Abstract
Introduction
Chronic complete paraplegic persons after spinal cord injury (SCI) maintain motor programs required for the control of foot movements. Previous neuroimaging experiments confirmed preserved cortical activation during attempted and imagined lower limb movements. 1,2
Observation of actions also activates neuronal networks involved in the performance of the same actions. In humans, neurophysiological, behavioral, and brain imaging techniques support the existence of a “mirror neuron system” (MNS). 3-10 These experiments demonstrated the consistent recruitment of a cortical network, including ventral premotor cortex (PMv), parietal lobes, and superior temporal gyrus (STG), 11 the core regions of the MNS. Activation of the MNS offers a possible explanation of how we understand the actions of others, reflecting the visual representation of the observed action onto our motor representation and their “internal” execution. 12,13 Most investigations have focused on the observation of hand movements, but overlapping networks for execution, motor imagery, and motor observation have also been demonstrated for the lower extremities. 14
Burgmer et al 15 have shown that the MNS is activated during observation of hand movements independently of the subjects’ actual motor abilities and, therefore, can provide information regarding the integrity of the motor network in patients with impaired motor function. In SCI patients, the reorganization of the sensorimotor system has been investigated using attempted or imagined motor behavior, but to our knowledge without considering movement observation. In the present study, we investigate the effects of complete long-term SCI on the activation of the observation–execution matching MNS. Based on studies showing overlapping networks for execution, motor imagery, and motor observation in healthy subjects, we hypothesized that complete SCI patients should engage the same sensorimotor foot representation during observation of foot movements that was found for attempted movements, if cortical motor programs have been preserved. The rationale for this study stems from previous reports that observation of specific movements can lead to subsequent gains in their performance. 16,17 Therefore, we looked for additional support for the assumption that movement observation can be applied as a tool for non-movement-dependent motor rehabilitation strategies when actual limb movements cannot, or not yet, be performed.
Methods
Participants
Nine right-handed paraplegic patients (6 males; mean age = 35 years, SD = 6) suffering from chronic complete SCI due to trauma (mean = 9 years, range = 2-20 years) were recruited from the outpatient clinic. All had clinically complete SCI between T3 and L3, as assessed with the International Standards for Neurological and Functional Classification of Spinal Cord Injury 18 and confirmed by repeated electrophysiological recordings of motor evoked potentials of the lower limbs and posterior tibial nerve somatosensory evoked potentials. Twelve age-matched, healthy right-handed volunteers (7 males, mean age = 29 years, SD = 3.7) were recruited as controls. In accordance with the Helsinki Declaration, the local ethics committee approved the experimental protocol. Subjects were reimbursed, and informed consent was obtained.
Instruction and Assessment
The motor task studied was an intransitive foot movement, that is, without any specific goal. It consisted of repetitive alternating dorsal and plantar flexions of the right foot (30°-0°-45°) at a self-paced frequency of approximate 0.5 Hz. In SCI patients, the perceived intensity and frequency of attempted movements was rated in a structured interview describing the intensity of the feeling to move the foot and the frequency of spontaneous attempts in daily life. Answers were noted as qualitative descriptors, and both frequency and intensity of the attempted movements were individually rated using a 6-point scale. 1 The verbal instruction for movement attempt in SCI patients was the following: “Try to move your right foot up and down at an approximate speed of 0.5 Hz.” Correct performance was controlled using an adapted version of the controllability of motor imagery described by Naito et al. 19 Instead of motor imagery as investigated by Naito et al, we asked the patients to attempt a foot movement. With eyes closed, the subjects were required to try moving their right foot as described above and, on command, to promptly give a verbal description of the foot position (dorsal or plantar flexion). The healthy volunteers had to execute the foot movements. For the observation task we used videos displaying repetitive flexion–extension foot movements presented from the side with a frequency of approximately 0.5 Hz. The participants were instructed to carefully observe the presented sequences while maintaining their gaze in the center of the screen.
Experimental Protocol
The experimental conditions were presented in a fixed-order sequence, consisting of execution (healthy subjects) or attempt (SCI patients) of the self-paced foot movements followed by the observation task. Functional magnetic resonance imaging (fMRI) data for each experimental condition were collected in a standard block design consisting of three 21-second periods of baseline alternating with three 21-second periods of activation. For the execution and attempt conditions, each activation period was signaled with the verbal commands “go” and “stop,” transmitted over the machine’s intercom system, and the baseline was rest. The eyes were kept closed during task performance. For the observation condition, videos of a person’s right foot were back-projected onto a screen in the field of view of the subject through a mirror mounted on the head coil. The baseline for the observation was a blank screen of the same color as the background of the observation videos. Subjects were instructed to maintain their gaze in the screen’s center.
Scanning Procedure
Blood oxygenation level dependent fMRI was carried on a Philips 1.5-T whole body scanner (Philips Healthcare, Andover, Massachusetts) equipped with a standard product transmit–receive head coil. FMRI data were obtained using a single-shot, gradient-echo, echo-planar imaging (EPI) sequence (TE [echo time] = 55 ms; TR [repetition time] = 3000 ms; flip angle = 90°). For each of the 126 time points, 30 contiguous, axial slices (voxel size 3.4 × 3.4 × 5mm) covering the entire brain were acquired. T1-weighted whole-brain anatomical reference volume data with an isotropic spatial resolution of 1.2 mm were acquired with a 3-dimensional spoiled gradient echo sequence (TE = 9 ms; TR = 50 ms).
Imaging Analyses
The first part of these experiments has already been published. 1 As we wanted to present these data as comparable with the previous publication, we applied an older version of SPM. The first 2 volumes of each fMRI time series were discarded. Using SPM99 (Welcome Department of Cognitive Neurology, London; http://fil.ion.ucl.ac.uk/spm), the EPI functional scans were realigned and normalized to the MNI template with a resolution of 3 × 3 × 3 mm. Thereafter, 10-mm smoothing was applied to the images. The statistical analysis was performed at 2 levels in the context of the general linear model. For individual analysis, data from each run were modeled using a delayed boxcar function convolved with the hemodynamic response function.
In the single-subject analysis, activations were considered significant at
Based on the known functional neuroanatomy of the MNS,
7,14
the following areas in both hemispheres were selected from the anatomical automatic labeling atlas
21
: for M1/S1, the paracentral lobule; for premotor regions, the supplementary motor area, cingulate motor areas (CMA), precentral gyrus and frontal operculum (PMv and PMd); for posterior parietal areas, postcentral gyrus (S1), superior and inferior parietal regions (IP and SP); for the temporal cortex, superior and middle temporal sulcus (STG); and subcortically, thalamus, basal ganglia, and cerebellum. For each activated cluster, the volume of activation and the maximal signal intensity were determined, and the localization in MNI coordinates was obtained using the “WFU-Pickatlas,” a Web-based interactive program, which provides the coordinates of a specified region of interest after implementation of small volume correction.
22
The volume of activation and the maximal signal intensity were determined and the activation in MNI coordinates was obtained with a threshold set at
Results
Behavioral Data
In the structured interview, all SCI subjects stated that they were able to attempt moving their right foot, as reported previously.
1
This was confirmed by the controllability of motor imagery,
19
showing that they were able to indicate the actual posture of their foot during random interruption of the movement attempt. The patients rated the intensity of their feeling during attempted movements on the 6-point rating scale as medium to very high (mean = 4.5, SD = 3.6, range = 3-6). The frequency of spontaneous daily movement attempts on the 6-point rating scale was lower (mean = 3.6, SD = 1.7). The perceived intensity and frequency of spontaneous daily movement attempts were significantly correlated (
Execution of Foot Movement in Healthy Controls and Attempted Movements in SCI Patients
For the controls performing movements with their right foot, the group analysis revealed significant activations contralaterally in M1/S1 foot representation and bilaterally in mesial and lateral premotor cortical regions and cerebellum. Additionally, ipsilateral activations were observed in the superior (SP) and inferior (IP) parietal areas, thalamus, and posterior putamen (Table 1, Figure 1).
MNI Coordinates of Significant Cluster Maxima,
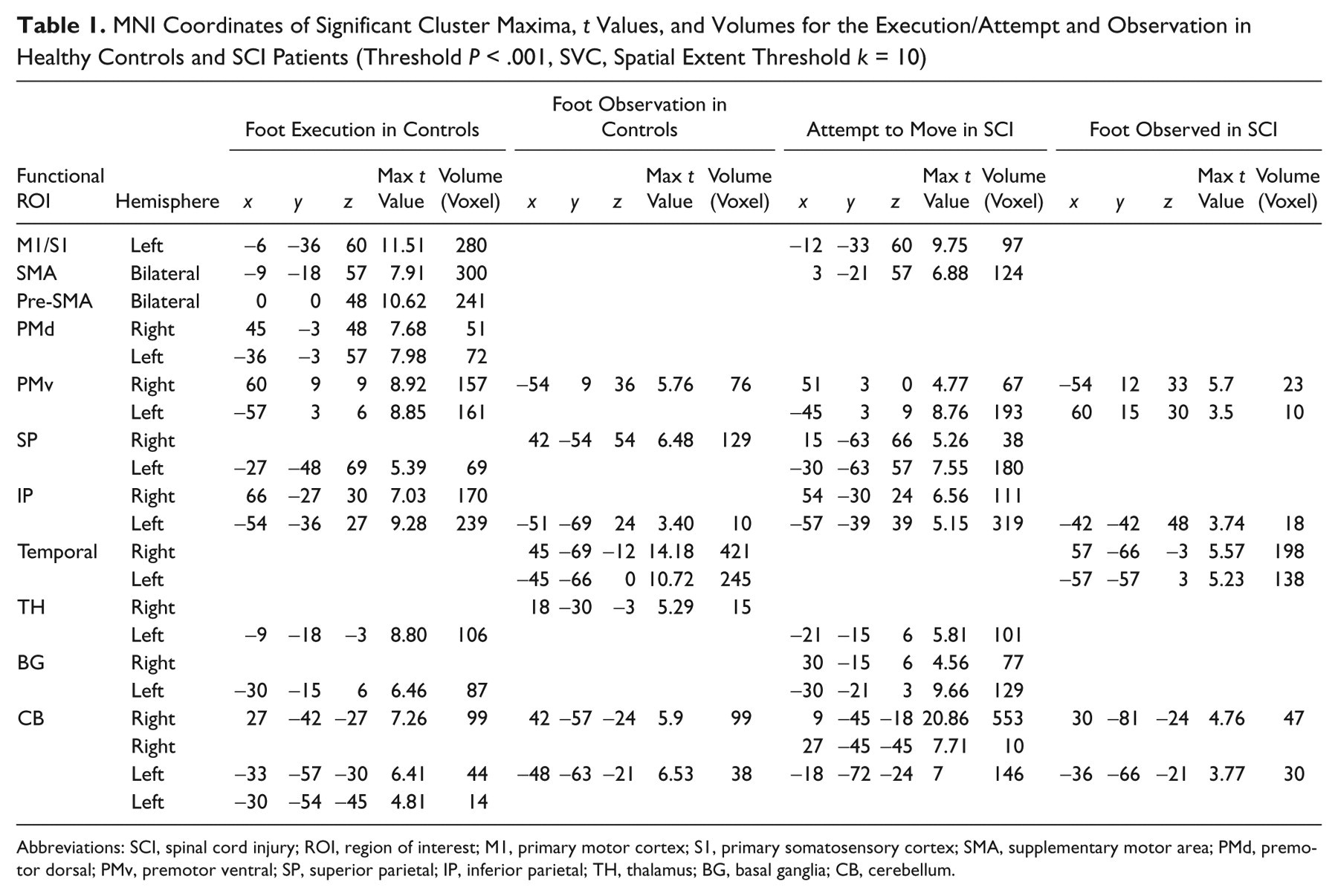
Abbreviations: SCI, spinal cord injury; ROI, region of interest; M1, primary motor cortex; S1, primary somatosensory cortex; SMA, supplementary motor area; PMd, premotor dorsal; PMv, premotor ventral; SP, superior parietal; IP, inferior parietal; TH, thalamus; BG, basal ganglia; CB, cerebellum.
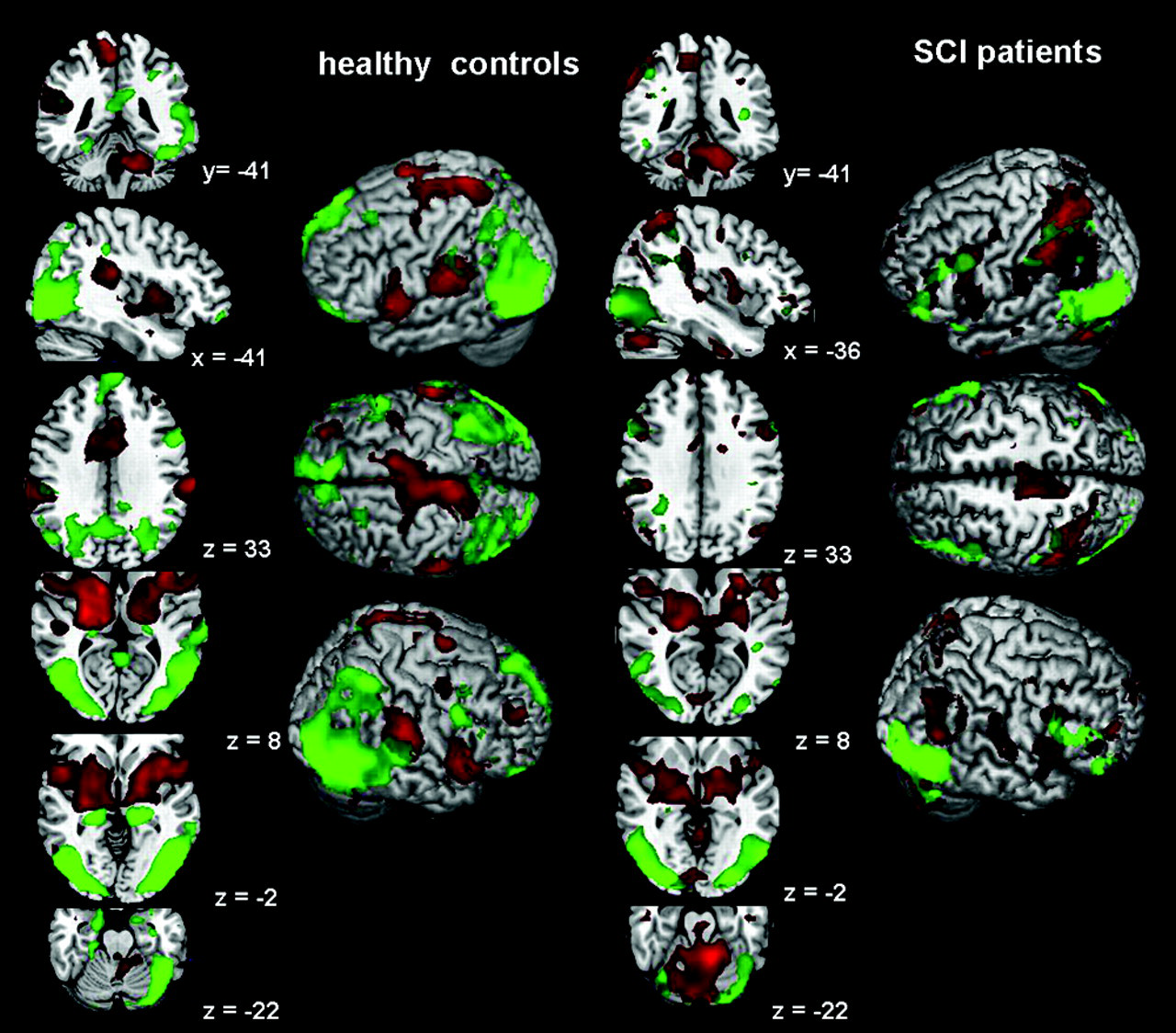
Group activations for observation and execution, respectively, attempt of right foot movements. Control group on the left, spinal cord injury patients on the right. In green: movement observation. In red: movement execution, respectively, attempt. Same average brain images to facilitate comparison between groups.
When the SCI patients attempted to move their right foot, the activated regions were comparable with those found during execution in the controls (Table 1). However, the activated cluster in the primary sensorimotor cortex was smaller and had less intensity when compared with the values in healthy subjects. Significant clusters were additionally found bilaterally in the prefrontal (PF) and SP cortex and ipsilaterally in PMv and the posterior putamen (Table 2).
Frequency of Single-Subject Activation in Specified ROIs During Observation of Foot Movements a
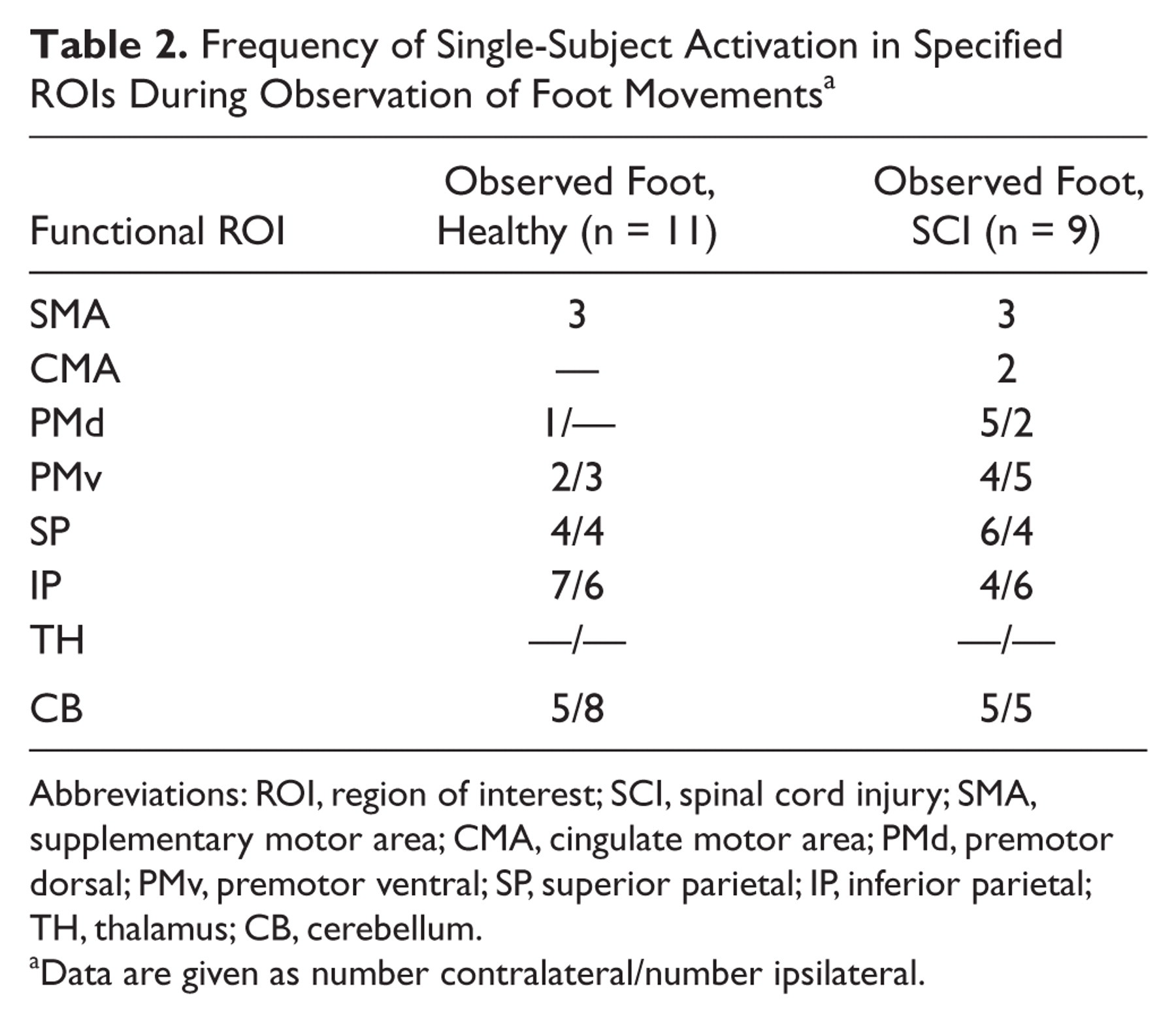
Abbreviations: ROI, region of interest; SCI, spinal cord injury; SMA, supplementary motor area; CMA, cingulate motor area; PMd, premotor dorsal; PMv, premotor ventral; SP, superior parietal; IP, inferior parietal; TH, thalamus; CB, cerebellum.
Data are given as number contralateral/number ipsilateral.
Observation of Foot Movement in Healthy Controls and SCI Patients
In healthy controls, observation of foot movements activated the core network of the execution–observation matching system including PMv, STG, and parietal regions. The PMv activation was located in the left inferior precentral gyrus at a higher location (
In SCI patients, group analysis showed activation clusters in PMv, IP, and posterior cerebellum, similar to the healthy controls, but none in the SP lobule. Moreover, the single-subject analysis revealed that during observation PMv and IP regions were more frequently activated in the SCI patients than in the controls (Table 2). Activations in SP were also frequently observed in the SCI patients; the clusters, however, were quite scattered and therefore did not reach the significance level in the group analysis. Figure 2 shows the activation patterns during movement attempt and observation in 4 SCI patients, demonstrating the large interindividual variability in the extent of activation areas. The individual analyses further revealed that 5 out of 9 patients did activate the MNS, whereas 3 patients showed activation of only parts of this network, and 1 had no significant activation, except in visual areas (Figure 2, left-hand side).
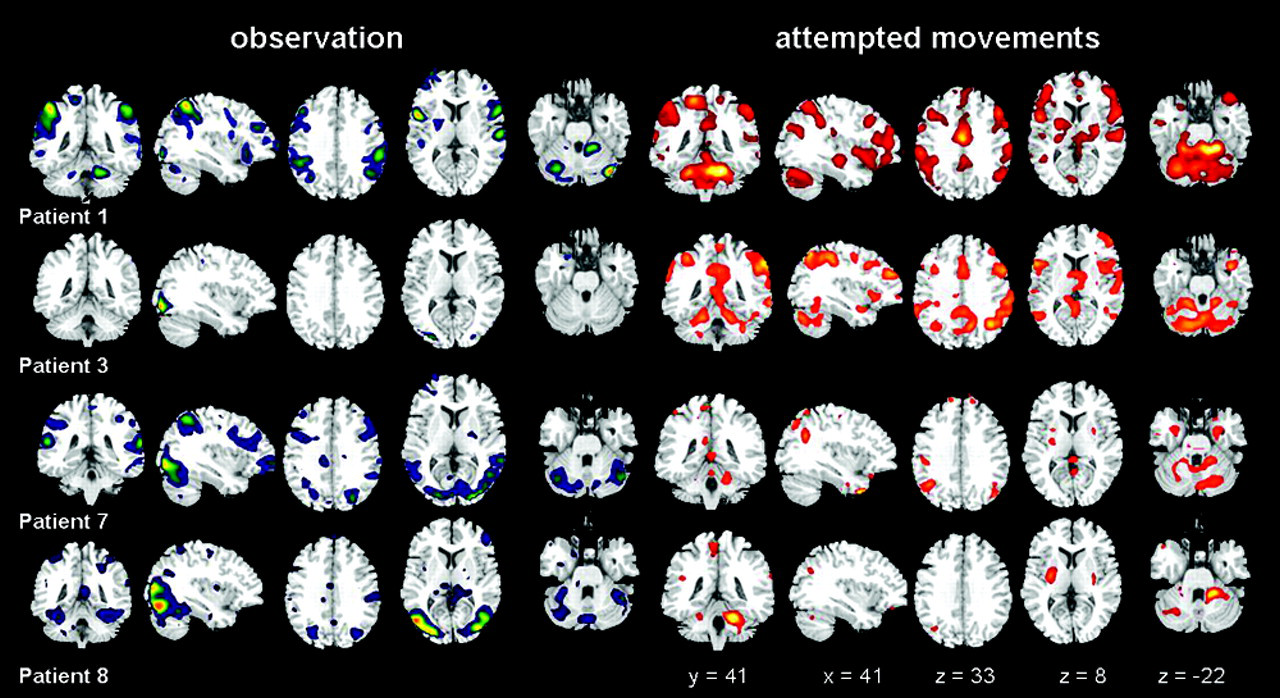
Individual activation patterns during observed and attempted movements in 4 spinal cord injury patients. Observation in blue (left-hand side) and attempt of right foot movements in red (right-hand side). Same slice levels as those in Figure 1.
In the SCI patients, the activated clusters during observation were compared with those during movement attempts; a decrease of activation (fewer and smaller clusters) was seen in PMv and parietal lobules. In the cerebellum, observation activated only posterolateral regions, and no anterior ones as during attempts.
Contrast and Conjunction Analyses
The contrast between SCI patients and controls during foot observation revealed that SCI patients activated more strongly the PMv and IP cortex in both hemisphere and subcortically, the basal ganglia, and the cerebellum (Table 3). The inverse contrast (healthy controls vs SCI patients) disclosed an additional cluster in the right lower PMv and bilateral activation in CMA and anterior cerebellum in the healthy subjects (Table 2).
MNI Coordinates of Significant Cluster Maxima,
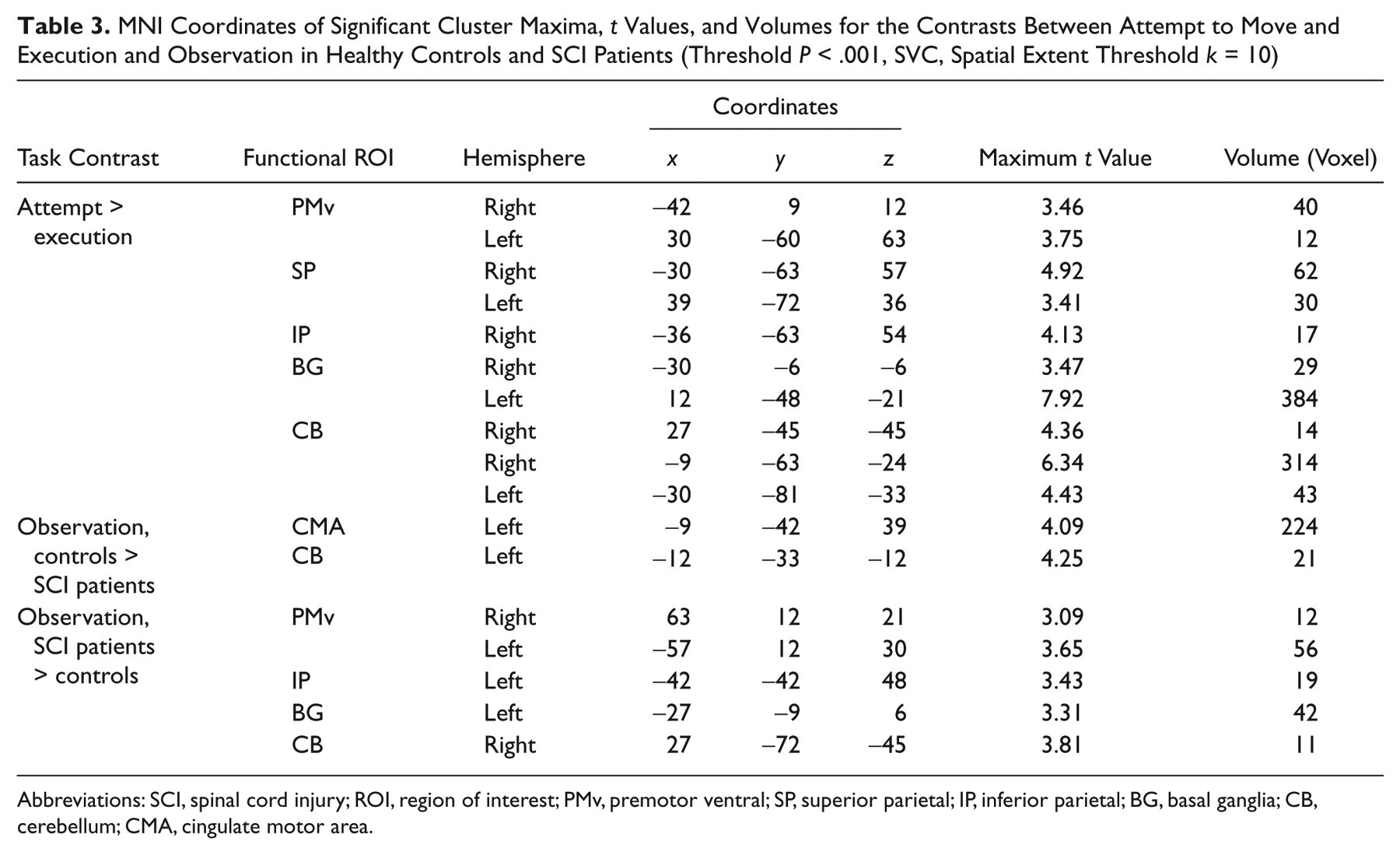
Abbreviations: SCI, spinal cord injury; ROI, region of interest; PMv, premotor ventral; SP, superior parietal; IP, inferior parietal; BG, basal ganglia; CB,cerebellum; CMA, cingulate motor area.
In the control group, the conjunction analysis confirmed that execution and observation of the same movement elicited activation clusters bilaterally in the PMv and IP cortex. In the SCI group, the conjunction analysis also showed common activated regions in bilateral PMv, IP and SP cortex, as well as in the cerebellum.
Discussion
Our study confirms the preserved excitability of the observation–execution matching “mirror” system in SCI patients during observation and attempted movements of the plegic lower limbs. This motor network is functional in the absence of actual movements and somatosensory feedback and thus remains intact after long-term nonuse. The regional activation triggered by observation partially overlapped the cortical and subcortical activations elicited during movement attempt. Moreover, 2 main regions of the MNS, namely the PMv and IP regions, were activated in both tasks in the SCI patients and healthy controls.
Observation–Execution Matching System After SCI
Our investigation confirms that in healthy subjects observation of foot movements induces activation in the core region of the cortical MNS in PMv and parietal cortex. 14,25 In addition, we extend these findings to subcortical regions, that is, the thalamus and posterior cerebellum. Activation elicited by movement observation was reduced compared with execution, as reported by Orr et al. 14 In our chronic complete paraplegic patients, the conjunction analysis revealed that observation and attempted foot movements activated the core regions of the MNS as in the healthy controls, with additional recruitment of the cerebellum. The activation during observation was reduced when compared with attempted movement, as well as when observation was compared with execution in the control group.
These findings have 2 logical consequences. First, the partially overlapping neural substrate for attempted and visually perceived movements confirms the retained integrity of the central representation of the plegic lower extremities, even after a long period of nonuse. The areas involved in action observation correspond to sensorimotor representations shaped by prior experience. Second, our study demonstrates that in complete SCI patients, neural systems involved in movement planning and control can be activated by observation only. This is an important finding in view of the possibility that a disturbed representation of movement generation could prevent the activation of the mirror system. This has been reported in patients with conversion disorders who had similar activation patterns during attempted hand movements as for executed ones by healthy subjects but no activation at all during movement observation. 15
Functional Implications
Our findings suggest 2 potential uses of movement observation in rehabilitative strategies. First, movement observation is a new type of passive paradigm to investigate the internal generation of a motor representation by means of neuroimaging or other neurophysiologic methods able to measure motor responses, when overt movements are not feasible. The partial preservation of motor control might be important when considering a restoration of spinal cord conductivity in complete SCI patients or for the use of brain–machine interfaces. 26,27
Second, movement observation might be a valuable complementary technique in neurorehabilitation. Recent investigations have shown that motor sequence learning in healthy subjects occurs in the absence of overt movements by observing, then imitating, the actions of others. 16,17,28,29 This imitation-based motor learning supports the hypothesis that the motor system can incorporate experiences of others in building a personal motor repertoire. In the future, patients will hopefully profit from clinical investigations, aiming directly to replace sensory deprivation by the mere visual activation of existing motor representations.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by the Swiss National Science Foundation (SNSF) grant numbers 3100-67168.01 and PMPDB-114395.
