Abstract
Background. Reorganization in motor areas have been suggested after motor imagery training (MIT). However, motor imagery involves a large-scale brain network, in which many regions, andnot only the motor areas, potentially constitute the neural substrate for MIT. Objective. This study aimed to identify the targets for MIT in stroke rehabilitation from a voxel-based whole brain analysis of resting-state functional magnetic resonance imaging (fMRI). Methods. Thirty-four chronic stroke patients were recruited and randomly assigned to either an MIT group or a control group. The MIT group received a 4-week treatment of MIT plus conventional rehabilitation therapy (CRT), whereas the control group only received CRT. Before and after intervention, the Fugl-Meyer Assessment Upper Limb subscale (FM-UL) and resting-state fMRI were collected. The fractional amplitude of low-frequency fluctuations (fALFF) in the slow-5 band (0.01-0.027 Hz) was calculated across the whole brain to identify brain areas with distinct changes between 2 groups. These brain areas were then targeted as seeds to perform seed-based functional connectivity (FC) analysis. Results. In comparison with the control group, the MIT group exhibited more improvements in FM-UL and increased slow-5 fALFF in the ipsilesional inferior parietal lobule (IPL). The change of the slow-5 oscillations in the ipsilesional IPL was positively correlated with the improvement of FM-UL. The MIT group also showed distinct alternations in FCs of the ipsilesional IPL, which were correlated with the improvement of FM-UL. Conclusions. The rehabilitation efficiency of MIT was associated with increased slow-5 oscillations and altered FC in the ipsilesional IPL. Clinical Trial Registration. http://www.chictr.org.cn. Unique Identifier. ChiCTR-TRC-08003005.
Keywords
Introduction
Motor imagery training (MIT) or mental practice, which relies on repetitive cognitive rehearsal of physical movements without overt execution, could be used as an adjunctive and noninvasive approach for stroke rehabilitation. Over the past 2 decades, dozens of clinical studies have demonstrated that additional use of MIT to conventional rehabilitation therapies (CRT) such as occupational therapy and physical therapy, can achieve a better outcome than CRT alone (see a review by Park et al 1 ), implying a distinct but complementary neural mechanism of MIT. Understanding the underlying mechanism and identifying the brain targets for MIT can help therapists optimize therapy plans and improve rehabilitation efficiency.
Previous neuroimaging studies have led to substantial advances in our understanding of MIT for stroke rehabilitation. Using task-based (motor execution [ME] and motor imagery [MI] tasks) functional magnetic resonance imaging (fMRI), reorganization after MIT has been observed mainly in the motor areas. Butler et al 2 found a stroke patient showing more focal activation in the contralateral primary motor cortex (M1) or in the ipsilateral ventral processing stream when executing or imagining flexion and extension of the affected hand after 2 weeks of constraint-induced movement therapy plus MIT. Page et al 3 found stroke patients showing increased activation to the affected hand movement in the bilateral premotor cortex and M1, and the contralateral superior parietal lobe after 10 weeks of task-specific practice plus MIT. Liu et al 4 observed more increased activation in the ipsilesional primary somatosensory cortex and attenuated activation in the contralesional M1 during MI and ME in stroke patients after 4 weeks of MIT combined with physical training, and the changes in activation were correlated with the improvement of Fugl-Meyer score. Bajaj et al 5 found enhanced effective connectivity in motor executive network during both MI and ME tasks in stroke patients after 60 hours of MIT or combined MIT and physical therapy, which was also correlated with Fugl-Meyer score. In addition to task-based studies, resting-state analysis, which selected the ipsilesional M1 as the seed, also observed reorganization in functional connectivity (FC) of M1 after MIT. Zhang et al 6 found increased FC with the contralesional M1 and decreased FC with the ipsilesional supplementary motor area and paracentral lobule in stroke patients after 30 days of combined CRT and MIT. Our previous study also found that FC with ipsilesional precentral and postcentral gyri, middle cingulate gyrus, and supramarginal gyrus increased after 4-week CRT combined MIT, but decreased after sole CRT. 7
Despite the existing evidence associating MIT-related brain reorganization mainly with the motor areas in stroke patients, it is not conclusive that the motor areas are the unique neural substrate of MIT for motor recovery. MI has been reported to involve a large-scale frontoparietal network as well as some subcortical regions, including the inferior frontal gyrus, precentral gyrus, middle frontal gyrus, supplementary motor area, insula, superior parietal lobe, inferior parietal lobe (IPL), putamen, thalamus, and pallidum (see a meta-analysis by Hétu et al 8 ). All these brain regions may play a role in MIT. In fact, the activation of M1 during MI remains in debate. Some studies reported less intense activation in M1 during MI than that during ME,9-11 or even no activation of M1 during MI. 12 Instead, some other regions, such as parietal lobe, have shown their importance for MIT. The dorsal pathway, which stretches forward into the parietal lobe, showed increased fractional anisotropy after 4 weeks of MIT in stroke patients. 13 Lesions in the parietal cortex might jeopardize the effectiveness of MIT. 2 Therefore, early studies which used motor tasks for activation analysis2-5 or arbitrarily chose M1 as the seed for FC analysis6,7 might miss the changes outside the motor cortex. On the other hand, the previously observed brain reorganizations in the motor areas might not be MIT-specific. These findings could not exclude the effect of other therapies as no control group was designed3,6 or compared, 5 or potentially compromised by the small or unbalanced sample sizes (eg, sample size in treatment group and control group = 1:12 and 10:54). Considering the complementary therapy efficiency of MIT, we assume that MIT-specific brain reorganization may take place in the brain regions outside the motor cortex, which can be observed by comparing MIT plus CRT with the sole CRT using a randomized design and a relatively large and balanced sample.
Therefore, we designed a randomized controlled study to investigate the brain reorganization that is specifically induced by MIT and potentially contributes to the rehabilitation. Resting-state fMRI was used for brain reorganization analysis. This modality allows to study the brain without the need of goal-directed task, and is more suitable for stroke population with various functional deficits. Compared with task-based fMRI, resting-state fMRI activity measures also showed higher test-retest reliability. 14 As no specific assumption was made about the target regions of MIT, we applied an approach that could quantify differences in resting-state activity across the whole brain, that is, fractional amplitude of low-frequency fluctuations (fALFF). fALFF measures the relative contribution of low-frequency fluctuations within a specific frequency band to the whole acquired frequency range. 15 In particular, the oscillations within the slow-5 range (0.01-0.027 Hz), which are most closely related to gray matter signal, 16 have shown the most extensive change after stroke. 17 Therefore, in this study, we focused on fALFF analysis in the slow-5 band, to identify brain areas with local functioning changes specifically associated with MIT. Furthermore, since the human brain is an integrated system, 18 local change in neural activity could propagate and modulate large-scale brain networks. Therefore, after the targets had been identified, we were also interested in their FC with other brain areas that are also associated with MIT.
Methods
Study Design and Participants
This study used a pretest-posttest, assessor-blinded, and randomized controlled design (see the flowchart of patients through the study in Supplementary Figure S1). Thirty-four patients (32 males, 2 females) were recruited from the inpatient services at Huashan Hospital. The inclusion and exclusion criteria are listed in the Supplemental Material. After the baseline clinical assessment, eligible patients were randomly assigned to an MIT group (16 males, 1 female) or a control group (16 males, 1 female) using a computer-generated random numbers table by an independent researcher. Each patient signed a written informed consent prior to his or her participation. This study was approved by the Review Board of Ethics Committee of Huashan Hospital and registered at the Chinese Clinical Trial Registry (ChiCTR-TRC-08003005).
Three patients (1 female in the MIT group and 2 males in the control group) were excluded from this resting-state fMRI study due to the excessive head motion during MRI scanning. Therefore, in the following analyses, there were 16 patients in the MIT group (16 males) and 15 patients in the control group (14 males, 1 female). To compare the demographic and baseline clinical information between the 2 groups, we performed chi-square tests (for sex, stroke type, and lesion side), independent t test (for age, days after stroke onset, lesion volume, and Modified Barthel Index [MBI]), and Mann-Whitney U test (for Fugl-Meyer Assessment Upper Limb subscale [FM-UL]). The 2 groups did not significantly differ in sex, age, stroke type, days after stroke onset, lesion side, lesion volume, MBI, and FM-UL score (all P > .05, Table 1). The lesion overlap maps for the MIT and the control groups (Figure 1) showed relatively focused lesions in basal ganglia (x = 29, y = −11, z = 10, all left lesions were flipped to the right side). Individual lesion maps were shown in Supplemental Figure S2.
Demographic and Clinical Information for the Participants.
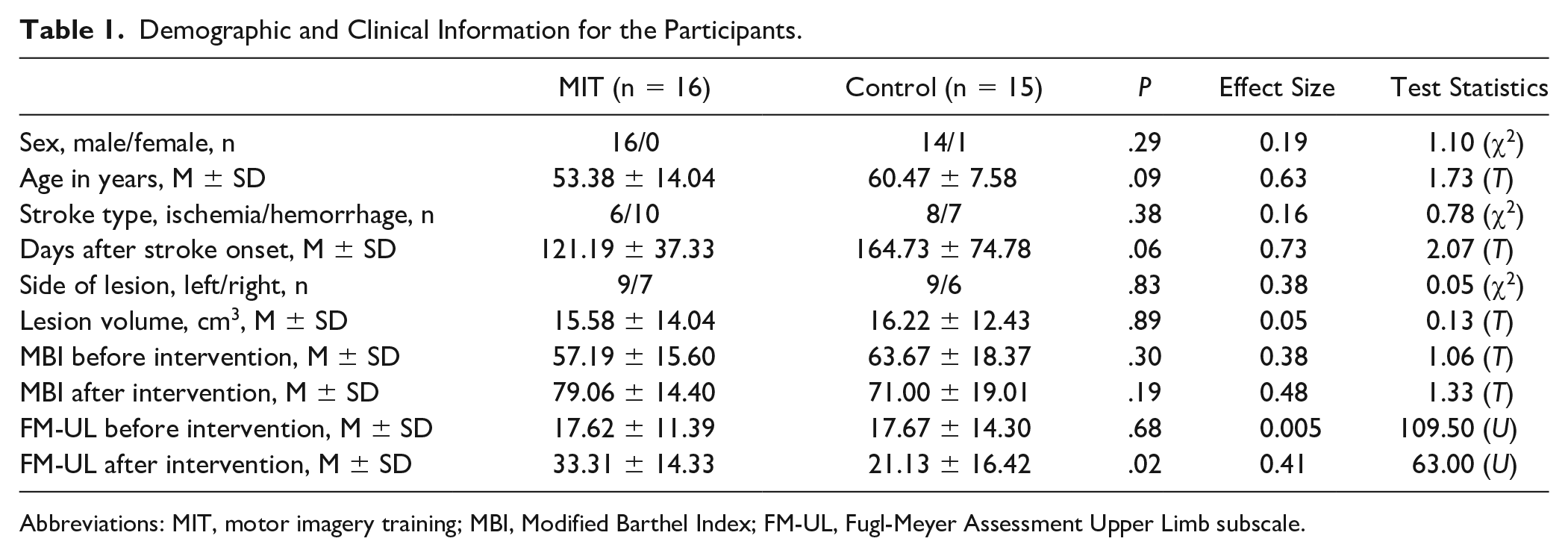
Abbreviations: MIT, motor imagery training; MBI, Modified Barthel Index; FM-UL, Fugl-Meyer Assessment Upper Limb subscale.
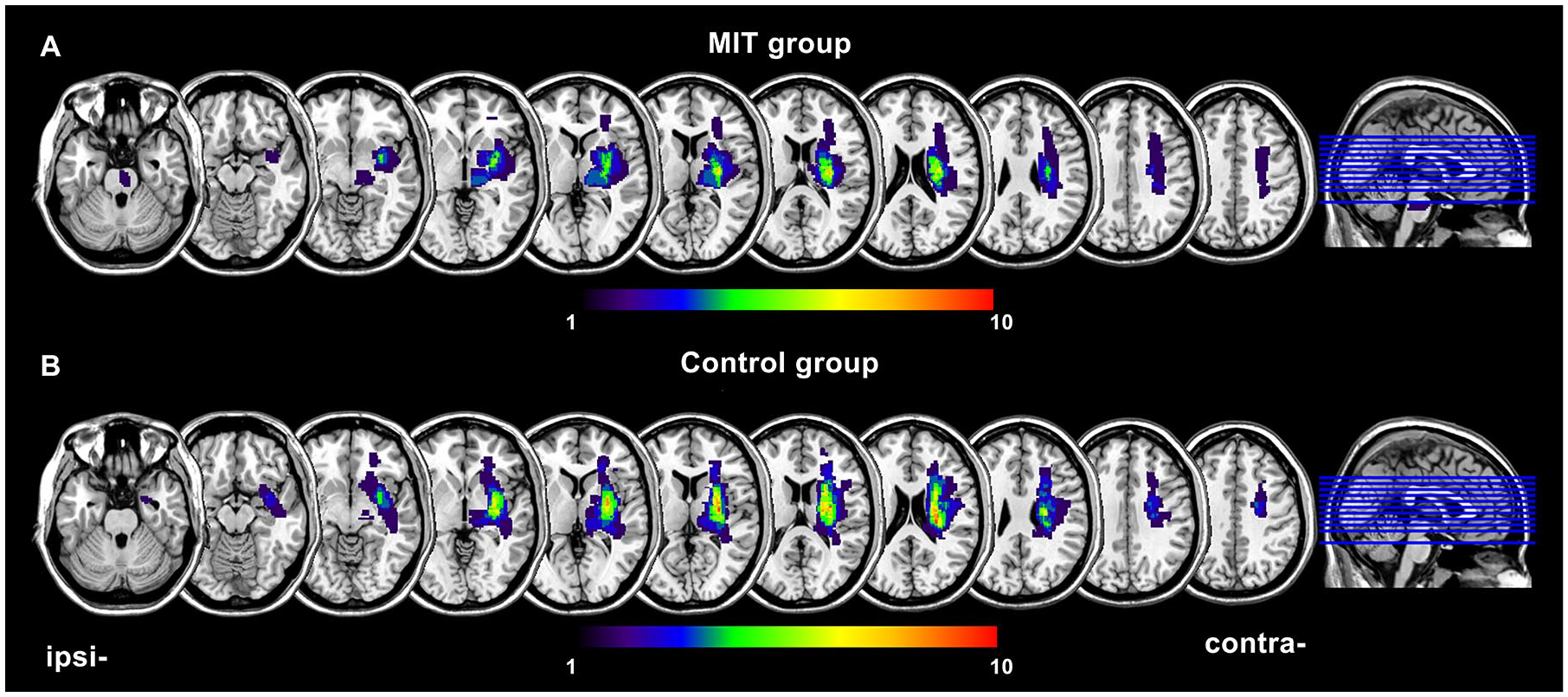
Lesion overlay maps for the motor imagery training (MIT) group (A) and the control group (B). All left lesions were flipped to the right side. Ipsi, ipsilesional; contra, contralesional.
Clinical Intervention
All patients received 3 hours of CRT per day, 5 days per week for 4 weeks by therapists who were blinded to the group assignment. After each day’s CRT, patients received another 30 minutes of treatments, that is, specific motion imagery training for the MIT group and health education on stroke information for the control group. Detailed intervention information is included in the supplemental material. The treatment intensity of MIT has also been adopted in previous clinical studies.19-21
Clinical Outcome Measurement and Imaging Data Acquisition
Before and after the intervention, each patient received an assessment of upper extremity motor function using the FM-UL scale and an assessment of independence in activities of daily living using MBI by an independent physician who was blinded to the treatment condition, and his or her images were collected with a 3.0-T MRI scanner (Siemens Tim Trio, Erlangen, Germany). The MRI scanning included a T1-weighted scan, a T2-weighted scan, and a resting functional scan. Detailed information for scanning and image preprocessing is included in the Supplemental Material.
fALFF and FC Analyses
The fALFF analysis was carried out using DPARSF toolbox. 22 The time series of each voxel from the preprocessed images was transformed into the frequency domain by the fast Fourier transform. The fALFF value in the slow-5 band at each voxel was calculated as the ratio of the power in the slow-5 frequency band (0.01-0.027 Hz) relative to the power in the full frequency range (0-0.25 Hz). The fALFF maps were then converted to Z-score maps by Fisher’s Z-transformation.
CONN toolbox was used for FC analysis. 23 The preprocessed images were band-pass filtered into 0.01 to 0.09 Hz. The voxel with the lowest P value in fALFF analysis (detailed coordination was listed in the results) was selected as the center of spherical seed (radius: 8 mm). FC between each voxel and the seed was calculated as the Pearson correlation coefficient between their time series.
Statistical Analyses
The normality of variables was tested by Shapiro-Wilk’s test, which showed that only FM-UL did not conform to the normal distribution. Therefore, we performed a robust repeated-measures analysis of variance (ANOVA) on FM-UL to verify the efficiency of MIT, taking group (2 levels: MIT and control) as the between-subject factor and time (2 levels: before and after intervention) as the within-subject factor. Post hoc analysis to compare FM-UL between the 2 groups before and after the intervention was conducted by performing the Mann-Whitney U tests. The MBI, which conformed to the normal distribution, were analyzed by repeated-measures ANOVA and t tests post hoc analysis. Using FSL Randomise (www.fmrib.ox.ac.uk/fsl/randomise), voxel wise 2 × 2 (group × time) repeated ANOVAs were carried out to investigate the brain regions with different alterations in fALFF/FC between the 2 groups, and voxel-wise correlation analyses were conducted between the change of fALFF/FC and the improvement of FM-UL. The statistical map was corrected by threshold-free cluster enhancement (TFCE corrected P < .01, H = 2, E = 0.5, number of permutations = 5000) with a cerebral gray matter mask to control the family wise error. The fALFF values from the clusters with significant time × group interaction were extracted and averaged for post hoc analysis, in which paired t tests were used to compare between before and after intervention in each group.
Results
Clinical Improvement
Significant group × time interactions were observed on FM-UL (robust repeated-measures ANOVA) F(1, 29) = 53.75, P < .001, and MBI (repeated measures ANOVA), F(1, 29) = 16.16, P < .001). FM-UL score before the intervention was comparable in the 2 groups (control: 17.7 ± 14.3, MIT: 17.6 ± 11.4, Mann-Whitney U test P = .68, U = 109.5). After the intervention, FM-UL of the MIT group was significantly higher than that of the control group (control: 21.1 ± 16.4, MIT: 33.3 ± 14.3, Mann-Whitney U test P = .02, U = 63.0, Table 1). However, the MBI did not show significant group differences either before or after the intervention (independent t test, both T < 1.33, P > .19).
fALFF in the Slow-5 Band
The repeated ANOVAs on fALFF in the slow-5 band showed significant time × group interactions in the contralesional inferior frontal gyrus, inferior temporal gyrus, and the ipsilesional IPL, and middle temporal gyrus (Figure 2A and B, Supplementary Table S1). Post hoc analysis found that after intervention (Figure 2E), the mean fALFF in the slow-5 band in the contralesional inferior frontal gyrus and the ipsilesional IPL was significantly decreased in the control group but increased in the MIT group; while the mean fALFF in the slow-5 band in the contralesional inferior temporal gyrus and the ipsilesional middle temporal gyrus was significantly decreased in the MIT group but increased in the control group. These areas did not show significant group differences in the baseline fALFF in the slow-5 band.
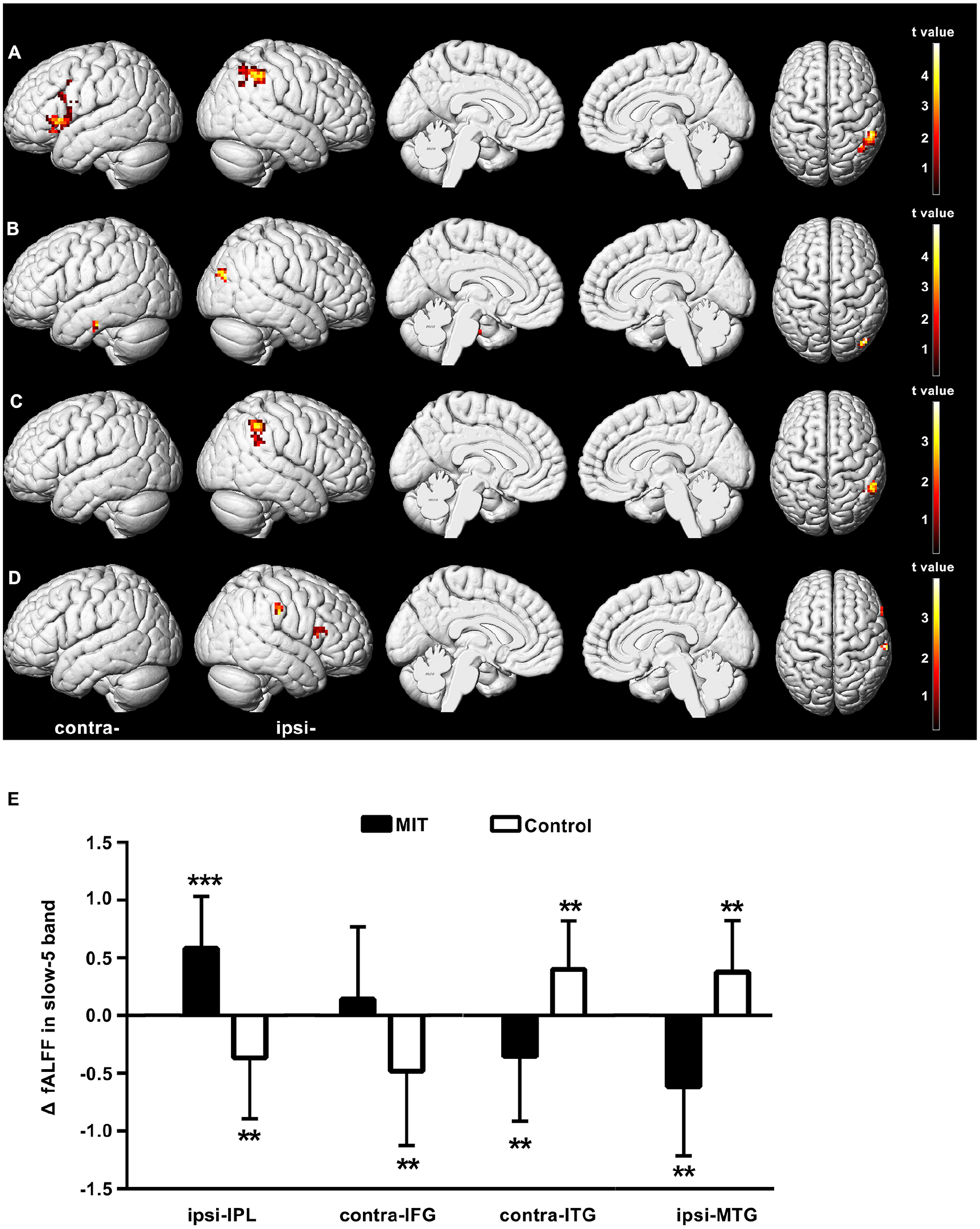
Brain regions in which fractional amplitude of low-frequency fluctuations (fALFF) in the slow-5 band shows significant time × group interactions or correlations with the improvement of Fugl-Meyer upper limb (FM-UL) (threshold-free cluster enhancement [TFCE] corrected P < .01). (A) Brain regions in which fALFF in the slow-5 band was increased in the motor imagery training (MIT) group but decreased in the control group after intervention. (B) Brain regions in which fALFF in the slow-5 band was decreased in the MIT group but increased in the control group after intervention. (C, D) Brain regions in which the change of fALFF in the slow-5 band is positively (C) or negatively (D) correlated with the improvement of FM-UL. (E) Averaged fALFF change in the slow-5 band (ΔfALFF in the slow-5 band) extracted from the clusters with significant time × group interactions. Significant changes after intervention in each group were shown as **P < .01, ***P < .001. IPL, inferior parietal lobule; IFG, inferior frontal gyrus; ITG, inferior temporal gyrus; MTG, middle temporal gyrus; ipsi, ipsilesional; contra, contralesional.
Correlation analysis showed that the change of fALFF in the slow-5 band was significantly positively correlated with the improvement of FM-UL in the ipsilesional IPL (Figure 2C, Supplementary Table S2). Significant negative correlations were observed in the ipsilesional postcentral gyrus and inferior frontal gyrus (Figure 2D, Supplementary Table S2). Importantly, we found that the ipsilesional IPL (cluster size = 44; peak MNI: 54, −45, 45) showed both significant time × group interaction and correlation with the improvement of FM-UL (Figure 3A). That is, in the ipsilesional IPL, fALFF in the slow-5 band was significantly increased in the MIT group (T = 4.22, P < .001) but decreased in the control group (T = 3.02, P = .05), and the change of fALFF in the slow-5 band was significantly positively correlated with the improvement of FM-UL (Spearman correlation, ρ = 0.67, P < .001, Figure 3B).
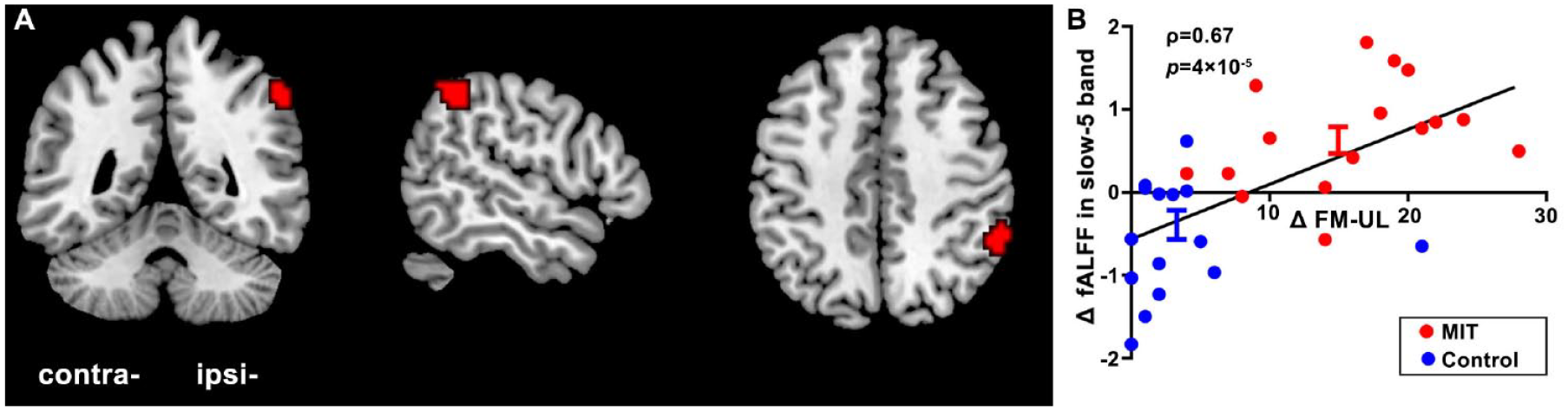
Fractional amplitude of low-frequency fluctuations (fALFF) in the slow-5 band in the ipsilesional inferior parietal lobule (IPL) shows both significant time × group interaction and positive correlation with the improvement of Fugl-Meyer upper limb (FM-UL) score. (A) The location of the significant cluster. (B) Scatterplot demonstrating the relationship between the improvement of FM-UL (ΔFM-UL) and the average fALFF change in the slow-5 band (Δ fALFF in the slow-5 band) extracted from the significant cluster. Ipsi, ipsilesional; contra, contralesional.
FC With the Ipsilesional IPL
The ipsilesional IPL (MNI: 54, −45, 45) was selected as the seed to perform the seed-based FC analysis. The baseline FCs were comparable between the 2 groups. However, FC in several brain regions displayed significant time × group interactions (Figure 4 and Supplementary Table S3). Post hoc analysis (Figure 4E) revealed that FC of the ipsilesional IPL with the bilateral parahippocampal gyrus, the ipsilesional IPL, middle cingulate gyrus, superior temporal gyrus, superior frontal gyrus, medial frontal gyrus, inferior frontal gyrus and angular gyrus, and the contralesional precentral gyrus, middle frontal gyrus, medial frontal gyrus, and middle temporal gyrus was increased in the MIT group but decreased in the control group after intervention. FC in the bilateral middle cingulate gyrus, the contralesional superior frontal gyrus, medial frontal gyrus, insula, and the ipsilesional inferior occipital gyrus was decreased in the MIT group while increased in the control group after intervention (Figure 4E). Moreover, the change of FC in the bilateral superior temporal gyrus, the contralesional postcentral gyrus and lingual gyrus, the ipsilesional fusiform gyrus and middle temporal gyrus, was significantly positively correlated with the improvement of FM-UL (Figure 4C, Supplementary Table S4), while the change of FC in the contralesional medial frontal gyrus and the bilateral middle cingulate gyrus was significantly negatively correlated with the improvement of FM-UL (Figure 4D, Supplementary Table S4). FC in the bilateral parahippocampal gyrus and middle cingulate gyrus, and the contralesional medial frontal gyrus showed both significant time × group interactions and correlations with the improvement of FM-UL (Figure 5, Supplementary Table S5).
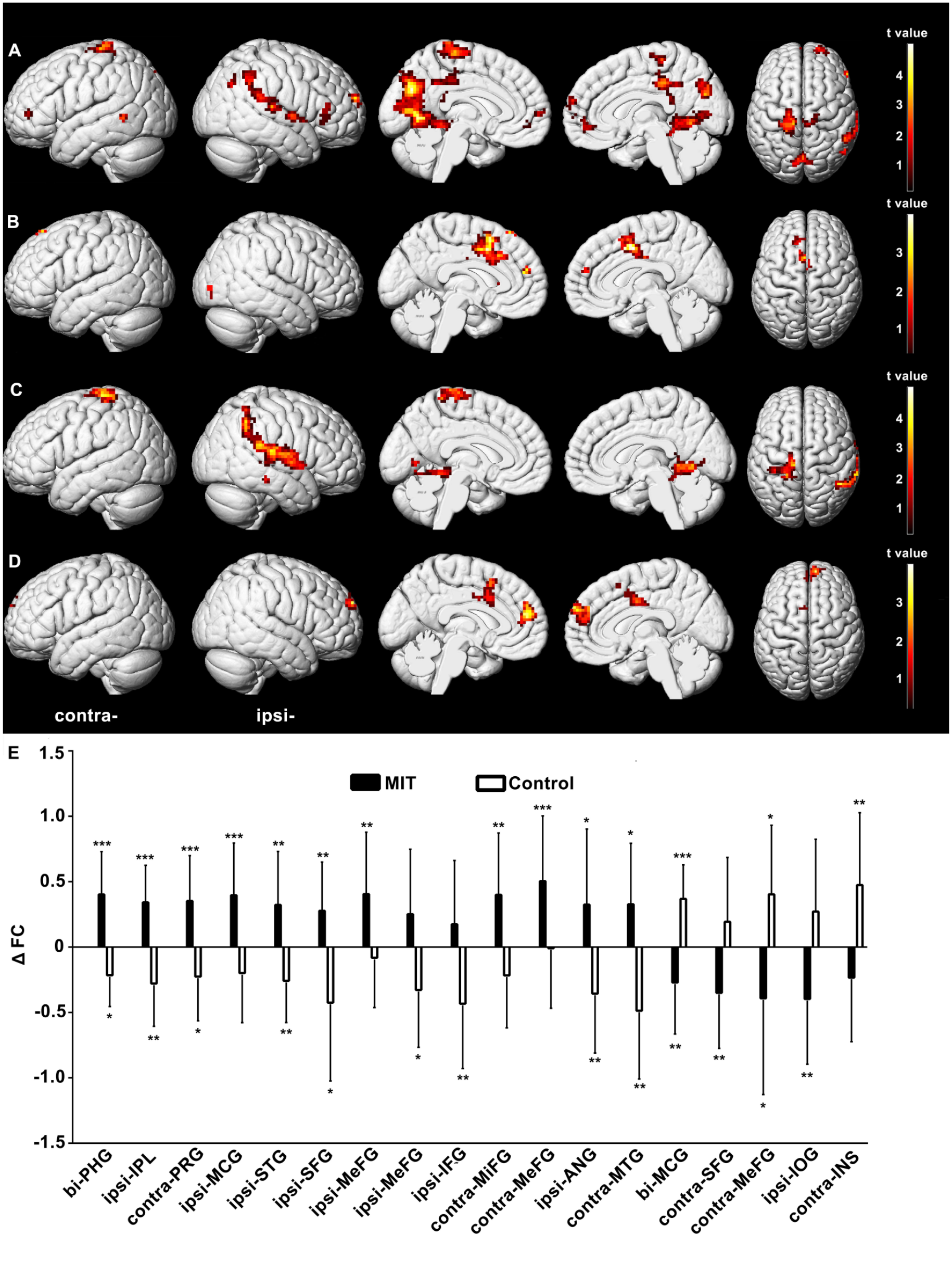
Brain regions in which the functional connectivity (FC) with the ipsilesional inferior parietal lobule (IPL) shows significant time × group interactions or correlations with the improvement of Fugl-Meyer upper limb FM-UL score (threshold-free cluster enhancement [TFCE] corrected P < .01). (A) Brain regions in which FC with the ipsilesional IPL was increased in the motor imagery training (MIT) group but decreased in the control group after intervention. (B) Brain regions in which FC with the ipsilesional IPL was decreased in the MIT group but increased in the control group after intervention. (C, D) Brain regions in which FC with the ipsilesional IPL is positively (C) or negatively (D) correlated with the improvement of FM-UL. (E) Averaged change in FC with the ipsilesional IPL (ΔFC) extracted from the clusters with significant time × group interactions. Significant changes after intervention in each group were shown as *P < .05, **P < .01, ***P < .001. PHG, parahippocampal gyrus; IPL, inferior parietal lobule; PRG, precentral gyrus; MCG, middle cingulate gyrus; STG, superior temporal gyrus; SFG, superior frontal gyrus; MeFG, medial frontal gyrus; IFG, inferior frontal gyrus; MiFG, middle frontal gyrus; ANG, angular gyrus; IOG, inferior occipital gyrus; INS, insula; bi, bilateral; ipsi, ipsilesional; contra, contralesional.
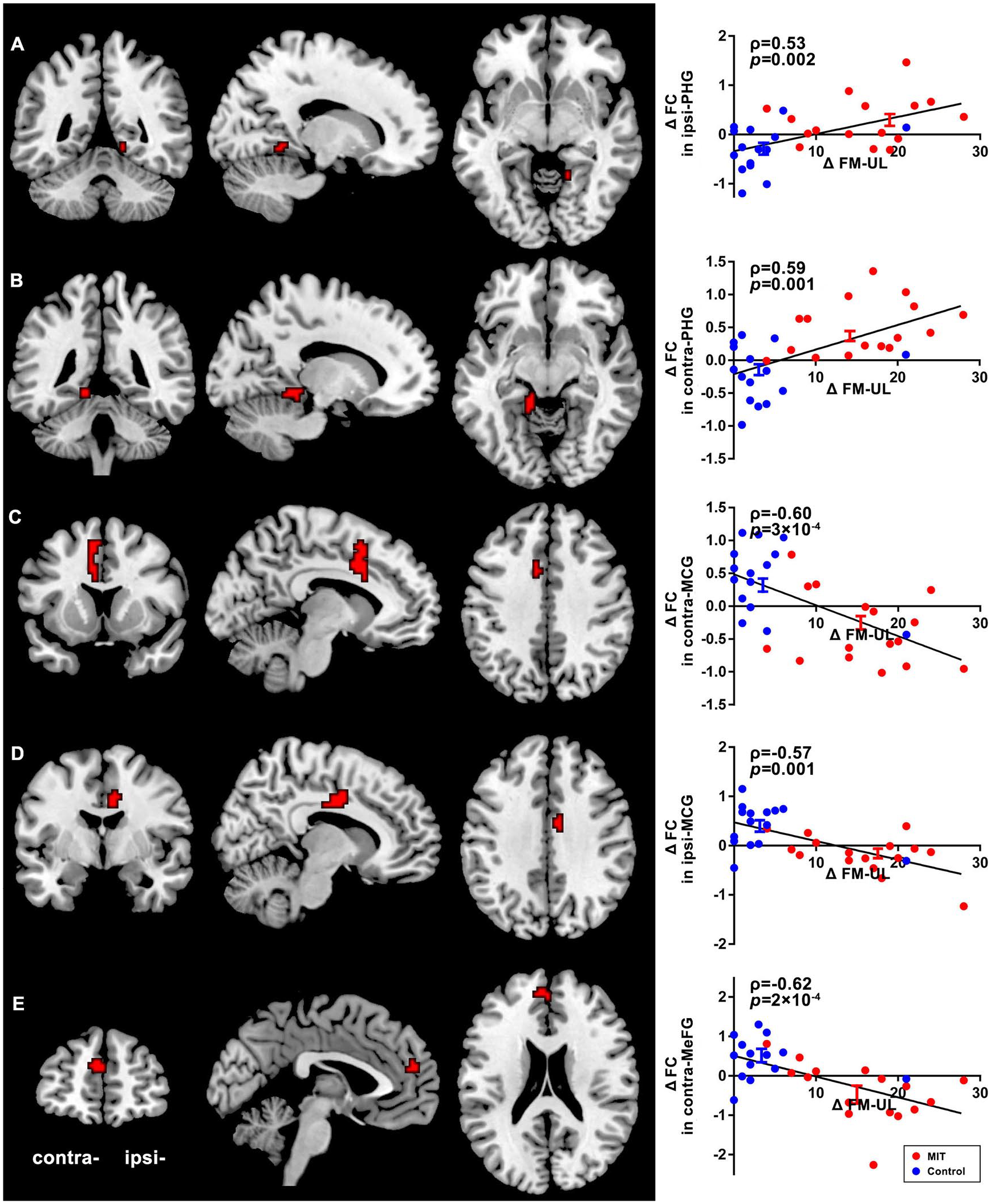
Brain regions in which the functional connectivity (FC) with the ipsilesional inferior parietal lobule (IPL) shows both significant time × group interaction and correlation with the improvement of Fugl-Meyer upper limb (FM-UL) score. (A) The ipsilesional parahippocampal gyrus (ipsi-PHG). (B) The contralesional parahippocampal gyrus (contra-PHG). (C) The contralesional middle cingulate gyrus (contra-MCG). (D) The ipsilesional middle cingulate gyrus (ipsi-MCG). (E) The contralesional medial frontal gyrus (contra-MeFG). The left panels highlight the location of each brain region. The right panels show the relationship between the improvement of FM-UL (ΔFM-UL) and the average change in FC (ΔFC) extracted from the significant clusters.
Discussion
In this study, we used resting-state fALFF to investigate the whole brain reorganization associated with MIT. The major finding is that the ipsilesional IPL rather than the motor cortex seems a potential target for MIT.
First, MIT showed a specific role in increasing the resting activities of the ipsilesional IPL. fALFF in the slow-5 band in the ipsilesional IPL was increased after MIT, but decreased after CRT. IPL is one of the key regions in the human mirror neuron system. 24 Activation of IPL has been observed during the MI task not only in healthy subjects but also in stroke patients.8,25,26 Inhibition of IPL by repetitive transcranial magnetic stimulation (rTMS) could result in an impairment of MI performance. 27 Given that repeated activation of certain brain areas could promote long-term neural plasticity, 28 it is no surprise that neural reorganization in the IPL was observed after MIT. The different IPL reorganization was only observed in the ipsilesional hemisphere. The lateralized result is reasonable since the activation of the ipsilesional IPL in stroke patients have shown more significant differences between MI and ME than that of the contralesional conterpart. 29 Considering that the ipsilesional IPL was more active during MI compared to ME in stroke patients, 29 it makes sense that the increase of the slow-5 oscillations in the ipsilesional IPL was only observed in the MIT group but not in the control group.
Second, the distinct reorganization in the ipsilesional IPL in the MIT group was demonstrated to contribute to the better motor recovery in this group. The increase of fALFF in the slow-5 band in the ipsilesional IPL was positively correlated with the improvement of upper limb motor function. During passive movement, activation of the ipsilesional IPL was also found to be positively correlated with clinical score and a predictor of faster or better recovery in stroke patients. 30 IPL, where sensory and motor information is integrated, 31 has been suggested to be associated with the forming of motor intentions, that is, high-level cognitive plans for movements. For example, fMRI studies demonstrated that the IPL was activated when shifting the motor intention in a response switching paradigm, 32 during the planning of tool use 33 or in selection of the required movement and the action preparation. 34 Lesions in the IPL could result in deficiency in planning of object-related hand posture 35 or motor prediction for visuo-motor temporal integration. 36 Disruption of the supramarginal gyrus (a structure in the IPL) by rTMS could cause interference with motor attention. 37 Thus, we speculate the increase of the slow-5 oscillations in the ipsilesional IPL as an indicator of motor planning refinements, implying that MIT may promote motor recovery by refining the performance of motor planning. This speculation is in line with previous behavioral studies demonstrating that the motor planning performance was correlated with the motor imagery ability, 38 which could be improved by MIT. 39 Given existing behavioral paradigm can help measure the patients’ ability of motor planning, 40 pretreatment assessment of motor planning could be used to select most beneficial patients from MIT. However, more direct evidence is required to demonstrate the relationship between motor planning and MIT.
It should be noted that MIT also induced a different reorganization in other brain regions, including the contralesional inferior frontal gyrus and inferior temporal gyrus, and the ipsilesional middle temporal gyrus. It is no surprise that the resting activities of these brain regions were changed after long-term MIT, as they were found to be activated during motor imagery.2,41,42 However, reorganizations in these brain regions did not show significant correlations with motor recovery.
Third, FC between the ipsilesional IPL and the bilateral parahippocampal gyrus was increased after MIT, which was also related to motor recovery. These 2 structures have been found functionally and structurally connected by previous neuroimaging studies. 43 IPL has been suggested to be associated with motor planning; while parahippocampal gyrus has been found to be involved in motor learning and motor memory consolidation. For example, activation of parahippocampal gyrus has been seen in learning and recall of a motor sequence, 44 and was positively correlated with the learning scores in a visuomotor tracking task. 45 Considering the important roles of IPL and parahippocampal gyrus in motor planning and motor learning, respectively, the increased FC between these 2 areas may imply an enhanced association between motor planning and motor learning after MIT. Our correlation analysis suggested that this enhanced association seems crucial for motor recovery. One possibility is that MIT could directly enhance motor planning, which promotes motor learning via motor-related memory consolidation. Another possibility is that motor-related memory learned by intense MIT could help patients optimize motor planning, and thus, achieve better motor outcome. Our explanation is reasonable as a more refined motor plan has usually been found to be coupled with better motor learning performance, 46 and MIT has been behaviorally demonstrated effective to facilitate motor skill learning (eg, perceptual-motor skills and sequential finger tapping) in healthy subjects.47-50
In addition to the increased FC with the bilateral parahippocampal gyrus, the ipsilesional IPL also showed decreased FC with the bilateral middle cingulate gyrus and the contralesional medial frontal gyrus after MIT. The IPL is one of the key nodes in multiple resting-state networks, such as the default mode network (DMN), the frontoparietal control network (FCN), and the executive control network (ECN) 51 ; while the medial frontal gyrus and middle cingulate gyrus are also included in the DMN and FCN, respectively. Therefore, our results implied a possibility of reduction in intra-DMN and intra-FCN connectivity after MIT. Consistent with our study, a previous longitudinal study found a decrease in the connectivity of the medial frontal gyrus in the DMN with stroke recovery. 52 One possible explanation is that more efficient reorganization brought by MIT may inhibit compensatory reorganization in the FCN or DMN after stroke. Our results indicated that MIT can recruit a large scale of brain networks outside the motor cortex to achieve better recovery.
Several limitations should be considered in the current study. First, we only used FM-UL to assess the upper limb motor function. More clinical assessments such as Wolf Motor Function Test, Action Research Arm Test, and Box and Block Test, could strengthen our study. Second, although the gender distribution was matched between the 2 groups, the low number of female participants (only 1 in the 2 groups) might impair the generalizability of our conclusion. 53 Future studies with more female participants are beneficial for further confirming our results. Third, the age and days after stroke are marginally different between the two groups. Considering that age was not a significant predictor for recovery outcome54,55 and our participants were all in chronic phase, we think the slight differences would not affect our results significantly. Nevertheless, more balanced participants should be further recruited to draw more solid results.
In conclusion, we found that the better motor recovery in stroke patients obtained by the additional use of MIT was related to the increased slow-5 oscillations and altered FC in the ipsilesional IPL. These resting-state brain reorganizations suggested that MIT might promote motor recovery through refinements in motor planning and facilitating motor learning. This study demonstrated the important role of the ipsilesional IPL for MIT, which could also provide valuable suggestions for training. Previous studies have suggested the necessity of parietal integrity for MI. Parietal lobe damage was shown to impair or altogether prevent the performance of MI. 56 A patient with a lesion restricted to the parietal cortex showed little improvement in upper-extremity function from MIT, in comparison with another patient with damage to nonparietal areas. 2 The current study suggested a more focused parietal area (ie, IPL), implying that the patients with lesions in the IPL maybe insensitive to MIT. However, whether use IPL lesions as an exclusion criterion for MIT, should be further demonstrated by clinical studies. On the other hand, this area could be also served as a potential target for brain stimulation techniques (like rTMS) to improve motor planning and motor recovery.
Supplemental Material
supplemental_material_clean – Supplemental material for Motor Imagery Training After Stroke Increases Slow-5 Oscillations and Functional Connectivity in the Ipsilesional Inferior Parietal Lobule
Supplemental material, supplemental_material_clean for Motor Imagery Training After Stroke Increases Slow-5 Oscillations and Functional Connectivity in the Ipsilesional Inferior Parietal Lobule by Xu Wang, Hewei Wang, Xin Xiong, Changhui Sun, Bing Zhu, Yiming Xu, Mingxia Fan, Shanbao Tong, Limin Sun and Xiaoli Guo in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank Stephen Kaputsos for his help in proofreading.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 61771313, 81974356, 81401859 and 81471651) and the Natural Science Foundation of Shanghai (No. 17ZR1445300).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
