Abstract
Keywords
Introduction
The recovery of walking is important to individuals who have sustained a spinal cord injury (SCI), particularly incomplete SCI. 1 It is therefore anticipated that future trials will target the improvement of lower-extremity motor function and walking. The Walking Index for Spinal Cord Injury (WISCI) is a hierarchical scale that measures improvement in walking following SCI. 2 Originally described with 19 levels, it was revised and expanded in 2001 to include 21 levels (see supplemental data available online). 3 The resulting WISCI II is intended for use in clinical trials and incorporates physical assistance as well as the use of ambulatory aids and braces. Conceptually, it captures the spectrum of adaptations required to walk with varying degrees of impairment following SCI (eg, lower-extremity weakness).
Within the context of clinical trials intended to improve lower-extremity motor function, it is important that outcome measures for walking demonstrate a linkage between the underlying impairment (weakness) and related functional activity (walking). During the development of the WISCI II, the ranking of items was driven by the severity of the underlying impairment (lower-extremity weakness) rather than the level of required physical assistance. 2,3 Prior studies have demonstrated a relationship between lower-extremity motor scores (LEMS) and walking in people with SCI. 4-10 An outcome measure that captures the relationship between the underlying impairment and the related functional activity would have convergent validity. Convergent validity is the degree to which a parameter (eg, lower-extremity strength) is similar to (converges with) other parameters to which it should be theoretically related (eg, walking).
An outcome measure should also have good reliability. Test-retest reliability indicates the level of measurement error, or “noise” for an outcome measure—in other words, how similar the values are when measuring an unchanged parameter on more than one occasion. The better the reliability, the easier it is to differentiate real change from noise. This can be objectively assessed by calculating the smallest real difference (SRD), a measure of test-retest reliability and within-subject variance. 11 The SRD yields a value with the same units as the outcome measure. Following an intervention, a change in score would need to exceed the SRD to be considered “real.”
The psychometric properties of the WISCI and WISCI II have been thoroughly evaluated following acute SCI. In this population, the WISCI has been demonstrated to have good international face validity, reliability, and concurrent validity (Functional Independence Measure [FIM], timed walking, Spinal Cord Independence Measure, Barthel Index, Rivermead Mobility Index, Berg Balance Scale). 2,4,12-16 There are, however, important distinctions between acute and chronic SCI: an important one being that, consistent with natural recovery, individuals progressively improve during the acute and subacute phases of injury. 17 In comparison, persons with chronic SCI are comparatively static. Because of this distinction, it is important to assess the psychometric properties of outcome measures independently in both populations. In this study, we were interested in determining the reproducibility and convergent validity of the WISCI II and walking speed in a cohort of individuals with chronic SCI.
Methods
Participants
Prior to study commencement, the protocol was approved by the Thomas Jefferson University institutional review board. Study participants were recruited from the patient pool at the Regional Spinal Cord Injury Center of the Delaware Valley, a partnership of Thomas Jefferson University Hospital and Magee Rehabilitation Hospital, Philadelphia, PA. Potential participants were identified through direct mailings, posters, and with the help of clinicians in outpatient clinics. Participants had to have sustained a traumatic SCI of at least 6 months’ duration at the time of screening. In addition, individuals had to weight-bear through the lower extremities at least once per week. Those with medical conditions that could limit their ability to safely ambulate were excluded from the study. Patients were also excluded if they had any history of heart disease and uncontrolled asthma or were taking medication that could affect heart rate (ie, β-blockers or calcium-channel blockers). The final study cohort consisted of 76 individuals.
WISCI Assessment
Data for this analysis were collected as part of a larger study protocol, for which the methods have been previously described. 18 Informed consent was obtained from all participants prior to testing. Motor and sensory levels as well as American Spinal Injury Association (ASIA) impairment scale (AIS) grade were determined in accordance with the International Standards for Neurological Classification of Spinal Cord Injury. 19 Efforts were made to assess walking twice for each participant; 63 participants completed 2 sessions, whereas 13 participants completed 1 session. Scheduling intervals between session 1 and session 2 varied from 0 to 437 days (mean = 35); however, participants were chronic and therefore felt to be relatively stable. A total of 5 people underwent both sessions on the same day, whereas 8 were tested more than 50 days apart.
Testing was performed by physical therapists trained in the determination of WISCI levels. For each participant, self-selected and maximum WISCI were determined. The self-selected WISCI is defined as the WISCI level the participant reports she/he uses to walk in the community or alternatively the household if community ambulation is not possible. The therapists confirmed that the participant could ambulate 10 m at the reported level. The maximum WISCI is defined as the highest level at which a participant can safely walk 10 m, as determined by the study therapists. To determine the maximum WISCI, the therapist advanced the participant through WISCI levels until the participant failed a level or was deemed unsafe for the next level. Only minimal assistance was allowed for WISCI levels that required the assistance of another person. 3
Following the determination of WISCI levels, the participants were instructed to ambulate 100 m at both their self-selected WISCI and maximum WISCI levels. Testing was performed on a hard, flat surface along a marked 10 m length for a maximum of 10 lengths (100 m). Turns were required at the completion of each 10 m length. Participants were instructed to begin walking at a comfortable speed when research personnel were in place and to continue through the final mark at a steady rate. They generally began walking at least a few feet behind the initial mark, and timing was initiated once she/he crossed the initial mark. There were no rest periods between lengths, and turning time was recorded and excluded from walking time. Maximum WISCI testing was performed first to optimize the chance that study participants would be able to complete the desired 100 m for the maximal WISCI level. Ambulatory capacity was then assessed at the self-selected WISCI level. To assign a WISCI level, a participant has to be able to walk 10 m, and only the first 10 m of the 100 m was used for the analyses presented here. Ambulatory velocity (in m/s) was calculated by dividing the distance walked (10 m) by time elapsed.
Statistical Analyses
The Spearman rank correlation coefficient (ρ) was used to determine the convergent validity between the assigned WISCI level and walking speed, LEMS, upper-extremity motor scores (UEMS), and combined upper- and lower-extremity manual muscle testing (MMT). For sessions 1 and 2, 1-way random effects model intraclass correlation coefficients (ICCs) 20 were calculated for WISCI level and walking speed. Additionally, the raw agreement between session 1 and session 2 was determined for the WISCI levels.
Reproducibility of the WISCI scale and walking speed was assessed by calculating the SRD, which is a function of the standard error of measurement (SEM). 11 The SEM quantifies the test-retest reproducibility of a measure by calculating the variability of measurements in the same individual and is the square root of the within-participant variance. The 95% confidence interval SRD is calculated as 1.96 × √2 × SEM. ICCs, SRDs, and SEM were calculated with variances generated from repeated-measures ANOVA for random effects. All analyses were conducted with the use of SAS v9.1 software for Windows (SAS Institute Inc, Cary, NC). Correlation coefficients were interpreted as weak (.2-.4), moderate (.4-.7), strong (.7-.9), and excellent (.9-1.0). 21 The threshold for significance was α ≤ .05.
Results
Demographics
A total of 76 people participated in the study, with 45% having paraplegia and 55% tetraplegia. Postinjury duration ranged from 8 months to 25 years at the time of testing. Seventy participants were ≥12 months postinjury, with only 1 being less than 10 months postinjury. In all, 79% of participants were men, which is consistent with the expected gender distribution of the SCI population in the United States. 22 The mean age for participants was 43.3 years (±13.8), and mean years from injury at the time of testing was 6.32 years (±5.99). The most common injury etiology was motor vehicle accidents (32%), followed by falls (26%). Sports/diving incidents and acts of violence represented 13% and 11% of injuries, respectively. Eighteen percent of injuries were due to other etiologies.
The distribution of AIS grades was A (3%), B (1%), C (8%), and D (88%), which reflects that participants had to ambulate a minimum of 10 m to be assigned a WISCI level and participate. This distribution is expected in a study of ambulation following chronic SCI, given that motor complete individuals (AIS A and B) typically do not walk, and many AIS C participants do not have sufficient strength for walking. The small number of individuals with motor complete injuries had either conus medullaris or cauda equina injuries with the preservation of volitional motor function in the proximal lower-extremity muscles, which facilitated gait. Mean LEMS was 38.97 (±9.52) with a range of 11 to 50.
Convergent Validity
To assess convergent validity for both self-selected and maximum WISCI levels and walking speeds, we assessed their relationships with LEMS, UEMS, and MMT (Table 1). The correlation between WISCI levels and 10-m walking speed was also assessed. For the entire cohort, self-selected and maximum WISCI levels correlated significantly with total MMT, LEMS, and walking speed. Apart from walking speed where the maximum WISCI demonstrated a stronger association than the self-selected WISCI, all correlations for self-selected and maximum WISCI levels were comparable. For both maximum WISCI and self-selected WISCI, the strongest correlations were with LEMS: ρ = 0.717 and ρ = 0.704, respectively.
Spearman Rank Correlations (ρ) a
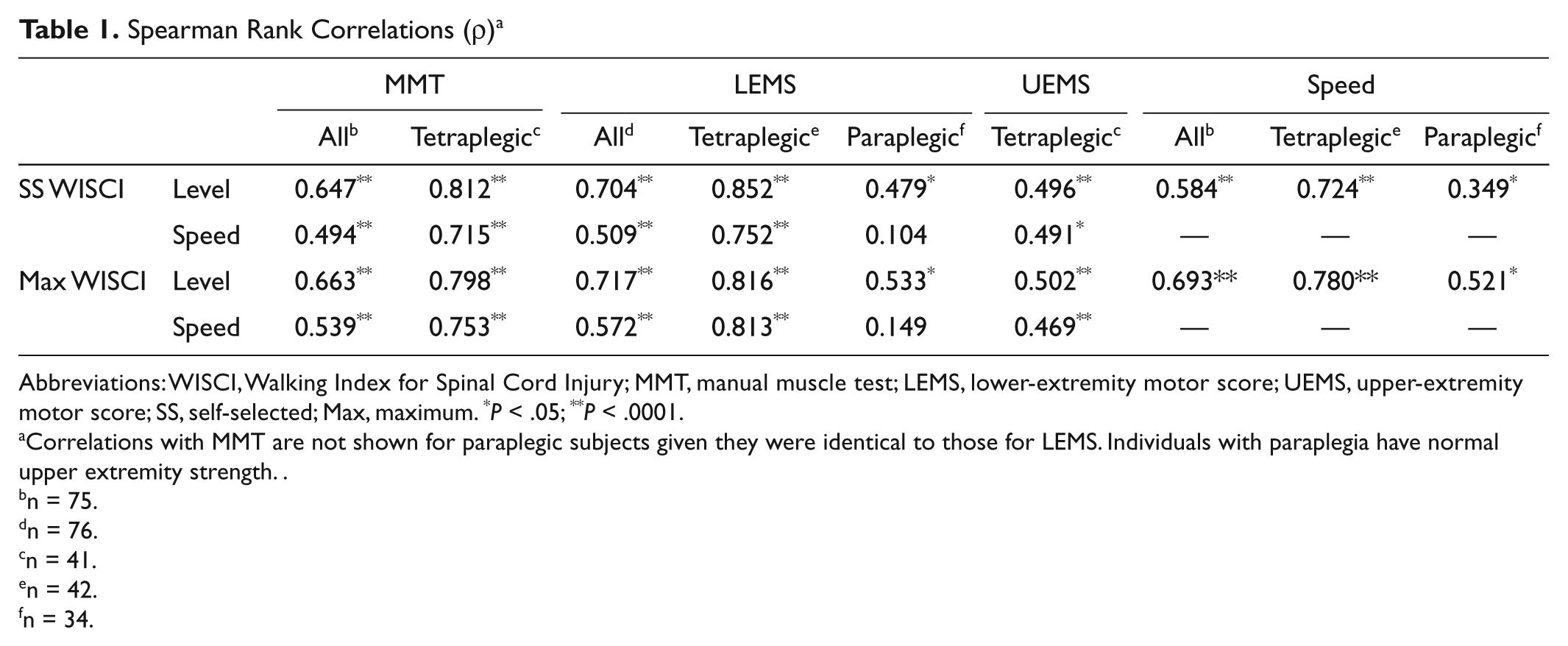
Abbreviations: WISCI, Walking Index for Spinal Cord Injury; MMT, manual muscle test; LEMS, lower-extremity motor score; UEMS, upper-extremity motor score; SS, self-selected; Max, maximum. *
Correlations with MMT are not shown for paraplegic subjects given they were identical to those for LEMS. Individuals with paraplegia have normal upper extremity strength.
n = 75.
n = 76.
n = 41.
n = 42.
n = 34.
Interestingly, there were profound differences when the composite cohort was split into tetraplegic (n = 42) and paraplegic (n = 34) cohorts (Table 1 and Figure 1). The correlations between self-selected WISCI level and LEMS were ρ = 0.852 (tetraplegic) and ρ = 0.479 (paraplegic), respectively. In the same vein, the correlations for maximum WISCI were ρ = 0.816 (tetraplegic) and ρ = 0.533 (paraplegic). For tetraplegic participants, there were also significant correlations between WISCI levels and UEMS: ρ = 0.496 (self-selected) and ρ = 0.502 (maximum). Similar analysis was not performed for paraplegic participants given their normal upper-extremity function.
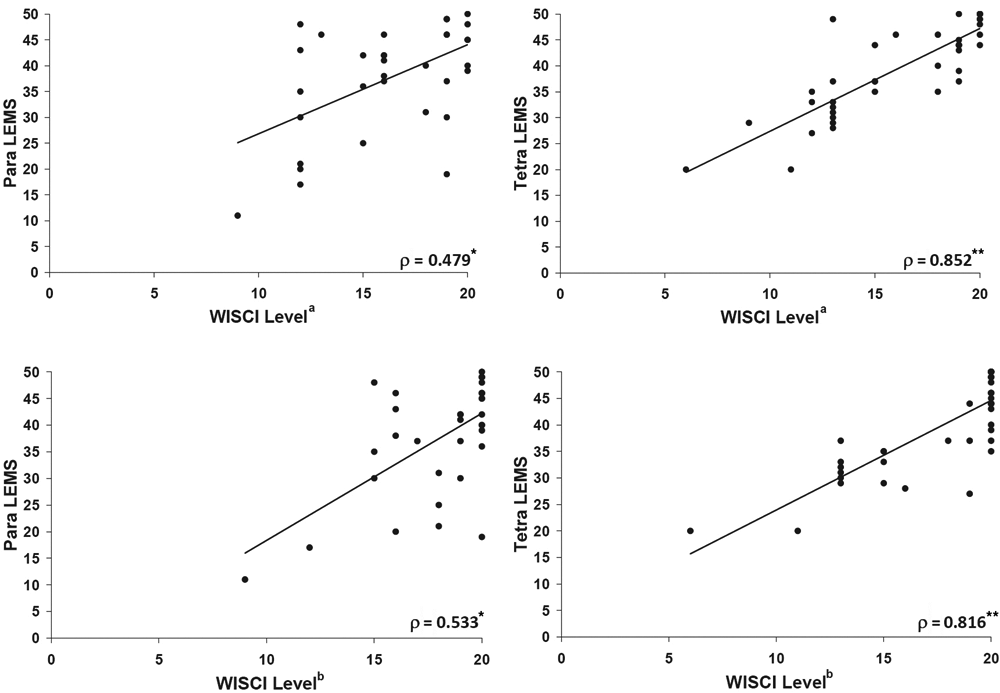
Scatterplots for relationships between WISCI level and LEMS in subjects with paraplegia (left) and tetraplegia (right)
When the entire cohort was analyzed, walking speed correlated significantly with MMT, LEMS, and WISCI (maximum and self-selected; Table 1). The correlation between walking speed and LEMS was lost when the analysis was restricted to paraplegic participants but not tetraplegic participants: ρ = 0.752 (self-selected) and 0.813 (maximum; Figure 2). For tetraplegic participants, significant correlations were also observed between walking speed and UEMS: ρ = 0.491 (self-selected) and 0.469 (maximum). The correlation with lower-extremity function (LEMS) was greater for both self-selected and maximal WISCI compared with walking speed, for all cohorts (composite, tetraplegic, and paraplegic).
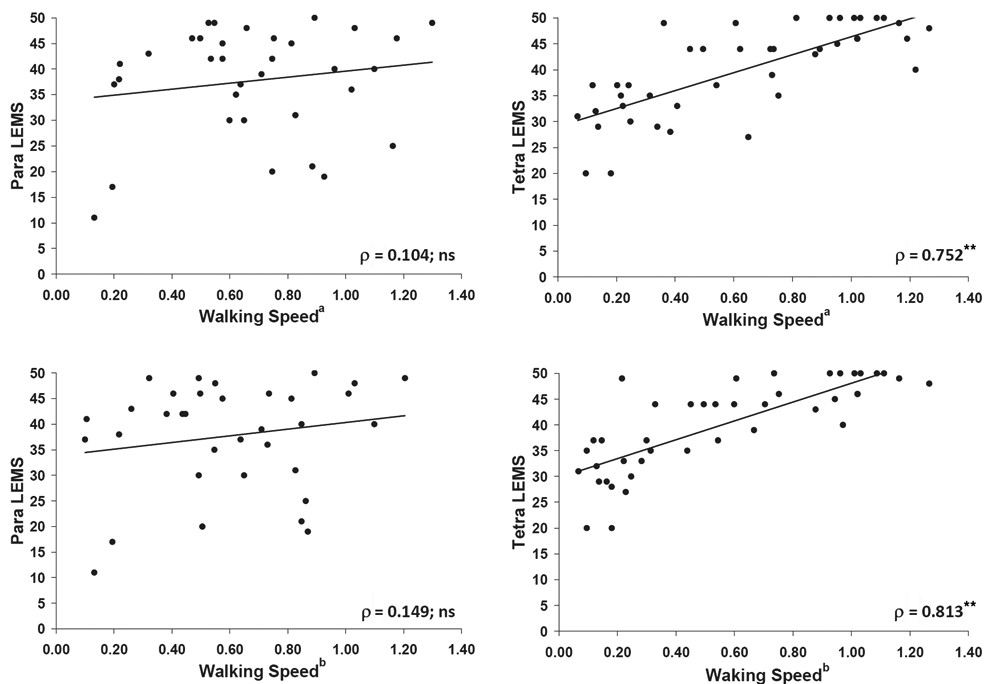
Scatterplots for relationships between walking speed and LEMS in subjects with paraplegia (left) and tetraplegia (right)
Test-Retest Reliability and Reproducibility
A total of 63 participants participated in both testing sessions. Mann-Whitney test and Pearson’s χ2 square did not reveal any significant differences between those who attended both sessions and those who only attended the first session for any of the demographic or impairment variables. ICCs revealed that both walking speed and WISCI level assignment remained consistent between sessions 1 and 2 (Figure 3). As expected, the majority of self-selected and maximum WISCI levels were the same during both sessions. For the self-selected WISCI, the assigned WISCI differed between sessions 1 and 2 for 2 cases. One participant increased from 16 to 19, whereas the other decreased from 20 to 19. Similarly, the assigned maximum WISCI differed between sessions for 3 cases. All cases decreased with 2 of them dropping from 20 to 19 and one from 20 to 18. There was 100% agreement for all other WISCI levels.
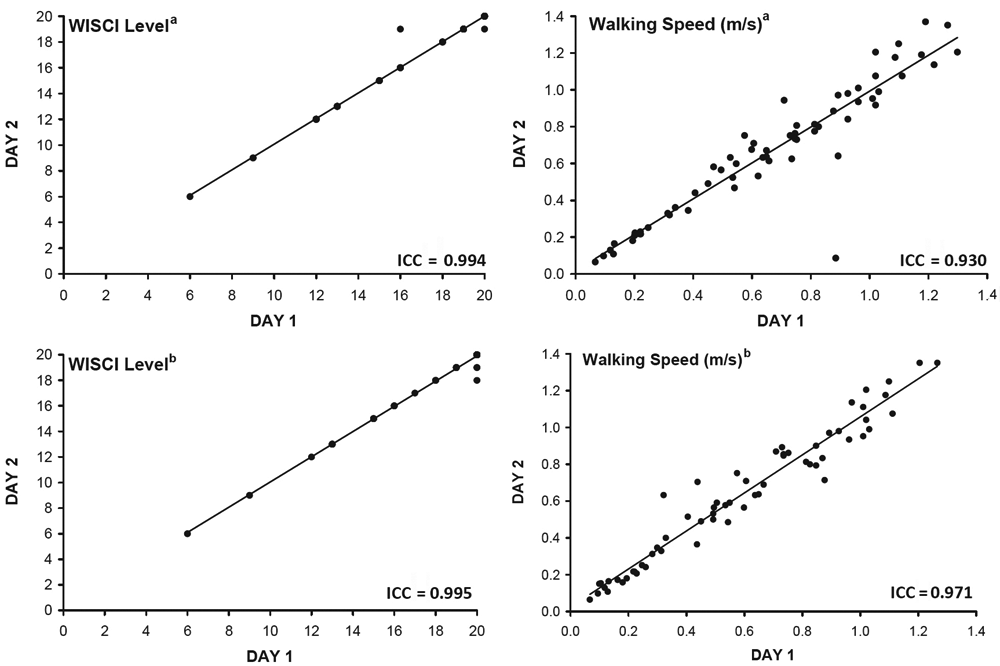
Test-retest reliability for WISCI score assignment and walking speed
The sensitivity of the WISCI for clinical trials was supported by the SRD (Table 2). The SRD was less than 1 for both self-selected (0.785) and maximum (0.597) WISCI levels, suggesting that a change of 1 WISCI level could be considered real in the context of a clinical trial. The range of walking velocities for the 10-m walk was (0.0668-1.2987 m/s) and (0.0668-1.2658 m/s) when assessed at the self-selected and maximum WISCI, respectively. The accompanying self-selected and maximum SRDs were 0.254 m/s and 0.163 m/s, respectively.
ICC and SRD for WISCI Level and Walking Speed (n = 63)
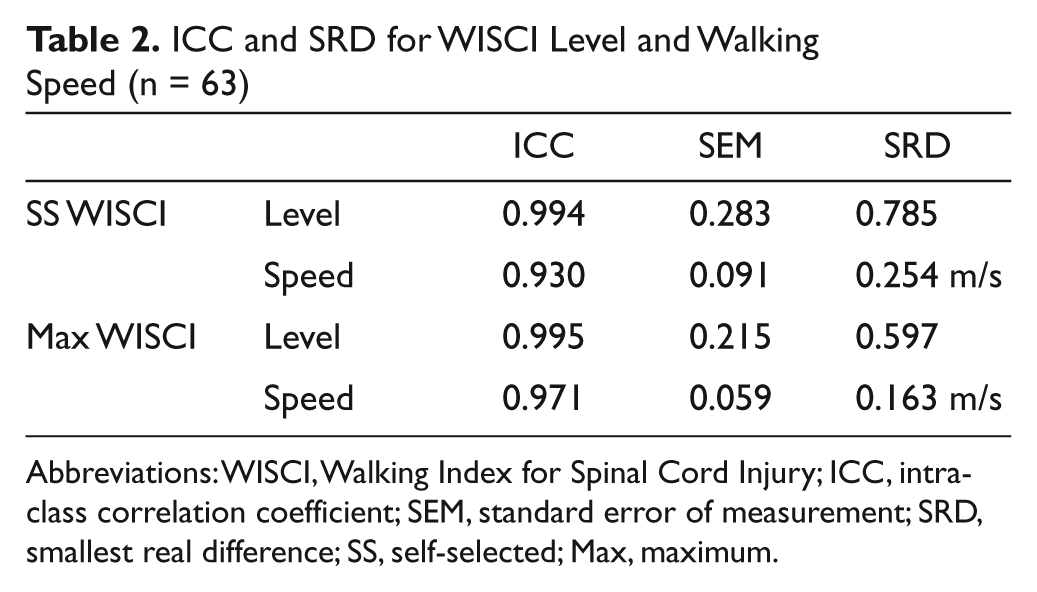
Abbreviations: WISCI, Walking Index for Spinal Cord Injury; ICC, intraclass correlation coefficient; SEM, standard error of measurement; SRD, smallest real difference; SS, self-selected; Max, maximum.
Discussion
It is anticipated that future interventions that target the recovery of motor function following SCI will also affect walking. Trials to determine the efficacy of these interventions will therefore need to incorporate outcome measures that capture changes in walking capacity. Prior studies have demonstrated a relationship between motor strength and walking. 4-8,10,13 Outcome measures therefore should demonstrate a clear relationship between the underlying impairment (motor function) and functional activity (walking)—convergent validity. It cannot, however, be assumed that outcome measures will exhibit the same psychometric properties following acute and chronic SCI, given the important distinctions. Consistent with natural recovery, individuals with acute injuries are expected to progressively improve during the acute and subacute phases of injury, whereas persons with chronic SCI are comparatively static.
Several recent studies have focused on the psychometric properties of outcome measures for walking capacity following acute SCI. 3,4,8,10,12-14,16,18,23,24 In participants with acute SCI, validity and reliability have been demonstrated for both timed walking tests (eg, 10 m, 6 minute) and categorical scales (eg, WISCI II). 4,8,12,13,25 In the context of acute SCI, the WISCI II has been shown to have concurrent validity with the FIM, FIM locomotor score, Spinal Cord Independence Measure indoor mobility score, Barthel Index, Rivermead Mobility Index, Berg Balance Scale, and timed walking tests (timed up and go, 10-m walk, 6-minute walk). 2,4,12-15
In comparison, the psychometric properties of these outcome measures still need to be assessed for chronic SCI. Recently, the WISCI II was reported to have good interrater and intrarater reliability following chronic SCI. 16 In this study of patients with chronic SCI, we found that compared with the 10-m walk, the WISCI II demonstrated a stronger correlation between the underlying impairment of lower-extremity motor function and walking—in other words better convergent validity. This relationship held true whether the cohort was analyzed as a whole or separated into tetraplegia and paraplegia. In fact, the correlation between walking speed and LEMS disappeared when analysis was limited to paraplegia. For tetraplegia, moderate correlations were present between UEMS and both timed walking and WISCI level (self-selected, maximum). This finding supports the contribution of the upper-extremity motor function to walking capacity following cervical SCI. Scivoletto and colleagues 9 have previously reported similar findings.
WISCI level and walking speed were strongly correlated with LEMS in tetraplegic patients, whereas such relationships were moderate or absent in individuals with paraplegia. Such findings suggest that lower-extremity motor function is a stronger contributor to walking capacity following cervical SCI than thoracic or lumbar SCI. This finding, however, should be interpreted in the context of the cohort, which consisted of predominantly AIS D participants. This finding is not surprising given that individuals with SCI usually require a certain threshold of lower-extremity strength to ambulate. 10 Waters and colleagues 26 have suggested that the ability of tetraplegic patients to use assistive devices is limited by impaired upper-extremity function, and they therefore require relatively greater lower-extremity strength when walking. It can also be speculated that the increased truncal stability associated with paraplegia might facilitate ambulation with comparatively less lower-extremity strength.
In all analyzed cohorts (composite, tetraplegic, and paraplegic), correlations of motor strength (LEMS, UEMS, MMT) with WISCI levels were stronger than with walking speed. This was most strongly contrasted in paraplegic patients, who demonstrated a moderate relationship between LEMS and WISCI level but no comparable relationship between LEMS and walking speed. The absence of a linkage between walking speed and motor strength in paraplegic patients brings into question the validity of using timed walking tests in interventions primarily targeting motor strength (as opposed to balance, coordination, spasticity, etc) in people with chronic paraplegia. In the setting of chronic paraplegia, individuals with greater lower-extremity strength may walk with less assistive devices or assistance, but they do not walk any faster (Figures 1 and 2).
Morganti and colleagues 13 previously demonstrated a correlation between LEMS and WISCI II following acute traumatic and nontraumatic SCI, although it was weaker (0.58) than what was observed here with chronic SCI. Conceivably, variables other than strength (eg, training effect/practice) might make a greater contribution to walking ability during the acute phase as compared with the chronic SCI phase, when the bulk of rehabilitation interventions have already occurred. In support of this, Ditunno et al 8 reported the correlations between LEMS and initial WISCI (within 1 month of injury) as well as final WISCI (6-12 months postrehabilitation discharge) in a cohort of 170 injuries. Spearman’s correlations were 0.47 and 0.91 for initial and final WISCI, respectively.
An interesting point is that another study that followed acute SCI patients serially for 1 year revealed that LEMS correlated better with timed walking tests (10 m, 6 minute) when compared with the WISCI II. 27 One possible explanation for the observed discrepancy is that individuals in this study were relatively high functioning and did not represent the full continuum of injury severity. To be included, participants had to be able to walk within 1 month of the injury. By 6 months postinjury, the patients reached normal walking speed, and all but 1 of the 22 participants reached the maximum WISCI level of 20. Furthermore, 21 of 22 participants had 12-month LEMS exceeding 40 (maximum 50). The resulting ceiling effect and accompanying limitations of the WISCI II for discriminating among high-functioning individuals would be expected to negatively affect correlations.
In one of the few studies specifically addressing chronic SCI, Scivoletto et al 9 reported significant WISCI correlations with MMT, UEMS, and LEMS. Significant correlations were also observed between the 10-m walk and MMT and LEMS but not UEMS. In this study, however, tetraplegic and paraplegic patients were grouped together for analysis, which complicates the interpretation of results. The durations postinjury were also not provided. The percentage of cervical patients (35%) was also lower than in our study. This would be expected to negatively affect the strength of observed correlations, and indeed correlations were less than those observed in our study.
In the absence of an intervention, parameters should be stable in chronic patients (reliability). This characteristic was demonstrated for the WISCI II. When evaluated on 2 separate occasions by the same research team, the level was unchanged at 97% and 95% for the self-selected and maximal WISCI, respectively. It is interesting to note that the only WISCI levels that showed fluctuations, from session 1 to 2, were 16 and 20. There was 100% agreement between the 2 testing sessions for all other WISCI levels.
The reproducibility of an instrument is a function of its test-retest reliability. Better reproducibility implies better precision of single measurements. In contrast, high variability is associated with poor reproducibility, and a larger difference is needed to detect a real change. SRD is the smallest measurement change that can be interpreted as a real difference and can be used to indirectly measure responsiveness of outcome measures. Because of excellent test-retest reliability, the SRD for self-selected and maximum WISCI was 0.785 and 0.597, respectively, which was less than 1 level. This means that a change of 1 WISCI level in a chronic patient can be interpreted as real. This property makes the WISCI II particularly useful for clinical trials for chronic SCI. In comparison, the SRDs for the 10-m walk were 0.254 m/s (self-selected) and 0.163 m/s (maximum). Given that the maximum observed walking speeds at the self-selected and maximum WISCI were 1.299 m/s and 1.266 m/s, respectively, individuals in our cohort would need to demonstrate changes of at least 19.6% (self-selected) and 12.9% (maximum) of the range in walking speeds to be confident that differences were real.
As with any study, there were limitations. The distribution of participants across WISCI levels was not equal; however, this is expected and has been previously described. 8,18 Furthermore, WISCI levels up to 5 require the use of parallel bars, and individuals requiring this level of support generally abandon ambulation following the completion of rehabilitation. Therefore, our inclusion criteria would have excluded such persons given the requirement to stand at least once per week. This prevents generalizability of the current findings to chronic SCI participants with very limited walking capacity. Little is known regarding the psychometric properties of outcome measures (timed walking or WISCI) in these low-functioning individuals, and future studies will need to address this issue. Regional differences in practice patterns could also affect observed WISCI levels. Indeed, differences have been described between North America and Europe; however, the hierarchical ranking and correlation of WISCI to impairment remained valid under these circumstances. 8 It should also be acknowledged that although the description of WISCI levels are self-explanatory, assessment should be performed by trained evaluators in the context of a clinical trial.
Conclusion
In the setting of interventions intended to improve strength following SCI, it is important that outcome measures for walking demonstrate a linkage between the underlying impairment (weakness) and related functional activity (walking). For chronic paraplegia and tetraplegia, the convergent validity of the WISCI II is supported by the correlations between LEMS and both self-selected and maximum WISCI levels. Less evidence was found for walking speed, where paraplegic participants failed to demonstrate a relationship with motor strength. When combined with excellent reproducibility (assigned WISCI levels), as demonstrated by the ICCs and the SRDs, our study results suggest that the WISCI II should be a very useful outcome measure for detecting changes in walking function following chronic SCI.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
This study was supported in part by the National Institute on Disability and Rehabilitation Research, Office of Special Education and Rehabilitation, US Department of Education, Washington, DC (Grant Number H133N060011).
