Abstract
Introduction
Multiple sclerosis (MS) is a progressive, chronic disease of the central nervous system, with symptoms such as weakness and fatigue. 1 Fatigue is often defined as a “subjective lack of physical and/or mental energy that is perceived by the individual or caregiver to interfere with usual and desired activities.” 2 Fatigue is thus a subjective symptom that interferes with task performance in daily life. Kluger et al 3 proposed a unified taxonomy, where the symptom of fatigue is affected by perceptions of fatigue and performance fatigability. Performance fatigability can be studied within different domains. When studying the motor domain, the following definition of motor fatigability (adapted from Kluger et al 3 ) can be used: “Motor fatigability is the magnitude or rate of change of motor performance on an objectively measured reference criterion after any type of voluntary activity or exercise” (p. 411). Several reviews on the assessment and treatment of fatigue in persons with MS (PwMS) or other neurological diseases exist.4-6 In contrast, no overview has been reported of protocols and outcomes that can identify pathological motor fatigability in PwMS. Motor fatigability is, however, important to consider in PwMS because it affects the ability to perform sustained activities of daily living. 7 Furthermore, it will have an impact on the exercise capacity of PwMS, limiting the possibility to exercise regularly. The assessment of muscle weakness is routinely performed, but muscle weakness and motor fatigability are different concepts. 8 PwMS might not show muscle weakness on a single assessment of muscle strength, but they might show increased motor fatigability with a fast decline in muscle strength during longer or repeated test protocols.
Importantly, self-reported measures for fatigue do not provide a valid indicator of motor fatigability after physical activities or vice versa 9 because the 2 concepts are most often unrelated.10,11 Consequently, it is important to quantify (pathological) motor fatigability applying objective, valid, reliable, and responsive outcomes. 10 To unravel the relation between motor fatigability and activities of daily living, it is, furthermore, important to include outcome measures on both the body function level and the activity level of the International Classification of Functioning, Disability and Health (ICF), when assessing fatigability. 12 It is not known if PwMS show motor fatigability after repeated and sustained muscle contractions of isolated joints, reflecting the “ICF body function level,” as well as after functional activities such as walking, reflecting the “ICF activity level.” From studies in healthy persons, it is known that motor fatigability is task specific and that protocol specifications affect the findings and the underlying mechanisms of motor fatigability. 13 In healthy individuals, different protocols are applied to assess different aspects of motor fatigability, where submaximal protocols most likely challenge the central nervous system and high-intensity exercises the peripheral neuromuscular system.14,15 It is, however, not known if PwMS express pathological motor fatigability during all tasks or whether the results of different protocols can be compared.
Therefore, the aim of this systematic review was (1) to provide an extensive overview of the applied motor fatigability assessment protocols and outcome measures within the ICF framework to detect the most optimal assessment to quantify motor fatigability in PwMS and (2) to summarize the available information on psychometric properties for the located protocols, including the discriminant ability and the effect of exercise interventions.
Methods
Data Sources and Study Selection
To review the literature, the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement were adopted. 16 The current review was registered on PROSPERO (CRD42016032320). Two databases (PubMed and Web of Science [WOS]) were searched using a combination of MeSH terms/keywords: multiple sclerosis AND (muscle fatigue OR fatigability OR motor fatigue). Literature searching started in September 2015, ended in November 2015, and was updated on August 23, 2016.
Studies were selected when they were written in English, published within the past 20 years, had undergone peer review, and enrolled ≥10 PwMS. Furthermore, studies had to generate/assess an objective parameter quantifying motor fatigability based on voluntary contractions. Interventional studies were only included when the intervention consisted of physical exercises. Descriptive, exploratory, or experimental full-length studies were included; conference papers, meeting reports, letters, and reviews were excluded.
All results were screened based on title and abstract by 2 independent reviewers (DS and CL). The entire text was read when the abstract did not provide sufficient information. In cases of disagreement between the 2 reviewers or if it was unclear whether a study should be included, a third reviewer (IL) was consulted. The reference lists of included articles were checked for further relevant articles.
Quality Assessment
The methodological quality was determined by the Downs and Black checklist. 17 Questions 6, 8, 14, 15, 19, 23, 24, and 26 were removed from the original scale when assessing observational studies because these questions were not applicable. A maximal score of 18 could, therefore, be assigned to observational studies as compared with 26 for randomized controlled trials. The score was converted to a percentage score for each study, with a score >65% indicating sufficient methodological quality. 18 Because the main goal of the review was to evaluate details of fatigability protocols, no studies were excluded based on the quality assessment.
Data Extraction
The following data were extracted from the selected articles: (1) study aim, ICF assessment level; (2) sample characteristics; (3) fatigability protocol specifications and outcome parameters; (4) information on psychometrics of outcomes; and (5) the main findings on motor fatigability in studies comparing healthy controls (HCs) with PwMS. Articles reporting protocols of a single muscle group or a single limb, during nonfunctional activities, were classified at the ICF body function level (category b 730-muscle power functions and b740-muscle endurance functions). Articles examining motor fatigability during functional activities were classified at the ICF activity level (d450-469-walking and moving). Details of the intervention and the effects on motor fatigability were documented from interventional studies.
Results
As presented in detail in Figure 1, 48 articles were included from the initial 494 hits (353 from WOS and 141 from PubMed). The data for quality assessment of the included studies are shown in Supplementary Tables 1 and 2. The results of the quality assessment for observational studies show that the scores range from 35% to 82% and for the interventional studies between 41% and 74%. The main problems of the observational studies were the lack of reporting of the following: the difference between asked and recruited participants (Q11 and Q12), the recruitment period (Q22), possible confounders (Q25), and the power of the study (Q27). For the interventional studies, all studies scored 0 on the representation of the study population (Q12), blinding of participants and assessors (Q14-15), and concealment of allocation (Q24). Furthermore, the reporting was also limited on the confounders (Q25), losses to follow-up (Q26), and the power of the study (Q27).
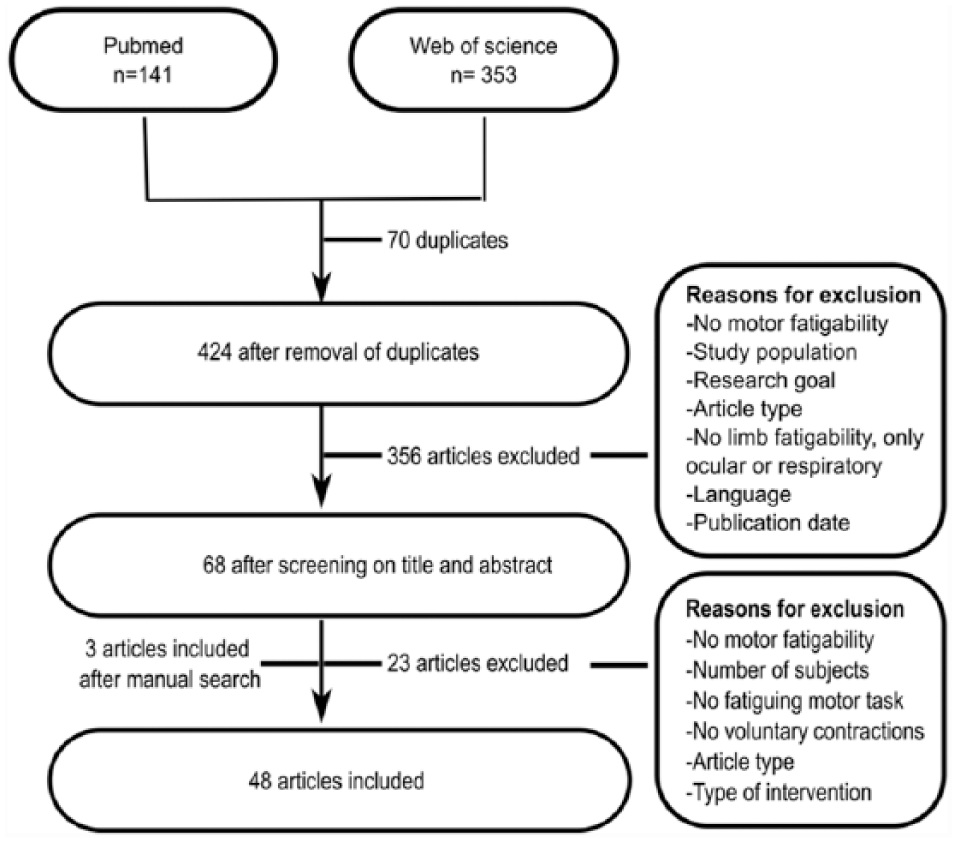
Schematic representation of the literature search.
Study Objectives and Patient Characteristics
Study objectives were heterogeneous (Supplementary Table 3). In 23 articles, the objective was to assess motor fatigability in PwMS, whereas 11 articles aimed to detect underlying mechanisms. Also, 8 articles aimed to study the correlation between self-reported fatigue and motor fatigability, whereas 7 reported the influence of motor fatigability on other outcomes such as dual tasking, balance, motor cortex excitability, and brain activation patterns; 5 articles reported the effect of an intervention on motor fatigability; and 3 reported on the reliability of the applied outcomes for fatigability.
Most articles (n = 36) investigated the ICF body function level. Two articles reported both at ICF body function level and ICF activity level. Of the protocols at body function level, 20 examined the upper, 16 the lower, and 2 articles both upper and lower limbs. Motor fatigability protocols were reported by 12 articles at the ICF activity level. One of these combined outcomes at body function level with an exercise on activity level.
Table 1 summarizes sample characteristics. The sample size ranged from 10 to 208; 13 articles included all types of MS, 11 articles only included relapsing-remitting MS (RRMS), and 3 articles only secondary progressive MS. Three articles by the same author reported on PwMS with clinically isolated syndrome.19-21 The average Expanded Disability Status Scale (EDSS) score ranged from 1.3 to 6. The studies that reported on self-reported subjective fatigue enrolled both fatigued and nonfatigued PwMS.
Participant Characteristics of the PwMS in the Included Studies.
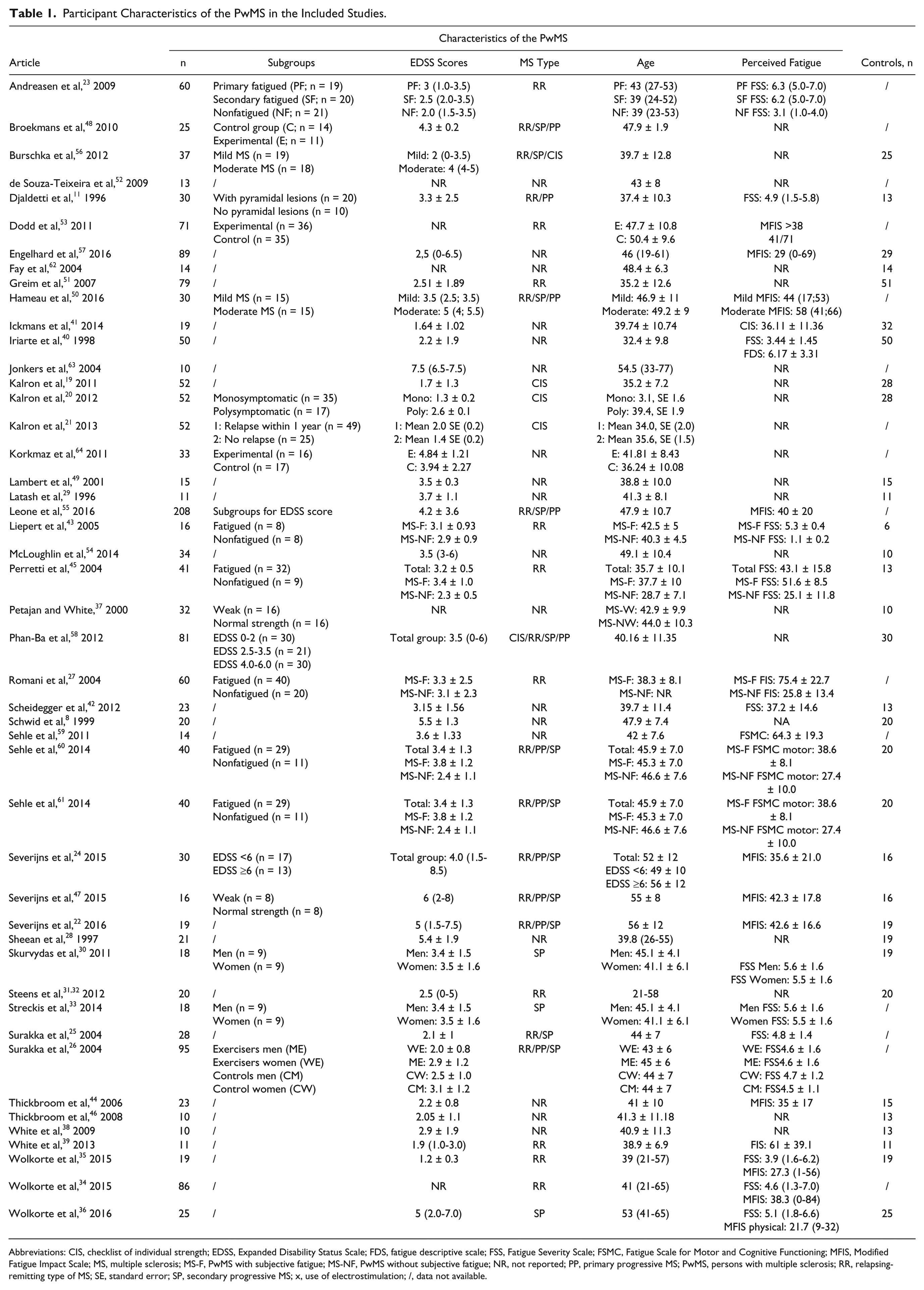
Abbreviations: CIS, checklist of individual strength; EDSS, Expanded Disability Status Scale; FDS, fatigue descriptive scale; FSS, Fatigue Severity Scale; FSMC, Fatigue Scale for Motor and Cognitive Functioning; MFIS, Modified Fatigue Impact Scale; MS, multiple sclerosis; MS-F, PwMS with subjective fatigue; MS-NF, PwMS without subjective fatigue; NR, not reported; PP, primary progressive MS; PwMS, persons with multiple sclerosis; RR, relapsing-remitting type of MS; SE, standard error; SP, secondary progressive MS; x, use of electrostimulation; /, data not available.
Fatigability Protocols
A schematic overview of type of protocols and frequency of application in clinical research in MS is provided in Figure 2. Table 2 documents the protocols’ specifications.
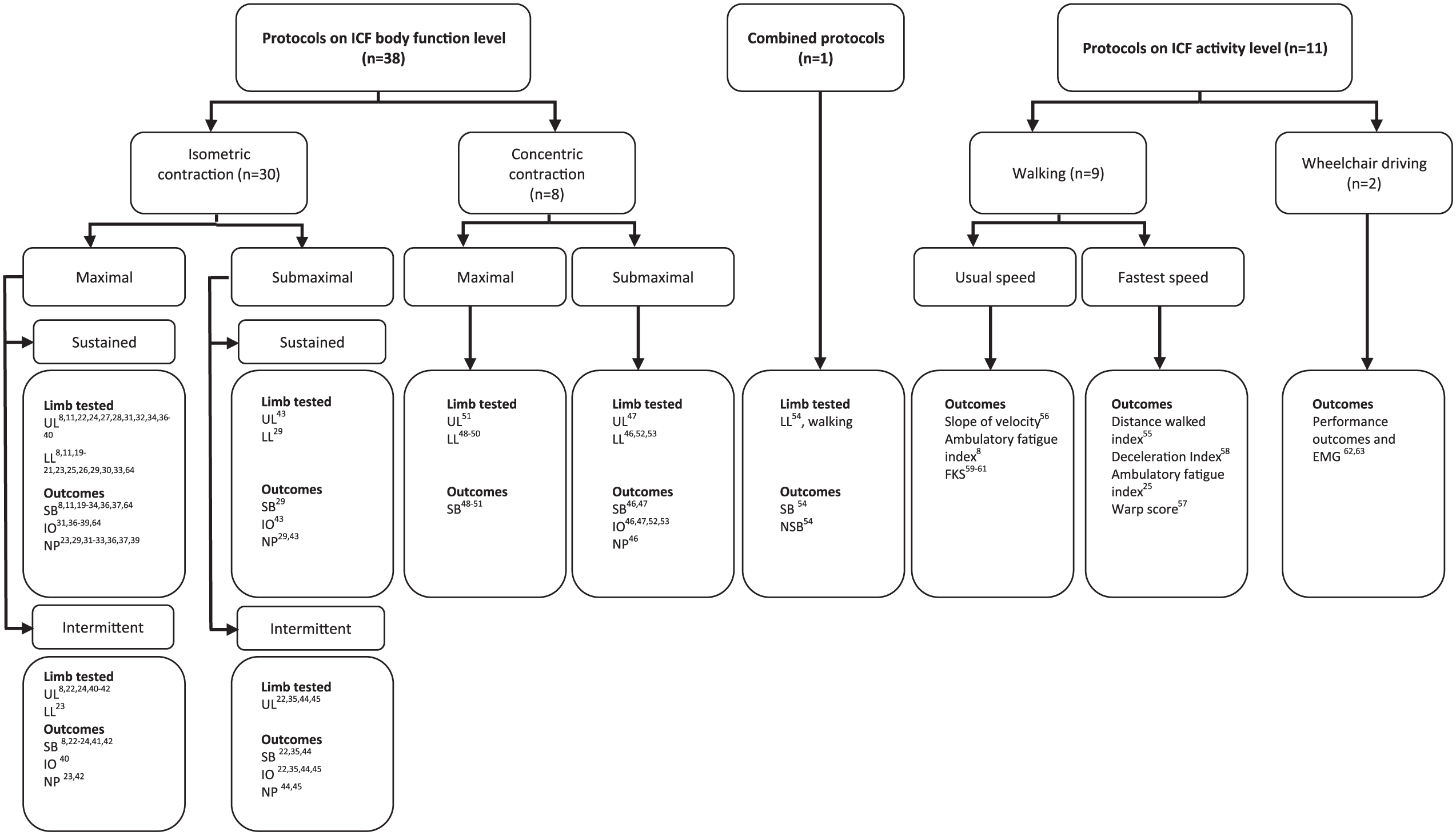
A schematic overview of the type of experimental protocols used to assess motor fatigability in multiple sclerosis.
Specifications of the Protocols and Outcome Measures for Motor Fatigability.
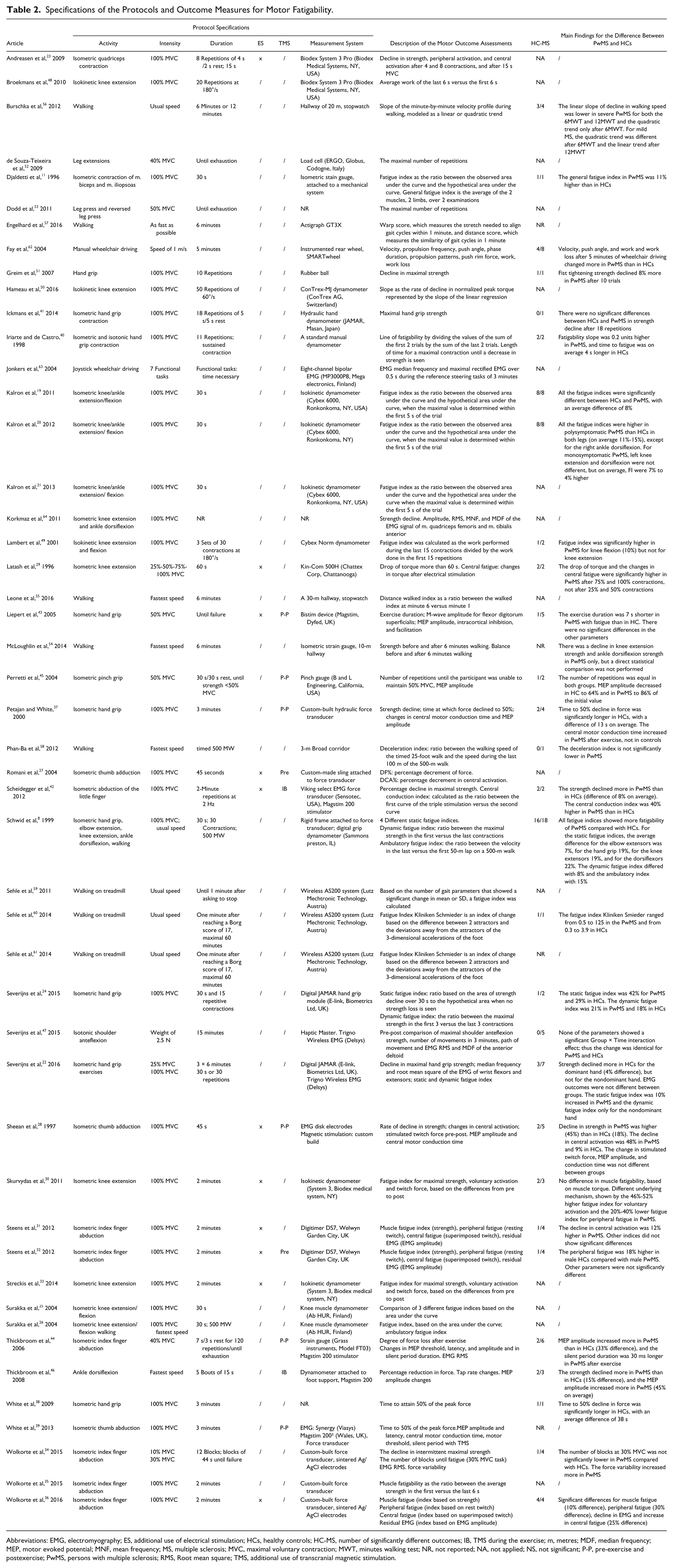
Abbreviations: EMG, electromyography; ES, additional use of electrical stimulation; HCs, healthy controls; HC-MS, number of significantly different outcomes; IB, TMS during the exercise; m, metres; MDF, median frequency; MEP, motor evoked potential; MNF, mean frequency; MS, multiple sclerosis; MVC, maximal voluntary contraction; MWT, minutes walking test; NR, not reported; NA, not applied; NS, not significant; P-P, pre-exercise and postexercise; PwMS, persons with multiple sclerosis; RMS, Root mean square; TMS, additional use of transcranial magnetic stimulation.
Protocols Assessing Motor Fatigability at the ICF Body Function Level
In 30/38 protocols, isometric protocols were used (ICF category b7300-7301, 7400-7401). In all, 10 studies examined the lower limb and 18 the upper limb; 2 studies assessed both the upper and lower limbs. Most studies applied maximal voluntary contractions (MVCs) to assess motor fatigability. One study reported both maximal and submaximal protocols. 22 Generally, the duration of an MVC was predetermined (15 s, 23 30 s,8,11,19-21,24-26 45 s,27,28 60 s, 29 120 s,30-36 180 s37-39). In 1 study, the end point was not clearly defined (“until strength declined”). 40 For protocols applying intermittent MVCs, the number of contractions ranged from 8 repetitions of 4 s 23 to 11 or 15 repetitive contractions without a specified duration,24,40 the number of contractions that can be performed in 30 s, 8 18 repetitions of 5 s, 41 and up to 2 minutes at 2 Hz (240 repetitions). 42
Six articles reported submaximal isometric tasks of 10% to 50% of the MVC. One study evaluated a 60-s sustained contraction at 25%, 50%, 75%, and 100% MVC, 29 whereas 1 study applied 50% MVC until failure. 43 The 3 other protocols used repetitive contractions until inability to produce target force (eg, 30 s vs 30 s rest, or blocks of 44 s with 5 s rest)35,44,45 or with a specified duration (6 minutes of exercises). 22
Eight articles reported nonisometric protocols (ICF categories b7401 or 7402). Two studies required as many contractions as possible in a certain time frame.46,47 Three other studies used an isokinetic protocol in the lower limbs, with 20 or 30 repetitions at 180°/s48,49 or 50 repetitions at 60°/s. 50 One study reported squeezing a rubber ball maximally 10 times. 51 Motor fatigability during multijoint movements of the lower limb was assessed with leg presses until exhaustion.52,53
A total of 10 studies applied additional electrical stimulation of the nerve innervating the target muscle to determine underlying mechanisms.23,27 -33,36,42 Two studies interspersed the voluntary exercises with transcranial magnetic stimulation (TMS) to gain further insight into corticospinal excitability,42,46 and 6 studies used TMS before and after a fatigability protocol.28,37,39,43-45
Protocols Assessing ICF Activity Level
In 10 studies, walking for a fixed time (6 or 12 minutes),54-57 a fixed distance (500 m),8,25,58 or until voluntary exhaustion59-61 at either comfortable or fastest walking speed was applied. Three walking studies used a treadmill59-61; 7 studies tested free overground walking.8,25,54-58 One study examined motor fatigability during 5 minutes wheelchair driving at a speed of 1 m/s in a manual wheelchair, 62 whereas another used a functional steering task with a joystick-driven wheelchair, mimicking daily life activities. 63
Outcome Measures: The Quantification of Motor Fatigability and Underlying Mechanisms
Table 2 summarizes the identified outcomes to document motor fatigability. Overall, the outcomes could be categorized as (1) strength-based outcomes, (2) indirect outcomes, and (3) neurophysiological outcomes.
Strength-Based Outcomes
The majority of the isometric protocols (29/48) used strength-based outcomes to assess motor fatigability. Despite the different names (eg, “percent decrement of force,” “static fatigue index,” “rate of decline in strength”), most outcomes directly measure the strength decline. Indices of motor fatigability are mostly based on the ratio between the initial and the final strength during sustained (23/48) or repetitive contractions (8/48). For the sustained contractions, an index was also based on the area under the curve or the slope of the strength decline.8,11,19-21,24-26 Some articles only reported a statistical comparison of the maximal strength, assessed before and after a specific task, rather than an index.22,29,47,64 This approach was also used in one study after walking. 54 During isokinetic protocols, the ratio between the work done during the first contractions versus the last contractions48,49 or the slope of torque decline 50 was used.
Indirect Outcomes
Four articles reported indirect outcomes based on endurance time. For example, 3 studies reported the time until MVC declined to 50% of the initial MVC.37-39 Five studies evaluated the “number of repetitions”: for example, the number of contractions until the participant was unable to maintain 50% MVC, 45 the number of repetitions performed in a fixed amount of time, 47 or until inability to maintain a target force35,52,53 were reported. The performance such as the change in tapping speed 46 and the trajectory of movements 47 during single-joint movements was described as an indirect measure of motor fatigability.
After walking, indices were based on walking velocity or distance, such as the deceleration index, 58 distance walked index, 55 and ambulatory fatigue index. 8 Four studies calculated an index based on gait kinematics.57,59-61 The performance outcomes after manual wheelchair driving were velocity and propulsion frequency. 62 Electromyography (EMG) was used in 9 studies.22,31,32,35,36,44,47,63,64 Four studies reported the median or mean frequency of the EMG signal,22,47,63,64 whereas the remaining 6 studies reported a measure of the EMG amplitude (eg, root mean square).
Neurophysiological Outcomes
Besides the assessment of motor fatigability as such, neurophysiological outcomes were reported in 17 studies to explore underlying mechanisms of motor fatigability (Table 2). The twitch interpolation technique 65 was frequently applied to determine the changes in central and peripheral activation of the muscles under investigation23,27-33,36,42,43 and used as an indication of a loss of central drive (central fatigue) or peripheral muscle fatigue. 66
Psychometric Properties
The only information on psychometrics in PwMS that was detected with the current literature search was related to reliability of the outcome measures. No information was detected on the validity and responsiveness of motor fatigability measures. The discriminant ability, however, might partially represent construct validity.
Reliability
Three studies investigated the reliability of motor fatigability outcomes. Schwid et al 8 and Surakka et al 26 reported on the reliability of static fatigue indices, based on a 30-s sustained isometric contraction. The static fatigue index with the best reliability in PwMS (intraclass correlation coefficients [ICCs] range = 0.71-0.96) that best discriminated between HCs and PwMS was calculated by dividing the total area under the strength curve by the hypothetical area under the curve, when no strength decline would occur (ie, the multiplication of maximal strength times contraction duration from 5 s after the start of the contraction). Other fatigue indices showed lower ICCs. 8 A fatigue index based on a ratio between the initial strength and the final strength had ICCs of 0.46 to 0.77. A fatigue index, based on the area under the first 5 s versus the last 5 s had ICCs of 0.5 to 0.73. A fatigue index, based on the area under the curve of the entire contraction period, showed ICCs of 0.64 to 0.93. A slightly modified static fatigue index also showed good reliability (ICC of 0.68-0.86). 26 Lambert et al 49 reported poor to moderate reliability of an index based on the work performed during the first 15 versus the last 15 contractions of an isokinetic protocol consisting of 30 maximal contractions at 180°/s (ICCs from 0.36 to 0.51). In accordance, poor reliability of a dynamic fatigue index was reported, with ICCs from 0.20 to 0.44. 8 One report showed poor reliability of an ambulatory fatigue index determined during a 500-m walk (ICC = 0.21-0.36). 8
Discriminant Ability Between (Subgroups of) PwMS and HCs
Table 2 shows that 34/48 articles enrolled PwMS and HCs. Not all studies compared groups statistically; 24 articles reported a statistically significant difference between HCs and PwMS, and 17 of these used isometric (repetitive or sustained) MVCs. Of these 17, strength-based outcomes showed a group difference for motor fatigability in the upper limb,22,24,28,36,40,42,51 the lower limb,19,20,29 and in both the upper and lower limbs.8,11 The number of times that an outcome showed a difference between PwMS and HCs is shown in Table 3. Three protocols with sustained MVCs investigated the difference between subgroups of PwMS and found that motor fatigability was related to disability level (EDSS score), 24 the presence of pyramidal signs, 11 and the involvement of more than 1 functional system. 20 Wolkorte et al 36 further found that secondary progressive PwMS show more motor fatigability compared with RRMS. Seven articles reported nonsignificantly different strength-based outcomes, and 6 of these studied the upper limb (hand grip or intrinsic hand muscles), with a sustained contraction of 2 minutes31,32 or 3 minutes37,38 or with intermittent isometric hand grip contractions.24,41 One article studied knee extension with a sustained contraction of 2 minutes. 30 Out of 6 studies with nonsignificant results, 4 included PwMS with RRMS or mild MS (average EDSS < 3.5). Of the latter studies, 5 using a sustained MVC did, however, report significantly different neurophysiological outcomes. Intermittent concentric maximal contractions elicited a greater decline in strength in the upper limb compared with HCs. 51 One study, using maximal concentric knee contractions, showed only a difference in total work, not for the work index. 49 One study reported different motor fatigability between mild and moderate MS, assessed with the decline in peak torque after 50 isokinetic knee extensions. 50
For the submaximal protocols contractions, only 1 strength outcome (force variability) differentiated HCs from PwMS for the upper limb, 35 whereas no difference in decline in strength, the number of contractions, or exercise duration was observed. One submaximal foot tapping protocol did detect a difference in strength decline between PwMS and HCs. 46 Significant differences were reported for motor fatigability during walking8,54,56,61 and after 5 minutes of wheelchair driving. 62 Three walking studies showed that PwMS with higher EDSS levels experienced more motor fatigability.55,56,58
Summary of the Number of Studies With a Statistical Comparison Between HCs and PwMS in Which Outcomes Indicated an Increased Motor Fatigability in PwMS.
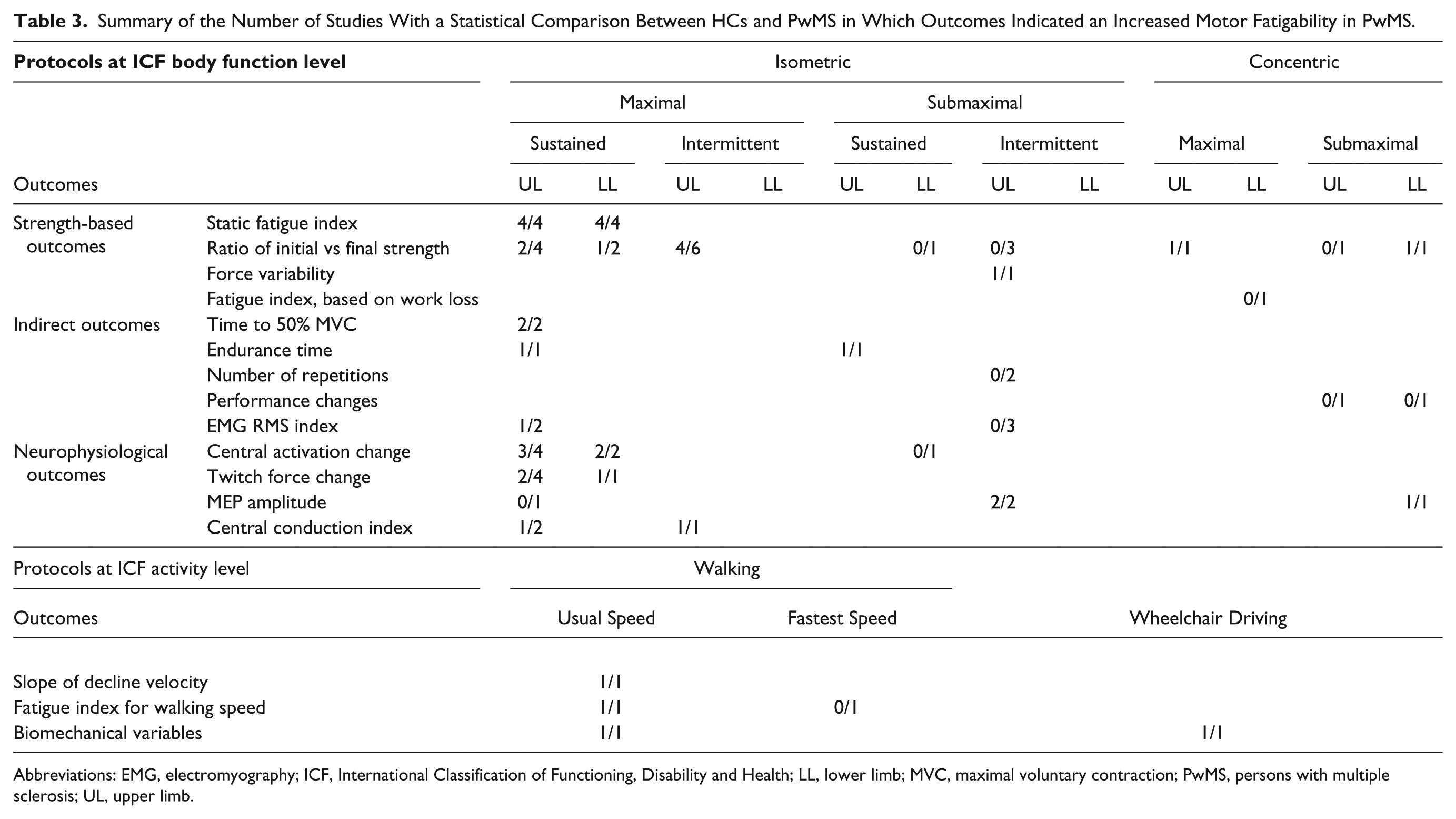
Abbreviations: EMG, electromyography; ICF, International Classification of Functioning, Disability and Health; LL, lower limb; MVC, maximal voluntary contraction; PwMS, persons with multiple sclerosis; UL, upper limb.
The Effect of Exercise Interventions on Motor Fatigability
Although there is no study investigating responsiveness of outcomes for motor fatigability, a limited number of exercise intervention studies revealed that motor fatigability can be improved. Five studies evaluated the effects of different exercise interventions on motor fatigability (Supplementary Table 4). Studies aimed to improve motor fatigability of the lower limb.25,48,52,53,64 Interventions included progressive resistance training,52,53 inpatient rehabilitation followed by home exercises, 25 electrical stimulation, 64 and whole body vibration. 48 Three intervention studies showed improvements of motor fatigability based on the number of repetitions, 52 the knee flexion fatigue index, 25 and the quadriceps peak torque after a 30-s sustained contraction. 64
Discussion
The main findings of this systematic review are that (1) most assessments of motor fatigability at the ICF body function level are based on isometric maximal contractions; (2) the assessments of motor fatigability at the ICF activity level are predominantly based on changes in performance during a functional activity; (3) the majority of the studies at both the body function level and activity level were able to discriminate between PwMS and HCs; and (4) information on the psychometrics of the applied fatigability protocols is limited.
Selecting the Right Protocol and Outcomes When Assessing Motor Fatigability
The included study protocols differed substantially on protocol components, such as the type, intensity, and duration of the fatiguing task; type of movement; and the assessed limb or body part. Because motor fatigability is task specific, 67 these components will influence the assessment of motor fatigability.
Body Function Level
Most protocols assessed isometric MVCs, which is probably explained by the physiological definition of fatigability that most of the studies adopted.14,68 Furthermore, isometric contractions have the advantage that they can be highly standardized. In addition, participants quickly fatigue when they perform MVCs, making strength deficits easier to detect. 69 The duration of most protocols is generally fixed, which is preferable, because protocols performed until volitional exhaustion show poor reliability. 70 The maximal contraction duration was 3 minutes,37 -39 but most applied 30 s. Most studies assessed motor fatigability with strength-based measures. These are easy to interpret, but results of different contraction times are difficult to compare because other mechanisms are likely responsible for the quick decline in strength within 30 s compared with over a longer period.34,71 Nonetheless, the reliability of fatigue indices does not seem to depend on protocol length in HCs. 72 Numerous calculation methods exist for fatigability indices. Using a ratio of the initial versus the final force level might cause problems because the force variability increases during the final stage of a sustained contraction. 73 This problem may, however, be overcome by looking at the total area under the curve after sustained contractions. 26 Studies evaluating the underlying mechanisms of motor fatigability require standardized (high-intensity) isometric contractions, often supplemented by TMS39,42-46 or peripheral nerve stimulation23,27,28,30-33,36,42 before and after the fatiguing task. An in-depth discussion on the underlying mechanisms of motor fatigability is provided in a recently published review. 74 Motor fatigability is most often assessed during sustained contractions; however, intermittent contractions seem more relevant for daily life. Although isokinetic protocols are sparsely used,48-50 these might be interesting because they are even more energetically demanding. 14 One should take into account that also here, results on motor fatigability of sustained contractions cannot be interchanged with intermittent contractions because the physiological reaction in the muscle tissue is different 14 and possibly, also, the brain activation patterns. 75 Some results might indicate that intermittent contractions are less suitable to determine pathological motor fatigability because these do not differentiate PwMS from HCs. 24
The majority of the protocols used maximal muscle contractions. Only 6 studies applied a submaximal isometric protocol.22,29,35,43 -45 One could argue that intermittent submaximal contractions more closely simulate real-world situations.76,77 Submaximal protocols might be difficult to standardize because these might be influenced more by problems with selective motor control requiring more concentration. To evaluate changes in muscle output caused by submaximal exercises, a submaximal fatiguing exercise interrupted with brief maximal contractions might be applied 78 as has been done in a few studies.35,46,47 Furthermore, PwMS with muscle weakness may already perform near their maximal strength during activities of daily living (ADL); thus, when asking a submaximal percentage of their strength, this might be underestimating the problem of fatigability in daily life. A further challenge to this approach relates to the higher absolute strength usually exerted by HCs. This causes reduced blood perfusion in HCs and, thus, increases the peripheral fatigue, although the proportional strength is identical, which contributes to sex-related differences in fatigability. 79 Indirect outcomes such as performance measures 47 have the advantage that they are directly related to the task. However, the interpretation is more difficult because they cannot be attributed to 1 muscle group.
Most protocols on body function level assess the upper limbs, which contrasts the fact that, clinically, motor fatigability is mostly reported in the lower limbs—for example, during walking.
Clinically, it could be hypothesized that fatigability in the lower limbs influences activities of daily living more profoundly and, thus, is reported more frequently because fatigued legs require rest by sitting down or lying. This fatigue effect might only be reported in the upper limbs after very specific tasks, such as sewing, which require continuous activation. One could further argue that investigating motor fatigability in the upper limbs is less relevant because the upper limbs are normally less impaired by the disease process. 8 The reason that most of the research is performed on the upper limbs, is probably caused by the fact that many studies included neurophysiological investigations for the mechanisms of fatigability. These investigations are technically more straightforward to perform accurately in the upper limbs than in the lower limbs because relevant muscles can more easily be accessed and stimulated. Furthermore, when investigating the lower limbs, a more profound effect of deconditioning (besides the disease effect of MS) can be expected. Until now, only 1 study compared motor fatigability in the upper limb and the lower limb, 8 stating that these are related in PwMS. This information is contrasted by a study in HCs, stating that there is no relation between hand grip fatigability and fatigability of the knee extensors. 80 In short, the generalizability of motor fatigability across upper and lower limb muscles is unknown. More can probably be learned in future studies comparing upper- and lower-limb fatigability.
Activity Level
Fatigability determined on the body function level might be more precise and has several advantages, such as the ability to determine underlying mechanisms within the same tasks (strength decline combined with neurophysiological measurements). However, the investigation at the ICF activity level might be more relevant for daily life because these activities are most often closer linked to quality of life. Most studies attempting to evaluate motor fatigability at ICF activity level have applied walking paradigms.8,25,56,58-61 These protocols seem relevant for clinical practice, but the interpretation is complicated because outcomes based on physical performance during functional activities, such as the deceleration in walking speed, give no information on underlying causes. Observing changes in gait dynamics seems an attractive approach and has been applied during walking on a treadmill at maximal speed.60,61 However, it is not straightforward to interpret changes in kinematics as an increase in motor fatigability (decreased neural drive) versus an increased manifestation of underlying impairments of spasticity or ataxia. To overcome this, some studies use EMG parameters as an additional indirect measure of motor fatigability. During sustained submaximal static contractions, notable changes occur as the signal amplitude (RMS) increases and shifts in the EMG spectrum toward lower frequencies take place. Both parameters are used as markers of muscle fatigue. However, EMG variables could not discriminate HCs from PwMS in several studies.31,47 Furthermore, it has been argued that amplitude characteristics are not reliable14,81,82 and that the use of EMG outcomes during dynamic contractions is often difficult to interpret. McLoughlin et al 54 tried to integrate the ICF body function level and the ICF activity level by assessing strength and balance before and after a 6-minute walking paradigm and showed that PwMS showed greater loss of strength and balance after 6 minutes walking than HCs.
Is Motor Fatigability Increased in MS?
Overall, the literature shows that motor fatigability is increased in PwMS. As discussed above, the ability to detect differences between HCs and PwMS seems to be related to the type of tasks (sustained or intermittent), contraction intensity (maximal or submaximal), and the limbs examined (upper or lower limb). Furthermore, it is likely that not all PwMS show abnormal motor fatigability, as reported earlier.24,55 Strength-based outcomes were not always sufficiently sensitive to detect differences in mildly impaired PwMS, despite different neurophysiological outcomes 32 ; 7 studies found no significant difference between HCs and PwMS for strength-based parameters. Maximal protocols, which could not discriminate HCs and PwMS, used a low number of repetitive hand grip contractions24,41 or a long isometric contraction for 2 or 3 minutes,30,31 where individuals are in the plateau phase of strength decline. Further explanations for the lack of discrimination might be the small samples, MS phenotype, 36 or disability level of the participants. 24
Of note, none of the submaximal protocols, assessing the upper limb with strength outcomes, discriminated HCs from PwMS, whereas 1 lower limb study did show a more profound force decline after a foot tapping exercise. 46 This may again suggest a different response between upper and lower limbs, caused by earlier and more substantial impairments in the lower limbs of PwMS. 8
Limitations in the Current Literature Review
The aim of this review was to detect the best protocol to assess and study motor fatigability. Therefore, we narrowed the literature search to included studies that specifically mentioned motor fatigue, muscle fatigue, or fatigability. This was done to avoid nonrelevant articles but might have excluded studies where motor fatigability was a secondary outcome. The quality checklist of the included studies showed that a substantial part of the included research suffered from methodological issues. Previously, Meyer et al 18 used a cutoff score of 65% for inclusion. However, if we used this value as a cutoff, only 22 studies would have been retained, leading to exclusion of several relevant protocols and outcomes.
Clinical Assessment of Motor Fatigability
In 1977, it was already stated that it was necessary to investigate muscle endurance as an additional index of paresis. 83 Later, it was stated that some PwMS do not show strength deficits acutely but experience abnormal motor fatigability over time. 37 To be able to use a protocol in a clinical setting, the protocol should be feasible, quick, easy to interpret, and reliable. Based on the current review, no single protocol can be recommended for clinical use because of the lack of comparison of protocols head to head and the lack of knowledge on psychometrics. To detect the PwMS prone to a slight increase in motor fatigability, a strenuous approach is needed by determining the change in strength during sustained MVCs. Currently, the only outcome that has documented sufficient reliability in PwMS is a fatigue index based on the area under the curve of a sustained MVC of 30 s. 8 Previously, it was stated that documenting the difference in strength before and after an exercise protocol is clinically feasible. 68 This is a feasible approach for physical exercises or daily life activities. With this approach, one should take into account that submaximal exercises might not elicit increased motor fatigability when they are executed at the participant’s own relative strength. Furthermore, PwMS are likely to use a higher percentage of their maximal strength in daily life compared with HCs. 84 Unfortunately, the current clinical tools to assess muscle strength do not include standard protocols and outcomes measures for motor fatigability. Furthermore, the lack of cutoff values for this kind of protocols hinders clinical interpretation.
Recommendations for Future Research
This literature review revealed a number of limitations that need to be kept in mind when interpreting the existing studies and designing future studies. To begin with, one should clearly define the concept that is assessed.3,7 Second, the existing psychometrics information only included limited information on the reliability of a few protocols.8,26 Related to this, no cutoff value exists to determine if motor fatigability is pathological. Future studies should identify norm values for motor fatigability obtained from a large, well-described population-based group of HCs. Third, before collecting norm data, a consensus has to be reached on the best protocol. This is not possible at the moment because of the almost complete lack of studies comparing different protocols head to head. Fourth, future intervention studies should evaluate patients with established motor fatigability at study entry, based on well-defined cutoff values, and perform comparisons of different interventions. Some research suggests that submaximal exercises are better to target the central nervous system. 85 Because pathological motor fatigability in PwMS seems more related to central mechanisms, submaximal exercises with a high number of repetitions or a long duration might attenuate motor fatigability. There is already (limited) information on the influence on white matter structure of, for example, task-specific training in PwMS. 86 However, for the improvement of strength, high-intensity exercises are advised. It was suggested that motor fatigability can be improved by increasing the muscle mass with strength training,87,88 which might also increase neural drive in PwMS 89 and, thus, improve motor fatigability indirectly. Fifth, there is limited information on the relation between motor fatigability in different muscle groups 8 and on the relation between motor fatigability on the different levels of the ICF. Although there seems to be a relation between motor fatigability on body function level and gait parameters, 19 there are no reports explicitly examining the relation between motor fatigability on body function level and activity level. It might be expected that motor fatigability affects functional tasks when sustained or high-intensity muscle activity is required. The clinical meaning of protocols and outcomes for motor fatigability (ie, the relation to quality of life or the participation in society) was not studied so far. It is, however, important that the influence of motor fatigability on daily functioning is elucidated because daily life functioning has a large impact on the quality of life of the patient. 90 Finally, the difference between PwMS and HCs in underlying mechanisms of motor fatigability have predominantly been studied in the upper limbs, with only 2 reports evaluating the lower limbs.
Conclusion
A variety of protocols and outcome measures are applied to study different aspects of motor fatigability in PwMS, thus challenging comparability across studies. Most protocols use maximal single-joint isometric contractions, with the advantage of high standardization. Protocols determining motor fatigability during submaximal or functional activities might, however, be more relevant for PwMS. Because of the lack of head-to-head comparisons of the different protocols and a lack of information on psychometric properties, no gold standard is currently available to determine increased motor fatigability in PwMS.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
