Abstract
Introduction
Ischemic spinal cord injury (iSCI) is considered to be less common than traumatic spinal cord injury (tSCI) 1 ; however, a recent study 2 reported that this condition represents the cause of about 20% of all nontraumatic lesions in the Western World (Europe and the United States). The admittance of patients with an ischemic event of the spinal cord to specialized spinal cord units occurs at a variable rate, between 1%2,3 and 8% 3 of all admissions.
Because of the relative rarity of these spinal lesions, there is a paucity of studies on the outcomes of ischemic lesions, in particular in comparison to tSCI. Most studies are based on the retrospective analysis of studies, with only a few involving more than 50 individuals and a limited set of outcome measures,4-16 which questions the representativeness of the obtained results. If valid conclusions are to be reached regarding differences in the recovery of iSCI and tSCI for clinical and research applications, then a rigorous comparison of a cohort with an adequate number of participants is warranted. For clinical purposes, the prediction of outcome after iSCI is important to guide clinical rehabilitative decision making (such as treatment methods, therapy duration, and goal setting). As patients with iSCI usually follow a rehabilitation regime originally designed for patients with tSCI, knowledge about differences in the rehabilitation potential of individuals with iSCI is important to decide on the validity of the same treatment approach. 17 Furthermore, precise knowledge about the course of recovery is essential to optimize the process of admitting patients from the clinical to the home environment.
For research purposes, the demonstration of similar occurrence and extent of neurological and functional recovery of iSCI compared with tSCI may permit inclusion of individuals with an iSCI into some clinical trials—for example, testing novel neuroregenerative 18 or neurorehabilitative approaches. 19
The aim of the following study is to quantify the extent and pattern of neurological and functional outcomes of iSCI in comparison to tSCI over the first year after SCI based on the analysis of a large prospectively documented representative European SCI cohort. The hypothesis is that, as it has been demonstrated for other nontraumatic spinal cord lesions, 20 the outcome of iSCI is not determined by the etiology but by other factors such as age, lesion level, and lesion severity. Once corrected by these factors, iSCI and tSCI would present the same amount and course of recovery.
Methods
Study Design and Patient Population
Data were derived from the European Multicenter Study about Spinal Cord Injury (EMSCI) database (http://www.emsci.org; ClinicalTrials.gov Identifier: NCT01571531). 21 Very briefly, EMSCI is a prospective longitudinal cohort study started in July 2001, conforming to the standards established by the Declaration of Helsinki and approved by the local ethics committees of all participating centers. Before their data were entered in the database, patients with acute tSCI or iSCI were thoroughly informed about the EMSCI database procedures and provided written informed consent to data recording. Assessments on their neurological and functional status were prospectively assessed per protocol within the first 15 days (very acute), between 16 and 40 days (acute I), and 3 months (acute II), 6 months (acute III), and 12 months (chronic) after SCI.
From the EMSCI electronic database, 22 we extracted the data of all patients aged 18 years or older, with a date of injury between July 2001 and April 2016. In particular, we screened the performance obtained by patients with either iSCI or tSCI within 3 time windows: within 45 days from date of injury (baseline/T0), within 3 months (T1), and within 6 months (T2).
Data on age, gender, and neurological level of injury (NLI; grouped as C1-C4, C5-C8, L, T1-T6, T7-T12) were collected at each time point along with other neurological and functional measures, derived from the neurological examination according to the most recent version of the International Standards for Neurological Classification of Spinal Cord Injury (ISNCSCI)23,24 and from the functional assessment according to the Spinal Cord Independence Measure versions II and III (SCIM 2/3). 25
The neurological assessment according to the ISNCSCI consisted of the assessment of Motor Scores of 10 key muscles per side of the body (total, Upper Extremity Motor Score [UEMS], Lower Extremity Motor Score [LEMS]) and light-touch (LT) and pin-prick (PP) sensation in all dermatomes of the body. According to these scores, the lesion severity is categorized into 5 American Spinal Injury Association Impairment Scale (AIS) grades (from AIS A, complete lesion defined by missing sensory and motor function in the most caudal sacral segment S4/5, to AIS E, normal sensation and motor function in all segments). Within the EMSCI database, AIS and all other classification variables are electronically calculated by a validated EMSCI-ISNCSCI calculator (http://ais.emsci.org). 26
The functional status of patients was assessed by SCIM clustered into 3 subscales: the self-care domain (with a score ranging from 0 to 20), including feeding, bathing, dressing, and grooming; respiration and sphincter management (score range 0-40), including respiration, bladder, and bowel management and use of toilet; and mobility (score range 0-40), including mobility in bed, transfers, mobility indoors and outdoors, and stair management. The total SCIM score ranges from 0 to 100, with higher scores reflecting higher levels of independence. 25
Finally, walking ability was evaluated by the Walking Index for Spinal Cord Injury II (WISCI II 27 ), a 0 to 20 scale that evaluates walking based on the need for physical assistance, braces, and devices, by the 10-Meter Walk Test (10MWT) 28 and the 6-Minute Walk Test (6MWT). 29
Statistical Analysis
Statistical analyses were performed with SPSS for Windows (version 21.0, Chicago, IL) and with R (version 3.3.1). Descriptive statistics were performed for all recorded measures, as mean (SD) for continuous measures and as frequency (percentage) for categorical measures.
The
However, after the first analysis of the entire group of patients and because of the strong size imbalance of the 2 groups (ie, 214 [6%] in the ischemic and 3301 [94%] in the traumatic group), a matching procedure was performed to correct for the concurrent effects of the different features of the 2 groups (ie, ischemic vs traumatic patients) on neurological recovery and functional rehabilitation outcomes.30,31
In particular, patients were matched according to AIS, NLI, and age at baseline (T0—ie, within 45 days from the date of injury). Gender was not considered in the matching procedure because it is well known that it neither affects the neurological nor the functional outcomes after SCI. 32
The match was exact for AIS and NLI, whereas for age, an interval within ±5 years was considered. After the matching procedure, 2 dependent samples from the original data were obtained, each composed of 191 patients (Figure 1 and Table 1).
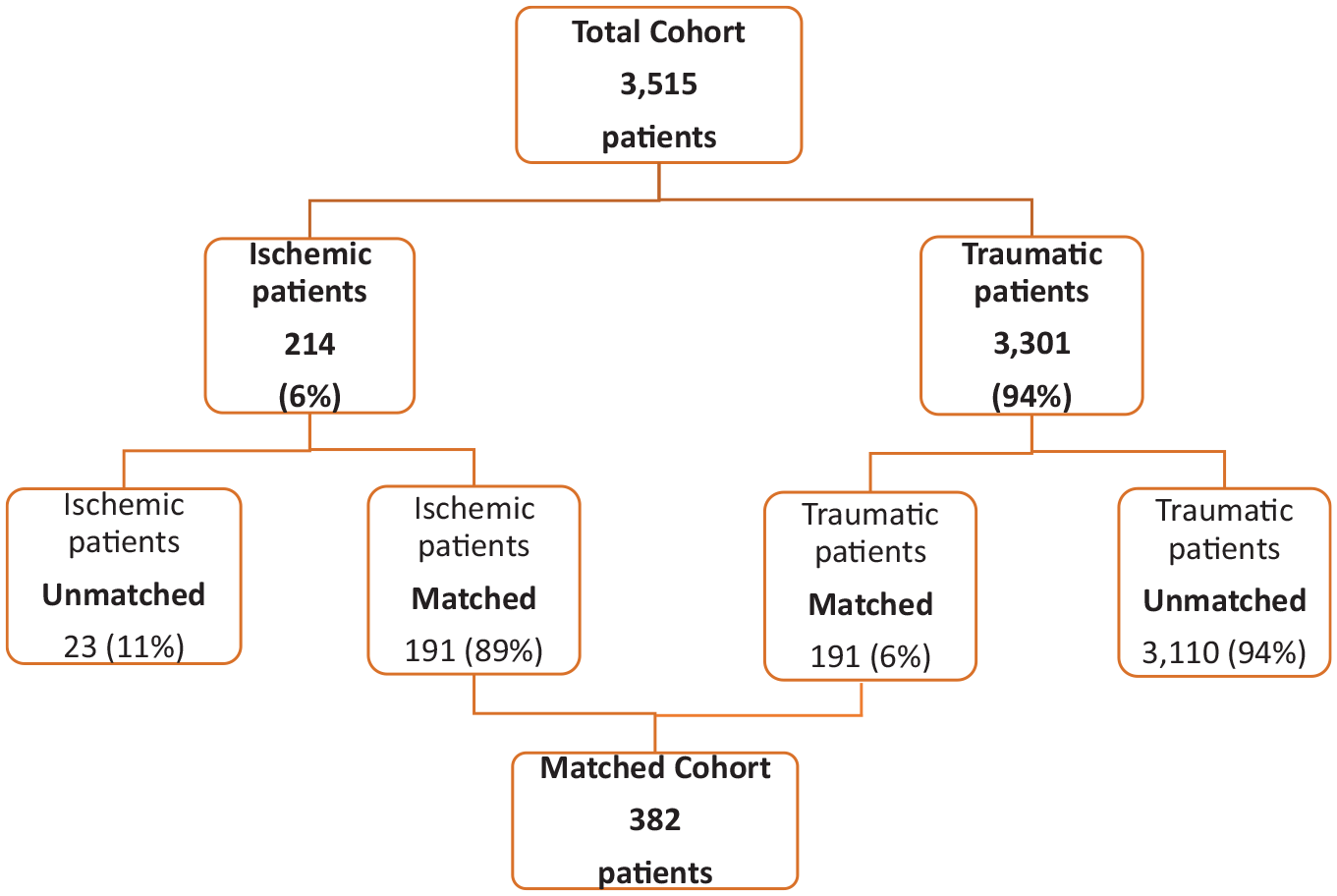
Flowchart describing the original cohort and the matched cohort.
Repeated-Measures ANOVA, Unmatched Cohort. a
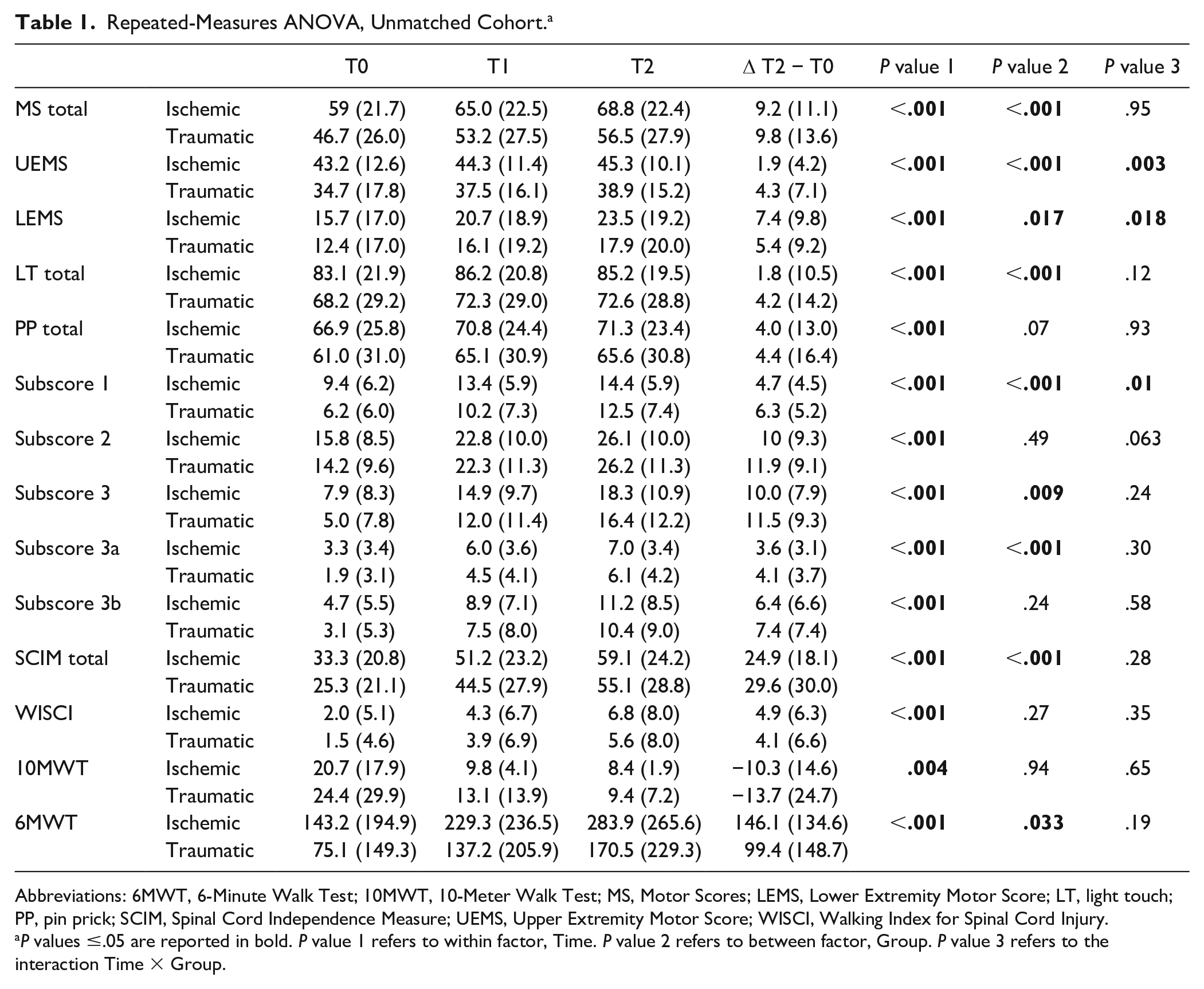
Abbreviations: 6MWT, 6-Minute Walk Test; 10MWT, 10-Meter Walk Test; MS, Motor Scores; LEMS, Lower Extremity Motor Score; LT, light touch; PP, pin prick; SCIM, Spinal Cord Independence Measure; UEMS, Upper Extremity Motor Score; WISCI, Walking Index for Spinal Cord Injury.
This matched cohort was again analyzed with a 2-way repeated-measures ANOVA (between factor: group, 2 levels [tSCI or iSCI]; within factor: time, 3 levels [T0, T1, T2]; outcomes: total, UEMS and LEMS, LT, PP, SCIM 2/3, WISCI II, 10MWT, and 6MWT. The aim of this analysis was to evaluate if the means of each score varied significantly over time and between groups. A
Results
During the selected period, the 25 EMSCI centers enrolled a total of 3515 patients (mean age 47.5 ± 18 years; 214 with iSCI and 3301 with tSCI) aged 18 years or older (2745 men) at baseline (Table 2). Ischemic and traumatic populations showed several differences. Ischemic patients were significantly older (57.6 ± 15.6 vs 46.9 ± 18.6;
Patient Characteristics at Baseline (T0, Within 45 Days From the Date of Injury). a
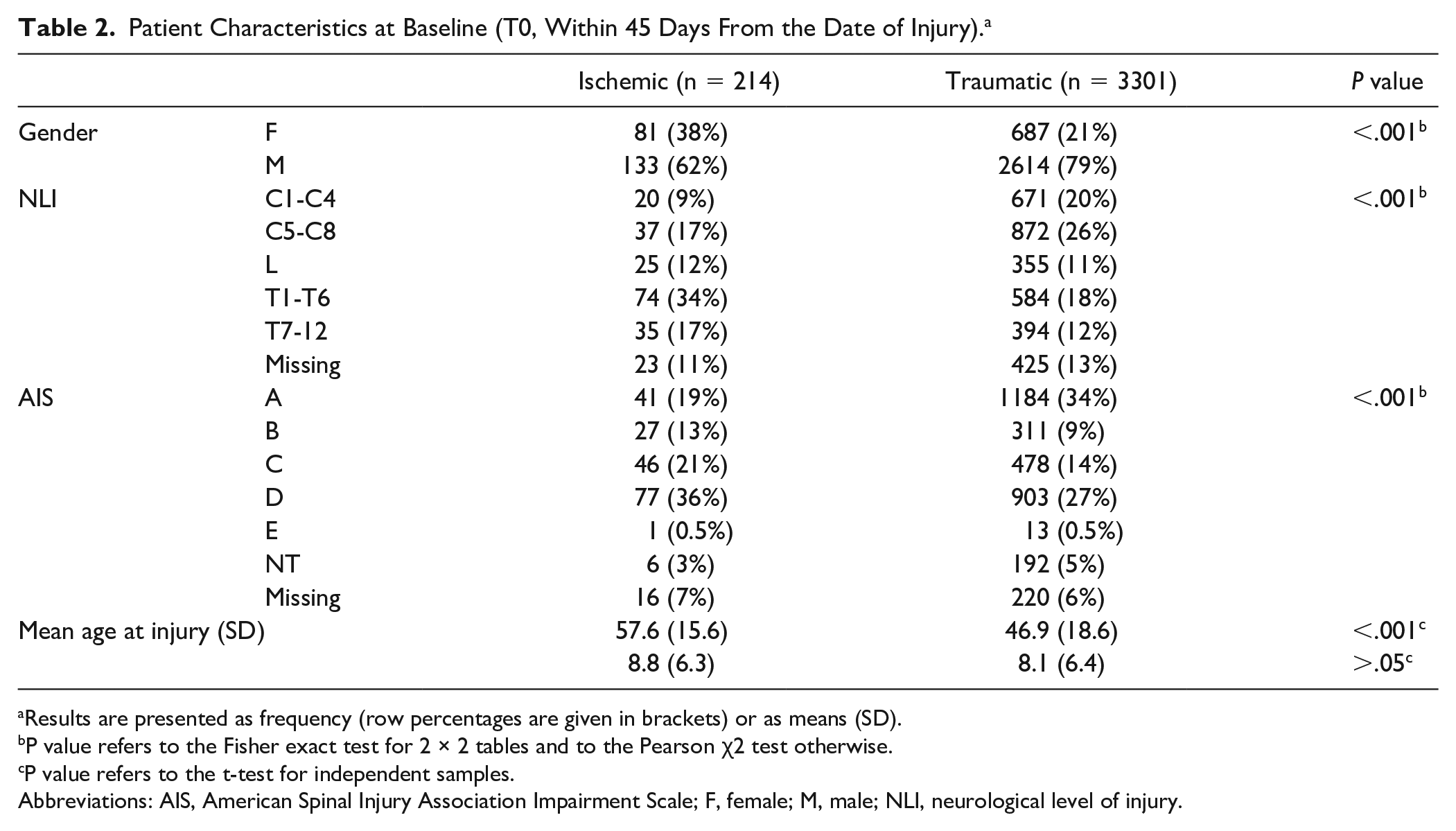
Results are presented as frequency (row percentages are given in brackets) or as means (SD).
Abbreviations: AIS, American Spinal Injury Association Impairment Scale; F, female; M, male; NLI, neurological level of injury.
The analysis of outcomes in the entire cohort of participants revealed that both groups (ischemic and traumatic) improved significantly from baseline (T0) to 6 months of follow-up (T2); however, ischemic patients had better neurological and functional status at T0 and at T1 and T2 (Table 1).
The matching procedure produced 191 pairs in respect to lesion level (29% tetraplegia and 71% paraplegia), lesion severity (21% incomplete), and age (56.6 ± 15.7 years in the ischemic and 56.9 ± 15.8 years in the traumatic group). Male gender was more frequent in the traumatic group compared with the ischemic group (Table 3). Mean distance between injury and first evaluation was 14.6 (10.7) days in the ischemic cohort and 14.5 (10.3) in the traumatic one.
Matched Cohort’s Characteristics at Baseline (T0, Within 45 Days From the Date of Injury). a
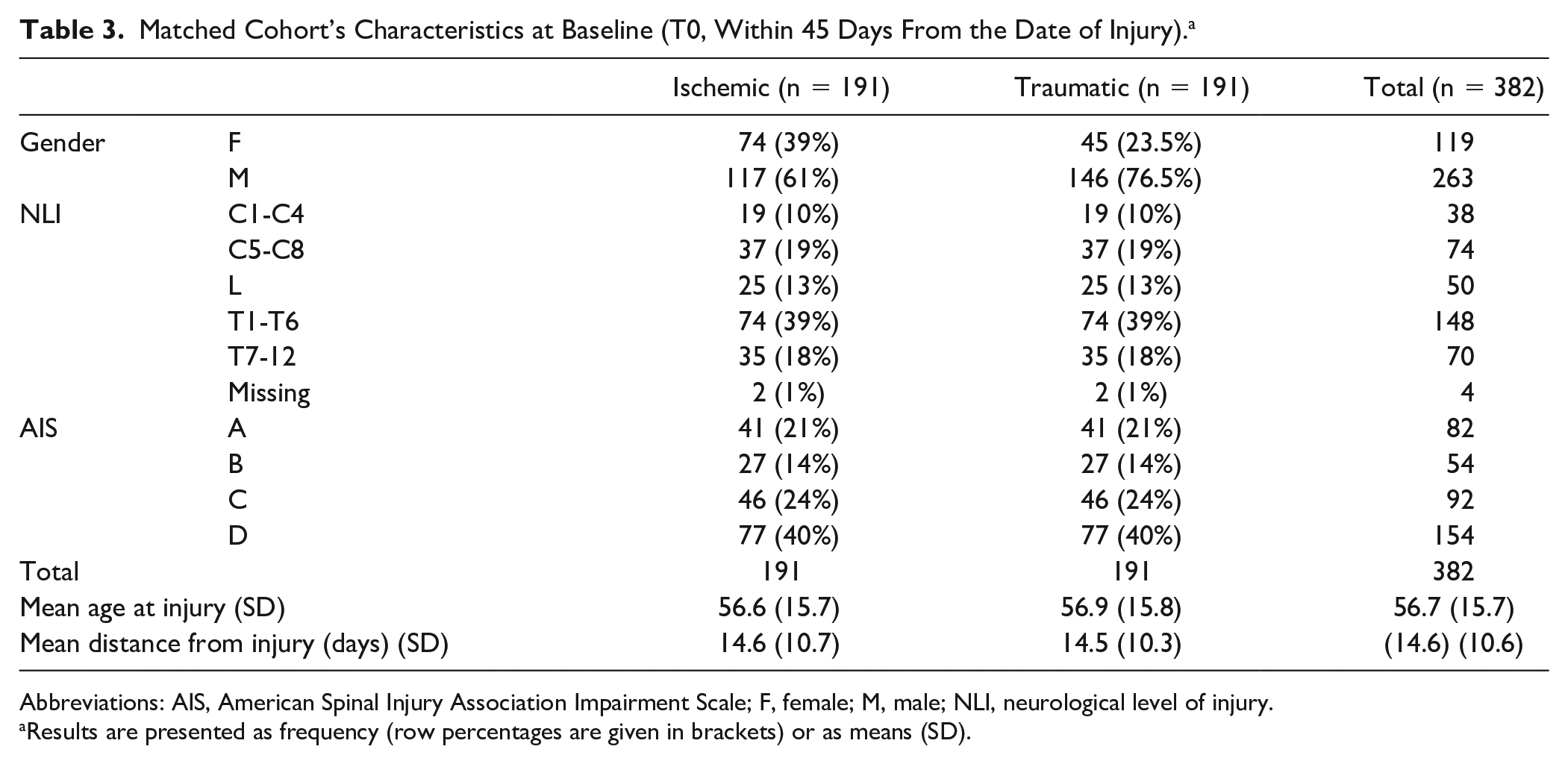
Abbreviations: AIS, American Spinal Injury Association Impairment Scale; F, female; M, male; NLI, neurological level of injury.
Results are presented as frequency (row percentages are given in brackets) or as means (SD).
The comparison of the matched cohorts revealed that the neurological status (motor and sensory scores) improved significantly in both groups over time between T0 and T2 (
Repeated Measures ANOVA, Matched Cohort. a
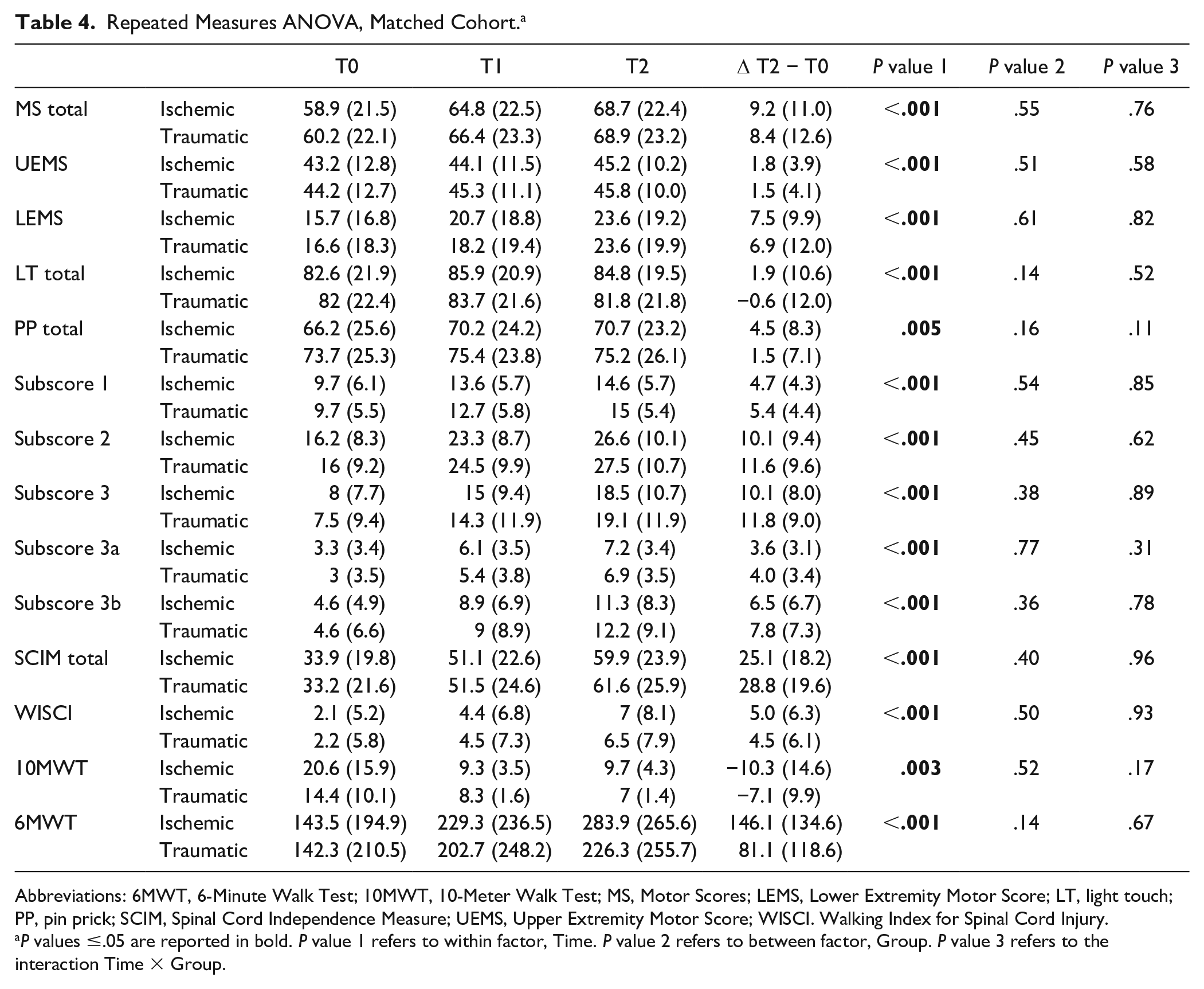
Abbreviations: 6MWT, 6-Minute Walk Test; 10MWT, 10-Meter Walk Test; MS, Motor Scores; LEMS, Lower Extremity Motor Score; LT, light touch; PP, pin prick; SCIM, Spinal Cord Independence Measure; UEMS, Upper Extremity Motor Score; WISCI. Walking Index for Spinal Cord Injury.
In the same way, the functional status (assessed by the SCIM) and the walking performance (assessed by WISCI II, 10MWT, and 6MWT) improved significantly in both groups between T0 and T2, but no differences were seen between iSCI and tSCI.
Discussion
With nontraumatic SCI receiving increased emphasis in recent years, etiologies that may mirror tSCI need to be identified for clinical and research purposes because they may complement our understanding of recovery following spinal cord damage.
The ischemic pathology of the spinal cord represents in the Western World (Europe and the United States) about 20% of all nontraumatic lesions 2 and is often secondary to aortic atherosclerosis or pathologies (with associated aneurysm and/dissection of the aorta) and surgical treatment required for aortic pathologies. 14 Other, less frequent etiologies are embolism, arteriovenous malformations, systemic hypotension, fibrocartilaginous embolism, vertebral artery dissection, coagulopathies, vasculitis, or decompression disease.14,15 However, a reasonable percentage of these lesions (20%-40%) is left to be called idiopathic while remaining without a clear explanation, especially in young individuals. 33 Patients with spinal cord ischemia typically present with a sudden onset of sharp pain (sometimes like a girdle around the chest) with rapid and progressive reduction of muscle strength accompanied by loss of bladder function and impaired nociception, whereas proprioception is relatively preserved (sensory dissociation). 6 Ischemic and traumatic spinal cord lesions share some underlying pathophysiological conditions. Since many years, it has been well known that a contusion of the cord inevitably induces ischemic/vascular damages. 34 Part of the secondary damage after a spinal cord contusion has an ischemic nature, with excitotoxicity phenomena, apoptosis, and calcium-mediated injury. 35 Both pathologies end with a serious loss of gray matter 36 and scar and cyst formation. 37 Ischemic and traumatic spinal cord lesions also share some neuroradiological features: it has been reported that lesion area, lesion length, and lesion width decreased over time and that, over time, intramedullary lesion changes are similar between patients with tSCI and iSCI. 38 Furthermore, the size of the lesion area and the presence and width of midsagittal tissue bridges early after traumatic and ischemic thoracic SCI have the potential to predict recovery after SCI, and this potential is the same in both etiologies. 38 With regard to the mechanisms of recovery, although not demonstrated, it is conceivable that the same phenomena of remyelination, neuroplasticity and neuromodulation 39 occur in both tSCI and iSCI. Furthermore, part of the recovery (at least of the functional one) is a result of compensation and search of new strategies related to the rehabilitation, which was the same for both iSCI and tSCI. 40
Based on these data, we compared 2 populations of individuals with iSCI or tSCI with 2 hypotheses: (1) iSCI and tSCI have the same amount and course of functional and neurological recovery and (2) recovery after iSCI and tSCI is not determined by the etiology but by other lesion features. To carry out this comparison, we examined the prospectively collected neurological and functional data of the EMSCI database.
With regard to the first hypothesis, in a matched cohort of 191 iSCI patients and 191 tSCI patients (Table 3), the 2 populations showed the same outcomes and the same time course of recovery (Figure 2). Whereas both populations showed significant improvements in all outcome measures between T0, T1, and T2, at each time point of our analysis, the neurological data, the independence of daily living, and walking capacity were comparable between the 2 groups (Table 4).
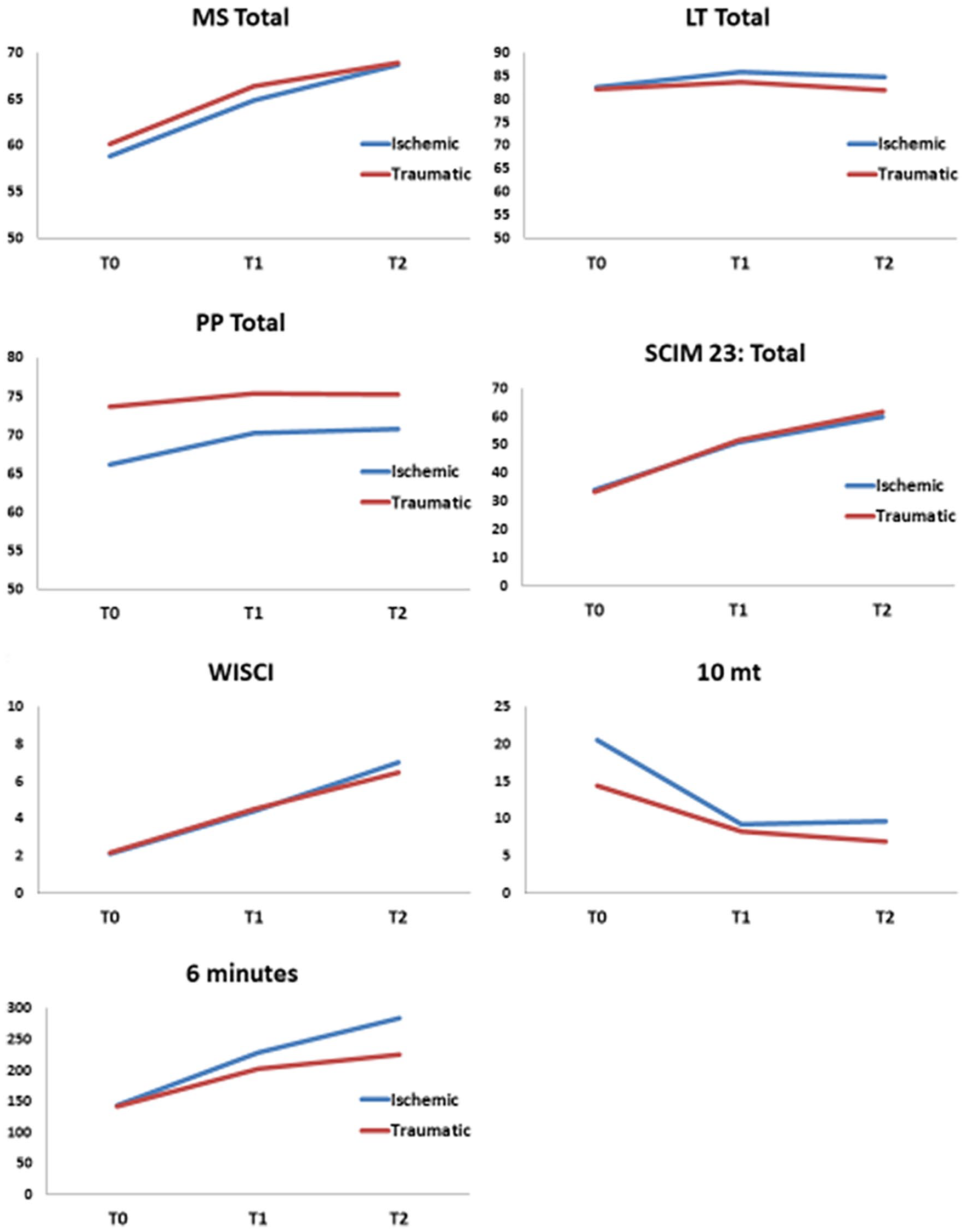
Course of recovery of the different outcomes.
To verify our second hypothesis, we compared the entire population of iSCI and tSCI of the EMSCI database (214 patients with iSCI and 3301 with tSCI). In agreement with previous reports, in the present study, patients with iSCI were older and more often presented with paraplegia and incomplete lesions compared with patients admitted with tSCI (Table 2). The evaluation of outcomes showed that, in this unmatched sample, patients with iSCI have better neurological and functional outcomes. However, after the matching procedure for lesion level and severity and age, outcome differences disappeared. This has been shown in other comparisons between tSCI and nontraumatic SCIs41,42 and demonstrates that etiology is not the major determinant of outcome. The outcome after an SCI, either traumatic or nontraumatic, is mainly dependent on age, level of lesion, and lesion completeness. With regard to age, it has been demonstrated that young SCI patients have better functional and perhaps neurological outcome than older ones. 43 This is probably because they have better fitness, higher vital capacity, and more motivation than their older counterparts. 44 Furthermore, it is well known that older patients have difficulties in turning neurological improvement into functional amelioration and deserve appropriate, specific training programs. 45 As for lesion completeness, it has been known for a long time that SCI patients with incomplete lesions (and in particular those with AIS B and C lesions) have a better chance of neurological recovery than those with complete lesions. 46 Finally, the effect of lesion level is intuitive: even in those with complete lesions, those with lower lesion level have better upper extremity functionality and better trunk control and could, therefore, reach a higher level of function. 47
Our results are comparable to those of other studies that evaluated the features and evolution of iSCI in comparison with tSCI by matching neurological variables. However, the majority of these studies are limited in terms of the number of involved participants, retrospective analysis of data, the choice of outcome measures, and the number of collecting centers. Iseli et al 5 demonstrated that iSCI and tSCI populations had comparable results in terms of neurological recovery (determined by AIS grade improvement) and walking ability (assessed by gross categorization in no ambulation/therapeutic ambulation/functional ambulation). However, the sample was small, was collected retrospectively, and was only from a single center. Furthermore, there was no correction for the confounding effect of lesion level and severity. Another report 10 compared the outcomes of acute iSCI paraplegic patients with tSCI patients. The neurological and functional recovery was measured by the ISNCSCI and SCIM, respectively, and linear regression analysis was used to correct for differences in injury characteristics. The authors concluded that etiology did not affect the outcomes, but this conclusion is based on a small sample of patients all treated in a single center. In another study 11 with a slightly larger sample size of 30 acute patients with iSCI, 18% of them tetraplegic, participants were matched for age, level, and completeness of injury. Similar to our results, no differences between the ischemia and trauma groups were found. However, the data were analyzed retrospectively over a 10-year period, and the Functional Independence Measure was used to determine functional recovery, which is less sensitive than the SCIM in capturing changes in patients with SCI. 48 In 2011, Scivoletto et al 13 compared a group of 68 individuals with iSCI with a cohort with tSCI. Using ANCOVA and logistic regression, they reported that etiology did not have an impact on the outcomes of their patients. However, in this case also the data were from a single center and were analyzed retrospectively, and there were a limited set of outcome measures, including the Barthel Index for assessing the functional outcomes, which has less validity and clinical utility for use in the SCI population. 25 One recent study 16 from 2 centers retrospectively collected data from 72 individuals with iSCI and compared them with those of 169 tSCI patients over a 5-year period; the difference of the total SCIM 3 scores from admission to discharge was analyzed as a measure of functional recovery. The patients were categorized according to the neurological level, and completeness of the lesion was assessed by the ISNCSCI examination; however, no detailed analysis of motor and sensory scores was performed. Differences with respect to age, neurological status, functional status, and length of stay were matched by multivariant analysis and linear regression. Their findings differ from our study in regard to the fact that patients with ischemic lesions showed lower levels of functional recovery than those with traumatic injuries. Although the study has many strengths such as the standardized assessments of neurological and functional status, such as those used in EMSCI, there were major drawbacks mainly related to the nonstandardized timing of examinations, which were only done at admission and discharge. It is possible that differences in lesion to admission time and in rehabilitation length of stay will alter the comparison of neurological recovery.
Compared with previous studies, our analysis has several strengths: it is the study with the largest number of patients with iSCI with no restrictions regarding severity and level of the spinal cord lesion. The data were collected prospectively in a multicenter setting (25 centers from all Europe), a factor that determines the possibility of generalizing the results. For this data analysis, full ISNCSCI data sets, which were obtained by trained examiners 49 and classified by a validated computer algorithm, were included, which has previously not been reported. Additionally, a large set of data on functional outcomes on both daily life activities and walking were analyzed. Furthermore, the EMSCI database provides a longitudinal collection of neurological status from the initial injury period to intervals of 3, 6, and then subsequent 12 months following injury, which has not been previously reported, allowing us to study the chronological trend of recovery of spinal cord lesions.
The study also has some limitations because of the nature of the database. Compared with previous studies, we were not able to analyze the impact of the presence and frequency of complications, the length of acute and rehabilitation stay, and the discharge dispositions because these data are not collected within the EMSCI study.
Additionally, the EMSCI database does not subclassify iSCI, in particular, in terms of spontaneous versus iatrogenic etiology, which could be relevant because patients with iatrogenic ischemia frequently have highly significant comorbidities, which may affect functional outcome.
Furthermore, it is well known that ischemic and traumatic brain injury show differences in time courses during the first month after injury. This could be the case also in the very acute phase (within the first 15 days) after SCI. In particular, it is probable that most patients with tSCI in this study would have had some surgical intervention, and that surgery as well as the initial pharmacological treatment of tSCI may have altered the course of the neurological and, consequently, functional outcomes. In the same way, although it is possible to assume that the progress in recovery will be similar between the 2 groups after the 6-month evaluation, we could not be sure of this trend. Both issues require further specific studies.
Conclusions
The results of our study demonstrated that patients with iSCI or tSCI have the same amount and course of recovery and same rehabilitation potential of tSCI and, therefore, could undergo the same treatment approach from 15 days following the event up to a further 6 months. This allows clinicians to apply the prognostic knowledge of tSCI to iSCI during the rehabilitation phase and to guide clinical rehabilitative decision making.
To date, patients with ischemic spinal lesions have not been considered as suitable participants in clinical trials because of the belief that the extent of recovery is different when compared with patients with tSCI. Our results demonstrate that from 15 days following the event onward, patients with ischemic lesions have a course of neurological and functional recovery similar to those with tSCI and are, therefore, suitable candidates for further interventional studies (most of all neuroregenerative or neurorehabilitative approaches).
Footnotes
Acknowledgements
We would like to thank Prof John Ditunno for his invaluable inputs and support of the manuscript. We are grateful to the support by the EMSCI network.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The article is partially supported by the European Union’s HORIZON2020 research and innovation programme Grant No. 681094 to Armin Curt and by the ERANET-NEURON grant to Giorgio Scivoletto.
