Abstract
Background
Motor fatigability (i.e. contraction-induced reduction in muscle strength) from a concentric task associate stronger to walking and perception of fatigue in persons with multiple sclerosis (pwMS), compared with an isometric task. However, the central and peripheral contributions of motor fatigability between these tasks have not been investigated.
Objective
Compare the central and peripheral contributions of motor fatigability in the knee extensors in a sustained isometric fatigability protocol versus a concentric fatigability protocol and in pwMS versus healthy controls (HCs).
Methods
Participants (n=31 pwMS; n=15 HCs) underwent neuromuscular testing before and immediately after two knee extensor fatigability tasks (sustained isometric and concentric) in an isokinetic dynamometer. Neuromuscular testing of fatigability consisted of maximal voluntary contraction, voluntary activation (central/neural contributor), and resting twitch (peripheral/muscular contributor) determined by the interpolated twitch technique.
Results
Sustained isometric and concentric fatigability protocols resulted in motor fatigability for both pwMS and HCs, with no between-protocols differences for either group. Regression analysis showed that motor fatigability variance in pwMS was mainly attributed to central fatigability in the sustained isometric protocol and to both central and peripheral fatigability in the concentric protocol. In HCs, the variance in sustained isometric and concentric fatigability were attributed to both peripheral and central fatigability.
Conclusion
Central and peripheral contributions of motor fatigability differed between sustained isometric and concentric protocols as well as between pwMS and HCs. These between-protocol differences in pwMS provide a neuromuscular dimension to the reported difference in the strength of associations of concentric and isometric tasks to walking and perception of fatigue in pwMS.
Introduction
Multiple sclerosis (MS) is a chronic immune-mediated disease of the central nervous system (CNS). 1 Among the most prevalent symptoms in MS is fatigue, which affects approximately two-thirds of all persons with MS (pwMS)2,3 and is considered one of the most disabling symptoms by pwMS.4,5 Furthermore, a relation between changes in fatigue and disability status in pwMS have been reported. 6 Because fatigue is a complex phenomenon, a unified taxonomy has been proposed by Kluger et al 7 (further elaborated by Enoka and Duchateau 8 ) to differentiate between the perceptions of fatigue and performance fatigability. Perceptions of fatigue have been defined as “subjective sensations of weariness, increasing sense of effort, mismatch between effort expended and actual performance or exhaustion (p. 411),” 7 and it is generally assessed via self-report using questionnaires such as the Modified Fatigue Impact Scale (MFIS) and the Fatigue Severity Scale (FSS). 9 Performance fatigability is defined as “the magnitude or rate of change in a performance criterion relative to a reference value over a given time of a task performance or a measure of mechanical output (p. 411)” 7 and is often quantified as the reduction in an objective performance test. 7 Specifically, motor fatigability is defined as “the rate of change in motor performance” such as muscle strength, 7 and most MS studies have investigated motor fatigability at the body function level of the International Classification of Functioning (ICF). 10 Nonetheless, no gold standard protocol exists for the assessment of motor fatigability at the body function level of the ICF, why a plethora of divergent protocols exists, differing in terms of contraction type (sustained vs concentric), duration of performance task (15-180 s), number of contractions (fixed or to exhaustion), and tested muscle groups (upper vs lower extremities). 10 Furthermore, only few studies have compared the effects of different protocols on motor fatigability, with further information elaborated below.
A systematic review reported increased upper- and lower-extremity motor fatigability in pwMS when compared with healthy controls (HCs),10,11 with the changes in neuromuscular function leading to increased motor fatigability (ie, reduction in muscle strength) being attributed to an interaction of motor fatigability in the nervous system (central contributor) and in the muscular system (peripheral contributor). 10 The majority (n = 15) of these studies have evaluated the upper extremity, 10 with 4 of them assessing neuromuscular function in pwMS and HCs using sustained isometric fatigability protocols.12-14 Their findings were somewhat divergent, with the central contribution of fatigability reported to be larger in pwMS12-14 or similar in pwMS and HCs 15 and with the peripheral contribution of fatigability reported to be larger in pwMS, 12 larger in HCs, 15 or similar in pwMS and HCs.13,14 In contrast, only a few studies (n = 2) have evaluated the lower extremity 10 (findings are reported below), despite its crucial role for mobility. 16
Among the methodological approaches being used to elucidate the central and peripheral contributions of motor fatigability in pwMS, the application of the interpolated twitch technique (ITT) is common. 10 The advantage of the ITT is that it provides a proxy measure not only of the central/neural input that reaches a given muscle, but also of the force producing capacity of the examined muscle. The existing ITT studies that have examined lower-extremity motor fatigability in pwMS with sustained isometric contraction protocols have reported a greater contribution from central fatigability and smaller or comparable contribution from peripheral fatigability in pwMS when compared with healthy controls (HCs). 17 However, to our knowledge, only 1 study has investigated the central and peripheral contributions of motor fatigability using a concentric protocol in pwMS, reporting contribution from central but also peripheral fatigability. 18 However, no studies have compared the central and peripheral contributions of motor fatigability in pwMS using different protocols (eg, sustained isometric vs concentric muscle contractions). As the ITT was also applied before and after the sustained isometric and concentric fatigability protocols in a subset of participants in a recent study from our group, 19 further insight into the contributors of motor fatigability can be gained.
Therefore, the aim of this exploratory study is to investigate the central and peripheral contributions of lower-extremity motor fatigability when comparing (1) a concentric fatigability protocol and a sustained isometric fatigability protocol as well as (2) pwMS and HCs.
It was hypothesized that (1) motor fatigability in pwMS mainly would be attributed to peripheral fatigability in the sustained isometric fatigability protocol, whereas it mainly would be attributed to central fatigability in the concentric fatigability protocol, and (2) that pwMS would show higher levels of central fatigability compared to HCs.
Methods
This is an exploratory cross-sectional study focusing on the central and peripheral contributions of knee extensor motor fatigability from 2 different protocols (concentric vs sustained isometric) in pwMS and HCs. For further information on the study design see Taul-Madsen et al. 19 A subset of pwMS (n = 31) from the published “original” study (n = 45) underwent neuromuscular examinations (ie, ITT) before and immediately after the 2 fatigability protocols. Moreover, HCs were included in the present study (n = 15) with an intended pwMS to HCs ratio of 2 to 1, matched by age (±5 years) and gender. All participants completed 2 different fatigability protocols, a sustained isometric fatigability protocol and a concentric fatigability protocol. The order of testing was randomized, and participants were allowed a minimum of 2 hours of rest between protocols. The study was approved by the regional ethics committee of Region Midtjylland, Denmark (Journal No. 1-10-72-287-15), and conducted in accordance with the Declaration of Helsinki. Prior to inclusion, all participants provided written consent.
Participants
Recruitment of pwMS was carried out through the MS Clinic of Southern Jutland, Denmark, the MS Clinic at Aarhus University Hospital, Denmark, and through the website of the Danish MS Society. To be included pwMS had to have been diagnosed with definite MS according to the McDonald criteria 20 without any acute exacerbation or relapse 8 weeks prior to testing. Furthermore, only ambulatory pwMS (Expanded Disability Status Scale 21 [EDSS] ≤ 5.5) were included. Additional exclusion criteria were pregnancy or comorbidities that could possibly affect perception of fatigue impact or performance. Recruitment of HCs was carried out via the network of participating pwMS—that is, spouses, family members, and friends.
Motor Fatigability Protocols
Both fatigability protocols were carried out using an isokinetic dynamometer (Humac Norm, CSMi, Stoughton, MA), in which pwMS and HCs were seated to perform knee extension, as described previously. 19 All participants completed a standardized warm-up prior to each of the 2 fatigability protocols. The warm-up prior to the isometric fatigability protocol consisted of 1 repetition (5-s isometric contraction) at 20%, 40%, 60%, 80%, and 100% of maximal muscle strength exertion (maximal voluntary contraction [MVC]), respectively. The warm-up prior to the concentric fatigability protocol consisted of 5 concentric contractions (30°/s) at 20%, 40%, 60%, 80%, and 100% of maximal muscle strength exertion, respectively.
Preceding and immediately following both fatigability protocols, an MVC trial combined with the ITT was carried out (Figure 1, details provided below). The sustained isometric fatigability protocol comprised a 2-minute sustained MVC at 70° knee extension. The concentric fatigability protocol comprised 40 maximal knee extensions at 30°/s with 90° range of motion (starting from 90°, ending at 0°). After each knee extension, the leg was passively brought back to the starting position, also at 30°/s. In this way, the active contraction time of each protocol was 120 s. A schematic presentation of both protocols can be seen in Figure 1. Participants received strict instructions to contract as forcefully as possible during the neuromuscular testing (MVC + ITT) and fatigability tasks (maximal sustained isometric and concentric muscle contractions). Furthermore, strong verbal encouragement to perform at maximal intensity was provided during the neuromuscular testing and fatigability tasks by the investigator along with visual feedback in real time of their force trace on a screen.
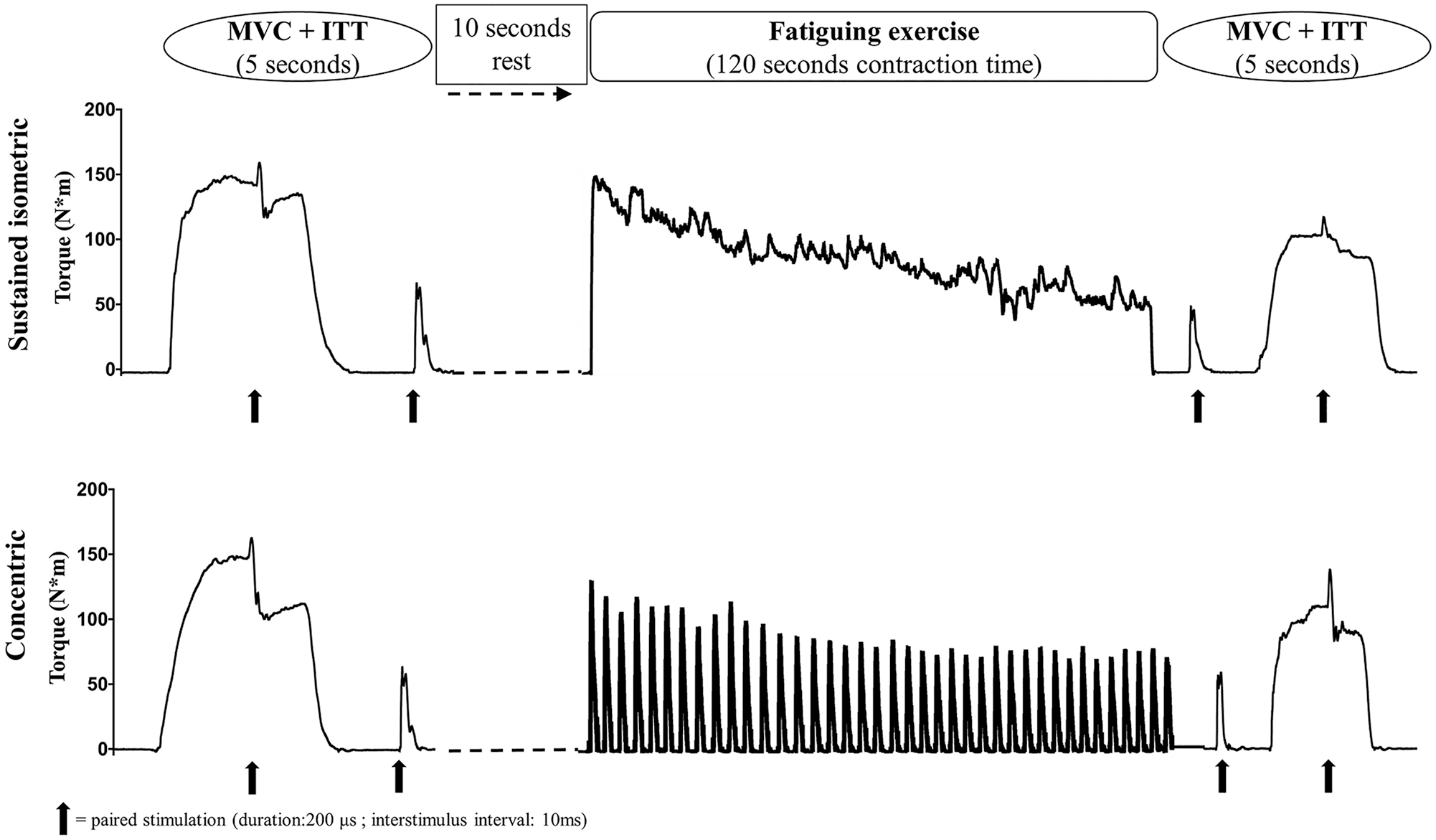
The 2 fatigability protocols are shown with the sustained isometric protocol above and the concentric protocol below. Neuromuscular testing was carried out before and after each fatigability protocol (sustained isometric and concentric), consisting of maximal muscle strength exertion (MVC) with an interpolated doublet and a resting doublet.
Neuromuscular Examinations
Electrical Stimulation
ITT was used to evaluate voluntary muscle activation (VA) as previously described by Hvid et al. 22 Initially, familiarization with electrical stimulation (direct current stimulator, model DS7A, Digitimer Ltd, UK) along with determination of individual maximal stimulation current (ie, until no further increase was observed in torque amplitude following systematic increment in stimulation current) were carried out. The electric stimulation consisted of doublet twitches (200-µs duration, 10-ms interstimulus delay) of quadriceps muscle by the use of two 5 × 10 cm electrodes (Valutrode Lite, Axelgaard, Denmark) positioned 15 cm above the basis of the patella and 15 cm below the anterior superior iliac crest. The ITT stimulation was applied at the force plateau of the MVC and immediately after the MVC at rest. This was carried out before and after the fatigability tasks in both the sustained isometric and concentric fatigability protocol (Figure 1).
Data Sampling
Muscle strength (torque) data were sampled at 1500 Hz using a TeleMyo Direct Transmission System and MyoResearch Software (Noraxon, Scottsdale, AZ) and subsequently low-pass filtered (6 Hz) and analyzed using custom-made software (MathWorks, MatLab 2017, Natick, MA).
Outcome Measures
Three main outcomes were assessed. First, motor fatigability was quantified as the percentage reduction in MVC torque normalized to body weight during the 2-minute sustained isometric or concentric fatigability task. This was termed the MVC fatigability index (FIMVC) and calculated as follows: FIMVC = [(MVCpost − MVCpre)/MVCpre] × 100. Greater (negative) percentage FIMVC values thereby represent a larger degree of motor fatigability. Second, central fatigability was quantified as the percentage reduction in VA assessed by the ITT, calculated as follows: Central fatigability = [(VApost − VApre)/VApre] × 100. VA was calculated as VA = 100 − {[(TITT − Tstim) − (Tstim/MVC)]/Trest} × 100, using torque at stimulation (Tstim), superimposed twitch peak (TITT), and resting twitch peak (Trest). This approach adjusts for situations where the superimposed stimulation is not delivered at the peak plateau of the MVC. 23 Third, peripheral fatigability was quantified as the percentage reduction in resting twitch torque, calculated as follows: Peripheral fatigability = [(Resting twitchpost − Resting twitchpre)/Resting twitchpre] × 100.
Statistics
Statistical analyses were performed using a linear mixed model in STATA (IC 14, StataCorp, College Station, TX). All data followed a normal distribution, based on visual inspection of histograms and quantile-quantile plots. Protocol (sustained isometric, concentric), group (HCs, MS), and time (Pre, Post) were set as fixed effects. To compare the demographic characteristics of pwMS and HCs, a linear mixed model was used.
A multivariate repeated measurements mixed model was used to investigate the effect of time (pre-to-post fatigability tasks; percentage changes) and protocol on MVC, VA, and resting twitch in pwMS and HCs. Furthermore, differences between groups (HCs, pwMS) were investigated with deficit percentage scores calculated for MVC (FIMVC), VA (central fatigability), and resting twitch (peripheral fatigability) and by a multivariate repeated-measurements mixed model.
Moreover, multiple linear regression analysis was carried out to determine if the independent variables VA change and resting twitch change were associated with the dependent variable FIMVC. The strength of the effect of each individual independent variable in the regression analysis was presented as standardized β (calculated by subtracting the mean from the variable and dividing by its SD), with values ranging from −1 to 1. Associations were considered weak if r2 <0.25, moderate if 0.25 ≤ r2 < 0.49, and strong if r2 >0.49.
All Pre and Post data are presented as mean (SD), whereas pre-to-post data (ie, changes following torque reduction) are presented as mean (95% CI).
Missing values were encountered for some of the outcome measures (ie, MVC, VA, resting twitch), either because some of the participating pwMS failed to perform the fatigability protocols correctly (yet only one of the protocols, with the other protocol being performed as instructed) or because of technical problems with the wireless data sampling system. Hence, fatigability data (Pre and Post data) were available for n =28 pwMS in the sustained isometric protocol and for n =27 pwMS in the concentric protocol. Because of technical problems with the sampling system, data were missing for some outcomes. Therefore, there were slightly different sample sizes for each outcome in the sustained isometric and concentric fatigability protocols, respectively, for both pwMS (MVC: n = 28 and n = 27; VA: n = 28 and n = 27; resting twitch: n = 25 and n = 25) and HCs (MVC: n = 15 and n = 15; VA: n = 15 and n = 15; resting twitch: n = 14 and n = 15). The statistical approach (linear mixed model) was, therefore, appropriate because it can handle missing data.
Results
Participants
As can be seen in Table 1, 31 pwMS with a mean EDSS of 2.4 (1.4; range 0-5.5) and 15 HCs were included in the present study. pwMS were comparable to the HCs group in terms of age, height, weight, and body mass index. As expected, the 2 groups differed in total MFIS score, with pwMS scoring 43.3 (12.8) and HCs scoring 4.8 (5.1).
Demographic data.
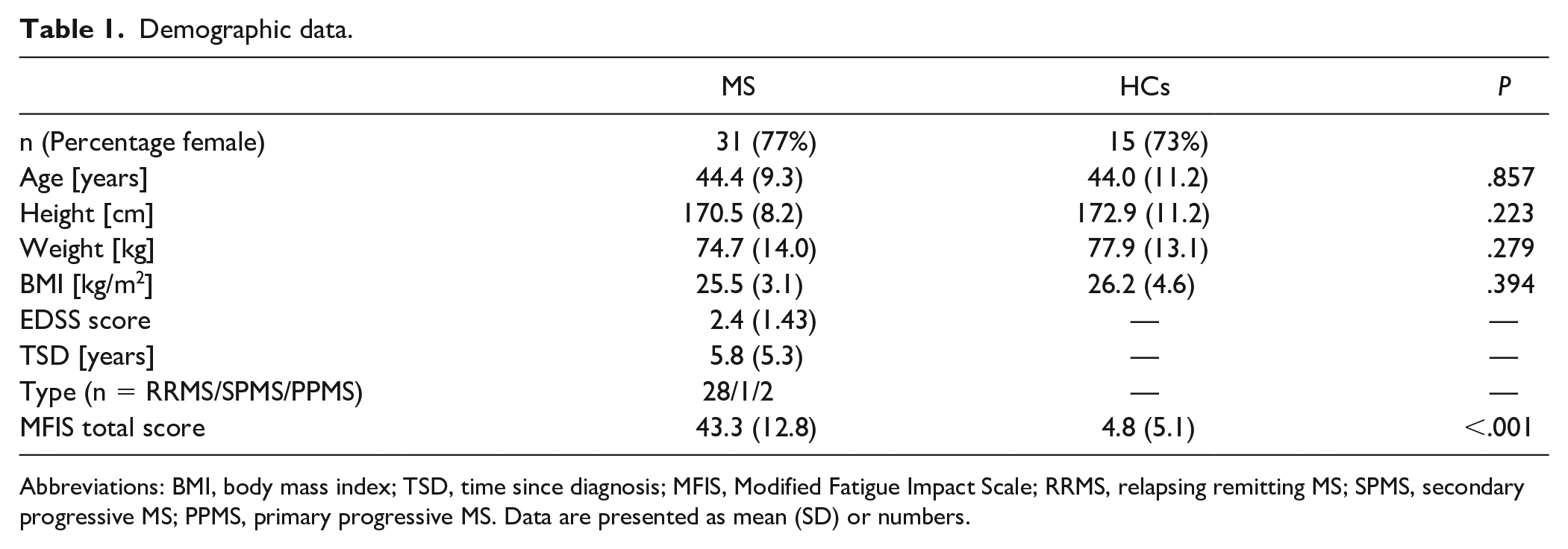
Abbreviations: BMI, body mass index; TSD, time since diagnosis; MFIS, Modified Fatigue Impact Scale; RRMS, relapsing remitting MS; SPMS, secondary progressive MS; PPMS, primary progressive MS. Data are presented as mean (SD) or numbers.
Motor Fatigability
In pwMS, a negative FIMVC (reduction in MVC induced by the fatigability task) was found in both the sustained isometric and concentric fatigability protocols. This motor fatigability was accompanied by a reduction in VA after both fatigability protocols, indicating central fatigability. Furthermore, resting twitch torque was also reduced in both fatigability protocols (Table 2), indicating peripheral fatigability. In HCs, a negative FIMVC was found in both the sustained isometric and concentric fatigability protocols. The VA did not change in either of the 2 fatigability protocols. However, the resting twitch torque was reduced in the sustained isometric fatigability protocol but not in the concentric fatigability protocol.
Fatigability of neuromuscular function.
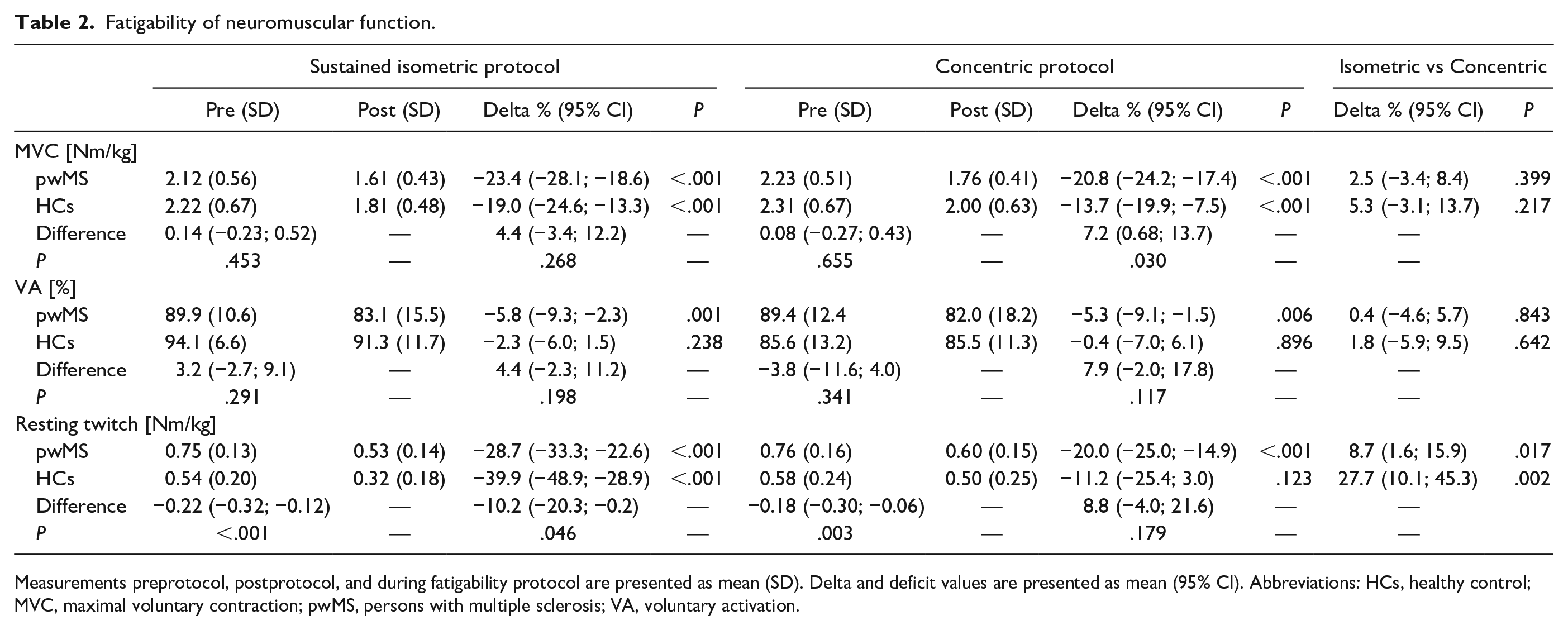
Measurements preprotocol, postprotocol, and during fatigability protocol are presented as mean (SD). Delta and deficit values are presented as mean (95% CI). Abbreviations: HCs, healthy control; MVC, maximal voluntary contraction; pwMS, persons with multiple sclerosis; VA, voluntary activation.
Between pwMS and HCs, a similar reduction in FIMVC was found in the sustained isometric fatigability protocol, whereas pwMS showed a greater reduction in FIMVC after the concentric fatigability protocol, when compared with HCs. The change in VA did not result in difference between the pwMS and HCs. The resting twitch torque change in the sustained isometric protocol was greater in HCs compared with pwMS but did not differ in the concentric fatigability protocol.
Between protocols, the only observed difference was a greater reduction of resting twitch torque in the sustained isometric fatigability protocol than in the concentric fatigability protocol. No difference was found between protocols in either FIMVC or VA (Table 2).
Supplementary to the presented results, the average recovery time from the end of the fatigability tasks to the resting twitch was 5.1 s (3.9 s) and to MVC was 9.0 s (4.0 s) after each fatigability protocol.
Association of Motor Fatigability to Neuromuscular Function
In pwMS, FIMVC in the sustained isometric fatigability protocol was associated with change in VA (β = 0.55; P = .002) but not with change in resting twitch torque (β = 0.21; P = .230), suggesting that variation in sustained isometric fatigability is mainly attributed to central fatigability. The complete model with both independent variables explained 25% of the variation in the dependent FIMVC (P = .010). In HCs, FIMVC in the sustained isometric fatigability tended to be associated with change in resting twitch torque (β = 0.48; P = .099) but not with change in VA (Table 3), suggesting that variation in sustained isometric fatigability is mainly attributed to peripheral fatigability, with the complete model explaining 14% of variance (P = .176) in FIMVC.
Multiple regression analysis of fatigabilities association to neuromuscular function.
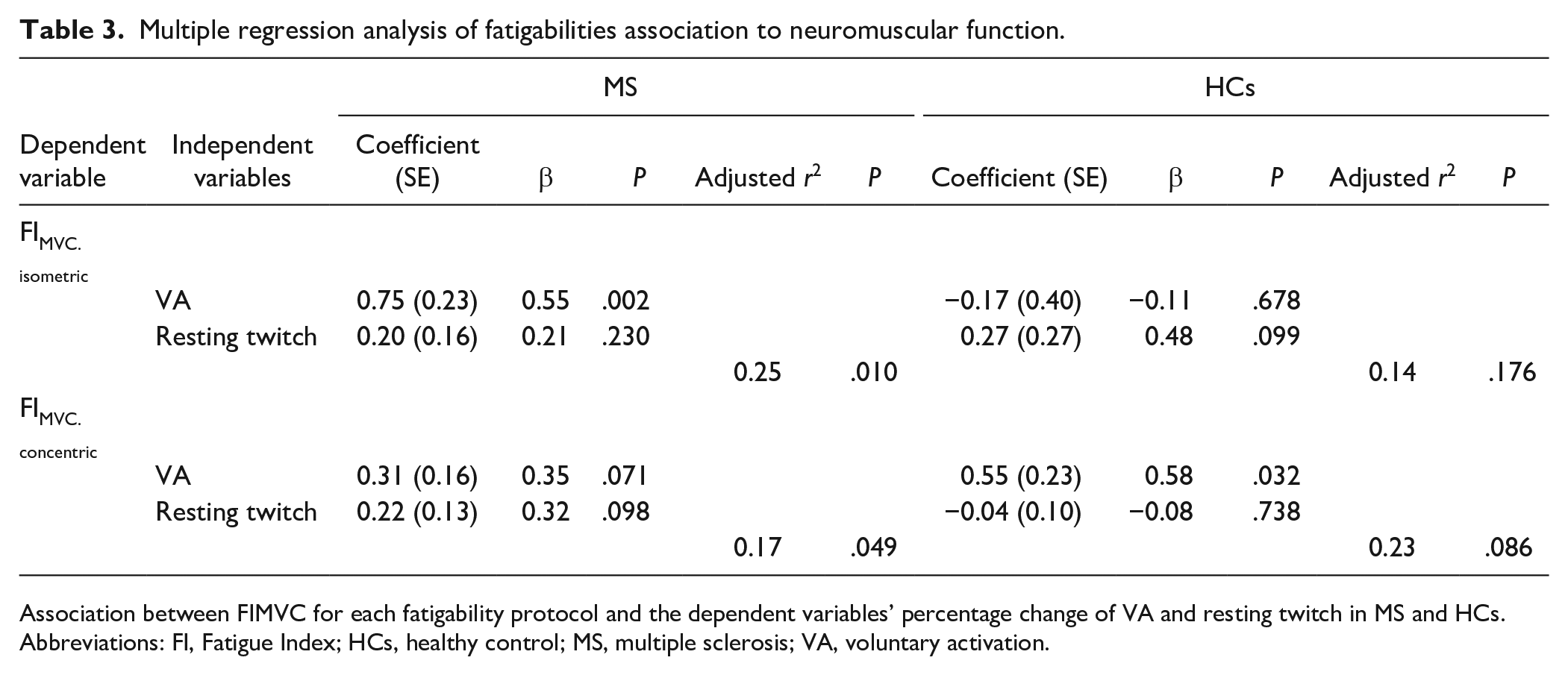
Association between FIMVC for each fatigability protocol and the dependent variables’ percentage change of VA and resting twitch in MS and HCs. Abbreviations: FI, Fatigue Index; HCs, healthy control; MS, multiple sclerosis; VA, voluntary activation.
In pwMS, contrary to the results of the sustained isometric fatigability protocol, FIMVC in the concentric fatigability protocol tended to be both associated with changes in VA (β = 0.35; P = .071) and in resting twitch torque (β = 0.32, P = .098; Table 3), suggesting that concentric fatigability is attributed to both central and peripheral fatigability. The complete model including both independent variables explained 17% of the variation in the dependent variable, FIMVC (P = .049). In HCs, also contrary to the results of the sustained isometric fatigability protocol, FIMVC in the concentric fatigability protocol was associated with change in VA (β = 0.58; P = .032) but not in resting twitch torque (Table 3), suggesting that concentric fatigability is attributed mainly to central fatigability. The complete model including both independent variables tended to explain 23% of the variation in the dependent variable FIMVC (P = .086).
Discussion
The findings of the present study suggest a complex interaction between the specific parts of the neuromuscular system and motor fatigability in sustained isometric and concentric fatigability protocols as well as in pwMS and HCs. The motor fatigability (negative FIMVC) observed from a sustained isometric fatigability protocol and a concentric fatigability protocol were similar in pwMS and HCs. However, in pwMS, the variance of the sustained isometric fatigability was mainly attributed to central fatigability, whereas concentric fatigability was attributed to both central and peripheral fatigability. In HCs, the variance of the sustained isometric fatigability was mainly attributed to peripheral fatigability, whereas concentric fatigability was mainly attributed to central fatigability.
The present study is a continuation of the study by Taul-Madsen et al 19 and adds a neuromuscular dimension to the observed associations of motor fatigability and clinical outcomes of the study. Taul-Madsen et al 19 reported a stronger association between the torque reduction in the concentric versus the sustained isometric protocol and pwMS perceptions of fatigue impact as well as measures of walking capacity.
Motor Fatigability in pwMS and HCs
To the knowledge of the authors, only 1 previous study has used reduction in maximal muscle strength (ie, assessed pre-to-post fatigability task, termed FIMVC) to quantify motor fatigability in pwMS, along with a detailed investigation of the central and peripheral contributors within the neuromuscular system. 18 In pwMS, the FIMVC in the concentric fatigability protocol was larger in the present study compared to that reported by Hameau et al. 18 This discrepancy in part is likely explained by the longer work time in the fatigability protocols used in the present study (ie, 120 s vs 75 s). Although motor fatigability can be calculated in different ways (see review for extensive exemplifications 10 ), the method of percentage change in MVC pre-to-post the fatigability task was used in the present study. However, this method includes a brief period of recovery between the last contraction of the fatigability task and the following contraction allowing a short recovery of MVC (Figure 1). This short resting period could potentially recover some of the motor fatigability, with rapid partial recovery of both peripheral and central parameters occurring within ~10s. 24 Such a rapid recovery seemed particularly evident following the sustained isometric fatigability protocol, with FIMVC being almost half the magnitude of FI. We can only speculate whether this affected the contribution of central and peripheral fatigability to the torque reduction found in both fatigability protocols. Future studies should strive to precisely report the period between fatigability task and neuromuscular testing to give further insights into the effects of neuromuscular recovery.
Central and Peripheral Contributors of Motor Fatigability: Difference Between Protocols
Central fatigability (reduction in VA) was found in both protocols in pwMS but not in HCs. Nevertheless, no between-group differences were observed, despite greater numerical changes in pwMS compared to HCs. In contrast to our observations, Skurvydas et al 17 did report greater central fatigability in pwMS versus HCs (45%-55% vs 20%-35%, respectively). These differences may be a result of the use of the ITT during the fatigability task, in contrast to the present study, which used the ITT before and after. This supports the notion that recovery of the neural system occurs rapidly.
Whereas no previous study has investigated change in knee extensor VA in pwMS by a concentric fatigability protocol, the fatigability task resulted in a reduction of VA in pwMS but not in HCs. However, no group difference between pwMS and HCs were present, which is in line with similar results using electromyographic activity following a concentric fatigability protocol of the lower extremities. 13
Peripheral fatigability (reduction in resting twitch) developed from the sustained isometric fatigability protocol in both pwMS and HCs, but to a greater extent in HCs. Similar findings have been reported in both pwMS and HCs (~25%-40% reduction in resting twitches).17,25 Unexpectedly, the resting twitch prior to both fatigability protocols was greater in pwMS compared to HCs; this contrasts earlier findings. 10 Although speculative, one explanation could be that muscles in MS patients compensate for a reduced neural drive, with a better distribution of the electrical signal between muscle fibers, thereby recruiting a larger number of muscle fibers when a twitch is delivered and ultimately leading to a greater force production in this state. The reduction in maximal muscle strength in both protocols was accompanied by peripheral fatigability in pwMS, whereas HCs only showed peripheral fatigability in the sustained isometric protocol. The sustained isometric protocol further resulted in a larger reduction in resting twitch torque in HCs than in pwMS, thereby confirming the findings by Skurvydas et al. 17
To further facilitate our understanding of the combined and separate involvement of central fatigability (VA change) and peripheral fatigability (resting twitch change) to overall motor fatigability (FI; ie, reduction in maximal muscle strength), we carried out multiple regression analysis, an approach that no study has previously taken in the lower extremities. This verified that in pwMS, sustained isometric fatigability is attributed mainly to central fatigability (as shown in the upper limbs 13 ) but mainly to peripheral fatigability in HCs. Contrary to our hypothesis, the level of motor fatigability in pwMS following the concentric protocol was not only attributed to central fatigability, but also to peripheral fatigability. In HCs, the level of concentric fatigability was mainly attributed to central fatigability.
The findings of the present study show that the contributors of motor fatigability from a concentric fatigability protocol in pwMS are both central and peripheral, whereas it is only central from a sustained isometric fatigability protocol. These findings could perhaps be part of the explanation as to why motor fatigability from the exact concentric fatigability protocol shows a stronger association to perceptions of fatigue and to measures of walking capacity than motor fatigability from the sustained isometric protocol. 19 Furthermore, the results could guide MS rehabilitation strategies to target both central and peripheral adaptations and not aim to emphasize one or another component of the neuromuscular system.
Methodological Considerations and Future Perspectives
A number of methodological aspects deserve mentioning. First, the present study relied solely on the ITT to determine the 2 contributors of motor fatigability: that is, VA and resting twitch. Whereas this technique and its limitations have previously been discussed in-depth,26-28 the ITT is a widely used and well-established technique to assess VA during MVC. To minimize the influence of the methodological limitations, we followed the best practice guidelines. 26 Second, a closer examination of the regression analysis revealed that central fatigability (VA) and peripheral fatigability (resting twitch) explained 14% to 25% of the variation in MVC reduction. Theoretically, motor fatigability should be 100% explained by changes in central and peripheral aspects of the neuromuscular system. A possible explanation is that the ITT only stimulates part of the muscle, whereas MVC represents the activity of the entire muscle along with synergistic muscles involved in the specific joint movement. Furthermore, it has previously been suggested that the motivation of an individual performing a fatigability task influences the level of motor fatigability: that is, the individual stops/fails at what they are willing to do and not at a physiological limit. 29 Furthermore, the recovery between fatigability tasks and neuromuscular testing (5.1 s [3.9] for the resting twitch and 9.0 s [4.0] for the MVC) could possibly alter the observed relation. However, the degree of recovery in such a short timeframe has not been investigated. 24 Third, the study sample size is somewhat small, especially in the HCs group (n = 15), with matching in a 1:2 ratio of pwMS only. This could further influence the interpretation of the results because of the low statistical power. Fourth, the assessment of 1 lower-extremity muscle group only (ie, knee extensors) limits the external/ecological validity as well as the clinical perspective of the study. It is well known that physical function relies on a synergy of multiple joints and muscle groups, all of which are of importance to tasks such as walking. 16 Fifth, the sample of pwMS were on average mildly disabled, limiting the generalizability of the findings.
Finally, future studies should take these methodological aspects into consideration and aim to improve the fatigability protocols, using more advanced measuring techniques or reducing the amount of recovery time between fatigability tasks and neuromuscular measurements, for instance by applying neuromuscular measurements during the fatigability task, thereby removing any recovery time (e.g. with transcranial magnetic stimulation). 24 Furthermore, recruitment of patients with a higher disability status and/or with fatigue levels above the clinical cut-point could also expand the current knowledge and move the field forward. Finally, studies comparing the extent of fatigability in upper and lower extremities are needed.
Conclusion
This study showed that when using a sustained isometric fatigability protocol and a concentric fatigability protocol, the reduction in maximal muscle strength (i.e. MVC) was comparable between protocols in both pwMS of average mild disability and HCs, but the central and peripheral contributions differed between protocols as well as between pwMS and HCs. The regression analysis showed that in pwMS of average mild disability, sustained isometric fatigability was mainly attributed to central fatigability (replicating previous observations), whereas concentric fatigability was attributed to both central and peripheral fatigability (novel observation).
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: UD has received research support, travel grants, and/or teaching honorary from Biogen Idec, Merck Serono, Novartis, Bayer Schering, and Sanofi Aventis as well as honoraria from serving on scientific advisory boards of Biogen Idec and Genzyme. All other authors declare no conflicts of interest in relation to the content of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
