Abstract
Keywords
Introduction
Repetitive transcranial magnetic stimulation (rTMS) has been used in many studies as a novel intervention to treat disorders associated with stroke, including paralysis or dysphagia, hemispatial neglect, pain, and aphasia.1-12 Approximately 20% of stroke patients have speech and language problems, including hesitant, poorly articulated, agrammatic speech with word-finding problems (nonfluent aphasia). 13
The language network includes Broca’s and Wernicke’s areas in the left hemisphere and homologous areas in the right side of the brain, the prefrontal and premotor areas in the frontal regions, and the lower part of the parietal region.14,15 In the subacute phase after stroke (8 weeks), the bilateral activation was detected by positron emission tomography in 7 out of 11 patients. In a separate investigation on aphasic patients 12 days after stroke, increased functional magnetic resonance imaging (fMRI) activity in both the inferior frontal gyri and the temporal gyri were detected. 16
An increasing number of studies have demonstrated that low-frequency rTMS over the unaffected hemisphere can be useful for enhancing recovery in aphasic patients.6-11 Excitatory rTMS over the damaged hemisphere induces improvements in poststroke aphasia.12,17
We hypothesized that simultaneous application of low-frequency rTMS over the nondominant speech area and high-frequency (facilitatory) rTMS over the dominant speech area would have a beneficial effect on improving speech performance, particularly if we applied it early after stroke in combination with language training at a time when neural plasticity might be maximal. The aim of this study was therefore to evaluate the therapeutic role of dual-hemisphere rTMS in subacute ischemic stroke aphasia, and to study the changes of cortical excitability after treatment sessions.
Methods
This trial is reported following 2010 CONSORT guidelines. A participants’ flow diagram is shown in Figure 1.
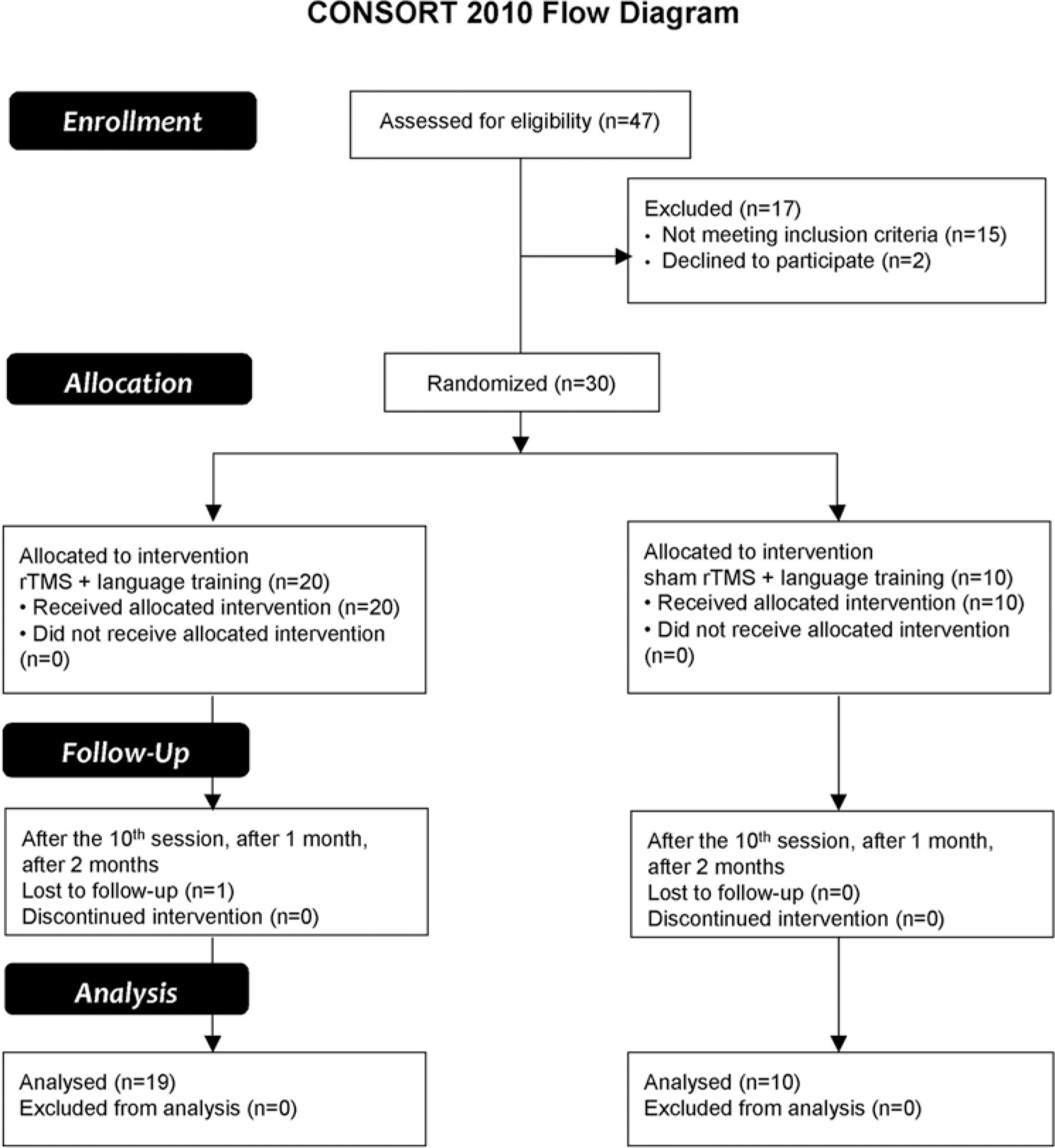
CONSORT 2010 flow diagram.
Patients
Forty-seven patients with poststroke nonfluent aphasia were recruited consecutively during the period from May 2010 to December 2012. All patients were admitted to the Stroke Unit, Department of Neurology, Assiut University Hospital, Assiut, Egypt. Each patient fulfilled the eligibility criteria as follows: subacute hemiplegia with nonfluent aphasia, single thromboembolic nonhemorrhagic infarction documented by computed tomography in the distribution of middle cerebral artery. Exclusion criteria included head injury or neurological disease other than stroke, unstable cardiac dysrhythmia, fever, infection, hyperglycemia, and prior administration of tranquilizer plus contraindications for rTMS according to the safety guidelines.18,19 The protocol was approved by the Assiut Medical School Ethical Review Board and all participants gave written consent.
Seventeen patients were excluded because 15 did not meet the inclusion criteria and 2 refused to participate. All patients were right-handed according to the Edinburgh Handedness Inventory. 20 Fourteen men and 16 women with subacute poststroke aphasia were admitted to the trial. The mean stroke interval was 5 ± 3.2 weeks, ranging from 1 to 12 weeks at study onset; aphasia was recognized in neuropsychological assessments with language deficits ranging from 0 to 5 points according to the the Aphasia Severity Rating Scale (ASRS). 21 Zero represents “no usable speech or auditory comprehension” and 5 represents “minimal discernible speech/ language handicaps.” The mean age was 57.3 ± 12.5 years. Twenty patients received real rTMS (one of them did not complete the follow-up) and 10 patients received sham rTMS.
Clinical Assessment
A detailed case history and neurological examination was performed for each patient. Hand strength and the language assessment section of Hemispheric Stroke Scale (HSS) 22 were evaluated for each patient. Additional scales used were the Stroke Aphasic Depression Questionnaire–Hospital Version (SADQ-H; asking about presence of depressive symptoms within the last week of stroke to be present all the days or frequent from 4 to 6 days, or infrequent from 1 to 4 days, or not at all) 23 and the National Institutes of Health Stroke Scale (NIHSS) 24 to assess the functional disability of each patient.
Cortical Excitability
Cortical excitability (resting motor threshold [rMT] and active motor threshold [aMT]) was measured with a monophasic magnetic stimulator (Magstim model 200; Magstim, Whitland, UK) connected to a 90-mm outer diameter figure-of-8 coil, which had a maximal output of 2.2 tesla. We located the optimal scalp location of each hemisphere from which TMS evoked motor-evoked potentials of greatest amplitude by moving the figure-of-8 coil systematically in 1-cm steps to determine the site of maximum peak-to-peak motor-evoked potentials in the first dorsal interosseous. The coil was positioned tangentially to the scalp and oriented so that the induced electrical currents would flow approximately perpendicular to the central sulcus, at a 45° angle from the mid-sagittal line. Single-pulse TMS was then delivered at the optimal location starting at suprathreshold intensity and decreasing in steps of 1% of the stimulator output to determine motor thresholds as defined by Rothwell et al. 25
We used silver–silver chloride surface electrodes, using a muscle belly-tendon montage, with a 3-cm diameter circular ground electrode placed on the wrist. A Nihon Kohden Machine model 9400 (Tokyo, Japan) was used to amplify and record the signals. Electromyography parameters included a bandpass of 20 to 1000 Hz and a recording time window of 200 ms. The electromyogram was monitored online for 20 seconds prior to stimulation to confirm muscle relaxation.
Randomization (Parallel Design)
Group allocations (real or sham with ratio 2:1) were placed in serially numbered opaque closed envelopes. Each patient was placed in the appropriate group after opening the corresponding sealed envelope.
Repetitive Transcranial Magnetic Stimulation Procedure
Real rTMS was applied for 10 sessions (5 days per week). A session of stimulation consisted of sequential stimulation of each hemisphere; one continuous 1-Hz train at 110% of the RMT over the unaffected right Broca’s area with 1000 total pulses (500 pulses over pars triangularis followed by 500 pulses over pars opercularis) followed by 10 trains of 20-Hz stimulation, each lasting for 5 seconds with an intertrain interval of 30 seconds given through a figure-of-8 coil (9-cm diameter loop) positioned over the left Broca’s area of the affected hemisphere (5 trains over pars triangularis followed by 5 trains over pars opercularis). We chose these areas based on the results of Gough et al, 26 who used TMS to define an anteroposterior division within the left inferior frontal cortex for semantic and phonological processing of words. The intensity of stimulation was set at 80% of the rMT for the first dorsal interosseous of unaffected hemisphere. Sham rTMS was applied using the same parameters, but with the coil held so that the edge was in contact with the head while the remainder was rotated 90° away from the scalp in the sagittal plane to reproduce the noise of the stimulation.27,28 Moreover, as the patients had never experienced rTMS previously, they did not know whether they were receiving real or sham rTMS. This was supported by self-report in a poststimulation survey. All patients, whether in the real or sham group, received single-pulse TMS to determine their RMT. Those patients in the sham group who had received single-pulse TMS were informed that the rTMS therapy would use a much lower intensity of stimulation than was required for testing, and that they would experience no muscle twitching during stimulation. During rTMS, all patients wore earplugs to protect the ears from the noise associated with the discharge of the stimulation coil.
We stimulated sequentially 2 parts of Broca’s area homologue: the anterior part (pars triangularis—PTr) and posterior part (pars opercularis—POp). To target the regions of interest precisely, we positioned the coil on the scalp according to the coordinates used by Gough et al. 26 The anterior stimulation site was 2.5 cm posterior to the canthus along the canther-tragus line and 3 cm superior to this line; the posterior stimulation site was 4.5 cm posterior and 6 cm superior to the canther-tragus line. The stimulation was applied in the same session with 500 pulses over pars triangularis followed by 500 pulses over pars opercularis in each hemisphere. The position of the TMS coil over the left Broca’s area and the right homologue of Broca’s area was validated in 3 patients using a 3-dimensional MRI sequence with vitamin E capsules in place 29 (see Figure 2).
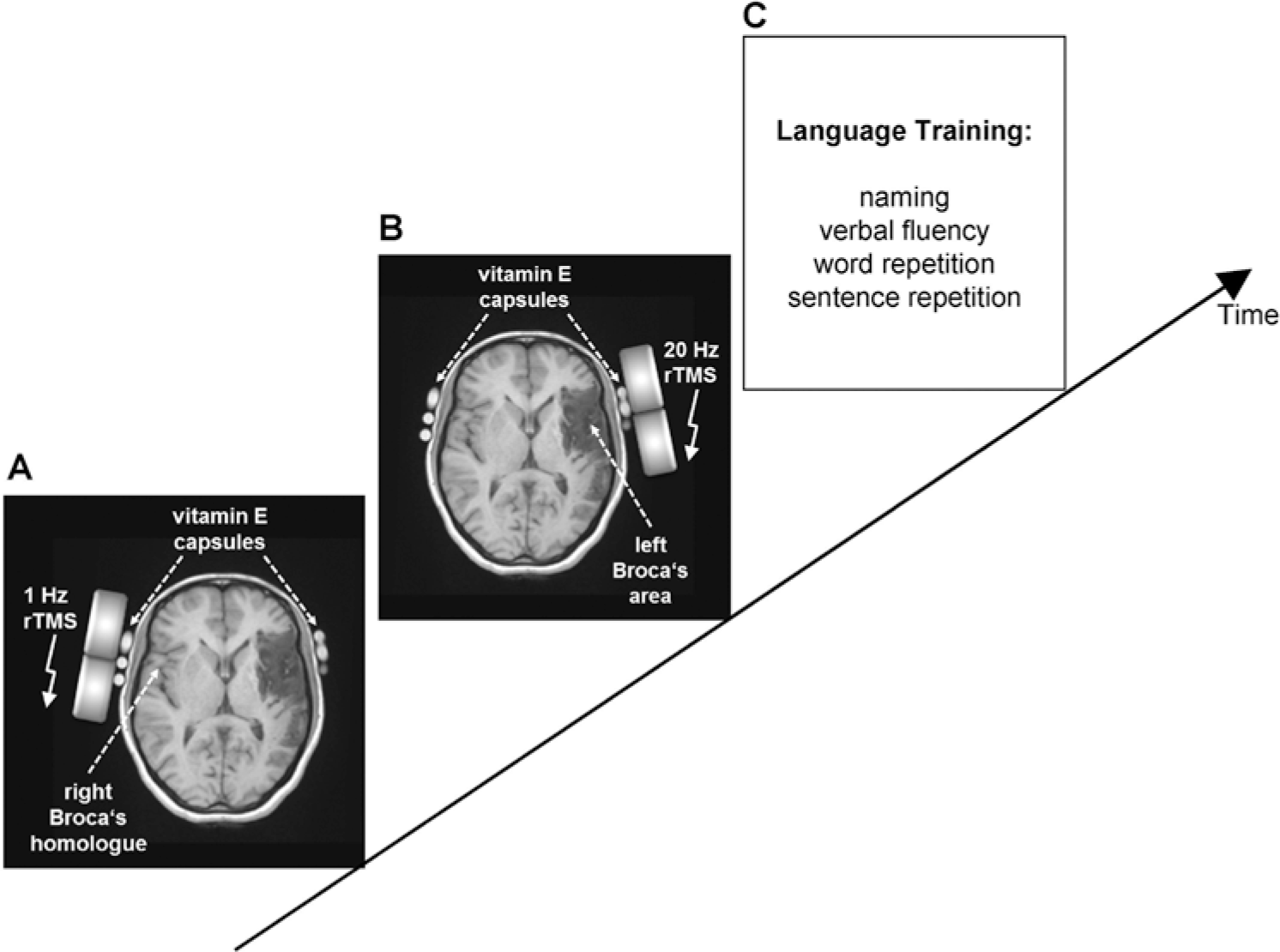
Experimental design. Real repetitive transcranial magnetic stimulation (rTMS) was applied for 10 sessions (5 days per week). A session consisted of one continuous 1-Hz rTMS train over the unaffected right Broca’s homologue with 1000 pulses (panel A) followed by 10 trains of 20-Hz rTMS over the affected left Broca’s area. Each train lasted for 5 seconds with an intertrain interval of 5 seconds (panel B). The position of the TMS coil was confirmed using a 3-dimensional magnetic resonance imaging sequence with vitamin E capsules in place. Immediately after the end of the applied stimulation protocol, each subject received speech and language training for 30 minutes (panel C). The control group received language training with sham rTMS as placebo control.
All the treatments were administered by only one investigator who was not involved in aphasia assessment and therapy. Immediately after the end of each session, each patient received speech and language training for 30 minutes.
Language Training Program
The rehabilitation program focused on specific training to stimulate various aspects of the language system. The following subtests of the Boston Diagnostic Aphasia Examination (BDAE)
21
were used for training: (
The general nature of the therapy method was similar for all patients, but the level of difficulty and types of exercises differed depending on the patients’ symptoms. Assessment and language training was performed by a speech and language therapist who was not aware of the patient’s group randomization. Therefore, any influence of the individual features of the therapist or differences in rehabilitation approach was minimized. The rational for combining rTMS with a training procedure is based on a previous study of Karim et al 30 showing for the first time that combining rTMS with operant learning can induce persistent after-effects not achievable neither with rTMS alone nor with operant learning alone. Figure 2A-C illustrates the experimental design.
Follow-up and Outcomes
All stroke patients were followed up clinically, after the end of last session and then at 1 and 2 months after the end of treatment with language rating scores as primary outcome and the other rating scales as secondary outcomes. All patients completed the evaluations except a male patient from the real group (see Figure 1). The evaluation was performed blindly by a neurologist who was unaware of the type of rTMS group. We also assessed resting and active motor thresholds and recruitment 1 hour after the last session. No patient developed seizures during the study or on follow-up.
Statistical Analysis
At the baseline assessment (ie, before rTMS), the mean values of different scales between both groups were compared using the Mann–Whitney test for independent samples. Means ± standard error were used to represent data. The level of significance was set at
Results
At baseline, there was no significant difference between the groups in age, sex distribution, duration of onset of stroke, site of infarction, HSS (language section “comprehension, naming, repetition, and fluency”), ASRS, SADQ-H, grip strength, and NIHSS (Table 1).
Demographic Data, Clinical Data, and Different Rating Scales of the Investigated Groups.
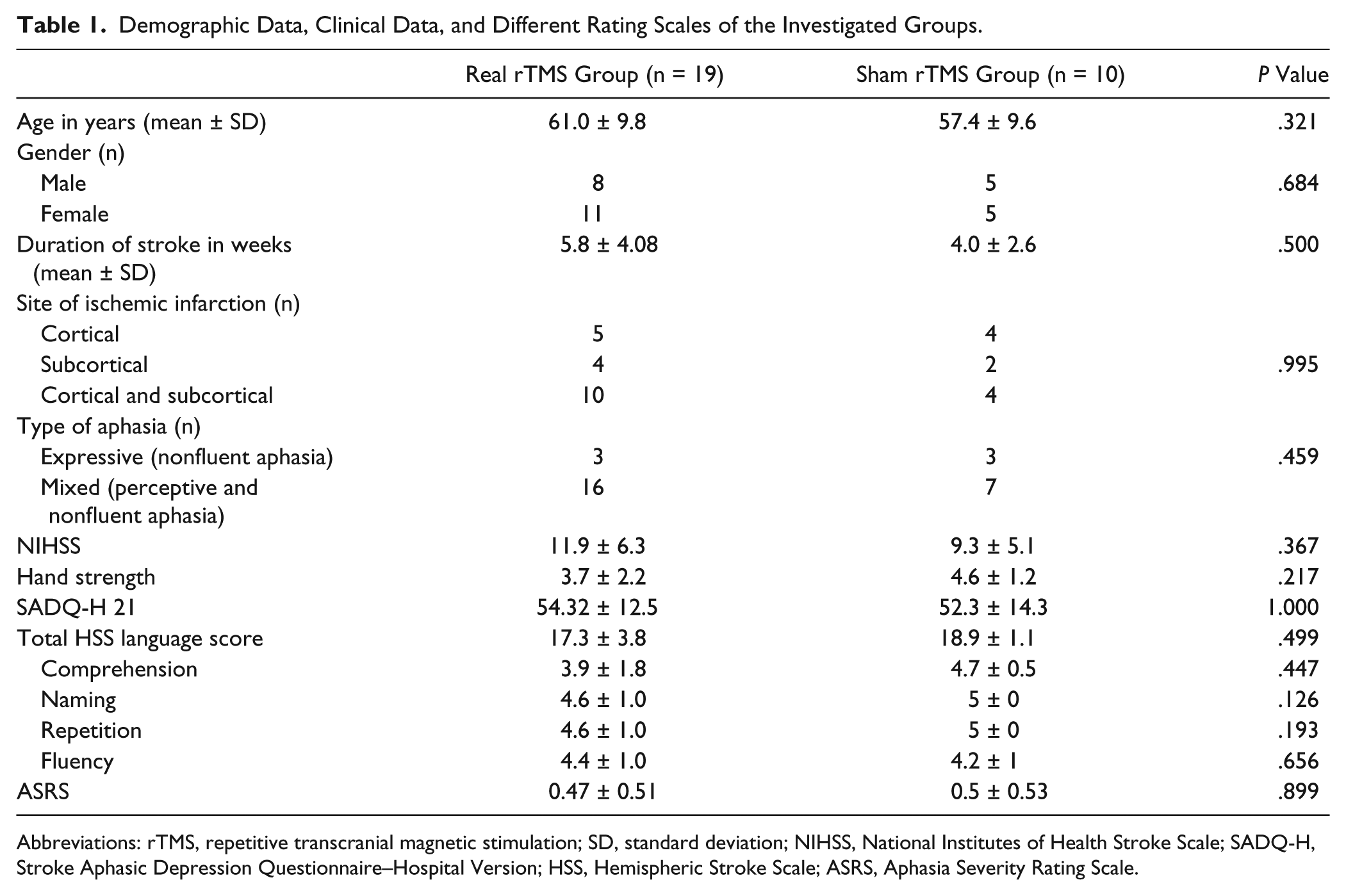
Abbreviations: rTMS, repetitive transcranial magnetic stimulation; SD, standard deviation; NIHSS, National Institutes of Health Stroke Scale; SADQ-H, Stroke Aphasic Depression Questionnaire–Hospital Version; HSS, Hemispheric Stroke Scale; ASRS, Aphasia Severity Rating Scale.
However, there was a significant improvement in the ASRS in the real rTMS group compared with the sham group. The ASRS in the real group increased by a mean of 1.8 ± 1.2 points (range 0-4), while in sham group it only increased by a mean of 0.9 ± 0.3 points (range 0-1). This was confirmed in a repeated-measures ANOVA that showed a significant Time (pre, post session, 1-month and 2-month postsession) × Group (real vs sham) interaction (
There was a significantly greater improvement in the HSS language score in the real rTMS group compared with the sham group both immediately after the end of treatment as well as at the 1- and 2-month follow up measurements (Figure 3A). This effect was confirmed in a repeated-measures ANOVA that showed a significant Time (pre, post session, 1- and 2-month post-session) × Group (real vs sham) interaction (F = 7.1, df = 1.6, 42.7 and P = .004). Post hoc
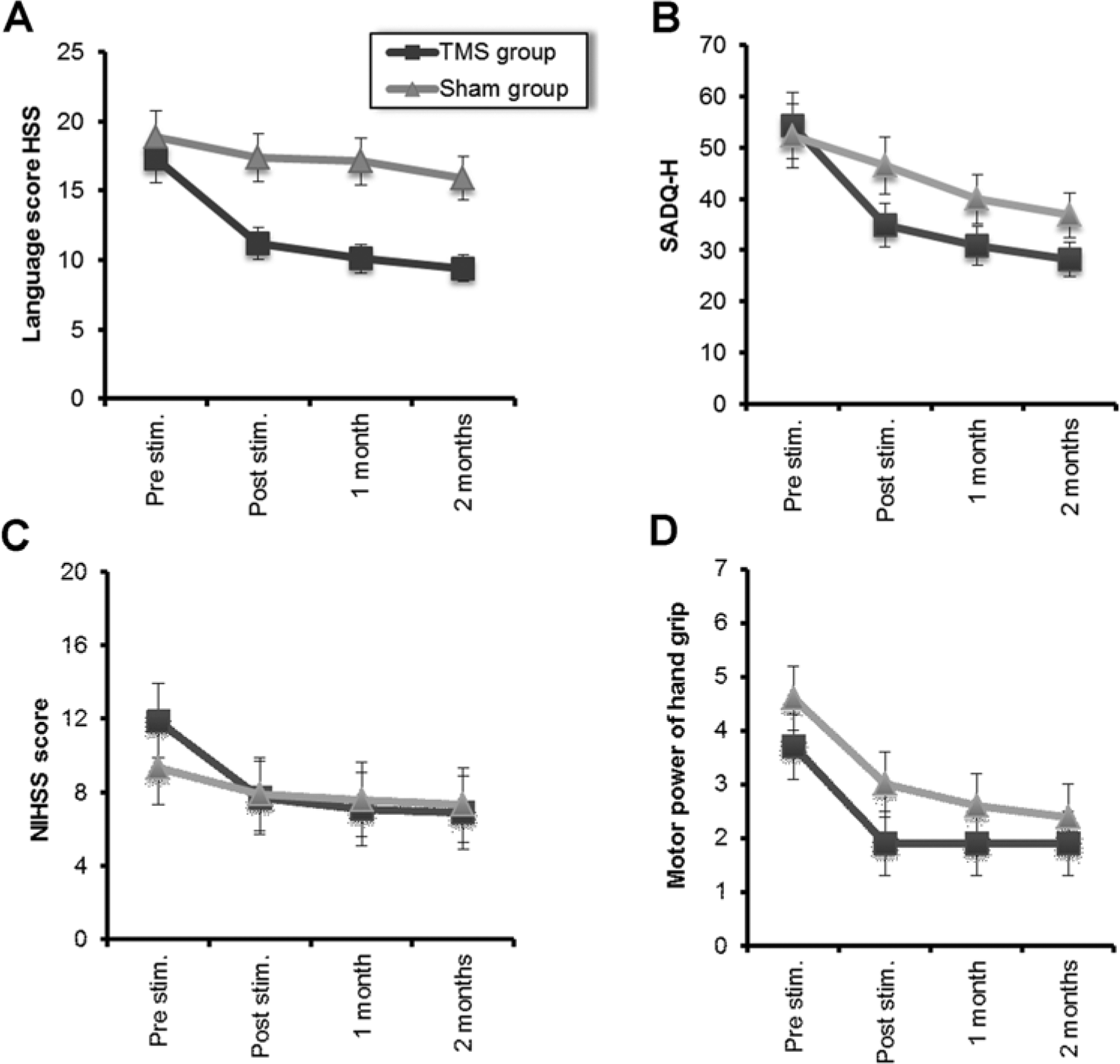
Changes in mean different rating scores of (A) Hemispheric Stroke Scale (HSS) language score, (B) Stroke Aphasic Depression Questionnaire–Hospital Version (SADQ-H 21), (C) National Institutes of Health Stroke Scale (NIHSS), and (D) hand grip strength at the 4 assessment points for the 2 groups of patients with stroke. The first assessment was immediately prior to commencing repetitive transcranial magnetic stimulation (rTMS) treatment (Pre), the second (Post session) was immediately after the end of the 10th session of rTMS, the third and fourth assessment were immediately after the end of the first and the second months, respectively. Each group separately shows significant improvement. However the mean scores of the patients who received real rTMS are significantly better than sham group over the course of the treatment in HSS language score and SADQ-H 21, while hand grip strength showed no significant differences (
The scores on the SADQ-H behaved similarly, with a significant Time × Group interaction (
Analysis of the subscores of the HSS language scale showed that there were significantly greater improvements in the rTMS than in the sham groups in all 4 main items of comprehension, naming, repetition, and fluency (Time × Group interactions:
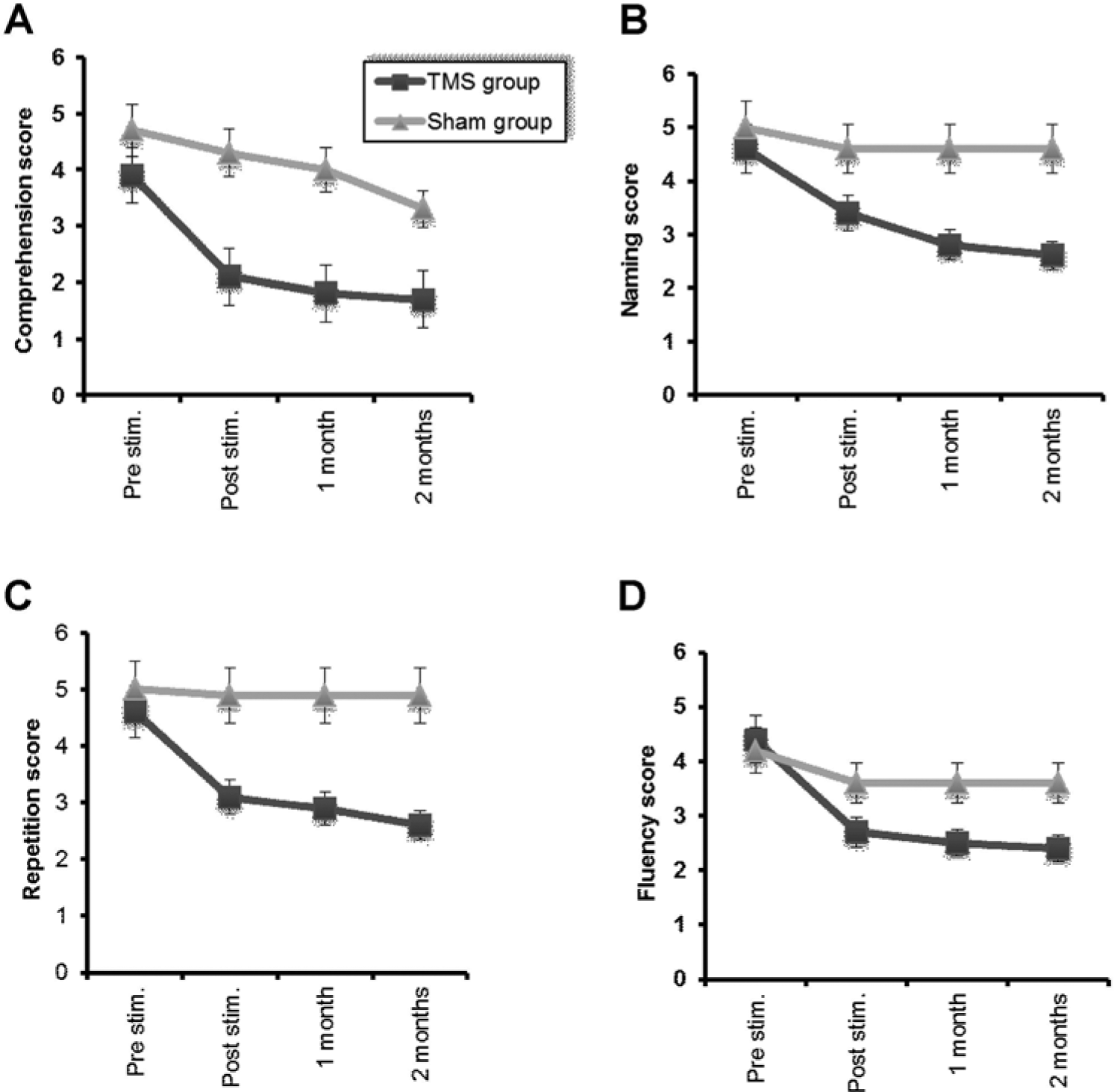
Changes in mean different rating scores of comprehension (A), naming, (B) repetition (C), and fluency (D) in real and sham groups along the course of the treatment and follow-up. The first assessment was immediately prior to commencing repetitive transcranial magnetic stimulation (rTMS) treatment (Pre), the second (Post session) was immediately after the end of the fifth session of rTMS, the third was immediately after the end of the first and the fourth assessment at the end of the second month. Each group separately shows significant improvement. However, the mean scores of the patients who received real rTMS are significantly better than those of the sham group (
Cortical Excitability
We assessed 13 patients in the real rTMS group and 7 cases in the sham group. Three cases in the real group plus 1 case in sham group had no motor-evoked potential from the affected hemisphere.
There was a significant increase in cortical excitability of the affected hemisphere when tested immediately after the last treatment session as demonstrated by reduced the rMT and the aMT in the TMS group (
In contrast, there was a significant mild inhibition in the unaffected hemisphere of the TMS group; however, the ANOVA did not show an interaction, just an effect of TIME, thus we have to conclude that there was a small increase in threshold in both groups (Figure 5).
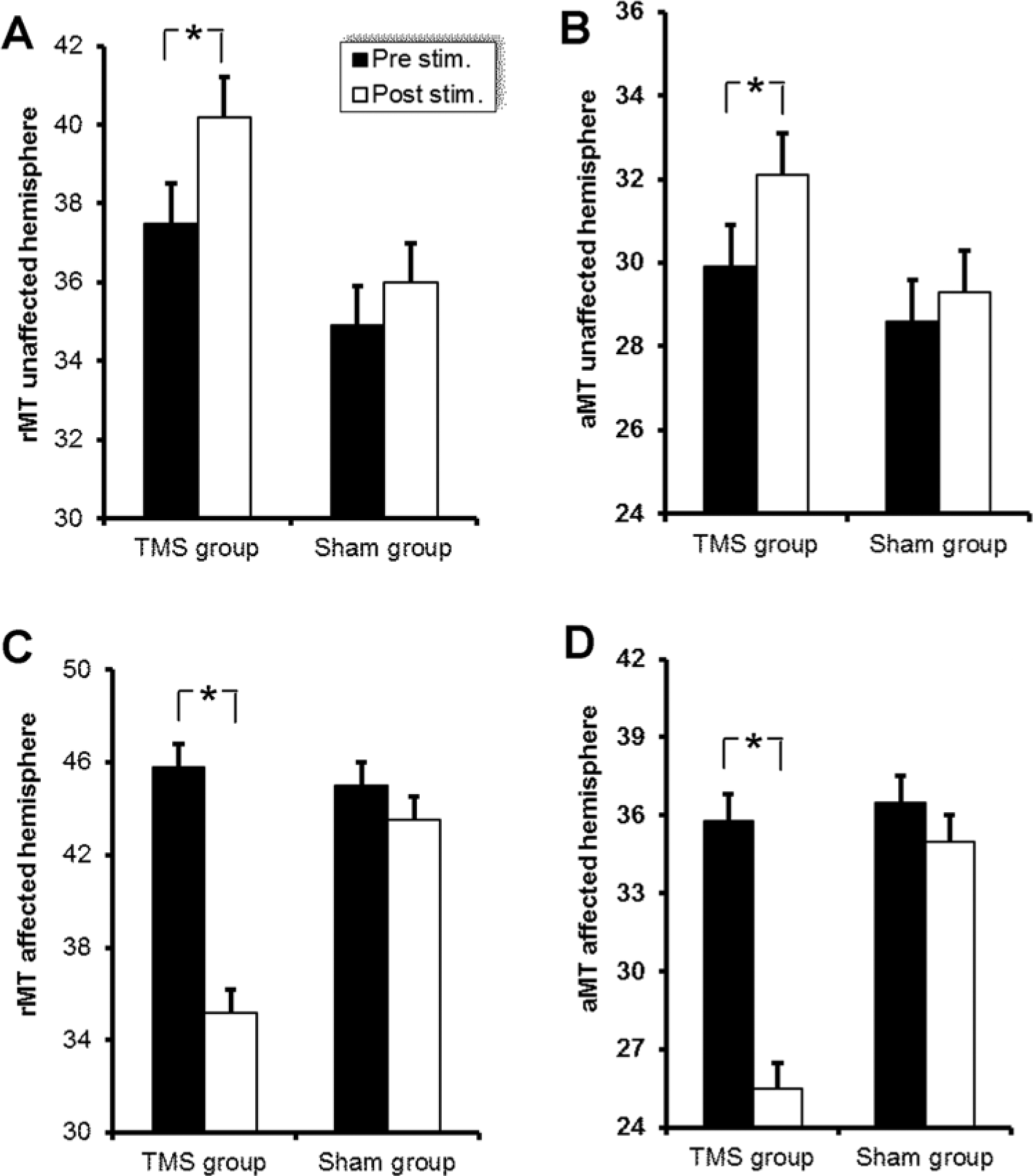
There was a significant increase in cortical excitability of the affected hemisphere as demonstrated in decreased resting and active motor thresholds in the TMS group compared with the sham group (
Moreover, there was a significant correlation between the change in the rMT (pre–post session) for all patients for the affected hemisphere and the change in the language score (pre–post session) with
Discussion
Direct manipulation of cortical brain activity by rTMS has the potential to serve as an efficacious complimentary rehabilitatory treatment for poststroke aphasia.
The main finding in this present study was the significantly greater improvement in language recovery (word comprehension, naming, repetition, and frequency) and in the aphasia severity rating scale (ASRS) in the group receiving real rTMS (dual stimulation) compared with the sham group. This effect occurred immediately after the treatment sessions and persisted for 2 months. We used a bihemispheric approach in which low-frequency rTMS (1 Hz) was given over the right Broca’s area in an attempt to remove the inhibitory effect from the nondominant to the dominant hemisphere, combined with high-frequency stimulation over left Broca’s area, in an attempt to strengthen neural connections within an area of metabolic dysfunction. The parameters that we chose for rTMS were within current safety guidelines. 19 Despite the absence of any rTMS side effects in this series, electroencephalography would be an important monitor to include in future studies to warn of possible seizure onset.
Our experimental design differs from previous work in the following ways. First, we targeted an early-stroke patients’ cohort, whereas most of the previous studies were done on chronic patients7,9,11,31-35 and few were conducted in the subacute stages.8,10 To maximize patient recruitment and compliance, this study was conducted during their initial hospital admission within the first few weeks of the stroke. Although functional recovery is not stable until several months after a stroke, early intervention might maximize the potential benefit. Second, we used the simultaneous bihemispheric approach (stimulating the unaffected then affected hemisphere) rather than limiting stimulation to one hemisphere. The rationale for keeping the stimulation order fixed was to reduce right hemisphere hyperactivity and transcallosal inhibition exerted on the left Broca’s area in order to prepare it for further facilitation (using high frequency rTMS) to return the balance between both hemispheres. Third, each stimulation session was followed by speech and language training. The rationale for combining rTMS with a training procedure is based on a previous study of Karim et al 30 showing for the first time that combining rTMS with operant learning can induce persistent after-effects not achievable neither with rTMS alone nor with operant learning alone.
A number of studies have examined the effect of 1 Hz rTMS of the contralesional right inferior frontal gyrus. Weiduschat et al
7
reported a beneficial effect of 2 weeks rTMS in a heterogeneous series of subacute aphasic patients. However, this was not replicated by Seniów et al
6
in their double-blind, placebo-controlled pilot study of 40 patients with subacute poststroke aphasia. However, they did report improvement (minimal word
Instead of inhibiting the right hemisphere, 3 rTMS studies attempted to restore the left hemisphere networks and perilesional neuronal activity in the injured Broca’s area by applying excitatory rTMS protocols (either intermittent theta burst stimulation or high-frequency rTMS) on the left inferior frontal gyrus of aphasic patients.12,17,35 They showed favorable effects in patients with nonfluent chronic aphasia. In the first study, intermittent theta burst stimulation trains were delivered under fMRI-guided navigation to the Broca’s area. 12 Clinical improvement (increased fluency) was associated with concomitant fMRI changes showing a relative increase in the activity of the lesioned inferior frontal gyrus, which was stimulated. 12 Cotelli et al 35 applied excitatory high-frequency rTMS over the left dorsolateral prefrontal cortex combined with speech therapy and observed a significant improvement in object naming.
Interestingly in the present study; 5 cases in the real rTMS group did not improve in HSS or NIHSS (4 cases had extensive infarction and 1 case had operculum infarction). This means that those patients with complete middle cerebral artery occlusion are not suitable to receive dual-hemisphere stimulation and perhaps would benefit more from high-frequency rTMS to the unaffected hemisphere to enhance activation of the right homologue of Boca’s area because the left Broca’s area is severely damaged.
Several neuroimaging studies with chronic, nonfluent aphasia patients during language tasks have observed high activation and possibly “overactivation” in parts of the right Broca’s area and other regions such as the right perisylvian language homologues, which may be maladaptive.31,36 When 1-Hz rTMS is applied to right hemisphere cortical regions of interest, including M1 mouth area, the superior temporal gyrus and subregions within the Broca’s area such as pars triangularis posterior and pars opercularis, rTMS may suppress transcallosal inhibition from these areas onto the damaged hemisphere, and thereby permit reactivation of some areas in the left hemisphere and facilitate functional recovery. 37 In contrast, high-frequency stimulation has been shown to cause regeneration of the left damaged part of the brain assessed by diffusion tensor imaging. 38 Stimulating neurons in the perilesional zone with appropriate parameters could increase the neuronal survival rate and facilitate clinical recovery. 37 Thus, both high- and low-frequency rTMS could induce neuroplasticity.39,40
Moreover, the observed improvement of speech and depression in this study could be explained on a reciprocal basis: improvement of speech could improve depression and vice versa. However, it should also be noted that stimulation of the left Broca’s area could spread to stimulate the left dorsolateral prefrontal cortex either directly or through functional connectivity. rTMS of dorsolateral prefrontal cortex has been reported to improve depression in previous studies. 41 Thus, rTMS may be able to enhance stroke recovery in general by acting directly on the underlying cortical regions or through its connections with other structures. Alternatively, there may have been indirect effects of rTMS in reducing patients’ depression as assessed by the SADQ-H, thereby increasing compliance with the treatment.
Conclusion and Recommendation
Repetitive TMS has some potential to be an adjuvant therapy for subacute poststroke expressive nonfluent aphasia. The present study was exploratory and followed only a small number of patients for a short time. Further studies are needed with a larger sample size to confirm the results. Cortical excitability changes and neuroimaging studies would be helpful to study bihemispheric changes in language recovery.
Footnotes
Acknowledgements
The authors would like to thank Prof John Rothwell for useful discussion during the preparation of this article and Prof Andreas J. Fallgatter for his valuable comments.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
