Abstract
Keywords
Introduction
Somatosensory function is central to the perception of limb position, to movement and particularly multijoint coordination,1,2 to adaptation following perturbation to movement, 3 and to the reacquisition of skills following stroke. 4 When passive somatosensory training is used in healthy adults and is paired with proprioceptive decision making and reinforcement, quite striking changes are observed in perceptual function and movement and in motor networks of the brain. Perceptual training conducted in this fashion is found to result in improvements in perceptual acuity, and it has also been shown to produce improvements to the rate and extent of subsequent motor learning 5 in normal individuals. The neuronal correlates of these behavioral changes have recently been investigated using resting-state functional magnetic resonance imaging (rs-fMRI). This task-free fMRI method detects training-induced changes in the synchronicity of the hemodynamic response of neuron groups throughout the brain, thus defining networks of high synchronous activity. Vahdat and colleagues have recently documented learning-induced changes in resting-state networks (RSNs) following motor 6 and perceptual 7 training using hypothesis-driven (seed-based) and hypothesis-free methods (independent component analysis [ICA]8,9). These changes were associated with improvements in behavioral task performance in healthy individuals (see Guerra-Carrillo et al 10 for a review). Sensory recovery also indicates a capacity for motor improvements in stroke patients and may be essential in the initiation of adaptive reorganization in motor systems. 11 In a series of PET experiments, we 12 and others13-15 investigated changes in the pattern of brain activation during passive movements in hemiplegic patients with subcortical stroke. These studies demonstrated the importance of passive training in inducing functional reorganization in bilateral sensorimotor areas. rs-fMRI has been used to study network disruption in acute and chronic stroke patients (see Thiel et al 12 for review). These studies seem to suggest that reduced interhemispheric connectivity correlates with functional impairment and that a well-balanced interhemispheric control of excitatory and inhibitory connections is required for optimal execution of sensorimotor function. 16 Decreased interhemispheric connectivity in the acute phase gradually increases during the subacute stage and tends to renormalize to prestroke values in well-recovered chronic patients.17-19 The effect of therapeutic intervention on RSN connectivity, however, remains to be studied.
Different forms of somatosensory stimulation such as peripheral nerve stimulation,20,21 muscle tendon vibration, 22 or paired associative stimulation 23 have been shown to enhance corticospinal excitability or induce changes in corticomotor representation in healthy individuals.20-22 In some cases, 24 improvements in functional measures of motor performance (such as muscle strength or number of correct key presses in motor sequence practice) have been reported in stroke patients. Compared with these forms of somatosensory stimulation, which generally involve focal afferent stimulation, passive perceptual training can access more of the distributed sensorimotor network.12,25 Somatosensory perceptual training can also be used in rehabilitation therapy whenever voluntary movement of a paretic limb is either too difficult or impossible. However, the effectiveness of passive perceptual training, specifically after a short training session, remains a matter of debate. 26
With this proof-of-principle study, we seek to answer the question of whether somatosensory training–induced changes in sensory motor connectivity can be detected in stroke patients and whether these changes result in improved movement accuracy, similar to normal controls. Robot-controlled passive perceptual training would have potential for developing new rehabilitation strategies that may specifically be applied in patients with prolonged stays in acute hospitals, who are too severely affected to participate in active training.
Methods
Patients
A total of 10 chronic stroke patients (>6 months after symptom onset) were recruited from the secondary stroke prevention clinic of the Jewish General Hospital, Montréal (see Table 1 for clinical and demographic data). Following informed consent, all patients underwent behavioral testing, including Fugl-Meyer Assessment for upper extremity (FMA) and Nottingham Sensory Assessment (NSA).
Patient Demographic and Clinical Data.
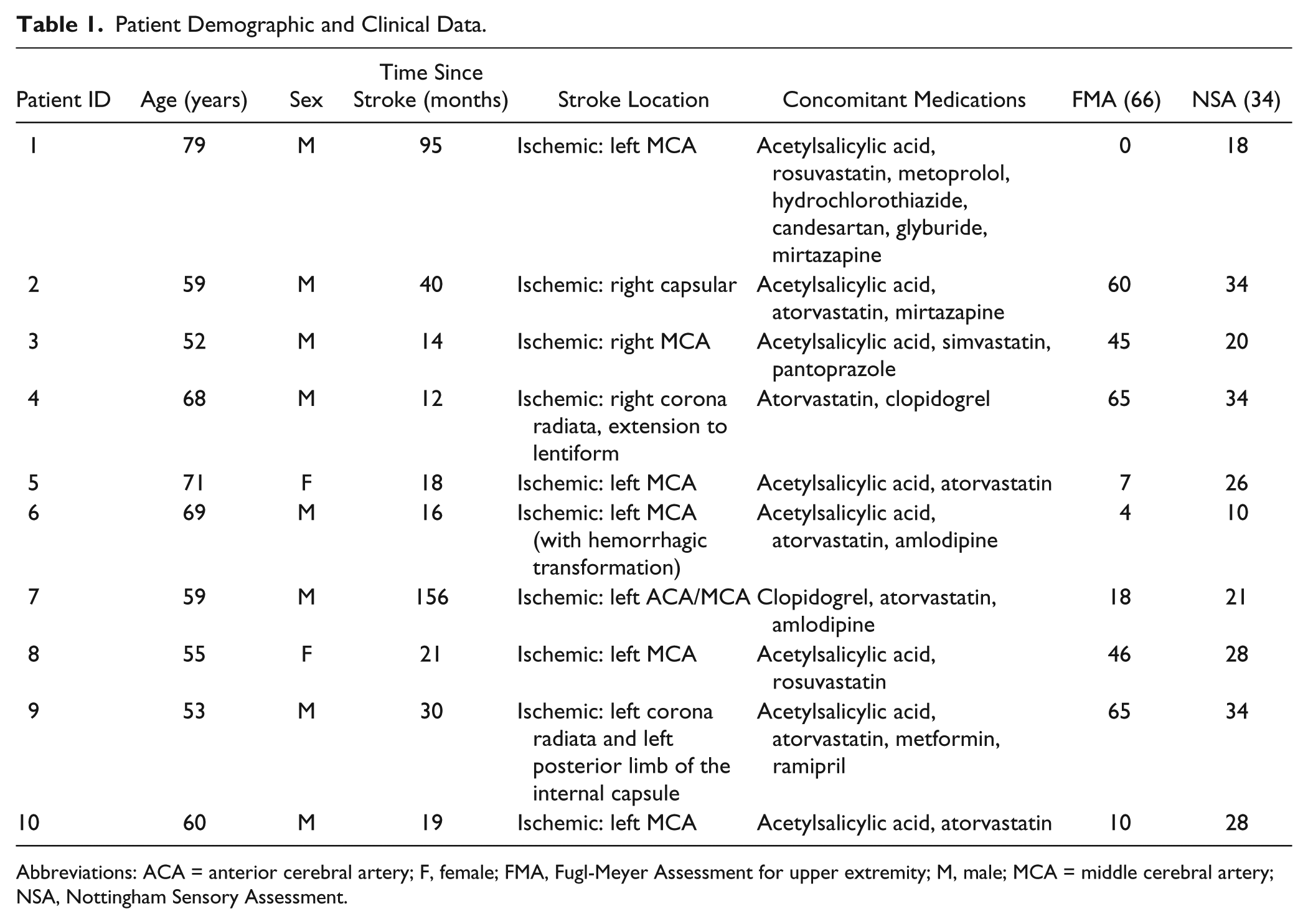
Abbreviations: ACA = anterior cerebral artery; F, female; FMA, Fugl-Meyer Assessment for upper extremity; M, male; MCA = middle cerebral artery; NSA, Nottingham Sensory Assessment.
Behavioral Procedures
Patients were seated in front of a computer screen, and the patient’s paretic arm was attached to a 2 degree-of-freedom robotic arm (In Motion2, Interactive Motion Technologies) using a custom-made plastic arm cast.
On the first day of the experiment (day 1), patients first underwent an initial test of reaching accuracy, in which they were required to move the robotic arm from a start to a target position as straight as possible (see Supplementary Methods for details). Six patients performed 2 to 4 blocks of 50 reaching movements each, depending on their comfort level. Patients rested for 10 minutes between each practice block. Four patients could not actively and steadily move the robotic arm. For these patients, the robot passively moved the arm along the straight line connecting the start and end positions in 4 blocks of 50 movements each. Afterward, patients were transferred to the brain imaging center for structural and functional MRI scans (see below).
The following day (day 2), patients underwent 1 session of 4 blocks of somatosensory training,
5
in which the paretic arm was again attached to the robotic handle. Each block consisted of 50 trials, in which the robot moved the arm outward on one of a set of fan shaped trajectories that deviated from the body midline by up to
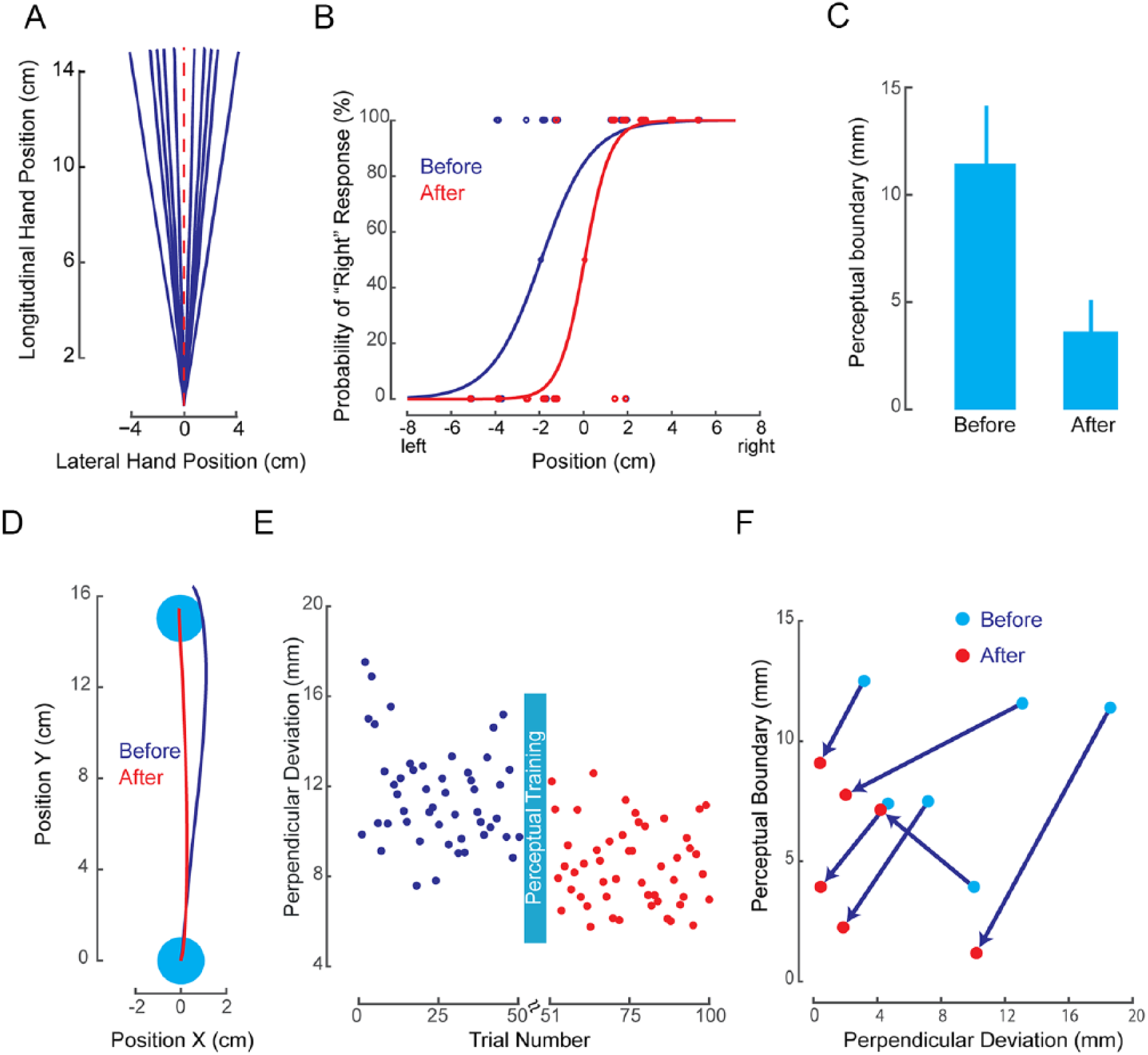
Somatosensory training procedure and behavioral results. A. In somatosensory training blocks, the robot passively displaced the individual’s arm along 1 of 10 trajectories (blue lines). Top-down view shows the entire fan-shaped displacement pattern. B. Systematic shift in the psychometric function of a representative patient as a result of somatosensory learning (dots show tested limb positions and binary responses; before and after denote the first and last block of perceptual training). C. The perceived boundary between left/right becomes more accurate over the course of somatosensory training (mean across all patients, error bars represent standard error of the mean). D. Sample reaching movement trajectories of a representative patient before (blue) and after (red) somatosensory training. Reaching movements become straighter following somatosensory training. E. Lateral movement deviation before and after somatosensory learning averaged across patients. The patients’ lateral deviation significantly decreased following somatosensory learning (
Behavioral Analysis
For reaching movements, the perpendicular deviation of the hand from a straight line, connecting start and end positions at the time of maximum tangential velocity was calculated (Figure 1D), obtaining quantitative estimates of movement straightness and, thus, accuracy. For somatosensory training, patient’s perception of the boundary between left and right was estimated using the method of constant stimuli. We obtained an estimate of the perceptual boundary between right and left for each patient separately by fitting a logistic function to that patient’s entire set of lateral deviations and associated binary (right/left) responses (Figure 1B). The 50% point of the psychometric function defined the perceptual boundary.
Image Preprocessing
All fMRI data were acquired using a 3 Tesla Siemens Trio MR scanner at the Montréal Neurological Institute (MNI). Data processing was carried out using the FSL software package (http://www.fmrib.ox.ac.uk, FMRIB, Oxford, UK), FSL version 4.1.27,28 We used the same preprocessing pipeline as described previously. 6 Data sets for the 2 patients with right hemisphere strokes were left-right mirrored along the anterior-posterior axis. Details of imaging procedure and preprocessing are reported in Supplementary Methods.
Functional Localizer Task
Patients underwent a passive arm movement task conducted in the scanner on day 2 to define the locations of seed coordinates used in the seed-based functional connectivity (FC) analysis. The details of the localizer task and analysis are reported in Supplementary Methods. For consistency across patients, we only selected contralesional seed regions, so that all patients had meaningful BOLD signals outside their lesion mask. The MNI coordinates of seed voxels, the
Seed-Based FC Analysis
We defined a spherical mask (radius = 6 mm) around each seed in MNI standard space. The average time course of the BOLD signal within the transformed region of interest (ROI) mask during each resting-state scan was then calculated and used as the regressor of interest in a subject-level GLM to assess the FC of each ROI with every other voxel in the brain (see Supplementary Method). This analysis produced maps of all voxels that were positively or negatively correlated with an ROI’s mean time course.
This was followed by between-subjects analyses using a mixed-effects model (FLAME) implemented in FSL.
29
To detect changes in FC related to patients’ clinical scores, FMA and NSA scores were used as regressors to obtain a weighted average of the difference between scans (posttraining compared to pretraining conditions) across patients.
6
Corrections for multiple comparisons at the cluster level were carried out using Gaussian random field theory (minimum
Shared and Specific ICA (SSICA)
Given the heterogeneity in infarct location and size in the sample, we also assessed training-induced FC changes in each individual patient. SSICA8,9 systematically classifies independent components (brain networks) that are specific to each experimental condition/group and represent a significant difference in FC across conditions.
Here, SSICA was applied on the resting-state data set to identify those brain networks
7
that are specific to the posttraining condition because these reflect the connectivity changes induced by the training. As part of SSICA preprocessing (see Supplemental Methods), the dimension of subject-level functional data for each session was reduced from 2000 (2 runs, 1000 volumes per run) to 50, corresponding to at least 80% of total variability per individual. To extract the components across conditions, for each patient, the reduced rs-fMRI data for the pretraining and posttraining sessions were time concatenated and fed to the SSICA algorithm. We extracted 30 networks using the SSICA algorithm and let the algorithm extract up to 2 specific networks per condition. Corrections for multiple comparisons at the cluster level were performed using Gaussian random field theory as implemented in FSL software (
Finally, for each specific network, we calculated the volume of activation in each hemisphere (NetVol), which is the sum of all voxels in each hemisphere showing training-associated FC change. An asymmetry index was defined based on NetVol values in the ipsilesional and contralesional hemispheres as follows:
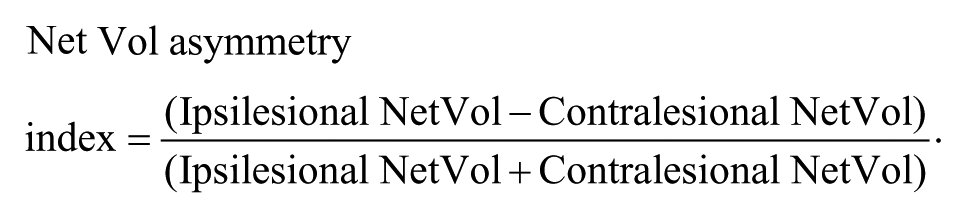
Results
Behavioral Results
Eight of 10 patients completed 4 blocks of somatosensory perceptual training. The training resulted in a more accurate perception of right-left boundary at the body midline [
For the 6 patients who could perform the active arm reaching test, we collected 2 blocks of reaching movements before and after the perceptual training. Figure 1E shows the group average of these data on a trial-by-trial basis. Patients’ reaching movements following perceptual learning became straighter than before [
We also investigated the relationship between improvement of movements and changes in perception of limb position. For 5 of the 6 patients who performed the active reaching test, a shift in somatosensory perception toward the body midline coincided with an improvement in movement (less curvature) from before to after perceptual training (Figure 1F). The sixth patient also showed an improvement in movement, but this patient had the best baseline perceptual estimates, and there was no room for improvement. A
Neuroimaging Results: Seed-Based Analysis
Motor function as determined by FMA scores was correlated with training-induced FC changes in a network comprising supramarignial gyrus (SMG) (seed) and ipsilesional SMG and S1 (Figure 2A). A post hoc analysis of FC values before and after training showed that these areas became more positively correlated posttraining (Figure 2B). Also, as confirmed by Pearson correlation (Figure 2C), those patients with a greater residual motor function showed a greater FC change between bilateral SMG and S1 following training.
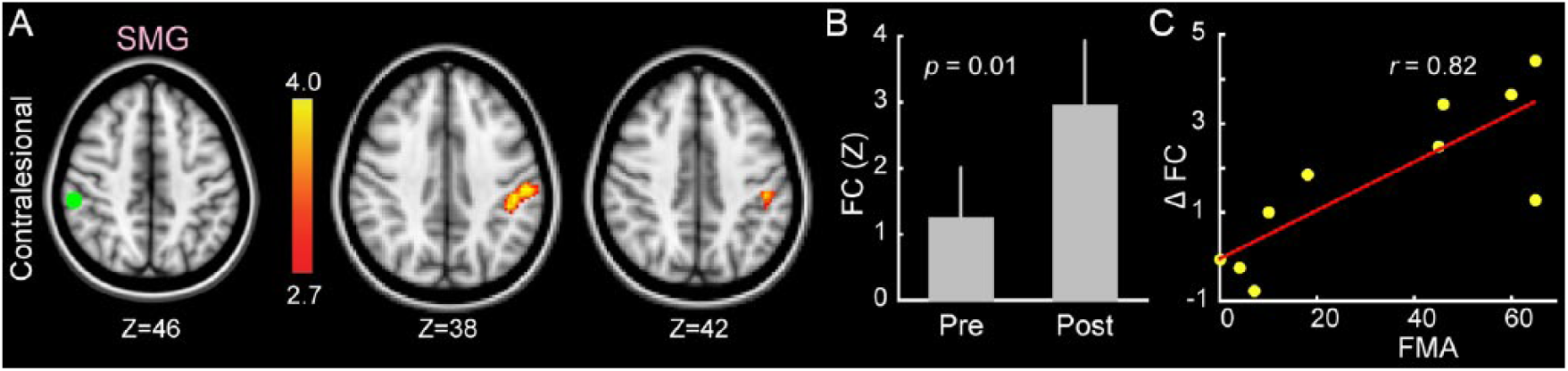
Training-induced functional connectivity changes associated with Fugl-Meyer Upper Extremity Scale (FMA). A. Functional connectivity between the bilateral supramarginal gyri increased significantly from pretraining to posttraining sessions in relation to patients’ FMA scores. The seed location is shown in green (left axial slice), and the color-coded map represents
Two networks showed a significant change in FC with regard to sensory function as measured with NSA scores. Contralesional S1 increased its connectivity with a network of bilateral SMA, M1, and pre-motor cortex (PMC) (Figures 3A-3C). Pearson correlation confirmed that those patients with greater NSA scores showed a greater FC increase within this network. Furthermore, a seed ROI in the ipsilesional cerebellar cortex (lobule VI) showed a significant change in FC (increased negative correlation) with a network of bilateral SMG and S1 areas (Figures 3D and 3E, bottom row). Again, the amount of increase in negative connectivity was related to patients’ residual somatosensory function (Figure 3F, scatter plot).
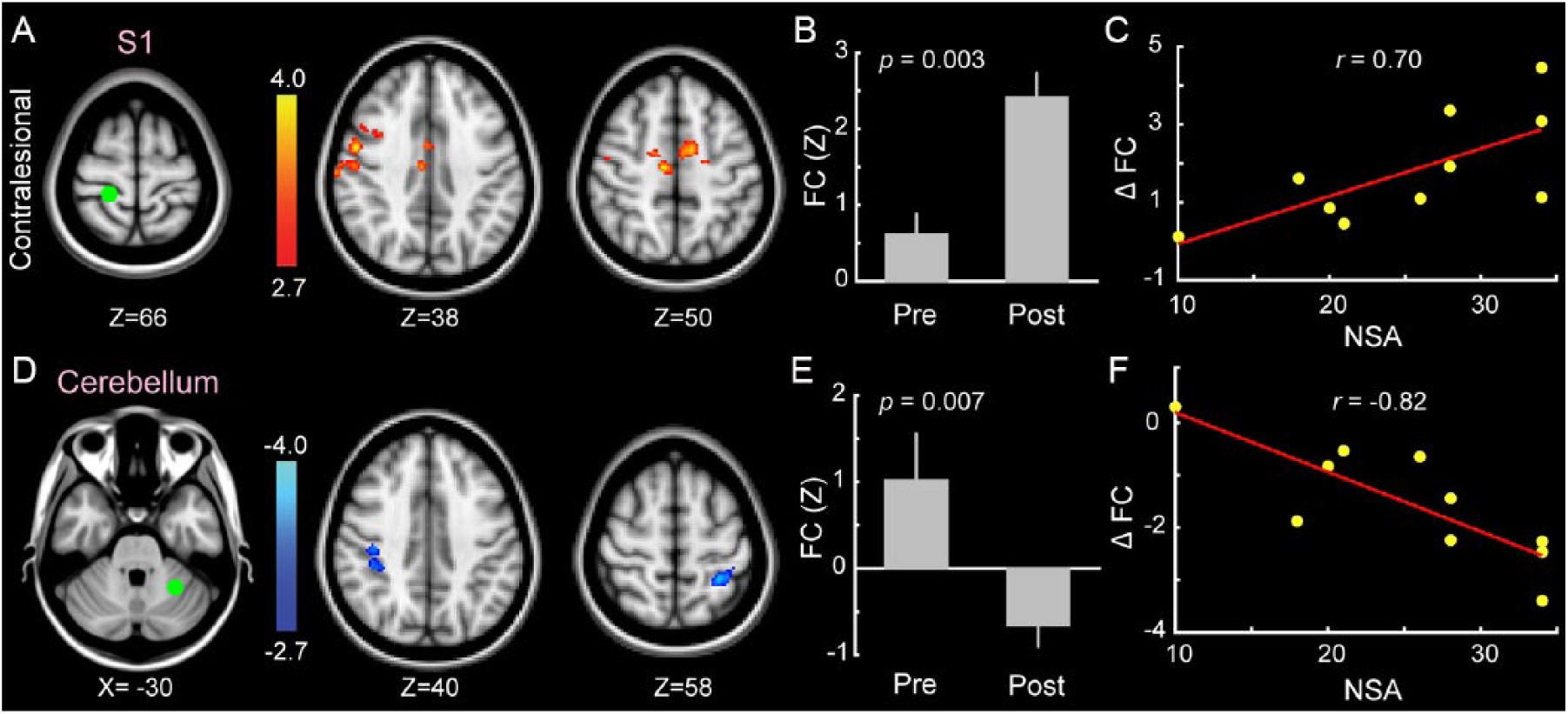
Training-induced functional connectivity changes associated with Nottingham sensory assessment (NSA). A, D: color-coded Z-score maps show clusters whose FC changed significantly from pre- to post-training sessions in relation to patients NSA score. The FC between the contralesional BA3 and contralesional M1, S1 and SMA significantly increased in association to NSA scores (A). Likewise FC changed significantly between the ipsilesional cerebellar cortex (lobule VI) and contralesional supramarginal gyrus and ipsilesional superior parietal cortex (D). B, E: bar plots show average Z-scores in the associated clusters in the pre- and post-training sessions. C, F: patients with greater change in FC in these connections (shown in A, D) showed a greater residual sensory function as measured by NSA.
We also performed the seed-based analysis to identify brain regions correlated with the behavioral changes. Only the 6 patients with kinematic and 8 patients with perceptual boundary data were included in this analysis, which resulted in no significant correlation between FC changes and changes in behavior (mixed-effect analysis, cluster level threshold
To investigate the specificity of our results, we performed additional analyses using seed locations outside the sensorimotor network (including visual [MNI coordinates:
Neuroimaging Results: ICA
Brain networks (components) with significantly higher FC in the posttraining compared with the pretraining condition are shown in Figure 4 sorted from top to bottom based on patient’s FMA scores. The 2 networks are color-coded in orange (C1) and blue (C2) for each patient. C1, identified in all patients (
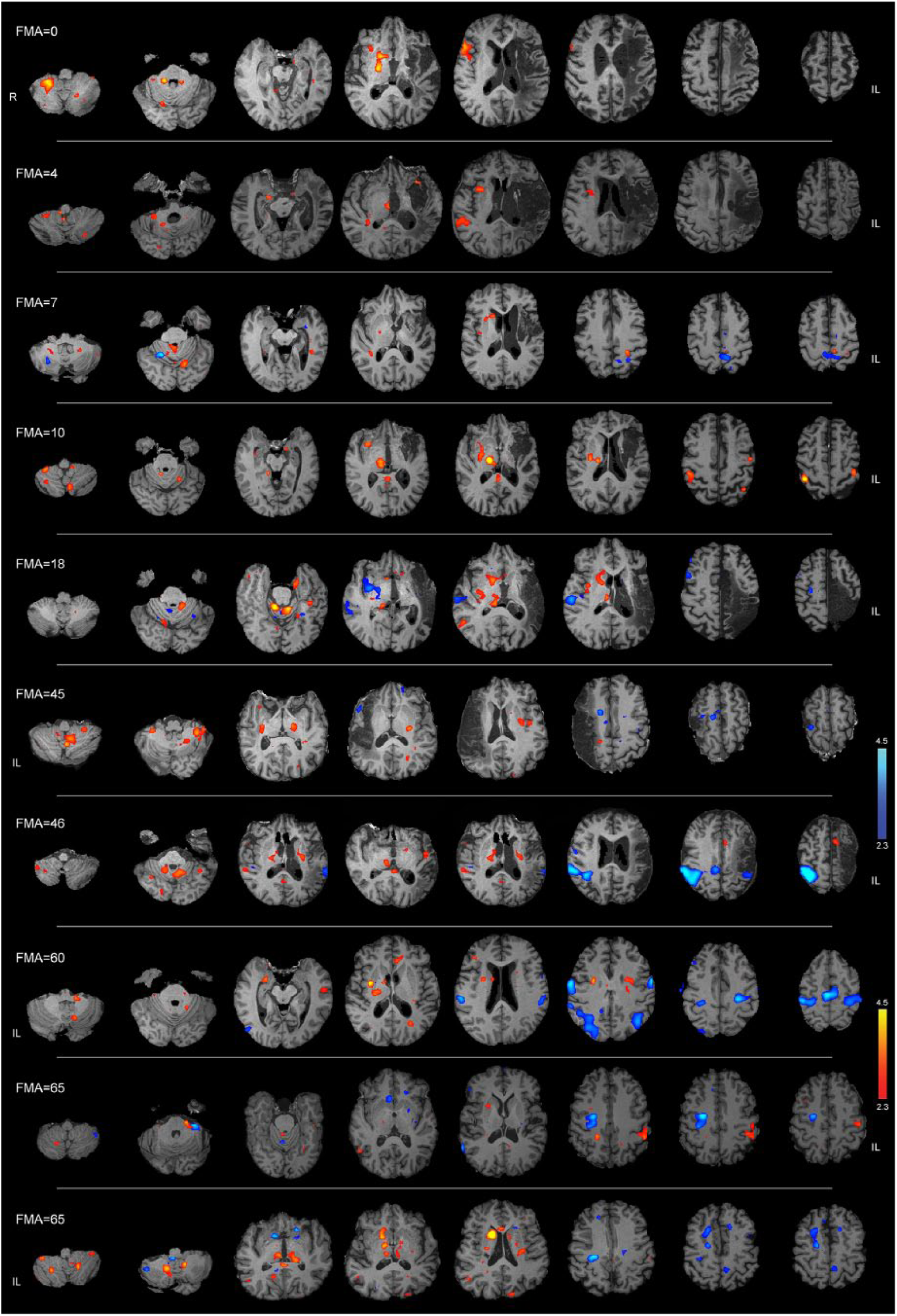
Brain networks in individual patients showing greater connectivity in the posttraining compared with pretraining condition. Each row shows the color-coded
We then calculated the network volume (NetVol) in each hemisphere of each patient for each network alone (10 patients and 7 patients) and for both networks combined (10 patients). There was no significant difference between the NetVol of the ipsilesional and contralesional hemispheres (
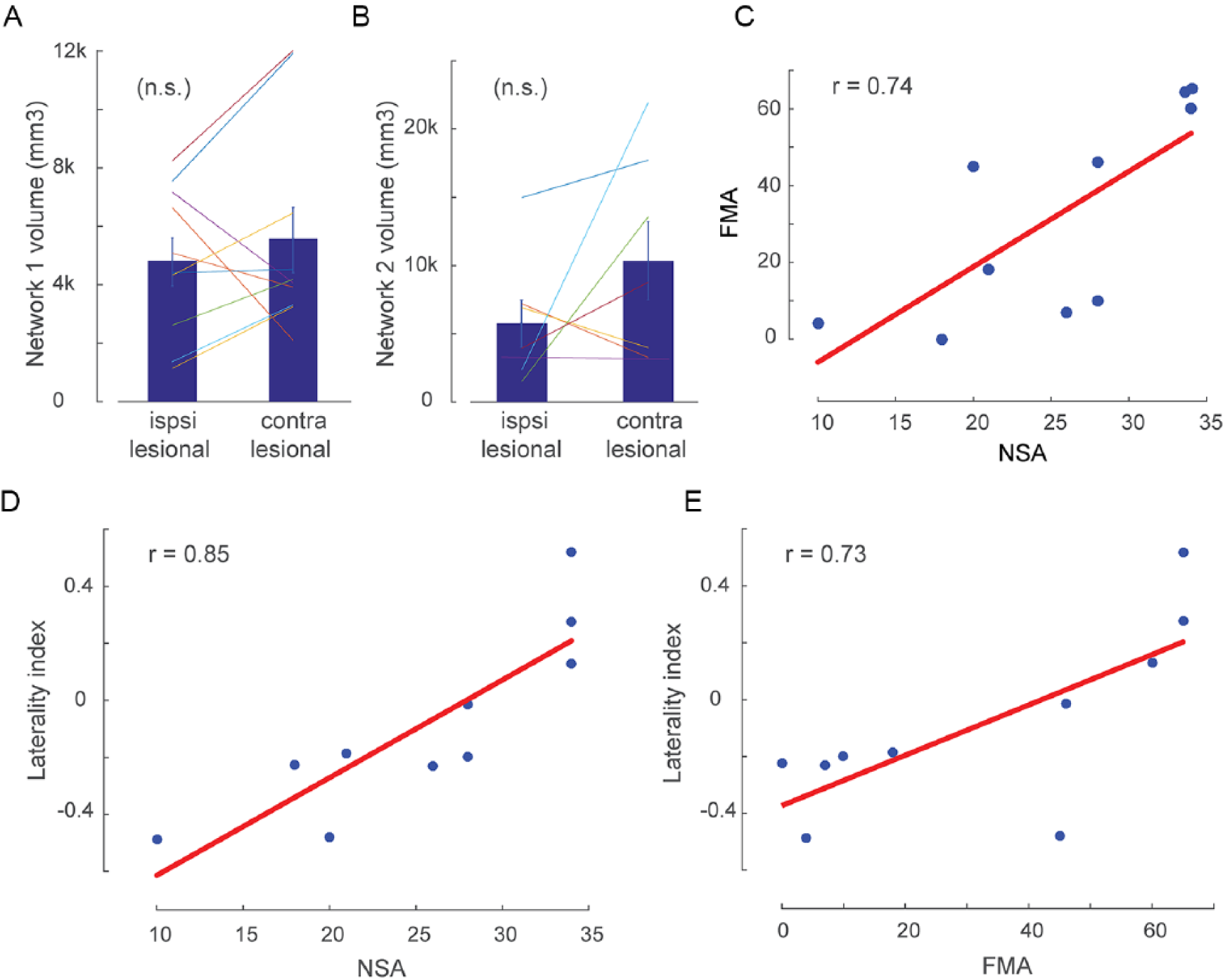
Ipsilesional and contralesional network volumes and their relationship to patients’ clinical scores. a and b, respectively, show the network 1 (C1) and network 2 (C2) volume of activation (shown in Figure 4 as red and blue components) in each hemisphere. Error bar shows standard error of the mean. c: significant linear relationship between the somatosensory and motor residual functions as measured by Nottingham sensory assessment (NSA) and Fugl-Meyer upper extremity assessment (FMA) respectively (p < 0.05). d and e show the linear relationship between the C1 network laterality index and the NSA and FMA scores, respectively. Patients with larger volume of activation in the ipsilesional compared to the contralesional hemisphere showed greater NSA (r = 0.85, p = 0.001) as well as FMA scores (r = 0.73, p = 0.01).
To assess interhemispheric differences in network extent, we calculated asymmetry indices for each network and patient (NetVol asymmetry index; see Methods). A positive or negative value of NetVol asymmetry index represents a larger training-related network in the ipsilesional or contralesional hemisphere, respectively. We then evaluated the relationship between the NetVol asymmetry index and motor and sensory residual functions on a per subject basis (note that at baseline, there is a significant correlation between FMA and NSA scores; Figure 5C; Pearson correlation
Discussion
It was the objective of this study to test the hypothesis that—similar to normal individuals—a single session of robot-controlled proprioceptive training with reinforcement and feedback would improve perceptual and motor performance in patients with chronic stroke and that FC changes could be measured in specific sensorimotor networks.
A close relationship between somatosensory and motor function31,32 as well as between proprioceptive function and motor learning 11 has previously been demonstrated in chronic stroke patients. In our sample, the degree of motor impairment (FMA) was also significantly related to the degree of somatosensory impairment (NSA). Proprioceptive training with feedback did improve proprioceptive function in our study population after 1 training session with 200 task repetitions. As observed in normal individuals, 5 chronic stroke patients’ reaching movements also improved with respect to movement curvature, indicating that mechanisms linking proprioceptive to motor function are still intact in patients with mild to moderate motor impairment and can potentially be used to improve motor function.
Using rs-fMRI, this study also aimed to identify the neuronal correlates associated with these changes. rs-fMRI has several advantages over task-based or -evoked fMRI when studying patients after stroke 33 : First, rs-fMRI is not subject to changes in task parameters during acquisition (eg, performance speed, power, movement trajectory) and, thus, allows evaluation of learning-related functional networks under similar experimental conditions before and after training. Second, rs-fMRI allows the study of more persistent effects of training beyond the periods of task performance. Finally, even patients with severe disabilities can undergo resting-state scans without causing excessive motion artifacts that potentially degrade image quality.
Training-associated FC changes related to residual motor function were detected in a network comprising bilateral SMG and S1. An increase in connection strength (meaning an increase in synchronicity of the BOLD signal fluctuations) within this network resulting from the training was positively correlated with upper-limb motor function. Several studies in normal healthy individuals have described an involvement of the SMG in motor learning,34,35 and it has been suggested that the SMG plays a key role in the suppression and correction of planned movements in heathy controls by suppressing movement plans from the dorsolateral prefrontal cortex if the plans do not match visual feedback. 36 In our proprioceptive training task, patients were deprived of visual input; thus, only proprioceptive information would be available to modulate FC in the SMG-S1 network. This proprioceptive feedback processing capability within a bilateral sensory network seems to be a crucial precondition for preserved motor function after stroke. Anatomical studies have shown that this network includes the SMG as well as the secondary somatosensory cortex, ventral premotor cortex, and ventrolateral prefrontal cortex.37,38
Two distinct networks were associated with sensory function. In the first network, comprising contralesional S1 (seed), contralesional M1, and PMC as well as bilateral SMA, a training-associated increase in FC was positively correlated with residual sensory function. Coherent FC increases in this network have been described in normal individuals using a nearly identical somatosensory training task (see Ostry and Gribble 39 for a review) and were found to be associated with motor learning. 7 The second network, in contrast, showed a negative association between residual sensory function and FC changes. The better the sensory function was preserved after stroke, the greater the negative correlation was in this suprainfratentorial network comprising ipsilesional cerebellum and bilateral S1 and SMG (Figures 3D to 3F). Negative FC indicates that resting-state BOLD fluctuations in this network were synchronously correlated prior to the training and became anticorrelated afterward (Figures 3E). Although there seems to be emerging consensus that anticorrelations in RSNs do in fact represent a neurophysiological signal40,41 rather than an artifact introduced by global signal regression, 42 the interpretation of these anticorrelations remains open. The seed region for this network was placed in the lateral parts of lobule VI in the ipsilesional cerebellar hemisphere. This region receives afferent input via contralesional corticopontine tracts and the pontocerebellar tract mainly from M1 of the upper limb 43 and, to a lesser extent, also from S1 44 and the contralesional spinal cord. Efferent inhibitory output projects via the ipsilesional dentate nucleus to the contralesional lateral ventral nucleus of the thalamus and from there to the cortex. 45 Cortical target areas comprise M1, PMC, the intraparietal lobule, and prefrontal cortex. 46 Similar to normal individuals, FC of this region is negative and anticorrelated with the supratentorial network nodes.6,7 This is consistent with the overall inhibitory influence of the cerebellar cortex on the cortical sensorimotor areas. Activation of lobule VI has been described in evoked fMRI studies of adaptive visuomotor learning 47 and may indicate an adaptation process similar to proprioceptive learning. Interestingly, lesions to the anterior lobe and adjacent lobule VI lead, among others, to dysmetria of the extremities,48-50 a symptom that is very similar to the deviation from the straight line in our reaching task.
Although many aspects of sensorimotor learning and the involved networks share similarities between chronic stroke patients and normal individuals, there are also differences such as the absence of networks originating from seed volumes in typical motor regions (such as M1, SMA, or ventral PMC). These differences may be explained by a lesion effect not present in controls.
To investigate this further, a non–hypothesis-driven ICA was performed. The aim of this analysis was to identify network components with increased training-associated connectivity in each individual patient. This resulted in 2 independent network components (C1 and C2), C1 being present in all patients independent of residual motor function and C2 only in patients with FMA scores greater 7 (Figure 4). Whereas C1 consisted mainly of subcortical regions (basal ganglia, thalamus), C2 comprised more cortical regions such as M1 and SMA. This demonstrates that our proprioceptive training induces connectivity changes in all patients, even those with severe motor deficits, and that more widespread connectivity changes, specifically in the cortex, appear to be associated with better residual motor function. Although the absolute volume of all network components across all patients was not different between both hemispheres, the volume asymmetry index, however, seems to suggest that better residual motor and sensory function is associated with relatively larger networks in the affected hemisphere, showing training-associated connectivity changes (Figure 5D-E) in the C1 network. These findings would be consistent with results from previous rs-fMRI studies in chronic stroke investigating spontaneous recovery, where a normalization of connectivity patterns in the affected hemisphere18,51,52 as well as an increased connectivity in cortical sensory motor networks was associated with recovery. 19
Some studies have reported that short periods of somatosensory training using passive movements are unable to induce plasticity in the human motor system.53,54 In contrast, others have shown that short periods of passive training can change the cortical motor representation25,55 but they did not report any behavioral improvements as a result of sensory training. Moreover, it has been shown that passive training for longer periods (~4 weeks) is able to induce plasticity in the motor system in healthy individuals 56 and stroke patients.13,57 However, in these studies, the passive training protocol did not include any cognitive engagement, and neither plasticity in the motor areas,53,54 nor detectable functional improvement in motor behavior25,55 was observed. Consistent with these observations, we previously found that passive movements alone were less able to change the characteristics of subsequent motor behavior, whereas passive movements together with decision making and feedback enhanced the following motor behavior and facilitated motor learning.5,7
This proof-of-concept study has some limitations that will have to be addressed in a larger trial before this training can eventually be developed into a possible future treatment for motor deficits after stroke. First, we performed this study in a limited number of individuals without a control group because we found it mandatory to demonstrate similar changes in FC and comparable behavioral effects after the robot-controlled training in stroke patients as we have observed in controls. Data from this study—even though it is in only 10 individuals—indeed suggest that this may be possible, under the assumption that chronic stroke patients would not show spontaneous and consistent changes in FC specific to the sensorimotor network (and not in other networks such as visual or default mode network (DMN)), which are related to residual function. In addition, very similar changes were detected in each individual, again arguing against the interpretation of these findings as unspecific random fluctuations. Second, we have not measured electromyographic activity during the task, so it cannot entirely be ruled out that some of the connectivity changes may have been attributable to muscle coactivation. However, it is known that in healthy individuals, muscle coactivation decreases over the course of training with a robot. Thus, it is reasonable to assume that on the first experimental day, patients experienced similar or even higher levels of muscle coactivation compared with the second experimental day. If improvements in movement were a result of muscle coactivation, it would have to increase rather than decrease. We have also measured the force applied by the patient to the robot handle, which was on average approximately 4N. A force of this magnitude would be expected simply because of the passive stiffness of the arm and as a result of the patient’s arm being cantilevered off the robot handle by the arm cast. This is consistent with the idea that an eventual effect of active force production in the passive movement procedure is very limited. Finally, we included patients with a large range in deficits. Despite the variable lesion size and deficits, we were able to detect connectivity changes in each individual, even those with very poor motor and/or sensory function. Given the variability of lesion size, we only investigated seed regions in the unaffected hemisphere that were present in all individuals. Future trials may specifically recruit patients with only subcortical lesions, so that seed regions in both hemispheres can be assessed.
In summary this proof-of concept study demonstrates that even a single session of robot-controlled proprioceptive training can induce connectivity changes in respective networks that are associated with residual motor and sensory function and translates into improved motor function in most individuals. Further studies are now required to test whether this approach can be developed into a rehabilitation strategy investigating important questions such as the effect of repeated training sessions as well as duration and possible generalization of treatment effects.
Supplemental Material
Supplemental_Material – Supplemental material for A Single Session of Robot-Controlled Proprioceptive Training Modulates Functional Connectivity of Sensory Motor Networks and Improves Reaching Accuracy in Chronic Stroke
Supplemental material, Supplemental_Material for A Single Session of Robot-Controlled Proprioceptive Training Modulates Functional Connectivity of Sensory Motor Networks and Improves Reaching Accuracy in Chronic Stroke by Shahabeddin Vahdat, Mohammed Darainy, Alexander Thiel and David J. Ostry in Neurorehabilitation and Neural Repair
Footnotes
Supplementary material for this article is available on the
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by a grant from the Heart and Stroke Foundation of Canada (G16-0014661) to AT and DJO and a grant from the National Institute of Child Health and Development (NICHD R01 HD075740) to DJO.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
