Abstract
Background. Robotic-assisted walking training after stroke aims to enable highly impaired patients to walk independently, but results have been mixed. Objective. The authors aimed to identify the characteristics of patients who may be most likely to benefit. Methods. A total of 48 participants with motor and gait dysfunction following subacute stroke were stratified by the motricity index into high (<29) and low (≥29) motor impairment groups. Each arm was randomized to a robotic or control group (RG or CG) at a mean of 20 days after stroke. All patients underwent 2 therapy sessions per day, 5 days per week for 3 months. Those in the RG underwent 20 sessions of robotic-assisted gait training in the first 4 weeks of inpatient therapy using controlled endpoint trajectories and abbreviated conventional therapy, whereas the CG received only conventional gait training. The primary outcome was the functional ambulation category (FAC), and secondary measures were the Rivermead mobility index (RMI) and 6-minute walking distance, all evaluated at hospital admission and at discharge. Results. The lower motricity group assigned to an electromechanical device significantly improved in the FAC (P < .001), RMI (P = .001), and walking distance (P = .029). Conventional and robotic therapies were equivalent in the higher motricity arm. Conclusion. Robotic therapy combined with conventional therapy may be more effective than conventional therapy alone in patients with greater motor impairment during inpatient stroke rehabilitation.
Introduction
Independent walking after stroke is one of the chief goals of rehabilitation to improve functional activity, 1,2 social participation, and perceived quality of life. 3 In patients with ischemic stroke who have been admitted to rehabilitation hospitals, the recovery of some degree of ambulation typically occurs in nearly 55% of patients. 1
Treadmill training with partial body weight support (BWS), which has limited evidence of efficacy in properly powered trials for stroke 4,5 and spinal cord injury, 6 led to the development of electromechanical steppers. Two commercial robotic gait machines are available to conduct ambulation training in hemiparetic patients: a Gait Trainer (GT), which controls endpoint trajectories (GT II, Rehastim, Berlin, Germany) 7,8 and the Lokomat, which integrates a robotic exoskeleton on a treadmill (Hocoma Medical Engineering Inc, Zurich, Switzerland). 9 The GT and Lokomat have been used for stroke 7,10 and spinal cord injury. 11,12 The high purchase cost, uncertainty about efficacy, and skepticism by some members of the rehabilitation team 13 limit use during inpatient care. A recently updated Cochrane review found that electromechanical-assisted gait training (Lokomat or GT) in combination with physiotherapy increased the odds of becoming independent in walking by the functional ambulation category (FAC) but did not increase walking velocity or walking capacity significantly more compared with those who receive conventional therapy alone. This report included nonambulatory and ambulatory patients with subacute and chronic stroke. 14
Hidler and colleagues 15 demonstrated that robotic-assisted therapy yielded less improvements for those who ambulate, whereas another study that investigated nonambulatory individuals found no differences between conventional and robotic-assisted training. 16 Conversely, Pohl and colleagues 17 found that robotic-assisted therapy plus physiotherapy achieved better gait ability for nonambulatory patients with stroke. Similar results were also found by Tong and colleagues 18 for nonambulatory patients, with greater improvement in mobility and walking speed achieved by robotic therapy in comparison with overground gait training. It is still unclear whether certain types of patients may benefit from robotic-assisted training. 19
We carried out a trial of patients with subacute stroke who could not walk independently at entry and stratified them by initial level of impairment to determine whether this parameter could help define the patients who might benefit more from the use of robotic therapy using the GT. We hypothesized that dependence in walking may be reduced in more impaired patients by training with the aid of robotic devices.
Methods
We screened all patients with recent stroke who were admitted to our rehabilitation unit during their first week of hospitalization. Immediately after screening, enrolled patients were stratified, randomized, and evaluated. The study was conducted between the second and sixth weeks poststroke. A second evaluation occurred at hospital discharge.
Inclusion criteria were as follows: hemiplegia/hemiparesis in the subacute phase (see Table 1) with significant gait deficits (FAC < 3) 20 caused by a first-ever stroke, lesions that were confirmed by computed tomography or magnetic resonance imaging, and age between 18 and 80 years.
Summary of Patient Characteristics a
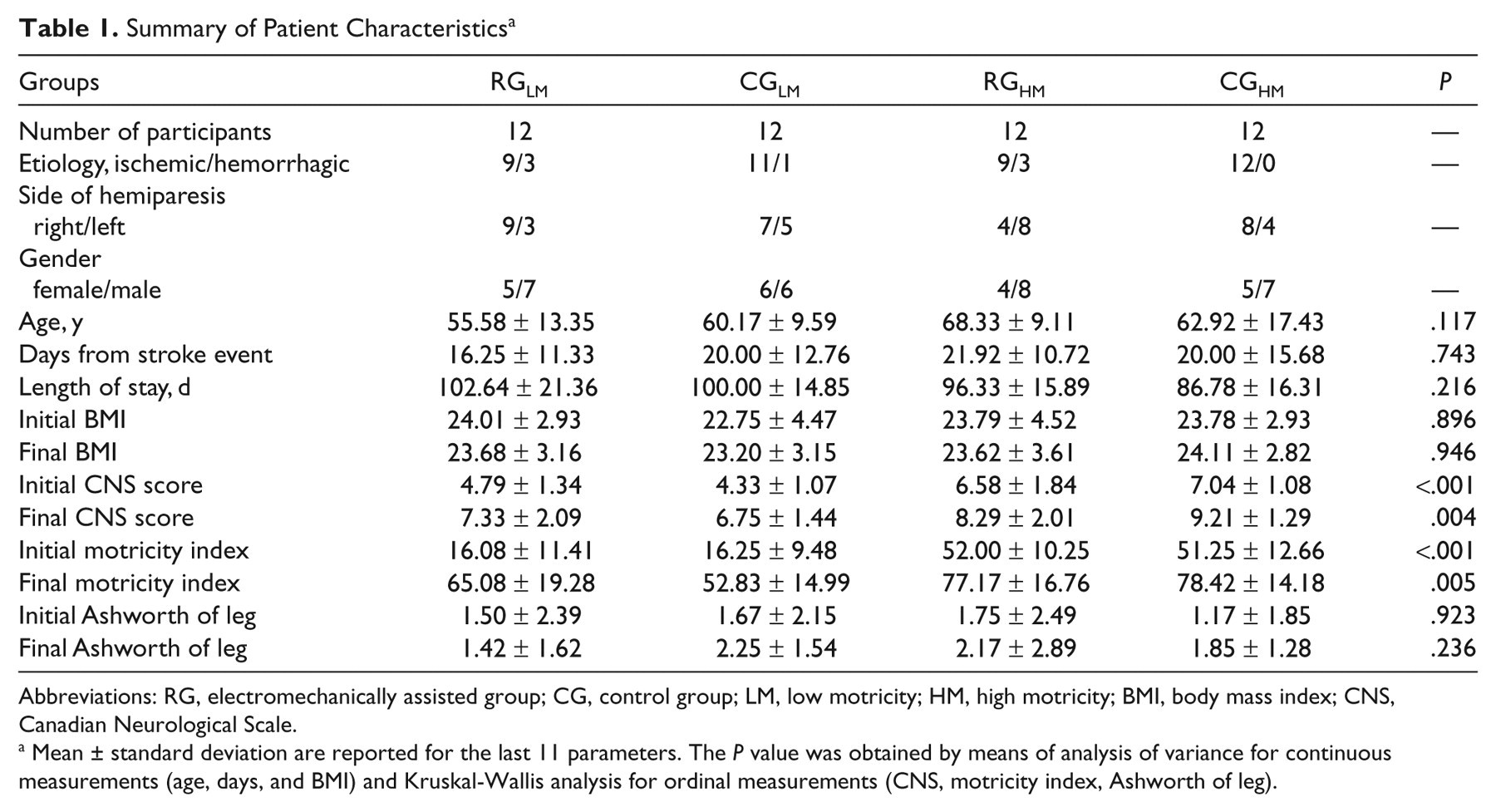
Abbreviations: RG, electromechanically assisted group; CG, control group; LM, low motricity; HM, high motricity; BMI, body mass index; CNS, Canadian Neurological Scale.
Mean ± standard deviation are reported for the last 11 parameters. The P value was obtained by means of analysis of variance for continuous measurements (age, days, and BMI) and Kruskal-Wallis analysis for ordinal measurements (CNS, motricity index, Ashworth of leg).
Exclusion criteria included the presence of subarachnoid hemorrhages, sequelae of prior cerebrovascular accidents or other chronic disabling pathologies, orthopedic injuries that could impair locomotion, spasticity that limited lower-extremity range of motion to less than 80%, sacral skin lesions, Mini-Mental State Examination (MMSE) score < 24, 21 and hemispatial neglect, as evaluated by a neuropsychologist. The protocol was approved by the local independent ethics committee, and all participants provided written informed consent.
Patients were stratified according to their leg motor score assessed by the motricity index (MI), a scale with well-known predictive value for ambulation. 22 We used a cutoff score of ≤29 to define a low motricity (LM) group of patients versus a group of patients with high motricity (HM; MI > 29). This threshold corresponds to (1) palpable contraction for all 3 joints without movement, (2) reduced movement in 2 joints and absent in the third, or (3) complete movement in 1 joint, contraction in another one, and no movement for the third joint. This threshold is a predictor of independent walking at 6 months. 23
At admission, 24 eligible participants with MI ≤ 29 entered the first arm of the study (LM), and 24 eligible participants with lower limb MI > 29 entered the second arm (HM). They were randomly allocated (per 2 blocked randomization lists, generated electronically by www.random.org) into the robotically assisted group (RG) or the control group (CG; see Figure 1). Thus, we defined 4 subgroups: RGLM, RGHM, CGLM, and CGHM.
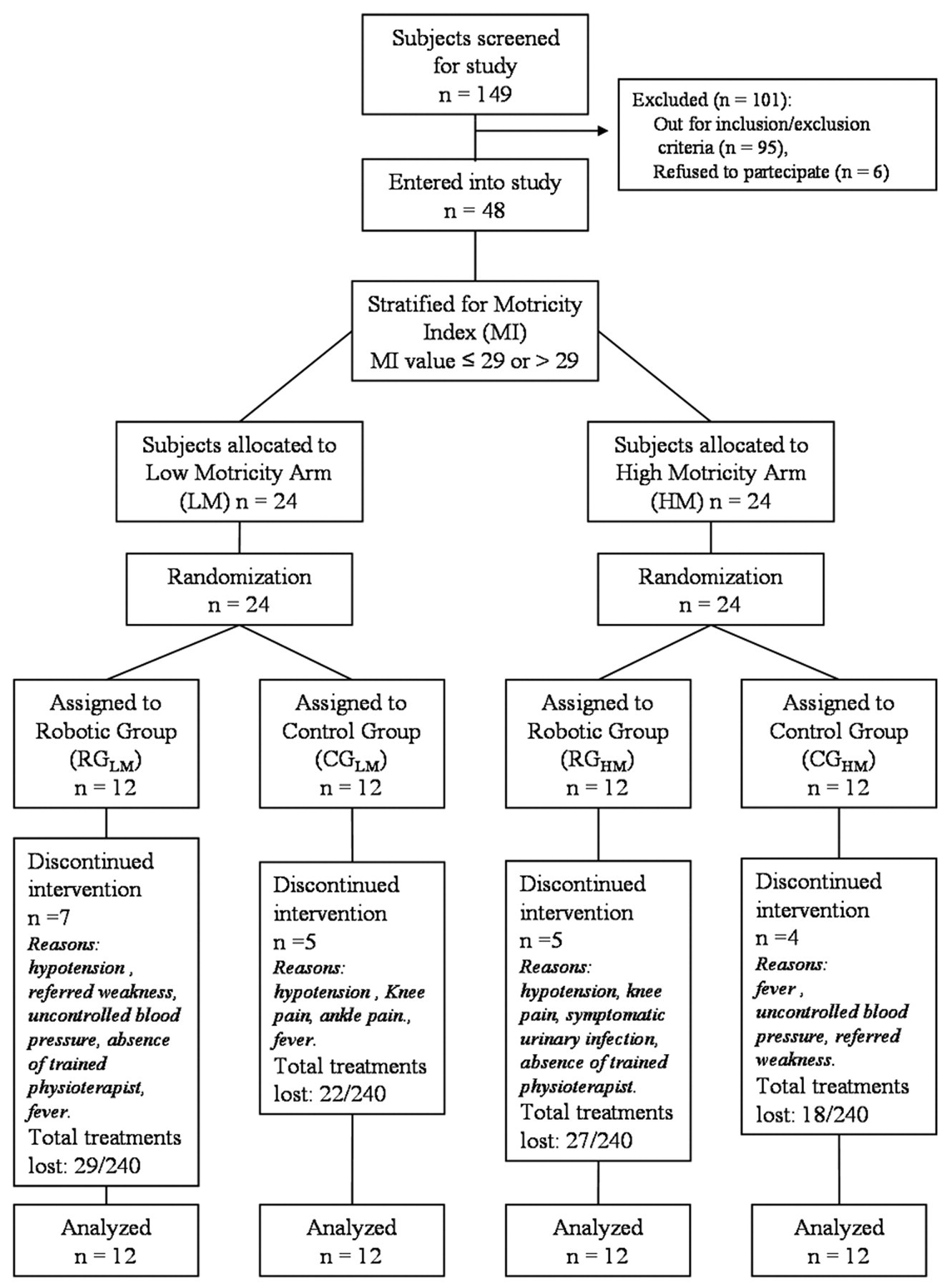
Study flow chart.
All groups had our standard rehabilitation treatment (nearly 3 h/d for 5 d/wk). In particular, 2 daily physiotherapy sessions were performed by all groups. After 1 week postadmission, RG participants performed 20 robotic sessions (5 times per week for 4 weeks) instead of a second session of standard physiotherapy; this session lasted 40 minutes, 20 of which were active GT therapy (the remaining 20 minutes were allocated for the patient’s preparation, parameter setting, and rest breaks as needed). The duration of robotic treatment was 4 weeks, in accordance with previous studies. 16-18
During all RG sessions, 1 physiotherapist manually assisted knee flexion and extension and verbally encouraged the patients to perform the task with correct posture, when necessary. However, 2 therapists were necessary to settle severely affected patients in the harness. Each RG session began after setting the parameters, based on motor ability and the patient’s comfort. Step lengths ranged from 38 to 44 cm. The walking speed was selected to be around 1 to 1.5 km/h at the first GT session and was increased as soon as possible in accordance with comfortable gait for each patient. 23 BWS was 0% to 50% of body weight. The BWS was measured while the patient was harnessed, after supporting straps were stretched, and after the nonparetic leg was extended and aligned to the paretic leg; hands were positioned on the handrail solely for balance. The BWS was selected and adjusted during therapy in line with 3 principles: a progressive decrease in BWS during rehabilitation, patient comfort, and the patient’s ability to extend his hip and sufficiently bear weight on the paretic leg. 24 During the GT session, a rest period was possible, if required by patients.
After 1 week of admission, the CG patients performed 2 daily physiotherapy sessions. One of them was dedicated to walking training, consisting of 20 sessions of 40-minute therapy (5 times per week), 25 instead of a second session of standard physiotherapy. In light of the patient’s ability, the walking therapy was focused on trunk stabilization, weight transfer to the paretic leg, and walking between parallel bars or on the ground. If necessary, the patient was helped by 1 or 2 therapists and walking aids.
The standard physiotherapy, shared by both groups, was focused on the facilitation of movements on the paretic side and upper-limb exercises, and improving balance, standing, sitting, and transferring.
The primary outcome measure of the study was walking ability (as measured by FAC), 20 assessed after the 4 weeks of treatment and at the end of the entire rehabilitation program. The remaining outcomes included assessments of mobility function and ability level, evaluated by lower-leg Ashworth (sum of scores for hip, knee, and ankle), Rivermead mobility index (RMI), 26 MI, 22 Trunk Control Test (TCT), 27 Canadian Neurological Scale (CNS), 28 Barthel index (BI), 29 Rankin Scale (RS), 30 6-minute walk test on a 20-m path (6mWT), 31 and 10-m walk test at a self-selected speed (10MWT). 32 A physician, blinded to the treatment, took measurements immediately after random allocation (which occurred in the first week after admission), after 4 weeks of the intervention, and at hospital discharge. These data were collected in the laboratory of our rehabilitation unit. Finally, we quantified the parameter “effectiveness” on all scales. Effectiveness reflects the proportion of potential improvement that was achieved during rehabilitation, calculated as [(discharge score − initial score)/(maximum score − initial score)] × 100. 33,34 Thus, if a patient achieved the highest possible score after rehabilitation, the effectiveness was 100%.
Statistical Analysis
An intent-to-treat analysis was performed. The mean and standard deviation of all outcome and effectiveness measurements were calculated. To assess the eventual initial differences in patient characteristics among the 4 subgroups, analysis of variance and Kruskal-Wallis analysis were performed on continuous measures and ordinal measures, respectively. Post hoc analyses were then performed using a Bonferroni correction.
We did not plan a power analysis before the beginning of the study. However, our sample size is similar to that used in some other studies in the same field, such as that of Hornby et al, 19 in which 48 patients were divided into 4 groups—robotic versus therapist assisted locomotor training—stratified for initial gait speed, similarly to our protocol.
Because the general effectiveness of this robotic device was already investigated in the Cochrane review, this study was designed to test 2 null hypotheses. Is the robotic GT useful in LM, nonambulant patients? Is it more useful in HM, nonambulant patients?
To determine the statistical differences between RGLM and CGLM (P LM), and RGHM and CGHM (P HM), parametric statistics (2-tailed Student’s t test) were used for continuous outcome measures (10MWT, 6mWT, evaluated only at discharge), and nonparametric statistics (Mann-Whitney U test) were used for ordinal outcome measures (scale scores, all evaluated at admission and discharge). To assess improvements during rehabilitation, as evaluated by scale scores, the Wilcoxon signed rank test was used for each subgroup. To determine which factor affected the effectiveness—that is, the proportion of potential improvement—a 3-way analysis of variance was performed for each scale.
To compare our results with those of the Cochrane reviews, 14 we evaluated the effects of the primary factor (RG vs CG and HM vs LM) on all patients, considering the level of motricity as a factor instead of using it to stratify patients. In particular, a forward-stepwise binary logistic regression was performed, and odds ratios (ORs, and their confidence intervals, CI95%) were calculated to determine the principal effects on all patients with regard to the possibility of walking independently at discharge (FAC > 3). For all tests, the significance level was set at .05.
Results
Between January 2008 and August 2009, we evaluated 54 eligible patients from 149 screened patients (of the excluded patients 38 had neglect or MMSE < 24, 20 had prior cerebrovascular accidents, 20 were older than 80 years, 11 had FAC ≥ 3 at admission, and 6 patients presented with severe comorbidity). We enrolled 48; 6 refused to give consent. The mean onset-to-admission interval was 19.54 ± 12.53 days (range 5 to 53 days) after the onset of stroke, and the mean length of hospital stay 96.88 ± 17.64 days. We did not use the length of stay as an outcome measure because it is also related to nonmedical problems, such as social ones. The length of stay did not differ among subgroups (P = .216), so each group underwent nearly the same amount of treatment.
In all, 24 patients were assigned to the LM group (age range between 36 and 77 years, 11 women, 46%), and the remaining constituted the HM group (age range between 29 and 80 years, 9 women, 38%). According to the study design, there were no statistically significant differences between groups, except for severity of stroke, as measured by CNS and MI (see Table 1).
RG patients performed 424 of 480 scheduled GT therapy sessions (88.34%); no differences were observed between RG subgroups. The number of those who discontinued treatment was similar in the CG and RG groups (see Figure 1). Some side effects developed during GT therapy. In particular, in 8 sessions involving 3 patients (13%), the physiotherapist shut off the electromechanically assisted therapy because of severe and symptomatic hypotension; 1 patient experienced knee pain in the paretic leg that required 1 day of rest and the use of a knee pad. The number of patients who experienced adverse events were 4 for the RG and 3 for the CG group (OR = 1.17; P = .683). A total of 16 GT sessions were not performed because of the absence of trained physiotherapists. In 9 sessions, patients refused to perform GT therapy because of perceived weakness. Uncontrolled blood pressure, fever, and urinary infection were contraindications for GT therapy in 22 sessions.
At the end of the 4 weeks of therapy, the mean FAC value was similar among RGLM (2.6 ± 0.9), RGHM (2.2 ± 0.8), and CGHM (2.5 ± 1.0), and lower for CGLM (1.2 ± 0.8). Kruskall-Wallis analysis highlighted a significant difference among the 4 subgroups (χ2 = 13.03; P = .005), and post hoc analysis revealed differences only between the 2 LM subgroups (P = .002) but not between the 2 HM subgroups (P = .429). At this step, the effectiveness of treatment on ambulation ability, calculated on FAC, showed a significant difference between the 2 main groups, related to treatment (F = 5.85; P = .020), and especially for the interaction between treatment and motricity impairment (F = 8.08; P = .007), whereas the main effect of motricity impairment was not significant (F = 1.73; P = .195). Post hoc analyses showed that the effectiveness of RGLM (50.8 ± 17.8%) was significantly higher than that recorded for CGLM (25.0 ± 15.1%; P = .001), whereas no significant differences were recorded between RGHM (43.3 ± 16.7%) and CGHM (45.4 ± 18.3%; P = .773).
Similar results were found at hospital discharge, with a significant difference on the FAC scores among the 4 subgroups (χ2 = 15.86; P = .001). Post hoc analysis showed a difference between LM subgroups (P = .001) but not between HM subgroups (P = .752). At the end of rehabilitation, we observed improvement in all groups on all scales (P < .05), with the exception of the Ashworth score.
At discharge, FAC values (primary outcome measure) were higher in RGLM than in CGLM (FAC, P LM = .001; see Figure 2). Similarly, some of the secondary outcome measures were significant (RMI, P LM = .001; TCT, P LM = .015; BI, P LM = .006; RS, P LM = .009; 6mWT, P LM = .029; see Figure 3). In particular, the mean distances walked in 6 minutes were 156 ± 78 m for RGLM and 91 ± 35 m for CGLM.
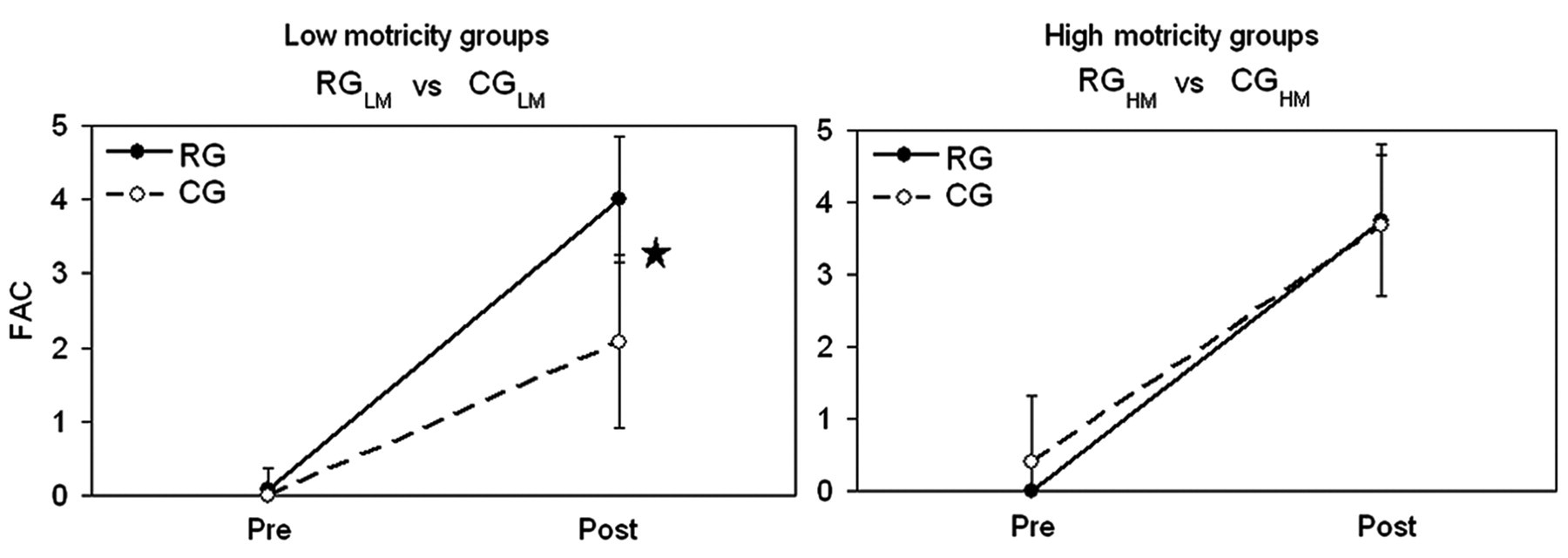
Primary outcome measures: mean and standard deviation at hospital admission (pre) and discharge (post) of Functional Ambulatory Category (FAC), selected as primary outcome measure. Left panel: low motricity groups. Right panel: high motricity groups. Filled dots and solid lines for RG, robotic group; empty dots and dotted lines for CG, control group. The star indicates a statistically significant difference (P < .05) between RG and CG.
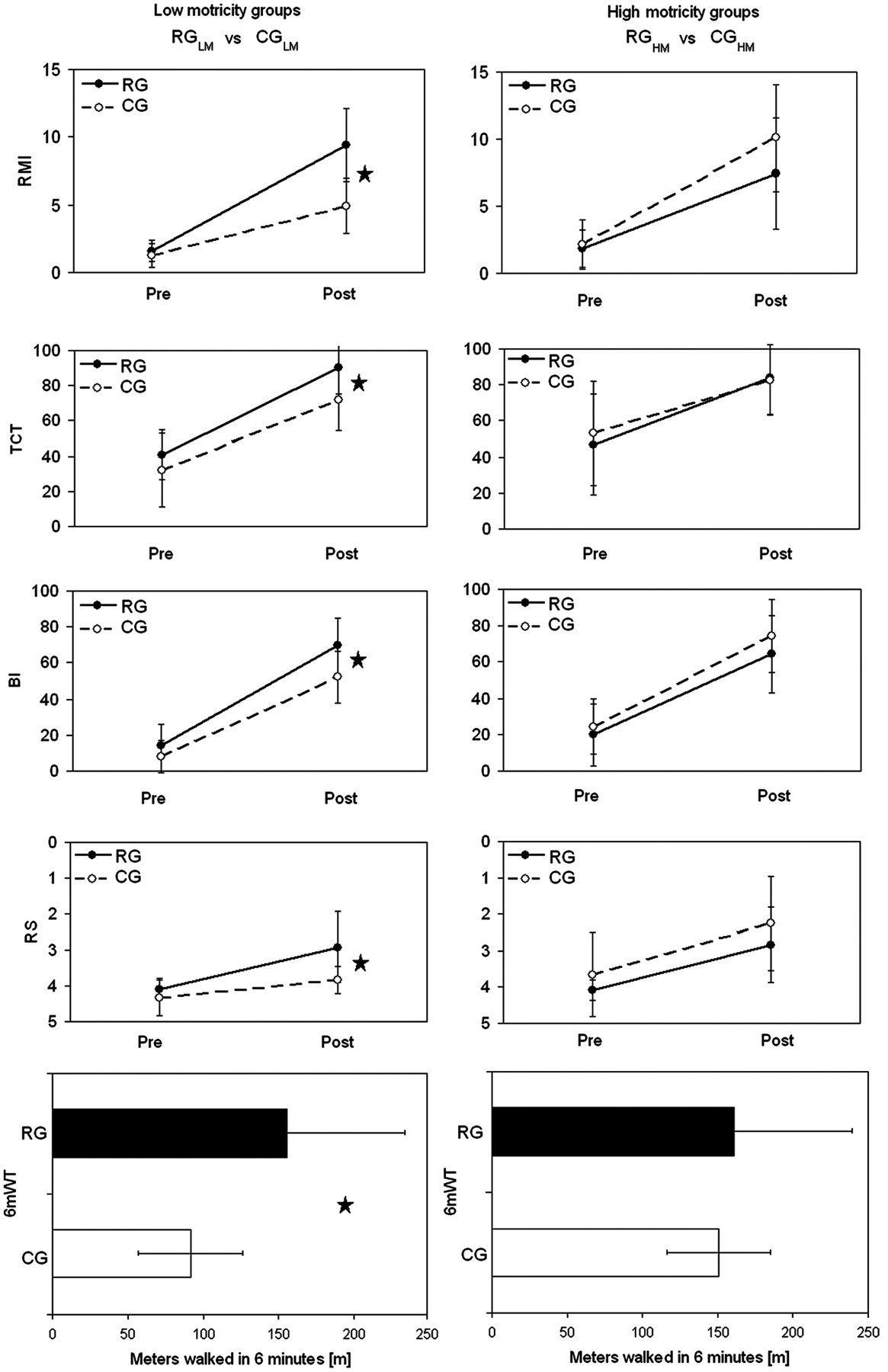
Secondary outcome measures: from above to below—mean and standard deviation at hospital admission (pre) and discharge (post) of Rivermead mobility index (RMI), Trunk Control Test (TCT), Barthel Index (BI), Rankin Scale (RS), meters walked in 6 minutes (6mWT, post). Left panels: mean and standard deviation at hospital admission (pre) and discharge (post) of secondary outcome measures for low motricity groups. Right panels: corresponding mean and standard deviation for high motricity groups. Filled dots or boxes and solid lines for RG, robotic group; open dots or boxes and dotted lines for CG, control group. The star indicates a statistically significant difference (P < .05) between RG and CG.
The final score differences between RGLM and CGLM participants for MI (P LF = .132) and the 10mWT did not differ (RGLM, 31.6 ± 13.8 s vs CGLM, 32.8 ± 10.8 s; P LM = .826, corresponding to walking speeds of 0.36 ± 0.11 m/s for RGLM and 0.37 ± 0.27 m/s for CGLM). The primary and secondary outcome measures did not differ between RGHM and CGHM (P HM > .05) for all administered scales and the 2 walk tests. The mean walking speeds for the 10MWT was 0.49 ± 0.21 m/s for RGHM and 0.52 ± 0.30 m/s for CGHM. The mean distances walked in 6 minutes were 161 ± 89 m for RGHM and 151 ± 89 m for CGHM (Figure 3). Higher values of effectiveness were observed for RGLM versus CGLM (Table 2).
Effectiveness: Proportion of Potential Improvement a
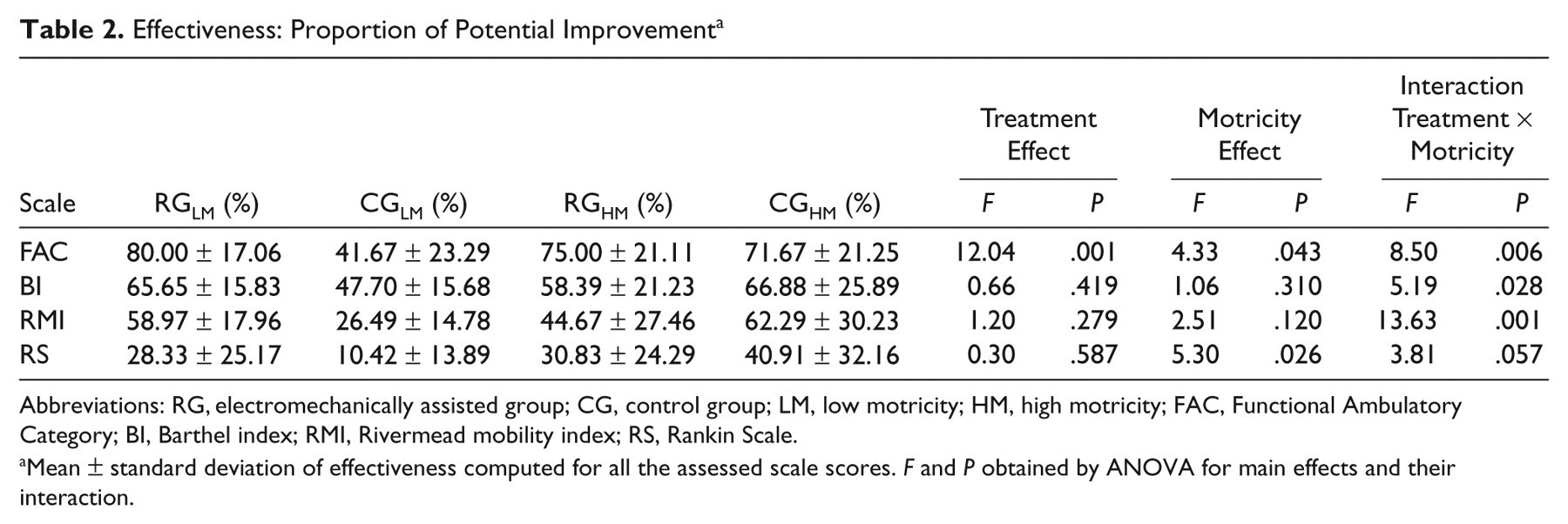
Abbreviations: RG, electromechanically assisted group; CG, control group; LM, low motricity; HM, high motricity; FAC, Functional Ambulatory Category; BI, Barthel index; RMI, Rivermead mobility index; RS, Rankin Scale.
Mean ± standard deviation of effectiveness computed for all the assessed scale scores. F and P obtained by ANOVA for main effects and their interaction.
At discharge, the following patients achieved independent walking (FAC > 3): 10/12 (83%) of RGLM, 2/12 (17%) of CGLM, 9/12 (75%) of RGHM, and 8/12 (67%) of CGHM. By logistic regression, the data demonstrated a significant effect of therapy (OR = 5.32; CI95% = 1.48 to 19.06; P = .010) but not of motricity (P = .110) or their interaction (P = .615). The overall percentage of prediction by the model was only 68.8%. Conversely, by analysis of variance on effectiveness, the interaction between treatment and MI was significant for FAC, BI, and RMI and nearly significant for RS (see Table 2).
Discussion
At hospital discharge, at a mean of 98 days after entry, all patients improved walking ability (FAC), motricity (MI, TCT), motor abilities (RMI), walking capacity (6mWT), ability (RS), and activities of daily living (BI). Our study suggests that the efficacy of robotic end-effector gait training therapy in stroke rehabilitation may be higher for patients with severe motricity impairments when it is combined with conventional therapy. This finding is likely a result of the positive effect of walking training that is augmented by the GT in terms of intensity and repetition for LM group. In fact, because of the severe disability of these patients, physiotherapists experienced overstraining on overground conventional therapy 24 because they often work in suboptimal postures for long periods, 35 and patients experienced discomfort and fear of falling when asked to perform many strides on the floor.
For patients with HM, there was no difference in improvement between robotic-assisted (RGHM) and conventional (CGHM) treatments. These patients had greater voluntary motor function in the affected limb that probably also allowed more intensive training overground. This is also suggested by the nonsignificant differences in walking speed and 6-minute walk distance for the HM subgroups.
By logistic regression analysis, we observed a high probability of independent walking recovery in the robotic-trained group, independent of their motor impairment. The model, however, explained approximately two thirds of cases. Conversely, by analysis of variance on the proportion of potential improvement, the interaction between the type of treatment and motricity impairment was significant.
Our results might be in disagreement with those of Hidler and colleagues, 15 which found higher efficacy of conventional gait training in patients who could walk for at least 5 m without physical assistance, and with those of Hornby and colleagues, 19 who noted a lack of interaction between the chief factors of therapy and initial locomotor impairment and demonstrated the superiority of therapist-assisted locomotor therapy in chronic ambulatory patients. In our study, the interaction between therapy and initial motor impairment was significant with regard to the proportion of potential motor improvement. This difference might be attributed to the fact that our patients had subacute stroke and were not ambulatory at the start of rehabilitation (for this reason, we used MI, not locomotor function, to stratify participants).
Moreover, our results should be considered with those of Hornby et al, 19 suggesting that in severely affected nonambulatory patients, robotic therapy could be used as an adjunct to abbreviated conventional therapy to improve the effectiveness of rehabilitation, but in less-affected nonambulant patients, the benefits of the 2 therapies are similar, and for ambulant chronic patients, therapist-assisted locomotor therapy is more effective alone than in conjunction with robotic therapy. This scenario is consistent with the results of a Cochrane analysis, 14 which concluded that the use of electromechanical devices for gait rehabilitation increases the likelihood of walking independently in patients with subacute stroke (OR = 2.56) but not in patients with chronic stroke (OR = 0.63).
Our results are consistent with small studies that have shown the efficacy of electromechanical GT in nonambulatory patients with stroke, 17,18,25 and we highlight the impact in patients with poor muscle activation. During our robotic-assisted therapy, the mean number of steps practiced by robotic-assisted patients was around 1000, similar to the values reported in the literature, 17 whereas Lang and colleagues 36 reported 350 steps performed in a session of conventional therapy addressing gait. Electromechanically assisted gait training can be used for cardiovascular reconditioning of extremely deconditioned patients after bedrest that reduced aerobic capacity. 37
In regard to safety, the GT caused pain at the knee and ankle levels as often as conventional therapy. Only 1 therapist may be needed during the GT session, 17,18,25 but it is preferable to use 2 therapists to set severely affected patients up in the harness. Of note, 6 out of 54 eligible patients (11.11%), mostly older women, did not consent to be treated with GT, essentially because of a fear of robotic devices.
The major limitation of our study is its small sample size by subgroups and our inclusion and exclusion criteria. Also, secondary outcome measures in our study were assessed only at the beginning and end of the entire rehabilitation pathway, which was a mean of 2 months longer than GT therapy.
Future research trials should examine the amount of training that is needed to achieve motor improvement with regard to level of strength and motor capacity. Moreover, future studies could investigate the role of early mobility training using electromechanically assisted gait training to reduce inpatient rehabilitation duration and to increase the proportion who return to home or community.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Italian Ministry of Health (grant
