Abstract
Background. Upper-limb chronic stroke hemiplegia was once thought to persist because of disproportionate amounts of inhibition imposed from the contralesional on the ipsilesional hemisphere. Thus, one rehabilitation strategy involves discouraging engagement of the contralesional hemisphere by only engaging the impaired upper limb with intensive unilateral activities. However, this premise has recently been debated and has been shown to be task specific and/or apply only to a subset of the stroke population. Bilateral rehabilitation, conversely, engages both hemispheres and has been shown to benefit motor recovery. To determine what neurophysiological strategies bilateral therapies may engage, we compared the effects of a bilateral and unilateral based therapy using transcranial magnetic stimulation. Methods. We adopted a peripheral electrical stimulation paradigm where participants received 1 session of bilateral contralaterally controlled functional electrical stimulation (CCFES) and 1 session of unilateral cyclic neuromuscular electrical stimulation (cNMES) in a repeated-measures design. In all, 15 chronic stroke participants with a wide range of motor impairments (upper extremity Fugl-Meyer score: 15 [severe] to 63 [mild]) underwent single 1-hour sessions of CCFES and cNMES. We measured whether CCFES and cNMES produced different effects on interhemispheric inhibition (IHI) to the ipsilesional hemisphere, ipsilesional corticospinal output, and ipsilateral corticospinal output originating from the contralesional hemisphere. Results. CCFES reduced IHI and maintained ipsilesional output when compared with cNMES. We found no effect on ipsilateral output for either condition. Finally, the less-impaired participants demonstrated a greater increase in ipsilesional output following CCFES. Conclusions. Our results suggest that bilateral therapies are capable of alleviating inhibition on the ipsilesional hemisphere and enhancing output to the paretic limb.
Keywords
Introduction
For stroke patients with hemiplegia, motor recovery typically plateaus by 6 months, and about 65% of patients are unable to incorporate the paretic arm and hand into their daily activities. 1 The motor deficit is thought to persist, in part, because of an imbalance between the ipsilesional and contralesional hemispheres often described using the model of “interhemispheric competition.” According to this model, paresis originates from the loss of corticospinal output to the impaired upper limb and persists because of additional interhemispheric imbalances. The contralesional (nonlesioned) hemisphere exerts unopposed inhibition on the weak ipsilesional (lesioned) hemisphere, which further reduces output to the paretic limb.2-4 Based on this model, one therapeutic strategy is to restrict activity of the nonparetic limb and encourage unilateral movement of the paretic limb; this is thought to limit interhemispheric inhibition (IHI) and improve the likelihood of functional recovery.
However, the interhemispheric competition model has recently been debated in the literature, where many studies have refuted its premise,5-8 suggest that the model is task specific, 9 or have demonstrated that the model only applies to a subset of the chronic stroke population.4,10-13 Therefore, it is important to consider therapeutic approaches that may engage alternative neurophysiological strategies. One such approach described in the literature is bilateral based therapies, where bilateral interventions have the potential to modulate the control and recovery of both the paretic and nonparetic limb and affect the recovery of both unilateral and bilateral tasks.
Several groups, including our own (for a review, please refer to Cunningham et al 14 ), have suggested that bilateral therapies may be a useful/feasible alternative for such patients who suffer from extensive ipsilesional damage. For example, some bilateral therapies, such as active passive bilateral training15,16 and contralaterally controlled functional electrical stimulation (CCFES)17-20 use movement of the nonparetic limb to drive/assist in the movement of the paretic limb. Movement of the nonparetic limb may help recruit viable, uncrossed pathways projecting from the contralesional hemisphere to the paretic limb, and this may support recovery of the paretic limb. Furthermore, bilateral movements, in general, may invoke mutual disinhibition of both hemispheres, which may support corticospinal output from the ipsilesional hemisphere.21-24 For example, Knutson et al 19 have shown that 12 weeks of training with CCFES, a bilateral therapy, reduces impairment and improves hand dexterity more than an equivalent dose of cyclic neuromuscular electrical stimulation (cNMES), a unilateral therapy.
Therefore, we sought to understand the differences in mechanisms of bilateral versus unilateral based therapies by comparing the neurophysiological effects of CCFES and cNMES. In addition, we investigated whether these effects vary as a function of severity of motor impairment. Patients with chronic stroke received single sessions of CCFES and cNMES in a randomized, crossover experiment. Transcranial magnetic stimulation (TMS) was used to assess IHI imposed on the ipsilesional hemisphere, ipsilesional corticospinal output, and ipsilateral (uncrossed) output to the paretic limb. Reliability of assessments was also studied by having a cohort of patients undergo a control condition, during which they received no stimulation/training. We hypothesize that bilateral CCFES will result in reduced IHI of the ipsilesional hemisphere and enhanced ipsilesional and ipsilateral corticospinal output to the paretic upper limb.
Methods
Participants
A convenience sample of 15 participants in the chronic phase of recovery (>6 months) following a first-ever ischemic or hemorrhagic stroke were enrolled. We identified the prestroke dominant hand based on the Edinburgh Handedness Inventory assessment. 25 We included participants with wide-ranging upper-limb impairment, including those with just trace movement at the fingers, thumb, or wrist as well as participants who were well recovered and still reported inadequate ability to use the paretic hand in daily life. Patients were excluded if they possessed any contraindications to the use of TMS, including cardiac pacemaker, metal implant in the head, history of seizures as an adult, or ongoing use of neuroactive or psychoactive medications. The institutional review board of the Cleveland Clinic approved the study. All participants provided written informed consent.
Clinical Assessment of Impairment of the Upper Limb
The Upper-Extremity Fugl-Meyer (UEFM) Scale was used to measure motor impairment of the paretic upper limb. The UEFM is a reliable and valid measure of poststroke upper-limb motor impairment.26-31 The UEFM items take into account synergy patterns, isolated strength, coordination, and hypertonia. Volitional movement of the upper limb (shoulder, elbow, forearm, wrist, and hand) is examined in and out of synergies. Each item is graded on a 3-point ordinal scale (0, cannot perform; 1, perform partially; and 2, perform fully) and summed to provide a maximum score of 66.
Design
Participants took part in a randomized, crossover experiment in which they received a 1-hour session of unilateral cNMES and a 1-hour session of bilateral CCFES, separated by a ≥1-week interval to allow for washout of effects. Before and after each session, indices of neurophysiology were collected using TMS (Figure 1). To establish within-session and between-session reliability of TMS indices, a cohort of 12 of the 15 participants also underwent an additional session (“control” condition), during which they received no intervention for an hour but underwent TMS testing.
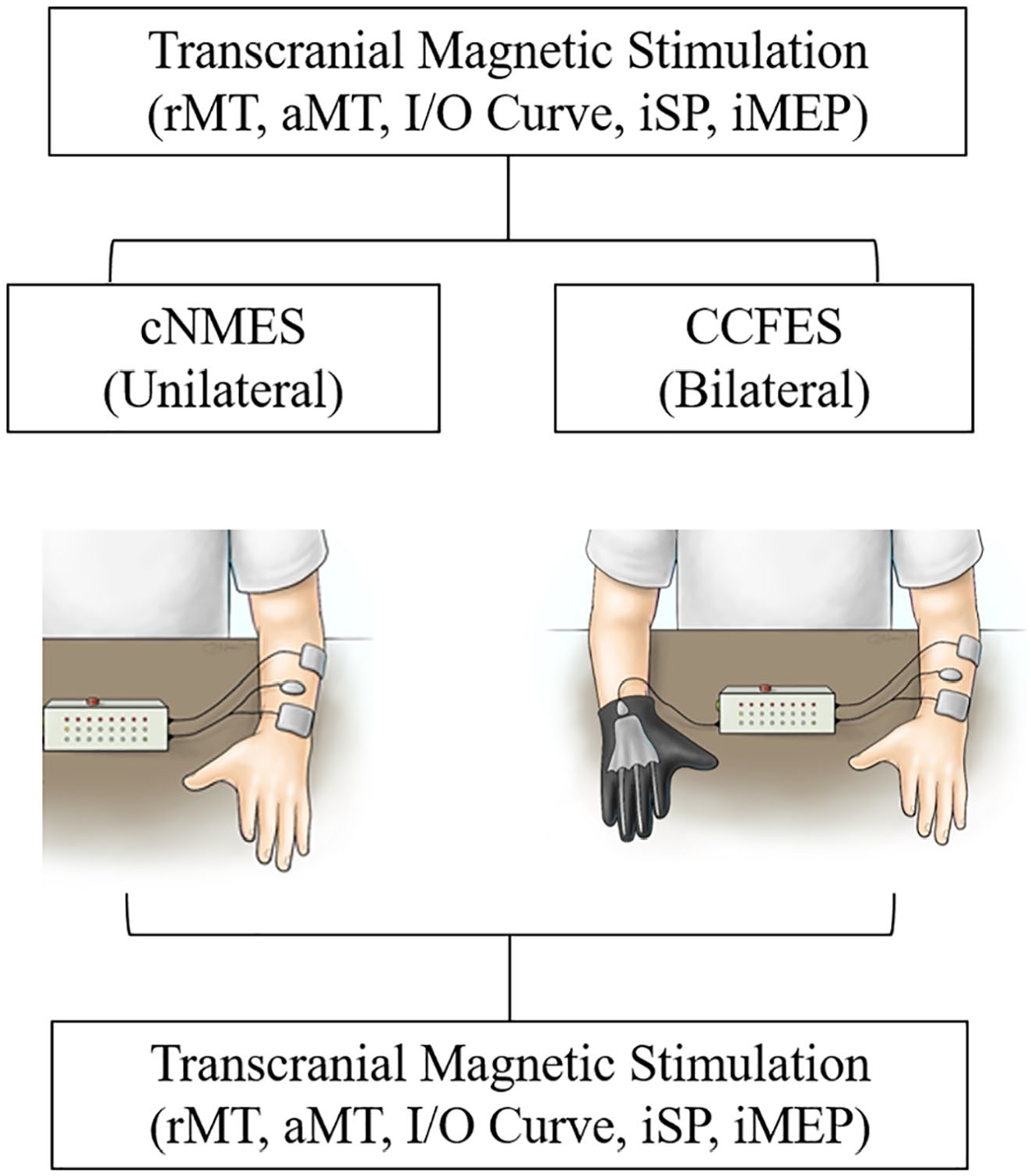
Participants took part in 2 separate sessions separated by a minimum of 1 week: cyclic neuromuscular electrical stimulation (cNMES) and contralaterally controlled functional electrical stimulation (CCFES). Transcranial magnetic stimulation was delivered immediately before and immediately following each session.
Interventions
For both cNMES and CCFES, we positioned surface electrodes (2-inch square and/or 1.25-inch round) on the forearm to produce finger and thumb extension. A picture of the electrode placement was used to ensure consistent electrode placement between sessions. We targeted the extensor digitorum communis (EDC) and extensor pollicis longus muscles. No more than 2 independent monopolar channels (using a common anode) of stimulation were used for any participant in this study. The stimulators used in this study delivered biphasic rectangular current pulses at a frequency of 35 Hz and amplitude of 40 mA. For each electrode, a maximum intensity (pulse duration) of stimulation was determined as that which produced maximum visible hand opening without pain.
For the cNMES session, the stimulator was programmed to automatically and repetitively ramp the pulse durations from zero to the predetermined maximum intensity, which produces functional hand opening in 1 s, maintains the stimulation at maximum intensity for 7 s, and then ramps down the stimulation to zero in 1 s, repeating this cycle every 24 s. Auditory and visual cues indicated when the stimulation ramped on and off, and the participants were instructed to attempt to volitionally open their paretic hand when the stimulation turned on and to not move the nonparetic hand. For the CCFES session, the stimulator was programmed to modulate the pulse duration from each stimulation channel from zero to maximum in proportion to the amount of opening of an instrumented glove worn on the nonparetic hand. The glove consisted of an assembly of 3 bend sensors in cloth sheaths attached to the dorsal side of the index, middle, and ring fingers (Figure 1, right). The stimulator issued auditory and visual cues to tell the participant when and for how long to attempt to open both hands during an exercise session. The visual cues instructed participants to open and then relax both hands with timing that was identical to cNMES. Participants were verbally encouraged to volitionally open both hands in response to the audio and visual cues. For both therapies, participants performed stimulation-mediated hand opening exercises for three 15-minute sets separated by 3-minute breaks. Both cNMES and CCFES resulted in 112 repetitions. During the control session, participants sat quietly with their hands resting on a flat surface for the same amount of time as sessions that involved training with cNMES and CCFES.
TMS Recordings
Using TMS, we measured 3 parameters: (1) inhibition imposed from the contralesional hemisphere on the ipsilesional primary motor cortex (iM1), abbreviated IHI upon iM1; (2) output of residual pathways from the ipsilesional hemisphere to the paretic EDC, termed ipsilesional output; and (3) output of pathways from the contralesional hemisphere to the paretic EDC (ie, via ipsilateral motor pathways), termed ipsilateral output. The procedures for these measurements are described below.
TMS was delivered to the primary motor cortex (M1) via a figure-of-eight coil (70 mm) connected to a monophasic Magstim device (2002 Magstim Co, Whitland, Dyfed, UK). The coil was placed tangential to the scalp, with the handle oriented backward and laterally at a 45° angle to the midsagittal axis. Magnetic resonance imaging (MRI)-based frameless stereotaxy was used to guide TMS application (BrainSight, v2, Rogue Research Inc, Montréal, Canada). 12 The participant’s MRI was used for stereotactic navigation if it was available; otherwise, a template MRI was used instead. Motor-evoked potentials (MEPs) were recorded in the EDC muscle using surface electromyography (EMG; PowerLab 4/25T, AD instruments, Colorado Springs, CO). The amplified (×500) EMG signals were filtered (bandpass, 10 Hz to 2 kHz, and notch filtered at 60 Hz), sampled at 4 kHz, and stored on a personal computer for offline analysis (LabChart, version 7.3, AD Instruments Inc, Colorado Springs, CO).
The TMS hotspot was identified as the scalp site eliciting reliable criterion MEP ≥50 µV in at least 5/10 trials at the lowest stimulator intensity. The lowest TMS intensity that elicited criterion-level MEPs while the subject was instructed to keep the paretic EDC relaxed was termed as the resting motor threshold (rMT; given as percentage maximum stimulator output [MSO]). Because resting state MEPs cannot be elicited in many participants with moderate to severe impairment, we also defined active motor threshold (aMT) as the lowest stimulator intensity required to elicit reliable criterion MEPs ≥200 µV in at least 5/10 trials in the slightly contracted EDC (~15% of maximum voluntary contraction [MVC]; given as percentage MSO).
MVC was collected during maximum isometric finger extension, whereas the EMG was recorded from the EDC. The root mean square value of the EMG was calculated and displayed in real time on a monitor. Participants performed 3 trials of MVC lasting 3 to 5 s each, and the maximum root mean square value was averaged. Participants were provided with a visual target to help them maintain adequate force production.
IHI on Ipsilesional Primary Motor Cortex
IHI was measured using the ipsilateral silent period (iSP). iSP is defined as the suppression of ongoing volitional EMG contraction with TMS given to the ipsilateral motor cortices.32,33 Here, we measured iSP during 50% MVC contraction of the paretic EDC. Suprathreshold TMS pulses were delivered to the hotspot in the contralesional hemisphere at an intensity of 150% to 200% rMT. Onset of iSP was defined when paretic muscle rectified EMG fell below the mean prestimulus EMG (defined as the activity recorded 100 ms prior to TMS) for 5 ms; the termination of iSP was defined when EMG returned to the mean for 5 ms. 34 Trials in which iSPs could not be elicited through visual inspection were discarded from the analysis. On average, stimulator output of 180% ± 40% rMT was required to generate iSPs and on average 19 ± 5 of 25 ± 4 trails were retained for analysis. The same stimulator output was used before and after each session. IHI was computed as a percentage decrease in mean rectified EMG during iSP relative to prestimulus rectified EMG (Equation 1). iSP was used rather than the other commonly used bihemispheric TMS technique for measurement of IHI because the bihemispheric technique is contingent on the availability of MEPs in the paretic muscle. But moderately to severely impaired patients cannot reliably elicit MEPs. Therefore, many groups, including our own, have chosen to use iSPs because it has been shown to be a relevant index of IHI.12,32
Ipsilesional Output
In participants who could elicit MEPs in the actively contracting paretic muscle, ipsilesional output was defined based on an input/output curve. During slight (~15% MVC) contraction of the paretic EDC, TMS pulses were delivered to the ipsilesional hotspot at incremental intensities (10%) between 90% and 150% aMT while amplitude of resultant MEPs was noted. Ten pulses were delivered at each TMS stimulus intensity. Order of intensities was randomized and changed after every 5 pulses. The same intensities were used before and after each session. Several periods of rest were given to avoid muscle fatigue. Peak-to-peak MEP amplitudes recorded at each intensity level were ranked in ascending order of size. The top 20% and bottom 20% of the ranked trials (4 of 10) were then discarded from the analysis.35,36 This method has been shown to give an accurate representation of centrality. 36 We normalized all MEP amplitudes (pre and post) to the size of the maximum MEP acquired at baseline (MEPMaxima). From this normalized recruitment curve, area under the curve (AUC) and percentage change in MEP amplitude at each intensity was calculated. 37 Because MEPs could only be evoked in the paretic EDC in 9 of the 15 participants—and of those, high aMT thresholds precluded testing at intensities higher than 100% MSO in 2 participants—input/output curve measurements were completed in 7 of 15 individuals.
Ipsilateral Output
Output of ipsilateral pathways dedicated to the paretic hand was studied as size of the ipsilateral MEPs (iMEPs) evoked in the contracting paretic EDC (50% MVC) with suprathreshold TMS (150% to 200% rMT) given to the contralesional hemisphere. The presence or absence of iMEPs was determined in responses collected over 25 TMS pulses. Based on the work of Tazoe and Perez, 38 we first visually inspected iMEPs, and those with amplitudes that exceeded mean prestimulus EMG amplitude were labeled as iMEPs. Then, an average of all iMEP waveforms was created. The onset of the averaged iMEP was defined as the time point at which its amplitude exceeded the mean rectified prestimulus EMG activity by 1 SD. Termination of the averaged iMEP was defined as the time point at which its amplitude fell below 1 SD of the mean rectified prestimulus EMG. Area (size) of the iMEP was then calculated using Equation (2):
Statistics
We tested normality of the data using a Shapiro-Wilk test. Within-session reliability was ascertained by comparing data collected before and after the control condition using the intraclass coefficient (ICC). ICC was conducted with absolute agreement at a P value of .05, where an ICC value of ≥0.6 indicates good to excellent reproducibility. Between-session reliability was also defined using ICC based on data collected at baseline for each session (CCFES, cNMES, and control; n = 12).
Effects of CCFES and cNMES on TMS metrics were compared using a 2-way repeated-measures ANOVA [Intervention (cNMES, CCFES) × Time] (n = 15). The percentage pre to post difference was compared between CCFES and cNMES using paired t-tests for post hoc analysis. Bonferroni correction was applied to adjust the P value for multiple comparisons. Secondarily, the influence of baseline impairment severity on changes in TMS parameters was explored for each condition using bivariate Pearson correlation analysis. All tests were 2-tailed, conducted at an α-level of significance of .05.
Results
Participant characteristics are presented in Table 1. Mean (±SD) UEFM was 42 ± 16.13 (range: 15 to 63). Lesions were primarily subcortical (9), followed by mixed cortical/subcortical (2), pontine (2), and cortical (1). We were unable to determine lesion location for 1 participant because imaging was unavailable. A total of 14 participants were classified as right-handed, and 9 participants had a stroke that affected their dominant hand.
Participant Demographics. a
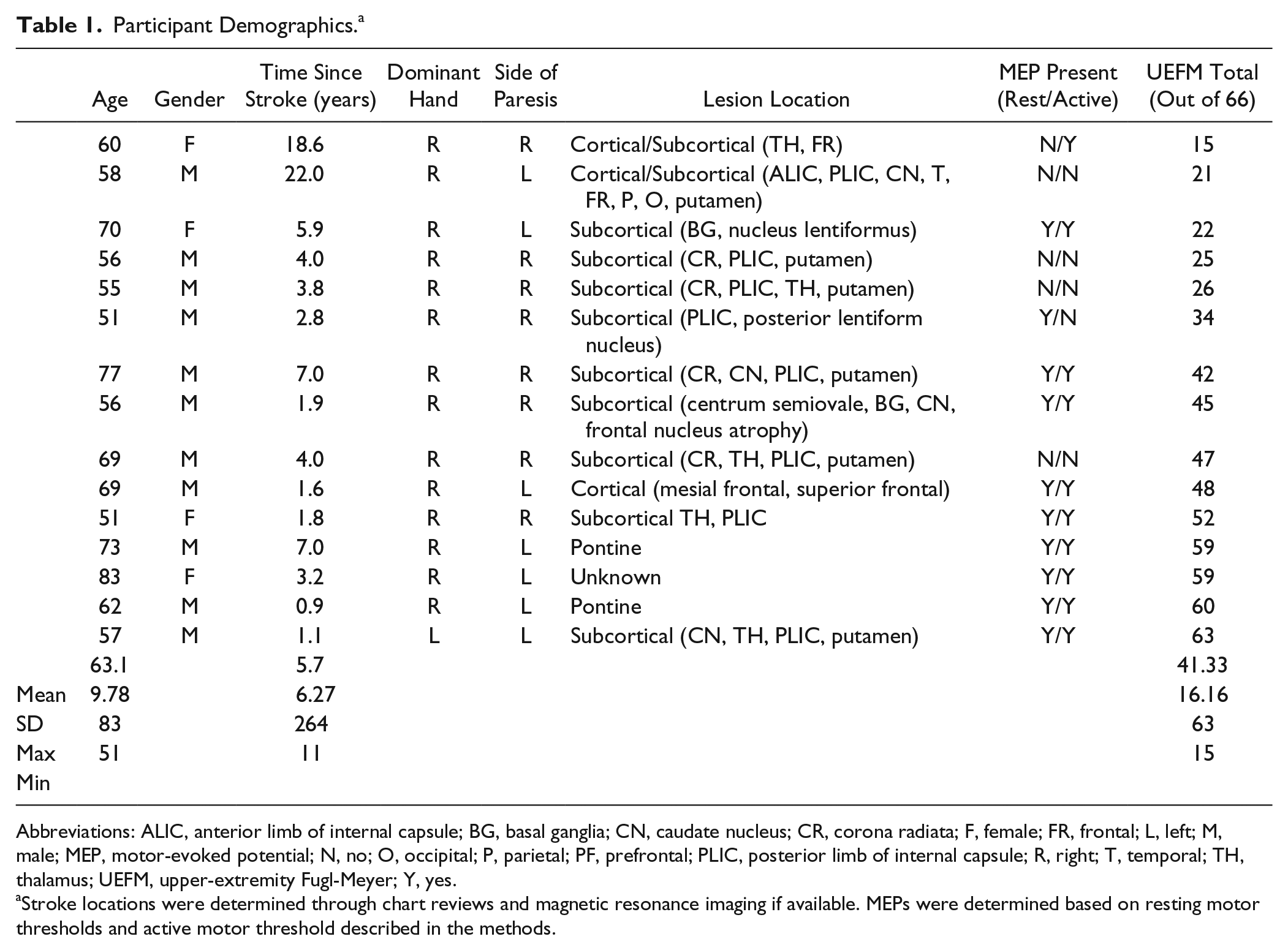
Abbreviations: ALIC, anterior limb of internal capsule; BG, basal ganglia; CN, caudate nucleus; CR, corona radiata; F, female; FR, frontal; L, left; M, male; MEP, motor-evoked potential; N, no; O, occipital; P, parietal; PF, prefrontal; PLIC, posterior limb of internal capsule; R, right; T, temporal; TH, thalamus; UEFM, upper-extremity Fugl-Meyer; Y, yes.
Stroke locations were determined through chart reviews and magnetic resonance imaging if available. MEPs were determined based on resting motor thresholds and active motor threshold described in the methods.
Within-Session Reliability of TMS Indices
The control condition demonstrated that there was excellent reliability of ipsilesional rMT and aMT (rMT ICC = 0.985, P < .001; aMT ICC = 0.996, P < .001) and MEPmaxima used for normalization of MEPs collected from the input/output curve measurements (ICC = 0.989; P < .001). AUC for the input/output curve also showed good reliability (ICC = 0.675; P = .09). IHI and size of iMEP showed excellent reliability (ICC = 0.982, P < .001, and ICC = 0.778, P = .023, respectively).
Between-Session Reliability of TMS Indices
There was excellent between-session reliability as well for ipsilesional rMT, aMT, MEPmaxima, and input/output curve AUC (ICC = 0.842, P = .001; ICC = 0.968, P < .001; ICC = 0.931, P < .001; ICC = 0.909, P = .001, respectively). IHI also showed excellent between-session reliability (ICC = 0.909; P < .001). iMEPs, however, showed poor between-session reliability (ICC = −0.922; P = .935).
CCFES Versus cNMES
There was no change in MVC following either intervention (CCFES Pre: 0.18 ± 0.03 mV, Post: 0.18 ± 0.03 mV, P = .84; cNMES Pre: 0.2 ± 0.03 mV, Post: 0.2 ± 0.04 mV, P = .87). Two-way ANOVA revealed that there was a significant Time × Intervention interaction for IHI on iM1: F(1, 14) = 8.44; P = .012. IHI was significantly reduced following CCFES compared with cNMES (−9.5% ± −3.7% vs +4.5% ± 5.2%; t = −2.2, P = .049; Figures 2A and 2B). Furthermore, ipsilesional output was significantly preserved following CCFES compared with cNMES. There was a significant Time × Intervention interaction for AUC: F(1, 6) = 9.18; P = .023. AUC was significantly reduced after cNMES compared with CCFES (−15.3% ± 6.67% vs +4.2% ± 5.62%; t = 3.2, P = .018; Figure 3A). CCFES also resulted in larger increases in normalized MEP amplitudes collected at 110% aMT and 120% aMT compared with cNMES (t = 3.3, P = .016, and t = 2.52, P = .045, respectively; Figure 3B). We were able to identify iMEPs in ~7 ± 3 (out of 25 ± 4) trials before and after each session (pre: 6.8 ± 2.7 [SD]; post: 7.1 ± 3.3 [SD]; P = .45). iMEP latencies were significantly later than contralateral MEPs evoked in the nonparetic EDC (5.75 ± 6.3 ms [SD]; P = .001). We observed a nonsignificant Time × Intervention interaction in iMEP amplitude: F(1, 14) = 2.59; P = .13.
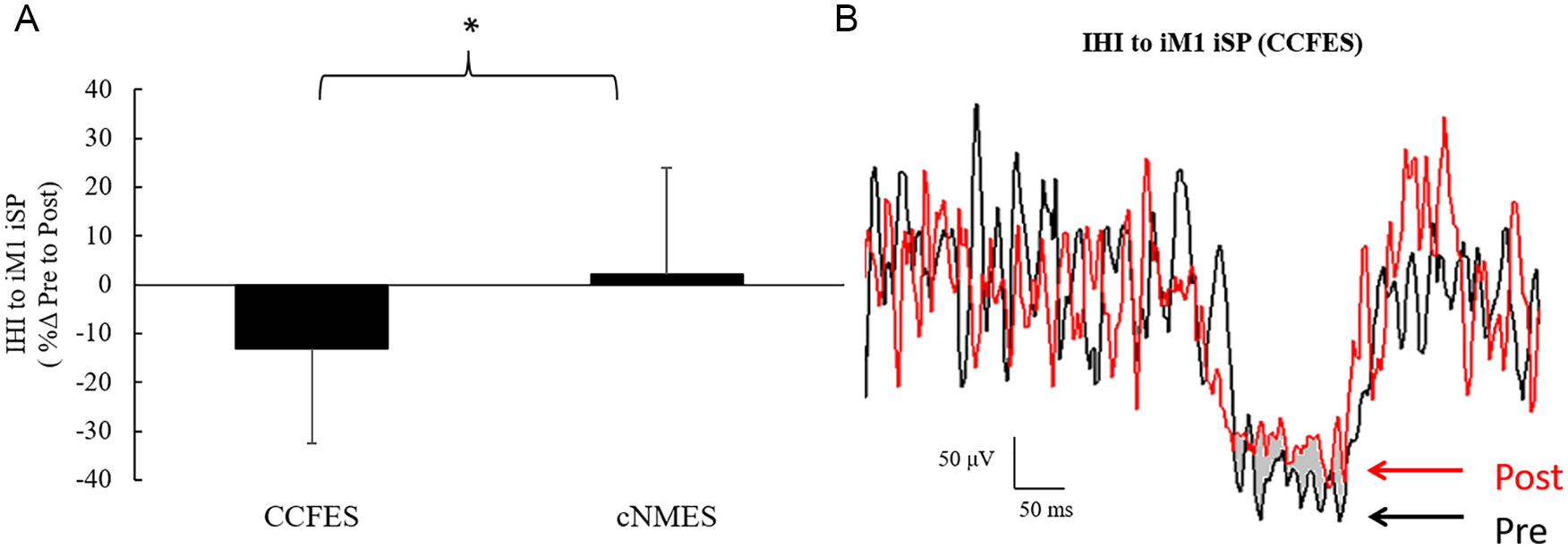
A. Interhemispheric inhibition (IHI) to ipsilesional primary motor cortex (iM1) decreased following CCFES, but not after cNMES. B. One patient representation of pre–ipsilateral silent period (iSP) and post-iSP (pre = black; post = red). *P < .05.
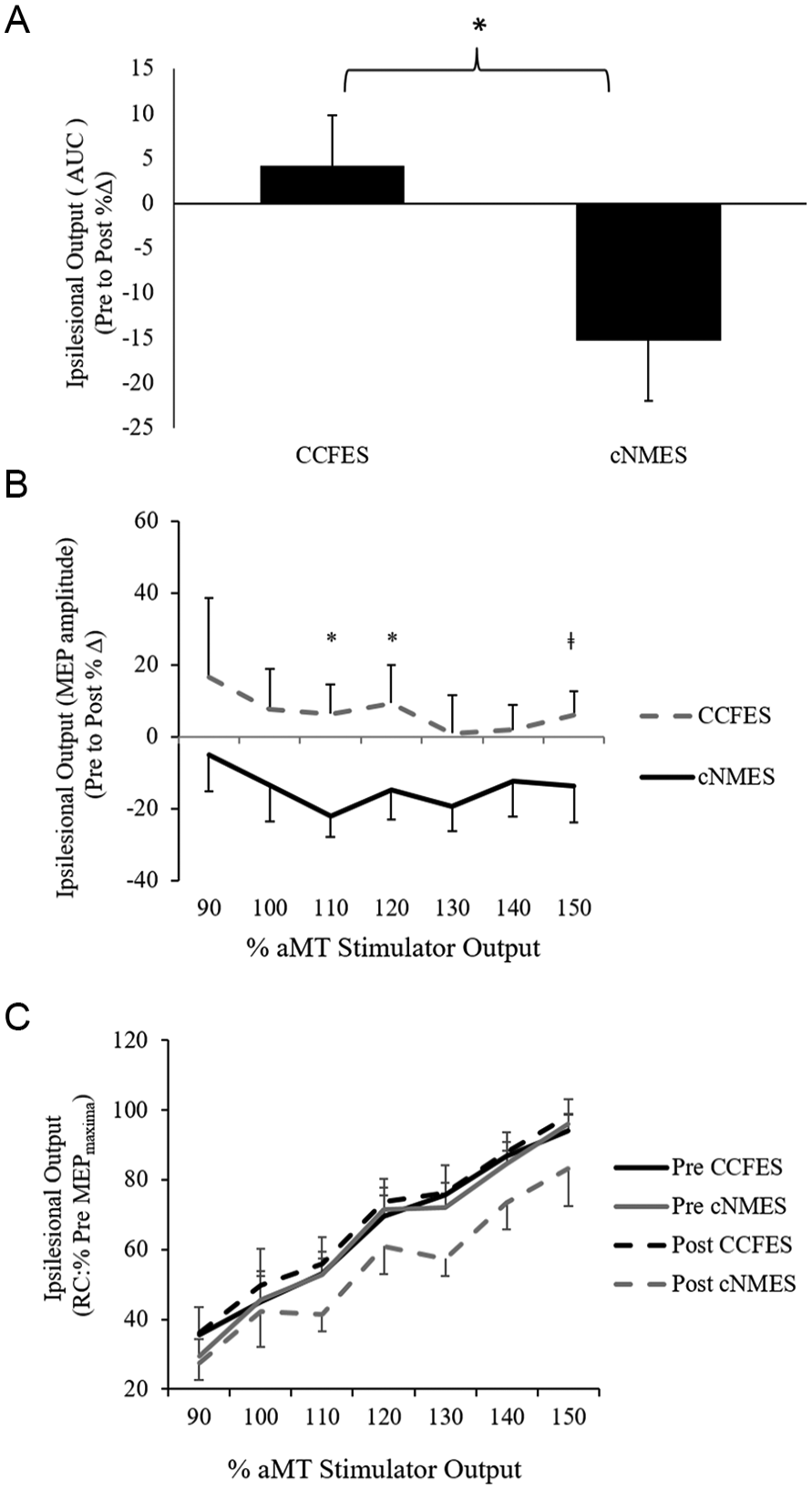
A. Ipsilesional output (recruitment curve area under the curve) decreased following cNMES when compared with CCFES. B. The percentage change in recruitment curves differed between CCFES and cNMES at 110%, 120%, and 150% aMT. C. Mean normalized ipsilesional recruitment curve for all participants’ pre and post cNMES and CCFES. Error bars are SEs. *P < .05, ‡P < .1.
Effect of Severity of Impairment
We studied how the change in TMS metrics observed with each session varied as a function of baseline severity of impairment. The changes in TMS metrics following cNMES did not vary as a function of impairment; however, change in IHI and ipsilesional output following CCFES did correlate with baseline impairment. Participants with more severe (ie, lower UEFM) motor impairment scores trended toward greater reduction in IHI (r = 0.503, P = .056; Figure 4A). In addition, participants with higher motor impairment scores showed the largest increases in ipsilesional output (AUC; r = 0.78, P = .04; Figure 4B). There was no correlation between baseline impairment and change in iMEP following CCFES, though participants with greater motor impairment tended to have higher iMEP amplitudes at baseline than participants with less impairment (r = −0.730, P = .002; Figures 4C and 4D).
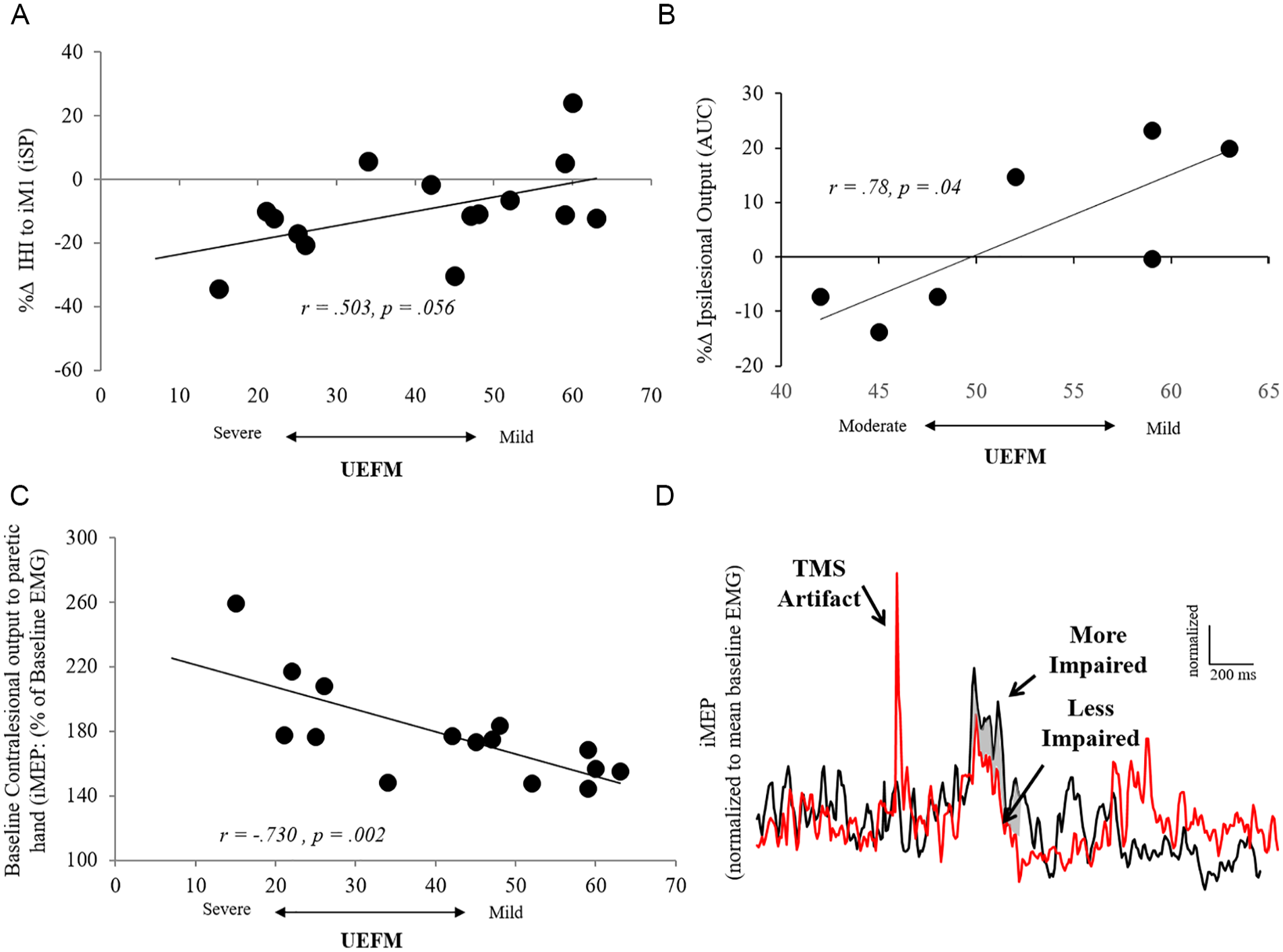
Neurophysiology as a function of motor impairment. A. Patients with more severe motor impairments demonstrated the greatest reduction of inhibition on the affected hemisphere following CCFES. B. Patients with more mild motor impairments showed the greatest increase in ipsilesional output following CCFES. C. We found no intervention effect of iMEP and its relationship to motor severity; however, patients with greater motor impairment showed greater iMEP output at baseline. D. Representation of 2 patients (more impaired [UEFM: 21] vs less impaired [UEFM: 63]) and their respective impaired upper-limb iMEP.
Discussion
This study has provided preliminary evidence that bilateral CCFES and unilateral cNMES have different neurophysiological mechanisms in chronic stroke participants. Unlike unilateral cNMES, IHI from the contralesional motor cortices decreased and ipsilesional output to the paretic limb was maintained following bilateral CCFES. Furthermore, we found that the less-impaired participants showed greater increase in ipsilesional output, and participants with more severe impairments showed a trend toward having greater reduction in IHI. However, we found no evidence that 1 hour of bilateral CCFES or unilateral cNMES movements differed in ipsilateral output to the paretic hand. Overall, our findings suggest that bilateral CCFES can recruit cooperative potential between the contralesional and ipsilesional motor cortices across different ranges of impairment.
Our most important finding is that bilateral CCFES reduces IHI to ipsilesional M1 across the majority of participants (mild to severely impaired; Figures 2A and 4A). This outcome would not be predicted by the interhemispheric competition model, which instead predicts that activity in the contralesional hemisphere will increase inhibition of ipsilesional M1. However, several groups have challenged the premise of the interhemispheric competition model.4,11,39 According to a more recent hypothesis called the bimodal balance-recovery model by Di Pino et al, 4 the degree of IHI imposed by the contralesional hemisphere varies based on the extent of damage to corticospinal output to the impaired upper limb. Our work only shows a trend for a relationship between change in IHI following CCFES and extent of baseline motor severity (more reduced IHI in those with more severe impairment; Figure 4A). Therefore, we are not able to confirm the bimodal hypothesis likely because of limited subsample size and insufficient power. Importantly, however, we have built on the premise of the bimodal balance-recovery model, where we have shown that the adaptive role of the contralesional hemisphere may be invoked using bilateral CCFES across the moderate to severely impaired as well as the mildly impaired participants. Future studies should incorporate multiple sessions of CCFES treatment to better understand the relationship between the individual neurophysiological mechanisms and degree of recovery seen across a wide range of motor severity.
Another important finding is that ipsilesional output stayed relatively the same after CCFES but decreased after cNMES. Peripheral nerve stimulation has been shown to promote recovery and either increase or decrease corticospinal excitability in stroke volunteers. 40 Peripheral nerve stimulation has been applied via electrodes over the skin overlying peripheral nerves or via a mesh-glove for whole-hand electrical stimulation. These changes appear to be dependent on lesion location, stimulus intensity, frequency, and duration.40-43 In our study, we showed an interaction between our 2 conditions, even though stimulus intensity, frequency, and duration were the same. One possible reason may be central fatigue among participants as a result of the 1-hour therapy. Kotan et al 44 have observed a significant reduction in corticospinal excitability after muscle fatigue induced by combined NMES and voluntary contraction, but not with NMES alone. The authors suggest that their findings may be a result of an increase in excitability of inhibitory networks. Central fatigue has been recorded in many studies that show that maintaining submaximal muscle contraction for an extended period of time results in what is known as postexercise depression of output devoted to fatigued muscles.45,46 In fact, Knorr et al 8 has shown that neuromuscular-induced central fatigue in the chronic stroke population is more prevalent in the paretic limb when compared with the nonparetic limb. These results, combined with Kotan et al, inform our findings because we observed reduction in ipsilesional output following cNMES and control conditions but no such decrease after CCFES. One possible reason may be that CCFES being a bilateral condition induced disinhibition, a mechanism characteristic of synchronous bilateral movements,21-24 as evidenced by reduction in IHI after training. This disinhibition may have helped overcome fatigue-related diminution of ipsilesional output otherwise observed after other conditions, an interpretation that remains to be tested in future studies.
Still, it is intriguing that relative to cNMES, CCFES resulted in CCFES-enhanced ipsilesional output to the impaired upper limb when compared with cNMES. In fact, participants with mild motor impairment (UEFM > 50) in our sample demonstrated the greatest gains in ipsilesional output following CCFES (Figure 4B). These results suggest that bilateral CCFES may not align with the interhemispheric competition model and even the bimodal balance recovery model, because movement of the nonparetic limb (in conjunction with the paretic) resulted in increases in ipsilesional output in mildly impaired patients. We are unable to corroborate evidence for the more severely impaired given their absence of paretic MEPs.
Our findings may have broad implications for bilateral and unilateral movements in general. One reason bilateral CCFES may not completely align with the interhemispheric competition model or the bimodal balance recovery model is that both theories are based on principles of unilateral movement. Bilateral movements, however, invoke neurophysiological mechanisms that are unique compared to unilateral movements. Studies have shown that by symmetrically moving both limbs for a common purpose, both hemispheres become coupled.47,48 As a result of coupling, Mudie and Matyas 47 explain that the contralesional hemisphere may offer a “template” of motor network recruitment to the ipsilesional hemisphere, allowing the impaired upper limb to learn from the nonimpaired upper limb. In fact, studies with fMRI show that bilateral movements elicit unique and greater activation of bilateral primary sensorimotor, premotor, and supplementary motor cortices in comparison to unilateral movements and that these distinctions amplify with therapy. 49 However, an important caveat is that previous studies have demonstrated that stroke motor deficits are hemisphere specific and, thus, differ between nondominant versus dominant hemisphere lesion locations. For example, Schaefer et al 50 have shown that patients with left hemisphere lesions experience reduced modulation of motor acceleration amplitude, where participants with right hemisphere lesions experience greater final motor position errors. Therefore, the template provided by the contralesional hemisphere is likely hemisphere specific as well.
Coupling between hemispheres could involve mutual disinhibition of both hemispheres that is characteristic of bilateral movements.21-24 It is important to note, however, that coupling between hemispheres appears to be more apparent during synchronous bilateral movements than during asynchronous movements. For example, bilateral synchronous movements result in a decrease in intracortical inhibition within M1,15,16,51 enhanced activation and connectivity between hemispheres, 22 and as demonstrated in this study, a decrease in IHI between M1s. A similar phenomenon has not been shown to occur during bilateral asynchronous movements. 51 Release of inhibition during synchronous movements could have contributed to the enhanced output of the ipsilesional M1 for the mildly impaired. Furthermore, the release of inhibition could have enhanced spared pathways, such as the higher motor areas, for patients with greater corticomotor damage (ie, no MEP present) not investigated in this study.12,15,48,49,52,53 Thus, it is possible that synchronous somatosensory feedback in bilateral motion and a single set of motor commands linking bilateral movements may help upper limbs become functionally coupled and help both hemispheres release their inhibition on one another to allow transfer from the contralesional to the ipsilesional hemisphere regardless of motor severity. In addition to the advantage of bilateral movements for the ipsilesional hemisphere, bilateral movements could engage the adaptive role of the contralesional hemisphere, especially for patients with greater corticomotor damage. Whitall et al 49 showed that 18 one-hour sessions of bilateral therapy result in greater contralesional hemisphere activation, and the enhancement of the contralesional hemisphere was associated with improved arm function. In the present study, however, we were not able to find greater ipsilateral output to the paretic hand after CCFES when compared with cNMES (P = .13) as originally hypothesized; this may be the result of our small sample size and large distribution of motor impairment. Still, we do show that the ipsilateral output to the paretic upper limb is primed in patients with moderate to severe motor impairment, results that are in line with Schwerin et al 54 (Figure 4C). Future studies will benefit from a more systematic investigation of the ipsilateral motor pathways originating from the contralesional hemisphere in patients with moderate to severe motor impairment.
Limitations and Conclusion
First, we were unable to quantify the volitional finger extension effort of the paretic limb during CCFES and cNMES. Ideally, we would have been able to record EMG; however, the electrical stimulus artifact makes this a challenging outcome to record. Second, although a crossover design was a strength of the study, allowing robust within-subject comparisons, applying only 1 hour of therapy would not likely result in any functionally relevant or impairment-based outcomes and is more likely to induce fatigue. Without functional and impairment-related outcomes and an assessment of the longevity of the effects on the TMS outcomes, we can only speculate that the changes measured in this study may help explain improvements in outcomes such as hand dexterity following CCFES, as shown by Knutson et al. 19 Future studies should incorporate multiple sessions of treatment to better understand the relationship between the individual neurophysiological mechanisms and patient recovery.
In conclusion, we show that bilateral CCFES may help recruit the recovery potential of the contralesional motor cortices by reducing competitive inhibition. Furthermore, bilateral CCFES may support residual ipsilesional corticospinal output in those with less-extensive damage and may not align with the interhemispheric competition model and the bimodal balance recovery model. Future studies will expand on these findings by including multiple sessions of CCFES in order to make direct comparisons between the neurophysiological benefit and the capacity to maximize recovery.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: David A. Cunningham: reports no disclosures. Jayme Knutson: coinventor on US patent 8 165 685 assigned to Case Western Reserve University: System and Method for Therapeutic Neuromuscular Electrical Stimulation. Vishwanath Sankarasubramanian: reports no disclosures. Kelsey A. Potter-Baker: reports no disclosures. Andre G. Machado has the following conflict of interests to disclose: being a consultant of functional neuromodulation at St Jude; having distribution rights at Enspire, ATI, and Cardionomics; and having fellowship support from Medtronic. Ela Plow: reports no disclosures.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Institutes of Health [1K01HD069504 (EBP), R01HD092351 (JSK) & R01HD061363 (AGM)] and the American Heart Association [13BGIA17120055, 16GRNT27720019] awarded to EBP, and Clinical and Translational Science Collaborative [RPC2014-1067] to DAC.


