Abstract
Background. Neural plasticity may contribute to motor recovery following spinal cord injury (SCI). In rat models of SCI with respiratory impairment, acute intermittent hypoxia (AIH) strengthens synaptic inputs to phrenic motor neurons, thereby improving respiratory function by a mechanism known as respiratory long-term facilitation. Similar intermittent hypoxia-induced facilitation may be feasible in somatic motor pathways in humans. Objective. Using a randomized crossover design, the authors tested the hypothesis that AIH increases ankle strength in people with incomplete SCI. Methods. Ankle strength was measured in 13 individuals with chronic, incomplete SCI before and after AIH. Voluntary ankle strength was estimated using changes in maximum isometric ankle plantar flexion torque generation and plantar flexor electromyogram activity following 15 low oxygen exposures (Fio2 = 0.09, 1-minute intervals). Results were compared with trials where subjects received sham exposure to room air. Results. AIH increased plantar flexion torque by 82 ± 33% (P < .003) immediately following AIH and was sustained above baseline for more than 90 minutes (P < .007). Increased ankle plantar flexor electromyogram activity (P = .01) correlated with increased torque (r2 = .5; P < .001). No differences in plantar flexion strength or electromyogram activity were observed in sham experiments. Conclusions. AIH elicits sustained increases in volitional somatic motor output in persons with chronic SCI. Thus, AIH has promise as a therapeutic tool to induce plasticity and enhance motor function in SCI patients.
Introduction
Most spinal cord injuries (SCI) are motor incomplete. 1 Although spinal plasticity in spared neural pathways contributes to recovery of limb function following SCI,2,3 the extent of recovery is often limited. 4 Thus, there is a need for strategies to further enhance spinal plasticity and improve limb function in persons with SCI. Acute intermittent hypoxia (AIH) induces spinal plasticity, strengthening synaptic pathways to respiratory motor neurons by a mechanism known as respiratory long-term facilitation (LTF).5,6 LTF is initiated by hypoxia-induced serotonin release on or near respiratory motor neurons,7-9 initiating de novo synthesis of brain-derived neurotrophic factor (BDNF) and activation of its high affinity receptor, TrkB. 10 Repetitive AIH (daily for 1 week) elicits functional recovery of breathing capacity in rats with high cervical spinal hemisection, an effect that lasts for many hours to days.11-13
We recently observed similar repetitive AIH-induced changes in the neurochemistry of respiratory and nonrespiratory motor nuclei, 14 suggesting the capacity of AIH to induce BDNF-dependent plasticity in nonrespiratory motor pathways.15,16 Here, we tested the hypothesis that AIH facilitates the ability to generate volitional motor output in pathways that innervate the legs of persons with chronic, motor incomplete SCI. Specifically, we assessed ankle torque generation and neural activation of muscles involved in plantar flexion.
Methods
We performed all experiments at the Rehabilitation Institute of Chicago, an affiliate of Northwestern University in Chicago. All participants gave written informed consent, which was approved by the Northwestern University Institutional Review Board.
Subjects
Thirteen adults with chronic SCI, graded as American Spinal Cord Injury Association (ASIA) Impairment Scale C or D 17 (Table 1), were included who had volitional ankle plantar flexion strength in at least 1 leg. Subjects were required to discontinue antispasticity medications 4 weeks prior to participation. Other potential subjects were excluded if they had concurrent medical illness, traumatic brain injury, cardiovascular or pulmonary complications, metabolic dysfunction, continued antispasticity medications, or were previously diagnosed with obstructive sleep apnea.
Demographic Subject Characteristics, Level of Lesion, and Clinical Spasticity Scores for Persons With Chronic, Incomplete Spinal Injury
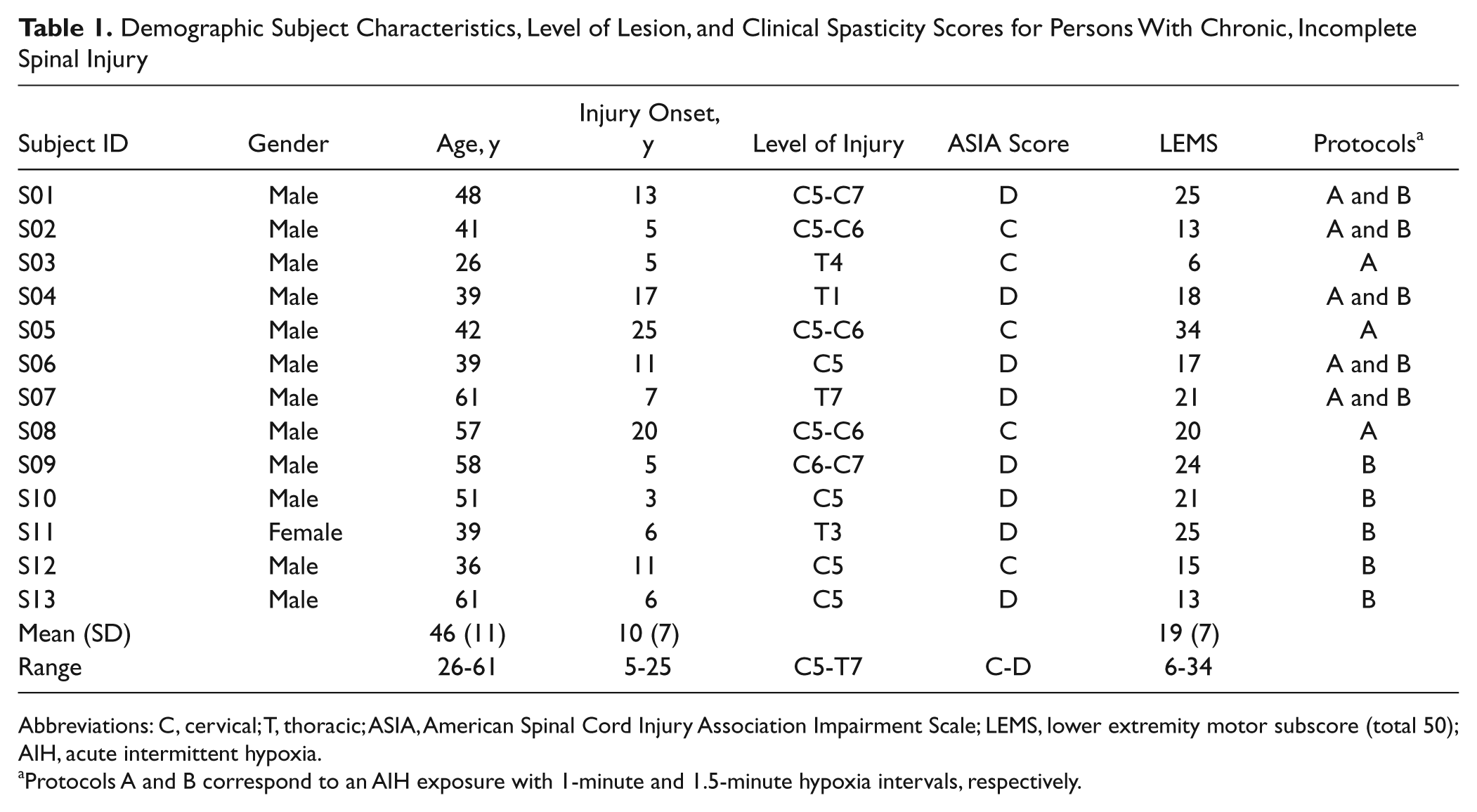
Abbreviations: C, cervical; T, thoracic; ASIA, American Spinal Cord Injury Association Impairment Scale; LEMS, lower extremity motor subscore (total 50); AIH, acute intermittent hypoxia.
Protocols A and B correspond to an AIH exposure with 1-minute and 1.5-minute hypoxia intervals, respectively.
Experiment Setup
Subjects maintained a fixed trunk posture assisted by secure straps attaching them to an adjustable chair (Figure 1A). The test ankle was attached to a 1 degree-of-freedom strength testing dynamometer (System 3; Biodex Medical Systems, Shirley, New York). The ankle joint axis was adjacent to the dynamometer’s center of rotation and plantar flexed 80° relative to the shank, while the knee joint was flexed 10° relative to the femur, and hip joint was flexed 80° relative to trunk (Figure 1D). To record volitional ankle torques, the dynamometer housed a 6 degrees-of-freedom load cell (Model 45E15A—JR3, Inc, Woodland, California; and Model F/T 3994—ATI Industrial Automation, Apex, North Carolina). Data from the load cell were anti-alias filtered at 500 Hz using custom firth-order Bessel filters and then sampled at 1000 Hz with an 18-bit analog-to-digital converter (NI PCI-6289; National Instruments, Austin, Texas). These data were acquired using custom programs in LabVIEW (National Instruments) and Matlab (Mathworks, Inc, Natick, Massachusetts).
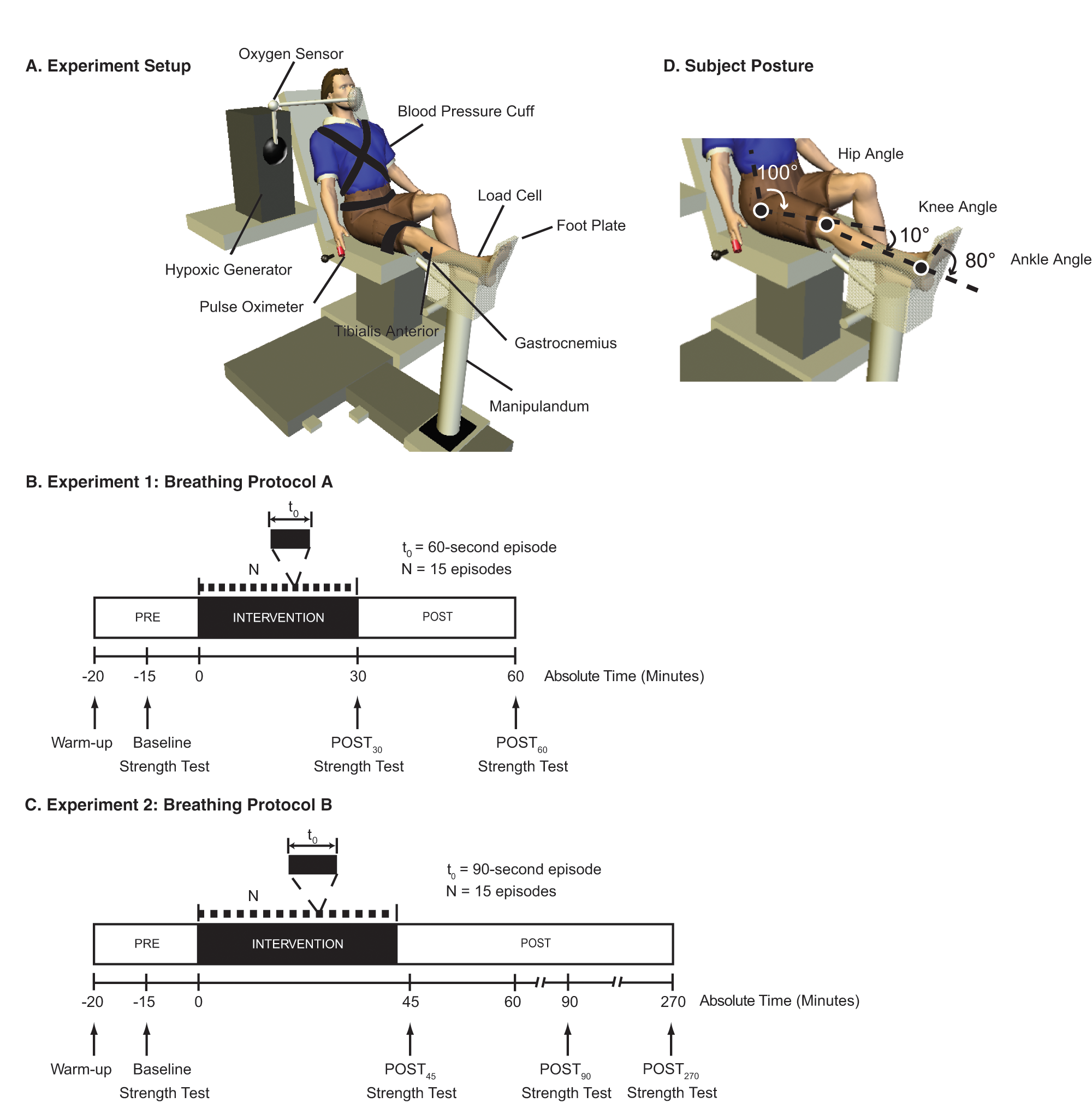
Experimental setup. Subjects were secured to a dynamometer with testing leg attached to a footplate (A). A 6 degrees-of-freedom load cell was mounted at the manipulandum motor head and was used to record ankle torque generation. Surface electromyography was used to record muscle activity from the plantar flexor muscles, gastrocnemius as well as the tibialis anterior muscle. Blood pressure, oxygen saturation, and heart rate were recorded continuously while subjects were wearing a rebreather mask connected to the hypoxic generator that delivered the air mixtures. While seated, subjects participated in a breathing intervention that consisted of a single sequence of mild bouts of acute intermittent hypoxia. In our first experiment (B), a single sequence lasted approximately 30 minutes and consisted of 15 iterations alternating 60-second bouts of breathing 0.09 Fio2 with 60-second bouts of breathing 0.21 Fio2. In our second experiment (C), a single sequence lasted approximately 37.5 minutes and consisted of 15 iterations alternating 90-second bouts of breathing 0.09 Fio2 with 60-second bouts of breathing 0.21 Fio2. An oxygen generator was manually adjusted to provide low oxygen (Fio2 = 0.09) or room air (Fio2 = 0.21). Subjects breathed the air mixtures using a latex-free full non-rebreather mask with a custom neoprene head strap. Ankle plantar flexion strength tests were made 15 minutes before the start of intervention, then again at the end of the intervention and then 30 minutes later.
To assess agonist muscle activity during ankle isometric plantar flexion torque generation, we recorded surface electromyograms (EMGs) from the medial head of the gastrocnemius (GAST) muscle using bipolar surface electrodes (Model DE-2.1; Delsys USA, Inc, Boston, Massachusetts). We also recorded EMG activity from the antagonist tibialis anterior (TA) muscle. EMGs were amplified 1000 times using a Bagnoli-16 system (Delsys USA, Inc), which has a bandwidth of 20 to 450 Hz. The filtered EMG signals were sampled at 2000 Hz with an 18-bit analog-to-digital converter (NI PCI-6289; National Instruments). A common clock was used to synchronize data acquisition from the EMG and dynamometer systems.
An oxygen generator (Model HYP-123, Hypoxico Inc, New York, New York) provided variable inspired oxygen mixtures (Fio2 = 0.09 or 0.21). Subjects breathed the gas mixtures using a non-rebreathing mask with a custom neoprene hexad strap. To complete the breathing circuit, the non-rebreathing mask attached to a reservoir bag that the generator filled between breaths; a 1-way valve prevented inhalation of room air or exhaled gas. Changes in inspired oxygen concentration were continuously monitored (Model OM-25RME; Maxtec Inc, Salt Lake City, Utah). Oxyhemoglobin saturation and heart rate were continuously monitored at the finger with a pulse oximeter for safety and to document the severity of hypoxic episodes at 0.25 Hz (Autocorr Plus; Smith Medical Inc, New York, New York). We recorded blood pressure using an electronic blood pressure monitor (HEM-780; Omron Healthcare Inc, Bannockburn, Illinois). We found no detectable change in systolic pressure before (109 ± 19 mm Hg) or immediately after (114 ± 21 mm Hg) AIH (paired t test; P = .6); similarly, we found no change in diastolic pressure before (71 ± 18 mm Hg) versus immediately after (79 ± 14 mm Hg) AIH (paired t test; P = .3).
Protocols
To determine the immediate effects of AIH on changes in voluntary ankle strength, 8 subjects participated in a 30-minute AIH intervention (protocol A, Table 1). We recorded baseline strength 15 minutes prior to intervention onset (Baseline), immediately after the breathing intervention (Post30), and again 30 minutes later (Post60). Subjects were exposed to a single sequence of mild AIH, consisting of 1-minute of 9% O2 alternating with 1-minute of 21% O2 for a total of 15 episodes (Figure 1B). In sham (SHAM), control experiments, subjects were exposed to continuous normoxia, which consisted of 15 iterations of 1-minute 21% O2 switching to another 1-minute 21% O2 exposure. To reduce potential carryover effects from AIH to SHAM testing, we randomized the order of single-dose exposures and required a minimum of 2 weeks between AIH and SHAM exposures for each subject. Subjects were unaware of the order of these exposures. To determine the prolonged effects of AIH on changes in voluntary ankle strength, we performed a second protocol (protocol B) in 5 of the original protocol A subjects and applied it in 5 new additional subjects (Table 1). Baseline strength recordings were made 15 minutes prior to AIH, and the subjects were then exposed to a slightly different intermittent hypoxia protocol: 1.5 minutes of 9% O2 alternating with 1-minute 21% O2 for a total of 15 episodes (Figure 1C). We increased the low oxygen exposure time from 1 to 1.5 minutes in this protocol to determine if the increase resulted in a significant change in oxyhemoglobin saturation during hypoxic episodes, which was tolerated by all participants. Similar to protocol A, we recorded voluntary ankle strength measurements (as detailed below) at approximately 45 (Post45), 90 (Post90), and 270 minutes post-AIH (Post270).
Strength assessment
For protocols A and B, a series of maximum voluntary ankle plantar flexion torque (MVT) measurements were used to assess ankle strength. 18 We recorded torque and EMG signals on the more dominant ankle as determined during clinical assessment using the ASIA lower extremity motor scores. 17 Subjects began each session by performing 3 baseline MVTs of the ankle plantar flexors. Subjects were instructed to push “as hard and as fast” as they could after they heard an auditory “go signal.” Each MVT was approximately 3 to 6 seconds in duration. A minimum of 1-minute rest was given between contractions. Subjects received consistent verbal encouragement during testing. Peak torque was defined as the average maximum value obtained across the 3 contractions. In the event that the peak torque values differed by more than 10%, additional contractions were performed. Following baseline MVT, all subjects were administered a breathing intervention (AIH or SHAM).
Torque and EMG data were processed off-line using Matlab. Torque data were digitally low-pass filtered at 10 Hz using fourth-order Butterworth filters with zero phase lag. Maximum torque was estimated for each MVT by averaging within a fixed time window corresponding to the maximum absolute value of the torque signal ±50 ms. An average maximum value obtained across the 3 contractions was used to define peak torque. EMG data were used to quantify the magnitude of GAST activity during each MVT. EMG signals were full wave rectified and smoothed using a zero-lag fourth-order Butterworth filter, low-passed at 5 Hz to create an EMG envelope. Peak EMG activity was determined for each MVT. We computed the peak EMG activity for each muscle corresponding to an averaged EMG activity level within a fixed ±50 ms time window, 100 ms preceding the maximum absolute value of the torque signal. We then subtracted the background level of EMG from the peak EMG activity. Background EMG was defined as a 100 ms time window of EMG activity 1 second prior to the auditory “go signal.” Average peak EMG activity for each muscle was obtained across the 3 contractions. 19
Statistical Analysis
A repeated-measure, randomized crossover statistical design was used to test the hypothesis that a single sequence of mild AIH increases ankle plantar flexion strength. A repeated-measures ANOVA was used to measure the AIH-induced change in plantar flexion torque and EMG activity, with treatment as the independent factor, subject as a random factor, and time as the repeated measure. Bonferonni corrections were made for post hoc comparisons between the 2 treatment conditions (AIH and SHAM) and 3 time periods (Baseline, Post30, and Post60). Using protocol B, we also tested the hypothesis that there would be significant changes in the capacity to generate a sustained increase in torque lasting hours after completion of a single sequence of AIH. Because of the nonnormal distribution of our data set, we tested this hypothesis using a nonparametric Friedman test on the group data from protocol B. We performed post hoc signed-rank comparisons between Baseline and each of the 3 postintervention measurements (Post45, Post90, and Post270). Maximum isometric torque can change substantially with changes in agonist and antagonist muscle EMG activity, and we wished to examine the strength of this relationship with the imposed breathing intervention at Post45, Post90, and Post270. Therefore, we assessed this relationship using linear regression models to determine if observed increases in capacity to generate torque correlate with increases in voluntary activity of agonist activity or decreases in voluntary activity of antagonist activity. Linear regression models were constructed to include normalized post-AIH data relative to pre-AIH data across all subjects to obtain a range of voluntary activation levels within each muscle. All results were analyzed using SPSS 16 statistical software (SPSS Inc, Chicago, Illinois) and were considered significant for P < .05. All reported statistical inferences were obtained at power levels greater than 0.80 and P values less than .05. Adjustments to P values were made for multiple comparisons.
Results
Isometric plantar flexion strength increased after AIH in all subjects that participated in protocol A (P < .002), an effect not found in SHAM experiments (Figure 2). Immediately post-AIH (Post30), maximal plantar flexion torque (ie, the MVT) increased 82 ± 33% (P < .003), and peak gastrocnemius EMG activity increased 43 ± 17% (P = .04). The MVT remained elevated above baseline at Post60 (P < .002). Values at Post30 and Post60 were not significantly different from one another, although both were greater than corresponding values in SHAM trials (P = .02). Medial gastrocnemius EMG activity increased 83 ± 23% at Post60 (Figure 2B; P = .007). There was no discernable relationship between AIH-induced facilitation of ankle strength and motor impairment as defined by the ASIA lower extremity motor score (r2 = .05; P = .6).
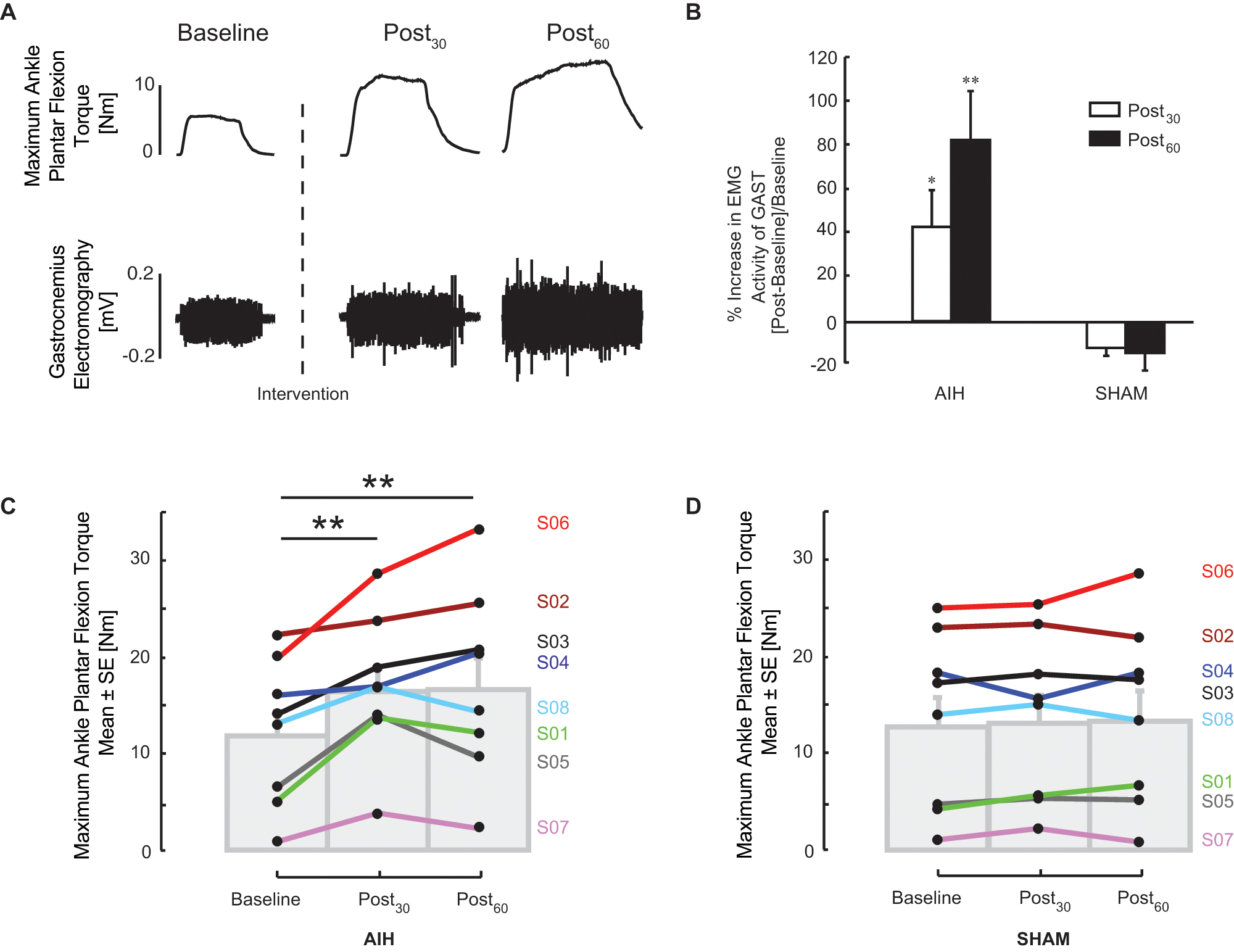
Effect of acute intermittent hypoxia (AIH) on ankle plantar flexion strength. Maximum plantar flexion torque and corresponding electromyogram (EMG) responses of the gastrocnemius (GAST) and tibialis anterior muscles from a spinal cord injury subject (S01) are shown in A. Subjects exerted maximum plantar flexion moment against a footplate prior to (Baseline), immediately after the AIH intervention (Post30), then again 30 minutes later (Post60). Ankle medial gastrocnemius EMG activity increased in all subjects following AIH. In B, peak medial gastrocnemius EMG activity increased following AIH in a heterogeneous group of subjects with spinal injury. Average plantar flexion torque (C) significantly increased from baseline values; increases were not observed in the same subjects when breathing air only (Sham). Average plantar flexion torque (D) was unaffected by Sham, normoxia protocols, but increased significantly relative to baseline measurements. Asterisks indicate statistical comparisons to Baseline, where * indicates significance at P < .05; ** indicates significance at P < .005.
In protocol B, AIH increased plantar flexion MVT in all subjects at Post90, and the MVT exceeded baseline in 6 of 10 subjects at Post270. An increase in plantar flexion MVT was observed post-AIH (Friedman’s χ2 = 9.5; P = .02). Typical single-trial MVT and EMG recordings from 1 SCI subject are shown in Figure 3A. In this subject, large torque increases were elicited at Post45, Post90, and, to a lesser extent, Post270. Similar responses were observed at Post45 and Post90 in our subjects, even though they exhibited a wide range of baseline plantar flexion torque generation (Figure 3B). Average MVT at Post45 (43.8 ± 7.8 N m) was greater than baseline (signed rank test, P = .005). The increase in MVT was sustained at Post90 (45.7 ± 8.0 N m), a level significantly greater than baseline (signed rank test, P = .007). Even 4 hours post-AIH, 4 of 10 subjects maintained at least a 50% increase in MVT versus baseline, although this effect did not reach statistical significance (signed rank test, P = .07). Similar assessments in the SHAM normoxia protocol from these same subjects revealed no change in MVT over time (Friedmans χ2 = 5.8; P = .1). To determine if background EMG activity contributed to this post-AIH enhancement, we quantified the EMG activity prior to MVTs for pre-AIH and post-AIH conditions and found no difference in the TA (P > 1.0) or GAST muscles (P > .7) across conditions. Isometric plantar flexion torque generation at the ankle is linked to changes in voluntary EMG activity of agonist (GAST) and antagonist (TA) muscles. We assessed these relationships using linear regression models (Figure 4). Data from all subjects were used to provide a range of normalized EMG activity levels within each muscle, corresponding to a range of normalized ankle plantar flexion MVT data. A significant regression between the change in GAST EMG activity and ankle plantar flexion MVT was observed (P < .001; r2 = 0.5; Figure 4A), which was not the case for the antagonist muscle. Although there was a negative trend in the relationship between TA activity and ankle plantar flexion MVT, this relationship was not significant (P = .2; r2 = .1; Figure 4B). Similar trends were observed between EMG activity and plantar flexion torque in 9 of 10 subjects.
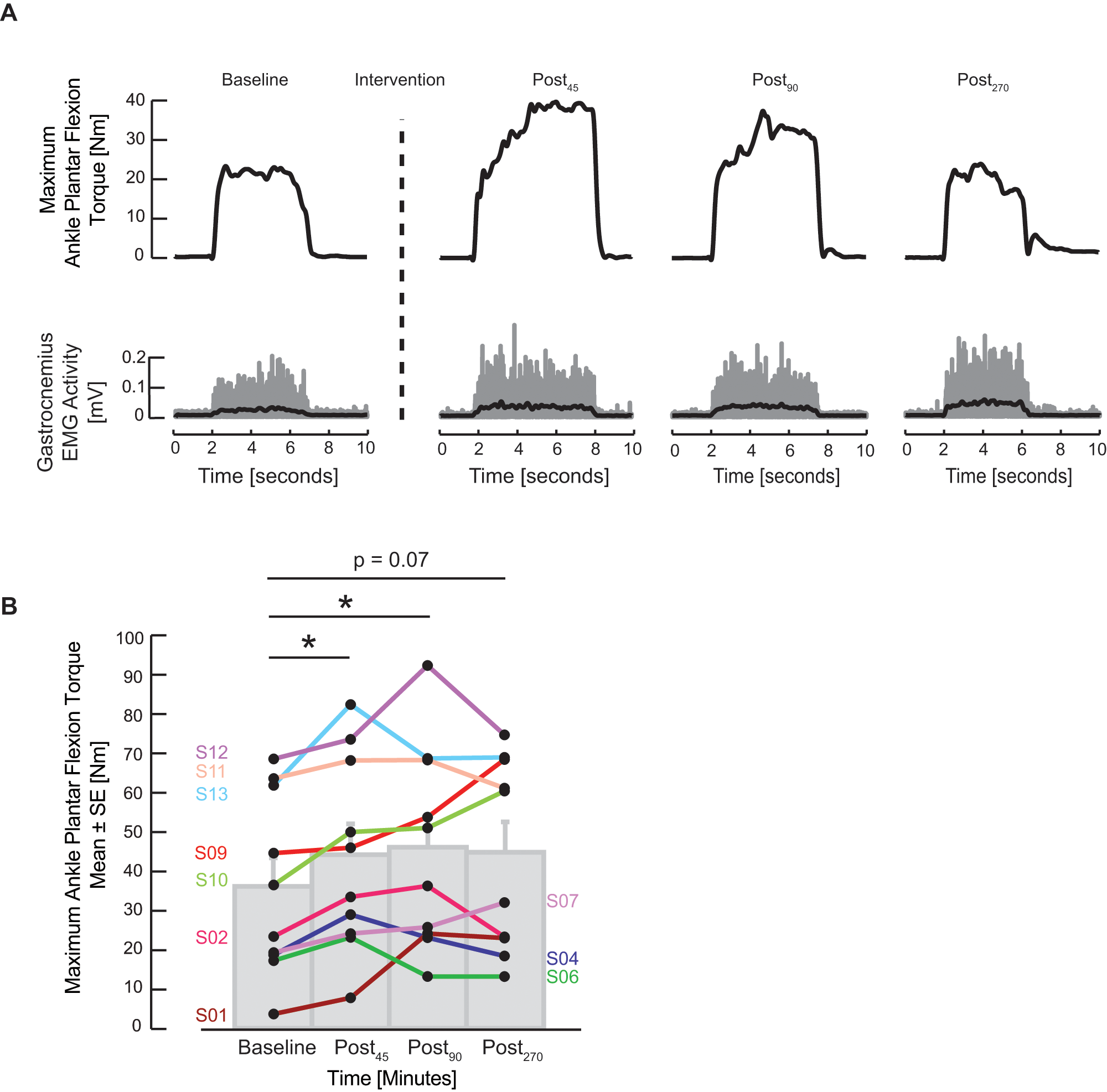
Sustained effect of acute intermittent hypoxia (AIH) on ankle plantar flexion strength. Subjects exerted maximum plantar flexion moment against a footplate prior to (Baseline), approximately 10 minutes after the AIH intervention (Post45), then again at 90 minutes (Post90) and 270 minutes (Post270) after AIH exposure onset. Maximum plantar flexion torque and corresponding electromyogram (EMG) responses of the gastrocnemius muscle from a spinal cord injury subject (S02) are shown in A; the superimposed black lines correspond to the EMG envelope at each time measurement. In B, ankle plantar flexion torque increased following AIH in a heterogeneous group of subjects with spinal injury. Torque values ranged from 3.8 to 92.2 N m. Asterisks indicate statistical comparisons to Baseline, where * indicates significance at P < .007.
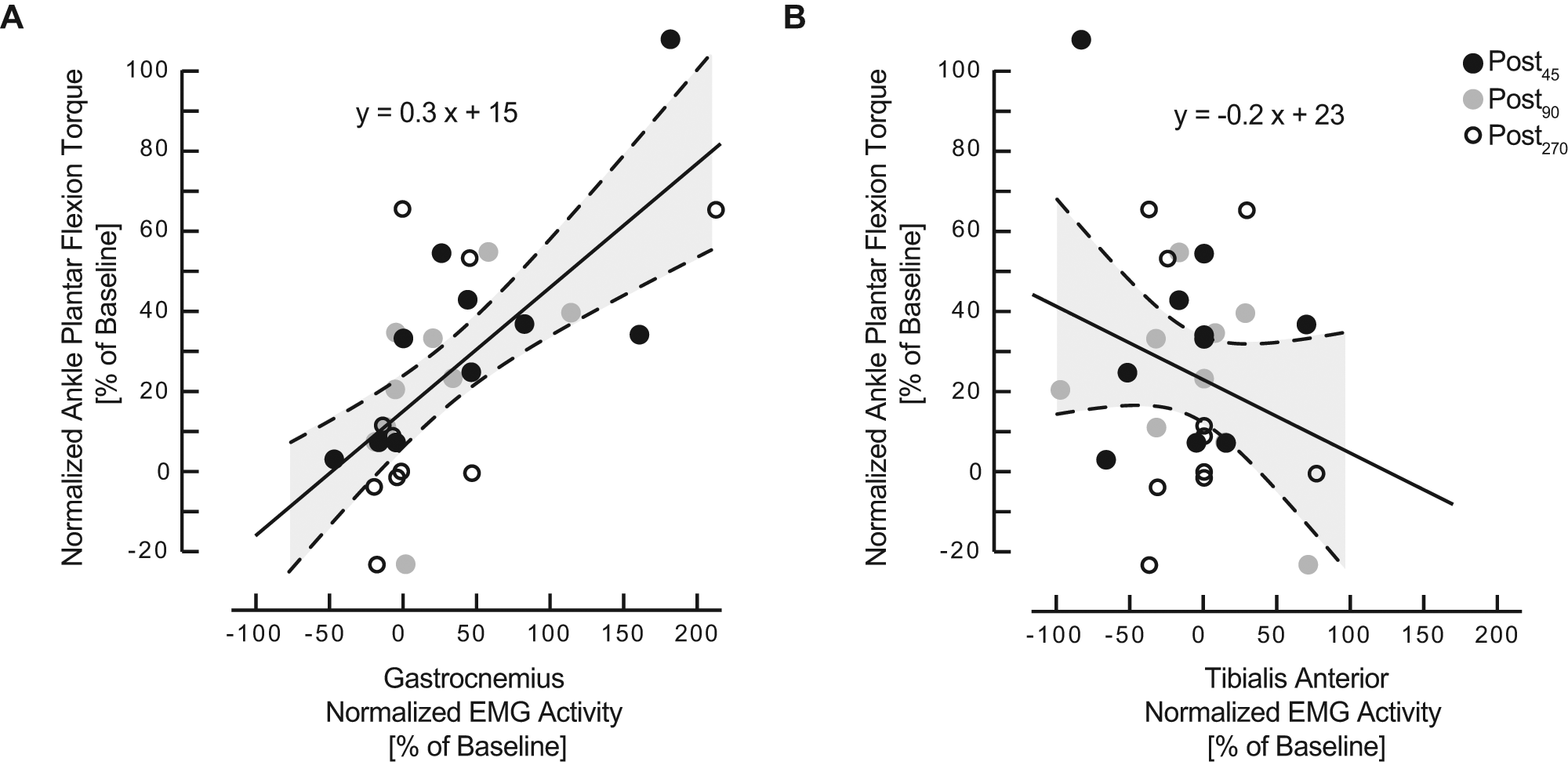
Relationship between electromyogram (EMG) ankle muscle activity and acute intermittent hypoxia–induced change in ankle maximum isometric plantar flexion torque. Linear regression models were used to describe the relationship between the change in ankle plantar flexion MVT relative to Baseline and the change in EMG activity relative to Baseline of the (A) plantar flexor (gastrocnemius) and (B) dorsi-flexor (tibialis anterior) muscles. Regression plots consisted of pooled responses from all subjects after the AIH intervention (black circle, Post45), then again at 90 minutes (gray circle, Post90) and 270 minutes (white circle, Post270). Thin dashed lines above and below the regression line define 95% confidence intervals. The models were significant for describing a relationship between ankle plantar flexor torque and corresponding agonist muscles; gastrocnemius had r2 value of .5 (P < .001). With increased isometric plantar flexion torque, there was a decreasing trend in EMG activity of the tibialis anterior muscle but was not significant (P = .2).
Acute intermittent hypoxia altered oxyhemoglobin saturation and heart rate during each hypoxic episode. Across all subjects, low oxygen exposures reduced oxyhemoglobin saturation after 60 seconds (~81 ± 1% [± 1 SEM]; P < .001). Similar responses were observed during protocol B with no difference in saturation between the 2 protocols (paired t test; P > .05). The low oxygen exposures increased heart rate across all subjects, with significant increases for all but 2 AIH exposures (P values < .03).
Discussion
Here we demonstrate for the first time that a single AIH exposure (15 episodes) improves lumbosacral motor output and maximal voluntary plantar flexion torque generation in persons with chronic, motor incomplete SCI. Improvements in leg strength correlated with increased activation of the medical gastrocnemius muscle. Improved muscle force generation was recorded for more than 4 hours in 4 of 10 subjects, suggesting the potential for long-lasting effects. Although the mechanism of functional improvement cannot be defined from this study, we speculate that similar cellular mechanisms give rise to respiratory LTF 6 and improved limb function in humans.
Individuals with incomplete injuries (ASIA C and D) were chosen for this study since residual spinal motor pathways are necessary as a substrate for AIH-induced spinal plasticity. Despite initial variability in the group’s capacity to generate voluntarily ankle torque, all 13 subjects showed improvement in maximal torque generation and gastrocnemius EMG activation. Our participants also had absolute ankle torque values that were comparable with earlier studies involving persons with incomplete spinal injury.18,20 Although the augmentation in ankle strength could be due to other mechanisms such as spasticity, the remarkable consistency of results provides reassurance that this is a fundamental response with potential as a therapeutic strategy in the treatment of motor deficits following SCI in humans. Reduced ankle strength is a major factor affecting mobility after spinal injury. A recent work 21 showed that persons with incomplete SCI had significant deficits in ankle strength when compared with healthy age-matched controls. Deficits in plantar flexor activity and corresponding speed of voluntary torque generation were also evident in those with incomplete spinal injury. 18 Plantar flexor muscle activation accounts for more than 50% of the gait cycle and contributes to the energetics of normal walking.22,23 We, therefore, speculate that repetitive leg strengthening protocols, which have been shown to promote strength and enhance walking in persons with spinal injury, 24 might further facilitate functional gains when coupled with AIH exposure.
Although only a single AIH presentation was studied here, repetitive or combined (AIH plus physical rehabilitation) treatments could potentially increase the magnitude, duration, and specificity of functional benefits. As a potential therapeutic tool, an important consideration is that repetitive intermittent hypoxia can be applied without physical activity and, thus, may be useful for patients with severe motor impairments. On the other hand, the potential benefits of intermittent hypoxia-induced plasticity may be diminished in cases with severe disruption of descending spinal serotonergic innervation.25,26 In such cases, it may be possible to elicit plasticity “downstream” from serotonin release via pharmacological treatments that activate phrenic 27 as well as nonrespiratory motor facilitation, including intermittent spinal serotonin receptor activation, BDNF administration, and/or TrkB transactivation.9,28,29
Establishing protocols of intermittent hypoxia to improve motor function in humans with SCI require knowledge of minimal hypoxic severity, the pattern and the number of hypoxic episodes, and the frequency of presentation to the patient. The pattern and number of episodes are key, since respiratory plasticity is highly pattern sensitive 30 ; neither continuous hypoxia13,31 nor spinal serotonin receptor activation 7 elicit similar motor plasticity.
Targeted investigations concerning the potential of repetitive AIH to facilitate restoration of limb function in safe and meaningful ways are needed. In parallel, we need to understand factors that undermine the capacity for AIH-induced plasticity, such as (a) systemic inflammation 32 or (b) medications to reduce spasticity (eg, baclofen), which may augment inhibitory neurotransmission and impair spinal plasticity. Finally, since exposure to severe, chronic intermittent hypoxia elicits morbidity such as hypertension, 33 hippocampal cell death, learning deficits,34-36 and metabolic syndrome, 37 it is imperative that repetitive AIH protocols be chosen that are above the threshold to elicit plasticity but below the threshold for serious morbidity. The protocol used here is considerably less severe than those shown to elicit morbidity.
AIH holds promise in promoting spinal motor excitability, an essential event to restore volitional movements following SCI. AIH, combined with exercise or other physical and pharmacologic, may offer a new direction to augment rehabilitation aimed at improving functional movements in persons with incomplete SCI.
Footnotes
Acknowledgements
The authors are very grateful to the participants.
The Craig N. Neilsen Foundation (the funding agency) had no role in study design, data collection and analysis, decision to publish, or preparation of the article.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Craig H. Neilsen Foundation (Grant No. 84118).
