Abstract
Objective. To test the hypothesis that an anti-inflammatory corticosteroid drug enhances spinal motor plasticity induced by acute intermittent hypoxia (AIH) in persons with chronic incomplete spinal cord injury (iSCI). Methods. Fourteen subjects with incomplete spinal cord injury (ASIA level C or D; mean age = 46 years) participated in a randomized, double-blinded, crossover, and placebo-controlled study. Subjects received either 60 mg oral prednisolone or a matching placebo, 1 hour before administration of AIH (15, 60-second hypoxic exposures; fraction of inspired oxygen [FiO2] = 0.09). Changes in voluntary ankle strength, lower extremity electromyograms (EMG), and serum inflammatory biomarkers were quantified. Results. Maximal ankle plantarflexion torque was significantly higher following prednisolone + AIH versus placebo + AIH (mean difference [MD] 9, 11, and 7 newton meter [N∙m] at 30, 60, and 120 minutes post-AIH, respectively; all Ps <.02). Soleus surface EMG during maximal voluntary contraction was also significantly increased following prednisolone + AIH (MD 3.5, P = .02 vs placebo + AIH), while activity of other leg muscles remained unchanged. Individuals had significantly higher levels of the anti-inflammatory serum biomarker interleukin-10 after prednisolone versus placebo (P = .004 vs placebo + AIH). Conclusions. Pretreatment with prednisolone increased the capacity for AIH-induced functional motor plasticity, suggesting that suppression of inflammation enhances the efficacy of AIH administration in individuals with spinal cord injury. Clinical trial registration number: NCT03752749.
Introduction
Harnessing neuroplasticity in persons with incomplete spinal cord injury (iSCI) remains an important quest for promoting recovery and enhancing rehabilitation. One unique and noninvasive approach to induce plasticity in spinal networks is via exposure to brief episodes involving inhalation of hypoxic gas mixtures, alternating with inhalation of ambient room air (known as acute intermittent hypoxia or AIH). In animal models, this protocol triggers the release of spinal serotonin, and increases the synthesis of plasticity-related proteins throughout the neuraxis, resulting in increased synaptic strength and improved motor function in people with chronic iSCI.1-3
Several recent human studies demonstrate that AIH induces plasticity in spinal pathways regulating limb muscle function, and represents a safe and noninvasive approach to supplement conventional neurorehabilitation after SCI.4-6 Although AIH holds promise as a priming tool for rehabilitation, recent reports in animal models indicate that systemic inflammation—a condition prevalent in persons with SCI7-10—can undermine hypoxia-induced spinal neuroplasticity. Even low-grade systemic inflammation inhibits the ability of AIH to induce respiratory motor plasticity in rodent models.11-13 Subsequent administration of anti-inflammatory drugs reverses this impairment. 14
The objective of this study was to determine if administration of a corticosteroid drug, prednisolone, prior to a single AIH session would enhance functional gains in motor output in persons with chronic iSCI. It was hypothesized that pretreatment with this anti-inflammatory agent would decrease baseline inflammatory activity, potentially enhancing the capacity of AIH to induce functional motor plasticity.
Methods
Subjects
All experiments were performed at the Rehabilitation Institute of Chicago (now known as the Shirley Ryan AbilityLab), an academic affiliate of Northwestern University. The study protocol was approved by Northwestern University’s Institutional Review Board, and informed consent was obtained from all study subjects (ClinicalTrials.gov ID = NCT03752749).
Subjects were recruited through the hospital’s Clinical Neuroscience Research Registry, via research flyers and by word of mouth. Fourteen subjects between the ages of 18 and 75 years were enrolled. The sample size was determined based on our experience with a similar and previously published study. 4 Based on a retrospective power analysis, a standardized effect size of δ = 0.9 can be detected with 80% power and 5% type I error rate in this crossover study, based on a paired t test comparing changes in torque (from Pre−120 to Post60) resulting from prednisolone versus placebo treatment for each subject. Subjects were English speaking, and had a history of chronic, motor-incomplete SCI at C4 or below, graded as American Spinal Injury Association Impairment Scale (AIS) C or D. An ability to achieve at least 30° voluntary plantarflexion in at least one ankle joint was also required. Exclusion criteria included: (1) tracheostomy, lower extremity peripheral nerve injury, significant pulmonary or cardiac disease, uncontrolled diabetes, or significant osteoporosis with post-SCI fracture; (2) inflammatory conditions such as pressure ulcers, active infections, heterotopic ossification, rheumatologic disease, cancer, or deep venous thrombosis; and (3) any prior adverse reaction to steroids and current use of steroids, disease-modifying anti-rheumatic drugs, intrathecal baclofen, or other medications known to affect actions of prednisolone. All subjects who met the inclusion/exclusion criteria were required to suspend all anti-inflammatory drugs and oral antispasticity agents for a minimum of 14 days prior to testing. Clinical and demographic profiles of the subjects are listed in Table 1.
Demographic Subject Characteristics.
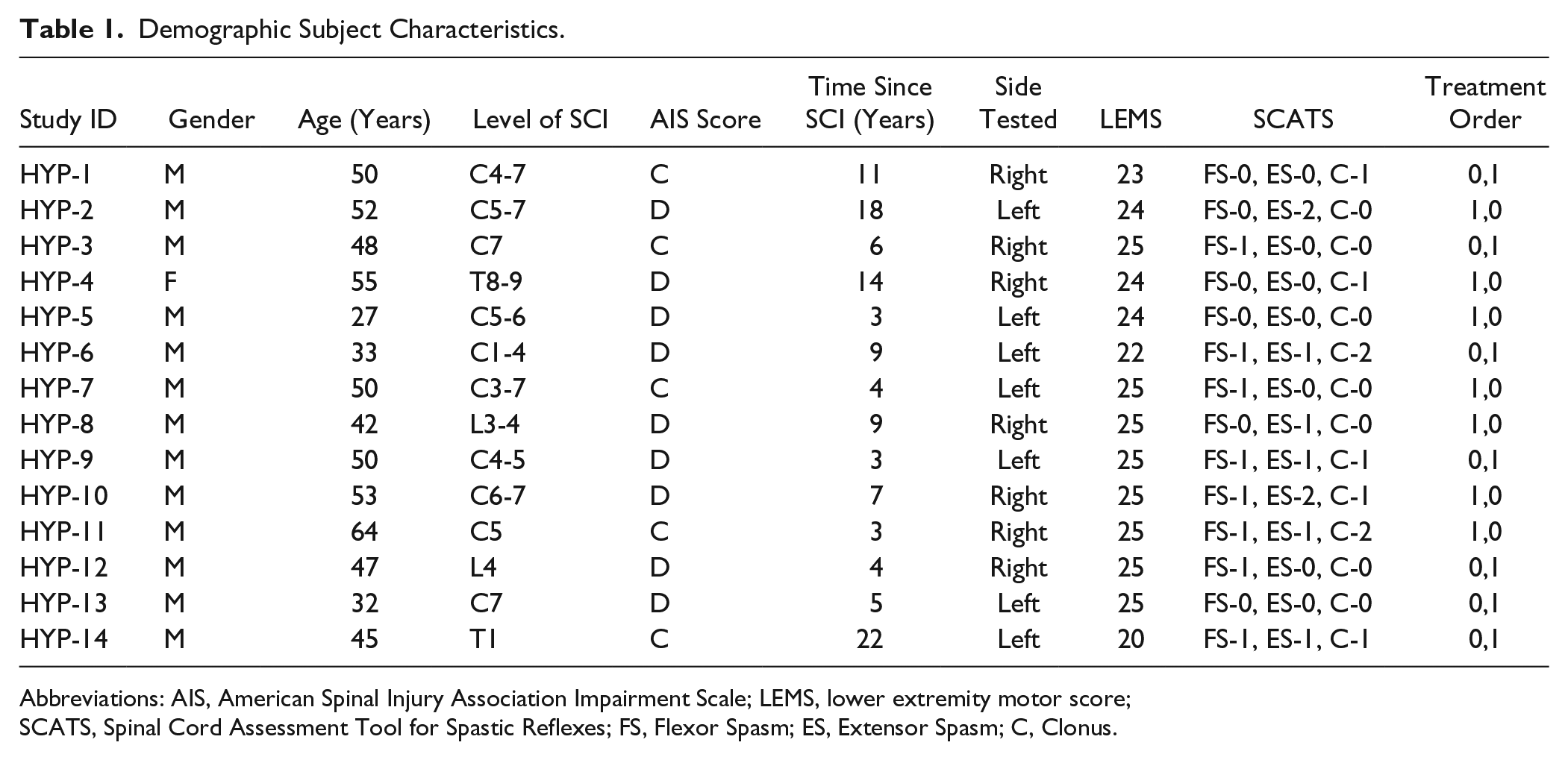
Abbreviations: AIS, American Spinal Injury Association Impairment Scale; LEMS, lower extremity motor score; SCATS, Spinal Cord Assessment Tool for Spastic Reflexes; FS, Flexor Spasm; ES, Extensor Spasm; C, Clonus.
Study Design
A randomized, double-blind, crossover, and placebo-controlled study design was utilized. Each subject visited the laboratory on two different occasions (study periods), at least 1 week apart, and received either 60 mg oral prednisolone (oral solution, 15 mg/5 mL, Hi-Tech Pharmacal Co, Amityville, NY) or a flavor-matched placebo (Ora-sweet, Paddock Laboratories, Perrigo Company, Allegan, MI). The prednisolone and placebo solutions were dispensed by a research pharmacist and administered by a research nurse. Both subjects and researchers were unaware of the order in which the study-agent was given. One hour after administration of the study agent, subjects underwent the AIH intervention (15, 60-second hypoxic exposures; fraction of inspired oxygen [FiO2] = 0.09). Outcome measures were assessed prior to delivery of the study-agent (Pre−120), prior to AIH intervention (Pre−60), and at 0, 30, 60, 90, 120, and 180 minutes post-AIH intervention (Post0 – Post180, Figure 1).
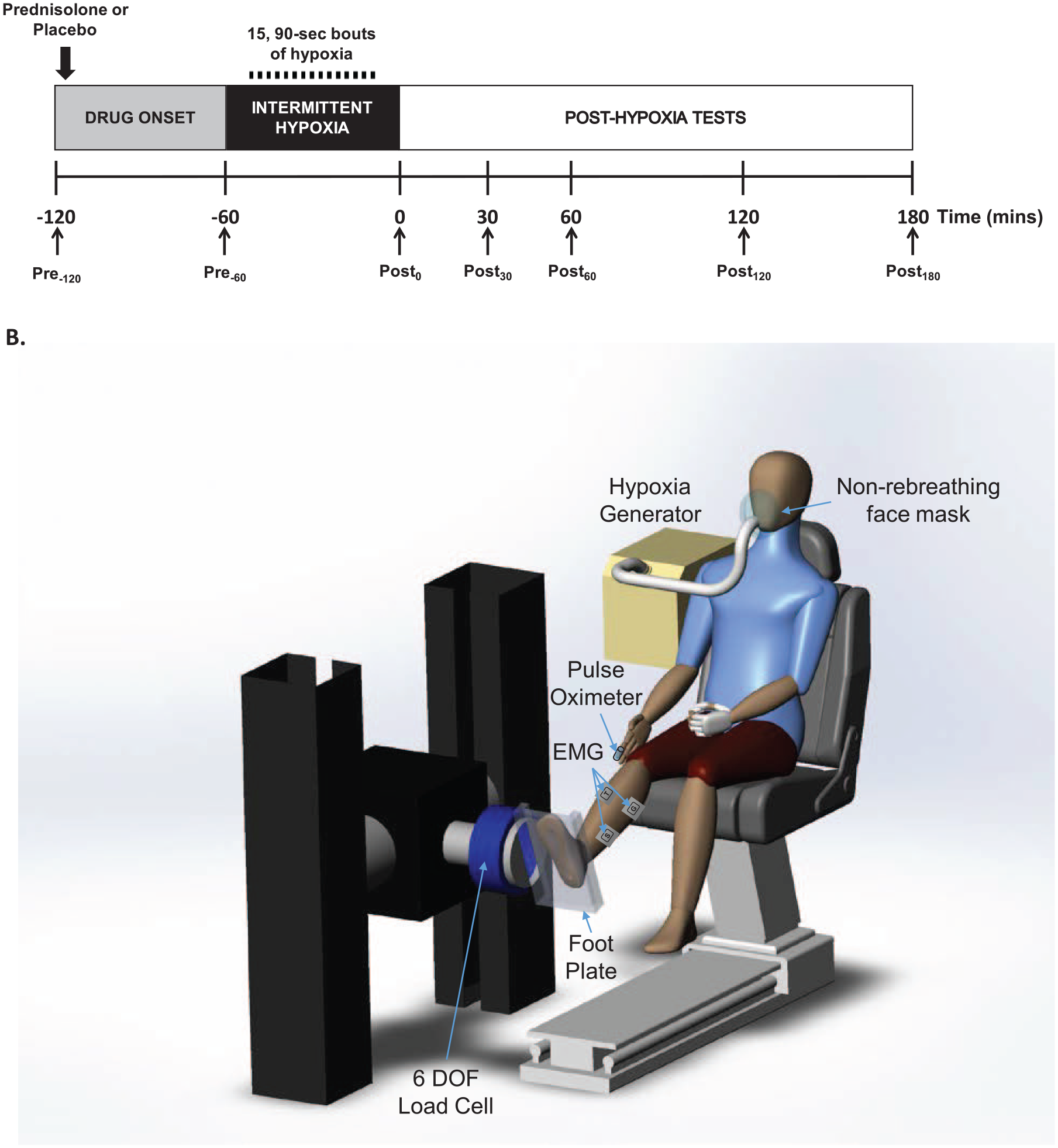
Study experimental design and setup. (Panel A) Ankle plantarflexion strength and surface electromyography was assessed prior to administration of prednisolone or placebo (Pre−120), prior to acute intermittent hypoxia (AIH; Pre−60), and at 0, 30, 60, 90, 120, and 180 minutes post-AIH (Post0 – Post180). The AIH protocol consisted of 15, 60-second alternating episodes of inspiring hypoxic air (~9% O2) with ambient room air (20.9% O2). Blood samples were taken before the start and at the end of each experiment. (Panel B) Subjects were seated and secured in an adjustable chair with the ankle strapped to the footrest and the thigh and trunk strapped to the chair. The seat and footrest were adjusted to align the ankle axis of rotation with the axis of the force sensor and the motor shaft. A 6 degrees-of-freedom load cell mounted on the motor head was used to record ankle torque generation. Electromyograms were recorded from soleus, medial gastrocnemius, tibialis anterior and quadriceps using bipolar surface electrodes. Oxygen saturation and pulse rate were recorded continuously while subjects received acute intermittent hypoxia intervention using non-rebreather face mask.
Experimental Setup and Delivery of AIH
On arrival to the laboratory, subjects underwent a screening interview during which basic clinical and demographic information was obtained. Lower extremity motor scores and Spinal Cord Assessment Tool for Spastic Reflexes (SCATS) scores were also measured prior to the intervention. Subjects were then seated in an adjustable chair and the designated foot was mounted on a footplate attached to a 6 degree-of-freedom load cell. Seating posture was maintained using padded straps across the chest, waist, and knee, as well as forefoot and ankle straps to secure the foot in place. The test side was positioned with 80° hip flexion, 10° knee flexion, and 0° plantarflexion (Figure 2). Surface electromyogram (EMG) electrodes were placed over medial gastrocnemius, soleus, quadriceps, and tibialis anterior muscles, according to Surface ElectroMyoGraphy for the Non-Invasive Assessment of Muscles (SENIAM) guidelines and secured with tape. One hour after administration of prednisolone or a placebo, AIH treatment was initiated using an oxygen generator (Model HYP-123, Hypoxico Inc, New York, NY, USA). Subjects were fitted with a non-rebreather mask; 15, 60-secon periods of hypoxia, alternating with 60-second periods of normoxia (21% O2). Continuous pulse oximetry (Smith Autocorr Plus, Smiths Medical, Dublin, OH, USA) was utilized to ensure target oxygen saturation of 82% to 85% during the hypoxia episodes.
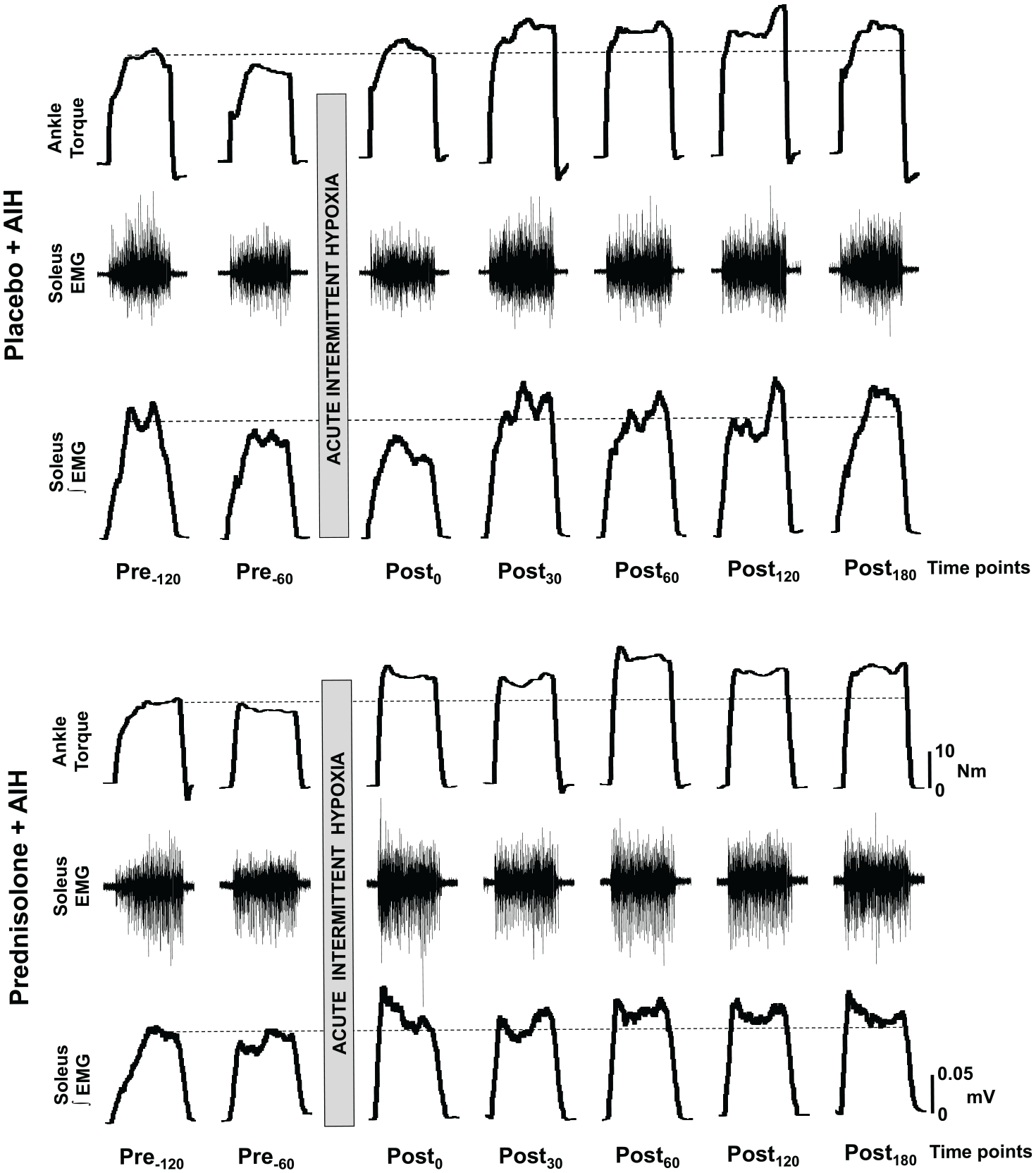
A representative example illustrating the impact of orally delivered study agent (prednisolone or placebo), prior to acute intermittent hypoxia (AIH), on ankle flexion strength and corresponding electromyogram (EMG) activity. Impact of placebo + AIH (panel A) and prednisolone + AIH (panel B) on maximal ankle plantarflexion torque (top row), raw EMG activity (middle row) and integrated EMG activity (bottom row) in a spinal cord injury subject are shown. Time points are in minutes prior to (Pre) or after (Post) AIH administration. N·m = newton meter; mV = millivolt.
Outcome Measures
Isometric Ankle Torque
The primary outcome measure was ankle plantarflexion strength. Subjects were secured in the chair and instructed to push against the dynamometer foot plate as hard as they could for a total of 5 seconds. Maximal voluntary contraction (MVC) during isometric ankle plantarflexion was assessed prior to administration of study-agent (Pre−120), prior to AIH (Pre−60), and at 0, 30, 60, 90, 120, and 180 minutes post-AIH (Post0 – Post180). A total of 3 trials were obtained at each time point, with a minimum of 1-minute rest in between. The resultant ankle torque was sampled at 1 kHz with 200-Hz low-pass filter, using an 18-bit analog-to-digital DAQ (NI PCI-6289; National Instruments, Austin, TX, USA) on a PC. Custom LabVIEW analysis software (National Instruments, Inc) was utilized for data acquisition and analysis. The mean torque of the 3 trials was used for analysis.
Electromyogram Recording
Surface EMG was obtained over the quadriceps, medial gastrocnemius, soleus, and tibialis anterior to confirm torque measurements. The Bagnoli-16 system was used to collect EMG data, sampled at 2000 Hz and synchronized with torque data using the analog-to-digital converter, amplified 1000 times and filtered with a bandwidth of 20 to 450 Hz. EMG data were processed and integrated off-line using Spike2 (Cambridge Electronic Design Limited, Cambridge, UK) software, and rectified and smoothed (20 ms smoothing window) before quantification. Peak EMG activity was computed from a 50-ms time window, 150 ms preceding the maximum absolute value of the torque signal for each contraction. Average peak EMG activity for each muscle was obtained across the 3 MVCs.
Serum Cytokine Measurement
To assess the relationship of serum inflammatory levels following prednisolone or placebo treatment with response to AIH, we analyzed the serum levels of inflammatory markers. Blood draws were obtained by a phlebotomist before the administration of agent, and at the end of each intervention (following Post180 time point). The serum samples were assessed using a high sensitivity immunoassay (9-Plex Human ProcartaPlex Panel, ThermoFisher Scientific). These assays were processed using Luminex xMAP technology for detection of the following proteins: interferon-γ (IFN-γ); interleukin-1β (IL-1β); IL-2; IL-4; IL-6; IL-10; IL-12p70; IL-17A; tumor necrosis factor–α (TNF-α).
Other Outcome Measures
Blood pressure was measured using an electronic blood pressure cuff, recorded before and immediately after the AIH. During and after AIH treatment, subjects were monitored for headaches, pain, lightheadedness, dizziness, respiratory distress, cyanosis, spasms, and autonomic dysreflexia, as reported by the subject or observed by the study administrator.
Statistical Analyses
Torque measurements were taken before the prednisolone or placebo administration (Pre−120), immediately prior to AIH (Pre−60), and at 0, 30, 60, 120, and 180 minutes post-hypoxia (Post0 − Post180). The primary endpoint was the relative change in torque from Pre−120 to Post60. The secondary outcome was relative change in surface EMG evaluated at the same time points.
For the initial analysis, the relative change from Pre−120 to subsequent time points was calculated for each subject during each treatment period. These changes were summarized as mean ± standard error (SE); Wilcoxon signed rank tests were used to compare within-subject changes during the prednisolone and placebo treatment periods for each time point.
Because this study utilized a crossover design, further analyses were conducted to account for the randomization of subjects to treatment sequences. Based on the comparison of different approaches for the analysis of crossover studies with baseline measurements, 15 model VII was chosen. This generalized linear mixed model, denoted by (Y1|X1) – (Y2|X2), estimates the difference in response between the 2 treatment periods at a given posttreatment time point, uses period-specific baseline measurements as covariates, and accounts for the within-subject correlation between treatment periods. Here Xj and Yj are the baseline and postbaseline responses in period j (j = 1, 2), respectively. The models were estimated using PROC MIXED in SAS statistical software using Kenward-Roger degrees of freedom adjustment. 15
For the primary outcome of torque, the model used posttreatment torque in periods 1 and 2 as the outcome variable, and predictors included period (1 and 2), treatment sequence (prednisolone/placebo and placebo/prednisolone), treatment in the period (prednisolone or placebo), baseline torque in the period, and the interaction between period and baseline torque. Treatment period was used as the repeated measure, and compound symmetry covariance structure was used to properly account for the within-subject correlation between the 2 periods. Separate models were fitted for each postbaseline time point. In addition, a more comprehensive analysis using an extension of this model was performed, this time including all time points together in a single repeated measures model. Surface EMG and the inflammatory biomarkers were analyzed using similar models.
Data Availability Statement
Anonymized data can be shared by request from any qualified investigator to the corresponding author.
Results
All subjects completed both experimental sessions and no adverse clinical events were recorded. Blood sample for 1 subject (S08) could not be collected due to technical difficulties. Demographics, lower extremity motor scores (LEMS) for the subject’s tested leg, Spinal Cord Assessment Tool for Spastic Reflexes (SCATS) and treatment order for all subjects are provided in Table 1.
Isometric Torque
Maximal voluntary torque increased significantly following AIH in both conditions, that is, with prior administration of placebo or prednisolone. A representative example of MVC recordings before and after AIH in each group is shown in Figure 2. Individual trajectories of changes over time, along with the average (±SEM), are shown in Figure 3. Table 2 shows average relative changes in torque during the 2 treatment periods, as well as the average of within-subject differences in relative torque change, along with the results of the Wilcoxon signed-rank test. Relative increases in torque were statistically significantly larger at Post30, Post60, and Post120 time points (P = .0012, P = .0004, and P = .0067, respectively), with the largest difference of 21.8% ± 4.4% observed at Post60.
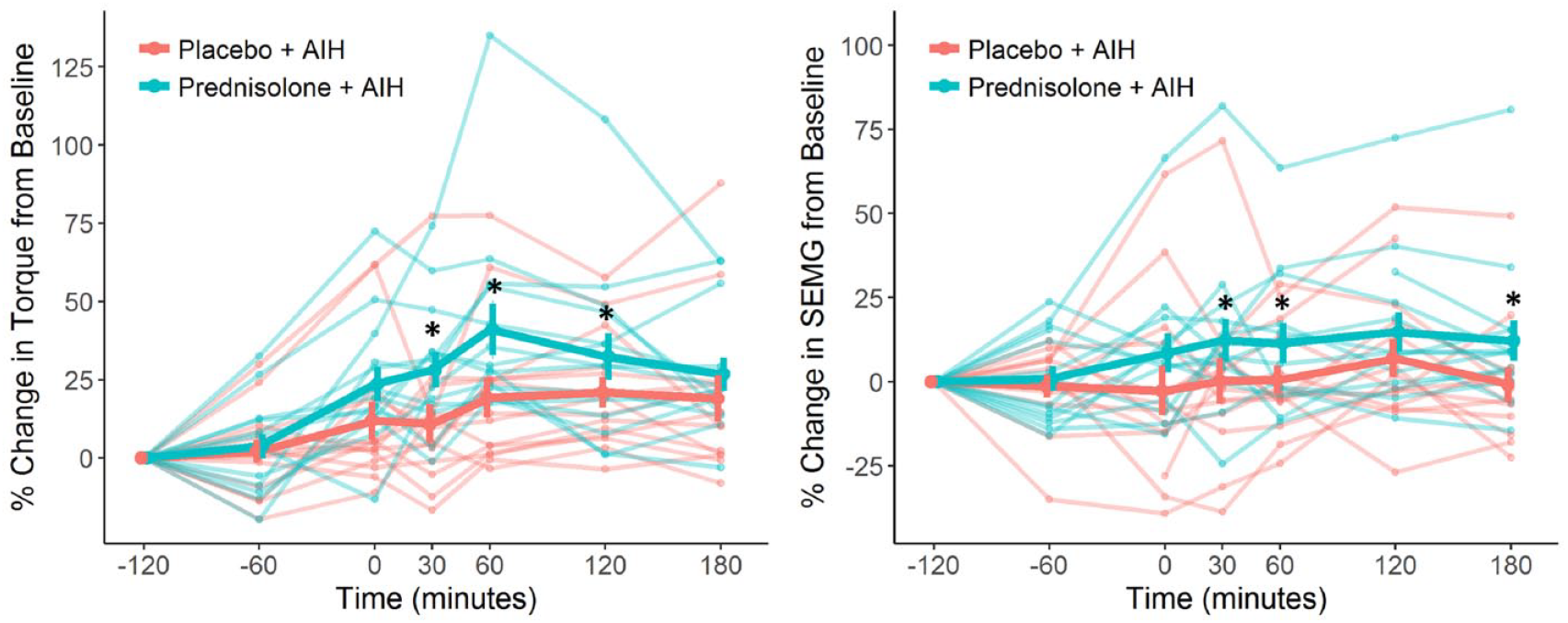
Changes in ankle torque and soleus electromyogram (EMG). Individual and mean changes in maximal ankle torque (panel A) and soleus EMG activity (panel B) following placebo + acute intermittent hypoxia (AIH) and prednisolone + AIH protocol are presented. Data points represent percent change from baseline (ie, Pre−120). Thin lines represent individual subjects, and the thick lines represent the mean values, with error bars representing standard error of the mean. *, P values <.05 vs. Placebo + AIH.
Average Relative Changes in Ankle Plantarflexion Torque. a
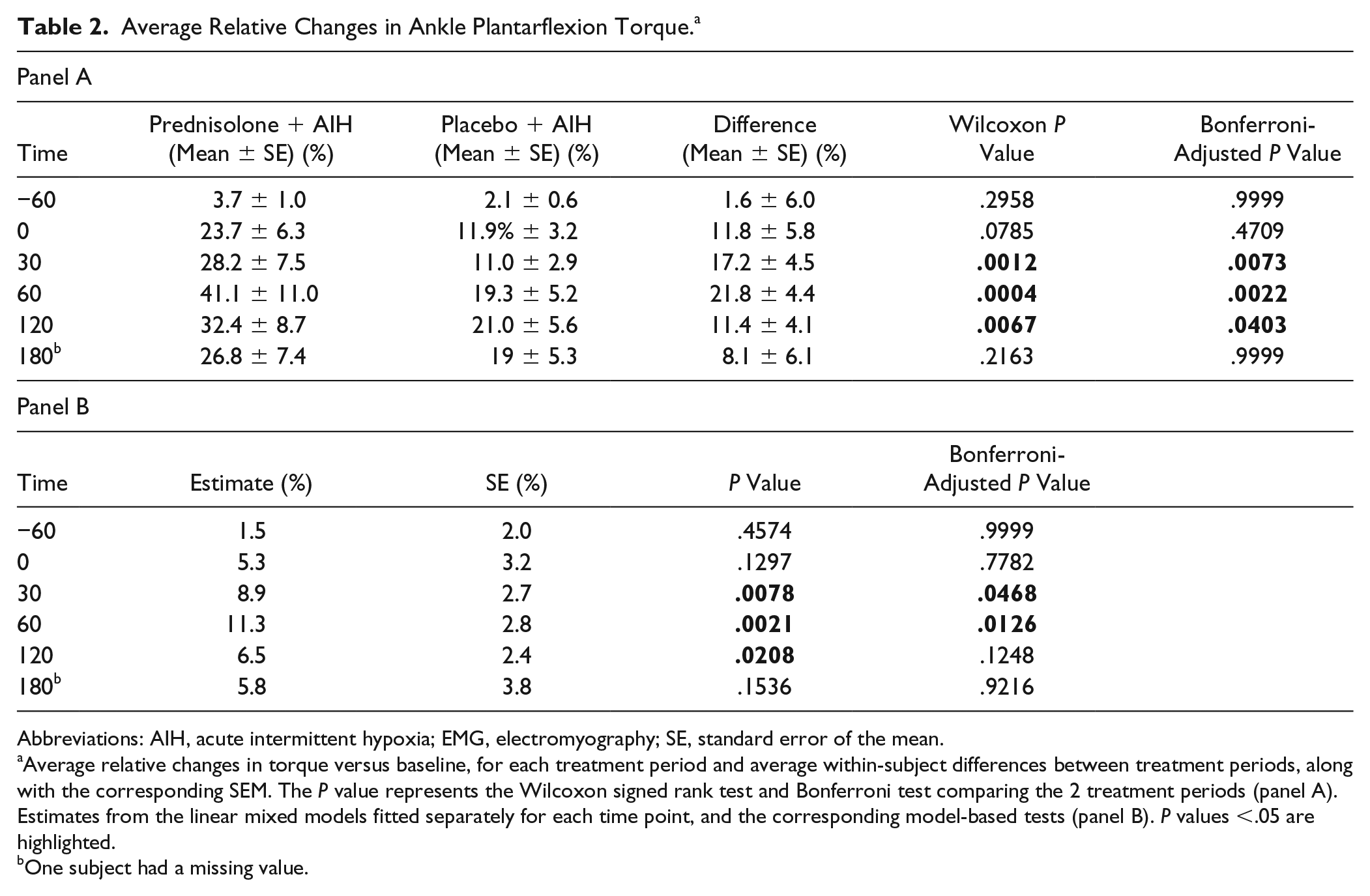
Abbreviations: AIH, acute intermittent hypoxia; EMG, electromyography; SE, standard error of the mean.
Average relative changes in torque versus baseline, for each treatment period and average within-subject differences between treatment periods, along with the corresponding SEM. The P value represents the Wilcoxon signed rank test and Bonferroni test comparing the 2 treatment periods (panel A). Estimates from the linear mixed models fitted separately for each time point, and the corresponding model-based tests (panel B). P values <.05 are highlighted.
One subject had a missing value.
The results of the repeated measures model comparing torque between prednisolone + AIH and placebo + AIH treatments at each post-baseline time point, adjusted for baseline (torque at Pre−120), treatment sequence, period, and their interaction, are summarized in Table 2, panel B. These findings are consistent with the analyses using Wilcoxon signed-rank test, and also show statistically significant differences between prednisolone + AIH and placebo + AIH treatment periods at Post30, Post60, and Post180. However, the adjusted model-based differences were smaller in magnitude, with the largest magnitude of difference observed at Post60 (11.3% ± 2.8%; P = .0021). The results from the linear mixed model including all time points were also consistent with these findings.
Electromyography Response
To quantify changes in muscle activity, EMG signals from the soleus, medial gastrocnemius, tibialis anterior and quadriceps were recorded during each MVC. A representative example of a raw and integrated EMG signal from the soleus is shown in Figure 3. Compared with placebo + AIH, the EMG activity was significantly higher in the soleus muscle 60 minutes following prednisolone + AIH intervention (
Average Relative Changes in Soleus EMG. a
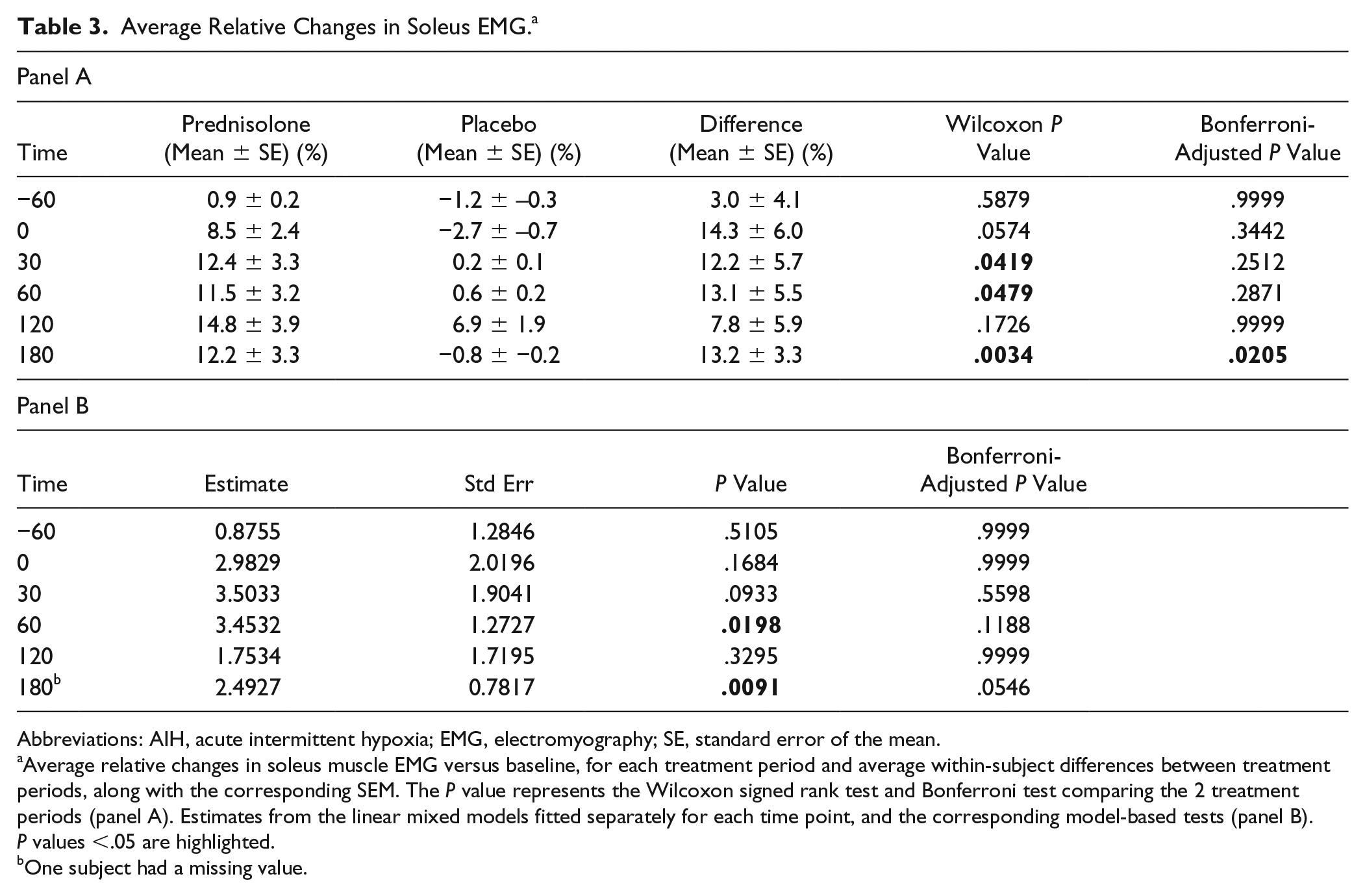
Abbreviations: AIH, acute intermittent hypoxia; EMG, electromyography; SE, standard error of the mean.
Average relative changes in soleus muscle EMG versus baseline, for each treatment period and average within-subject differences between treatment periods, along with the corresponding SEM. The P value represents the Wilcoxon signed rank test and Bonferroni test comparing the 2 treatment periods (panel A). Estimates from the linear mixed models fitted separately for each time point, and the corresponding model-based tests (panel B). P values <.05 are highlighted.
One subject had a missing value.
Inflammatory Serum Markers
Serum analyses were performed in 13 out of 14 subjects. Within-subject changes in each biomarker are summarized using boxplots and are shown in Figure 4. For the majority of subjects, biomarker levels were low or below the limit of detection meaning that there was little or no change before versus after treatment, resulting in boxplots where the median, first and third quartiles were equal. The only exception was IL-10, for which the median (first, third quartiles) change was 0 (0, 0) following placebo, and 1.04 (0.59, 1.10) following prednisolone. We used a linear mixed model with repeated measures as described above to compare IL-10 levels between placebo and prednisolone treatments; IL-10 expression was log-transformed to satisfy the normality assumption. Based on the model, the difference between treatments was statistically significant (P = .004).
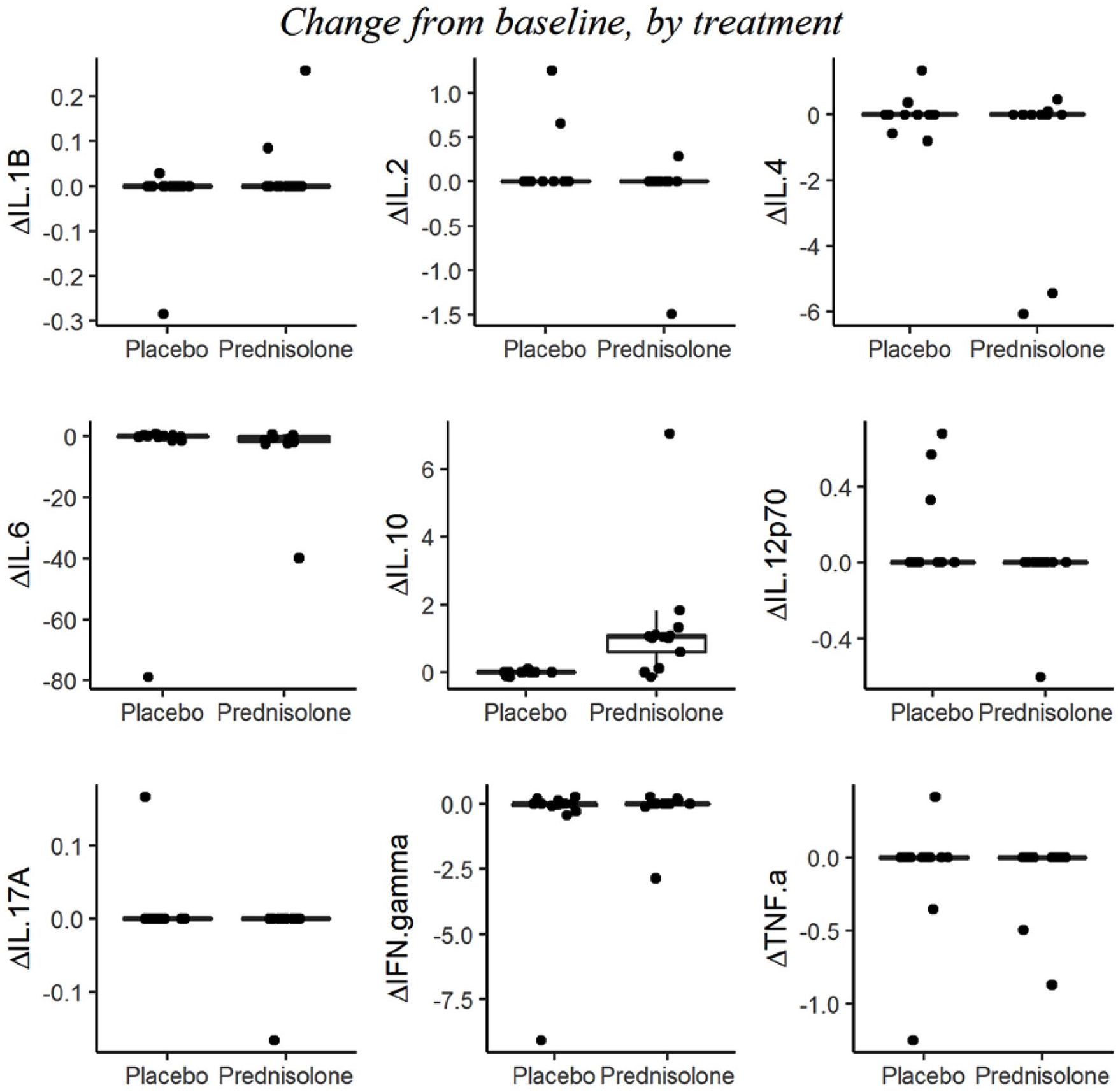
Serum inflammatory biomarker data. Boxplots of absolute change from baseline (Pre−120) in serum inflammatory biomarker levels during placebo + acute intermittent hypoxia (AIH) and prednisolone + AIH treatment periods. The bottom and top of the box represent the first and third quartiles of the data, the horizontal bar within the box represents the median, and whiskers extend to ±1.5 IQR (interquartile range). Because of low variability for several biomarkers, their first and third quartiles are equal producing a single line rather than a box. Serum samples were assessed using a high sensitivity immunoassay (9-Plex Human ProcartaPlex Panel) and processed using Luminex xMAP technology for detection of the following proteins: interferon-γ (IFN-γ); interleukin-1β (IL-1β); IL-2; IL-4; IL-6; IL-10; IL-12p70; IL-17A; tumor necrosis factor–α (TNF-α).
Discussion
AIH is a promising, simple, and potentially safe strategy to promote recovery of motor function in individuals with partial paralysis due to iSCI. We demonstrate that pretrial administration of a corticosteroidal anti-inflammatory drug can enhance AIH-induced somatic motor plasticity. A significant change in ankle torque following combined prednisolone and AIH treatment was observed, accompanied by a higher soleus muscle EMG activity as well as increased mean IL-10 serum expression (anti-inflammatory cytokine). This study suggests that pretreatment with anti-inflammatory drugs may enhance the capacity for AIH-induced functional plasticity in persons with chronic iSCI.
The ability of AIH to improve motor function in individuals with chronic iSCI has been reported in several studies. For example, a single AIH sequence of 15 episodes increased ankle torque in individuals with iSCI by 30% to 40%. 4 In a follow-up study, daily AIH combined with walking training for 5 days increased both walking speed and endurance in a randomized, placebo-controlled, double-blinded, crossover design study; these beneficial effects lasted for at least 1 week. 5 In a similar study, it was shown that AIH combined with treadmill training for 4 weeks is more beneficial than AIH or treadmill walking alone. 16 No deleterious side effects were reported with the AIH exposures, suggesting that AIH is a clinically feasible therapeutic approach for chronic SCI. 17 More recently, a study using transcranial magnetic stimulation showed that AIH changes corticospinal function in humans, likely by altering corticospinal-motoneuronal synaptic transmission. 6
One source of concern is that the impact of AIH therapy in humans may be mitigated by systemic inflammation. Several studies suggest that individuals with chronic SCI often have elevated levels of inflammatory markers in the serum,7-10 indicating that the SCI population is predisposed to nonetiological, low-grade systemic inflammation. The immune system and central nervous system (CNS) exhibit bidirectional crosstalk across the blood-brain barrier. There is an active immune surveillance within the CNS and, in turn, systemic inflammation elicits CNS inflammatory responses that can influence synaptic function. Circulating immune cells and proinflammatory molecules can infiltrate the CNS by crossing the blood-brain barrier or induce inflammatory signaling within the CNS via cross-talk between endothelial cells, neurons, and glia.18,19 Thus, systemic or neuronal inflammation associated with iSCI may affect AIH-induced synaptic plasticity. Indeed, numerous studies indicate that systemic inflammation undermines other forms of CNS synaptic plasticity; for example, hippocampal long-term potentiation20-22 and spinal instrumental learning 23 are impaired by systemic inflammation.
The impact of inflammation on AIH-induced plasticity has recently been acknowledged. A commonly studied form of AIH-induced motor plasticity in animal models, phrenic long-term facilitation, is remarkably sensitive to systemic inflammation.11-13 For example, Huxtable et al 11 showed that systemic administration of a low dose of lipopolysaccharide abolishes phrenic facilitation for at least 24 hours. Another model of inflammation induced by a high dose of intermittent hypoxia (eg, 8 hours of continuous intermittent hypoxia) impairs AIH-induced respiratory motor plasticity. Inflammation in both models is accompanied by a transient but substantial increase in inflammatory gene expression within the rat cervical spinal cord. A key molecule underlying impairment of AIH-plasticity by inflammation appears to be spinal p38 MAP (mitogen-activated protein) kinase. 14 Severe intermittent hypoxia-induced spinal inflammation is accompanied by increased p38 MAP kinase phosphorylation within phrenic motoneurons and nearby microglia, and spinal p38 MAP kinase inhibition restores AIH-induced phrenic long-term facilitation.
In both lipopolysaccharide- and chronic intermittent hypoxia–induced inflammation rat models, subsequent application of a high dose of ketoprofen restores AIH-induced phrenic motor plasticity. Ketoprofen is a nonsteroidal anti-inflammatory drug that directly inhibits cyclo-oxygenase (COX) and, at high doses, the pro-inflammatory transcription factor nuclear factor kappa B (NF-κB). Lynch et al 24 investigated the effect of a similar nonsteroidal anti-inflammatory drug, ibuprofen (800 mg single dose), on AIH-induced leg plasticity in humans with SCI. The low ibuprofen dose permissible in humans had no significant effect on AIH-induced leg plasticity. This finding is not unexpected since 800 mg in humans is sufficient to inhibit COX-2 enzymatic activity, but not NF-κB. COX-2 enzymatic activity alone is not sufficient to restore AIH-induced phrenic long-term facilitation in rats, 25 suggesting that other factors (such as NF-κB transcriptional activity and increased IL-1β levels) suppress AIH-induced motor plasticity.26,27
In this study, pretreatment with a high prednisolone dose (ie, 60 mg) significantly increased the impact of AIH on leg strength. Prednisolone was chosen because it is a potent anti-inflammatory drug, is rapidly absorbed after oral administration reaching peak plasma concentrations after 1 to 3 hours and suppresses NF-κB activity. Its effects are primarily mediated by binding to the glucocorticoid receptor complex and downregulating inflammatory responses via inhibition of NF-κB activation. 28 In addition, it represses the COX pathway, which generates inflammatory prostaglandins. 29 It is highly likely that the effect of prednisolone on AIH-mediated plasticity resulted from diminished NF-κB-mediated inflammation as described above, either within the CNS or periphery, thereby decreasing circulating pro-inflammatory molecules. 30 Increased circulating levels of the anti-inflammatory cytokine, IL-10, following prednisolone administration are consistent with the anti-inflammatory actions of prednisolone.
Enhanced motor plasticity following prednisolone could also have been nonspecific, independent of anti-inflammatory effects. The interaction between neurotrophins and glucocorticoids has been a focus of studies on neuroplasticity for many years. For example, glucocorticoids selectively activate tropomyosin receptor kinase B (TrkB) after in vivo administration in the brain and to cultured neurons. 31 Since this activation does not require increased neurotrophin production, glucocorticoids may directly activate brain-derived neurotrophic factor (BDNF)/TrkB signaling, enhancing subsequent AIH-induced motor plasticity. A recent study by Arango-Lievano et al 32 showed that BDNF receptors facilitate glucocorticoid receptor (GR)-mediated signaling and enable glucocorticoids, primed by BDNF, to activate gene transcription and possibly other cellular actions mediated by the GR 32 following acute treatment. 31 In vitro studies suggest that BDNF and dexamethasone have a different time-course of pTrkB activation. BDNF results in immediate activation whereas the dexamethasone response is smaller and more persistent. These effects are independent of the release of other neurotrophins. 31 Together these studies suggest that multiple mechanisms of glucocorticoid mediation of BDNF/TrkB-signaling exist, which could account for increased AIH-induced plasticity after prednisolone treatment.
The results of this study should be considered in light of some potential limitations. One is a relatively low sample size, which was determined based on our experience with a similar and previously published study. 4 Our subjects exhibited a large variability of AIH response following both prednisolone and placebo treatment, which could be due to genetic variability between our study participants, gender, age, assessment tools, or time post-SCI. Further studies with larger sample size may be informative in understanding the influence of the various factors. However, the significance of the improvements following prednisolone pretreatment are supported as a smaller sample size would be expected to result in low statistical power and a lower likelihood of finding a change. The second limitation concerns lack of serum inflammatory marker analysis from age-matched able-bodies individuals, and a lack of change in most inflammatory markers following prednisolone or placebo administration. Biomarker levels from SCI subjects in our study were either low or below the limit of detection, which could be addressed in future studies via utilizing high-sensitivity luminex kits or assays, which are superior to the classical assays for detecting low levels of inflammatory markers indicating subclinical inflammation in serum of individuals with SCI.
Conclusions
A growing number of studies support the potential of AIH to promote functional motor recovery in humans with chronic iSCIs. Currently, there are more than 20 active clinical trials investigating the effects of AIH therapy in humans (clinicaltrials.gov search dated October 22, 2018). In order to realize the full potential of AIH therapy, it will be critical to identify and eliminate factors that may undermine its therapeutic efficacy. This study provided evidence that pretreatment with an anti-inflammatory drug enhanced the efficacy of a single AIH session. These findings suggest that inflammation, whether systemic or neuronal, affects the expression of hypoxia-induced plasticity in humans, and should be recognized as a probable factor in individuals who do not respond to AIH therapy.
Footnotes
Acknowledgements
We are immensely grateful to Sheila Burt for assistance with editing the document that greatly improved the manuscript.
Author Contributions
Milap S. Sandhu, contributed to development of the study concept and design, subject recruitment, data collection, analyses, and interpretation, literature search, figures, and manuscript writing. As corresponding author, he has full access to all data and final responsibility for the decision to submit for publication. Elizabeth Gray and Masha Kocherginsky contributed to statistical analyses, data interpretation, and manuscript writing. Arun Jayaraman contributed to study concept and design. Gordon S. Mitchell and William Z. Rymer contributed to development of study concept and design, data interpretation, literature search, figures, and manuscript writing and approval of final submission.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
