Abstract
Introduction
The organization of the sensory and motor cortices and their connections to the musculature provide the basis for the control of voluntary movement. Learning novel tasks and practicing motor skills can lead to measurable changes in this organization. 1-8 It is thought that this adaptation is not only critical for learning new skills but also for recovery after a neural insult such as stroke. 9 The goal of the current research is to develop paradigms to improve motor function for hemiplegic patients who have minimal control of the hand. We investigated a bilateral task in which movement by one hand triggers electrical stimulation of the other hand, to determine if the concurrent performance of a motor task would modulate the effect of the stimulation on corticospinal excitability. This task was designed to take advantage of activity-based adaptation, 3,7 stimulation-induced adaptation, 10,11 and the benefits of bilateral tasks. 12
Adaptations within the motor cortex can be observed after optimizing performance of a motor task through repeated practice. Typical tasks consist of maximizing the acceleration of a single joint of the thumb or index finger, 2-4,8 practicing a novel direction of thumb movement, 1,7 learning an isometric force task, 13 or maximizing tapping speeds. 14 Adaptation is also observed after practicing more complex tasks such as the Purdue Pegboard 5 or training in a novel physical environment. 6 Transcranial magnetic stimulation (TMS) applied to the motor cortex is used to assess these task-induced adaptations by either measuring corticospinal excitability 2,5,6,14 or the stimulation-induced direction of movement. 1,7 Many of these unilateral tasks also demonstrate effects on the untrained cortex/limb, such as changes in corticospinal excitability, 4,8 induced movement direction, 15 and performance. 4,8
Motor cortex adaptations are also observed after the application of pure stimulation paradigms, demonstrating the strong link between the sensory and motor components of the motor system. These paradigms consist of either repeated peripheral electrical stimulation or a combination of peripheral electrical stimulation and central stimulation (TMS). Purely peripheral paradigms consist of either a single point of stimulation 16 or 2 locations. 10,11 Paired stimulation appears more efficient than single point stimulation, producing alterations in TMS measured cortical excitability 10 and in cortical motor maps 11 after an hour of exposure to paired stimuli. In contrast, 2 hours are required to induce changes in single point stimulation paradigms. 16 The combination of a peripheral stimulation (electrical) and a central stimulation (TMS), often referred to as paired associative stimulation (PAS), also induces adaptations in cortical excitability 17-21 and can induce measurable adaptation in exposures as short as 20 minutes. 2,22 These paradigms are thought to modulate cortical responses to TMS by adaptation of synaptic strength in a Hebbian-like manner to the converging peripheral and central input. 17,18,23
There is evidence that the effects of PAS can be modified by concurrent motor tasks. Small focal contractions of the PAS targeted hand muscle lead to increased facilitatory effects of PAS on that muscle. 24 Furthermore, a bilateral task that combined contraction of wrist flexor muscles of the left arm with PAS applied to the right arm demonstrated increased PAS-induced facilitation and muscle specificity. 25 The current experiment investigated whether pairing peripheral stimulation of one hand with a voluntary motor task performed by the other hand will induce measurable adaptation in primary motor cortex of the stimulated hand. Two control experiments were performed to determine what adaptation results from the individual performance of the motor or stimulation aspect of the combined task. Understanding how the motor system adapts and what drives this adaptation is of interest for both research and rehabilitation. Although some evidence of improved motor function after the application of plasticity inducing paradigms in healthy individuals 26 and those with underlying movement disorders 27,28 has been observed, the future therapeutic potential of these paradigms is not yet known.
Methods
A total of 15 neurologically intact participants (7 women, mean age 26.8 ± 7.8 years, 1 left-handed) participated in the motor and sensory task (MStask) and stimulation only task (Stask). A total of 11 participants were recruited for the motor only task (Mtask; 4 women, mean age 27.3 ± 7.8 years, 2 left-handed). Six participants participated in all 3 tasks. All participants were recruited from the university community and gave written, informed consent. The experimental procedures were approved by the University of Tasmania’s Human Research Ethics Committee.
Tasks
The MStask, Stask, and Mtask are illustrated in Figure 1. The motor component consisted of repeated isometric thumb abductions of the right hand (10 blocks of 50 repetitions at 0.5 Hz). Participants were asked to make brief efforts using visual feedback so that the peak of each effort matched a magnitude target (35% of maximal effort) and a temporal target (see Figure 1). The hand and forearm were supported and secured. The stimulation component consisted of electrical stimulation (5 pulses, 100 µs width, at 30 Hz) applied simultaneously to the motor point of the first dorsal interosseus (FDI) and abductor pollicis brevis (APB) of the left hand using 2 Digitimer DS7A constant current stimulators (Digitimer Ltd, Hertfordshire, UK). Stimulation intensity was adjusted to elicit a visible movement of the digit. 10,16,29 The MStask combined the motor task with electrical stimulation that was triggered when force reached 2.5% of maximum. The Stask was performed 1 week later. Stask participants viewed a replay of the visual feedback from the MStask while receiving equivalent sensory stimulation to that received during the MStask. To ensure attention to the visual feedback, participants verbally indicated particularly good and bad trials to the experimenter. The Mtask was conceived as a further control after analysis of the MStask and Stask and consisted of only the motor component. Three maximal effort trials were performed in the MStask and Mtask to establish the isometric force target levels. These efforts were performed before all TMS testing in the MStask, whereas initial TMS sessions were performed before and after the maximal efforts in the Mtask.
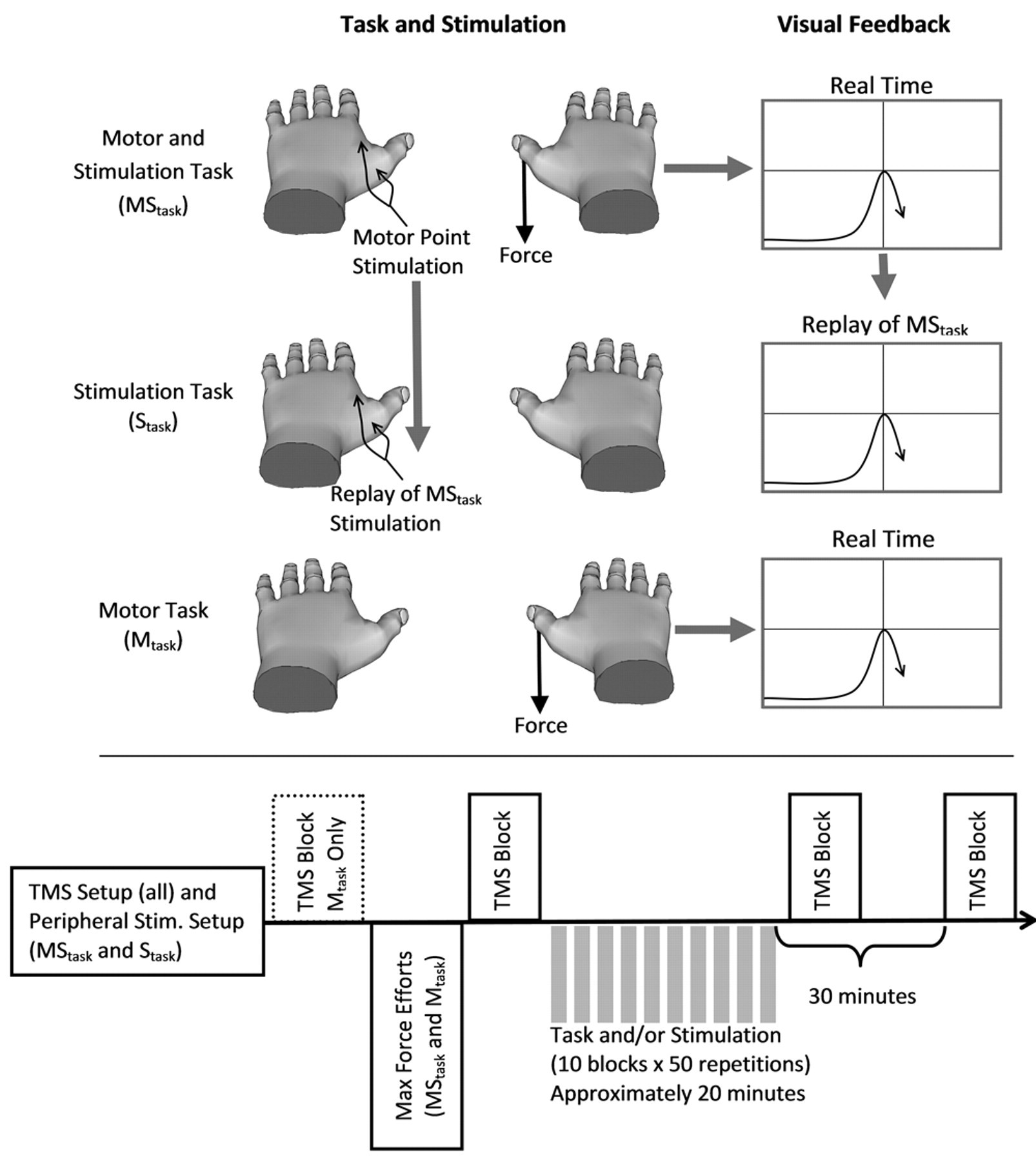
Tasks and timeline. The top panel demonstrates the 3 tasks. The motor and stimulation task (MStask) consisted of a targeted isometric thumb abduction force that triggered simultaneous 30-Hz stimulation of the motor points of the abductor pollicis brevis (APB) and first dorsal interosseous (FDI) of the left hand. The stimulation only task (Stask) consisted of a replay of the sensory stimulation from the MStask while viewing a replay of the visual feedback from the MStask. The motor only task (Mtask) was the same as the MStask but with no sensory stimulation. The bottom pane shows the time course of each experiment. Transcranial magnetic stimulation (TMS) measures were collected for the right cortex/left hand for all tasks. An additional TMS testing block was included in the Mtask to assess the effect of performing the maximum force efforts.
Transcranial Magnetic Stimulation and Electromyography
The effect of task performance on corticospinal excitability, short latency intracortical inhibition (SICI), and intracortical facilitation (ICF) was assessed by measuring the amplitude of motor evoked potentials (MEPs) resulting from TMS stimulation of the motor cortex. Excitability was assessed using test stimulus intensity of 130% of each participant’s resting motor threshold (RMT). SICI and ICF were tested with a paired-pulse technique consisting of a small (70% RMT) conditioning TMS stimulus applied before the 130% TS. SICI is measured using 3-ms delay between stimulations and results in a reduction in the response size, 30 which reflects GABAergic inhibition. 31,32 ICF is measured with a 15-ms delay and results in an increased response size thought to result from glutamate activity. 30,33 Test stimulus intensity was maintained for all testing blocks, despite possible changes in MEP size, as previously recommended. 34 Three TMS testing blocks were performed for the MStask and Stask, whereas a fourth block was added to the Mtask to test the effect of the maximum force efforts. For the MStask and Stask, each block consisted of 45 stimulations (15 TS, 15 SICI, 15 ICF) at 0.2 Hz; 12 repetitions of each was used in the Mtask.
Electromyogram recordings were obtained from 3 intrinsic hand muscles (FDI, APB, and abductor digiti minimi [ADM]) of the left hand using pairs of disposable Ag/AgCl electrodes in a belly-tendon montage. Electromyogram data were recorded at 5000 Hz, bandpass filtered (20 to 500 Hz), and amplified (1000×) using a CED 1902 signal conditioner and Power 1401 data acquisition system (Cambridge Electronic Design, Cambridge, UK). TMS was applied to the right cortex with a figure-of-eight coil (70-mm diameter) using a pair of Magstim 200 stimulators connected through a BiStim module (Magstim Company, Whitland, UK). The coil was orientated with the handle pointing backward at ∼45° angle to the sagittal plane. The hotspot for the APB was determined and marked on the scalp. The RMT was defined as the minimum stimulator intensity that elicited 3 of 5 responses greater than 50 µV in the relaxed APB muscle. Muscle activity was visually monitored during the experiment to ensure the test muscles were relaxed.
Data Analysis
Behavioral data from the MStask and Mtask were quantified for each block with mean time error and mean force error normalized to each participant’s maximal force. Stimulation parameters between tasks were compared with paired
Results
Stimulation Parameters
The electrical stimulation applied to the FDI was similar for both the MStask and the Stask (13.2 ± 3.2 mA and 13.4 ± 2.0 mA, respectively
Behavioral
All participants were able to perform the motor task and improved on both force accuracy and timing accuracy across the 10 blocks (Figure 2). Performance in the MStask and the Mtask was nearly identical. For the participants (n = 6) who performed all 3 tasks, the time between performing the MStask and Mtask ranged from 218 to 519 days (mean 365 days). No carryover in performance was observed for these participants (block 1 mean force error of the MStask 4.67% ± 1.03% vs block 1 mean force error of the Mtask 5.09% ± 1.95%; paired
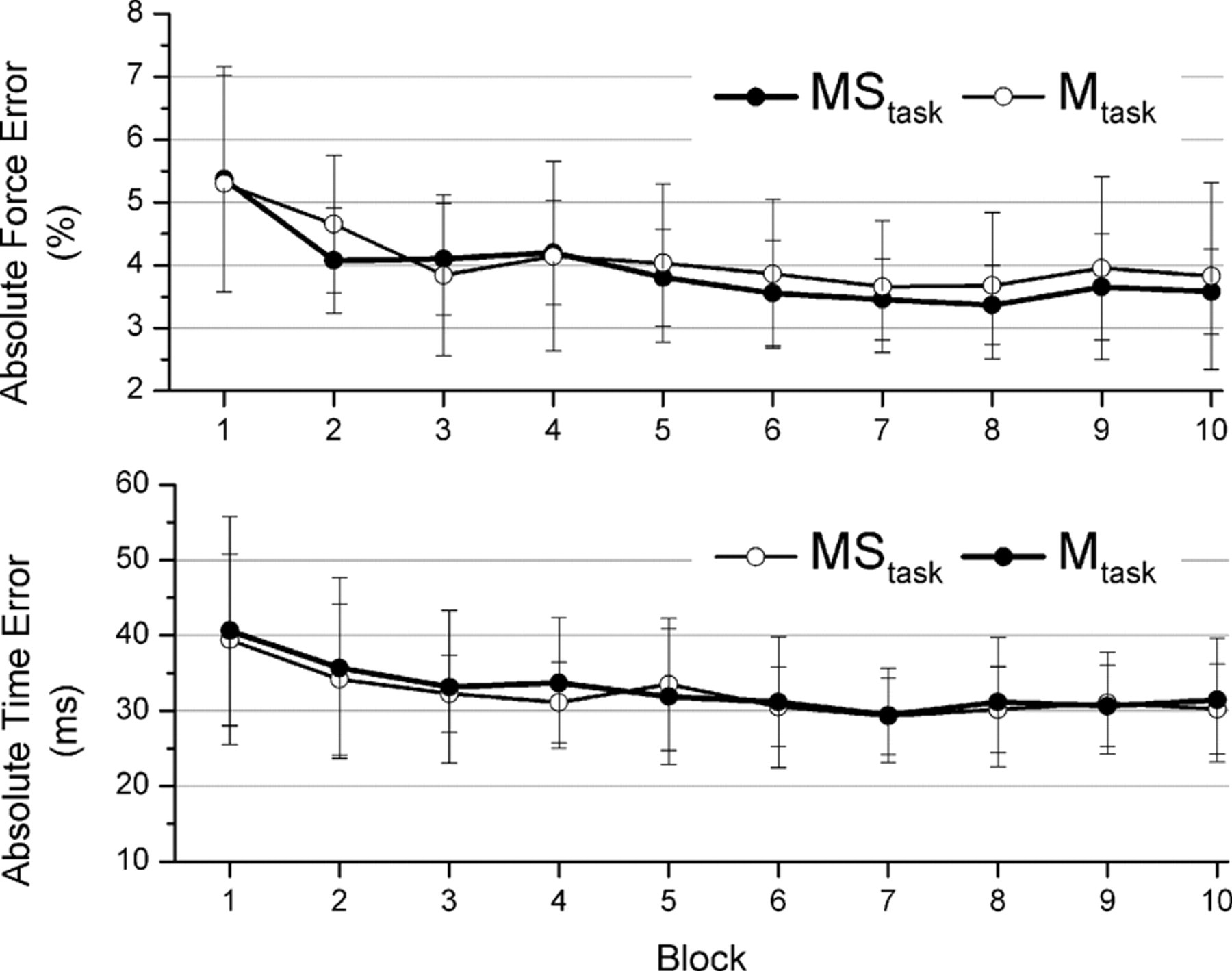
Force and time error by block for the motor and stimulation task (MStask) and motor only task (Mtask). Top panel shows force error and the bottom panel shows time error. Points are means with error bars representing standard deviations.
Corticospinal Excitability
The MStask resulted in a task-specific facilitation of the FDI and APB muscles, which was maintained 30 minutes after task completion (Figure 3). For the MStask the mean facilitation for the FDI was 152%, 95% CI [112%, 204%] Post 1, and 187%, 95% CI [123%, 256%] Post 30. The APB was facilitated 185%, 95% CI [108%, 303%] Post 1, and 154%, 95% CI [105%, 212%] Post 30. In the MStask, the ADM confidence interval spanned 100% but was largely above it for both Post 1 (130%, 95% CI [91%, 172%]) and Post 30 (131%, 95% CI [92%, 180%]) suggesting the task may have had a facilitatory influence on the ADM. Examination of Figure 3 demonstrates that the response to the MStask was not consistent across participants, with some participants showing a small decrease in excitability. This range of responses is reflected in the width of the confidence intervals. The Stask and Mtask had minimal effect on excitability with all confidence intervals overlapping 100%. The confidence interval for APB in the Stask was shifted up at Post 1 (121%, 95% CI [90%, 162%]) and Post 30 (129%, 95% CI [93%, 173%]), suggesting that facilitation is more likely than inhibition. Initial MEP amplitudes are presented in Table 1.
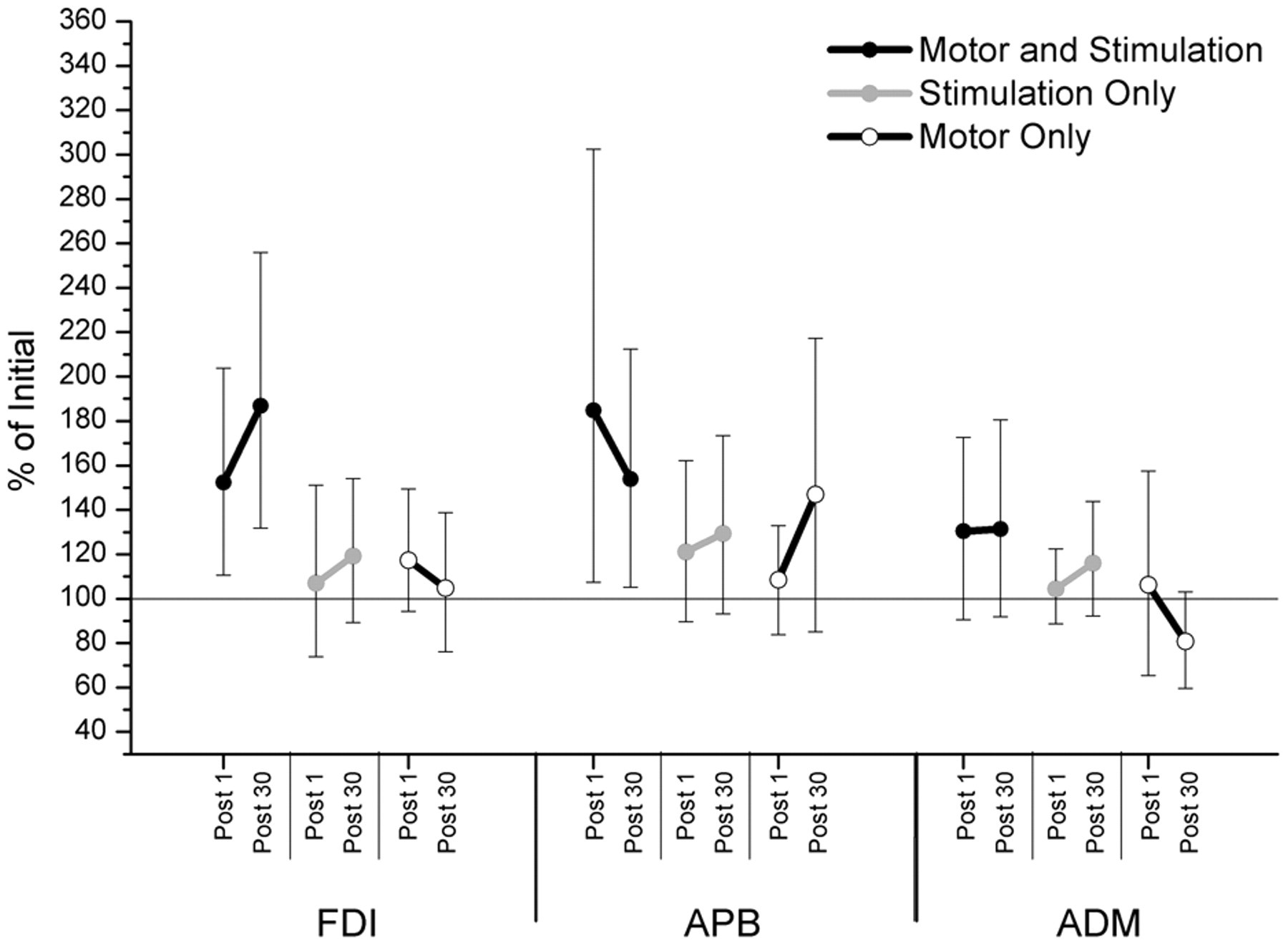
Effect of task on excitability change for 3 muscles: first dorsal interosseous (FDI), abductor pollicis brevis (APB), and abductor digiti minimi (ADM). For each muscle, time, and task 95% confidence intervals of the ratio to the initial test session are presented. Facilitation is observed after performance of the motor and stimulation task but not the stimulation only or motor only tasks.
Initial Excitability, Inhibition, and Facilitation Values for Each Task
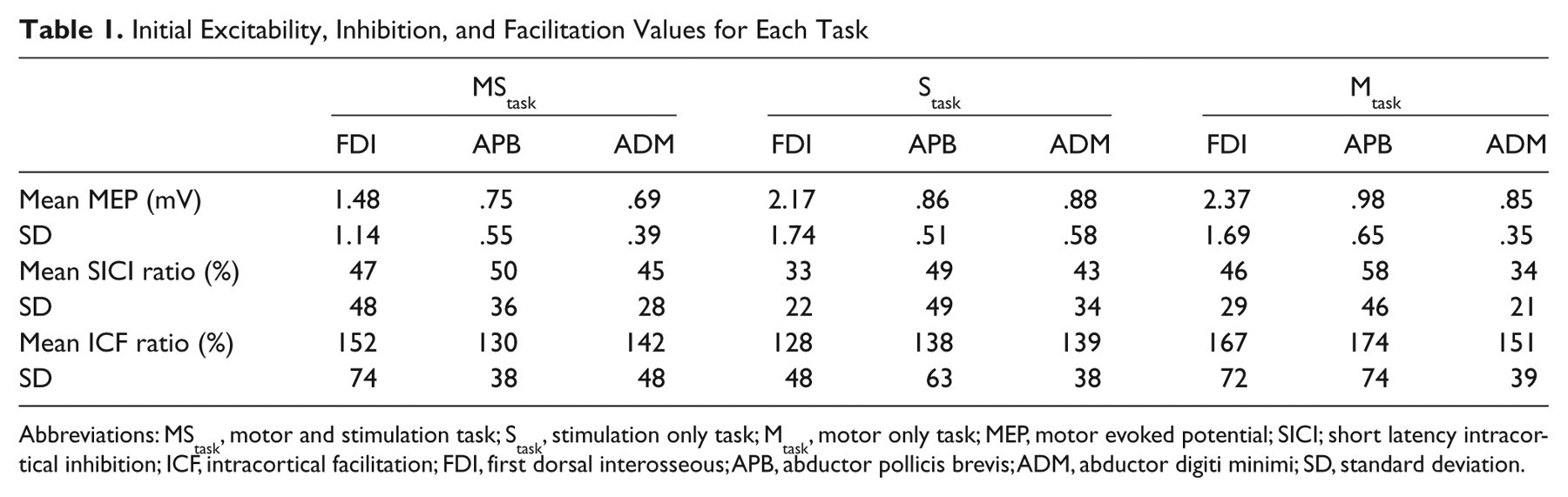
Abbreviations: MStask, motor and stimulation task; Stask, stimulation only task; Mtask, motor only task; MEP, motor evoked potential; SICI; short latency intracortical inhibition; ICF, intracortical facilitation; FDI, first dorsal interosseous; APB, abductor pollicis brevis; ADM, abductor digiti minimi; SD, standard deviation.
Increased MEP Responses 1 Week After the MStask
Comparison between measurements of corticospinal excitability in the initial TMS block of the MStask and Stask revealed an increase in response size 1 week after performance of the MStask (Figure 4). Mean increases in excitability were FDI 200% (95% CI [131%, 292%]), APB 170% (95% CI [117%, 227%]), and ADM 144% (95% CI [112%, 175%]). It is possible that the performance of the maximal efforts before the MStask but not before the Stask led to the observed increase in responses. Thus the Mtask was designed with TMS blocks before and after the maximum efforts. No effect of the maximum effort on excitability was observed in the Mtask (Figure 4).
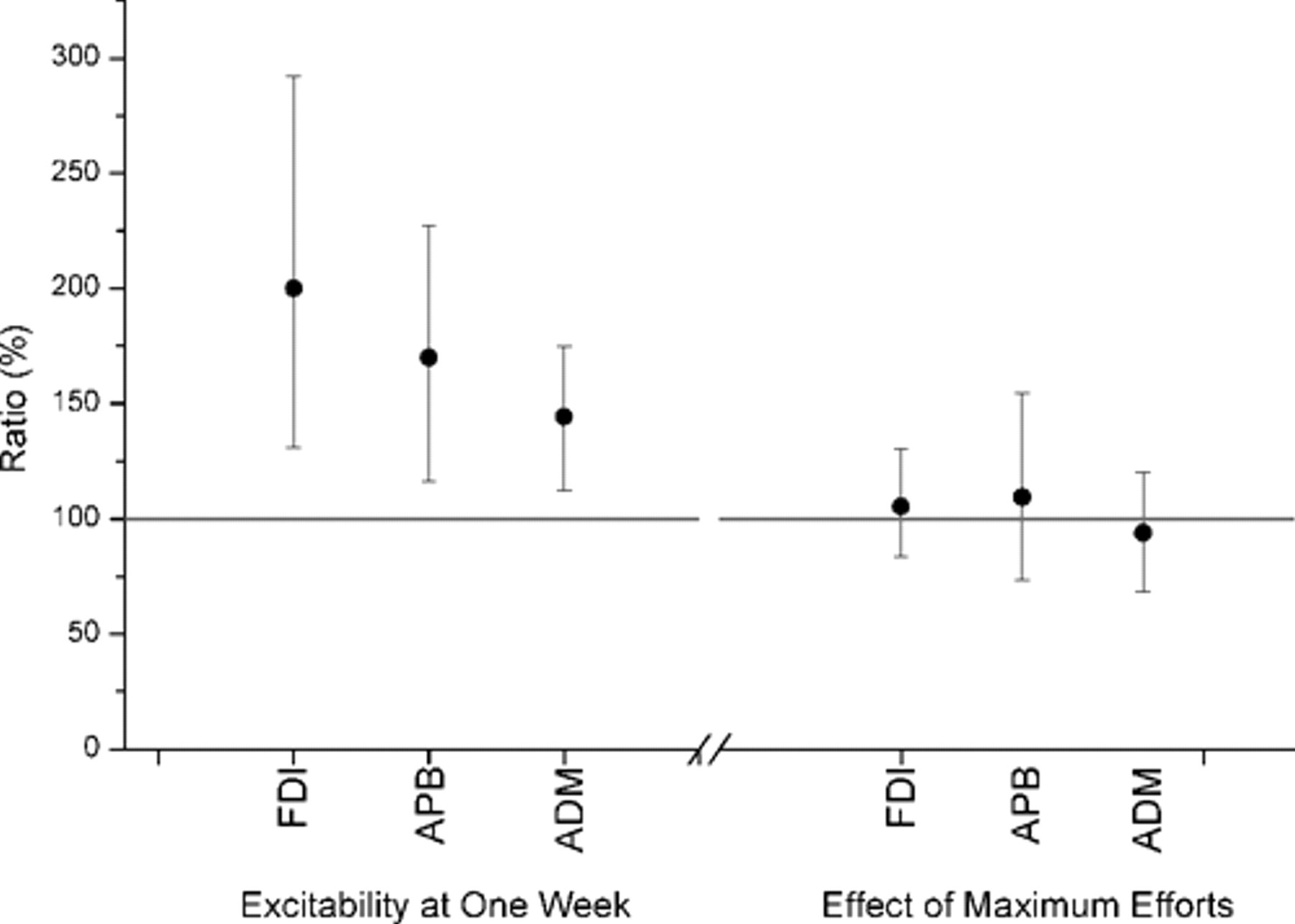
Effect of motor and stimulation task on excitability 1 week later and the immediate effect of maximum force efforts on excitability. Left: 95% confidence intervals demonstrate an increase in excitability for all 3 muscles (first dorsal interosseous [FDI], abductor pollicis brevis [APB], and abductor digiti minimi [ADM]) 1 week after performing the motor and sensory task. Right: 95% confidence intervals demonstrate that performing the maximal force efforts had no immediate statistically significant effect on excitability.
Correlations were performed between excitability changes in the MStask and alterations in excitability observed one week later (Figure 5). No significant correlations were observed with the Post 0 block of the MStask; however, for the Post 30 block both the APB (
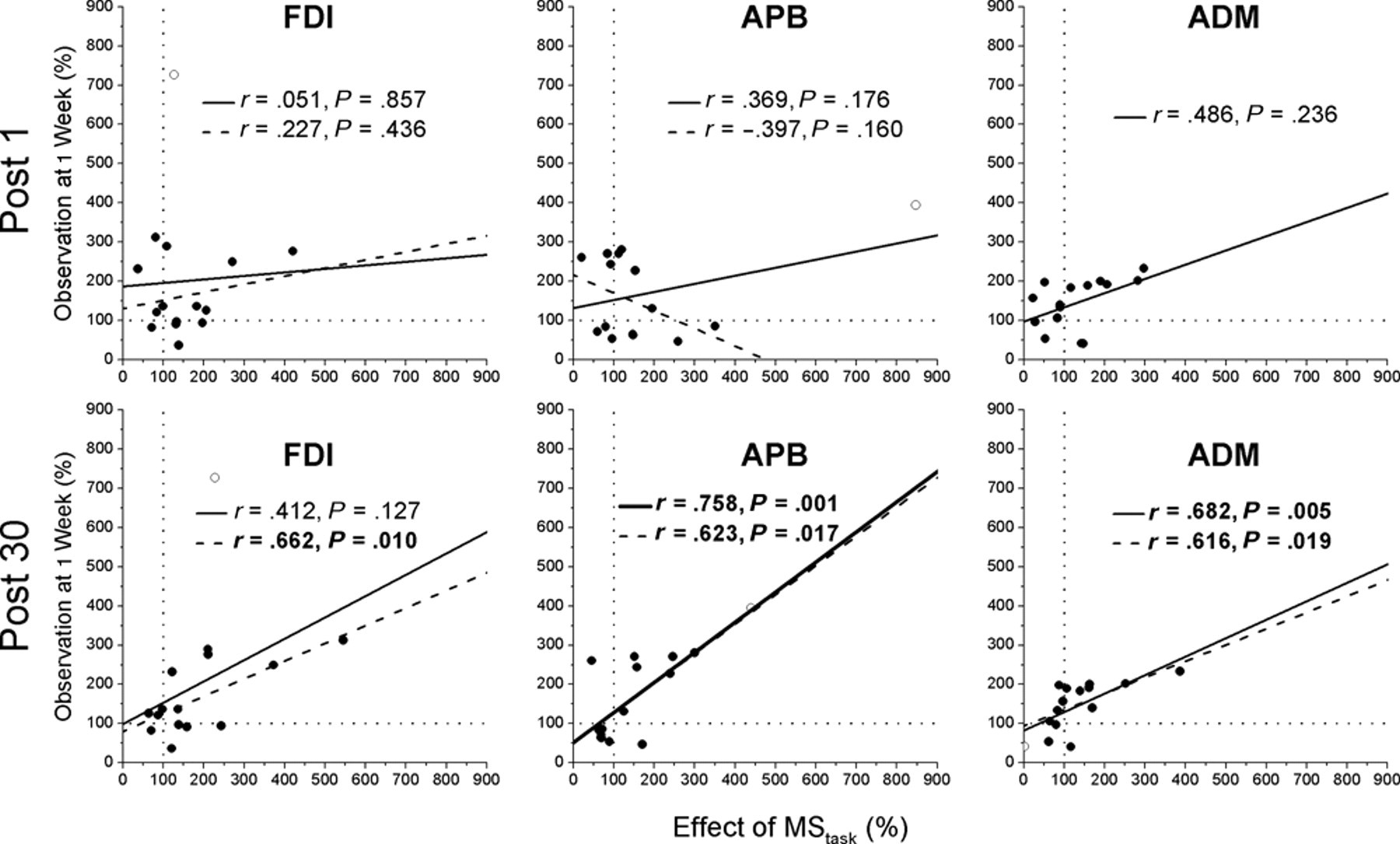
Correlation between effect of the MStask on excitability and excitability observed 1 week later: first dorsal interosseous (FDI), abductor pollicis brevis (APB), and abductor digiti minimi (ADM). No correlation was observed between the immediate effects of the MStask and excitability 1 week later for all muscles (top row). The effect of the MStask on excitability 30 minutes after task performance was correlated with excitability at 1 week (bottom row). Each plot shows Pearson’s correlation coefficient (
Short Latency Intracortical Inhibition/Intracortical Facilitation
All 3 tasks had minimal effects on SICI (Figure 6). A decrease in ICF was observed in the FDI immediately after the MStask −23.8%, 95% CI [−57.8%, 2.8%] and further decreased 30 minutes after task performance (37.0%, 95% CI [−69.4%, −4.7%]). The Mtask ICF confidence interval was consistent with a decrease immediately after task performance (−27.3%, 95% CI [−65.0%, 0.5%]), but this was not maintained 30 minutes later (9.0%, 95% CI [−31.2%, 60.9%]). ICF measurements in the APB muscle were mostly unaffected by task with a trend toward a decrease observed at Post 1 of the Mtask (−39.3%, 95% CI [−82.3%, −0.7%]), which ceased 30 minutes later (−25.3, 95% CI [−60.7%, 8.7%]). Mean and standard deviation of initial SICI and ICF ratios are presented in Table 1.
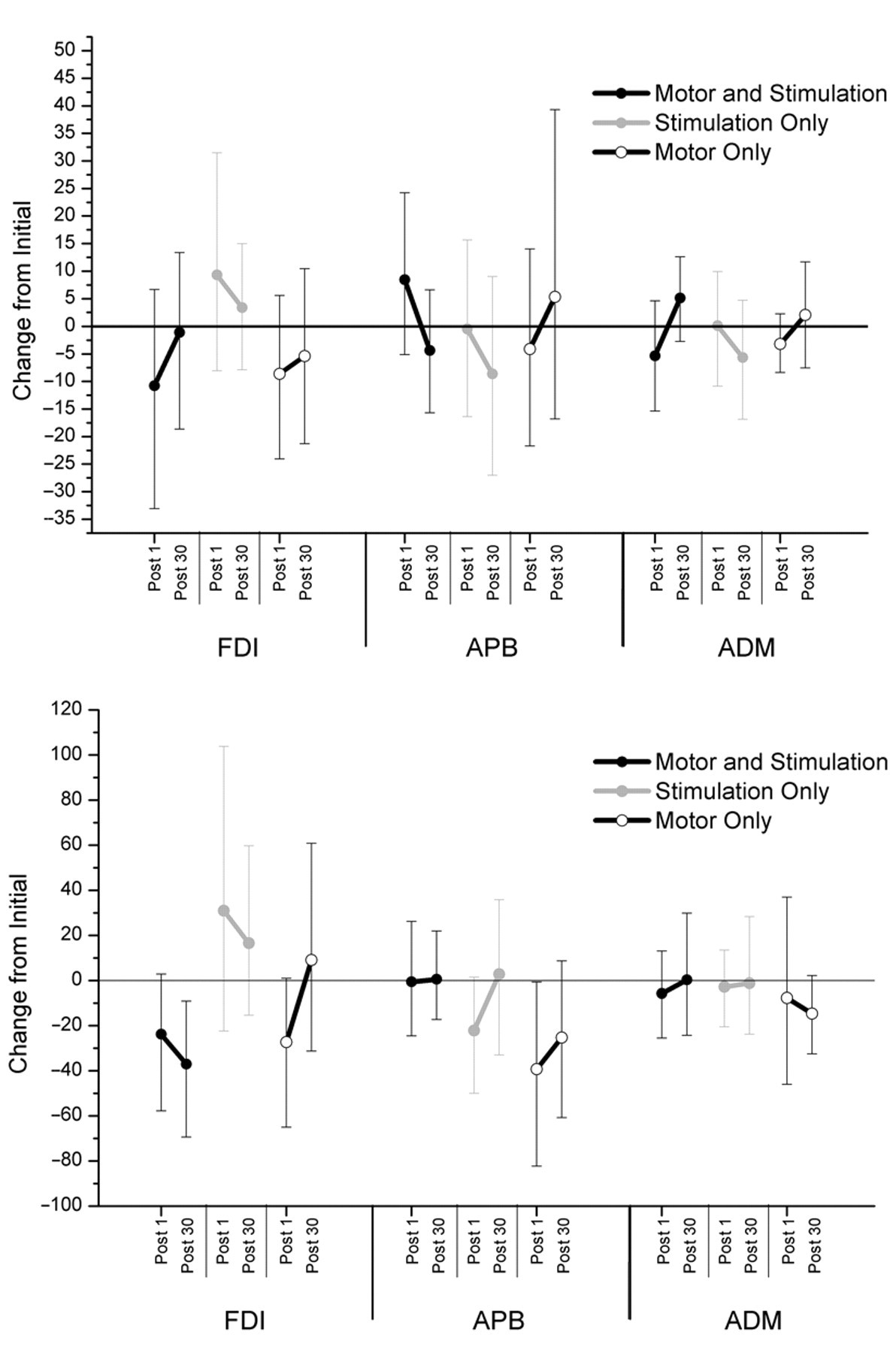
Effect of task on alterations in short latency intracortical inhibition (SICI; top panel) and intracortical facilitation (ICF; bottom panel). Figure shows 95% confidence intervals of the difference between measures of SICI of the presented time points and the initial for each task. Positive values indicate increased facilitation or less inhibition, whereas negative values indicate more inhibition or less facilitation.
Discussion
Prior work demonstrates that the corticospinal system adapts to the repeated performance of simple motor tasks or the repetitive application of peripheral or central stimulation. This experiment tested the effect of a bilateral combination of a motor and contralateral electrical stimulation task and compares it to the separate performances of the individual components. The results indicate that the combined MStask increased corticospinal excitability in the task-related muscles, but neither the Mtask nor Stask did. This increase was present immediately after task performance and was maintained 30 minutes later. In addition, the effect of MStask was correlated with measures of corticospinal excitability one week after performance of the MStask. These results have implications for understanding the interaction between sensory and motor components of behavior within the corticospinal system and offer possible therapeutic interventions aimed at modifying excitability and recovery of function after a neural insult.
Comparison Between the 3 Tasks
The adaptation observed in the MStask was specific to the muscles receiving electrical stimulation (FDI and APB). The effect of the MStask is consistent with a previous study that demonstrated that contractions on one side of the body can modify PAS induced changes in corticospinal excitability in the other cortex. 25 Many studies have documented the influence of unilateral task performance on the ipsilateral cortex. 4,8,36-38 This influence comprises a complex pattern of changes in excitability, 36 inhibitory networks, 39 and ICF. 38 The net result of this influence is thought to be increased facilitation and reduced inhibitory drive to homologous muscles, and a diffuse inhibition to proximate heterologous muscles. 40 These documented effects of unilateral motor tasks on the ipsilateral cortex could provide the basis for the interaction of the motor and stimulation task components within the right cortex.
The effect of the MStask may be analogous to the adaptations observed in PAS paradigms where the central stimulus is provided by TMS, whereas the peripheral stimulation is provided by electrical stimulation. 17,20,22,41,42
Increase in MEP Size 1 Week After the MStask
Measurements of corticospinal excitability were increased in all muscles 1 week after performance of the MStask. This increase was positively correlated with the delayed (by 30 minutes) measurements of the effect of the MStask but not with the immediate effects (Figure 5). This result is of considerable interest as inducing a long-term adaptation is critical for therapeutic potential. This result must be interpreted with caution as the experiment was not designed to compare absolute MEP size between tasks. Small changes in TMS coil position, electrode position, and skin conductance can lead to alterations in elicited MEP size. The overall influence of these effects should be negligible over a sample of 15 participants, however. An alternate explanation is that participant experience before the initial TMS block could have either decreased responses in the MStask or increased them in the Stask. The only difference in preparation between the 2 tasks was the 3 maximal force efforts performed in the MStask. Thus if performing maximum force trials depresses MEP measurements in the ipsilateral cortex, this could explain the observed increase from one session to the next. This hypothesis was tested in the Mtask by performing an additional TMS block before the max force efforts. No effect of the maximum force efforts was observed. Furthermore, neither variations in setup nor performance of the maximal force efforts would explain the observed correlations between MStask effects and alteration in excitability 1 week later.
The increased excitability of the ADM observed 1 week after the MStask (Figure 4) along with the correlation observed between weeks (Figure 5) suggests that the MStask may have had an immediate facilitatory effect on the ADM. The confidence interval that estimated this immediate effect of the MStask on ADM excitability was predominantly greater than 100%, but also included values <100% (Figure 3). Although this confidence interval suggests that the true effect of the task was likely facilitation, it is also consistent with the true effect being nil or even a slight inhibition. However, interpreting the aforementioned increased excitability and correlations observed one week after the MStask along with confidence interval of the immediate effect, suggests that the MStask may have had an immediate facilitatory effect on the ADM. A similar spread of increased excitability to muscles distant from the site of stimulation was observed in a study of the long-term effect of somatosensory stimulation 43 and noted in PAS paradigms. 17
Neither the Stask nor Mtask Altered Corticospinal Excitability
Neither the Stask nor the Mtask elicited changes in corticospinal output. The stimulation parameters used in the Stask, 30-Hz stimulation applied synchronously to 2 motor points, have proven effective in modifying TMS-induced excitability measures. 10,11 The duration of the stimulation may have prevented measurable changes from taking place. In the Stask, stimulation was applied with same time course that it was received in the MStask. A consequence of this pairing was stimulation durations of approximately 20 to 25 minutes compared with 1 hour plus that is more typical in stimulation paradigms. 10,16,29 Caution must be used in generalizing the results of the Stask as there may have been an order effect or a ceiling effect of the MStask that prevented further facilitation. The Mtask, performed by the right hand, also did not elicit any changes in corticospinal excitability of the right cortex. In contrast, repeated performance of unimanual ballistic finger movements, rather than the isometric task used in this study, has modified the excitability of the ipsilateral cortex. 4,8
Role of Attention
The direction and magnitude of attention has been shown to be a critical factor in tasks that induce cortical adaptation. 44-46 Furthermore, in peripheral stimulation paradigms, participants are often instructed to “attend to the stimulated hand.” 47 The lack of adaptation observed in the Stask is consistent with these results as attention was not explicitly focused on the stimulated hand. However, the adaptation observed in the MStask occurred despite a similar lack of attention to the peripheral stimulation. In the MStask the electrical stimulation was triggered by the motor task and thus provided stimulation temporally linked to the efferent motor command. This differs from the externally timed stimulation used in most adaptation-induced paradigms. We conjecture that attention may not be critical for adaption to occur in this scenario. If validated, this has implication for the design of therapeutic paradigms for participants who have reduced control of attentional resources, such as hemineglect.
Conclusion
A bilateral task that combines a motor task performed with one hand that triggers electrical stimulation of the other induces long-lasting changes in corticospinal excitability of the cortex receiving electrical stimulation. Application of just the stimulation or performance of only the motor task did not elicit any effect. The observed adaptations may result from the temporally linked motor and sensory components of the combined task, which in many ways recreates the relationship between the motor and sensory components of natural movements. Furthermore, this combination of tasks may be suitable for stroke survivors who retain minimal motor function and thus cannot practice tasks with their affected hand.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this work was provided for by the National Health and Medical Research Council of Australia grant number 435708.
