Abstract
Background. No single intervention restores the coordinated components of gait after stroke. Objective. The authors tested the multimodal Gait Training Protocol, with or without functional electrical stimulation (FES), to improve volitional walking (without FES) in patients with persistent (>6 months) dyscoordinated gait. Methods. A total of 53 subjects were stratified and randomly allocated to either FES with intramuscular (IM) electrodes (FES-IM) or No-FES. Both groups received 1.5-hour training sessions 4 times a week for 12 weeks of coordination exercises, body weight–supported treadmill training (BWSTT), and over-ground walking, provided with FES-IM or No-FES. The primary outcome was the Gait Assessment and Intervention Tool (G.A.I.T.) of coordinated movement components, with secondary measures, including manual muscle testing, isolated leg movements (Fugl-Meyer scale), 6-Minute Walk Test, and Locomotion/Mobility subscale of the Functional Independence Measure (FIM). Results. No baseline differences in subject characteristics and measures were found. The G.A.I.T. showed an additive advantage with FES-IM versus No-FES (parameter statistic 1.10; P = .045, 95% CI = 0.023-2.179) at the end of training. For both FES-IM and No-FES, a within-group, pre/posttreatment gain was present for all measures (P < .05), and a continued benefit from mid- to posttreatment (P < .05) was present. For FES-IM, recovered coordinated gait persisted at 6-month follow-up but not for No-FES. Conclusion. Improved gait coordination and function were produced by the multimodal Gait Training Protocol. FES-IM added significant gains that were maintained for 6 months after the completion of training.
Keywords
Many stroke survivors are unable to execute the precisely coordinated movements for the swing and stance phases of gait. 1,2 Recovery of gait coordination depends on sufficiently addressing the impairments underlying those gait deficits, such as weakness and impaired coordination. 3,4
Three rehabilitation strategies seem especially potentially useful for improving the therapeutic targets inherent in gait deficits. These 3 promising rehabilitation strategies include exercise to improve strength and coordination; 3 body weight–supported treadmill training (BWSTT) to practice stance phase hip, knee, and ankle control 5,6 and faster gait speed practice 7 and to mitigate fear of falling; 6 and over-ground gait training to integrate volitional control of all the components of gait. Examples of specific therapeutic targets include the following: isolated single joint movements such as ankle dorsiflexion while the knee is maintained in the extended position; out-of-synergy multijoint movements such as ankle dorsiflexion while extending the knee; muscle strength; stability and movement control of hip, knee, ankle joints during forward translational movement of the center of mass over the stance limb; and speed of execution of coordinated movements.
Considering the combined rehabilitation strategies of exercise, BWSTT, and over-ground gait training, we identified the additional need to provide practice of more normal muscle activation latencies and movement patterns than could otherwise be practiced. We identified functional electrical stimulation (FES) as a promising exercise-assist and gait training-assist method to test in a randomized trial of those with persistent gait deficits. 8 FES-assisted exercise has shown improved volitional muscle activation and reduced spasticity, improved out-of-synergy coordination, 9-11 and reduction of abnormal co-contraction. 12
We studied a gait training protocol (composed of 3 treatment aspects: exercise, BWSTT, and over-ground gait training), and compared intramuscular FES (FES-IM) with no FES. The goal was to restore the coordinated movement components of gait. The secondary investigative objectives were 2-fold: (a) functional response to treatment and (b) benefit of the gait training protocol, with and without FES-IM.
Methods
Participants
A total of 88 candidates were assessed for eligibility (Figure 1), and 53 qualified for randomization to 1 of 2 treatment groups according to accepted guidelines 13 (Figure 1). Inclusion criteria included onset >6 months; first stroke; inability to execute normal swing phase in the sagittal plane using hip, knee, and ankle flexion; hyperflexion or hyperextension of knee during stance; passive joint range of motion of hip, knee, and ankle equal to normal excursion needed for walking; and not participating in gait rehabilitation. Exclusion criteria included inability to follow 2-level commands; pacemaker; peripheral neuropathy; and debilitating illness (eg, cancer). In all, 27 did not meet the criteria, and 8 declined to participate after the screen (Figure 1). The 53 remaining candidates were accepted and enrolled. The study was approved by the Medical Center Institutional Review Board and a written informed consent process was conducted. Subjects were stratified 13 according to stroke severity using the Fugl-Meyer Lower Limb Scale (FM) with 0 to 19 as severe, 20 to 28 as moderate, and ≥29 as mild and excluded. Within this framework, 13 we randomly allocated subjects to either FES-IM or No-FES treatment. Randomization was conducted in the 3 following steps by 3 separate individuals: generate an unpredictable treatment allocation sequence; record enrollment date; allocate treatment to a given subject based on a cross-matching of enrollment date with the unpredictable treatment allocation sequence (open-list method). Subject screening, enrollment decision, and allocation to group were conducted by different individuals.
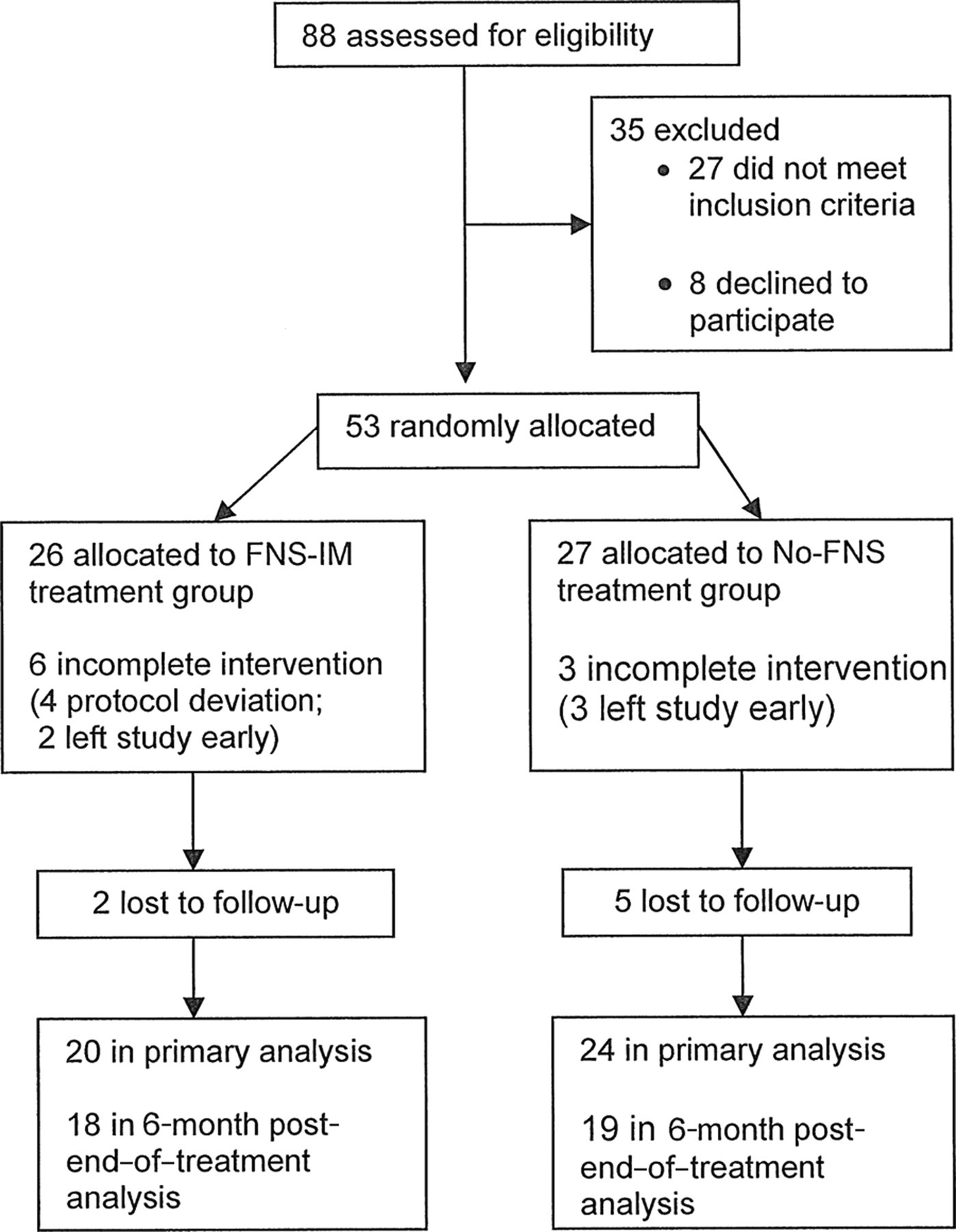
Study flowchart. Abbreviations: FNS, functional neuromuscular stimulation; IM, intramuscular.
Treatment Methods
Both groups were treated for 1.5 hours per session, 4 sessions per week, for 12 weeks. All received (a) strengthening and coordination exercise, (b) BWSTT, and (c) over-ground gait training. The treatment intensity and duration were based on prior work. 6,10 Exercise included practice of coordinated joint movements underlying gait components. 14 BWSTT was conducted using the BIODEX500 (Biodex, Shirley, New York), beginning with 30% BWS, and decreasing to 0%, according to ability to maintain normal, neutral alignment of torso and stance limb. Walking training speed was increased up to 0.894 m/s, as tolerated. Over-ground gait training included training in torso, pelvic, hip, knee, and ankle position control during loading and weight bearing; swing hip, knee, and ankle flexion; and terminal swing knee extension/ankle dorsiflexion. Home exercises emphasized coordination exercises for 1 h/d.
Exercise, BWSTT, over-ground gait training, and the home program were identical for both groups, but FES-IM was used during all treatments in one arm of the trial, including home program. Template FES patterns from prior work 9,10 were individualized for each subject. Custom software 15 was used to create the individualized FES patterns. This program enabled a stimulation pattern using up to 8 muscles in multiple combinations and timing variations for the precisely coordinated movements required for motor learning of gait components. 16,17
The FES-IM system included a V-40 stimulator worn on a belt and custom Vortex software 15 on a personal computer. Each customized pattern was downloaded to the subject’s own stimulator for gait practice. A given exercise was selected from a menu by a 4-button hand switch worn as a finger ring or attached by Velcro to a cane or exercise bar. The electrodes were constructed of a stainless steel wire configured in a double helical coil for flexibility, with a polypropylene core for durability. 18 These electrodes have a good safety and performance record. 19 Electrodes were placed at the motor point, under monitored anesthesia care. The lead wires exited the skin (percutaneous) at a common site on the anterior proximal thigh and were crimped into a connector, which communicated via ribbon cable to the external stimulator, during FES-assist exercise and gait training. When the FES-IM system was not in use, the external portion of the lead wires and their connector were taped to the skin. Electrodes were removed at the end of the study in a 30-minute outpatient procedure.
We implanted electrodes in 8 muscles from among the following: tibialis anterior, peroneus longus, gastrocnemius lateral head, biceps femoris short head, semimembranosis, semitendenosis, vastus lateralis, and gluteus medius, which remained in place throughout the treatment protocol. For implantation of a given muscle, we used the following criteria: movement capability either less than or equal to movement against gravity through only partial normal range and no ability to take resistance 20 and inability to perform coordinated movements according to the FM.
The electrical stimulation parameters included amplitude, 4 to 20 mA; pulse width, 1 to 150 µs; and frequency, 15 to 50 Hz, within comfort. FES-IM was used to treat pelvic stability during stance phase, knee extension at loading, ankle dorsiflexion during swing, knee flexion at toe-off, and knee flexion and extension during swing, 6,9,10 and swing and stance phase components during combination BWSTT + FES-IM. Because the purpose was restoration of volitional gait control, we incrementally reduced the level of FES, as volitional control improved.
Measures
The primary outcome measurement was the Gait Assessment and Intervention Tool (G.A.I.T.). 21 The G.A.I.T. measure items pertain to the spatial coordinated movement components of gait (movement excursion), including upper extremity and trunk, pelvis, hip, knee, and ankle movements at specified temporal events of stance and swing phases or across the time of subphases. Deviations from normal are scored for each movement that is assessed within the gait cycle, for example, pelvic position control in the coronal plane during stance; mid-stance knee control; and swing phase hip flexion. Intrarater reliability of the G.A.I.T. was good 22 (intraclass correration [ICC] = .98; P = .0001; 95% confidence interval [CI] = 0.95-0.99). Interrater reliability of the G.A.I.T. was good (ICC = .83; P = .007; 95% CI = 0.32-0.96), including between an experienced and an inexperienced clinician (ICC = .996; P = .0001; 95% CI = 0.986-0.999). The G.A.I.T. items measured our targeted interventions. The G.A.I.T. was scored using a video record of gait.
In secondary analysis, we investigated measures of impairment and function. We acquired data at pre-/mid-/posttreatment and 6 months after the end of treatment (follow-up). Secondary outcomes included leg muscle strength (manual muscle testing [MMT] on a 5-point scale, 20 isolated leg joint movement coordination (FM)), and the 6-Minute Walking Distance Test (6MWT). Subjects used usual assistive device (constant at pre-/posttreatment) and a 30.5-m walkway. Functional Independence Measure subscales for Locomotion and Mobility (FIM-L&M) 23 were acquired. A questionnaire was used to assess subjective response to the comfort of the system. For all outcome measures, volitional movement was tested (no FES and no BWSTT), and was assessed by a blinded examiner.
Data Analysis
The Mann–Whitney test was used to test baseline equivalency between the 2 groups. Group difference at follow-up, according to the G.A.I.T. measure, was investigated using the plum ordinal regression model, with either posttreatment or followup score as dependent variable, group as independent variable of interest, and pretreatment score as covariate. We generated descriptive statistics to describe the distribution of G.A.I.T. gain scores and the number of subjects having gains in individual items. The Wilcoxon signed rank test was used in secondary analysis to test within-group, pre-/posttreatment effect, and the Bonferroni correction was applied to control for multiple comparisons for impairment measures. Effect size for pre-/posttreatment was calculated using Cohen d. The Wilcoxon signed rank test was used in exploratory analysis to investigate mid-/posttreatment benefit (continued benefit of the last half of treatment for each group), as well as maintenance of coordinated gait components for each treatment group from posttreatment to follow-up.
Results
No baseline differences were found between the 2 treatment groups (Tables 1 and 2), including age (P = .80), time since stroke (P = .897, with FES-IM mean 2.8 [±3.3 years] and No-FES mean 2.6 [±3.0 years]).
Participant Characteristics
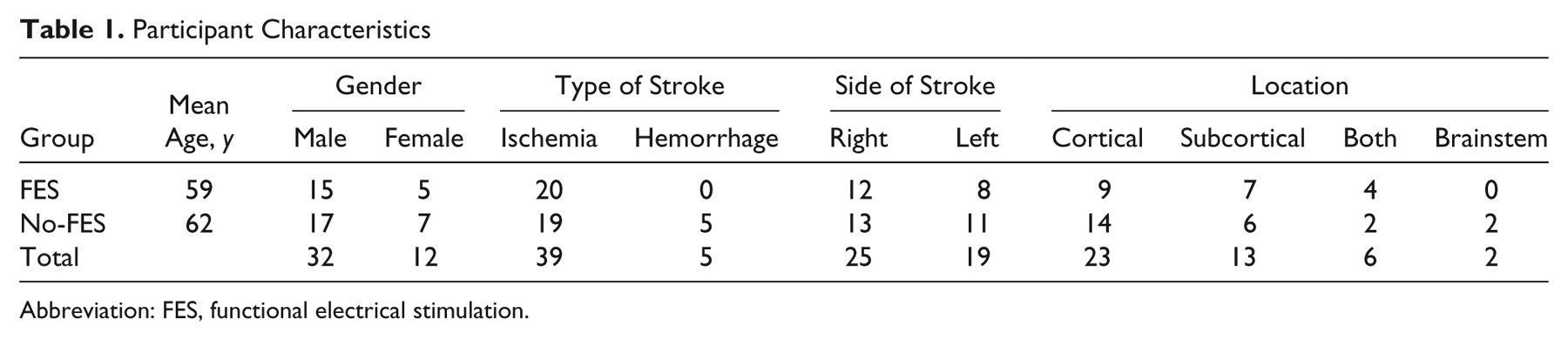
Abbreviation: FES, functional electrical stimulation.
Pretreatment Group Means/Medians (SD, IQR)
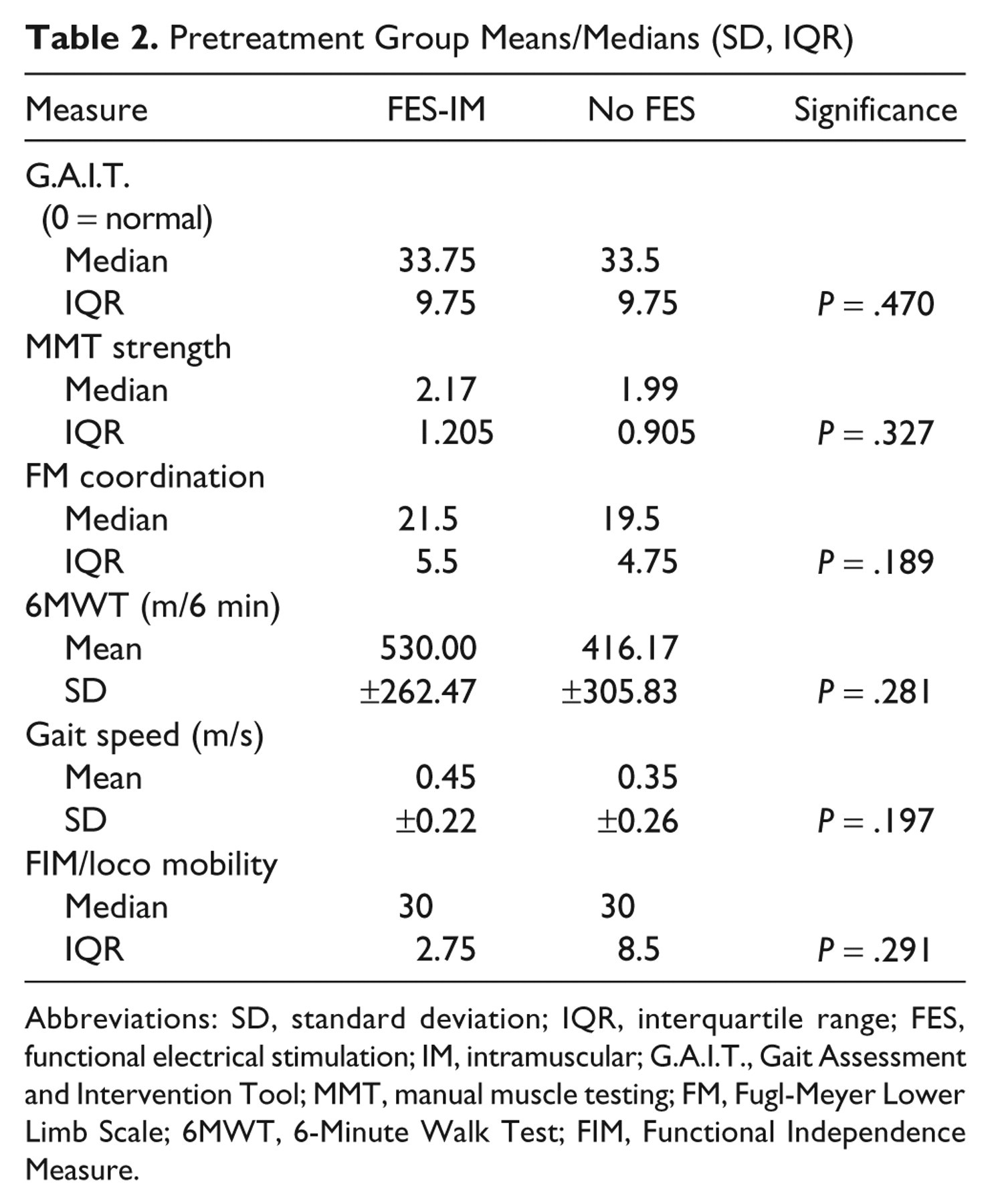
Abbreviations: SD, standard deviation; IQR, interquartile range; FES, functional electrical stimulation; IM, intramuscular; G.A.I.T., Gait Assessment and Intervention Tool; MMT, manual muscle testing; FM, Fugl-Meyer Lower Limb Scale; 6MWT, 6-Minute Walk Test; FIM, Functional Independence Measure.
For the primary outcome, FES-IM had a significant additive effect (G.A.I.T. parameter statistic = 1.10, P = .045, 95% CI = 0.023-2.18; Figure 2A). Figure 3 shows an example of recovery of hip, knee, and ankle flexion at mid-swing phase. A video document is available as supplementary file A on the journal Web site (http://nnr.sagepub.com/supplemental), showing an example of dyscoordinated gait pattern prior to treatment and recovery of coordinated movements of gait after treatment. Exploratory analysis showed that 10/20 (50%) FES-IM treatment subjects improved ≥10 points on the G.A.I.T. whereas only 5/24 (20.8%) No-FES subjects improved ≥10 points (P = .043).
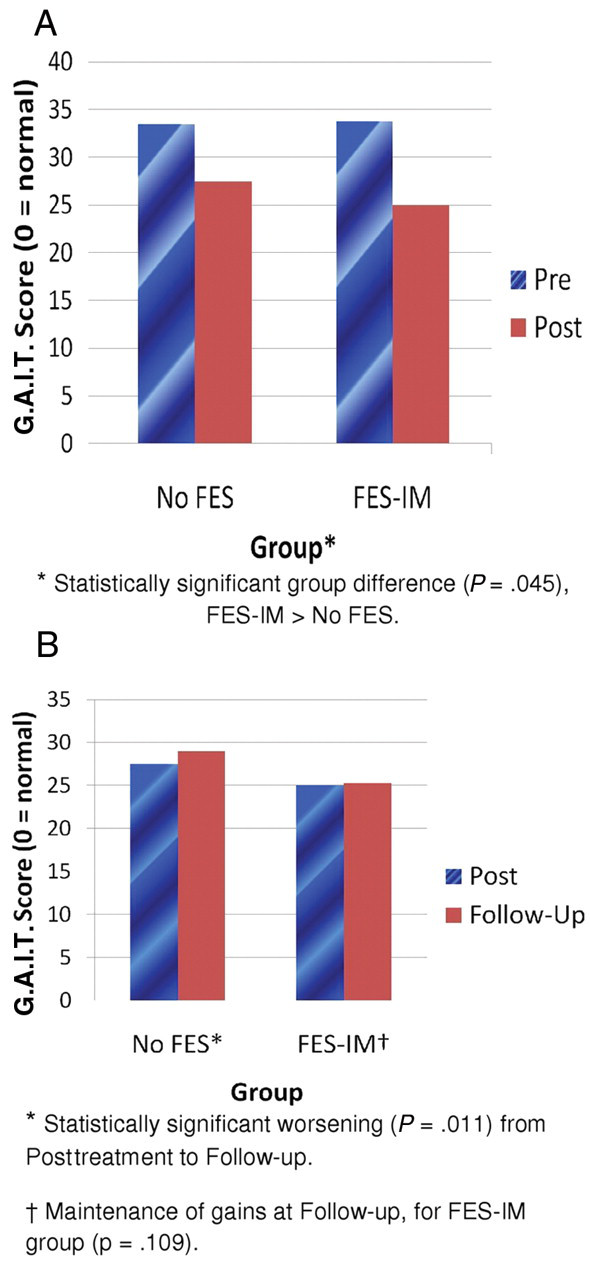
(A) Significant additive advantage of FES-IM for recovery of coordinated gait components and (B) follow-up worsening in the No-FES group and maintenance of G.A.I.T. gains in the FES-IM group. Abbreviations: FES, functional electrical stimulation; IM, intramuscular; G.A.I.T., Gait Assessment and Intervention Tool.

(A) Pretreatment absent hip, knee, and ankle flexion at mid swing. (B) Posttreatment recovery of hip, knee, and ankle flexion at mid-swing.
Descriptive Statistics
Figure 4 shows the distribution of gain scores. The FES-IM group had a range of G.A.I.T. gain scores from 0 to 17, whereas the No-FES group had worsening or gain scores ranging from −1 to 12. In the lower performing range, in the No-FES group, 45% had change scores ≤5 points; the FES-IM group had only 25% of subjects with gains ≤5 points. There were 3 subjects in the No-FES group who had either no gain or a worsening; in the FES-IM group, there was 1 subject who had no change (none had a worsening).
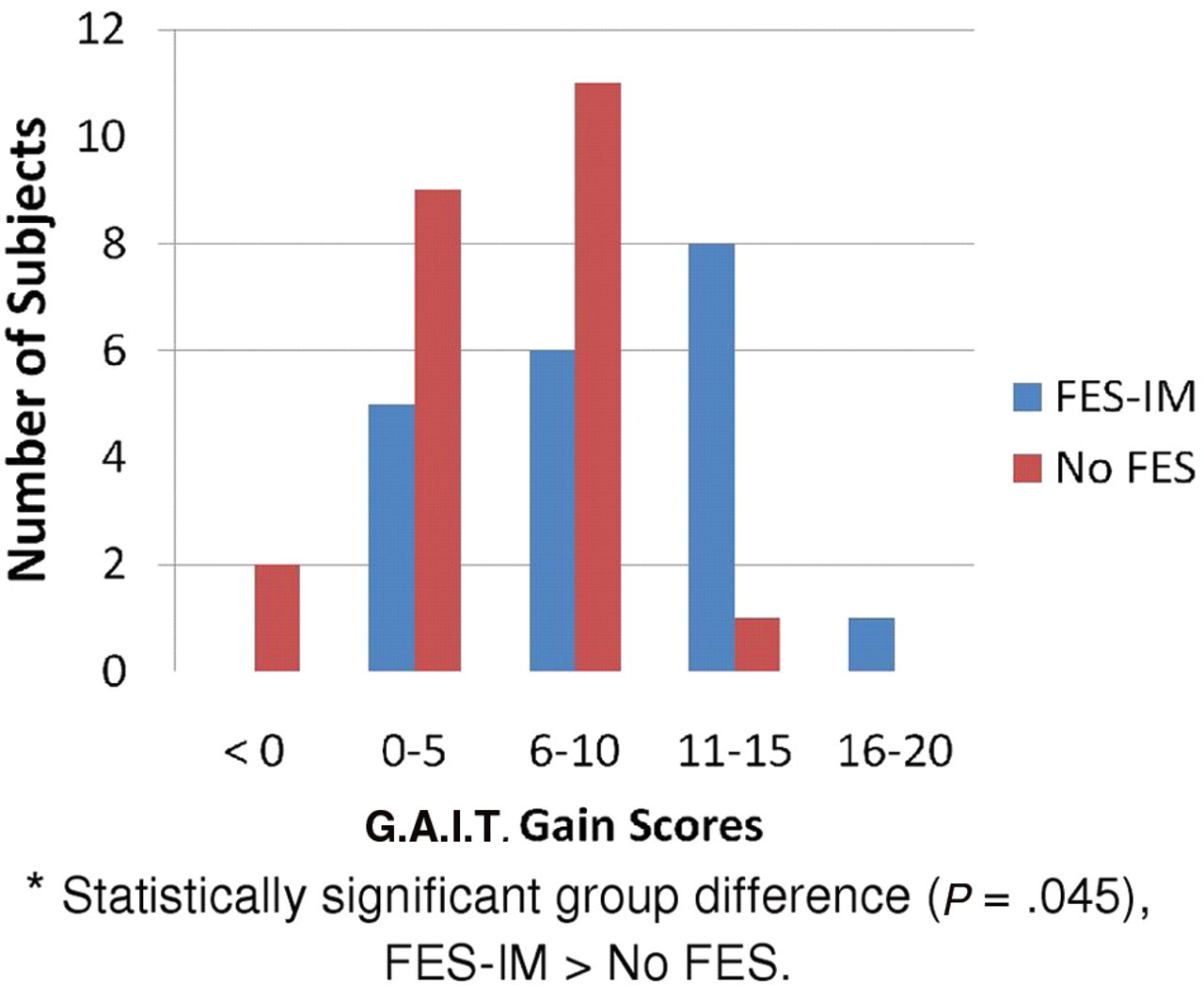
Distribution of G.A.I.T. gain scores for FES-IM and No-FES groups. Abbreviations: FES, functional electrical stimulation; IM, intramuscular; G.A.I.T., Gait Assessment and Intervention Tool.
We found that 40% more subjects in the FES-IM versus the No-FES group had recovery in the following gait components: stance phase trunk postural control in the sagittal plane, stance phase trunk postural control in the coronal plane, weight shift during stance phase, late stance phase hip extension, stance phase thigh position without internal/external rotation, late stance plantarflexion from heel rise to toe-off, swing phase hip flexion, swing phase thigh position, elbow flexion, and shoulder position.
As a method for placing perspective on the clinical relevance of the G.A.I.T. measure results, we used the value of 0.16 m/s gait speed, which has shown functional significance in that this minimal clinically important difference (MCID) of 0.16 m/s was associated with a gain in the functional measure, the Rankin Scale. 24 The FES-IM group improved by 0.16 m/s, a MCID. 24 Of possible clinical importance, the G.A.I.T. score and the MCID of gait speed were correlated at r = .61 (P = .0001; Pearson correlation).
Secondary Analyses
For both FES-IM and No-FES, there was a significant pre- to posttreatment gain within each group for all measures, with generally high effect sizes (Table 3, panels A and B). In addition, from mid- to posttreatment, for both FES-IM and No-FES, we found continued significant improvement (Table 4, panels A and B).
Significant Pre-/Posttreatment Gains for Both Groups
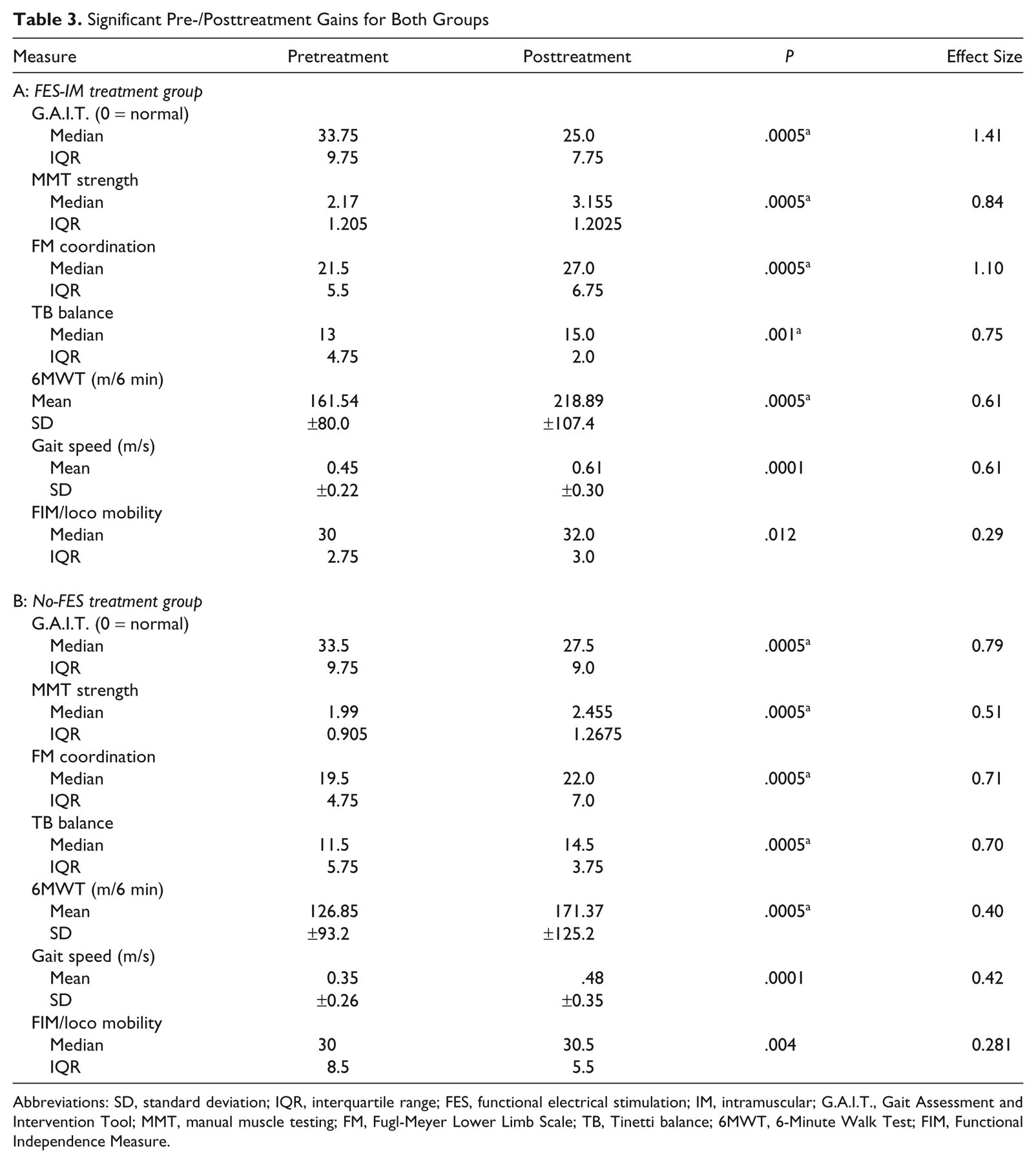
Abbreviations: SD, standard deviation; IQR, interquartile range; FES, functional electrical stimulation; IM, intramuscular; G.A.I.T., Gait Assessment and Intervention Tool; MMT, manual muscle testing; FM, Fugl-Meyer Lower Limb Scale; TB, Tinetti balance; 6MWT, 6-Minute Walk Test; FIM, Functional Independence Measure.
Bonferroni corrected P values.
Change From Mid- to Posttreatment
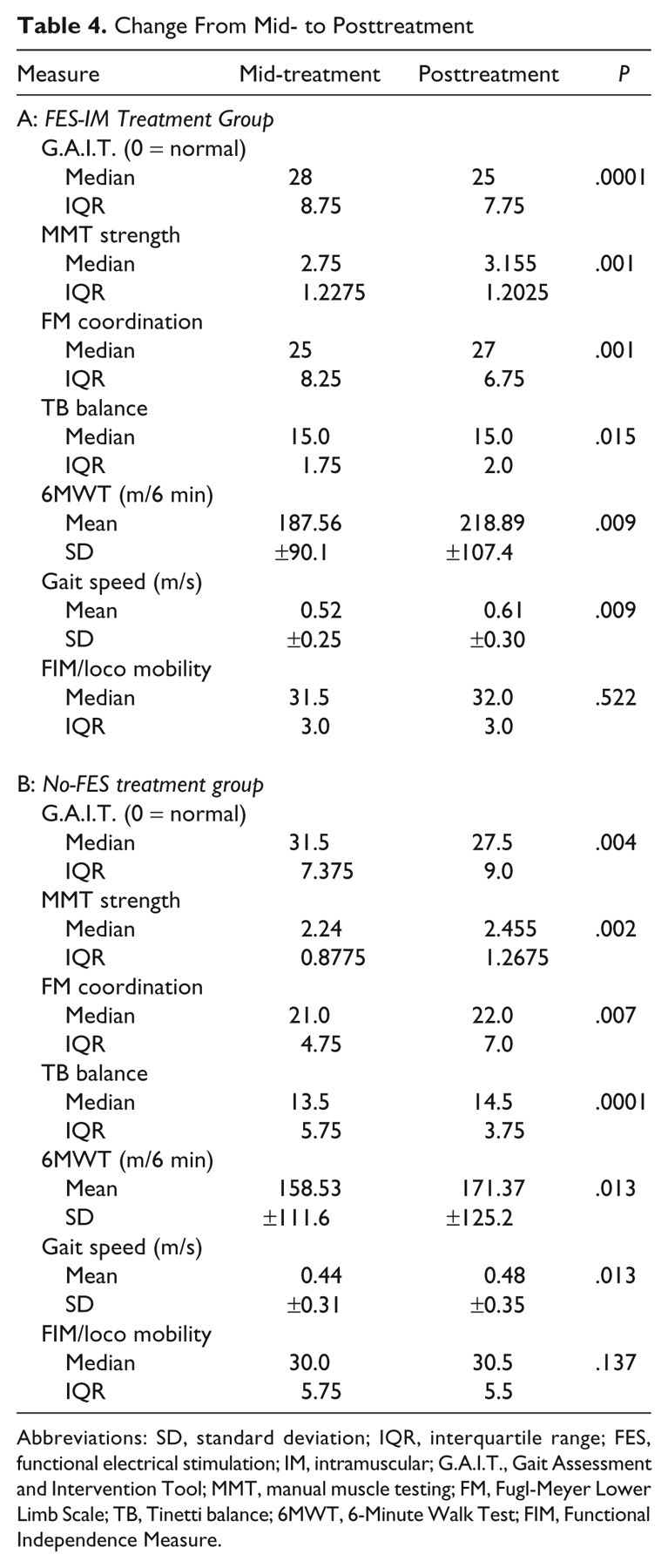
Abbreviations: SD, standard deviation; IQR, interquartile range; FES, functional electrical stimulation; IM, intramuscular; G.A.I.T., Gait Assessment and Intervention Tool; MMT, manual muscle testing; FM, Fugl-Meyer Lower Limb Scale; TB, Tinetti balance; 6MWT, 6-Minute Walk Test; FIM, Functional Independence Measure.
From posttreatment to follow-up, for the FES-IM group, coordinated gait components were maintained, but no additional recovery was observed (G.A.I.T. P = .109). In contrast, the No-FES group worsened at follow-up, according to the G.A.I.T. (P = .011; Figure 2B; Table 5). For the FES-IM group, there was a median pretreatment G.A.I.T. score of 33.75 (interquartile range [IQR] = 9.75) and a follow-up median score of 25.25 (IQR = 14.5). For the No-FES group, there was a pretreatment median G.A.I.T. score of 33.5 (IQR = 9.75) and a follow-up G.A.I.T. score of 29 (IQR = 9.5). At follow-up, there was an additive advantage of FES-IM versus the No-FES (parameter statistic = 1.896, P = .003, 95% CI = 0.648, 3.145).
Follow-Up Maintenance for FES-IM Group, According to the G.A.I.T. Measure
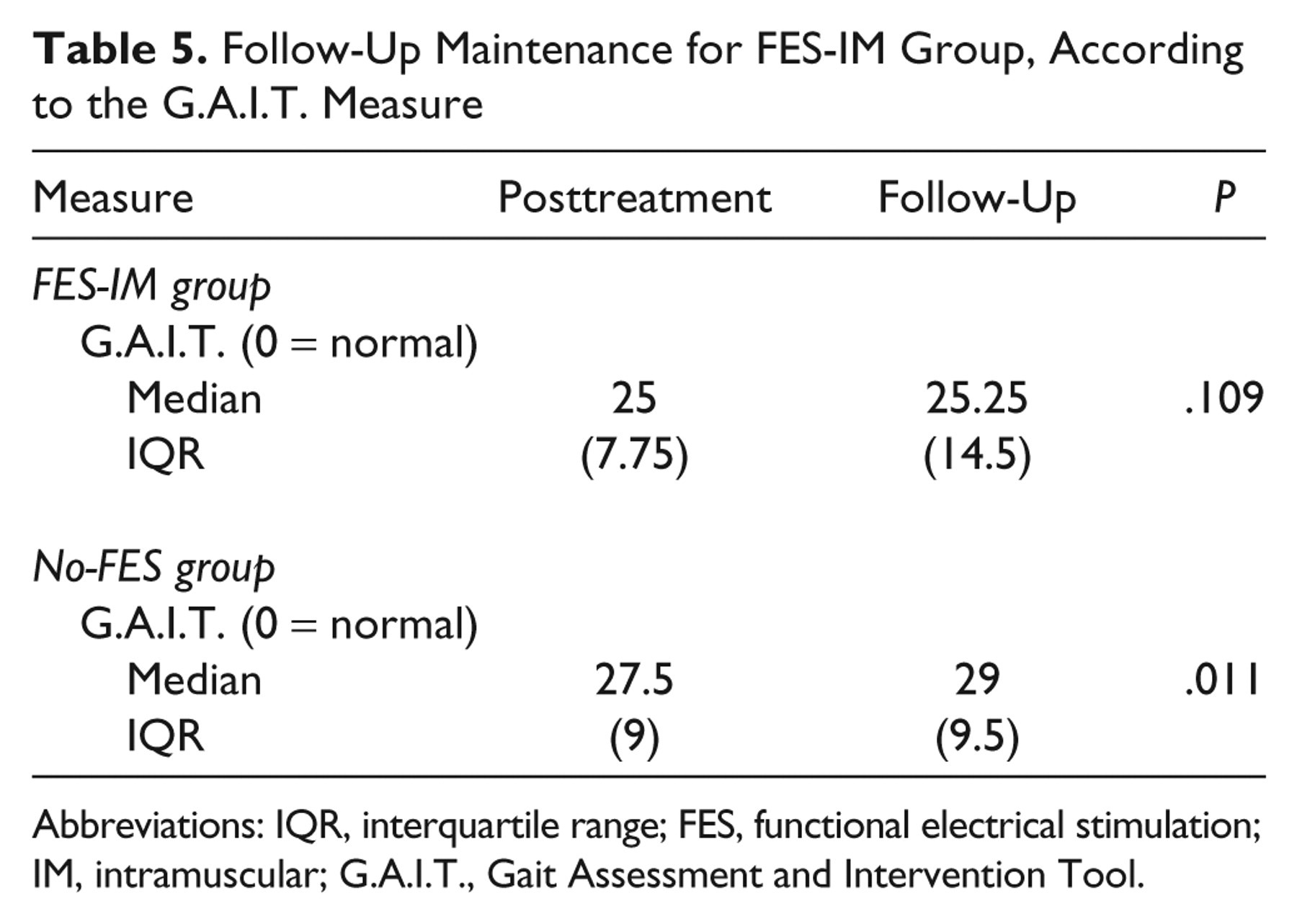
Abbreviations: IQR, interquartile range; FES, functional electrical stimulation; IM, intramuscular; G.A.I.T., Gait Assessment and Intervention Tool.
Adverse Events
After surgical electrode placement under monitored anesthesia care, subjects reported no discomfort or a discomfort level that accompanies a bruise. Discomfort resolved within 48 hours. Expected adverse events occurred during the months of use, with erythema near electrode lead exit site (18/161 electrodes) that resolved with warm compress (10) or prophylactic oral antibiotics (8). No infections occurred. No adverse events were associated with placement or removal of electrodes. When the system was deactivated and during stimulation by the FES-IM system, no subject reported discomfort.
The 44 subjects who completed the study did not significantly differ from the 53 who started the study, according to the following baseline characteristics: age, P = .765; time since stroke, P = .728; and stroke severity P = .965). Social issues resulted in the incomplete participation (such as family moving out of town), except for 2 who had back pain prior to study enrollment (but failed to report it at enrollment) and one who developed a Baker cyst in the uninvolved limb.
Discussion
Gait Training Protocol With FES-IM
Results showed that FES-IM provided a significant additive advantage versus an otherwise comparable comprehensive gait training protocol without FES, with regard to coordinated movement components of gait. Several advantages of FES-IM gait training could have contributed to produce this result. After stroke, many suffer from inability to volitionally activate muscles, resulting in inactivity and weakness. The FES-IM system induced muscle contractions that were not possible under volitional control alone, thereby assisting to strengthen muscle. A second advantage was that FES-IM assisted the training of coordination. Strength is necessary, but not sufficient, to produce recovery of voluntary control of coordinated movements. Indeed, recovery of coordinated movements depends on recovery of central nervous system motor control function, sufficient to control simultaneous and sequential multiple joint movement latencies, joint torques, speed of movements, and extent of movement excursions. After stroke, practice of more progressively normal complex gait components is difficult, because mass limb flexion or extension reflex patterns of movement interfere with the intended movement. Increasingly stronger effort to produce a coordinated movement may result in abnormal muscle activations and dyscoordinated movements. FES-assisted movement can mitigate the abnormally strong (and counterproductive) effort to control a simple movement. The FES may have elicited not only an efferent but also an afferent feedback effect that could have contributed to inducing a more coordinated pattern. In addition, the efficiency of a stiff-legged gait is only 76% of normal gait; 25 in that regard, FES-IM enabled coordination practice and a less demanding walking pattern.
FES-IM was incorporated into gait training in a purposeful manner. Our protocol for both groups uses the paradigm of test–treat–test within a given session, as well as a retest procedure at the beginning of the subsequent session. These clinical practice procedures provide information regarding the subject’s need for intervention at a specific level of difficulty, the immediate response to intervention (within the session), and the carry-over effect between sessions. We applied this to each gait component within the session. The subject’s ability, to demonstrate carry-over between the 2 sessions, served as the criterion for progression to the next level of difficulty for practice of a given gait component. This information was not formally recorded but was used to progress treatment. The FES-IM system served to assist motor practice, specifically in providing practice of more normal movement patterns than would have been possible with volitional effort alone. We observed this FES-assisted training advantage in all of the FES-IM group subjects; 26% to 100% of each subject’s muscle groups (2 to 8 muscle groups) and associated movement patterns exhibited this practice advantage using FES-IM. For some muscles, the FES-IM system did not induce a muscle contraction beyond volitional capacity. Rather, in those cases, the FES-IM system was used to provide cues for the practice of more normal timing of muscle activation. FES-IM thus provided more normal, productive practice of coordinated single and multiple joint movements than otherwise possible. As we found in our prior work, 10,11 both the FES-assisted and volitional movements improved over time. The most typical pattern of recovery was that FES-assisted movement recovered faster, with volitionally controlled movement lagging behind. 11
Both groups had statistically significant, within-group gains in all measures, indicating that the gait training protocol either with or without FES had a positive impact on gait and function for those in the chronic phase after stroke. The majority of publications regarding gait recovery in the chronic phase after stroke describe gait recovery in terms of gait speed or temporal measures of gait, such as stride time, gait speed, and symmetry of stance and swing phase latencies. 26-29 However, gait coordination can be considered a “higher order property” of the locomotor system,30(p213) “having both spatial and temporal components.” 30(p213) In that case, temporal measures (gait speed and symmetry) are incomplete descriptors of coordinated gait, and faster gait speed does not necessarily imply a more coordinated gait pattern. 30 The G.A.I.T. measure assesses spatial gait components either at specified time points in the gait cycle, or across the time domain of a subphase of the cycle. To our knowledge, few prior studies reveal recovery of coordinated gait components. The current study provides evidence that the multimodal gait training protocol can produce a statistically significant gain in coordinated gait components with or without FES-IM. The separate contribution of each aspect of the comprehensive treatment (exercise, BWSTT, over-ground gait training) was not directly measured.
The protocol included 48 visits of 1.5 hours over 12 weeks. With some exceptions (eg, treadmill training 28 ), this is a greater intensity and longer duration than has been provided in many other studies. This intensity and duration afforded the opportunity to use a finely incrementalized approach to coordination retraining of the gait pattern. These advantages may have allowed the gait training protocol to produce a sufficiently robust improvement in coordinated gait so that the 6MWT significantly improved, as well as the functional measure of FIM Locomotion and Mobility. These findings are important because this intensity is not currently being delivered in postacute rehabilitation. Both FES-IM and No-FES showed within-group, continued improvement in gait coordination from the mid-treatment to posttreatment. This continued improvement during the last half of the protocol suggests that still longer treatment duration, beyond the 12 weeks provided in the current study, may show additional gains.
For the FES-IM group, during the time interval between posttreatment and follow-up, there was no significant further improvement in the G.A.I.T. score, suggesting that for most subjects, no additional recovery would occur without additional gait training. During the follow-up period, in the absence of the gait training practice, coordinated gait components worsened for the No-FES group. This difference in follow-up maintenance could have occurred because the FES-IM group had a greater gain in coordination at posttreatment, which may have resulted in their continued use of relatively more normal coordination during walking.
Limitations
FES-IM is an invasive procedure that uses a research stimulation system, which is not yet clinically available. We used this system because it was the only available system that provides an 8-channel system that is portable and programmable, and easy to don. The cost of implementing the FES-IM gait training protocol in clinical practice would include some costs beyond the No-FES protocol, including the outpatient surgical procedure (4 hours) and the FES technology. Others have used surface FES in conjunction with BWSTT 31 and with an electromechanical gait trainer. 32 We found, however, a number of advantages of FES-IM in comparison with surface FES, including greater specificity in muscle stimulation, especially in deeper muscles; short setup time (8 surface electrode pairs may require 40 minutes to set up); and more comfortable stimulation at higher levels of stimulation, because of bypassing the cutaneous pain fibers. As surface FES technology improves, surface FES may prove to be a more clinically viable alternative.
Conclusions
In the chronic phase after stroke with moderate to severe impairment in the coordination of gait components, function can improve in response to a multimodal gait training protocol, and more so with the addition of FES-IM. At least 12 weeks of gait training was necessary.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by the Department of Veterans Affairs, Office of Rehabilitation Research and Development, Grant Nos. B2226R, A3102R, B5080S.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
