Abstract
Background. Alterations in the function of spinal neuronal circuits underlying locomotion after a spinal cord injury (SCI) are associated with changes in the behavior of spinal reflexes (SRs) in both rats and humans. In healthy subjects, the SR consists of a dominant early reflex component, whereas in chronic, severely affected SCI subjects, a later component dominates. Objective. The aim of this study was to investigate the relationship between SR behavior and walking ability in para-/tetraplegic subjects. Method. The SR was evoked by nonnoxious tibial nerve stimulation. Walking ability was assessed by functional tests and questionnaires. Results. There was a correlation between walking ability and SR behavior in chronic SCI: Severely affected SCI subjects unable to walk showed dominant late SR components, whereas in ambulatory SCI subjects an early SR component dominated. A functional training with an improvement of locomotor ability was accompanied by both a shift from a dominant to a smaller late and the appearance of an early SR component. Conclusions. Our findings indicate that SR can serve as a marker for the locomotor ability of SCI subjects. Neuronal plasticity exploited by a functional training is reflected in both an improvement of locomotor ability and a change in balance of SR components toward the early SR component.
Introduction
Spinal polysynaptic reflex recordings in mammals have been used to identify interneuronal circuits that are involved in spinal locomotion.1,2 There is evidence for a relationship between the time course of these spinal cord reflexes (SR) and locomotor capacity after a spinal cord injury (SCI) in rats 3 and humans. 4
A severe complete SCI leads to a dysfunction of spinal neuronal circuits. This dysfunction is reflected in the exhaustion of locomotor electromyographic (EMG) activity during assisted locomotion and is associated with a change from a dominant early to a dominant late SR component.4-6 However, subjects with sensorimotor incomplete SCI, who regularly perform stepping movements, show no EMG exhaustion and the early SR component dominates.
The focus of this study was to investigate whether SR behavior can be used as a marker for the functional state of spinal locomotor circuitries underlying walking ability in SCI subjects. In addition, we evaluated the plasticity of neuronal circuits and investigated whether an improvement in locomotor ability following functional training is accompanied by a shift from a late to an early SR component. The SR was evoked by cutaneous afferents. 7 Since a noxious stimulus cannot be determined in SCI subjects, the term spinal reflex was chosen as a response perceived by healthy subjects to be below the nociceptive stimulation threshold. Although we use the term spinal reflex, we cannot exclude that latter parts of the SR are mediated by supraspinal pathways.
Methods
General Procedures and Subjects
The study protocol was approved by the local ethics committee, and all patients gave informed written consent before data recordings. Altogether, SR, walking ability, and muscle strength were assessed in 28 subjects with chronic (>1 year postlesion) SCI. In total, 10 subjects suffered a motor complete SCI (AIS [American Spinal Injury Association Impairment Scale] A/B) 8 and 18 subjects a motor incomplete SCI (AIS C/D). The mean age of the 28 subjects was 44.8 years (SD = 14.6 years), and the neurological level of lesion ranged from C4 to L3. All SCI subjects showed slightly increased muscle tone and normal tendon reflexes, that is, preserved lower motoneuron function. The lesion duration ranged from 1.1 to 37.4 years (10 ± 9.3 years). A total of 12 SCI subjects had tetraplegia, 16 had paraplegia, and all showed signs of spasticity. The clinical data of all subjects are given in Table 1.
Characteristics of the SCI Subjects Included in the Study
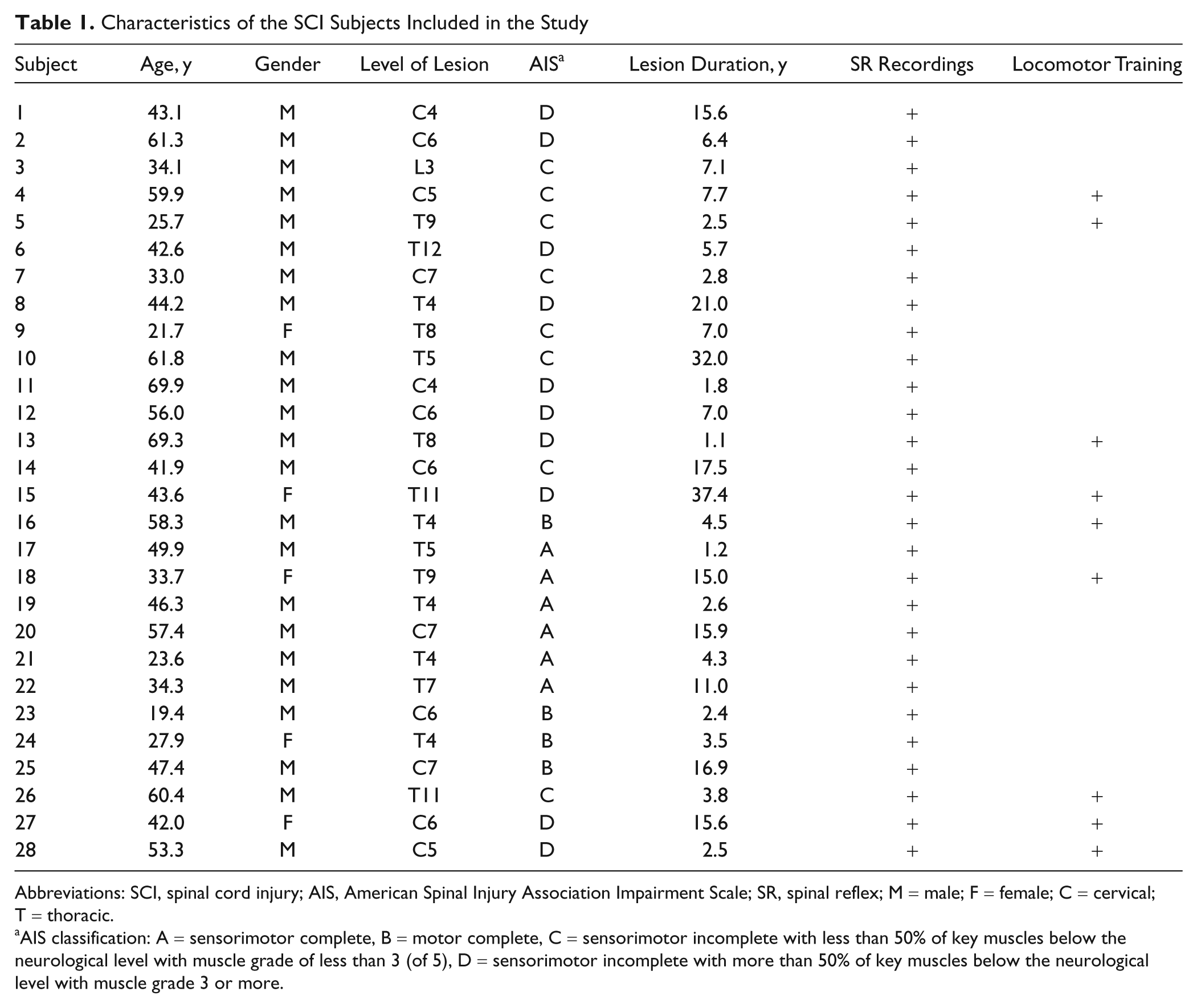
Abbreviations: SCI, spinal cord injury; AIS, American Spinal Injury Association Impairment Scale; SR, spinal reflex; M = male; F = female; C = cervical; T = thoracic.
AIS classification: A = sensorimotor complete, B = motor complete, C = sensorimotor incomplete with less than 50% of key muscles below the neurological level with muscle grade of less than 3 (of 5), D = sensorimotor incomplete with more than 50% of key muscles below the neurological level with muscle grade 3 or more.
Walking ability was evaluated using the Walking Index for Spinal Cord Injury II (WISCI II) to determine ambulatory capacity. 9 It is scored from 0 (cannot stand or walk) to 20 (can walk more than 10 m without walking aids or assistance). In addition, we used the 10-meter walking test (10MWT). 10 The results of the 10MWT were also converted into walking speed with a speed of 0 m/s recorded when the WISCI II score was 0 or 1. Walking ability during daily life can be validly assessed by the Spinal Cord Independence Measure II (SCIM II) mobility part. 11 This questionnaire has several items that quantify mobility and assesses mobility in the subject’s daily environment. Scores of the SCIM II mobility part range from 0 to 40 with higher scores reflecting better daily mobility. We also assessed the lower extremity motor scores (LEMS) by manual muscle testing according to the AIS guidelines. 8
Spinal Reflexes
Spinal reflexes were elicited by electrical stimulation of the distal tibial nerve at the dorsal aspect of the medial malleolus with the electrical stimulator AS 100 constant-current source (ALEA Solution GmbH, Zurich, Switzerland). The stimulus consisted of a train of 8 biphasic rectangular pulses with single stimulus duration of 2 ms and a frequency of 200 Hz, that is, the total duration of the stimulation was 40 ms; see Muller and Dietz. 12 The motor threshold (MT) was determined in a sitting position by a gradual increase of stimulation intensity in steps of 2 mA until the first visible contraction in abductor hallucis muscle was observed. The stimulation intensity was set to 2 times MT threshold. Electrical stimulation was elicited 10 times in both legs in a supine position and was randomly released every 30 to 45 seconds to minimize habituation.13,14
Locomotor Training
Seven motor incomplete (AIS C/D) and 2 motor complete (AIS A/B) SCI subjects underwent intense locomotor training. They each received individually adapted locomotor training over 4 weeks, 3 to 4 times a week. One training session lasted 30 to 45 minutes using either the driven gait orthosis (DGO) Lokomat 15 or conventional treadmill training with partial body weight support. Two SCI subjects were differentially trained: First, in the most impaired incomplete SCI subject, assisted treadmill training was combined with functional electrical stimulation (FES) of the lower limbs, stimulating the tibialis anterior (TA) and peroneus muscles as well as the peroneal nerve behind the caput fibulae. This approach facilitates the swing phase by increasing foot clearance. 16 Stimuli were manually triggered at the end of every stance phase in both legs to enhance hip, knee, and ankle flexion during the swing phase. Second, one motor complete SCI subject (AIS B) was trained using the Lokomat over several years once a week.
Spinal reflex and functional tests were assessed at the beginning and end of the training period and also 6 months after the end of training. One incomplete SCI subject did not participate in the follow-up assessments. In the most severely affected, incomplete SCI subject the EMG activity of rectus femoris (RF), biceps femoris (BF), TA, and gastrocnemius medialis (GM) muscles were assessed during 15 minutes of assisted walking in the DGO to detect changes in EMG amplitudes, 17 before and after 1 month of locomotor training.
Data Analysis
In correspondence with earlier recordings, 12 only SRs in the ipsilateral TA muscle were analyzed. The early and late components of the SR response were analyzed separately. 4 Time windows were set from 60 to 120 ms after stimulation onset for the early and from 120 to 450 ms after stimulation onset for the late component. The presence of reflex responses within these time windows was determined by an increase of EMG activity 3 times above standard deviation of the mean baseline activity for at least 5 samples. If a reflex response were detected, the highest peak amplitude within the corresponding time window was determined and the root mean square value of 25 ms before and 25 ms after the peak amplitude was calculated. If no response were detected the value was set to zero.
Because of large interindividual variability in reflex amplitudes, the relationship between the 2 components was calculated. The amplitude relationship between the early and late SR component within a subject was the focus of interest. Therefore, the 2 reflex components were compared and the greater component was set to 1 and then subsequently used for normalization of the other component. The normalized values of each subject were averaged and thereafter the difference between the normalized early and late SR component was calculated. This difference is used here as the term SR behavior. For further analyses, only the normalized values of the functionally worse leg (lower ASIA motor score) were used.
Statistics
Correlations between the functional tests and the SR behavior were statistically tested using the Spearman rank correlation test. Correlation coefficients between 0.5 and 0.75 and with the number of observations n = 28 indicate moderate to good correlations and correlation coefficients higher than 0.75 indicate very good correlations. 18 Differences in SR behavior and functional outcome measures before, after, and at the 6-month follow-up following locomotor training were tested using the Friedman and Wilcoxon signed-rank test. The significance level for all tests was set at P < .05.
Results
As expected, the results of the walking tests varied among the 28 SCI subjects investigated: The SCIM II mobility score varied between 15 and 40 (median = 20.5); the WISCI II score varied between 0 and 20 (median = 12); the gait speed of the SCI subjects varied between 0.0 and 2.95 m/s (median = 0.21 m/s); and the LEMS varied between 0 and 49 (median = 24).
Walking Ability and Spinal Reflex Behavior
Figure 1 shows the SR behavior in relation to the 3 walking tests and questionnaires from all SCI subjects. There was a moderate to good significant correlation (Spearman correlation coefficient ρ = 0.626) between SR behavior and SCIM II mobility score (Figure 1A). This positive correlation coefficient indicates that the more dominant the early SR component in TA muscle, the higher the independence in daily mobility of the SCI subject (high SCIM II mobility score). Moderate to good correlations were found between the SR behavior and the WISCI II score (Figure 1B; ρ = 0.703), and very good correlations were found for the gait speed (Figure 1C; ρ = 0.772). These positive correlation coefficients indicate that the more dominant the early SR component, the less the SCI subjects required assistive devices to walk 10 m (Figure 1B) and the higher was the subject’s gait speed (Figure 1C).
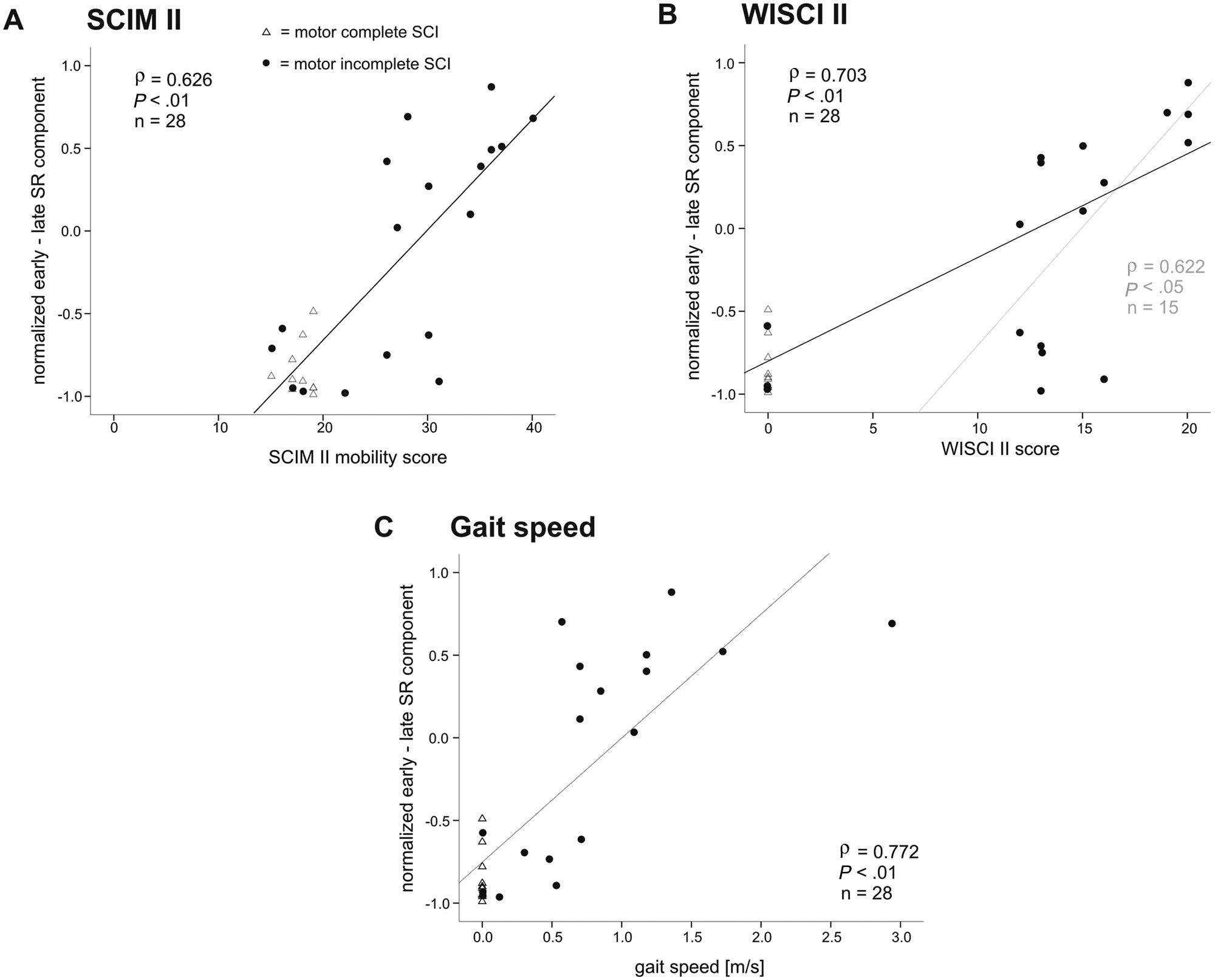
Spinal reflex behavior and walking ability. Mean values of all SCI subjects are displayed. Correlation of the SR behavior (normalized early − late SR component) and (A) the SCIM II mobility score, (B) the WISCI II score, and (C) the gait speed. SCI subjects were divided into 2 groups according to the completeness of injury, motor incomplete (AIS C/D) and motor complete SCI (AIS A/B) subjects. A second trend line (B, gray interrupted line) was added to show the correlation when the subjects with a WISCI II score = 0 were excluded. Abbreviations: SCI, spinal cord injury; SR, spinal reflex; SCIM II, Spinal Cord Independence Measure II; WISCI, Walking Index for Spinal Cord Injury II; AIS, American Spinal Injury Association Impairment Scale.
The correlations in Figures 1A and 1C seem to be split up into 2 clusters of incomplete SCI subjects consisting of 8 subjects showing dominant late SR components (SR behavior < −0.5) associated with no or poor walking ability and 10 SCI subjects with dominant early SR components (SR behavior > 0.0) associated with moderate to good walking ability. In addition, one might argue that the high correlation between the SR behavior and the WISCI II score could be solely attributed to the SCI subjects who were not able to walk (WISCI II score of 0). However, if these subjects were excluded from data analysis, the correlation was still good (Figure 1B; ρ = 0.622). There was a moderate to good correlation between SR behavior and LEMS (ρ = 0.611, P < .01) indicating that greater muscle strength in muscles of the lower extremity is associated with a dominant early SR component.
Effect of Locomotor Training
Seven incomplete SCI subjects underwent individually adapted locomotor training lasting 4 weeks. Figure 2 shows the changes in SR behavior and SCIM II mobility score from pretraining, posttraining, and follow-up assessment for all 7 SCI subjects. In every assessment, stimulation intensity for SR was kept constant at 2 times the MT of the abductor hallucis muscle. There were improvements in both the SR behavior (P = .018) and the SCIM II mobility score (P = .038) after the locomotor training sessions. However, neither of the other walking tests (WISCI II, 10MWT) nor the LEMS changed significantly over the 4 weeks of training or at the follow-up assessment.
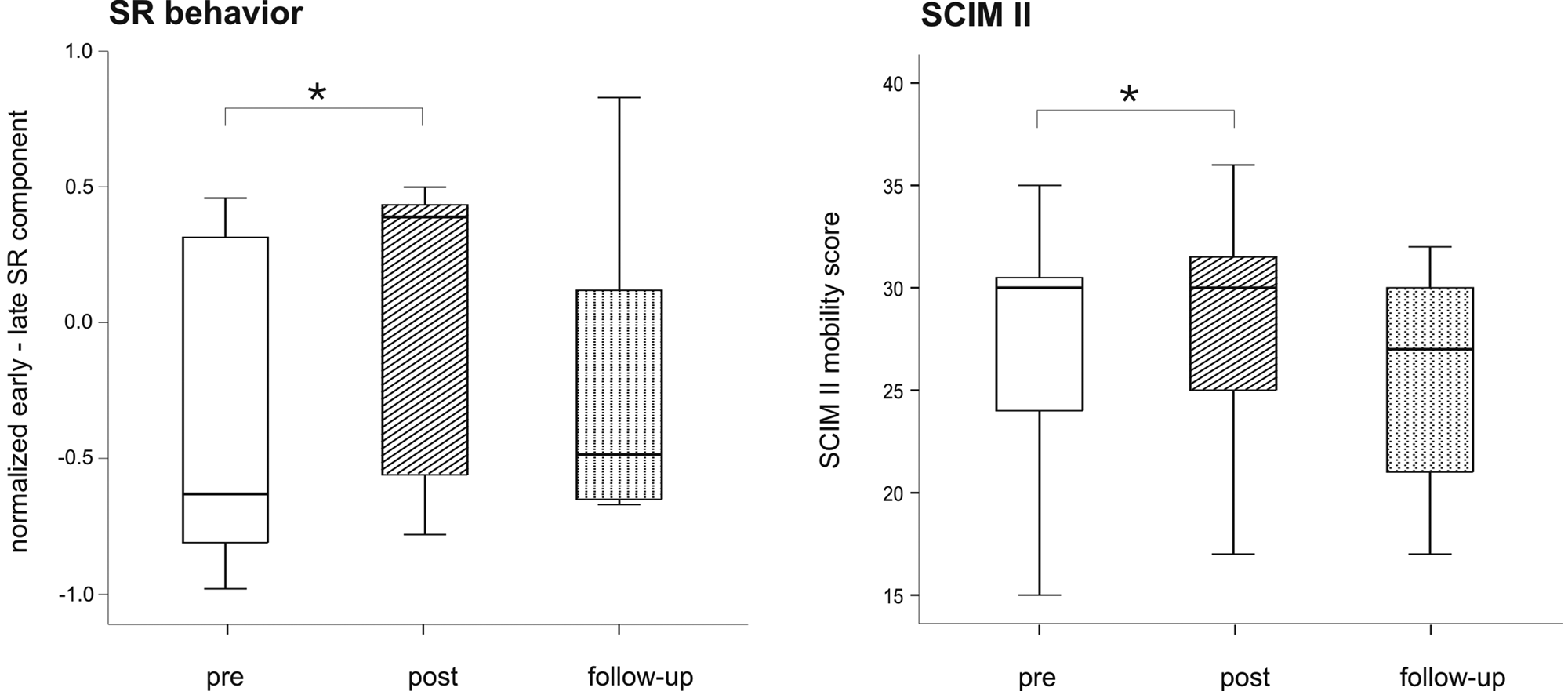
Influence of locomotor training on SR behavior and SCIM II score. The descriptive statistic of the SR behavior and the SCIM II mobility score is depicted in boxplots for the pretraining, posttraining, and follow-up assessments for all trained, incomplete SCI subjects (n = 7, n = 6 for follow-up). Asterisks above the columns define the level of significance of the between-assessments comparison (*P < .05). Abbreviations: SR, spinal reflex; SCIM II, Spinal Cord Independence Measure II; SCI, spinal cord injury.
Figure 3 shows the effect of a locomotor training on SR behavior and functional outcome in a severely affected AIS C subject. The mean values of 10 evoked SR at the beginning of (pretraining), after (posttraining) training, and 6 months after the last training session (follow-up) of this severely affected, incomplete SCI subject are displayed in Figure 3A. A late SR component dominated in the pretraining assessment in both TA muscles (latency left 201 ms, right 165 ms). After 4 weeks of an assisted, body weight supported, treadmill training combined with FES of TA and peronaeus muscles and stimulation of a flexion reflex by strong stimuli applied to the peroneal nerve behind the caput fibulae, an early SR component (latency left 90 ms, right 92 ms) appeared in addition to the late SR component. Six months after the last training session and without stepping practice no early SR component could be elicited (only a late component: latency left 196 ms, right 226 ms). Figure 3B shows the functional changes in walking ability of the same subject. The SCIM II mobility score, the 10MWT and the LEMS improved after 4 weeks of locomotor training but worsened after 6 months of no locomotor practice at home. In particular, the proximal leg muscles (hip flexors and knee extensors) benefited from the locomotor training.
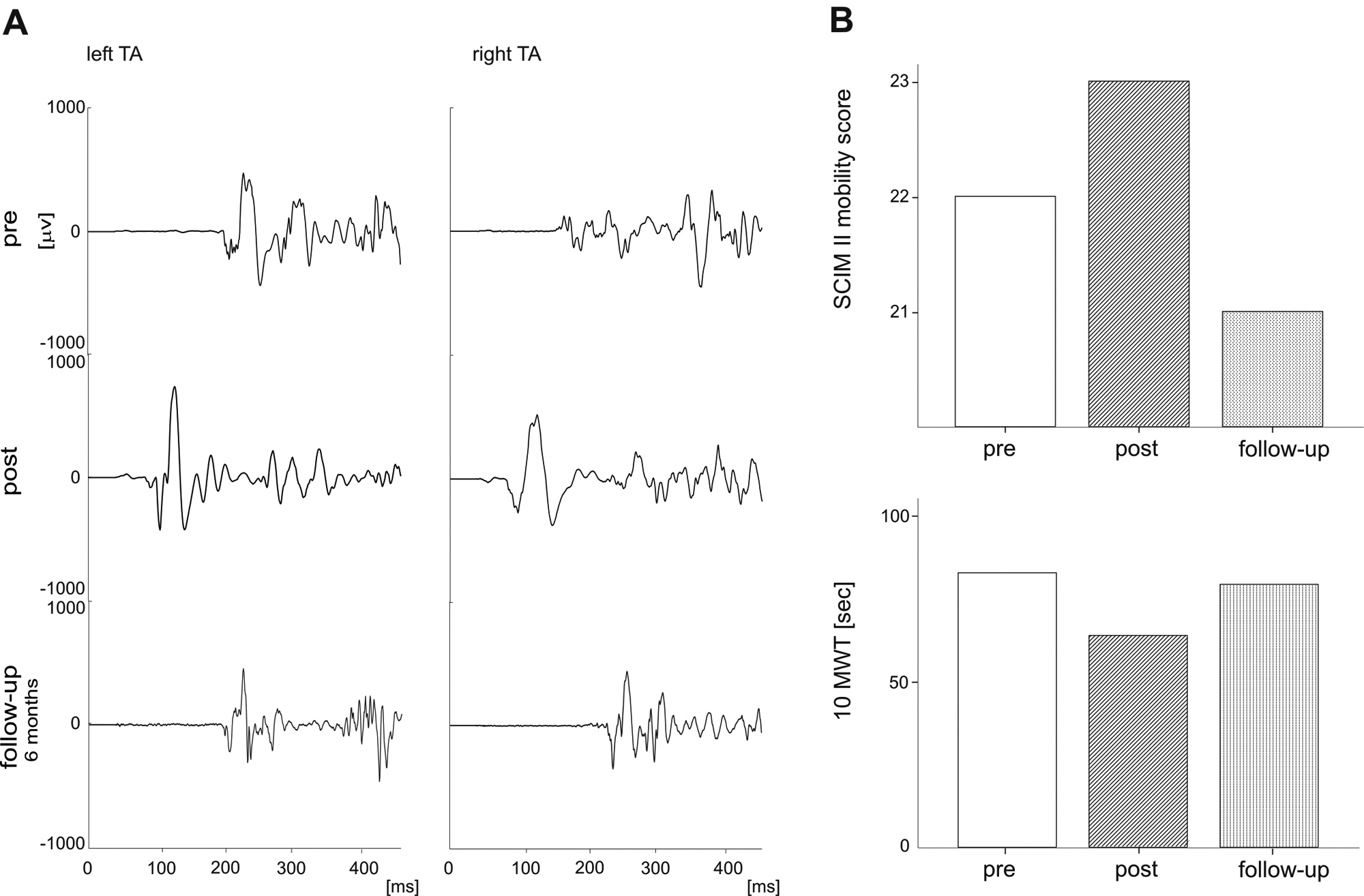
Changes in SR and walking ability after a locomotor training. A representative example of the effect of locomotor training in an incomplete SCI subject (male, 59.9 years; AIS C; level of lesion C5; duration of lesion 7.7 years) is depicted. (A) The SR responses in the TA muscles of the left and the right leg were recorded at 3 different time points: pretraining, posttraining, and at a follow-up 6 months after the end of the training (training duration was 4 weeks). (B) Changes in SCIM II mobility score and 10 MWT from pretraining to posttraining and follow-up assessments. Abbreviations: SR, spinal reflex; SCI, spinal cord injury; AIS, American Spinal Injury Association Impairment Scale; TA, tibialis anterior; SCIM II, Spinal Cord Independence Measure II; 10 MWT, 10-meter walking test.
Figure 4 shows the changes in leg muscle EMG activity in this severely affected incomplete SCI subject before and after locomotor training. The EMG recordings during assisted walking within the DGO before (Figure 4A) and after 1 month of a locomotor training (Figure 4B) are shown. At the beginning of the training period, the leg muscle activity considerably decreased during 10 minutes of assisted walking (RF, −67%; BF, −80%; TA, −12%; and GM, −84%). After the training sessions (Figure 4B), EMG activity showed less of a decrease (RF, −21%; BF, +10%; TA, +8%; and GM, +2%).
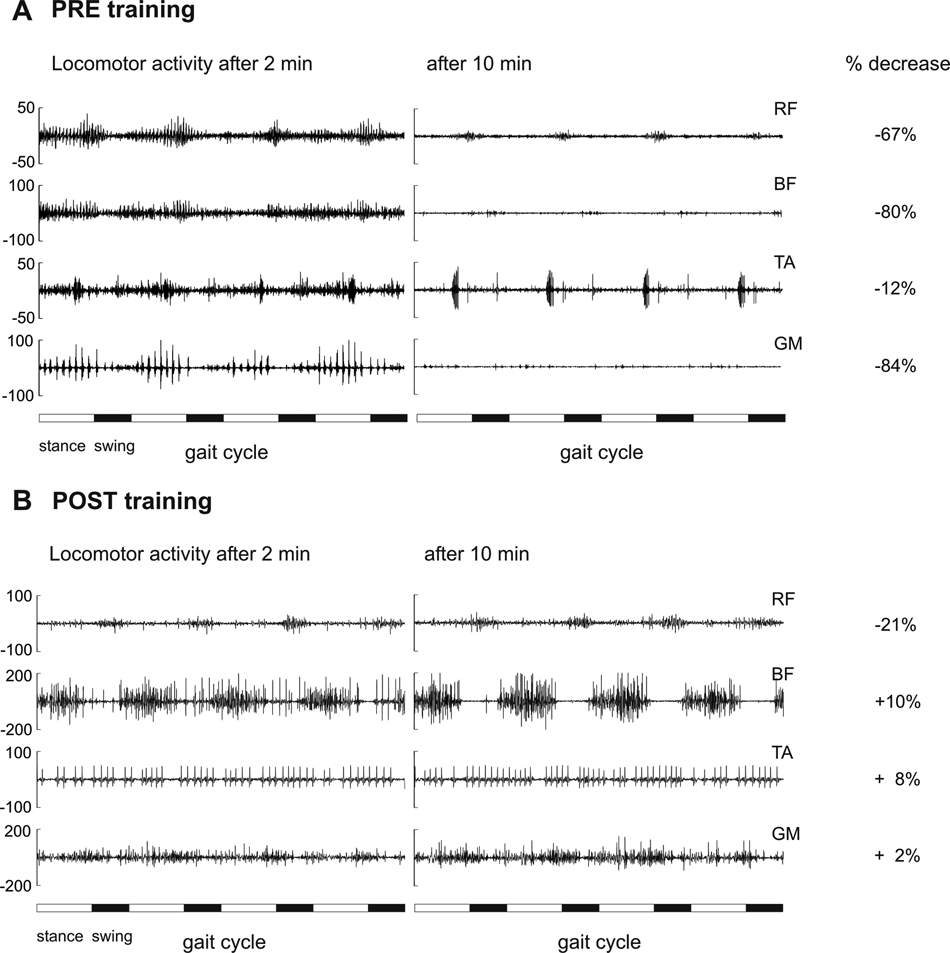
Effect of locomotor training on EMG exhaustion. Representative EMG recordings of 4 leg muscles during assisted walking at 2 and 10 minutes are displayed for pretraining (A) and posttraining assessment (B). Relative decreases of RMS value (in percentage of the initial value) of 4 gait cycles from 2 to 10 minutes of assisted walking are depicted on the right line. Abbreviations: EMG, electromyography; RMS, root mean square; RF, rectus femoris; BF, biceps femoris; TA, tibialis anterior; GM, gastrocnemius medialis.
Two motor complete paraplegic subjects served as control subjects. Neither the SR nor the functional outcome was affected by the locomotor training. The late SR component as well as the walking ability assessed by the SCIM II mobility score, the WISCI II and the gait speed persisted over the training period whereas the LEMS remained at baseline values.
Discussion
This study investigated the relationship between the SR behavior and walking ability in SCI subjects. The main findings were as follows: (a) The more dominant the early SR component, the better the locomotor ability of an SCI subject. This was reflected in increased daily mobility, less need for assistive walking devices, faster walking speed and higher leg muscle strength. (b) Intense locomotor training was followed by both a change in balance of SR toward the early SR component and an improvement of walking ability. Thus, SR behavior might be a useful marker for individual walking ability, which is modifiable by locomotor training after an SCI.
Spinal Reflex and Walking Ability
A close relationship between the generation of SR and locomotor activity has been shown for rats
3
and cats19,20: First, the late SR response in rat with transected spinal cord is associated with the recovery of locomotor function,
3
and this is assumed to correspond to the appearance of the early SR component described here. Second, the long-latency reflex discharges in the spinal cat evoked by administration of
Correspondingly, in humans the neuronal circuits underlying the SR have been suggested to share the same spinal circuitries for locomotion.24,25 In chronic nonambulatory SCI subjects, locomotor EMG exhaustion during assisted walking was associated with a shift from an early to a late SR component, whereas in incomplete SCI subjects the presence of an early SR component was associated with a preserved and stable locomotor EMG activity during 10 minutes of assisted walking. 4
Our present findings suggest that in chronic, incomplete SCI subjects, the presence of an early SR component is associated with walking ability whereas in chronic, severely affected SCI subjects, immobility leads to dominance of the late SR component. Hence the SR component might serve as a marker to identify chronic SCI subjects with “functional” spinal locomotor circuitries that have the potential to ambulate.
The functional state of spinal locomotor circuitries, reflected in the SR behavior, seems to depend rather on the regular practice of locomotor movements in chronic SCI subjects than on the “completeness” of the SCI. Thus, an appropriate afferent input is essential to maintain spinal locomotor circuitries. 26 The loss of this input, because of immobility after an SCI, leads to a neuronal dysfunction of spinal locomotor circuits, specifically comprising a dominant late SR component and EMG exhaustion.5,6
Plasticity of Locomotor Circuits
Neuronal plasticity within the central nervous system can contribute to improved walking ability after SCI and plays an essential role in the recovery of locomotion.27,28 Neuroplastic changes can occur at cortical, brainstem, and spinal levels. One conceptual basis for specific types of locomotor training is based on enhancing neuronal plasticity 29 as well as the reorganization of sensorimotor pathways. 30
In our study, the 6 incomplete SCI subjects who received gait training without FES showed only mild gains, whereas the 1 SCI subject who received FES in addition showed good improvements of walking ability. Nevertheless, a functional improvement after the training period was always associated with a strengthening of the early SR component.
The rather limited effects on locomotor improvements might be explained by the following aspects. First, the training period over 4 weeks might have been too short to induce stronger long-lasting changes in functional outcome. Second, the changes induced in spinal locomotor circuitries were less reflected in the functional outcome measures used, and third, 4 (AIS D) out of the 7 SCI subjects already had a moderate to good walking ability (floor effect) at the beginning of the study prior to training. However, training with the DGO Lokomat is more effective in AIS C SCI subjects. 31 Correspondingly, the most severely affected incomplete SCI subject with dominant late SR components at the beginning of the intervention profited the most from a combined locomotor and FES training in terms of neuronal function, locomotor ability, and SR behavior. Such a combined training approach provides more appropriate input to “nonfunctional” neuronal circuits that might turn them into a “functional” state. This is in line with the observation in rats that a combination of locomotor training, epidural stimulation, and pharmacological intervention leads to a conversion of spinal circuits from a “nonfunctional” into a “functional” state. 32
Regular training combined with afferent input to spinal neuronal circuits seems to prevent neuronal dysfunction and is even able to regain normal neuronal function in severely affected, incomplete SCI subjects. In contrast, no change in locomotor capacity occurs in motor complete SCI subjects after intense locomotor training. 31 Correspondingly, a dominant late SR component as a marker of no walking ability persisted even after the training in motor complete paraplegic subjects.
This study shows that the SR changes in parallel with ambulatory improvement following locomotor training. The effects on SR behavior can be because of activation of spinal neuronal circuits or, alternatively, because of an influence of the training on supraspinal centers. For example the application of FES has been shown to increase the excitability of the cortex or/and corticospinal pathways.33,34
Physiological Considerations
In vertebrates, an interaction between excitatory and inhibitory neuronal circuits shapes the locomotor pattern. 35 Following SCI in cats, an imbalance between excitatory and inhibitory inputs to the spinal neuronal circuits occurs.36,37 As a consequence, blocking inhibitory transmission can markedly improve walking capacity. 38 In immobilized SCI subjects, inhibitory circuits might become dominant, while the function of excitatory interneurons weakens.5,6 As a consequence, there was an exhaustion seen in the EMG amplitude during assisted walking, associated with a suppression of the early SR component and a facilitation of pathways mediating the late SR component. In contrast, in motor incomplete, mobile SCI subjects proprioceptive feedback information drives excitatory spinal neuronal circuits reflected in a dominance of the early SR component.
Conclusions
We show that the SR could serve as a marker for the functional state of spinal locomotor circuitries and could be used as a new tool to assess changes within these circuits. The SR might be used in addition to clinical measures in SCI subjects to estimate the dysfunction of neuronal circuits or the absence of dysfunction, for example, in psychogenic plegia or plegia due to biomechanical rather than neuronal constraints.
Footnotes
Acknowledgements
We thank R. Labruyère and E. Limacher for help in data acquisition and M. Starkey for her editorial assistance.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the European Commission by the seventh framework program through the project “Spinal Cord Repair” (HEALTH-F2-2007-201144) as well as by the EMDO and Hartmann Müller Foundation.
