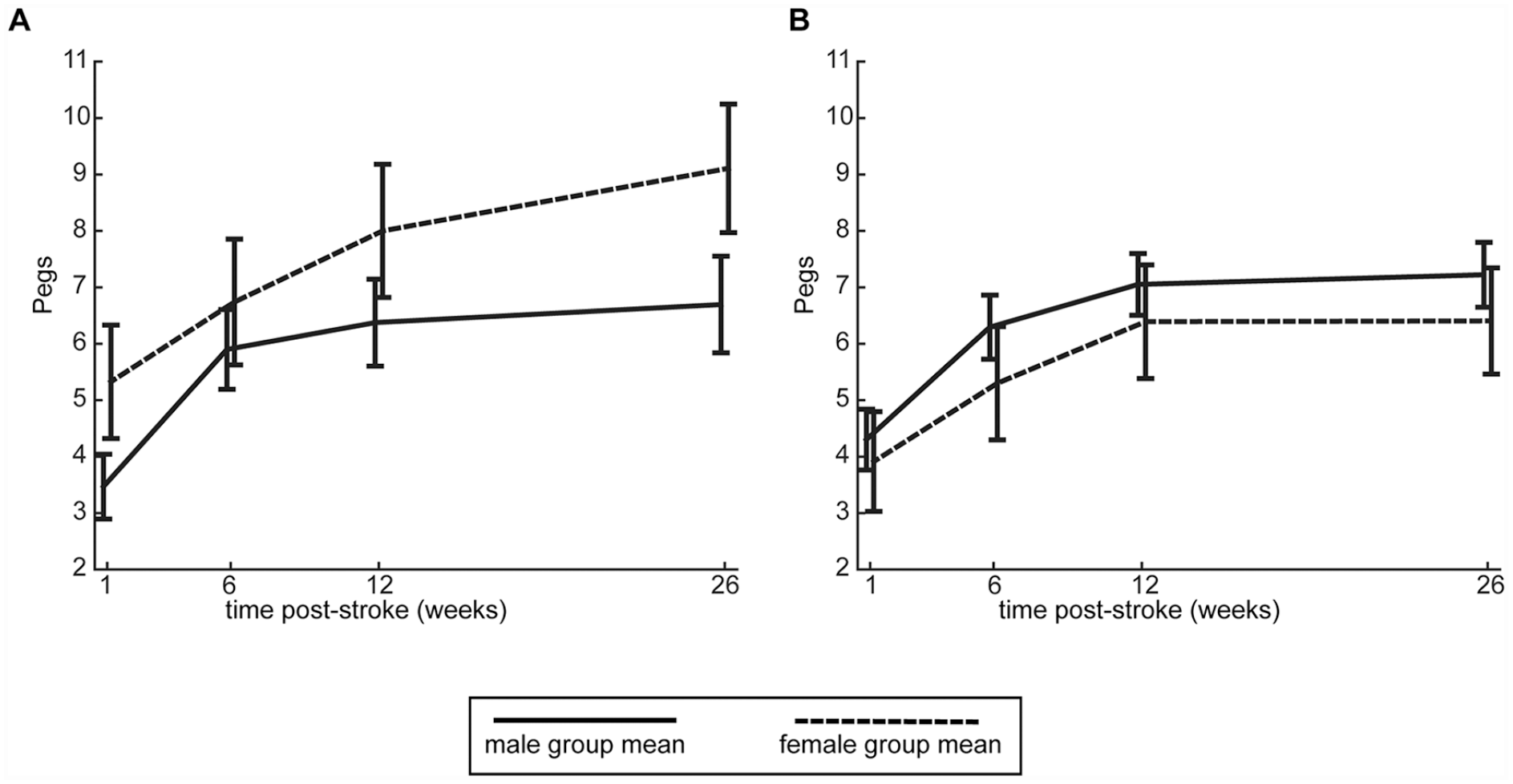
Interaction between age and sex for Purdue Pegboard Assessment. Mean values for the Purdue Pegboard Assessment at each timepoint are depicted for males and females, with error bars representing standard error. Males and females <62 years old (49 males, 28 females) are depicted in A, while males and females ≥62 years old (59 females, 24 females) are shown in B. Analysis of the coefficients demonstrates that in younger participants (<62 years), females outperform males, however in older participants (≥62 years), males outperform females. Note that age was treated as a continuous variable in all analyses, and the subgroups based on age are only being shown in this figure for visualization purposes.
Results for clinical assessments including only the participants who completed all four timepoints are shown in Supplemental Table S2. In general, the same patterns are observed with the exception that we do not find the sex × age interaction significant for the Purdue Pegboard Assessment.
Discussion
This study aimed to examine sex differences in stroke recovery over the first 6 months poststroke using robotic and clinical measures. Unlike most previous studies on sex differences that used functional outcomes,10-13,42 we were primarily interested in determining if differences exist at the impairment level. With the exception of the Purdue Pegboard Assessment, we did not find sex differences in stroke recovery when accounting for age and lesion volume.
An important aspect of the robotic task scores was that they were based on normative values which account for age and sex differences in a large sample of healthy controls. This adjusts for baseline differences in performance that may be at play in previous studies. The importance of using normative models that account for age and sex can be illustrated with the object hit task, where males may hit more balls due to generally better visuospatial skills than females. 43 If we then found male stroke survivors hit more balls than females, this could be misconstrued as males having a better outcome, when it can actually be attributed to baseline differences. The same misinterpretation for age differences could be made as well. While we believe that normative models accounting for baseline differences in sex and age are important to understand stroke recovery, we found the same pattern of results (no age or sex differences) in the clinical assessments that were not based on age or sex adjusted norms.
The Purdue Pegboard Assessment was the only assessment to have a sex × age interaction as well as a main effect of sex. Available norms for the Purdue Pegboard may help to explain this finding. Across adulthood, normative data show that females outperform males,44,45 which has been attributed to females having smaller fingers, which is advantageous on the task. 46 Both sexes in the normative data demonstrate a decrease in number of pegs with age. Our finding that younger females have better performance on the Purdue Pegboard Assessment than younger males is in line with sex differences shown in the normative data. The fact that we found older males then outperform older females support previous findings of poorer functional poststroke outcomes for females compared with males.11,12,17,42 We did not find the sex × age interaction to be significant when only including individuals who had completed all 4 time points, which may be due to the smaller sample size.
Our results differ from previous studies that have found females have poorer outcomes than males poststroke.10,17-19 It is possible that the factors that influence sex differences in functional outcomes do not have the same influence on impairment measures. For instance, overall frailty could contribute to poor functional outcomes, 47 but not have the same magnitude of impact on specific impairment measures such as a proprioceptive position matching task. While our study focused on sex and not gender, gender-based norms may affect functional measures to a greater extent than impairment measures. For example, women may report functional limitations in activities such as cooking and cleaning, whereas some men would not report such limitations since they may be less likely to complete those activities. 48 However, we also did not find sex differences in the FIM, which is contrary to previous findings and our hypothesis.9-12,18 This is likely due to recruitment biases in our study that are discussed in detail below. To further bridge potential discrepancies between sex differences at the impairment and function levels, measures of the upper limb at the activity level would be beneficial. While our work was focused on impairment-based measures, future work assessing sex differences in recovery at the activity level are warranted.
The demographics of our sample do not match the statistics of stroke at large. First, our sample was heavily weighted toward male participants. While the incidence of stroke is greater in females, 6 recent work has documented larger numbers (56.8%) of males in stroke rehabilitation in Alberta.49,50 Second, we found no difference in age between males and females in our study. These differences are likely due to a recruitment bias. While females may have strokes at older ages in general, older participants may be less interested in participating in research or be excluded from participation due to being less medically stable or having additional comorbidities such as other neurologic diagnoses (Parkinson’s disease, dementia) or orthopedic diagnoses such as shoulder osteoarthritis. The older participants in our study were those with milder strokes and better outcomes than their general age-matched cohort, as older individuals with severe strokes were less likely to enter into the study. In general, female participants may be less likely to participate in a trial due to balancing caretaking roles at home, or transportation issues with making study appointments if they have outlived a partner. 51 Women have been found to be more likely to refuse participation in a research study, which may be due to being more risk-adverse. 52 While one must be careful comparing our findings to the general population of individuals undergoing stroke recovery due to the factors above, we believe our sample is well representative of other research studies that are often influenced by the same biases (RATULS, 39% female 53 ; EXCITE, 34.7% female 54 ; VA-Robotics, 4.0% female 55 ; ICARE, 43.8% female). 56 We also acknowledge that to make definitive statements on sex differences at the impairment level, a larger sample may be necessary.
We did not find evidence to suggest that sex affected the trajectory of recovery post-stroke on specific upper limb tasks. We advocate for being proactive in recruiting female participants for research studies, accounting for factors that may negatively influence their desire to participate. We also need to be aware that when looking at impairment, study methodology will often introduce a recruitment bias, since such studies are often more involved than studies where functional outcomes can be determined with an interview. In addition to sex differences, ethnicity and race may also influence stroke risk, treatment, and recovery; however, these factors were beyond the scope of this study. In conclusion, our results do not demonstrate a clear need to account for sex in study design and analysis when examining similar impairments to this study poststroke. Future research is needed to further determine why female stroke survivors may have worse functional outcomes if sex differences in impairment do not exist.
Supplemental Material
supplemental_materials – Supplemental material for Assessment of Sex Differences in Recovery of Motor and Sensory Impairments Poststroke
Supplemental material, supplemental_materials for Assessment of Sex Differences in Recovery of Motor and Sensory Impairments Poststroke by Rachel L. Hawe, Tyler Cluff, Dar Dowlatshahi, Michael D. Hill and Sean P. Dukelow in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We acknowledge Janice Yajure and Mark Piitz for assistance in data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by a Canadian Institutes of Health Research operating grant (MOP 106662) and a Heart and Stroke Foundation of Canada Grant-in-Aid.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
