Abstract
Background:
Motor impairment in the arms is common after stroke and many individuals participate in therapy to improve function. It is assumed that individuals with stroke can adapt and improve their movements using feedback that arises from movement or is provided by a therapist. Here we investigated visuomotor adaptation in individuals with sub-acute and chronic stroke.
Objective:
We examined the impact of the stroke-affected arm (dominant or non-dominant), time post-stroke, and relationships with clinical measures of motor impairment and functional independence.
Methods:
Participants performed reaching movements with their arm supported in a robotic exoskeleton. We rotated the relationship between the motion of the participant’s hand and a feedback cursor displayed in their workspace. Outcome measures included the amount that participants adapted their arm movements and the number of trials they required to adapt.
Results:
Participants with stroke (n = 36) adapted less and required more trials to adapt than controls (n = 29). Stroke affecting the dominant arm impaired the amount of adaptation more than stroke affecting the non-dominant arm. Overall, 53% of participants with stroke were impaired in one or more measures of visuomotor adaptation. Initial adaptation was weakly correlated with time post-stroke, and the amount of adaptation correlated moderately with clinical measures of motor impairment and functional independence.
Conclusion:
Our findings reveal impairments in visuomotor adaptation that are associated with motor impairment and function after stroke. Longitudinal studies are needed to understand the relationship between adaptation and recovery attained in a therapy setting.
Introduction
Humans perform a variety of movements and activities while interacting with different objects and environments (e.g., bathing, dressing, and feeding). While most people perform activities of daily living with ease and proficiency, even healthy adults have to adapt when changes in properties of the task, environment, or body disrupt accuracy and produce movement errors. These errors can arise from transient disturbances, like a bus traveling over a bump when we are sipping from a drink, or systematic changes in properties of the body or environment, such as a loss of muscle strength, wearing a cast, or adjusting to new eyeglasses. Motor adaptation enables the nervous system to accommodate for changes in the body or environment that would otherwise produce errors and jeopardize task performance. 1
Approximately 70% of individuals with stroke have motor impairments that compromise their ability to perform everyday tasks. 2 Stroke rehabilitation often involves working with therapists to practice arm movements that are important for activities of daily living. It is assumed that individuals with stroke are capable of modifying their arm movements using error feedback that arises from movement or is provided by a therapist.3–6 This may be problematic as motor adaptation is reduced, on average, in chronic stroke and impedes the ability to use error feedback to interact with different objects and environments.7–11
Visuomotor rotations are a common method for assessing how the nervous system uses error feedback to adapt arm movements. The rotation creates a systematic discrepancy between the motion of the participant’s arm and a virtual feedback cursor displayed in their workspace. The resulting errors engage the nervous system in adapting arm movements. 12 Adapting to visual transformations may also capture challenges encountered in daily life after stroke. Many tasks, like using a computer mouse to move a cursor or a mirror to facilitate grooming or dressing, alter the relationship between arm movements and their visual consequences.
Lesions in the left hemisphere of the brain reduce average levels of visuomotor adaptation in participants with chronic stroke, whereas lesions in the right hemisphere seem to have little influence on adaptation.8,10 It remains unclear if motor adaptation is impaired in the weeks and months after stroke (i.e., early to late sub-acute phases of recovery), 13 when many individuals participate in rehabilitation. Beyond this, we have yet to establish the relationship between visuomotor adaptation and motor impairment measured by common clinical scales (e.g., Fugl-Meyer Assessment – Upper Extremity, FMA-UE), as well as how adaptation relates to difficulties performing activities of daily living (e.g., Funtional Independence Measure, FIM). Answering these questions will broaden our understanding of stroke impairment within the time period that many therapists work with patients.
Here we used a robotic task to assess visuomotor adaptation in individuals with sub-acute to chronic stroke. First, we hypothesized that participants with stroke would adapt less and require more trials to adapt than controls. Second, based on past studies,8,10 we expected that participants affected in their dominant arm would adapt less and slower than participants affected in their non-dominant arm. Finally, we examined how adaptation related to time post-stroke, and clinical assessments of motor impairment and functional independence.
Methods
Participants
We recruited a convenience sample of participants with stroke from rehabilitation units at the Foothills Medical Centre and Dr. Vernon Fanning Centre in Calgary, AB, Canada. Participants with stroke were also recruited from the community after spending time at these facilities and consenting to be contacted for research post-discharge.14,15 Our inclusion criteria were as follows: 18 years or older with clinical diagnosis of first-time ischemic or hemorrhagic stroke. Our exclusion criteria were as follows: prior diagnosis of secondary neurological conditions (e.g., Parkinson’s disease), musculoskeletal injuries that could interfere with the ability to perform the task, apraxia as identified by clinical assessment, 16 or the inability to understand instructions for the task and clinical assessments. 17 Control participants were recruited from the University of Calgary and surrounding community.14,15 Controls were considered eligible if they had no history of neurological conditions or recent upper limb musculoskeletal injuries. The control and stroke samples were matched for overall age and sex. The study protocol was approved by the Conjoint Health Research Ethics Board at the University of Calgary. Participants provided written informed consent before the experiment.
Experimental Apparatus and Task
Participants made planar reaching movements while seated with their arm supported against gravity by a robotic exoskeleton (Kinarm, Kingston, ON, Canada; Figure 1A). The robot recorded shoulder and elbow joint motion and was paired with a visual display that enabled participants to interact with virtual targets while moving their arm in a near frictionless environment. A small feedback cursor (0.8 cm diameter) was displayed in the participant’s workspace throughout the task (Figure 1B). Direct vision of the arm and hand was occluded by a metal shutter.
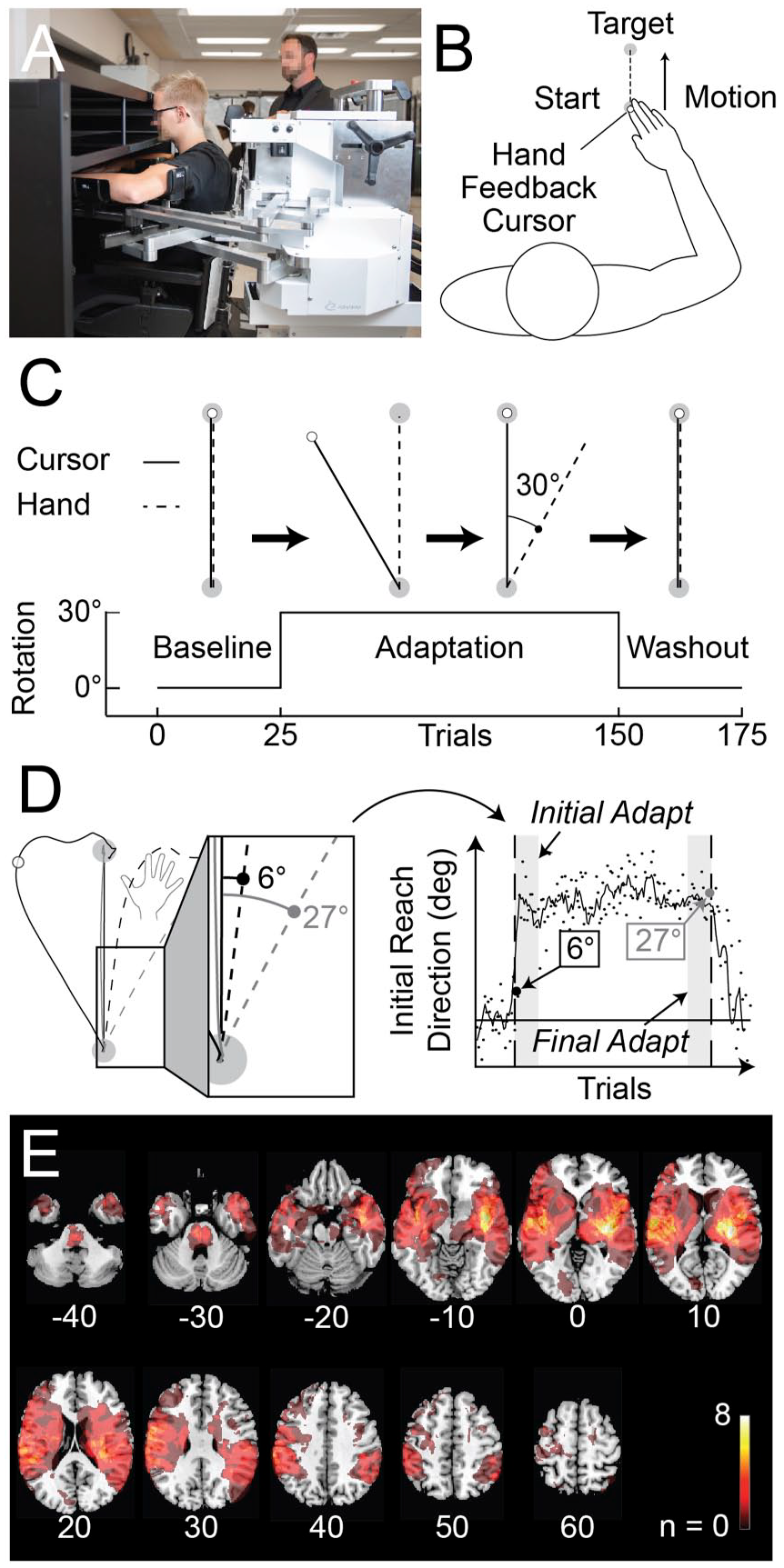
Experimental apparatus, protocol, and analysis. (
The experimental task is shown in Figure 1B. We asked participants to make accurate movements from the start position to goal target. Trials were self-paced and began when participants moved their feedback cursor into the start position. Participants with stroke performed the task with their more affected arm as this is the arm that typically undergoes rehabilitation. 18 Controls performed the task with their dominant arm as studies have revealed similar patterns of adaptation across both arms in healthy adults.19,20
The task was divided into baseline, adaptation, and washout phases (Figure 1C). Participants performed 25 baseline movements with the feedback cursor aligned with the tip of their index finger (Figure 1C). The position of the feedback cursor was rotated 30° counter-clockwise around the center of the start position in the adaptation phase. As a result, forward motion of the hand caused the feedback cursor to move 30° leftward of a straight line joining the center of the start position and goal target (Figure 1C). Participants were exposed to the rotation for 125 movements. The feedback cursor was then unexpectedly re-aligned with the participant’s index fingertip in the washout phase. Participants performed 25 movements to washout adaptation (Figure 1C).
Data Analysis
Adaptation was measured by quantifying the direction of each participant’s hand movements at the start of every trial (Figure 1D). The initial reach direction was calculated as the angular deviation of the hand relative to a straight line connecting the centre of the start position and goal target at 150 ms after the onset of movement. This method has been used to understand how healthy adults adapt their planned arm movements to a visuomotor rotation while minimizing the role of corrective movements. 21 Initial reach directions were normalized to the average of each participant’s baseline movements. Initial Adaptation was defined as the average reach direction during the first 3 blocks of the adaptation phase (5 trials/block). Final Adaptation was defined as the average reach direction during the last 3 blocks of the adaptation phase. Movement variability was calculated throughout the baseline phase (SD). Finally, the normative range of Final Adaptation was defined as the interval that contained 95% of the control sample. Using the lower limit of the range as a threshold, we determined the number of trials that controls and participants with stroke required to achieve 15 consecutive trials (3 blocks) above this criterion. The first trial to meet the criterion was taken as the number of trials that each participant required to attain the level of Final Adaptation expressed by controls (Trials to Adapt). Participants who did not meet this criterion by the end of the adaptation phase were assigned a value of 125 trials (length of adaptation phase).
Normative ranges (containing 95% of control data) were determined for Initial Adaptation, Final Adaptation, and Trials to Adapt. Participants who scored outside the normative ranges were considered to have performed poorly on a specific measure. 17 The change in performance between Initial and Final Adaptation (ΔAdaptation) was used to quantify practice effects and the extent to which participants with lower performance in Initial Adaptation adapted by Final Adaptation (see Supplemental material 1).
Imaging and Lesion Delineation
Most participants with stroke underwent clinical MRI (n = 30). The clinical MRI scans were taken 1 day [median, range = 0–36 days] post-stroke-onset on a 1.5 T Siemens or 3.0 T GE Medical Systems scanner. The scans were acquired as part of standard acute stroke imaging protocols at the Foothills Medical Centre.17,22,23 Scans included T2-weighted fluid-attenuated inversion-recovery (FLAIR), diffusion weighted imaging (DWI), and apparent diffusion coefficient (ADC) sequences. Participants with hemorrhagic stroke also underwent susceptibility weighted imaging (SWI) or gradient echo (GRE) sequences. A smaller portion of participants with stroke received noncontrast CT (n = 6) at 1 day [median, range = 0–9 days] post-stroke onset using a Siemens CT system or one of three GE CT scanners. 23 As part of clinical care at Foothills Medical Centre, MRI is typically not performed if a well-defined infarct is seen on CT.17,22
Stroke lesions were delineated by a trained assessor on T2-FLAIR or noncontrast CT using MRIcron software (https://www.nitrc.org/projects/mricron). 24 The procedure provided a volume of interest (VOI) describing the extent of brain damage. 22 For participants with MRI, DWI and ADC images were used to identify areas of acute brain damage due to ischemia. Either SWI or GRE sequences were used to identify the extent of intracranial hemorrhage in the brain. 22 Lesion delineations were verified by an experienced stroke neurologist blinded to the purpose and results of the study. The lesions were registered to Montreal Neurological Institute (MNI) space using the ch2bet anatomical brain template in MRIcron. 24 Image registration was performed using the clinical toolbox 25 in SPM12. 26 We avoided distortion and warping of the damaged tissue by applying cost function masks to the areas damaged by stroke prior to registration. 27 The registered VOIs were compared to the original images to ensure accuracy. The VOIs were used to create an overlap map that characterizes brain lesions in our stroke sample.
Clinical Assessments
Participants self-reported their dominant arm prior to stroke. We acquired National Institutes of Health Stroke Scale (NIHSS, assessment for stroke severity) 28 scores performed by the stroke neurologist as a part of standard clinical care upon admission to the acute stroke unit.29,30 All other clinical assessments were performed by a certified research therapist blinded to the purpose and results of the study within 2 days [median, range = 0–11 days] of when participants performed the experimental task. The following assessments were performed: Fugl-Meyer Assessment of Motor Recovery – Upper Extremity Motor Assessment (FMA-UE, assesses motor impairment of the arm and hand), 31 Chedoke–McMaster Stroke Assessment – Arm Impairment Inventory (CMSA, measures motor impairment in the arm), 32 Thumb Localization Test (TLT, assesses for proprioceptive impairment), 33 Modified Ashworth Scale (MAS, assesses spasticity of the elbow flexors), 34 Montreal Cognitive Assessment (MoCA, assesses for cognitive impairment), 35 conventional sub-tests of the Behavioral Inattention Test (BIT, screens for hemispatial neglect), 36 and Functional Independence Measure (FIM, measures independence in performing activities of daily living). 37
Statistical Analysis
Adaptation measures were compared across stroke and control groups using two-sample bootstrap hypothesis tests (one-tailed, resampled 99 999 times with replacement).38,39 The proportion of participants who performed poorly on each adaptation measure were compared using chi-square tests. Kruskal–Wallis H-tests were used to compare the adaptation of control and stroke participants affected in their dominant arm (dominant affected, DA), or non-dominant arm (non-dominant affected, NDA). We also performed an ANCOVA to rule out the influence of time post-stroke on each adaptation measure. Chi-squared tests were used to compare the proportion of controls, DA, and NDA participants with stroke who fell outside of the normative range for each adaptation measure.
The relationship between time post-stroke (days) and measures of adaptation was assessed using Spearman’s partial rank correlations (two tailed) to account for the side of the stroke-affected arm. Spearman’s partial rank correlations (two tailed) 40 assessed the relationship between clinical assessments of motor impairment (FMA-UE), functional independence (FIM) and each adaptation measure. Time post-stroke was used as a covariate in this analysis. The analysis was repeated using ordinary Spearman’s correlations unadjusted for time post-stroke. P-values for statistical tests were corrected for multiple comparisons using Bonferrroni–Holm methods. 41 The corrected P-values are presented throughout the text. The threshold for significance was set to α = .05. Analyses were performed using custom scripts created in MATLAB 2018a (MathWorks, Natick, MA).
Results
Visuomotor adaptation was examined in 36 participants with stroke and 29 controls. We excluded 2 participants with stroke who consented to the study, but were unable to understand task instructions. Of the participants with stroke, 25 were inpatients in stroke rehabilitation units at the time of assessment (sub-acute stroke). The remaining 11 participants completed the experiment after they had left the rehabilitation units (chronic stroke). Demographics and clinical scores are displayed in Table 1. Lesion characteristics for participants with stroke are shown in Figure 1E.
Demographics and clinical characteristics of participants with stroke.
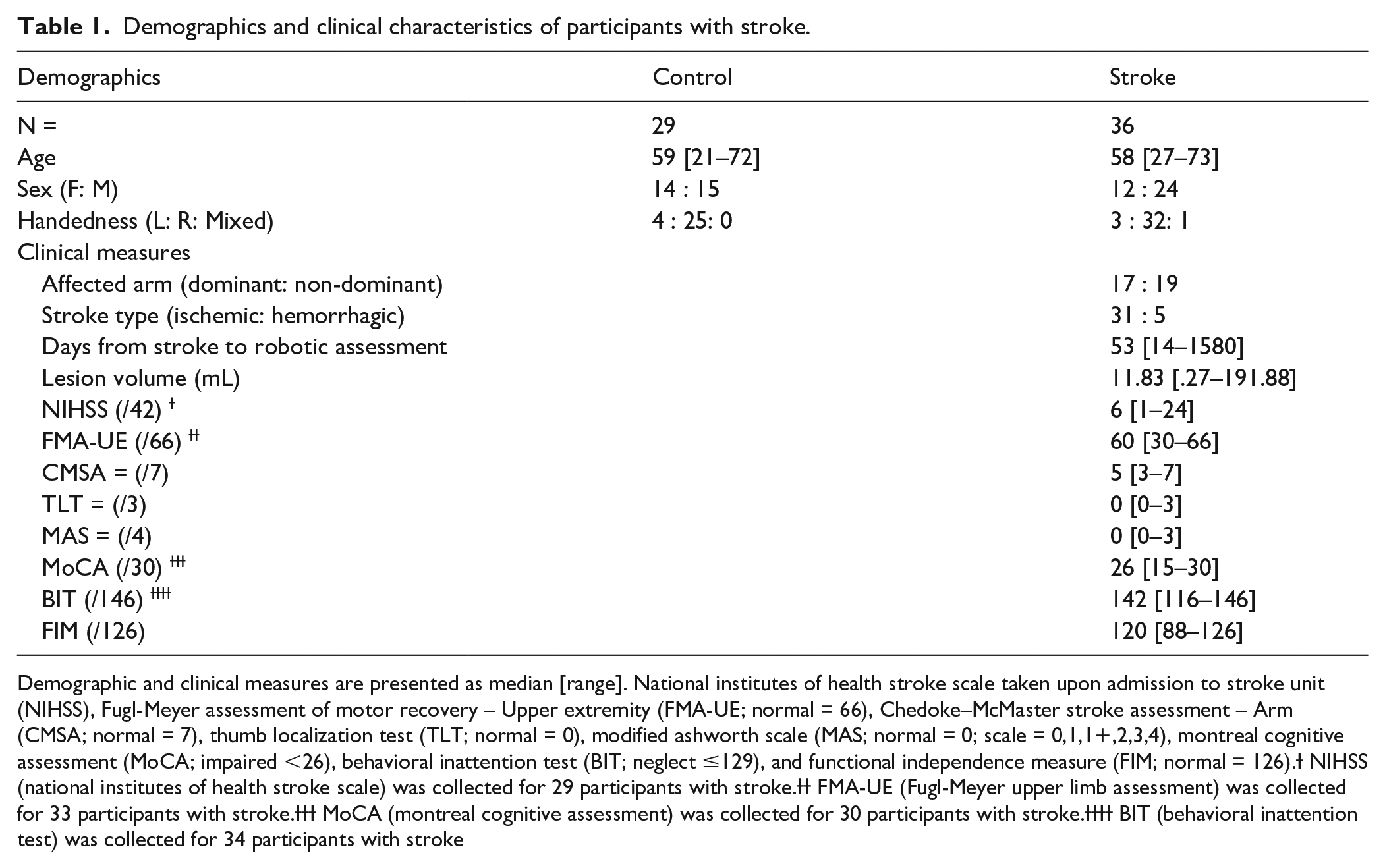
Demographic and clinical measures are presented as median [range]. National institutes of health stroke scale taken upon admission to stroke unit (NIHSS), Fugl-Meyer assessment of motor recovery – Upper extremity (FMA-UE; normal = 66), Chedoke–McMaster stroke assessment – Arm (CMSA; normal = 7), thumb localization test (TLT; normal = 0), modified ashworth scale (MAS; normal = 0; scale = 0,1,1+,2,3,4), montreal cognitive assessment (MoCA; impaired <26), behavioral inattention test (BIT; neglect ≤129), and functional independence measure (FIM; normal = 126).Ɨ NIHSS (national institutes of health stroke scale) was collected for 29 participants with stroke.ƗƗ FMA-UE (Fugl-Meyer upper limb assessment) was collected for 33 participants with stroke.ƗƗƗ MoCA (montreal cognitive assessment) was collected for 30 participants with stroke.ƗƗƗƗ BIT (behavioral inattention test) was collected for 34 participants with stroke
Visuomotor Adaptation Is Impaired After Stroke
Figure 2A shows a representative control, a participant with stroke who performed poorly on the task, and a participant with stroke who performed well on the task. Participants made relatively accurate movements throughout the baseline phase. Initial reach direction did not differ significantly between stroke and control participants in the baseline phase (bootstrap: P = .14). Participants with stroke displayed more variable baseline movements than controls (bootstrap: P < .01).
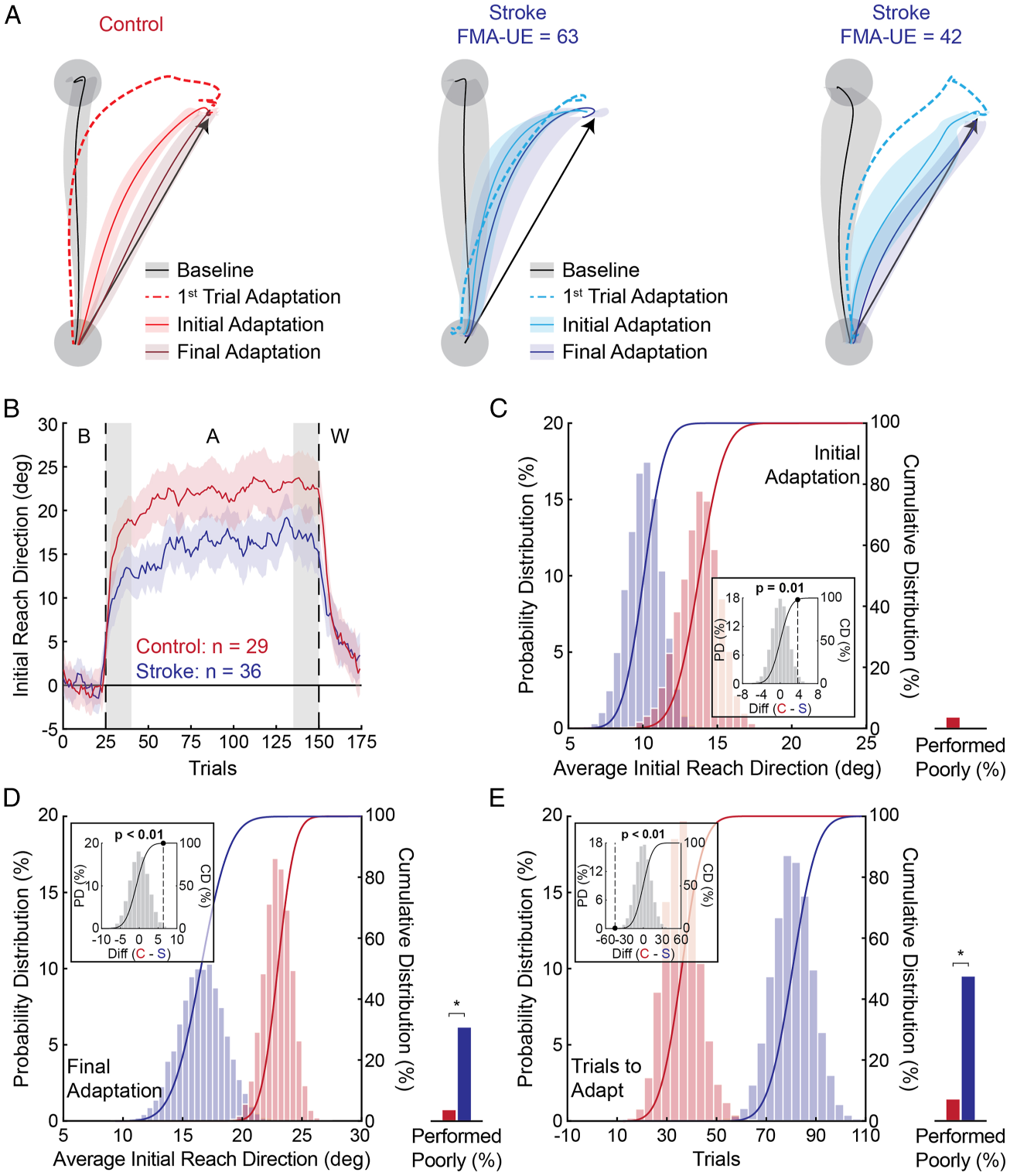
Hand paths, adaptation patterns, and bootstrap hypothesis tests. (
Participants reached directly towards the target on the first adaptation trial. Due to the rotation, forward hand motion resulted in a leftward cursor deviation (i.e., counter-clockwise) and the participants made a rightward correction to direct their cursor to the goal target (Figure 2A). In Initial Adaptation, the control participant adapted their initial reach direction more than the representative participants with stroke (Figure 2A). We found a similar result at the group level (bootstrap: P = .01; Figure 2B and 2C). The proportion of participants who performed outside of the normative range did not differ significantly between stroke and control participants (chi-square: X 2 (1) = 1.26, P = .26; Figure 2C).
The representative control participant displayed near complete adaptation to the rotation (Final Adaptation; Figure 2A). The range of adaptation was more variable in participants with stroke. Despite mild motor impairment, one of the exemplar participants with stroke displayed incomplete adaptation. The other representative participant with stroke had moderate motor impairment but adapted to a level similar to controls (Figure 2A). At the group level, participants with stroke displayed an overall reduction in Final Adaptation (bootstrap: P < .01; Figure 2D). A greater proportion of participants with stroke performed poorly compared to controls (chi-square: X 2 (1) = 7.84, P < .01; Figure 2D).
Participants with stroke required more Trials to Adapt (81 [5–125]) than controls (36 [3–125]; bootstrap: P < .01) and close to half of the participants with stroke were unable to adapt to the normative range (47%; Figure 2E). As expected, only 2 controls were outside of this range (chi-square: X 2 (1) = 12.63, P < .01; Figure 2E). Collectively, the participants with stroke adapted less and required more Trials to Adapt than controls.
Final Adaptation Is Reduced When the Dominant Arm Is Affected by Stroke
Figure 3A shows visible differences in adaptation between individuals who were affected in their dominant arm (DA) vs non-dominant arm (NDA). Initial Adaptation did not differ significantly across groups (H-test: X 2 (2) = 5.68, P = .06). There were no differences in the proportion of controls, DA, and NDA participants with stroke who performed poorly on this measure (chi-squared: all X 2 (2) = 1.26, P = .53).
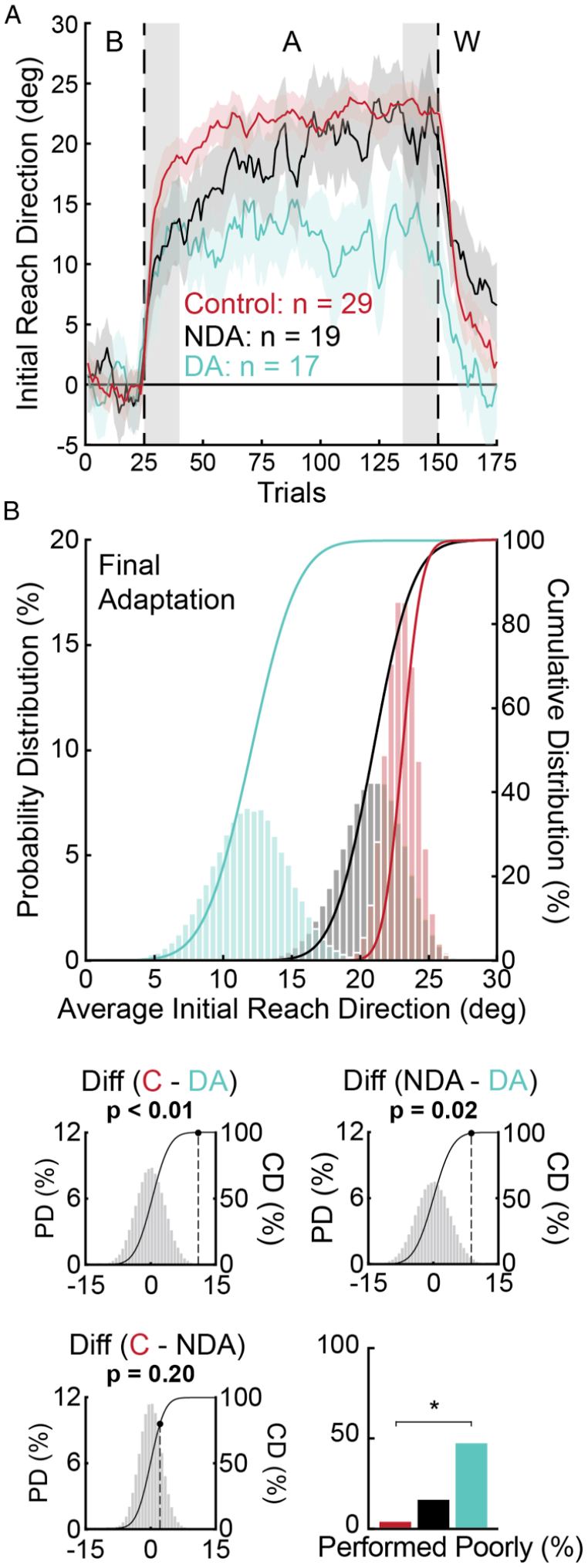
Visuomotor adaptation curves and bootstrap hypothesis tests examining the influence of the stroke-affected arm. (
Final Adaptation differed across groups (H-test: X 2 (2) = 9.96, P < .01) and was significantly lower in the DA group relative to controls (bootstrap: P < .01), and DA relative to NDA participants (P = .02; Figure 3B). Final Adaptation did not differ significantly between NDA participants and controls (P = .20). We also observed a significant difference in the proportion of individuals who performed poorly in Final Adaptation (chi-squared: all X 2 (2) = 13.67, P < .01; Figure 3B). A greater proportion of DA participants performed poorly compared to controls (chi-square: X 2 (1) = 12.95, P < .01; Figure 3B).
The number of Trials to Adapt differed across groups of controls, DA, and NDA participants with stroke (H-test: X 2 (2) = 14.54, P < .01). DA and NDA participants did not differ significantly in the number of Trials to Adapt (bootstrap: P = .44), and both groups required more trials than controls (both P < .01). The proportion of participants who performed poorly on this measure also differed across groups (chi-squared; all X 2 (2) = 12.63, P < .01). A larger proportion of DA participants performed poorly on the Trials to Adapt measure than controls (chi-squared: X 2 (1) = 10.16, P < .01). ANCOVA analysis did not reveal any interactions between time post-stroke and the stroke-affected arm for any measure of adaptation (all P > .05).
Participants in the DA group scored significantly lower on the FIM than NDA participants (bootstrap: P < .01). Initial stroke severity (NIHSS: P = .49), motor impairment (FMA-UE: P = .34; CMSA: P = .23), proprioception (TLT: P = .47), spasticity (MAS: P = .52), and cognitive impairment (MoCA: P = .37; all bootstrap hypothesis tests) did not differ significantly across DA and NDA participants. BIT scores were worse in the NDA group (bootstrap: P = .04). The difference in BIT scores was perhaps unsurprising as hemispatial neglect typically arises from damage to the right parietal cortex and most of our sample was right-hand dominant prior to stroke (17/19 or 89% of NDA participants had right hemisphere lesions).
Correlations With Time Post-Stroke
Spearman partial correlations revealed a weak negative relationship between time post-stroke and Initial Adaptation when adjusting for the side of the stroke-affected arm (ρpartial = −.36, P = .04; Figure 4A; Supplementary material 2). There was no significant relationship between time post-stroke and Final Adaptation (ρpartial = −.11, P = .54) or Trials to Adapt (ρpartial = .15, P = .38). Qualitatively similar relationships were observed in the unadjusted correlations (Supplementary material 3).
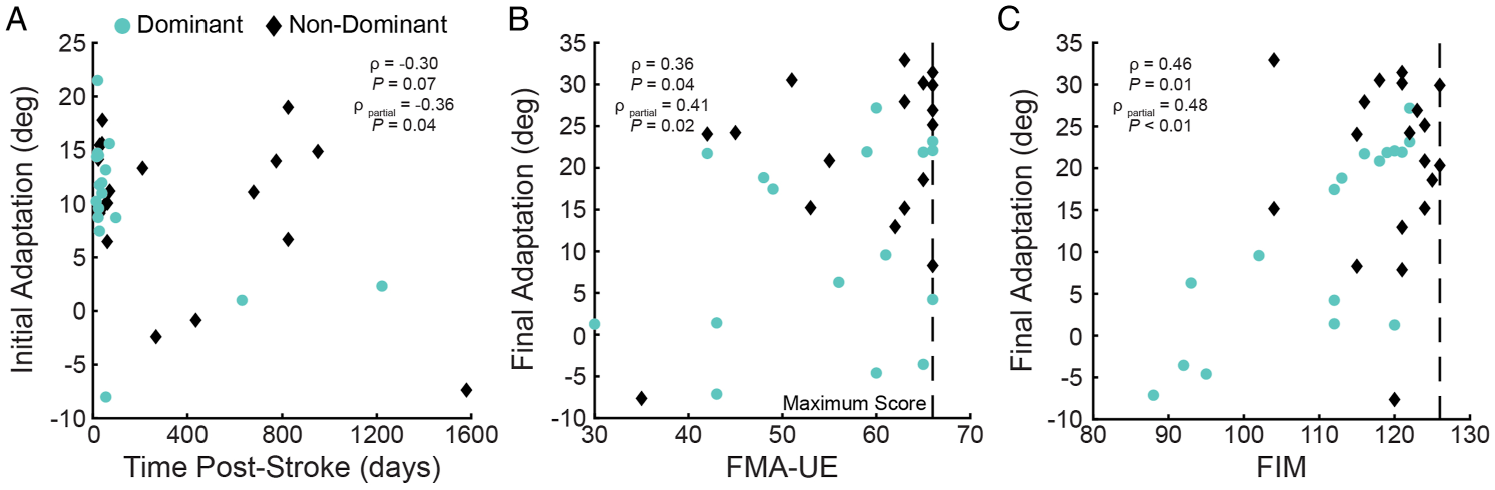
Scatter plots of (
Correlations With Clinical Assessments
We performed separate Spearman partial correlations to examine the relationship between each measure of visuomotor adaptation and motor impairment (FMA-UE). The same analyses were used to examine the relationship between visuomotor adaptation and functional independence (FIM). FMA-UE scores did not correlate with Initial Adaptation or Trials to Adapt when controlling for time post-stroke (Table 2). However, there was a significant positive relationship between FMA-UE scores and Final Adaptation (Table 2; Figure 4B; Supplementary material 2), such that individuals with lesser motor impairment adapted better to the rotation. FIM scores did not correlate with Initial Adaptation or Trials to Adapt when controlling for time post-stroke (Table 2). We did find a positive relationship whereby greater functional independence was associated with greater Final Adaptation (Table 2; Figure 4C; Supplementary material 2). Qualitatively similar results were observed with Spearman’s correlations that were not adjusted for time post-stroke (Supplementary material 3).
Spearman partial correlations accounting for time post-stroke. Fugl-Meyer assessment of motor recovery – Upper extremity (FMA-UE; n = 33) and functional independence measure (FIM; n = 36). Data are presented as the rho (P-value) for each correlation. P-values were Bonferroni–Holm corrected. *Indicates corrected P < .05

Discussion
Motor adaptation is thought to be important for understanding the health and function of the nervous system across a variety of contexts including stroke rehabilitation.1,3,4 We used a robotic task to profile impairments in individuals ranging from 14 days to roughly 4 years post-stroke (median = 53 days). The task imposed a systematic discrepancy between hand motion and a feedback cursor displayed in the participant’s workspace. The task engages adaptation processes in healthy adults that are reduced in chronic stroke.8–11 Out of 36 participants with stroke, 25 were within 6 months post-stroke (sub-acute stroke) and participating in rehabilitation. The results reveal widespread impairment in visuomotor adaptation after stroke.
Visuomotor Adaptation After Stroke
The results highlight the heterogeneity of visuomotor adaptation and the utility of robotic technologies in quantifying impairments after stroke. Impairment levels ranged from 0–47% on individual parameters, with 53% of participants with stroke displaying impairment in one or more measures of visuomotor adaptation. Despite a significant reduction in Initial Adaptation at the group level, the participants with stroke did not perform poorly enough to be classified as impaired based on the range of control data.
Impairments in Final Adaptation (31%) and Trials to Adapt (47%) were common amongst participants with stroke. The results are consistent with the finding that the ability to adapt to visuomotor rotations8–11 or forces applied to the arm during point-to-point reaching movements7,42,43 is reduced in chronic stroke. The findings are also broadly consistent with skill learning studies that revealed a reduction in the ability to learn motor sequences,14,15 navigate through virtual environments with varying accuracy demands,44,45 and track moving stimuli with the arm after stroke. 46
Participants with stroke were most extensively impaired in the number of repetitions they required to adapt (81 trials vs 36 trials in controls). Close to half of the participants (47%) did not attain normal levels of adaptation after performing 125 movements in the rotation. While we do not know the relationship between the number of trials an individual requires to adapt and the number of functional movements required to reduce motor impairment in therapy, it does lead one to consider the following. Therapy is a resource intensive process. Knowing the number of trials that each participant needs to adapt may provide a means to adjust for an individual’s needs while minimizing the cost of therapy.
The Importance of Arm Dominance and Side of the Lesioned Hemisphere for Visuomotor Adaptation
Arm dominance, recovery, and the side of the lesioned hemisphere have received considerable attention after stroke. While some studies report better functional and strength gains in the dominant arm after therapy 47 and less motor impairment in DA participants, 48 other research has reported little difference in motor and functional recovery. 49 We did not observe significant differences in stroke severity (NIHSS), motor (FMA-UE, CMSA), sensory (TLT) or cognitive impairment (MoCA) between DA and NDA participants in our sample. We did note greater functional independence (FIM) in the sample of NDA participants (9 point difference between NDA and DA participants).
Average levels of Final Adaptation were reduced in DA participants compared to NDA participants and controls. This finding is consistent with research in chronic stroke that reported selective impairment in visuomotor adaptation following left-hemisphere damage in right-hand dominant participants. 8 The majority of our sample was right-hand dominant prior to stroke (89%) and we found similar results when limiting our analyses to these participants (Supplementary material 4). Despite normal levels of Final Adaptation, NDA participants required more Trials to Adapt than controls. Larger studies may better inform how clinicians account for arm dominance and the side of hemispheric damage in stroke therapy.
Visuomotor Adaptation and Time Post-Stroke
We observed a negative relationship between time post-stroke and Initial Adaptation when accounting for the side of the stroke-affected arm. This suggests that participants who were farther from the date of their stroke adapted less within the first 15 trials of being exposed to the rotation. The relationship was weak, however, and likely shaped by other factors, including variation in the location and size of stroke lesions, and impairments in other facets of sensory, motor, and cognitive function. Another possibility is that changes in motor impairment influence motor adaptation when assessed at different time points after stroke. Longitudinal studies are needed to determine the relationship between motor adaptation, time post-stroke, and motor impairment during therapy (see Limitations for further discussion).
Relationship Between Adaptation and Clinical Assessment
The robotic measures of adaptation were not only useful in identifying impairments in motor adaptation, but Final Adaptation was also related to motor impairments (FMA-UE) and function (FIM). The relationships between motor adaptation, motor impairment, and functional independence are consistent with studies in motor sequence learning,14,15,46 reaching, 50 elbow force production, 51 and force-field adaptation,7,42,43 ranging from a single session to several weeks of training. We build upon research in motor skill learning by showing that deficits in visuomotor adaptation are also linked with independence in performing activities of daily living. 46
It is worth noting that FMA-UE and FIM varied greatly in how they related to Final Adaptation. Some participants expressed mild motor impairment but adapted poorly to the rotation (FMA-UE = 63; Final Adaptation = 50%; Figure 2A), and others with moderate motor impairment performed well on the task (FMA-UE = 42; Final Adaptation = 80%; Figure 2A). Similar results were noted for the FIM. Neither measure was associated with Initial Adaptation or Trials to Adapt. Thus, while the extent to which participants adapted was related to levels of motor impairment and function, how quickly individuals adapted was not. This may reflect different underlying processes and brain structures that contribute to Initial and Final Adaptation. 52 Another important consideration is that the FIM does not consider nor penalize compensatory strategies that may emerge after stroke (e.g., increased use of the less or unimpaired arm in daily activities). Thus, some participants may adopt compensatory behaviors that allow them to score higher on the FIM despite impairments in motor adaptation. 6 In short, impairments in adaptation are complex and seem to depend on the interplay of a number of factors related to stroke.
Implications for Stroke Rehabilitation
The ability to adapt arm movements based on error feedback is an important component of stroke rehabilitation with potential to serve as a biomarker of stroke recovery. 7 The growing impetus for quantitative biomarkers of stroke impairment has been paired with acknowledgment that the ability to identify impairments may help to inform therapy decisions. 53 We observed that 53% of the participants with stroke that we tested had deficits in visuomotor adaptation. The assessment of motor adaptation deficits is not typically performed in a therapy setting because we lack standardized bedside tools to perform these assessments.
There are currently no treatment options that accommodate for deficits in the ability to modify movements using error feedback. This is perhaps unsurprising as we lack empirical data linking motor adaptation, learning, and improvements in motor function in human participants with stroke. 3 Recent work has reported a lack of confidence amongst therapists who incorporate principles of motor adaptation and learning into clinical practice.54–56 Despite growing consensus surrounding the importance and clinical utility of motor learning principles, experts still lack consensus regarding what principles best promote improvements in motor performance.57,58
Studies in healthy adults have reported a relationship between motor adaptation within a single session, retention at 24 hr, and long-term motor learning.59,60 Prior exposure to a visuomotor rotation can generate long-term motor memories in healthy adults for up to a year without practice. 60 It is unclear if deficits in adaptation after stroke impact long-term motor learning and therapy outcomes. Addressing the relationship between motor adaptation and learning after stroke will require studies on retention, testing the relationship between adaptation and motor skill learning, and determining whether lesions in distinct brain regions are associated with unique impairments in adapting and retaining motor skills. In a treatment setting, once deficits have been identified, it is possible that strategies that are thought to promote motor adaptation and learning, such as non-invasive brain stimulation, pharmaceutical intervention, or mirror therapy, could be attempted to determine if they enhance recovery from stroke. We believe there is much to gain by probing for deficits in motor adaptation and learning as they may capture different challenges or limitations experienced by individuals with stroke.
Limitations
Our study was cross-sectional and can only provide a snapshot of adaptation, impairment, and function after stroke. It is also difficult to rule out other factors that may mediate this relationship, including sensory and cognitive impairment, location and size of stroke lesions, comorbidities and other lifestyle-related factors. The level of impairment measured using common clinical scales ranged from mild to moderate. Finally, the sample of left-handed participants was limited. Prospective longitudinal studies in larger samples, including left and right-hand dominant participants matched for impairment, will provide deeper insight into the relationship between motor adaptation and recovery across a broader range of stroke impairments.
Conclusion
Visuomotor adaptation can be impaired after stroke and relates to some clinical assessments of motor impairment and functional independence. Our results build on existing literature8–10 and reveal a range of impairments with only a portion of the variability in performance captured by common clinical measures of motor impairment and functional independence.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683221095166 – Supplemental material for Assessing Impairments in Visuomotor Adaptation After Stroke
Supplemental material, sj-docx-1-nnr-10.1177_15459683221095166 for Assessing Impairments in Visuomotor Adaptation After Stroke by Robert T. Moore, Mark A. Piitz, Nishita Singh, Sean P. Dukelow and Tyler Cluff in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-2-nnr-10.1177_15459683221095166 – Supplemental material for Assessing Impairments in Visuomotor Adaptation After Stroke
Supplemental material, sj-docx-2-nnr-10.1177_15459683221095166 for Assessing Impairments in Visuomotor Adaptation After Stroke by Robert T. Moore, Mark A. Piitz, Nishita Singh, Sean P. Dukelow and Tyler Cluff in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-3-nnr-10.1177_15459683221095166 – Supplemental material for Assessing Impairments in Visuomotor Adaptation After Stroke
Supplemental material, sj-docx-3-nnr-10.1177_15459683221095166 for Assessing Impairments in Visuomotor Adaptation After Stroke by Robert T. Moore, Mark A. Piitz, Nishita Singh, Sean P. Dukelow and Tyler Cluff in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-4-nnr-10.1177_15459683221095166 – Supplemental material for Assessing Impairments in Visuomotor Adaptation After Stroke
Supplemental material, sj-docx-4-nnr-10.1177_15459683221095166 for Assessing Impairments in Visuomotor Adaptation After Stroke by Robert T. Moore, Mark A. Piitz, Nishita Singh, Sean P. Dukelow and Tyler Cluff in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We aknowledge the work of Janice Yajure for participant assessment.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Canadian Institutes of Health Research operating grant [grant number MOP 106662], as well as the Faculty of Kinesiology and Calgary Health Trust.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
