Abstract
Background. Neuroimaging studies of spinal cord injury (SCI) have mostly examined the functional organization of the cortex, with only limited focus on the subcortical substrates of the injury. However, thalamus is an important modulator and sensory relay that requires investigation at a subnuclei level to gain insight into the neuroplasticity following SCI. Objective. To use resting-state functional magnetic resonance imaging to examine the functional connectivity (FC) of thalamic subnuclei in complete SCI patients. Methods. A seed-based connectivity analysis was applied for 3 thalamic subnuclei: pulvinar, mediodorsal, and ventrolateral nucleus in each hemisphere. A nonparametric 2-sample t test with permutations was applied for each of the 6 thalamic seeds to compute FC differences between 22 healthy controls and 19 complete SCI patients with paraplegia. Results. Connectivity analysis showed a decrease in the FC of the bilateral mediodorsal nucleus with right superior temporal gyrus and anterior cingulate cortex in the SCI group. Similarly, the left ventrolateral nucleus exhibited decreased FC with left superior temporal gyrus in SCI group. In contrast, left pulvinar nucleus demonstrated an increase in FC with left inferior frontal gyrus and left inferior parietal lobule in SCI group. Our findings also indicate a negative relationship between postinjury durations and thalamic FC to regions of sensorimotor and visual cortices, where longer postinjury durations (~12 months) is associated with higher negative connectivity between these regions. Conclusion. This study provides evidence for reorganization in the thalamocortical connections known to be involved in multisensory integration and affective processing, with possible implications in the generation of sensory abnormalities after SCI.
Introduction
Traumatic spinal cord injury (SCI) results in permanent or progressive deterioration of motor, sensory, and autonomic functions leading to an overall decline in physical and psychosocial health. As of 2018, there are about 17 700 new incidences of SCI every year in the United States of which nearly 48% have incomplete tetraplegia, 20% have incomplete paraplegia, 20% have complete paraplegia, and 11% have complete tetraplegia (National Spinal Cord Injury Statistical Center. Birmingham, AL). Complete functional recovery in these individuals is rare and relies on the ability of the central nervous system to recover through spontaneous injury-induced and therapy-induced reorganization (plasticity). Reorganization is the complex process of molecular events contributing to the evolution of adaptive and maladaptive pathological changes at both the site of injury and the multi-systemic level of the brain.1-3
Cortical reorganization after SCI in humans is well documented using noninvasive neuroimaging techniques. Studies using magnetic resonance imaging (MRI) and task-based functional magnetic resonance imaging (fMRI) have shown an expansion in the somatotopic cortical representation of the body rostral to the level of injury in individuals with SCI.4-8 More recently, resting-state fMRI has emerged as a useful tool in measuring systems-level injury-induced alterations in the intrinsic functional architecture of the brain. Previous research using resting-state fMRI has reported a decrease in functional connectivity (FC) between cortical sensorimotor regions after SCI.9-15 Such alterations in resting-state properties have been shown by a previous study to hold significant implications for the prognosis of SCI at 6 months postinjury. 15 Findings from alternative approaches such as graph analysis provide evidence for preserved small-worldness with reduced local efficiency and greater network modularity and characteristic path length in the network architecture of SCI patients.10,16 However, the translation of cortical reorganization into clinical biomarkers is yet to be established. This gap in our knowledge of neuroplasticity and functional recovery may pertain to the subcortical substrates of SCI. Bridging this gap could provide better understanding of the adaptive or maladaptive changes in the brain.
Maladaptive outcomes after SCI such as neuropathic pain and dysesthesia are prevalent in ~53% of the population with SCI. 17 Neuropathic pain has long been associated with abnormal increase in the spontaneous and evoked activity of the mediodorsal nucleus of the thalamus. 18 Research also points to a reduction in gray matter volume, perfusion, and concentration of N-acetyl aspartate and γ-aminobutyric acid in thalamic subnuclei of individuals with SCI and neuropathic pain when compared with healthy controls (HC).19,20 Outside the role of the thalamus in pain, research suggests that reorganization within the thalamus and brain stem may be mediating body movement related cortical reorganization to recalibrate the integration of multisensory information appropriate to the altered body state.21-23 The ventrolateral nucleus of the thalamus is known to modulate the motor cortex for complex locomotion based on the integration of visual information and cerebellar feedback.24,25 Similarly, evidence from primate and human anatomical studies corroborate the possible roles of the thalamus (specifically, the pulvinar nucleus) in synchronizing spatially distinct cortical areas 26 allowing fast multisensory interplay through thalamic subnuclei 27 and even providing a feed-forward circuit through the cortico-thalamic-cortical relays.28-30 Given the imbalance in sensory input following SCI, the new perspective of the thalamus as both a relay and modulator in cortico-cortico processing leads us to hypothesize that a compound alteration must occur in the thalamocortical pathways of higher-order subnuclei that integrate multiple cortices after SCI. Reorganization in the connectivity of different subnuclei may, therefore, uniquely contribute to and compensate for the adaptive or maladaptive cortical changes observed after SCI.
With only suggestive evidence in the literature, the consequences of SCI on different thalamic subnuclei in humans remain mostly unexplored. Although, fMRI, diffusion tensor imaging, and other advanced MR techniques have enabled the parcellation of the human thalamus to understand the functions of individual subnuclei.31,32 Hence, the objective of this study is to investigate reorganization in the cortical connections of different thalamic subnuclei in individuals with complete SCI using fMRI. We used resting-state fMRI to compare the cortical FC of the motor subnuclei (bilateral ventrolateral [VL] nucleus) and 2 major higher-order subnuclei (bilateral pulvinar [PUL] and bilateral mediodorsal [MD] nucleus) between patients with complete SCI with paraplegia and HC subjects. Additionally, we investigated the effect of time since injury on the FC between thalamus and other brain regions.
Methods and Materials
Participants
Twenty-three healthy controls (33.5 ± 8.6 years, 12 females) and 20 right-handed individuals with complete SCI (35.2 ± 10.9 years, 4 females) at the thoracolumbar level were scanned as part of a larger study. A comprehensive neurological examination was performed on SCI patients as well as HC subjects to exclude accompanying neurological disorders of the peripheral and central nervous system, which could potentially act as confounding variables. Participants with SCI were evaluated based on the International Standards for the Neurological Classification of Spinal Cord Injury (ISNCSCI) published by the American Spinal Injury Association (ASIA). Pinprick and light touch were applied on the 28 dermatomes of each side of the body (left and right) to assess the level of sensory impairment. For every dermatome, a normal sensory response to pinprick or light touch was assigned a score of 2, impaired response was 1 and absent response was 0. Similarly, movement and range of motion was tested in 10 myotomes on each side of the body. For every myotome, muscle function was graded as 0 for paralysis; 1 for visible contraction; 2 for active movement and full range of motion (ROM) without gravity; 3 for active movement and full ROM against gravity; 4 for active movement, full ROM, and moderate resistance; and 5 for normal movement, ROM, and resistance. Neurological level of injury was defined as the caudal-most level of the spinal cord with intact sensation and movement against gravity. The injury was classified as complete when the individual had no residual sensation in the sacral segments of the spinal cord. Additionally, none of the individuals with SCI or HC subjects had concomitant psychiatric disorders or a history of brain trauma. The study was approved by the Ethics Committee of the Third Hospital of Hebei Medical University, China, and all participants gave their written consent to participate in the study.
Imaging Parameters
High-resolution structural images were acquired on a 1.5-T Siemens MAGNETOM Avanto scanner using magnetization-prepared rapid gradient echo sequence (MPRAGE). The imaging parameters for MPRAGE were as follows: field of view (FOV) = 240 mm, 512 × 512 matrix, repetition time (TR) = 2300 ms, echo time (TE) = 2.91 ms, flip angle = 90°. The whole brain was imaged in the sagittal configuration where 176 slices were collected, and each slice was 1 mm thick. The spatial resolution of all the anatomical scans was 1 mm × 0.469 mm × 0.469 mm. The resting-state fMRI scans were acquired using echo planar imaging (EPI) sequence with the following imaging parameters: FOV = 240 mm, 64 × 64 matrix, TR = 2000 ms, TE = 27 ms, and flip angle = 90°. The whole brain was scanned in an axial configuration where 20 slices were collected, and each slice was 6 mm thick. The spatial resolution was 3.75 mm × 3.75 mm × 6 mm for all functional scans. During the scan duration, the subjects were instructed to remain motionless and avoid falling asleep. A total of 195 volumes were collected over a scanning period of 390 seconds.
Data Processing
The data processing was performed using Statistical Parametric Mapping 12 toolbox (http://www.fil.ion.ucl.ac.uk/spm/) within the MATLAB environment (Mathworks Inc, Natick, MA, USA). Before preprocessing, the first 5 time points of the resting-state fMRI scan were removed to reduce transient scanner artifacts and the origin of the functional and anatomical images of all participants was manually reoriented to the anterior commissure of the brain. Preprocessing workflow included the following procedures. First, head motion correction using a least squared approach and 6 parameters (rigid body) spatial transformation with respect to the first image of the scan was performed. One individual with SCI and 1 HC had more than 2 mm maximum head motion in any direction and were therefore removed from subsequent analysis. Nineteen individuals with SCI (34.7 ± 10.9 years, 4 females) and 22 HC (33.4 ± 8.8 years, 11 females) were used for the remainder of the analysis. After eliminating subjects, the mean motion in any direction was 0.041 mm in HC and 0.014 mm in SCI group. Additionally, the average framewise displacement of the 2 groups was also comparable at 0.0519 mm in HC and 0.0577 mm in SCI group. Second, co-registration of anatomical image to the mean functional image of each subject was performed. Third, segmentation of anatomical images into gray matter, white matter (WM), and cerebrospinal fluid (CSF) tissue probability maps was conducted. Fourth, spatial normalization was done using deformation vectors obtained from the segmentation procedure of each subject and resampling to isotropic voxel size of 3 × 3 × 3 mm3. Fifth, regression of the average WM and average CSF time series extracted from voxels of WM and CSF probability maps with probability >0.98 was performed. Sixth, regression of head motion noise using Friston 24-parameter model was performed (6 head motion parameters, 6 head motion parameters from previous time point, and their 12 corresponding quadratic parameters). 33 Lastly, spatial smoothing was conducted on the data using a Gaussian kernel of 8 mm full-width at half-maximum followed by temporal bandpass filtering between 0.01 to 0.1 Hz using a second order Butterworth filter.
Image Analysis
Seed-Based Analysis
Thalamocortical FC maps were generated using seed-based analysis technique. Six 3-dimensional spherical seeds of 8 mm radius were generated using MNI coordinates of bilateral PUL (±15, 31, 5), bilateral MD (±6, 16, 9) and bilateral VL (±15, 13, 9) nucleus previously described by Zou et al. 34 A comparison of the spherical seeds with the masks of each of the subnuclei from stereotactic atlas of human thalamus (Morel atlas) is demonstrated in the Supplementary Material (Figure S1). In general, the spherical seeds used in this study are larger but greatly overlap with the stereotactic definition of the different thalamic subnuclei. The seeds of left/right PUL, MD, and VL defined spherically are each 81 voxels in size whereas the subnuclei masks based on the Morel atlas constitute of 84/89, 44/49, and 40/41 voxels, respectively. Of note, the spherical seeds of MD and VL have 18 out of the 81 voxels overlap in the left-right direction. Average time series of each of the 6 spherical thalamus seeds were extracted as the reference time series for each of the participants. Using AFNI’s 3dfim+ function, the whole brain voxel-wise connectivity map of all 6 seeds in the thalamus were computed for all participants. 35 Each of the connectivity maps were then converted to z-score maps using r to z Fisher transformation. For next steps of the analysis a 4D dataset for each of the 6 seeds was generated by concatenating the z maps of all subjects.
Statistical Analysis
For each of the 6 thalamic seeds, FSL’s randomise was used to perform nonparametric voxel-wise 2-sample t-test using 5000 permutations to assess connectivity differences between the 2 groups (FMRIB Analysis Group, Oxford, UK). The statistical design included gender and age as covariates of no interest. Significant effects were obtained at α = .05 by thresholding the voxel-based probability maps (output of Randomise) at uncorrected (uncorr) P < .005 followed by a Monte Carlo simulation–based cluster-extent thresholding to correct for multiple comparisons. The Monte Carlo simulation–based cluster-extent thresholding was performed using AFNI’s 3dClustSim with the autocorrelation function to compute cluster-size threshold for each of the 6 thalamic seeds. Furthermore, to exclude regions outside of the brain from statistical analysis, a spatial mask comprising of only the voxels present across all subjects was used as an inclusive mask.
Effect of Injury Characteristics on FC of Thalamus
For each of the 6 thalamic seeds, FSL’s randomise was used to model the effect of injury duration on FC measures utilizing a nonparametric voxelwise estimation using 5000 permutations. The general linear model design included duration of injury as a covariate of interest, and gender and age as covariates of no interest. Significant effects were obtained at α = .05 by thresholding the voxel-based probability maps at voxelwise uncorr-P < .005 followed by cluster-extent based multiple comparison correction described in the previous section.
The aforementioned analysis was repeated to investigate the relationship between FC measures of the 6 thalamic subnuclei and severity of injury (total sensory and motor function computed based on ASIA scale) in the SCI group.
Results
Functional Connectivity of Thalamic Subnuclei in Both SCI and Control Groups
The average age of the SCI group is 34.7 ± 10.9 years (4 females) with an average injury duration of 194 ± 187 days; the average age of HC group is 33.4 ± 8.8 years (11 females). Chi-square test showed no significant differences in gender distribution between the HC and SCI groups (χ2 = 3.68, f = 1, P = .055). Table 1 summarizes the demographical information of the patients in detail.
Demographics of the SCI Patients. a
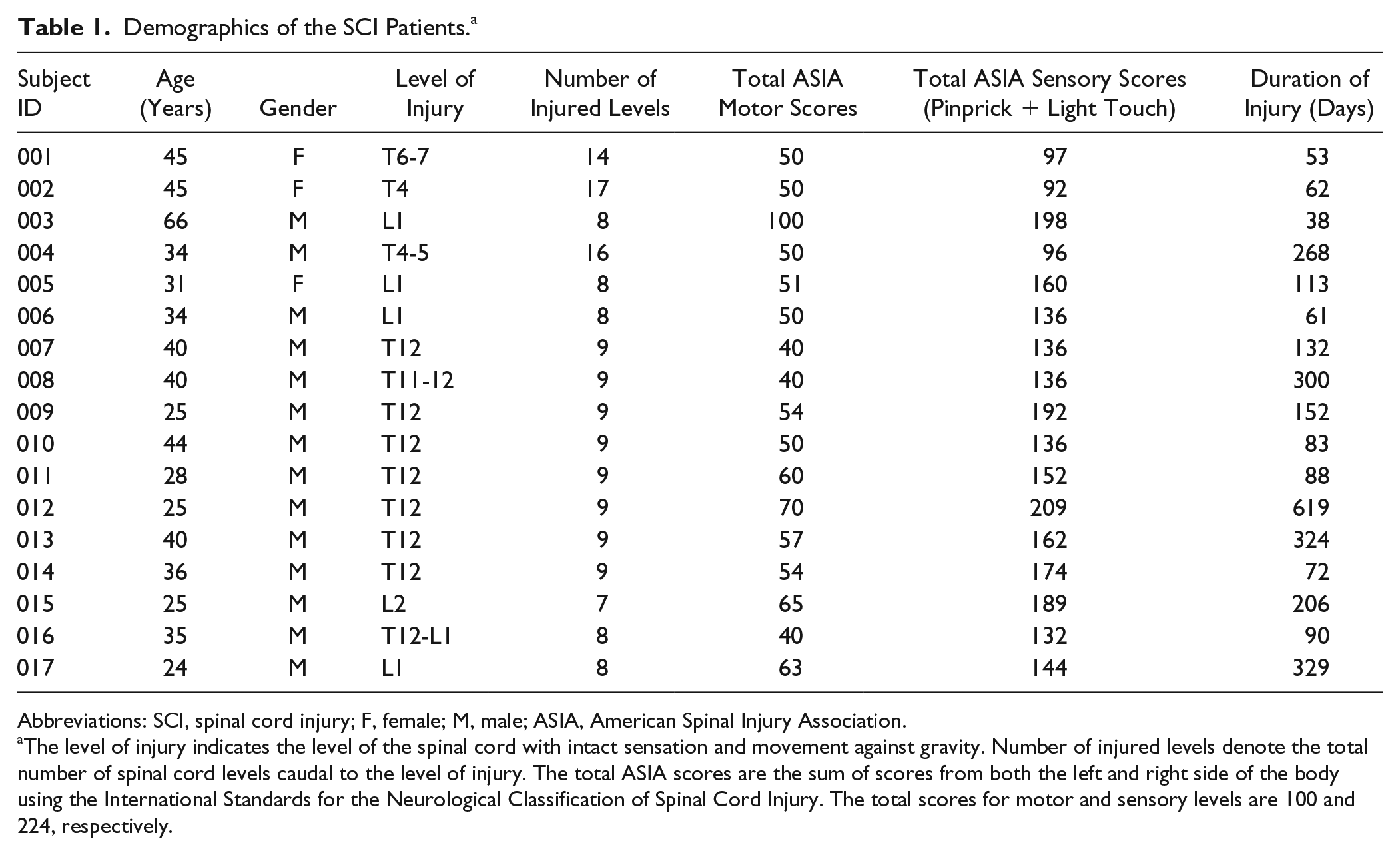
Abbreviations: SCI, spinal cord injury; F, female; M, male; ASIA, American Spinal Injury Association.
The level of injury indicates the level of the spinal cord with intact sensation and movement against gravity. Number of injured levels denote the total number of spinal cord levels caudal to the level of injury. The total ASIA scores are the sum of scores from both the left and right side of the body using the International Standards for the Neurological Classification of Spinal Cord Injury. The total scores for motor and sensory levels are 100 and 224, respectively.
As seen in Figure 1, the mean FC of left and right PUL nucleus in both groups are localized to areas of bilateral PUL nucleus and bilateral hippocampus with only marginal extension to precuneus and posterior cingulate cortex regions. The mean FC of MD nucleus in HC subjects is spatially widespread, where it extends from regions of bilateral thalamus to anterior cingulate cortex, bilateral middle temporal gyrus, middle cingulate gyrus, and precuneus. However, the mean FC of MD nucleus in SCI group appears to be spatially limited to regions of bilateral thalamus. Likewise, the mean FC of VL nucleus in HC encompasses regions of bilateral thalamus, basal ganglia, anterior cingulate cortex, bilateral middle frontal gyrus, insula, superior temporal gyrus, and middle cingulate cortex. The mean FC of VL nucleus in SCI group was sparse with connections mostly extending to bilateral thalamus, basal ganglia, and cingulate gyrus.
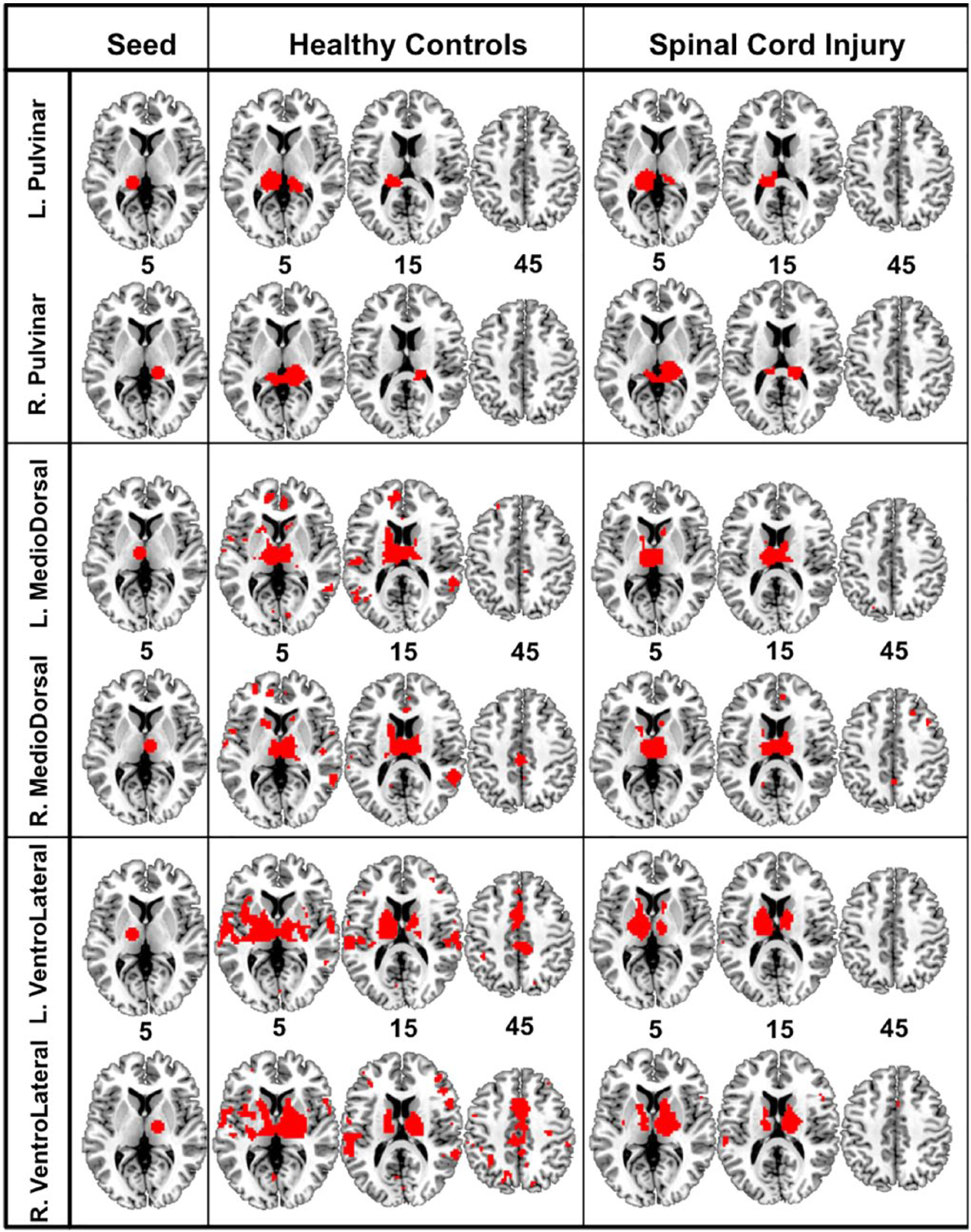
Mean group connectivity map of left and right pulvinar nucleus (top), left and right mediodorsal nucleus (middle) and left and right ventrolateral nucleus (bottom) for healthy controls (HC) and spinal cord injury (SCI) group. Functional connectivity (FC) maps are the result of mean group effect thresholded at voxel-based familywise error corrected at P < .05 with cluster-volume threshold = 270 mm3. Seed column on the left illustrates the thalamic seeds. The number indicates the z coordinates of the slices shown.
Statistical tests comparing the voxelwise connectivity maps between SCI and HC groups resulted in significant effects in four out of the 6 seeds investigated (shown in Figure 2). Two-sample t-test at false discovery rate (FDR)-P < .05 revealed a widespread increase in the FC of left PUL nucleus to regions of left inferior parietal lobule and left inferior frontal gyrus–pars triangularis. Right PUL nucleus conversely showed no significant difference between the 2 groups. Comparing the connectivity of MD nucleus between the 2 groups revealed decreased connectivity of left MD with regions of left anterior cingulate cortex and right superior temporal gyrus in SCI group (shown in Figure 2c). Similarly, right MD exhibited a decrease in connectivity with right rolandic operculum extending to right superior temporal gyrus in SCI group. Furthermore, the results showed a decrease in functional connectivity of left VL nucleus with left superior temporal gyrus in SCI group.
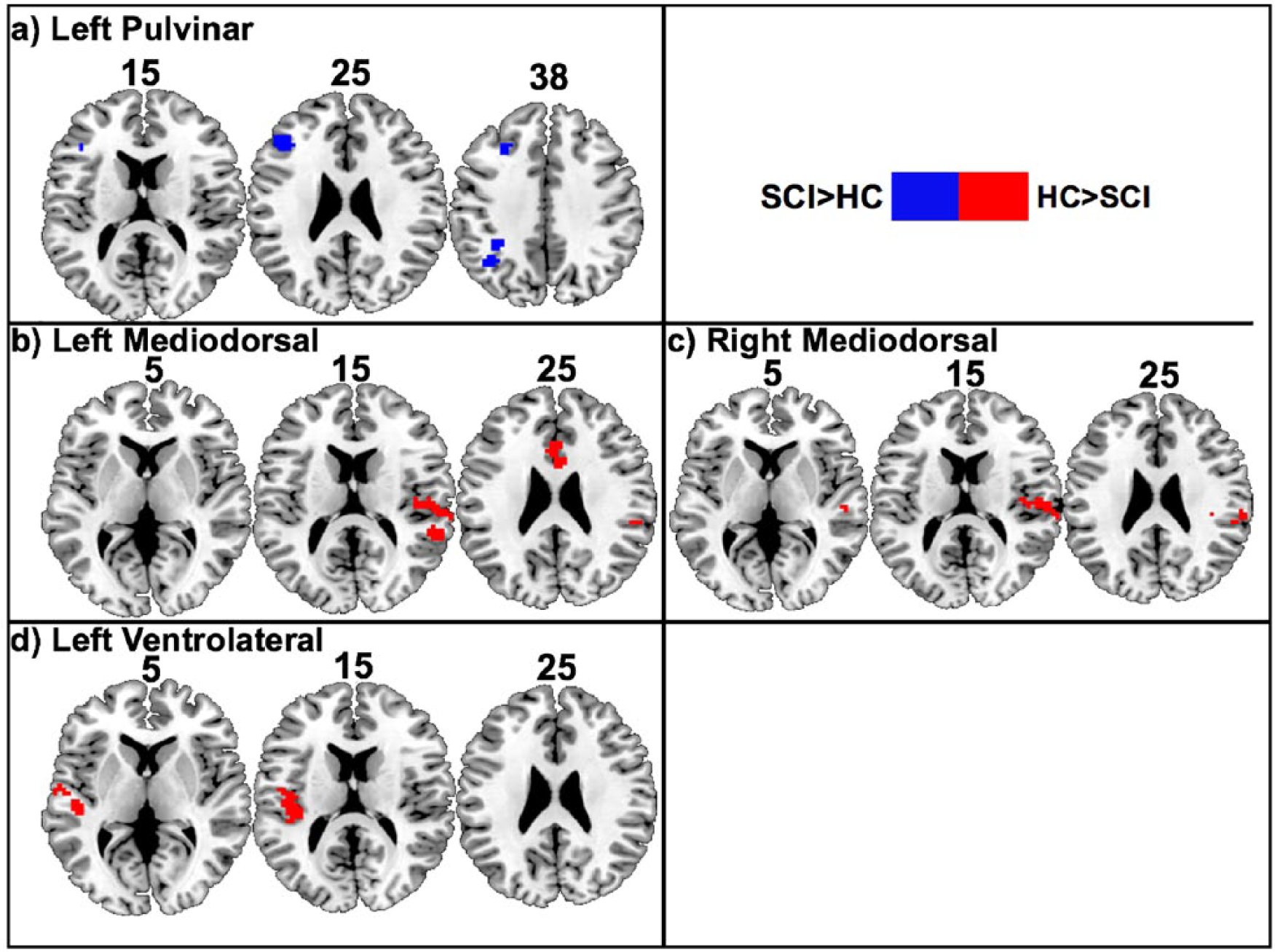
Result of 2-sample t-test displaying regions with altered functional connectivity (FC) to the different thalamic subnuclei using 8-mm spherical seeds in patients with spinal cord injury (SCI) when compared with health controls (HC). (a) Two clusters with peak statistical difference at the left inferior parietal lobule and left inferior frontal gyrus pars triangularis exhibits increased FC with left pulvinar (PUL) nucleus in SCI at uncorrected P < .005 with cluster-volume threshold = 2292.3 mm3. (b) Two clusters with peak statistical difference at the right superior temporal gyrus and left anterior cingulate cortex exhibit decreased FC with left mediodorsal (MD) nucleus in SCI at uncorrected P < .005 with cluster-volume threshold = 2335.5 mm3. (c) One cluster with peak statistical difference at the right superior temporal gyrus exhibit decreased FC with right MD nucleus in SCI at uncorrected P < .005 and cluster-volume threshold = 2348 mm3. (d) One cluster with peak statistical difference at the left superior temporal gyrus exhibit decreased FC with left ventrolateral (VL) nucleus in SCI at P < .005 and cluster-volume threshold = 2313 mm3. Red indicates regions that are decreased in SCI in comparison with HC; blue indicates regions that are increased in SCI in comparison with HC. The slice index represents the z coordinates of the slices shown. Note: FC analysis of right PUL nucleus and right VL nucleus exhibited no significant differences between the 2 groups (P > .05).
A list of all the regions with significantly altered FC to the different thalamic subnuclei in the SCI group is summarized in Table 2. The average Pearson’s correlation values (r) of these connections are further illustrated in Figure 3. The HC group has a mean positive FC for connections of MD and VL nucleus while SCI group exhibits positive but reduced connectivity in these regions. Conversely, the connectivity strength of PUL nucleus with regions of inferior frontal gyrus and inferior parietal lobule in SCI was positive when compared with a negative connectivity in HC.
List of Regions With Altered FC Differences With Thalamus. a
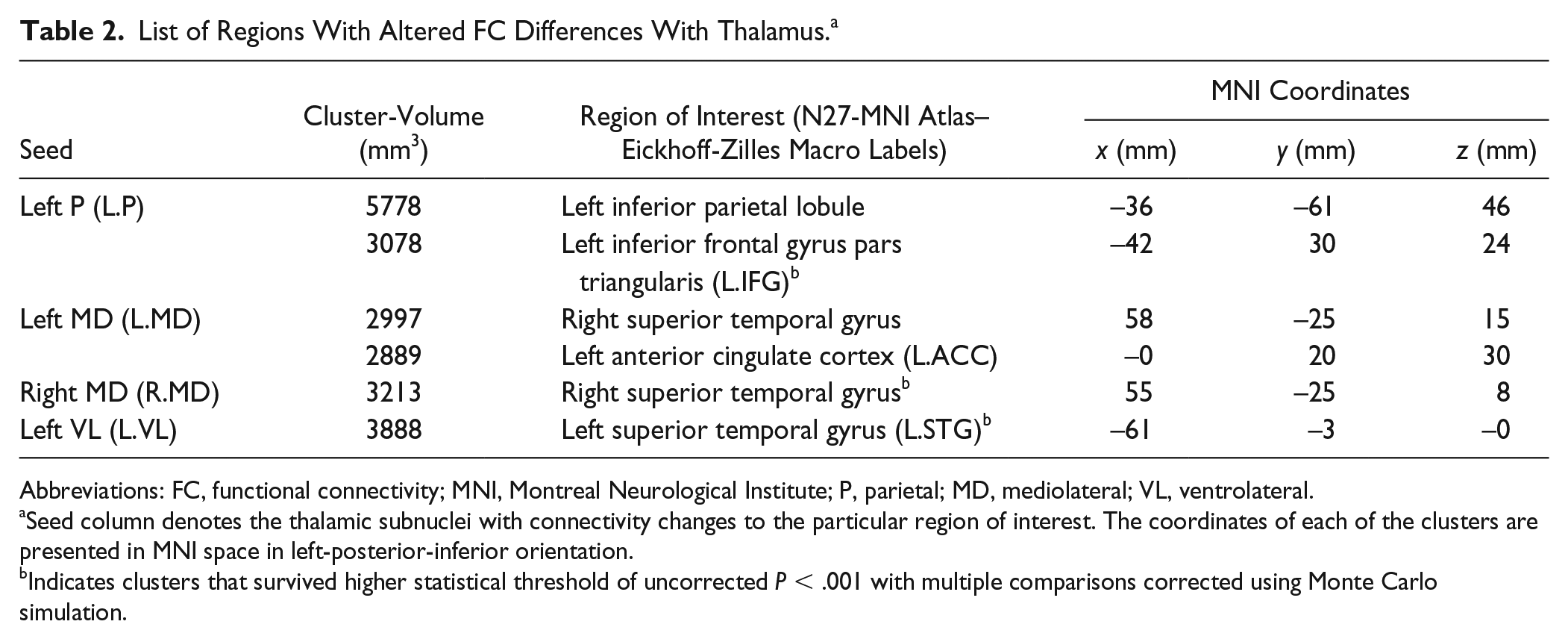
Abbreviations: FC, functional connectivity; MNI, Montreal Neurological Institute; P, parietal; MD, mediolateral; VL, ventrolateral.
Seed column denotes the thalamic subnuclei with connectivity changes to the particular region of interest. The coordinates of each of the clusters are presented in MNI space in left-posterior-inferior orientation.
Indicates clusters that survived higher statistical threshold of uncorrected P < .001 with multiple comparisons corrected using Monte Carlo simulation.
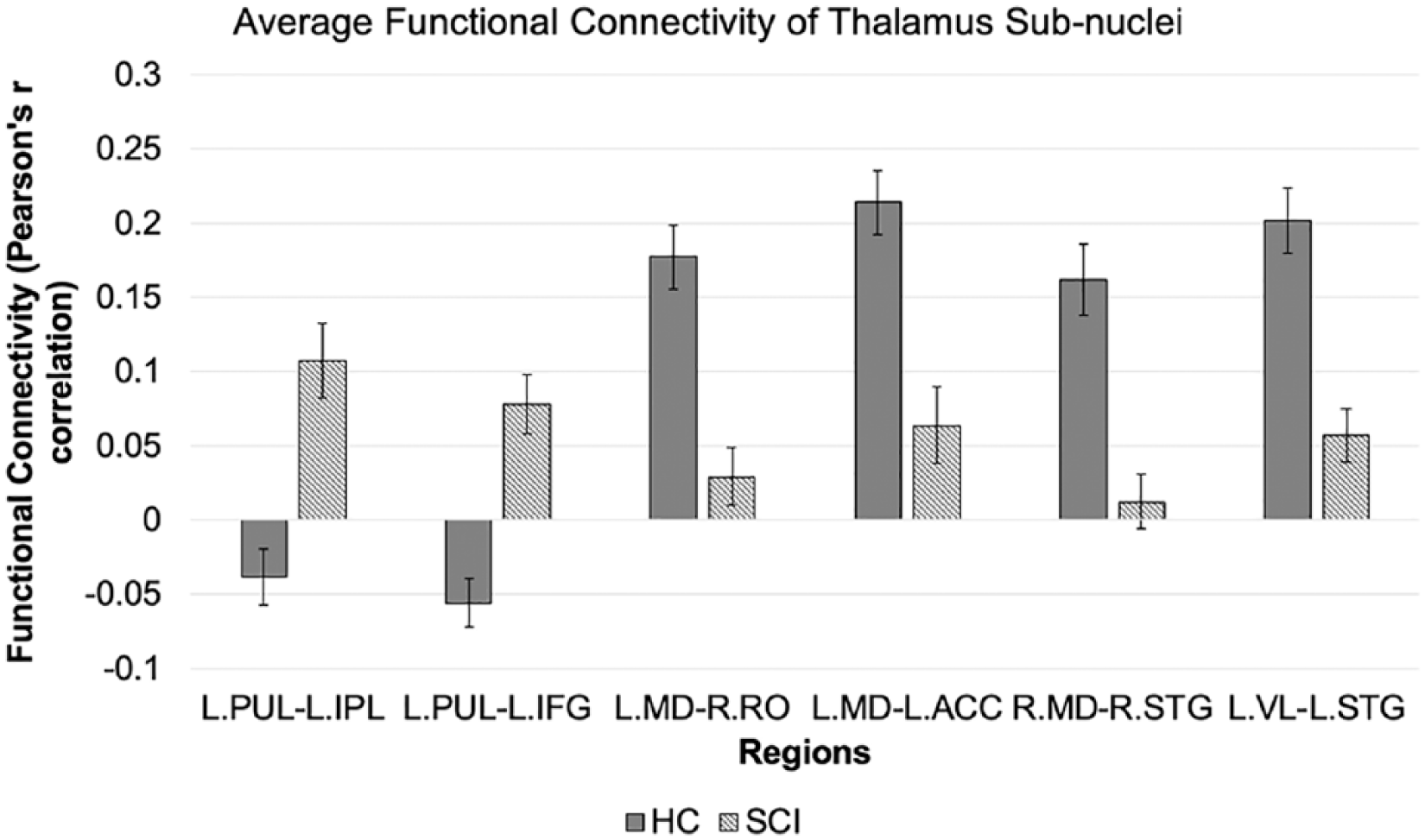
Average functional connectivity (FC) measures of the regions with significantly altered connectivity to the different thalamic subnuclei in health controls (HC) and spinal cord injury (SCI) groups. Statistical significance was computed at uncorrected P < .005 with cluster-based thresholding using Monte Carlo simulation. Error bars indicate standard error of the mean.
Additionally, 2-sample t-test results comparing seed-based connectivity maps obtained using stereotactic atlas masks (Morel atlas) and spherical seeds with 6 mm radius as seeds are provided in Figure S2 and Figure S3 of the Supplementary Material. Reducing the size of the spherical seed from 8 to 6 mm or alternatively using masks from atlas did not affect the overall effect in connectivity changes between the 2 groups, where PUL nucleus is associated with increased connectivity to frontoparietal regions and MD nucleus is associated with decreased connectivity to salient network regions. However, there exists some differences in the clusters that survive multiple comparison correction (Figure 2 vs Figure S2 vs Figure S3).
Effect of Injury Characteristics on FC of Thalamic Subnuclei
Models of the mean effect of injury duration on FC of the thalamus in SCI resulted in statistically significant effect in all the 6 subnuclei studied. Figure 4a presents the regions whose FC with different thalamic subnuclei (indicated in different colors) are significantly associated to the duration of injury in individuals with SCI. In general, as shown in Figure 4b, the FC of the different thalamic subnuclei to regions of the visual cortex, premotor cortex, and secondary somatosensory cortex appears to be dynamic and progressively decreasing with time following injury in patients with SCI. The specific coordinates and cluster information of the regions are further listed in Table S1 of the Supplementary Material.
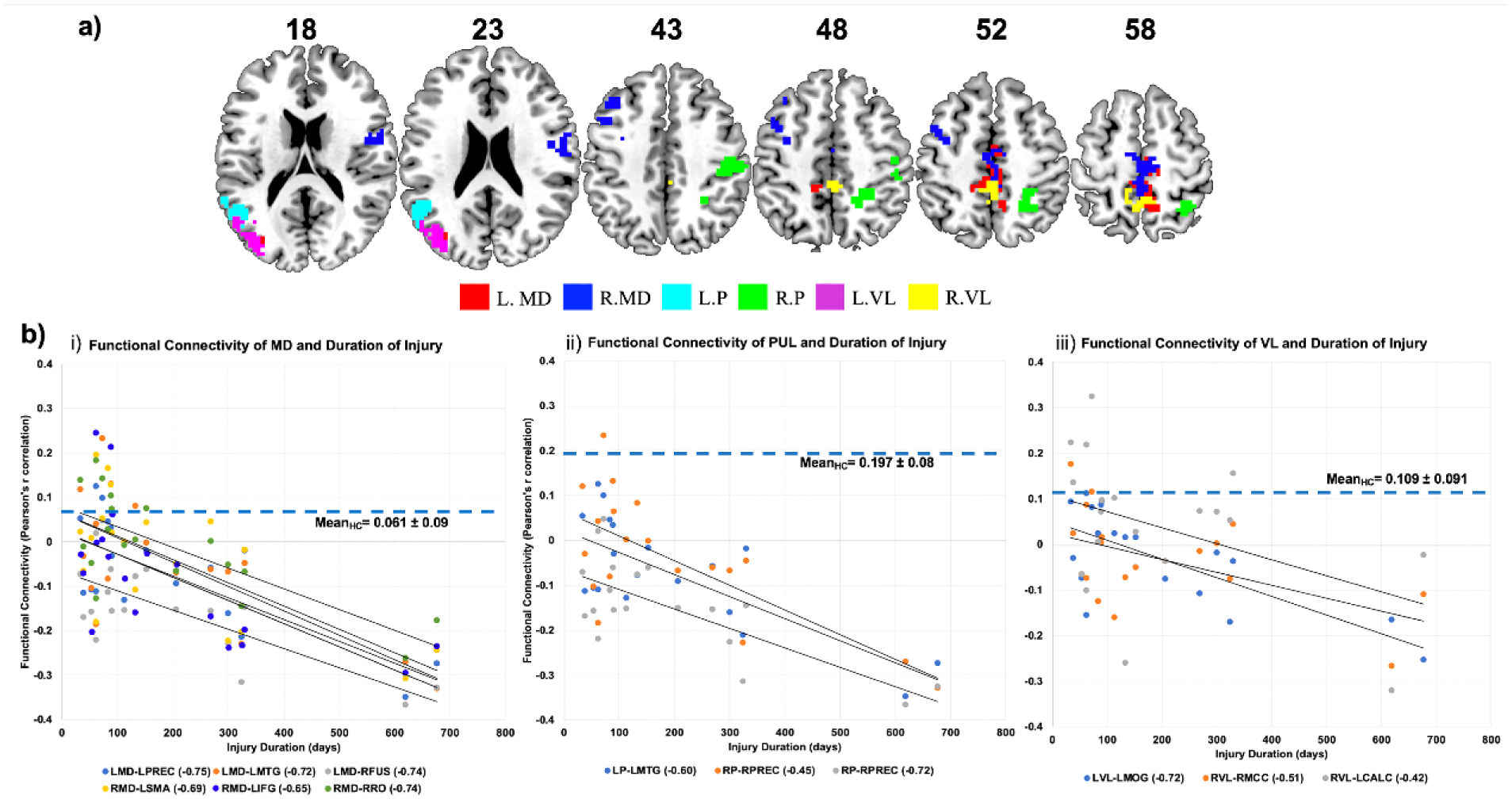
Effect of duration of injury on the functional connectivity (FC) of the different thalamic subnuclei in the spinal cord injury (SCI) group: (a) Result of t-test displaying clusters whose FC to the thalamic subnuclei is significantly correlated to the duration of injury. Red indicates clusters whose FC to the left mediolateral (MD) nucleus is significantly associated with the injury duration in the SCI group at uncorrected P < .005 with cluster-volume threshold = 2335.5 mm3; similarly, blue indicates clusters whose FC to the right MD nucleus is significantly associated to the injury duration in SCI group at uncorrected P < .005, with cluster-volume threshold = 2349 mm3, light blue indicates clusters whose FC to left pulvinar (PUL) nucleus is significantly associated to injury duration in SCI group at uncorrected P < .005, k = 2292.3 mm3, green indicates clusters whose FC to the right PUL nucleus is significantly associated to the injury duration in SCI group at uncorrected P < .005, with cluster-volume threshold = 2257.2 mm3, violet indicates clusters whose FC to left ventrolateral (VL) nucleus is significantly associated to injury duration in SCI group at uncorrected P < .005, with cluster-volume threshold = 2313.9 mm3, and yellow indicates clusters whose FC to the right VL nucleus is significantly associated with the injury duration in SCI patients at uncorrected P < .005, with cluster-volume threshold = 2300.4 mm3. The number represents the z coordinates of the slices shown. (b) Negative relationship between duration of injury and FC measures of (i) MD nucleus, (ii) PUL nucleus, and (iii) VL nucleus with clusters shown in a). Pearson’s r correlation values of the relationship between FC and duration of injury is given in parentheses in the legend of each plot next to the region of interest. Dotted lines in the scatter plot indicates the mean and standard deviation of functional connectivity (Pearson’s r measure) between all significant clusters and thalamic subnuclei (MD, PUL, or VL) in HC group. LMD, left MD; RMD, right MD; LVL, left VL; RVL, right VL; LPUL, left PUL, RPUL, right PUL; LMTG, left middle temporal gyrus; RFUS, right fusiform gyrus; LPREC, left precentral gyrus; RRO, right rolandic operculum; LIFG, left inferior frontal gyrus; LSMA, left supplementary motor area; RPREC, right precentral gyrus; LMOG, left middle occipital gyrus, RMCC, right middle cingulate cortex; LCALC, left calcarine gyrus.
Voxelwise 1-sample t tests examining the relationship between FC of thalamic nucleus with sensory-motor scores and injury level revealed significant correlation between sensory-motor scores and the different thalamic subnuclei in the left hemisphere. A positive relationship was observed between the motor scores and FC of left MD nucleus with right parahippocampal gyrus and left PUL with right supplementary motor area. An opposite (negative) relationship was observed between the sensory scores (pinprick + light touch) and FC of left VL nucleus with right lingual gyrus. Significant clusters and their relationship with ASIA sensory-motor scores are illustrated in Figure S4 of the Supplementary Material.
Discussion
Classically, the thalamus is seen as a major relay switch while the modern view suggests a modulatory and an indirect transthalamic role with numerous pathways between the cortex, thalamic subnuclei, and lower motor centers.36,37 Identifying the functional alterations of the thalamus at a subnuclei level in response to an imbalance in cortical computation and peripheral reactions (as in the case of SCI) could provide insight into the neuroplasticity following SCI. Furthermore, previous research in SCI suggests an association between thalamus dysfunction and chronic pain.18,19,38 Therefore, in this study, we performed a voxelwise seed-based analysis to identify reorganization in the connectivity of 3 major thalamic subnuclei (PUL, MD, and VL nucleus) after complete SCI. By estimating resting-state FC of the thalamus to the whole brain, we identified specific alterations in connectivity of left PUL nucleus, bilateral MD nucleus, and left VL nucleus. Furthermore, the FC of bilateral MD nucleus, bilateral PUL nucleus, and bilateral VL nucleus to regions of the visual cortex, motor cortex, and secondary somatosensory cortex was found to have a negative relationship with injury duration.
FC findings indicated decreased connectivity between the bilateral MD nucleus and right superior temporal gyrus and between left MD nucleus and left anterior cingulate cortex in the SCI group. We reproduced these alterations using spherical seeds of 6 mm radius and stereotactic thalamus atlas as alternative masks. The MD nucleus consisting of medial and lateral subdivision is known to influence cognitive processes, including working memory, executive function, emotional perception, and decision making.39,40 Rodent and nonhuman primates show extensive reciprocal connections between the MD nucleus and prefrontal cortex with ablation of one resulting in dysfunction of the other.41-44 Furthermore, some literature suggests that sympathetic and parasympathetic afferents from the tissues in the body project to the insular cortex and dorsal anterior cingulate cortex (ACC) through the ventromedial and MD nucleus, respectively. 45 Taken together, disconnection of autonomic afferents from lower motor centers to the thalamus in SCI could explain the hypoconnectivity of the MD nucleus with the ACC, and insula. These apart, MD nucleus may also be involved in the emotional-affective processing of pain. 18 Dorsal ACC, insula, striatum, amygdala along with the prefrontal cortex constitute the salience network responsible for integrating internal sensory information (interoception and visceromotor signals) with external sensory information to generate meaningful neural responses, including emotional awareness. 46 The salience network is also posited as the pain matrix of the brain where the occurrence of central pain condition is considered as a behavioral response to an event of imbalance in the internal and external sensory information or bodily states. 47 But since we do not have information regarding the pain status of the individuals with SCI, no direct conclusion of the results can be drawn regarding pain. Besides, impairment of emotional perception in SCI although subtle is greatly debated and overlooked due to the overbearing psychological consequence of a life-altering injury. Emerging evidence in SCI indicates impaired emotional perception 48 and enhanced activity in dorsal ACC, temporal gyrus, and periaqueductal gray regions during an emotional fear conditioning task. 49 Therefore, if the same cortical regions represent the visceral, emotional, and pain states of the body, an alteration in connectivity of MD thalamus to these cortical regions could be associated to both emotion and pain processing deficits observed in SCI.
Results also revealed an increase in FC between left PUL nucleus and regions of left inferior frontal gyrus and left inferior parietal lobe in the SCI group. The PUL nucleus is a higher order nucleus that receives the majority of its inputs from the cortex and sends efferent connections to the cortex. 50 It is also an important component of the visual attention network with connections mostly extending to regions of multisensory association cortices. 51 For example, PUL nucleus connects to posterior parietal lobe and frontal gyrus for visuosomaesthetic processing and superior temporal gyrus for visuoauditory processing. Besides, the PUL is believed to regulate and facilitate large-scale synchronization of cortical regions through cortico-pulvinar-cortical pathways. 52 Therefore, a lack of afferents from lower motor centers could create an imbalance in sensory weighting, initiating a compensatory increase in synchronization between the multisensory association cortices through PUL. After all, the accuracy of spontaneous or goal-directed movements depends on the accurate and continuous processing of visual information updated by the body’s spatial representation through proprioception. By the same token, SCI patients rely more on visual, vestibular, and auditory sources for spatial navigation. Hence, an increase in connectivity between the left PUL and the regions of the frontoparietal network in SCI patients could be a compensatory mechanism contributing to spatial awareness and body image in individuals with a lack of proprioceptive feedback.
The third subnuclei left VL nucleus exhibited decreased connectivity to left STG in SCI group. The VL nucleus along with other ventral subnuclei constitute the motor thalamic nucleus that receives afferent signals from lower motor centers and relays sensory information to the cortical layers of M1, primary somatosensory cortex, supplementary motor area, and premotor cortex.53,54 Specifically, indirect inputs from cerebellum and basal ganglia are linked to the somatosensory cortex through the VL nucleus. 55 However, with no evidence in literature for direct anatomical links between temporal gyrus and VL nucleus, it is unclear what the consequences of such connectivity changes could be and whether these changes are a reflection of somatotopic reorganization of the motor cortex. Perhaps, examining the FC of the sensorimotor network and auditory network in the same cohort would help with the interpretation of these results. Furthermore, since the right and left sub-nuclei of PUL and VL result in asymmetrical differences in FC, it raises the question of whether the thalamus dysfunction in SCI patients manifests in one hemisphere due to asymmetrical thalamic function. From an evolutionary point of view, the lateralization of thalamus function is expected to reflect the asymmetric organization of the cortex, where the left hemisphere primarily dictates language processing and the right hemisphere governs visuospatial attention processing. 56 But there is a notion that almost all thalamic relays to the neocortex are likely regarding ongoing cortical and subcortical activity related to efferent motor commands. 57 Hence, a left lateralization of thalamus dysfunction observed in this study may not reflect the conventional lateralization of cortex but could merely be the result of investigating right-handed (or left hemisphere motor dominant) individuals with complete SCI.
A significant association between FC measures of different thalamic subnuclei and duration of injury indicates that the functional alterations of the thalamus are likely dynamic and varying through the recovery process after SCI. Our findings indicate that the average FC of thalamic subnuclei to the regions of the dorsal visual attention stream, precentral gyrus, postcentral gyrus, and visual cortex are similar to HC in individuals with shorter postinjury duration (~3 months), that declines and approaches higher negative FC in individuals with longer disease duration (~12 months). The decline in FC postinjury could be attributed as a spontaneous response to deafferentation though its functional implication is unclear. Rao et al 58 reported a decreased FC between the thalamus and parieto-occipital association cortex of rhesus monkeys with right hemitransection at 8 weeks postinjury, which however increases to normal at 12 weeks. In contrast to our findings, Hou et al 9 reported an increase in FC of M1 and cerebellum to anterior and MD portions of the thalamus, respectively, in incomplete SCI at ~12 weeks postinjury. Several reasons could explain the inconsistency in our results, such as completeness of the injury, intersubject variability in residual functionality, rehabilitation techniques, comorbidities, and nonlinear recovery patterns. All of these emphasize the need for longitudinal studies that investigate brain markers immediately following injury, at acute, subacute, and chronic phases to understand the evolution of the condition.
Furthermore, a recent study in individuals with complete SCI report a decrease in FC between the visual cortex and the motor cortex simultaneous with a compensatory increase in FC of visual cortex with sensory parietal cortex. 59 Perhaps this compensatory increase between the visual and sensory parietal cortex is reflected by or is a reflection of alterations in the potential visuo-thalamo-parietal pathways. A significant inverse relationship between ASIA sensory scores and FC of left VL nucleus with cerebellum/occipital gyrus in this cohort may also be associated to the increased dependence on visual inputs arising due to the loss of proprioception in higher levels of injury. Besides, a significant relationship between the ASIA motor scores and the FC of left MD and left PUL nucleus further indicates that the level of function retained could influence the extent of thalamic reorganization in other cortical systems and may differ in higher injury levels (i.e. tetraplegia).
Our study has a few limitations. First, by performing a priori selection of the larger thalamic regions, we limit the scope of the study to the three thalamic subnuclei. Alternatively, we could apply a data-driven approach to investigate the resting-state properties of all the thalamic subnuclei; however, image resolution based on the scanning sequence used could not support these analyses. It would be ideal to have a high-resolution echo planar sequence to study some of the smaller subnuclei of thalamus. Second, the lack of patient information regarding pain status, functional recovery levels and physical therapy limits the functional and behavioral interpretation of the results. Future studies may wish to explore the effect of neuropathic pain levels and somatosensory abnormalities on the thalamic connectivity of the SCI population. Furthermore, we do not account for structural changes found in this cohort as a result of the injury. 60 For future research, we will investigate the effect of gray matter changes on the FC of subcortical structures after SCI.
To our knowledge, this is the first study to investigate and quantify resting-state FC patterns of the thalamus and its subnuclei in a complete paraplegic SCI population. We demonstrate significant changes in the FC of the PUL, MD, and VL nucleus of the SCI group when compared with the HC group. The FC of thalamic subnuclei to sensorimotor and visual attention regions also varies depending on the postinjury duration in these individuals. In conclusion, the present study provides evidence for potential new roles of thalamic subnuclei (MD, VL, and PUL nucleus) in SCI recovery, bringing us one step closer to understanding the functional underpinnings of SCI and paving the way to successful clinical recovery.
Supplemental Material
Supplementary_File – Supplemental material for Resting-State Functional Connectivity of the Thalamus in Complete Spinal Cord Injury
Supplemental material, Supplementary_File for Resting-State Functional Connectivity of the Thalamus in Complete Spinal Cord Injury by Keerthana Deepti Karunakaran, Rui Yuan, Jie He, Jian Zhao, Jian-Ling Cui, Yu-Feng Zang, Zhong Zhang, Tara L. Alvarez and Bharat B. Biswal in Neurorehabilitation and Neural Repair
Footnotes
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the New Jersey Commission on Spinal Cord Research (CSCR15FEL002).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
