Abstract
Background. Stroke often involves primary motor cortex (M1) and its corticospinal (CST) projections. As hand function is critically dependent on these structures, its recovery is often incomplete. Objective. To determine whether impaired hand function in patients with chronic ischemic stroke involving M1 or CST benefits from the enhancing effect of Hebbian-type stimulation (pairing M1 afferent stimulation and M1 activity in a specific temporal relationship) on M1 plasticity and hand function. Methods. In a double-blind, randomized, sham-controlled design, 20 patients with chronic ischemic stroke affecting M1 or CST were randomly assigned to 5 days of hand motor training that was combined with either Hebbian-type (trainingHebb) or sham stimulation (trainingsham) of the lesioned M1. Measures of hand function and task-based M1 functional magnetic resonance imaging (fMRI) activity were collected prior to, immediately following, and 4 weeks after the intervention. Results. Both interventions were effective in improving affected hand function at the completion of training, but only participants in the trainingHebb group maintained functional gains. Changes in hand function and fMRI activity were positively correlated in both ipsilesional and contralesional M1. Compared with trainingsham, participants in the trainingHebb group showed a stronger relationship between improved hand function and changes in M1 functional activity. Conclusions. Only when motor training was combined with Hebbian-type stimulation were functional gains maintained over time and correlated with measures of M1 functional plasticity. As hand dexterity is critically dependent on M1 function, these results suggest that functional reorganization in M1 is facilitated by Hebbian-type stimulation. ClinicalTrials.gov Identifier: NCT01569607.
Keywords
Introduction
Despite rehabilitation treatment, compromised hand function often persists and is one of the most common long-term deficits after stroke. 1 Hand function depends critically on the primary motor cortex (M1) and its corticospinal tract (CST) projections.2,3 Lesions to these structures result in reduced M1 output 4 and detrimental effects on the function of the hand contralateral to the lesion.4-6 In nonhuman primate stroke models, M1 reorganization of viable neuronal tissue surrounding the lesion (perilesional tissue of ipsilesional M1) is critical in the recovery of hand motor function,7,8 and recovery of independent finger movements is not observed after lesion of the CST. 9 Targeting these structures for restorative treatment of hand function in stroke patients is therefore neuroanatomically and physiologically justified.
In healthy humans, motor training induces functional M1 reorganization and improvement of motor function, likely through mechanisms that are similar to memory formation and include modification of synaptic efficacy, such as long-term potentiation (LTP).
10
One technique that has been used to induce LTP in M1 slice preparations is Hebbian stimulation, where stimulation of cortical afferents is paired with depolarization or stimulation-induced firing of the targeted postsynaptic pyramidal tract neuron (PTN).11,12 In healthy adults13-15 and patients after stroke,
16
the importance of the temporal relationship between afferent stimulation and discharge of PTNs has been demonstrated using peripheral nerve stimulation and/or low frequency repetitive transcranial magnetic stimulation (rTMS) of M1. Subthreshold rTMS is used to stimulate the intracortical connections targeting PTNs below the level of discharge.
17
We have previously demonstrated in an NMDA (N-methyl-
Methods
Participants
Twenty-two individuals (11 men, age 61.6 ± 11.0 years) in the chronic phase of ischemic stroke were randomized to the treatment and sham conditions (Table 1, Figure 1). All participants gave informed consent and experimental procedures were approved by the Emory University and Georgia Tech/Georgia State University Institutional Review Boards. Two participants (1 male and 1 female) were subsequently excluded because they failed to follow training instructions (n = 1) or did not complete postintervention measurements (n = 1). Of the remaining 20 participants, fMRI data were not available from 2 participants due to claustrophobia (n = 1) or inability to perform the scanner task (n = 1). All participants met the inclusion criteria of (1) single ischemic infarction affecting M1 and/or CST more than 6 months prior to study enrollment, as determined by MRI review; (2) motor deficit in the hand contralateral to the infarct and ability to perform the training task; (3) no other neurological disorder or aphasia-related inability to understand and follow the instructions of the research protocol or communicate effectively to give consent; (4) no contradiction to TMS or MRI 22 ; (5) no intake of medication that interfered with TMS measures (eg, benzodiazepines); (6) a measurable motor-evoked potential (MEP) of >50 μV amplitude in the extensor carpi ulnaris (ECU) muscle with TMS of ipsilesional M1 as measured during the screening visit; (7) absence of dementia as determined by a score of >70 on the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) 23 ; and (8) the ability to give informed consent. Comorbidity was determined from medical records and interview by a board-certified neurologist. Upper extremity muscle strength and tone were determined using the Medical Research Council Scale (MRC) 24 and the modified Ashworth Scale. 25 Subjects were screened for depression with the Hamilton Depression Scale. 26 The Edinburgh Handedness Inventory was used to determine handedness. 27
Characteristics of Stroke Patients.
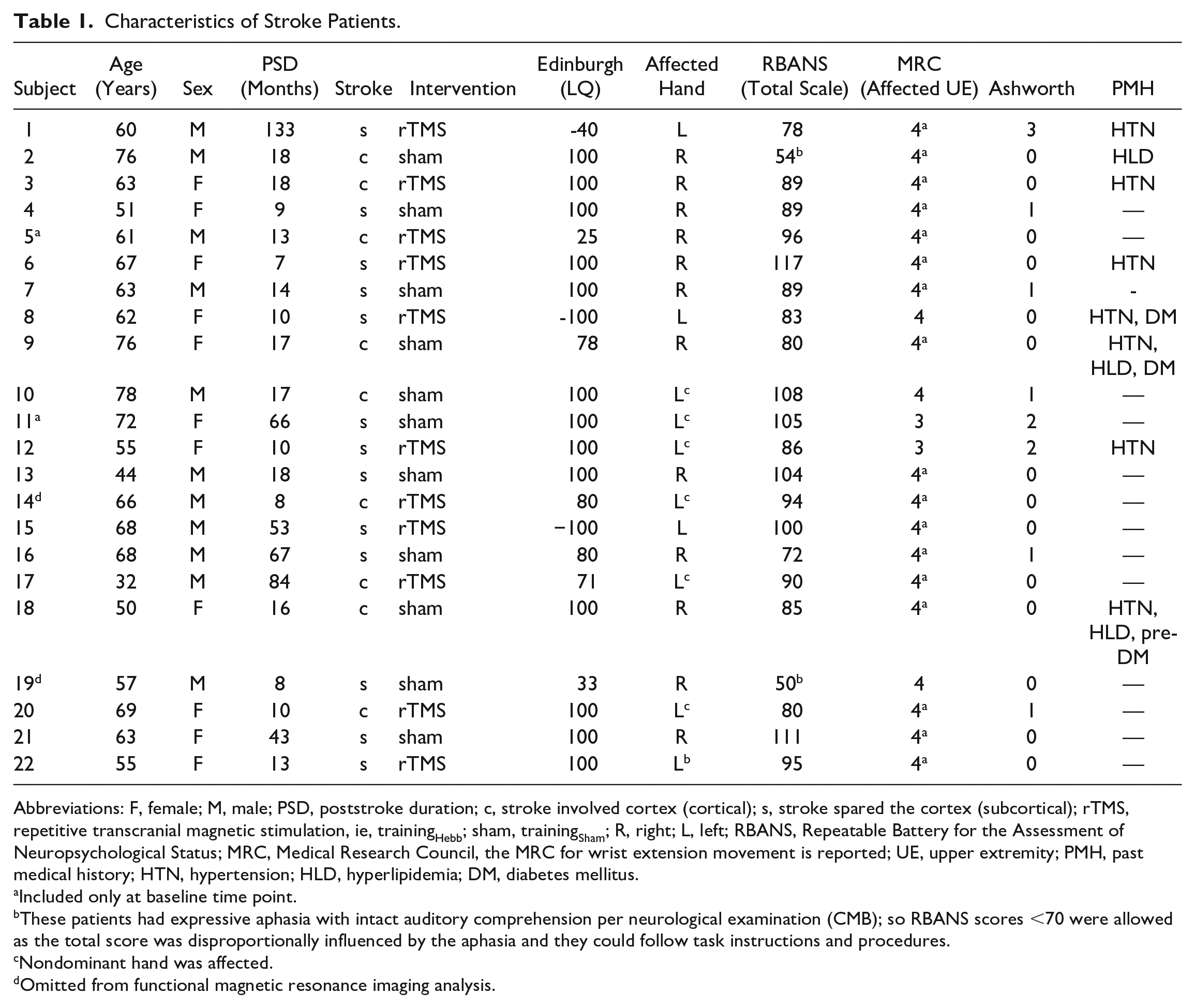
Abbreviations: F, female; M, male; PSD, poststroke duration; c, stroke involved cortex (cortical); s, stroke spared the cortex (subcortical); rTMS, repetitive transcranial magnetic stimulation, ie, trainingHebb; sham, trainingSham; R, right; L, left; RBANS, Repeatable Battery for the Assessment of Neuropsychological Status; MRC, Medical Research Council, the MRC for wrist extension movement is reported; UE, upper extremity; PMH, past medical history; HTN, hypertension; HLD, hyperlipidemia; DM, diabetes mellitus.
Included only at baseline time point.
These patients had expressive aphasia with intact auditory comprehension per neurological examination (CMB); so RBANS scores <70 were allowed as the total score was disproportionally influenced by the aphasia and they could follow task instructions and procedures.
Nondominant hand was affected.
Omitted from functional magnetic resonance imaging analysis.
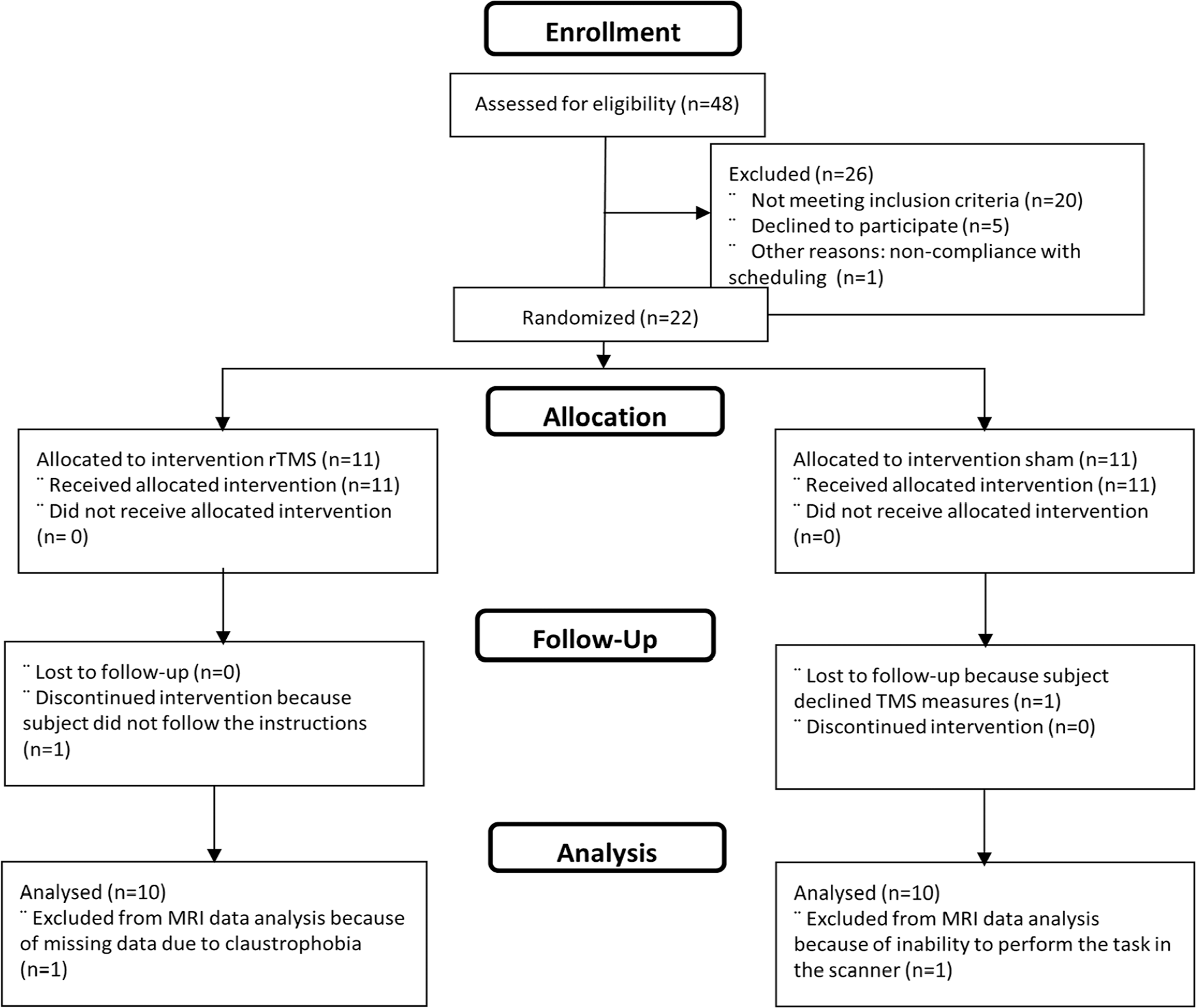
CONSORT 2010 flow diagram.
Study Design
The effect of Hebbian-type ipsilesional M1 stimulation on motor training-related improvement in affected hand function and M1 plasticity was determined in a double-blind, randomized, sham-controlled study of patients with chronic ischemic stroke. M1 plasticity was defined as increases in the blood oxygen level–dependent (BOLD) response to hand movements. Motor training-related improvement of hand function was defined as improved performance on the Jebsen Taylor Test (JTT), 28 a standardized test of hand function.
At an initial screening visit, all procedures were explained to the participant and informed consent was obtained. Participants were screened for the inclusion criteria listed above and received a neurological examination. Following screening, participants were allocated in a randomized predetermined order to receive hand motor training that was combined either with Hebbian-type rTMS (trainingHebb; n = 10) or sham stimulation (trainingSham; n = 10) over ipsilesional M1 during motor training for 5 consecutive days. fMRI measures of functional M1 plasticity and motor function were obtained at 3 time points: 2 to 4 days prior to the intervention (baseline), 2 to 4 days after the intervention (posttest), and 4 weeks after the end of the intervention (follow-up) (Figure 2A). All participants and all investigators involved with the data analysis were blinded to the treatment conditions.
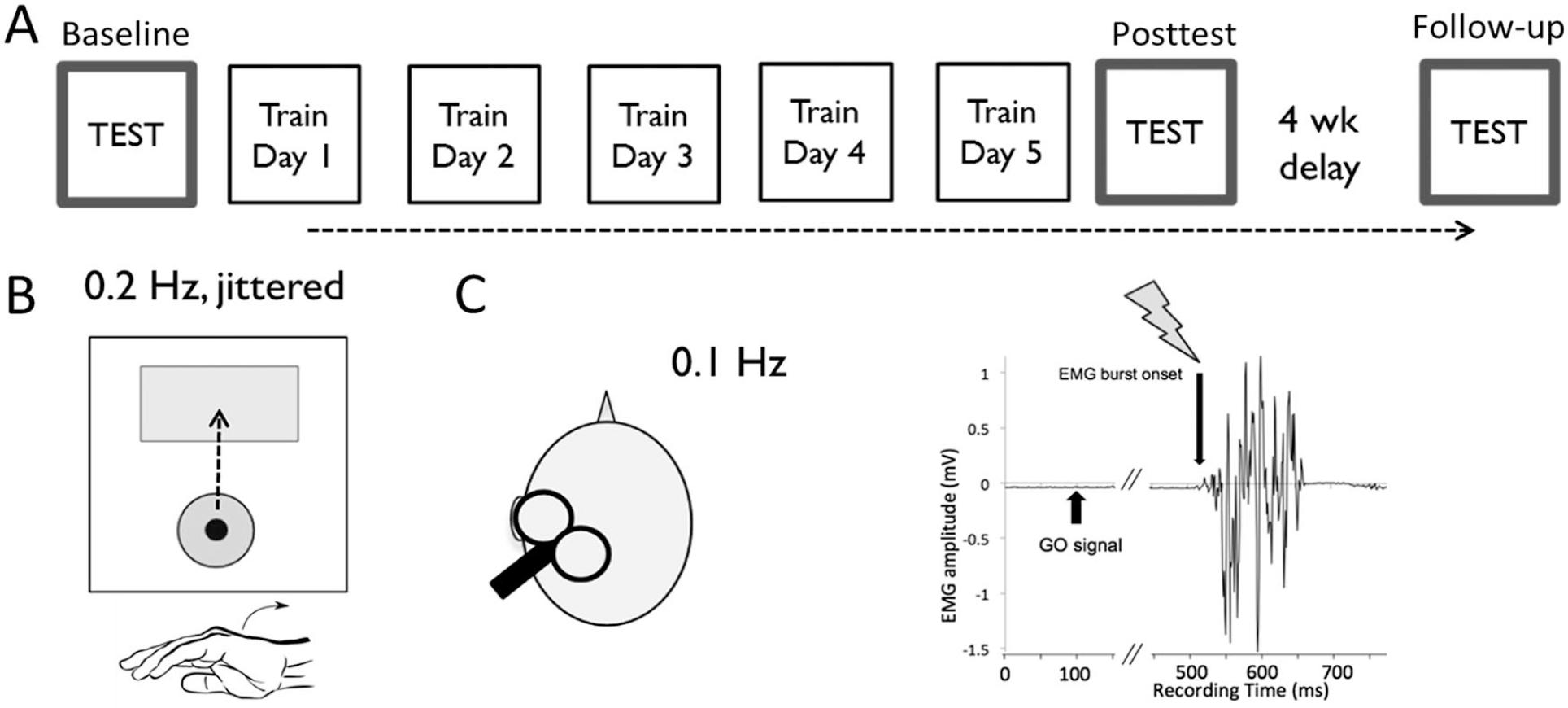
Study design. (A) Study timeline. (B) Training task. A 2-dimensional accelerometer was mounted on the dorsum of the affected hand to measure acceleration in the extension/flexion and abduction/adduction movement planes; the forearm was restrained to ensure that the cursor was only driven by hand movements. Acceleration in the abduction/adduction plane moved the cursor to the right/left along the horizontal axis. The landing position of the cursor was determined by the peak acceleration along the extension and abduction/adduction axes. An encouraging auditory tone sounded if the cursor landed in the target box and the movement was scored as a hit. (C) Repetitive transcranial magnetic stimulation (rTMS) protocol. rTMS and sham stimulations were triggered by movement related increases in electromyographic (EMG) activity of the muscle supporting the training movement (training agonist, extensor carpi ulnaris [ECU]). rTMS or sham stimulation was delivered at the onset of the EMG burst of the ECU muscle (arrow). The onset of movement related EMG activity is clearly identified by the software program using a preset threshold of about 10% to 20% maximum muscle contraction EMG amplitude (horizontal bar in grey). TMS application was triggered when the amplitude of the EMG activity of the ECU muscle exceeded the preset threshold. 21
Measurement of Hand Motor Function
For the purpose of assessing upper extremity and hand motor function at baseline, 3 categories of motor function measurements were evaluated at each time point: motor impairment (altered motor kinematics), motor function, and overall function in activities of daily living. Details of these measurements were summarized in a previous article. 4 Hand function as measured by the JTT 28 was used as the primary outcome measure. Patients completed 7 motor tasks as quickly as possible (capped at 120 seconds). The raw score was calculated by summing the time to complete all but 2 subtests (writing and simulated feeding), which were omitted due to low test-retest reliability. 29 The raw score was normalized to age- and sex-matched standard scores that accounted for hand dominance.28,30 A normalized score greater than 0 indicated abnormal hand function, with higher values indicating more severe impairment. As a secondary outcome measure, the overall function of the affected hand in 30 activities of daily living was measured at each time point with the “How Well” subtest of the Motor Activities of Daily Living (MAL) 31 by averaging the scores for each activity of daily living. All measures were assessed by a trained physical therapist.
Brain Imaging
All imaging data were collected on a Siemens 3T Trio scanner at the Georgia Tech/Georgia State University Center for Advanced Brain Imaging using a 12-channel receive head coil. T1- and T2-weighted structural scans were acquired for each participant at each time point (T1: repetition time [TR] = 2250 ms, echo time [TE] = 4.18 ms, fractional anisotropy (FA) = 9°, integrated parallel acquisition techniques [iPAT] = 2, field of view [FOV] = 256 × 256 mm, matrix size = 256 × 256, 176 one-mm-thick sagittal slices, voxel size = 1 mm3, acquisition time 6:17; T2-weighted: TR = 3200 ms, TE = 402 ms, iPAT = 2, FOV = 256 × 256 mm, matrix size 256 × 256, 176 one-mm-thick sagittal slices, voxel size = 1 mm3, acquisition time 4:43). Participants also completed two 176-volume runs of cued wrist extension movements with the affected hand (echo-planar imaging [EPI]: TR = 1920 ms, TE = 30 ms, FA = 90°, iPAT = 2, FOV = 204 × 204 mm, matrix size = 68 × 68, 36 3-mm-thick axial slices, interslice gap 20%, voxel size = 3 mm3, acquisition time 5:45 per run). Correct unilateral movements, including contraction of the corresponding extensor carpi ulnaris (ECU) muscle, were confirmed outside the scanner during initial task familiarization. Each wrist extension movement block consisted of 6 movement cues of 2 seconds each. The intertrial interval between cues was variable (100, 600, or 1100 ms) so that participants could not adopt a strategy of rhythmic wrist extension and flexion. Each run contained eleven 15-second blocks of wrist extension alternating with 15-second blocks of rest. Execution of strictly unilateral wrist extension movements inside the scanner were visually confirmed for all participants by the experimenters.
Intervention
Motor training: Participants completed 5 consecutive days of 30 minutes of hand motor training (Figure 2A). Each day, patients performed 360 auditory-cued ballistic wrist extension movements with their affected hand at a rate of 0.2 Hz with a 1000-ms jitter (Figure 2B). This type of training has been shown to effectively induce M1 plasticity and to improve the kinematics of hand movements in healthy participants10,13,21 and patients with stroke.16,32 Furthermore, wrist extension is essential for wrist stabilization, which is a prerequisite for hand and finger function, and is weak and recovers poorly in patients and nonhuman primates after stroke involving M1 and CST. 9 These clinical considerations are supported by evidence from nonhuman primate studies of M1 where digit and wrist representations are localized in proximity to one another,33,34 have monosynaptic CST projections 9 and reorganize in response to skilled hand training after lesion of the M1 hand area. 8
During motor training, patients were seated in front of a computer screen with the arm, wrist and hand supported. Participants were instructed to move a cursor from its home position to a target located along the same vertical axis by extending the wrist as quickly as possible (Figure 2B). A 2-dimensional accelerometer mounted on the dorsum of the hand controlled cursor position. To reduce fatigue, training was broken into 3 blocks of 120 movements with 1 to 3 minutes of rest between blocks. To promote motor learning, target distances were adjusted according to performance, 8 with target distance increased once performance exceeded 75% hits in 2 consecutive training blocks.
rTMS was delivered through an air-cooled figure-of-eight coil (7-cm wing diameter) connected to the Magstim Super Rapid (Magstim Company, Whitland, UK). The rTMS coil was positioned with the handle toward the back of the head and at a 45° angle to the midline. 21 The optimal site (hot spot) for the contralateral ECU muscle, a muscle that supports the training movement, was determined on day 1 prior to rTMS or sham application. The motor threshold (MT) was defined as the minimum stimulus intensity to evoke an MEP of >50 µV for at least 5 of 10 trials and was determined to the nearest 1% of the maximum stimulator output (MSO). 35 To ensure that the same site was stimulated within and across sessions, the hot spot identified at the initial time point (day 1) was marked on the T1 image of the participant’s brain using a neuronavigation system (BrainSight, Rogue Research, Montreal, Canada). For participants in the rTMS treatment group, rTMS was administered over this hotspot according to a previously established effective rTMS protocol at a frequency of about 0.1 Hz and at a subthreshold intensity of 80% MT13,21 (Figure 2C). The mean stimulation strength across all participants in the rTMS treatment group was 62.9% ± 12.62% MSO. The TMS pulse was applied at the onset of movement-related increases in the ECU EMG activity, a timing that was most effective in enhancing motor learning in healthy participants13,21 and is also consistent with the temporal relationship between M1 pyramidal tract neurons (PTN) discharges and movement in nonhuman primates. 36 Sham stimulation was applied through an air-cooled sham coil, which mimics the experience of subthreshold stimulation with the real coil.
Data Analysis
Analysis of Hand Motor Function Data
The scores from the JTT and MAL were separately modeled as the outcome of 2 factors, intervention type (trainingHebb or trainingSham) and time point (baseline, posttest, and follow-up), in a generalized linear model (GLM) with an unconstrained correlation structure and pooled variance. The test quantified the effects of intervention, time point, and their interaction. We performed the test both with and without covariates and performed a likelihood ratio test to control for potential confounders (age, sex, stroke location, and RBANS). In no case did including covariates improve the fit of the model, so we only report results from the models without covariates. Planned tests for post hoc differences between time points (posttest vs baseline, follow-up vs baseline, and follow-up vs posttest) were measured via the appropriate contrast matrix using the Wald statistic. Due to hypothesized differences between treatment types, post hoc tests were also conducted for each treatment group separately.
Analysis of Brain Imaging Data
An unbiased structural volume was created for each participant by combining the T1 images from the baseline, posttest, and follow-up time points using the Freesurfer longitudinal pipeline. 37 The resulting longitudinal structural image was registered to the TT_N27 structural template in AFNI. 38 Transformation between an individual’s structural image and a template in atlas space can be challenging when patient or aged populations are used since atrophy or significant damage may be present, and standard affine or nonlinear normalization techniques may result in poor template matches or overnormalization. While there are several approaches for normalizing lesioned brains, we chose enantiomorphic normalization 39 to reduce the effects of varying lesion size that may occur when normalization is restricted in certain brain areas by lesion masks.
Functional data preprocessing and statistics were performed using AFNI. 38 Preprocessing steps included slice time correction, head motion correction, 12-parameter affine alignment between the time point’s functional images and T1 and the longitudinal T1, smoothing with a full-width at half-maximum 6.0-mm kernel, and conversion to percent signal change. Following preprocessing, GLM analysis was performed using AFNI’s 3dDeconvolve tool. In addition to the main task regressor, 6 head movement vectors were included as regressors of no interest. Movement censoring excluded volumes with more than 0.5-mm total head movement. This resulted in a loss of 4% of the data on average.
A region of interest (ROI) approach was used to analyze the relationship between hand function and functional activation during cued wrist extension of the affected hand. Prior to ROI analysis, we examined individual activation maps for each participant to verify that significantly more activity was observed during cued wrist extension than at rest. Data were categorized based on whether they were from the lesioned hemisphere (ipsilesional M1) or the intact hemisphere (contralesional M1; Figure 3A). These terms should not be confused with ipsilateral and contralateral as used in studies of motor function in healthy participants; here, the ipsilesional hemisphere is contralateral to the performing hand and the contralesional hemisphere is ipsilateral to the performing hand. Robust task activation was seen in the ipsilesional precentral gyrus in all participants, extending into postcentral gyrus and pre- and supplementary motor areas (Figure 3B). Additionally, activation was seen at the group level in contralesional M1 anterior to the precentral gyrus, in postcentral gyrus, and in parietal areas. Anatomical M1 ROIs were created using the maximum probability masks from the Juelich atlas of motor areas 40 by combining the 4a and 4p masks. The ROI from ipsilesional M1 was masked with the participant’s lesion mask so that lesioned tissue was not included in the ROI. The effects of intervention and time point on ROI activation values were modeled with GLMs as described above.
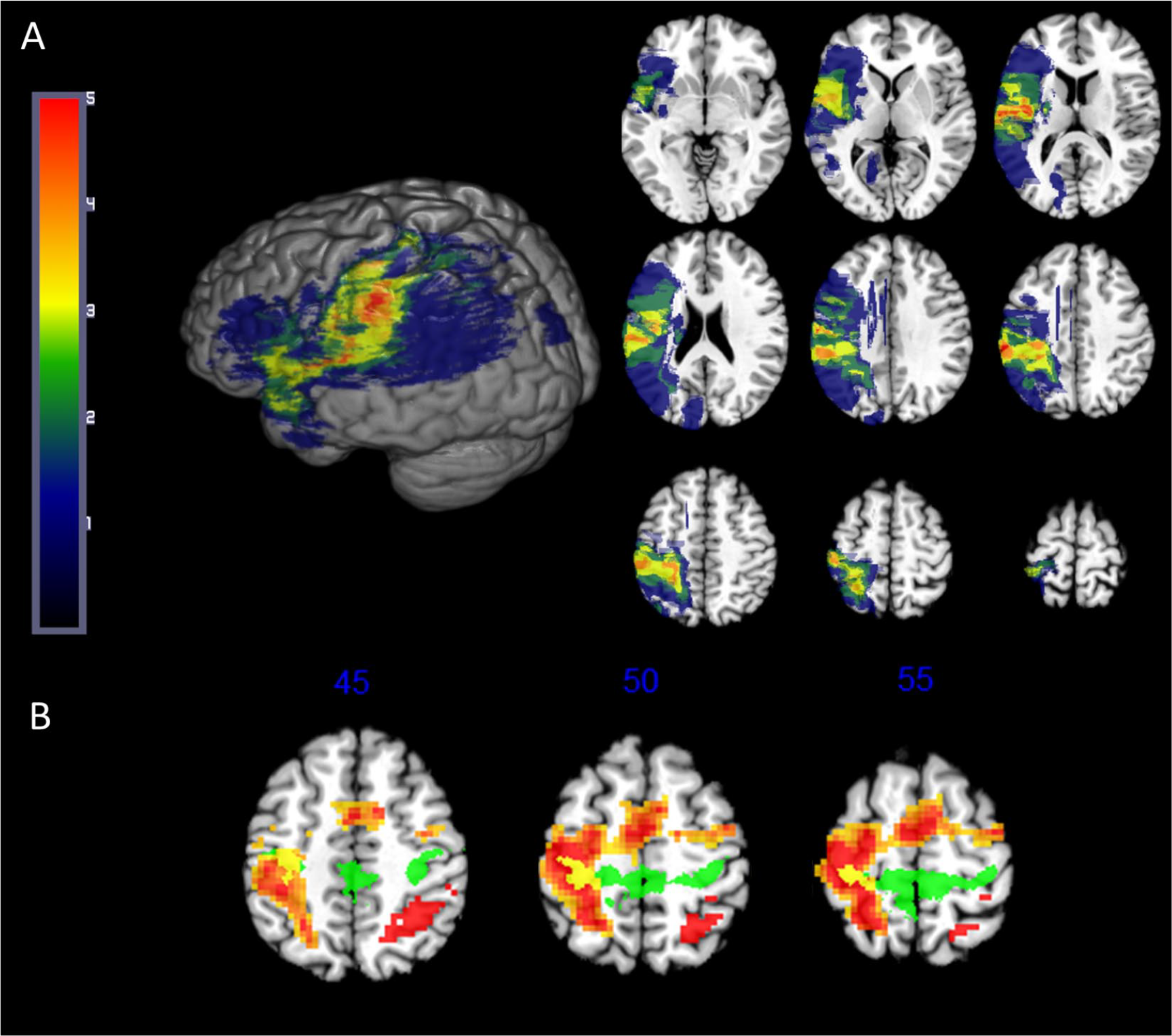
(A) Group lesion overlap map; lesioned brains were flipped when necessary so that all participants’ lesions appear in the same hemisphere. (B) Group average activation for wrist movement > rest (cluster size corrected P = .001) for all subjects. Warmer colors indicate higher t statistic values. The M1 region of interest (ROI) mask (green, overlap in yellow) is from the Juelich maximum probability atlas. 42
Results
Baseline Performance
Prior to the intervention, the trainingHebb and trainingSham groups did not differ on performance on the JTT or MAL, amount of baseline brain activity, or demographic details (Supplemental Table 1). At baseline, there was a significant correlation between JTT score and contralesional M1 activity during wrist extension, with poorer hand function being associated with greater activation in contralesional M1 (R2 = 0.25, P < .05, Figure 5D). The relationship between hand function and ipsilesional M1 activity was in the same direction but the correlation was not significant (R2 = 0.10, P > .1, Figure 5A.)
Changes in Hand Function Following Motor Training
Participants’ performance on the JTT improved over time. Mixed model analysis with JTT score as the dependent variable and time point (baseline, posttest, follow-up) and intervention type (trainingHebb/trainingSham) as independent variables showed a significant effect of time point, F(2, 54) = 8.12, P < .001 (Figure 4A). The effect of intervention type was not significant, F(1, 54) = 1.37, P > .1, and did not interact with time, F(2, 54) = 0.44, P > .1. Post hoc contrast testing indicates that participants’ affected hand function at posttest was significantly better than at baseline,
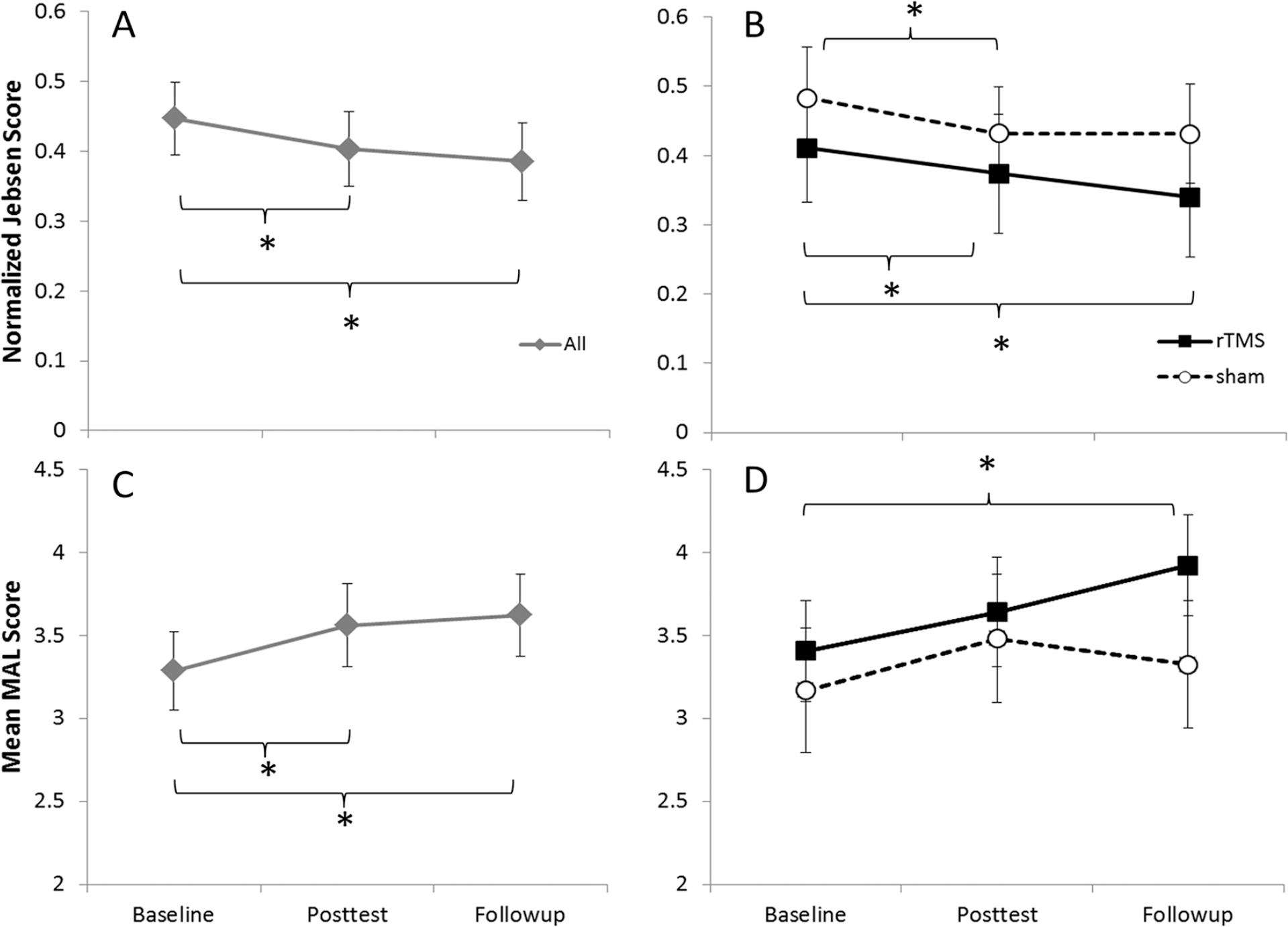
Measures of change in hand function and functional activation over time across all participants (panels A and C) and for the trainingHebb (rTMS) and trainingsham (sham) groups (panels B and D). (A and B): Jebsen Taylor Test (JTT) performance with the affected hand. (C and D) Mean “How Well” score on the Motor Activities of Daily Living (MAL) instrument. *Indicates a contrast significant at P < .05. Error bars represent standard error of the mean (SEM).
Participants’ MAL scores also improved over time, with the improvement conditioned by intervention type. Mixed model analysis with the MAL scores as the dependent variable and time point and intervention type as independent variables showed a significant effect of time, F(2, 54) = 5.23, P < .01 (Figure 4B) and a significant interaction between intervention and time, F(2, 54) = 3.67, P < .05. The main effect of intervention was not significant, F(1, 54) = 0.36, P > .1. Post hoc testing indicated that, at posttest, patients were better able to use the affected hand in ADLs relative to baseline,
Changes in M1 Activation Following Motor Training
Unlike measures of hand function, M1 activation was not significantly different across the 3 measurement time points or between intervention groups, as mixed model analysis with percent signal change as the dependent variable and time and intervention type as independent variables did not show significant effects of time, iM1 F(2, 54) = 0.48, P > 0.1; cM1 F(2, 54) = 0.29, P > .1; intervention, iM1 F(1, 54) = 2.98, P = .09; cM1 F(1, 54) = 1.82, P > .1; or an interaction between time and intervention, iM1 F(2, 54) = 1.25, P > .1; cM1 F(2, 54) = 2.21, P > .1.
Relationship Between Hand Function and M1 Activation Following Motor Training
There was no significant relationship between JTT score and task-related fMRI activity in either ipsilesional M1 (R2 = 0.01, P > .1, Figure 5B) or contralesional M1 (R2 = 0.01, P > .1, Figure 5E) at posttest. However, at follow-up, the relationship between hand function and M1 activity was reversed relative to baseline, with a significant positive correlation between Jebsen score and ipsilesional M1 activity (R2 = 0.268, P < .05, Figure 5C), such that participants with better hand function showed greater activity in ipsilesional M1 during the wrist extension task. This direction of correlation was present in contralesional M1, but did not reach significance (R2 = 0.14, P = .12, Figure 5F). These correlations are significant only when we consider all patients; no significant correlations between hand function and M1 activity emerge when only trainingHebb participants or trainingSham participants are considered.
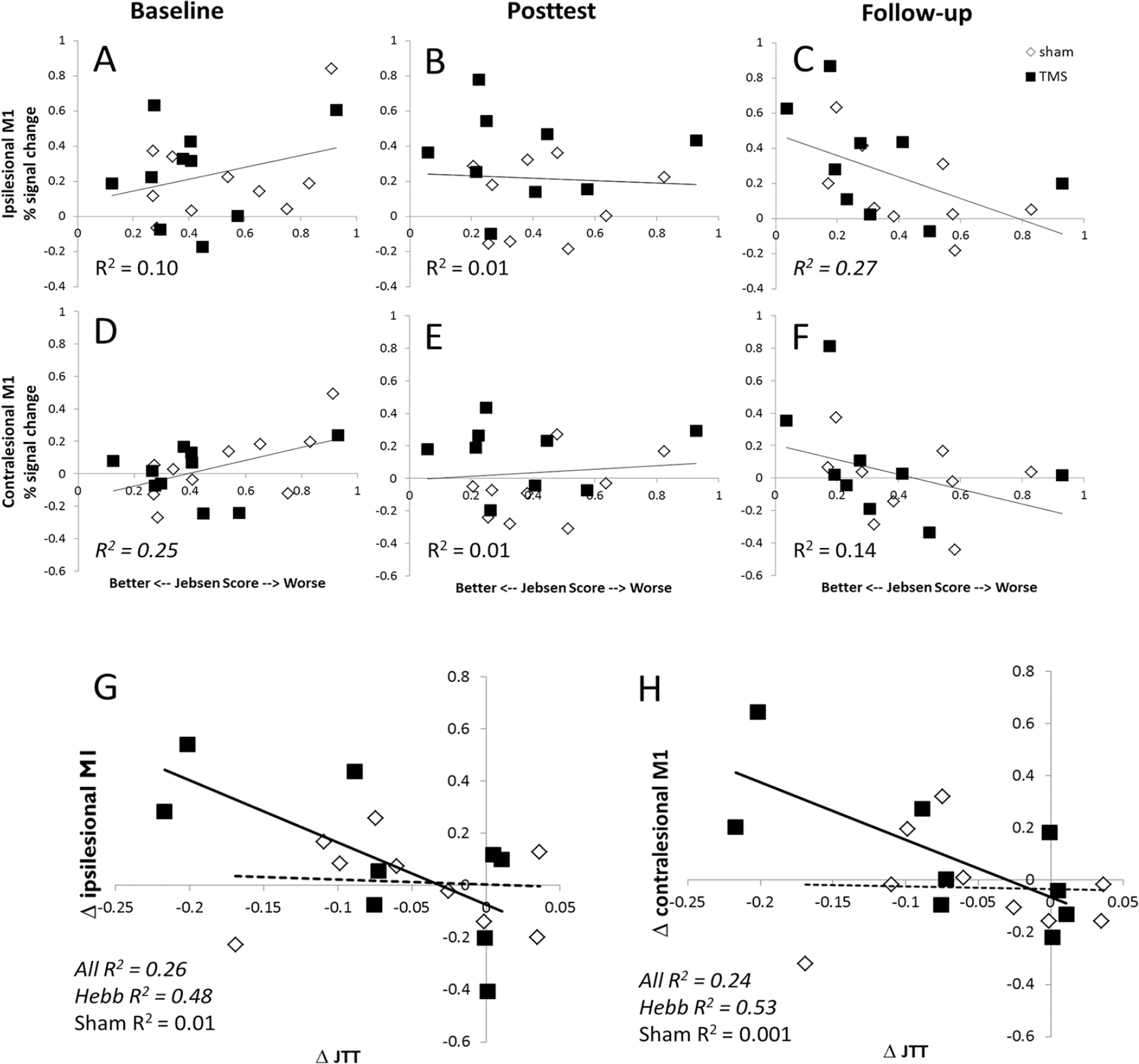
Relationships between hand function and primary motor cortex (M1) activity. Panels A-C (ipsilesional M1) and D-F (contralesional M1) show the relationship between an individual’s M1 activity and performance on the Jebsen Taylor Test (JTT) at the baseline, posttest, and follow-up time points, respectively. R2 values corresponding to significant correlations are italicized. The best-fitting line reflects data from all participants; no significant differences were found when the trainingHebb and trainingsham groups were considered separately. Panels G (ipsilesional M1) and H (contralesional M1) show the relationship between the change in hand function across the study (Follow-up JTT score – Baseline JTT score) and the change in M1 task activation across the study (Follow-up M1 activation – Baseline M1 activation). Bold lines correspond to the best-fitting line for the trainingHebb group, dotted lines for the trainingsham group. A significant correlation between hand function change and fMRI activation change is present for the group as a whole (best-fit line not shown) and the trainingHebb group but not the trainingsham group.
Relationship Between Training-Related Improvement of Hand Function and Change in M1 Activation
As indicated above, hand function and M1 activity are related both at baseline and at follow-up, but a change in the sign of the correlation between these 2 time points indicates a systematic effect of training on M1 activity (Figure 5). While M1 activity at baseline does not predict hand function at follow-up (iM1 R2 = 0.03, P > .1; cM1 R2 = 0.142, P > .1), the amount of change in M1 activity and in hand function are related. Significant correlations between the degree of improvement in hand function and the change in M1 activity between baseline and follow-up emerged in both hemispheres (iM1 R2 = 0.257, P < .05; cM1 R2 = 0.236, P < .05), with participants who showed a greater increase in hand function also showing a greater increase in task-related M1 activity (Figure 5G and H). This relationship is carried by the trainingHebb group, as there are significant correlations in both hemispheres for the trainingHebb group (iM1 R2 = 0.480, P < .05; cM1 R2 = 0.526, P < .05) but not the trainingSham group (iM1 R2 = 0.006; cM1 R2 = 0.001, both Ps > .1).
Discussion
In the present study, we tested the hypothesis that in patients with chronic ischemic stroke involving M1 and/or its CST projections, training combined with Hebbian-type stimulation of lesioned M1 enhances M1 reorganization and hand function when compared to training alone. We found that hand motor training, whether alone or combined with Hebbian-type stimulation, improved hand function significantly when tested immediately after training, consistent with previous results in healthy adults,10,13,21 patients with subacute 32 and chronic stroke, 41 and stroke models of nonhuman primates 34 and rodents. 42 While there was no difference between the 2 interventions immediately after completion of the training, gains in hand function were maintained or further improved over the 4 weeks following the intervention only in the trainingHebb group. The magnitude of improvement in hand function and change in task-dependent fMRI activity were positively correlated in both ipsilesional and contralesional M1 in the trainingHebb group. This correlation was not seen in the trainingSham group. While the follow-up time in the present study is only 1 month, the trajectory for paretic hand function in the trainingHebb group is distinctly different when compared with the trainingsham group, with a tendency toward further improvement in both the JTT and MAL. This is an important finding, considering the reported decline in function over time after initial gains from neurorehabilitation in mice 43 and stroke patients.44,45 The finding is also consistent with the report of a lack of reorganizational changes in the rostral forelimb area in aged mice undergoing upper extremity rehabilitation treatment and could indicate that longer training sessions are needed. 42
The results are consistent with previous results in healthy participants where Hebbian-type stimulation combined with training resulted in enhanced encoding of a motor memory. 13 In these single exposure experiments, motor training leads to encoding of the kinematic details of the practiced movements, a form of plasticity that involves NMDA receptor function as well as GABAergic neurotransmission and shares similarities with mechanisms involved in LTP. 10 In a similar study with healthy older adult participants, wrist extension training paired with M1 Hebbian-type stimulation resulted in enhancement of training-related gains in hand kinematics and M1 excitability in a muscle supporting the training movement, further supporting the notion of enhanced induction of M1 plasticity. 21 In the present study, the tighter link between improved hand function and increased M1 functional activity in the trainingHebb group was not observed immediately after training but at 4 weeks after completion of training. This is consistent with previous reports 46 where evidence for M1 reorganization with rehabilitation-related behavioral improvement is not demonstrated immediately after training 42 and may indicate that the reorganizational processes in M1 that support improved hand function develop over time and are related to long-term reorganizational processes. 47 Taken together, the stronger link between improved hand function and increased ipsilesional M1 functional activity in the trainingHebb group at 1 month suggests that Hebbian-type stimulation induces reorganizational processes in ipsilesional M1 that support improvement and maintenance of hand function through its CST projections. While the causal relationship between Hebbian-type stimulation, M1 activity changes, and maintenance of hand function cannot be established from the data gathered here, similar functional processes have been reported for the injured M1 in rodents 19 and nonhuman primates. 18 An alternative explanation or additional contributing factor for ongoing reorganization in M1 could be the effect of the documented increase in the use of the paretic hand (MAL) in the trainingHebb group that parallels the improvement in the hand function. However, because both groups had similar gains in hand function immediately after the training and the reported long-term improvement and the association between hand function improvement and M1 activity changes are seen only in the trainingHebb group, this is unlikely to be the underlying mechanism.
Consistent with previous reports,48,49 superior hand function was negatively correlated with contralesional M1 functional activity at baseline. Following the intervention, there was a positive relationship between ipsilesional M1 functional activation and hand function, so that the patients with the least impaired hand function had the highest level of ipsilesional M1 activation. The flip from a negative correlation between fMRI activity and better hand function at baseline to a positive correlation at follow-up may reflect the change in M1’s involvement due to motor learning. While several studies did not find a relationship with recovery or showed decreases in ipsilesional M1 activity following intervention in the subacute or chronic periods, 50 others have found that ipsilesional M1 activity increases during recovery, 48 perhaps indicating a “normalization” of M1 activity. After training, Cramer et al 51 showed the largest behavioral improvements in patients with the lowest initial ipsilesional M1 activity. While there was no relationship between M1 activation at the baseline time point and hand function at the final time point in this study, we did see that patients with the largest changes in ipsilesional M1 activity between baseline and follow-up also had the largest changes in hand function.
The role of contralesional M1 in the support of paretic hand function after stroke is a topic of debate and is of interest as contralesional M1 may serve as a target for rehabilitation treatment. 52 Similar to healthy older controls, 53 the involvement of the contralesional M1 in stroke patients depends on the motor challenge. 54 A meta-analysis 48 showed no consistent relationship between contralesional M1 activity and degree of impairment or time since stroke. Some previous reports 49 suggest that contralesional M1 activity in the chronic stage is associated with poor motor function, while others show no relationship 55 and others suggest that contralesional M1 activity persists in chronic stroke patients with good recovery and increases following training.50,54 While this question is not the main topic of the study, our findings indicate that in our patients, contralesional M1 activity supports training-related gains in hand function.
Sample size limitations prevent us from examining possible differential effects of cortical or subcortical strokes. In addition, individual or group differences in performance, attention, and strategy can create confounders in task-related fMRI, making characterizing the underlying causes of changes in activation difficult. 56 In the present study, individual-level confounders remain relatively stable over time due to the longitudinal approach and the chronicity of the stroke. As there were no differences in hand function or brain activity between the 2 groups at baseline, the superior gains in hand function in the trainingHebb group cannot be explained by differences in their baseline measurements. In the current study, we report a positive correlation between change in ipsilesional and contralesional M1 activity and change in hand function between baseline and a follow-up time point 4 weeks after the end of training. As the fMRI task remained the same across time points, it is unlikely that the changes in activity are related to changes in task difficulty or attentional demands. If task difficulty did change over time due to the observed hand function improvements, decreases rather than increases in task activation would be predicted. Furthermore, the concurrent changes in measures of motor function (JTT) and overall function in activities of daily living (MAL) in the trainingHebb group supports the robustness of the treatment effect in this group.
In conclusion, we demonstrate that Hebbian-type stimulation applied to ipsilesional M1 results in better maintenance of motor training–related gains in the hand function of patients with chronic ischemic stroke involving M1 and its corticospinal projections. As hand function is critically dependent on these structures, the tighter association between M1 activation and treatment-related gains in hand function suggests that M1 plasticity supports these behavioral gains. These results help to fill a recognized gap in knowledge 57 by offering biological insight into the dose-response relationship for the targeted CNS effects of noninvasive neuromodulation. Considering the impact of compromised hand function as one of the most common long-term deficits after stroke, 1 targeting ipsilesional M1 with Hebbian-type stimulation in this patient group seems to be a clinically promising approach that deserves further development. While the design of this study does not rule out the possibility that other rTMS protocols may have had similar effects, the frequency, intensity, and timing of the protocol used for Hebbian-type stimulation in the current study were informed by preclinical data 11 and developed in previous studies of young and age-matched healthy controls13,21 and are consistent with the evidence for the importance of the temporal relationship between afferent stimulation and discharging of PTNs.13-15
Supplemental Material
NNR899911_Supplemental_Table_CLN – Supplemental material for Hebbian-Type Primary Motor Cortex Stimulation: A Potential Treatment of Impaired Hand Function in Chronic Stroke Patients
Supplemental material, NNR899911_Supplemental_Table_CLN for Hebbian-Type Primary Motor Cortex Stimulation: A Potential Treatment of Impaired Hand Function in Chronic Stroke Patients by Kate Pirog Revill, Marc W. Haut, Samir R. Belagaje, Fadi Nahab, Daniel Drake and Cathrin M. Buetefisch in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We would like to thank our subjects for their participation and M. Bidgood for clinical assessment of motor function.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Neurological Diseases and Stroke and the National Institutes of Child Development and Health at the National Institutes of Health, Bethesda, MD, USA (R21HD067906, R01NS090677), and the American Heart Association (15PRE25760023).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
