Abstract
Background. A recent Cochrane Review showed that early robotic training of the upper limb in stroke survivors can be more effective than other interventions when improving activities of daily living involving the arm function is the aim of therapy. Objective. We tested for efficacy of the study a protocol which involved the use of the NeReBot therapy in partial substitution of standard upper limb rehabilitation in post–acute stroke patients. Methods. In this dose-matched, randomized controlled clinical trial, 34 hemiparetic participants with movement against gravity in shoulder, elbow, and wrist muscle groups were enrolled within 15 days of the onset of stroke. All participants received a total daily rehabilitation treatment for 120 minutes, 5 days per week for 5 weeks. The control group received standard therapy for the upper limb. The experimental group received standard therapy (65% of exercise time) associated with robotic training (35% of exercise time). Muscle tone (Modified Ashworth Scale), strength (Medical Research Council), and synergism (Fugl-Meyer motor scores) were measured at impairment level, whereas dexterity (Box and Block Test and Frenchay Arm Test) and activities of daily living (Functional Independence Measure) were measured at activity level. All assessments were performed at baseline, at the end of therapy (time T1), at 3 months (time T2), and at 7 months (time T3) after entry. All between-group analyses were tested using nonparametric test with Bonferroni’s adjustments for multiple testing. Results. No significant between-group differences were found with respect to demographic characteristics, motor, dexterity, and ADLs at baseline, postintervention (T1) and at follow-up (T2 and T3). Conclusions. The robot therapy by NeReBot did not lead to better outcomes compared with conventional inpatient rehabilitation.
Introduction
Disability in activities of daily living (ADLs) and motor impairment often remain in patients who survived to stroke, a pathology with high social impact. 1 The recovery is partial in 85% of stroke survivors, 2 while in 35% of them a serious disability remains. In 30% to 60% of patients treated with traditional rehabilitation, a residual functional impairment of the paretic arm and consequently of ADLs is common.3,4 The number of people who require rehabilitation after a stroke is increasing because of the aging of population. 5
The goal of rehabilitation in paretic patients is to promote the recovery of lost functions, of independence, and reintegration in the activities of social life as soon as possible. Clinical trials have shown a better motor outcome with various sensorimotor programs, 6 including repetitive intensive mobilization, 7 forced use of the paretic limb or constraint-induced movement therapy, 8 biofeedback, 9 functional electrical stimulation, 10 and transcranial magnetic stimulation. 11 Scientific studies on this topic have given evidence of greater effectiveness, in poststroke recovery, if rehabilitation treatment is administered in the early stage (within 6 months). Early rehabilitation leads to improved functional outcome, 12 especially when it is based on intensive and multisensory stimulation.13,14 An improvement of brain plasticity in early poststroke stages has been described in combination with the aforementioned type of stimulation.15-18
The recent introduction of robotic devices to integrate the multidisciplinary programs of the standard rehabilitation in poststroke is a new approach that looks very promising. Robotic systems can offer a high-intensity, repetitive, task-specific, interactive treatment (passive and/or active assisted exercises) of the paretic limb and can give an objective evaluation of motor progress in monitored patients, measuring changes in kinematics movement and forces.19,20 Mehrholz et al, 21 in a review published in the Cochrane Library, showed that the use of robotic devices in rehabilitation may improve ADLs and paretic arm function, but not arm muscle strength. However, the results must be interpreted with caution because of the high variability between the trials.
An important issue still to be explored is the effectiveness of robotic therapy in the post–acute stroke phase. In this regard, many robotic instruments were created to treat the paretic limb, 22 but only few of these have been tested in at least one randomized controlled trial on stroke patients in acute (ie, the days immediately following the onset of illness) or post–acute stroke phase (ie, after the first days following stroke when patients are discharged from acute care). These devices are the MIT-Manus,23,24 the Mirror-Image Movement Enabler,25-27 the Bi-Manu-Track, 28 and the Neuro-Rehabilitation-roBot (NeReBot).29-31 The result of these trials is that robotic training in the early phase of upper limb paresis can significantly improve the ADLs versus other forms of intervention.21,32,33
Studies in the early phase, in which the robotic treatment has been used in addition instead of substitution of conventional rehabilitation of paretic upper limb, suggest that an early start of rehabilitation exercise may benefit patients 33 ; however, appropriate trials are lacking. This new clinical trial, whose preliminary results were presented by Masiero et al, 33 has the aim to assess the effects of NeReBot on arm function and ADLs, when NeReBot training is used in substitution of conventional rehabilitation therapy. Our hypothesis is that substitution of upper extremity training with the NeReBot for one third of conventional rehabilitation time will bring at least equivalent outcomes compared to conventional therapies alone.
Methods
Patients
We enrolled subjects from the Stroke Unit, after a first, single, ischemic or hemorrhagic cerebrovascular accident in postacute phase, which is defined as “the period of time immediately after discharge from acute care (it is known to be average within 15 days after stroke)” and with upper limb impairment. At this point, the stroke patient has achieved medical stability and rehabilitation becomes the focus of care. 34 The following inclusion criteria were adopted: (a) diagnosis of recent single-sided stroke (ischemic or hemorrhagic) demonstrated by brain computed tomography or nuclear magnetic resonance, (b) sufficient cognitive and language capacities to understand the instructions of the operator (assessed with Modified Mini-Mental State Examination score >18), 35 and (c) patients with paralysis or paresis who were not capable of active movement against gravity or weak resistance (Motor Power score between 8 and 12). 36
The following exclusion criteria were adopted: (a) cardiovascular instability (severe uncontrolled hypertension, severe coronary artery disease, etc) or orthopedic or neurological conditions; (b) presence of multiple cerebrovascular lesions (usually results of many past ischemic episodes); (c) early appearance of marked spasticity (≥3 on the Ashworth Scale 37 ); (d) upper limb joint that would have limited the subject’s ability to complete the protocols; (e) severe neuropsychological impairment (global aphasia, severe attention deficit, or severe spatial inattention, evaluated with neurological examination and, when necessary, with the following battery tests—for the language, the neuropsychological aphasia test and the Gellanza-Coen test; for selective and sustained attention, the Bells test; for the spatial attention the Barrage test); (f) use of functional electrical stimulation or botox injection in their upper extremity; and (g) older than 85 or younger than 18 years.
Randomization
Forty-one patients (28 males, 13 females) with stroke were eligible for the study. Seven of them declined to participate to the study (Figure 1). The 34 remaining patients (23 males and 11 females) were randomly allocated by a computer program to the experimental group (EG; n = 16; 11 males, 5 females) or to the control group (CG; n = 18; 12 males, 6 females).
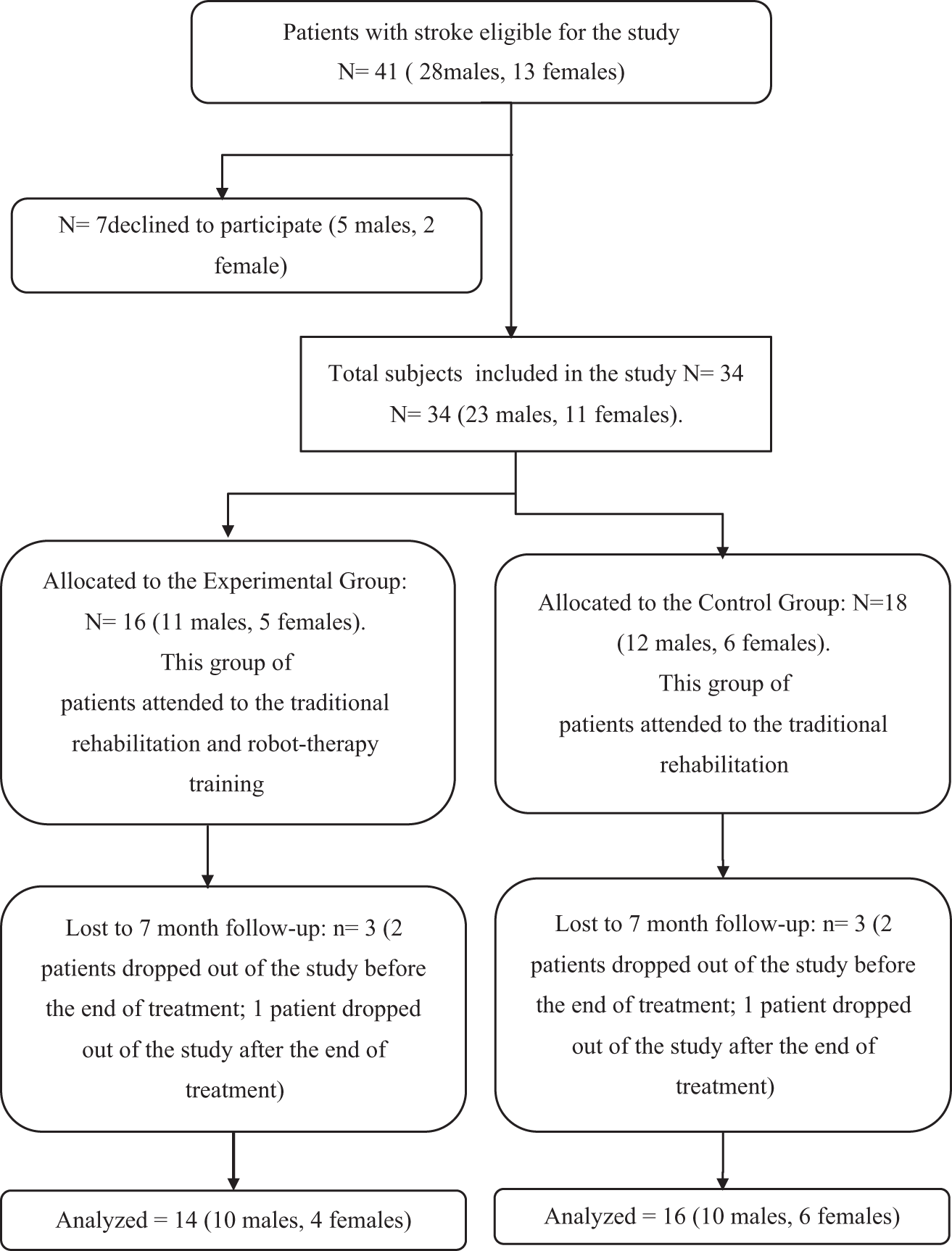
CONSORT flowchart of participants.
The study was approved by the ethics committee for clinical trials of the Provincial Agency for Health Services of Trento, Italy. Informed consent was obtained from all patients and from one family member before randomized assignment. The informed consent from one of the family members was required by ethics committee for clinical trials.
Intervention
This is a dose-matched clinical trial; patients of both groups received an equal dose of rehabilitation treatment. The EG received conventional therapy (for 65% of exercise time) and substitutive robotic treatment (for 35% of exercise time), while the control group (CG) received only conventional treatment (for 100% of exercise time). Patients of both groups received a total daily rehabilitation treatment time of 120 minutes, for 5 days a week and for 5 weeks (in total, 3000 minutes per subject). CG patients performed conventional functional rehabilitation 80 minutes a day (partly based on the Bobath technique and including proprioceptive exercises, functional reeducation, gait training, occupational therapy, and passive and active assisted mobilization of the hand and wrist, but without specifically exercising the upper paretic arm), whereas the upper paretic arm was actively exercised for 40 more minutes with the assistance of a therapist. For the EG, conventional treatment of the upper paretic arm was substituted by NeReBot training for 40 minutes a day (divided into two 20-minute sessions).
NeReBot is a 3-degrees-of-freedom robotic device for upper extremities that can be easily used in the acute and post–acute stroke phase thanks to its portability and to the possibility of being used at bedside.29,30,33 The robot is based on direct-drive wire actuation: 3 actuated nylon cables are used to sustain and move the patient’s forearm, which is fastened on a splint (Figure 2). In this way, a traditional rehabilitation therapy of the upper limb is simulated with imperceptible differences in patient’s sensorimotor experience. NeReBot allows to train the patient on 3-dimensional movements of the arm (flexion and extension, pronation and supination, adduction and abduction, circumduction), not only while sitting but also in the supine position (Figure 2). In this study, NeReBot training was administered under the supervision of a physiotherapist, who defined the exercises (flexion and extension, adduction and abduction, pronation and supination, circumduction) according to the protocol, adapted robot parameters to the patient’s progress and, in agreement with the rehabilitation team, defined the treatment schedule weekly. Each robotic session lasted 20 minutes, and consisted of robotic training interrupted by a 30- to 60-second resting period every 3 to 4 minutes. Patients actively contributed to exercises in all sessions, and were verbally encouraged by the therapist to do so. 33 The same therapist supervised the NeReBot training for all EG patients, whereas a different therapist delivered standard rehabilitation to all patients. Treatment was delivered in an exercise room in the same rehabilitation center for all the subjects during hospitalization.

NeReBot training with the patient in supine position.
Measures
We performed a standard assessment procedure on all the recruited patients using scales of proven reliability, validity, and sensitivity to change during poststroke recovery. All patient assessments were performed by the same blinded clinician trained to the use of the scales, who was not directly involved in the delivery of neither robot-aided nor standard rehabilitation therapy within the study. The clinician that performed the assessments of the rehabilitation treatment was not aware of the type of treatment of the patients, previously assigned by a different physician investigator.
Assessments were done immediately before treatment start (baseline), 5 weeks after treatment start (time T1) and 3 months (time T2) and 7 months (time T3) after the end of treatment.
Measures of primary outcome were Fugl-Meyer, Motor-Functional Independence Measure, and Frenchay Arm Test.
The following impairment, disability, and activity rating scales were used:
Medical Research Council (MRC, range 0 [no muscle contraction] to 5 [normal strength]), 38 used to measure the strength of the paretic arm during shoulder abduction (MRC deltoid), elbow flexion (MRC biceps), elbow extension (MRC triceps), wrist flexion (MRC wrist flexors), and extension (MRC wrist extension).
Fugl-Meyer assessment (FM, range 0-66), 39 used to measure motor impairment of the upper limb; we considered the shoulder/elbow and coordination subsection (FM-SEC = 42/66), the wrist/hand subsection (FM-WH = 24/66), and the total score (FM-tot = 66/66).
Motor-Functional Independence Measure (FIM-mot, range 13-91), used to measure abilities in basic motor ADLs and degree of self-care, and Functional Independence Measure (FIM, range 18-126). Both scales do not provide a direct measure of the function or ability of the upper extremity; nonetheless, they were used to allow comparison with other studies. 40
Modified Ashworth scale (MAS, range 0-5), used to measure muscle spasticity in several upper limb joints. A lower MAS score indicates a lesser augmented muscle tone. 37
Frenchay Arm Test (FA-T, range 0-5), used to evaluate 5 ADLs, reflects both hand and arm functions. 41
Box and Block Test of manual dexterity, used to assess upper limb disability. 42
Tolerability of treatment, evaluated by noting the number of medical complications in the 2 groups (shoulder-hand syndrome, shoulder pains).
The degree of acceptance of robotic training in the EG was evaluated by a visual analogue scale (VAS; 0 = poor tolerance; 10 = high tolerance). Subjects were asked to mark a point on the line describing their average degree of acceptance during robot training.
Statistical Analysis
Descriptive statistics were produced for all variables. Before analyzing each variable, data distribution normality was verified by the Shapiro-Wilk test. Statistical analysis was conducted with the intention-to-treat principle according to the CONSORT statements (Figure 1). Between-group comparisons at baseline were evaluated by Mann-Whitney test for age and t test for time between stroke and treatment start; by χ2 test for sex, stroke side and type, and medical complications. No participants changed group allocation data, because lost patients had dropped out after the baseline assessment that is before posttreatment evaluation. The variables MRC deltoid, MRC biceps, MRC triceps, MRC wrist flexor, MRC wrist extensor, FM-SEC, FM-WH, FM-tot, FIM-tot, FIM-mot, Box and Block Test, FA-T, and MAS were analyzed after rank transformation with a nonparametric analysis of covariance, as proposed by Bathke and Brunner, 43 including in the model the baseline value and the effects treatment, time (posttherapy, 3 and 7 months follow-up) and treatment by time interaction and considering an unstructured variance-covariance matrix. In Brunner’s approach,44,45 the data are rank transformed, but this is not regarded as a technique for the derivation of statistics, as it is with the Conover and Iman method, but as a property of a statistic that can be useful for computational purposes. The statistic used for testing the null hypothesis in term of normalized distribution function in case of small sample size is the so-called analysis of variance type statistic, which has, based on asymptotic theory, an approximate F distribution (with dfN [numerator] and dfD [denominator] degrees of freedom) under the null hypothesis.
The P values for the group comparisons are also reported after Bonferroni’s adjustment to take into account the multiple comparisons due to the variables analyzed. Data analysis was implemented using SPSS version 17.0 (SPSS, Inc, Chicago, IL).
Results
Baseline Evaluation
Out of the 34 patients randomized, 6 patients (3 in the EG and 3 in the CG) were lost to follow-up (Figure 1). Four subjects (2 patients in the EG and 2 in the CG) did not complete the rehabilitation therapy because of a complication (ie, pneumonia [n = 1], death [n = 1] or withdrawal from trial (n = 2) and they performed only one assessment at baseline; 2 subjects (1 subject of EG and 1 of CG) were lost after the second assessment for withdrawal from trial, and they have 2 assessments). Demographic characteristics and baseline motor and functional assessments of the 30 subjects are shown in Table 1. The 2 groups were comparable in terms of age, gender, side of stroke, type of stroke, time between stroke, and rehabilitation treatment start. NeReBot training of EG patients started on average 8.36 days after stroke (SD = 3.3), while the CG started on average 10.13 days after stroke (SD = 2.3).
Patients’ Demographic and Clinical Characteristics at Baseline.
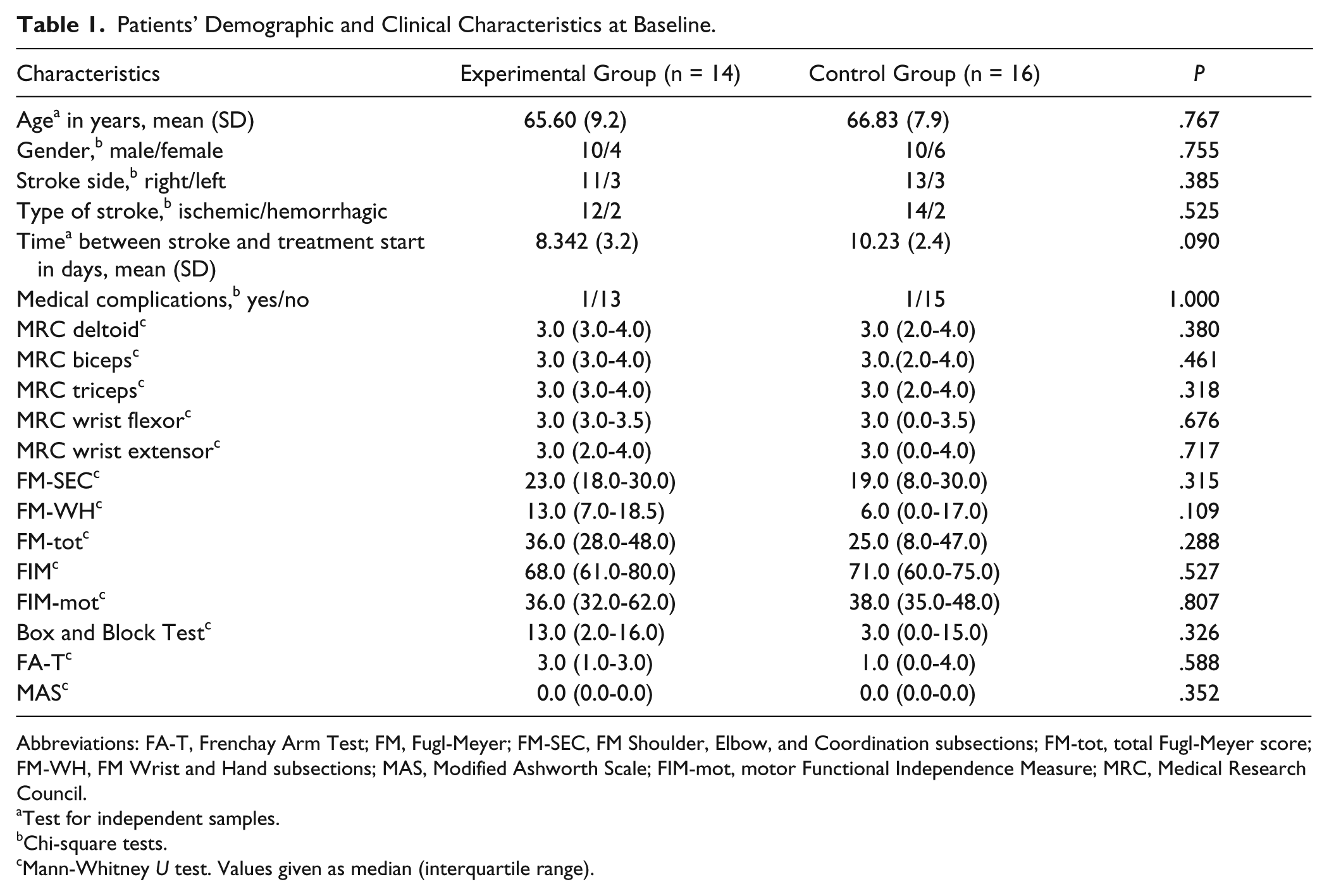
Abbreviations: FA-T, Frenchay Arm Test; FM, Fugl-Meyer; FM-SEC, FM Shoulder, Elbow, and Coordination subsections; FM-tot, total Fugl-Meyer score; FM-WH, FM Wrist and Hand subsections; MAS, Modified Ashworth Scale; FIM-mot, motor Functional Independence Measure; MRC, Medical Research Council.
Test for independent samples.
Chi-square tests.
Mann-Whitney U test. Values given as median (interquartile range).
Effects of Rehabilitation Treatment
Patients of both groups received an equal amount of rehabilitation therapy (3000 minutes per subject). Assessment of EG and CG groups at posttherapy, 3-month follow-up, and 7-month follow-up are summarized in Table 2. Before-after training improvements in motor and functional scales within each group were all statistically significant at the end of robot therapy and the gains at follow-up continued to be statistically significant. In both groups, the MRC test of deltoid, biceps, triceps, wrist flexor, and wrist extensor show strength increase at times T1, T2, and T3, motor impairment and abnormal hypertonia were reduced (FM and MAS results), and basic ADLs and dexterity were improved (FIM-mot, FA-T, and Box and Block Test results). No significant between-group differences were found with respect to demographic characteristics, motor function (ie, MAS, FM, and MRC), dexterity (FA-T and Box and Block test), and ADLs (ie, FIM or FIM-mot) at baseline, postintervention (T1), and at follow-up (T2 and T3).
Nonparametric Analysis of Covariance Including in the Model the Baseline Value, and the Effects Treatment, Time (at the End of Rehabilitation and at 3 and 7 Months After the End of Rehabilitation Treatment) and Interaction Treatment × Time and Considering an Unstructured Variance-Covariance Matrix.
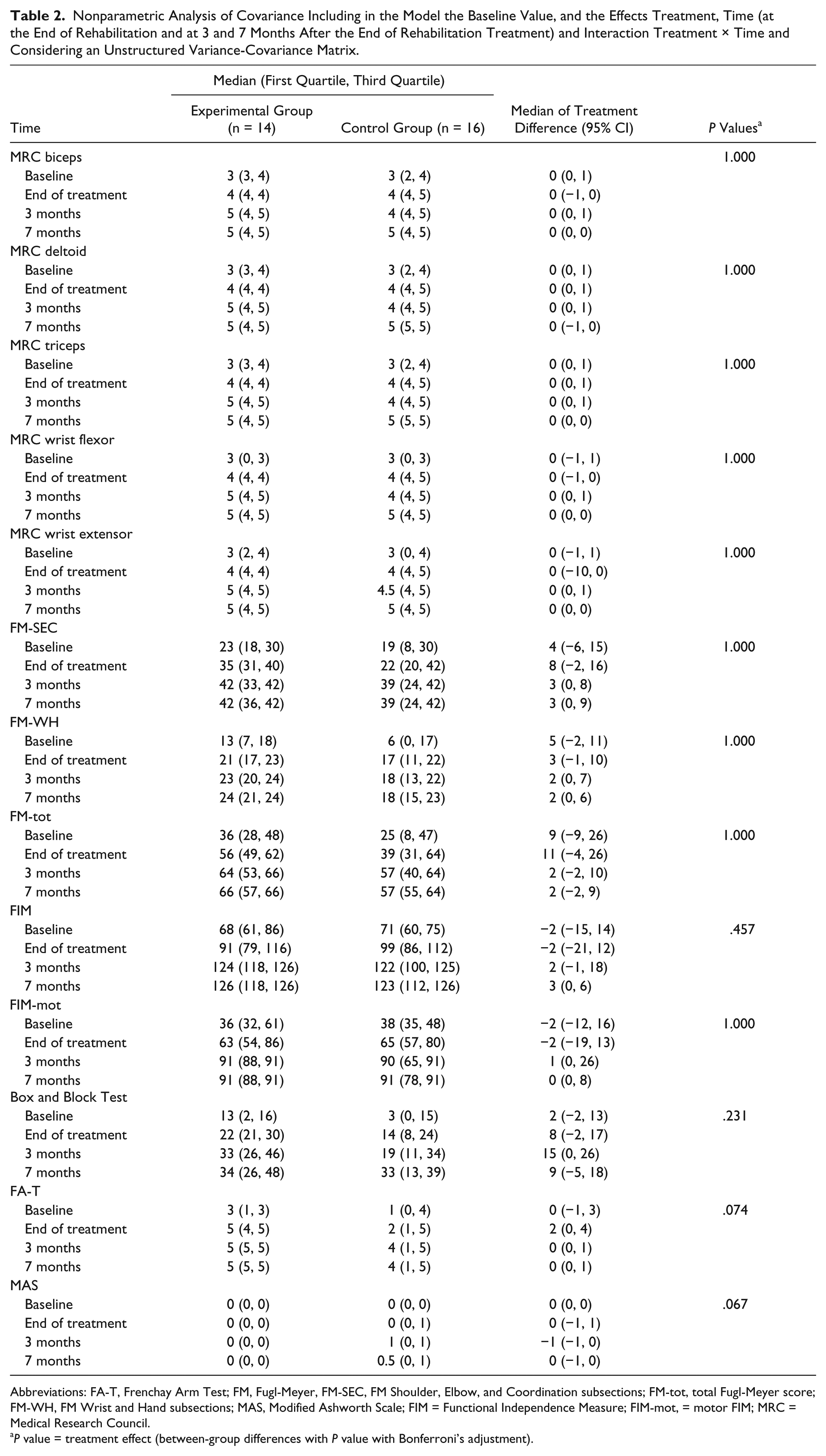
Abbreviations: FA-T, Frenchay Arm Test; FM, Fugl-Meyer, FM-SEC, FM Shoulder, Elbow, and Coordination subsections; FM-tot, total Fugl-Meyer score; FM-WH, FM Wrist and Hand subsections; MAS, Modified Ashworth Scale; FIM = Functional Independence Measure; FIM-mot, = motor FIM; MRC = Medical Research Council.
P value = treatment effect (between-group differences with P value with Bonferroni’s adjustment).
No differences were found between the 2 groups in terms of joint or tendon pain in the shoulder, wrist, or hand, or any other complications, including shoulder-hand syndrome: Only 1 patient of the CG developed a mild complication (shoulder-joint pain) that did not, however, influence performance in the rehabilitation program. The questionnaire administered to the EG patients at the end of robot therapy showed that this form of intervention was well accepted and tolerated by patients (mean score = 8.0/10, question A), as already reported by the first study.31,33 The patients in EG showed a good degree of acceptance of the robotic training with NeReBot evaluated by a VAS (mean score = 8.2/10).
Discussion
In this study, very early robotic therapy (starting between the first and second week after stroke) was investigated and both groups spent comparable time in training (dose-matched therapy) and both groups used active elements of therapy. We tested a protocol which involved the use of robotics as partial substitute for traditional rehabilitation therapy of the paretic upper limb, for a period of 35% of the total daily rehabilitation treatment time. The results of the study demonstrate that robotic therapy by NeReBot, when used as a partial substitute for traditional rehabilitation therapy, is at least as effective as conventional therapy for treatment of patients with hemiparesis after acute stroke, because capable of providing comparatively similar gains on both functional and motor scales. This result may be read as a lack of efficacy of our device. However, as stated in the introduction, our aim was to demonstrate that the NeReBot can be used in partial substitution of conventional therapy while preserving gains on motor and functional scales. This result is very important, as it allows to design rehabilitation protocols in which therapy substitution can be used to reduce costs without losing efficacy of the rehabilitation protocol. On the other hand, since additional robotic training with our device has proven to bring greater gains with respect to conventional therapy,31,46 a proper mix of substitutive and additional robotic therapy would allow to get greater gains but comparable costs with respect to conventional therapy (see below).
The fact that NeReBot therapy was no better than conventional therapy when used in in place of it, may appear to contradict the general finding that robotic therapy can be more effective than other interventions when improving ADLs is the aim of therapy, as reported by the most recent Cochrane Review on upper limb robot-assisted therapy by Mehrholz et al. 21 However, the Cochrane Review did not group studies on the basis of the type of intervention (additional or substitutive). On the other hand, substitutive robotic treatment in the acute phase poststroke proved itself to be no better than conventional therapy, in terms of ADLs, in at least 2 studies (see review 33 ), which is in accordance with the results of this study.
The study suffers from several limitations, including small sample size and dropouts, in particular at follow-up. Another limitation of this study is that, although both groups did not show statistically significant scores at baseline on the different tests, the CG looked much more involved than the EG (see FM-tot, Box and block test and FA-T), and this difference between both groups could have affected the study results. Also, the subjects enrolled in this study, according to the inclusion criteria, had poor/moderate upper extremity motor control as reported in the “Patients” section.
This study suggests that NeReBot can be used in the treatment of patients in acute and post-acute phases of stroke, in replacement of the upper arm portion of traditional treatment. Lo et al 47 showed instead that in patients with moderate-to-severe upper limb impairment 6 months or more after a stroke, robot-assisted therapy did not significantly improve motor function at 12 weeks, as compared with usual care or intensive therapy. In chronic stroke survivors with moderate/severe hemiparesis, Housman et al 48 showed that in 28 chronic stroke survivors with moderate/severe hemiparesis, arm exercise by arm orthosis (Therapy Wilmington Robotic Exoskeleton [T-WREX]) was associated with modest sustained gains at 6-month follow-up when compared with the conventional approach.
As reported by the personnel who have been working with the robotic device, the rehabilitation with a device such as NeReBot does not represent a difficulty for access to the ward, both for inpatient or outpatient treatment. The size of the device is compatible with transport through the doors of patients’ rooms, and it can fit in elevators that are used in the transport of stretchers. Therefore, this device can be easily used at the bedside, with the patient lying or in sitting position. This operating modality requires one to one supervision and can work in the first week of treatment, while 2 or 3 robots can be managed by a single operator in the same room during the course of treatment from the second week on. We tested the 1-to-3 supervision with 1 patient using the robot and 2 healthy subjects simulating robot-assisted movements: The therapist was able to set the robotic exercise in 5 minutes, then he simulated the setting of 2 additional robots; for the rest of the session, the therapist provided supervision to the 3 subjects in the same room.
The use of multiple devices under the control of the same physiotherapist brings an advantage for patients, who can receive further intensive treatment with a reduction of costs proportional to the number of robots that a single operator can handle. On this premise, we proposed 33 that an optimized, cost-effective robotic protocol for patients with acute and postacute stroke may be distinguished into 2 phases, an initial phase of additional robotic therapy, followed, as a next step, by the replacement of conventional therapy with robotic training. Thus, the intensity of treatment would be increased in the recovery phase in which the improvements are likely to be greater. Based on Italian National Healthcare System standard costs, we calculated that the cost of each hour of additional robotic training with one-to-one supervision can be compensated by the savings deriving from 3 hours of therapy substitution with 1-to-3 supervision. 49 In this way, for instance, one could deliver 5 hours of additional NeReBot training to all patients in the first week without increasing treatment costs, provided that 30 hours of conventional treatment of the upper limb are substituted by robotic therapy in the following weeks. Further substitution of conventional therapy would result in reduced costs with respect to standard intervention. On the basis of the results of our clinical trials, we expect such a training protocol to bring greater gains in both motor and functional scales; however, a dedicated randomized controlled trial is needed to test the real viability and efficacy of such protocol. Of course, more investigations are needed to assess the exact dose response to robotic therapy with our device, with the aim of finding the optimal mix of additional and substitutive robotic therapy. Nonetheless, we believe that our approach with the NeReBot in the early stage of recovery may lead to a favorable trade-off between the needs of improving outcome of poststroke rehabilitation and those of reducing treatment and personnel costs. 50
Footnotes
Acknowledgements
We express our deepest appreciation to all subjects who participated in the study, and to Alice Chiasera, MD, from the Neuromotor Rehabilitation Unit, Eremo Hospital, Arco, Trentino, Italy, who contributed to patients’ enrollment. We also thank Joanne Fleming for her help with the text.
Authors’ Note
All authors contributed equally to the article. Statistical analysis was performed by Dr Masiero with our statistics consultant (Dr A. C. Frigo from University of Padua). Registered with ClinicalTrials.gov; NCT01102309, “Robot-assisted rehabilitation of the upper limb in acute and subacute poststroke patients” ![]() .
.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Autonomous Province of Trento, Italy, within the 2007-2008 Health Research Program (Grant Doc No. 2008-S143-00178).
