Abstract
Background. Clinical trials have demonstrated some benefits of electromyogram-triggered/controlled neuromuscular electrical stimulation (EMG-NMES) on motor recovery of upper limb (UL) function in patients with stroke. However, EMG-NMES use in clinical practice is limited due to a lack of evidence supporting its effectiveness. Objective. To perform a systematic review and meta-analysis to determine the effects of EMG-NMES on stroke UL recovery based on each of the International Classification of Functioning, Disability, and Health (ICF) domains. Methods. Database searches identified clinical trials comparing the effect of EMG-NMES versus no treatment or another treatment on stroke upper extremity motor recovery. A meta-analysis was done for outcomes at each ICF domain (Body Structure and Function, Activity and Participation) at posttest (short-term) and follow-up periods. Subgroup analyses were conducted based on stroke chronicity (acute/subacute, chronic phases). Sensitivity analysis was done by removing studies rated as poor or fair quality (PEDro score <6). Results. Twenty-six studies (782 patients) met the inclusion criteria. Fifty percent of them were considered to be of high quality. The meta-analysis showed that EMG-NMES has a robust short-term effect on improving UL motor impairment in the Body Structure and Function domain. No evidence was found in favor of EMG-NMES for the Activity and Participation domain. EMG-NMES had a stronger effect for each ICF domain in chronic (≥3 months) compared to acute/subacute phases. Conclusion. EMG-NMES is effective in the short term in improving UL impairment in individuals with chronic stroke.
Keywords
Introduction
Upper limb (UL) hemiparesis is one of the most common sequelae following stroke, which significantly affects performance of activities of daily living and quality of life. 1 Neuromuscular electrical stimulation (NMES) is recommended in clinical practice for poststroke UL rehabilitation. 2 NMES is a technique that produces contraction of innervated muscle by applying external electrical current transcutaneously. It can be applied with or without patient participation. The term “cyclic NMES” is used when patient participation is passive, that is, no voluntary effort is required to assist the muscle contraction. In another type of NMES, called “EMG-NMES,” electrical stimulation of a target muscle is directly controlled or triggered by volitional EMG (electromyogram) activity from the same or a different muscle.3,4
According to a recent review, several clinical trials have demonstrated that EMG-NMES can strengthen muscles, reduce spasticity, increase range of motion, and reorganize neural circuits after stroke. 4 However, there is contradictory evidence from 3 previous systematic reviews about the effects of EMG-NMES on motor recovery of arm and/or hand functions in patients with stroke.5-7 In a systematic review of 5 studies, Bolton et al 5 found a large mean effect size, indicating a positive influence of EMG-NMES on reducing arm/hand impairment and improving functional outcomes. In contrast, Van Peppen et al 6 reviewed 13 studies and found no evidence to support the use of EMG-NMES in paretic UL rehabilitation. Similarly, Meilink et al 7 found no differences between the effects of EMG-NMES and usual care on arm/wrist function.
Considering the large number of trials published since the last review, 7 the aim of this review was to provide an updated analysis and clinical perspective of the comparative effectiveness of EMG-NMES for wrist and hand rehabilitation in poststroke patients. The novelty of our approach is the focus on the effect of EMG-NMES on UL recovery based on each of the International Classification of Functioning, Disability, and Health (ICF) domains. 8
Methods
Literature Search and Selection Criteria
The systematic review and meta-analysis were performed by KMS and NNG according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement guidelines. 9 An extensive literature search was conducted using 10 databases available through the McGill University library system: Allied and Complementary Medicine Database (AMED), Cumulative Index to Nursing and Allied Health Literature (CINAHL), Cochrane Database of Systematic Reviews, Database of Abstracts of Reviews of Effects (DARE), Excerpta Medica Database (EMBASE), Medical Literature Analysis and Retrieval System Online (MEDLINE), Physiotherapy Evidence Database (PEDro), Psychological Information Database (PsycINFO), Disability and Rehabilitation Literature Database (REHABDATA), and Occupational Therapy Systematic Evaluation of Evidence (OTseeker). Databases were searched from their inceptions until mid-February 2018.
Additional studies were identified by examining reference lists of all relevant articles and previous reviews. The search strategy included combining keywords (free text words) and, if applicable, corresponding controlled terms based on the chosen database (eg, Medical Subject Headings [MeSH]). The “OR” and “AND” Boolean Operators were used to combine terms. MeSH terms were “exploded” for the search. Terms and search strategies used are shown in Table 1. Only articles in English regardless of year of publication were considered.
The Terms and Search Strategy Used in the Study a .
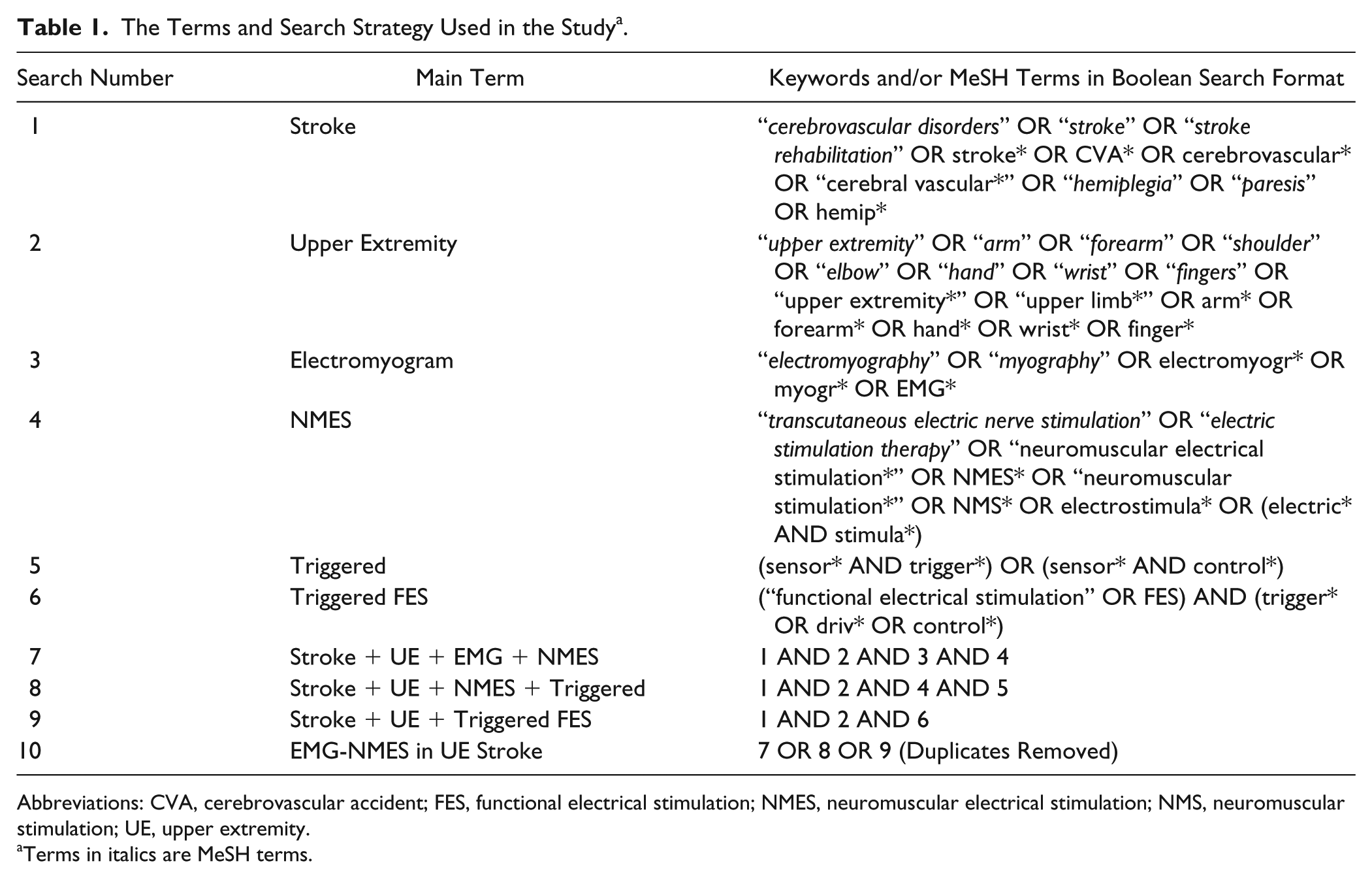
Abbreviations: CVA, cerebrovascular accident; FES, functional electrical stimulation; NMES, neuromuscular electrical stimulation; NMS, neuromuscular stimulation; UE, upper extremity.
Terms in italics are MeSH terms.
Following the searches, the same 2 reviewers independently screened all abstracts and titles of studies and ranked each trial as “included” or “excluded” according to the selection criteria below. If the reviewers were unsure whether to include or exclude a study, a final consensus decision was made by both reviewers after reading the full-text version.
Only randomized controlled trials were included. Quasi-randomly controlled and crossover trials with or without blinding, case reports, case series, and uncontrolled pre-post studies were excluded. Since the main principle of EMG-triggered NMES is that a person has to produce EMG activity in the affected muscle to receive the stimulation, contralaterally controlled EMG-NMES studies were excluded. We included studies conducted on adults (>18 years old) with a clinical diagnosis of either ischemic or hemorrhagic stroke regardless of their initial level of impairment, at any time after stroke. EMG-NMES for wrist/finger extension regardless of duration of therapy were considered. Trials in which all groups received EMG-NMES or which used implanted electrodes for stimulation were excluded.
Studies were included if the control group received either no treatment, any kind of physical or occupational therapy, placebo, or different types of electrostimulation (eg, transcutaneous electrical stimulation or cyclic NMES). Studies with concomitant therapies (eg, physical or occupational therapy) in both groups (EMG-NMES and control) were included.
Primary and Secondary Outcomes
Results were made more meaningful to clinical practice by using the ICF conceptual framework of Body Structure and Function, and Activity and Participation, 8 to classify outcome measures used in the included studies according to Salter et al. 10 The primary analyses focused on Body Structure and Function outcomes and the secondary analyses focused on Activity and Participation outcomes (see Supplementary Table).
Data Extraction
Data from each trial were extracted by KMS, summarized in tables, and independently checked by a second reviewer (DP). Data collected were the following: study design, participant characteristics (age, time since stroke, initial impairment level), intervention/control protocol details (EMG-NMES parameters; stimulated muscles, number of sessions, and training hours), outcome measures, measurement time points, and main results. The phase of stroke was categorized as acute (<3 weeks), subacute (3 weeks to 3 months), and chronic (≥3 months).
Stroke severity was categorized as severe (initial Fugl-Meyer Assessment of the Upper Extremity [FMA-UE] 11 score <30), moderate (initial FMA-UE score between 30 and 50), and mild (initial FMA-UE score >50). 12
Methodological Quality Assessment
PEDro scores (www.pedro.org.au) 13 were used to determine the methodological quality of each study. When scores were not available on the PEDro website, 2 reviewers (KMS and MABP) independently assessed the methodological quality using the PEDro-10 item system (random allocation, allocation concealment, discrepancies between groups at baseline, blinding of subjects, therapist and assessors, dropouts, incomplete outcome data, selective outcome reporting, and use of a measure of variability), each requiring a dichotomous response (yes = 1 or no = 0). The final study quality score was determined by summing scores of all items for a maximal score of 10. Studies with PEDro scores of 6 to 10, 4 to 5, and ≤3 were categorized as “high,” “fair,” and “poor,” respectively. Disagreements in PEDro scores between reviewers were resolved by a third reviewer (DP).
Data Analysis
Included studies were pooled in a meta-analysis using Review Manager software (RevMan 5.3, Copenhagen). When the original raw data (mean and standard deviation [SD]/standard error of the mean [SEM] or median and interquartile range) were not provided, numerical data were extracted from graphs or figures using a digitizing computer program (WebPlotDigitizer 14 ). Studies that did not provide sufficient information or graphs for data extraction were excluded from the meta-analysis but still included for the descriptive review. Attempts to obtain missing information by contacting authors were unsuccessful.
To avoid entering several comparisons into the meta-analysis from studies with multiple EMG-NMES arms, groups were combined as recommended by the Cochrane Handbook for Systematic Review of Interventions (CHSRI), Section 16.5.1. 15 For trials with more than one control group, only one of the control groups was selected to compare to the EMG-NMES group.
Means and SDs of each group (EMG-NMES and control) for each included study were analyzed separately at posttest (short-term) and follow-up. When only median values were reported, these were considered to be equal to mean values when data were normally distributed. To obtain the associated SD, interquartile ranges were divided by 1.35 as suggested by CHSRI, Section 7.7.3.5. 15
Since improvements in different outcome measures could be indicated by higher or lower scores, between-group differences could be expressed as either positive or negative numbers, which would complicate results interpretation. Thus, as recommended in CHSRI Section 9.2.3.2, 15 we converted negative difference scores to positive ones. For example, improvements were indicated by reduced scores for reaction time, and by higher scores for FMA-UE. Thus, reaction time scores were multiplied by −1 so that better scores were indicated by higher numbers.
Overall effect sizes included data from studies of acute/subacute and chronic patients. Using mean and SD for each group, group effect sizes were analyzed by calculating standardized mean differences (SMDs) and 95% confidence intervals (CIs) using a fixed-effects model. Heterogeneity was reported using the τ2 test and the I2 statistic. For I2 values >50%, indicating a substantial heterogeneity, the random-effects model was substituted. Sensitivity analysis was done for each analysis by removing lower quality studies rated as poor or fair quality (PEDro score <6).
Separate analyses were done for each of the 3 ICF domains and for posttest and follow-up time periods. In studies with several follow-up periods (eg, at 8, 12, and 24 months), only a single time point (longest follow-up) was considered since the longest follow-up period was considered more clinically important. Since all rehabilitation effects cannot be attributed only to EMG-NMES, we performed additional subanalyses to compare EMG-NMES to cyclic NMES, EMG-NMES without concomitant therapies to control, and EMG-NMES with concomitant therapies to control. These analyses were done only at the Body Structure and Function level. Subgroup meta-analyses were also conducted based on stroke chronicity (acute/subacute and chronic stroke) at each ICF domain.
Results
Study Identification and Characteristics
The search strategy, following duplicate reference removal, resulted in a total of 1212 references, 1204 from electronic databases and 8 from additional search strategies. Screening by title resulted in removal of 888 unrelated articles. The remaining 324 references were screened by abstract and 129 were selected for full-text analysis. Of these, 2616-41 matched our eligibility criteria. Studies were published between 1998 16 and 2017.38-41 Figure 1 shows the selection of studies included in the systematic review and the main reasons for exclusion.
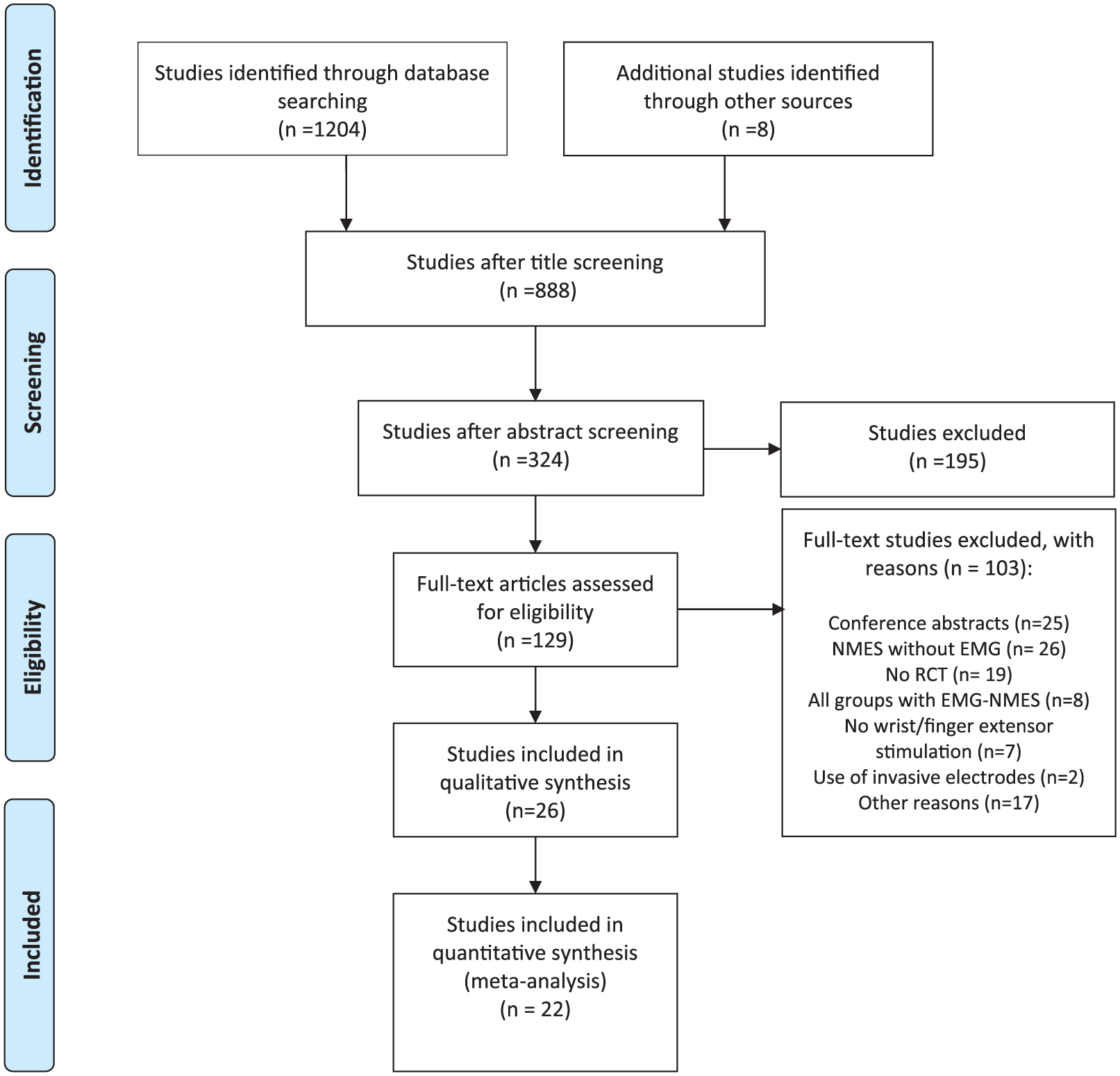
Study flow diagram.
Patient Characteristics
The studies included a total of 782 patients with stroke (Table 2). Sample sizes ranged from 9 16 to 122 37 participants. A majority of studies (n = 19) were conducted with chronic patients (536 patients),17-23,25-28,30-33,35,37-39 while 7 involved acute/subacute patients (246 patients).16,24,29,34,36,40,41 Mean age ranged from 42.6 32 to 71.1 23 years, and stroke duration ranged from 9 days 36 to 4.8 years. 28 Only one study 40 provided no details on age and time since stroke. According to initial FMA-UE scores, UL impairment levels ranged in severity from moderate (n = 6)24,25,31,35,37,38 to severe (n = 8).16,29,30,33,36,39,40,41 Twelve studies17-23,26-28,32,34 provided no FMA-UE score and therefore were not classified.
Summary of Characteristics of Included Studies.
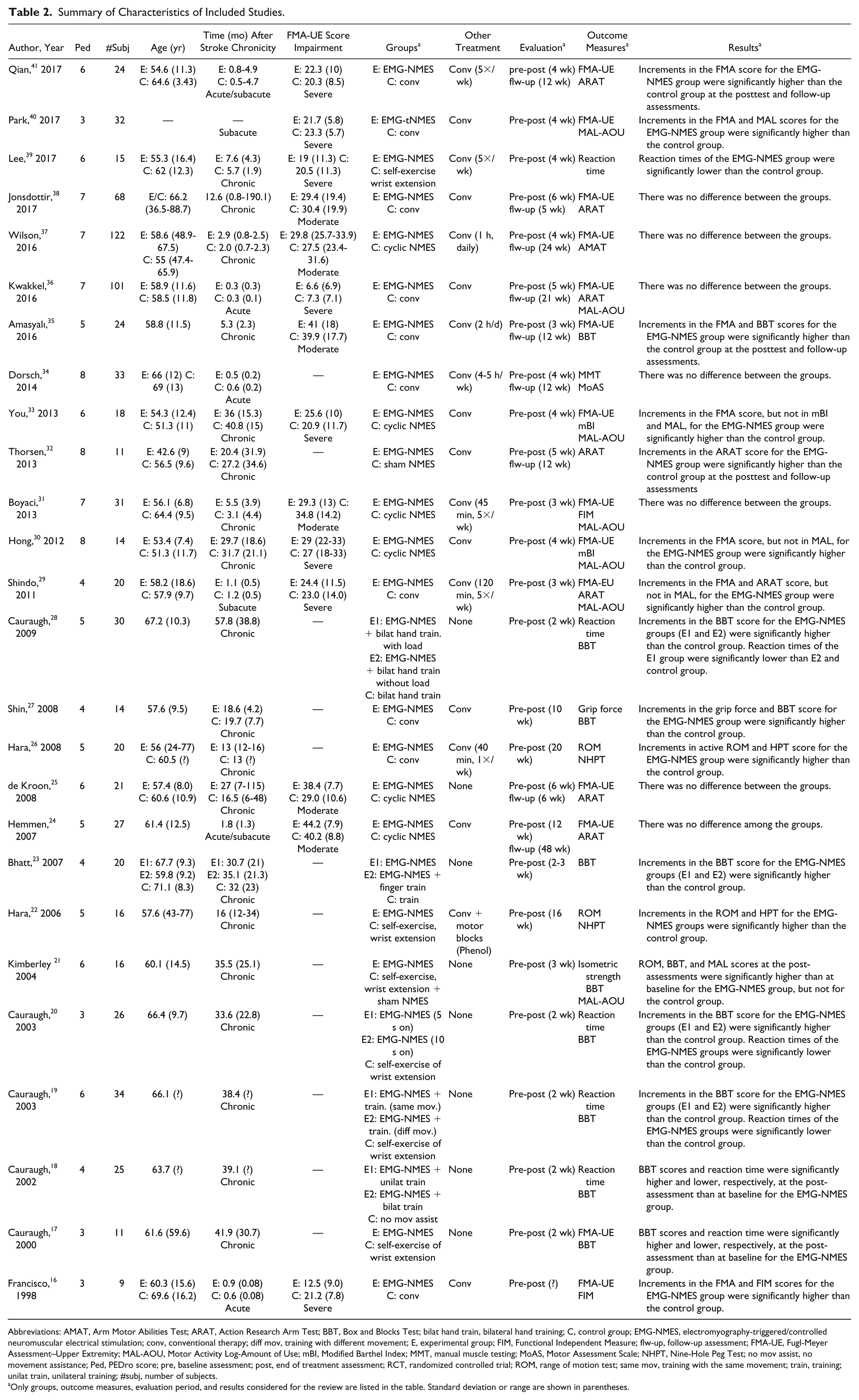
Abbreviations: AMAT, Arm Motor Abilities Test; ARAT, Action Research Arm Test; BBT, Box and Blocks Test; bilat hand train, bilateral hand training; C, control group; EMG-NMES, electromyography-triggered/controlled neuromuscular electrical stimulation; conv, conventional therapy; diff mov, training with different movement; E, experimental group; FIM, Functional Independent Measure; flw-up, follow-up assessment; FMA-UE, Fugl-Meyer Assessment–Upper Extremity; MAL-AOU, Motor Activity Log-Amount of Use; mBI, Modified Barthel Index; MMT, manual muscle testing; MoAS, Motor Assessment Scale; NHPT, Nine-Hole Peg Test; no mov assist, no movement assistance; Ped, PEDro score; pre, baseline assessment; post, end of treatment assessment; RCT, randomized controlled trial; ROM, range of motion test; same mov, training with the same movement; train, training; unilat train, unilateral training; #subj, number of subjects.
Only groups, outcome measures, evaluation period, and results considered for the review are listed in the table. Standard deviation or range are shown in parentheses.
Interventions
Table 3 summarizes the intervention characteristics and stimulation parameters of the included trials. The most commonly stimulated muscles were the extensor digitorum communis (EDC), the extensor carpi radialis (ECR), and the extensor carpi ulnaris (ECU). The dosage of electrostimulation (total hours) combined the number of sessions (per day and per week) and number of weeks of treatment. In the majority of the studies (n = 18), treatment was applied for less than 20 hours.17-20,22,23,28,30-36,38-41 Treatment dosage ranged from 617-20,28 to 168 hours. 29 Treatment duration ranged from 217-20,28 to 20 weeks 26 with frequency varying between 1 22 to 725,29 times a week and 1 to 3 times 25 a day. One trial 16 provided no details about treatment dosage.
EMG-NMES Intervention Characteristics of Trials.
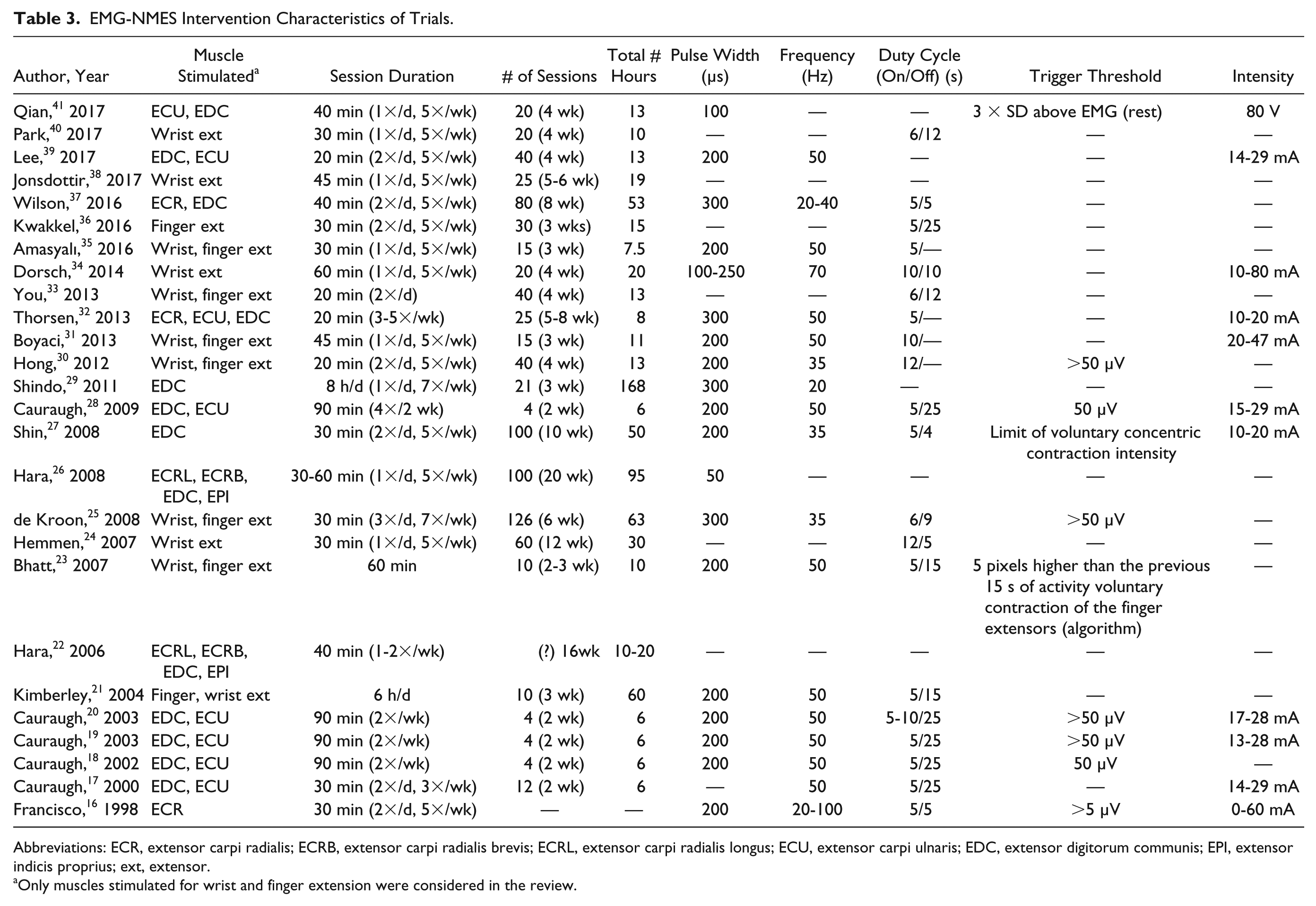
Abbreviations: ECR, extensor carpi radialis; ECRB, extensor carpi radialis brevis; ECRL, extensor carpi radialis longus; ECU, extensor carpi ulnaris; EDC, extensor digitorum communis; EPI, extensor indicis proprius; ext, extensor.
Only muscles stimulated for wrist and finger extension were considered in the review.
Control interventions included cyclic NMES,24,25,30,31,33,37 sham NMES, 32 self-exercise of wrist extension,17,19,20,21,22,39,45 unilateral or bilateral hand training,23,28 or traditional occupational and/or physical therapy.16,26,27,29,34-36,38,40,41 In general, control interventions had similar dosages relative to the EMG-NMES groups. In most studies, except for 8 RCTs,17-21,23,25,28 NMES and control interventions were applied in addition to conventional care.
Outcomes
Several different outcome measures were used (see Supplementary Table). Twenty-four studies evaluated components of the Body Structures and Function domain,16-22,24-31,33-47 24 of the Activity domain,16-22,24-38,41 and 7 of the Participation domain.21,29,30,31,33,36,40 Some studies assessed outcomes in more than one ICF domain and/or used more than one measure to analyze the same ICF component. The measure that was most often used in studies was chosen for the meta-analysis. Only those outcome measures listed inTable 2 were considered in our meta-analysis. All studies included assessments before (pre) and after (post) the intervention. However, only 9 studies24,25,32,34,35-38,41 included assessments at follow-up, ranging from 5 weeks 38 to 1 year 24 posttreatment.
Quality of Included Studies
Based on the results of the quality of evidence, 13 trials were considered to be of high quality,19,21,25,30-34,36-39,41 9 of fair quality,18,22-24,26-29,35 and 4 of poor quality16,17,20,40 (seeTable 2). The median PEDro score was 6, with scores ranging from 316,17,20,40 to 8.30,32,34
Effects of EMG-NMES on Measures in the ICF Body Structure and Function Domain
Twenty-four studies (751 patients), 7 with acute/subacute patients (246 patients),16,24,29,34,36,40,41 and 17 with chronic patients (505 patients)17-22,25-28,30,31,33,35,37-39 investigated the effect of EMG-NMES on Body Structure and Function. Of these, 20 were included in the meta-analysis (617 patients; Figure 2). Four studies16-18,30 were excluded because of missing information (mean and/or SD) needed for data pooling. The following outcome measures were identified from the studies and pooled: (1) FMA-UE,24,25,29,31,33,35-38,40,41 (2) reaction time,19,20,28,39 (3) force,26,32,39 and (4) range of movement (ROM).22,26 Since 6 studies compared more than 2 groups, additional grouping was done. In 3 studies with more than one EMG-NMES treatment group,19,20,28 groups were combined to create a single pairwise comparison in the meta-analysis. In 3 studies involving more than one control group,31,35,37 only data from the control group that underwent cyclic electrical stimulation or conventional therapy were used.
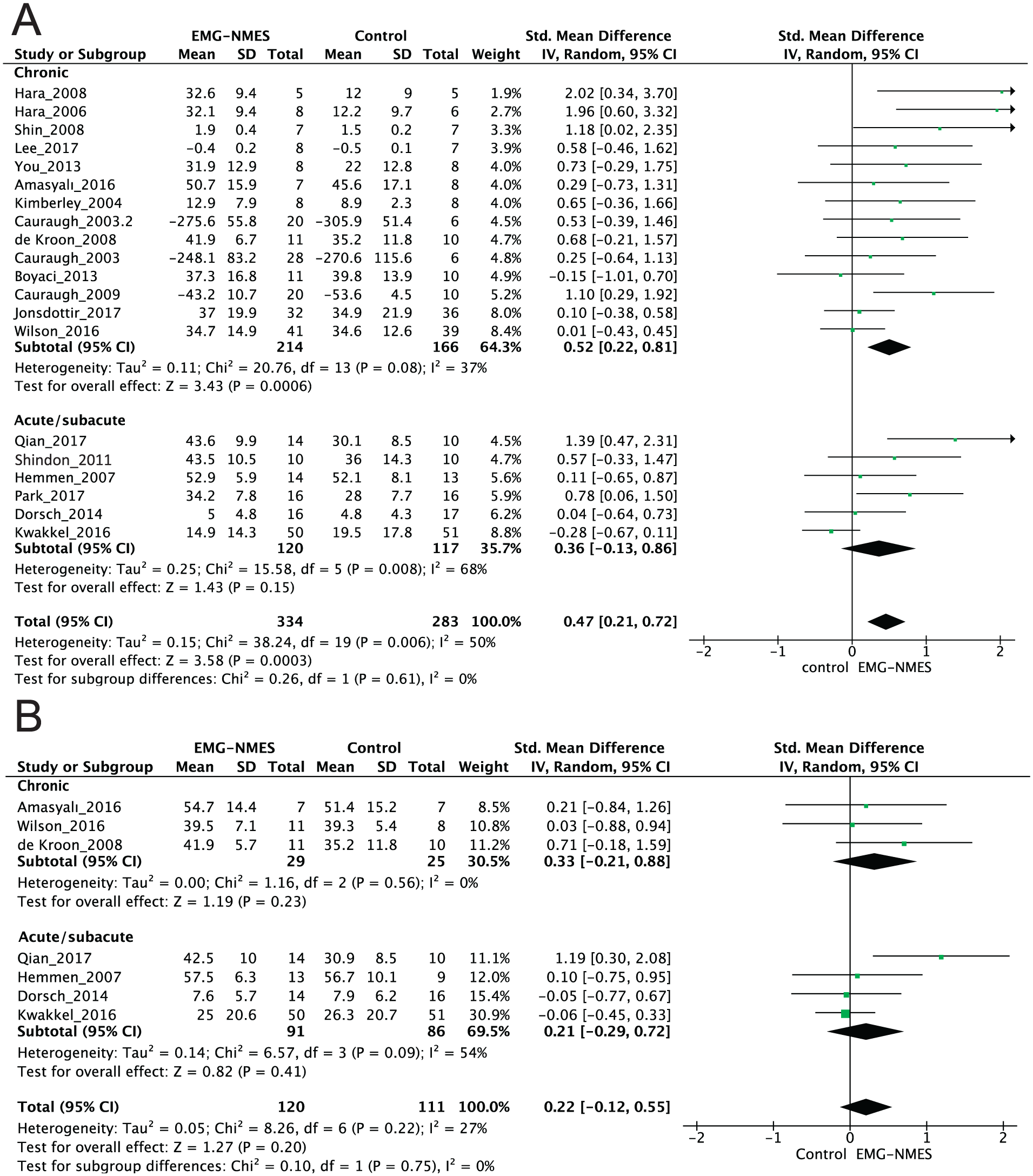
Meta-analyses of the effect of EMG-NMES versus control interventions on the Body Structure and Function domain of the International Classification of Functionality, Disability, and Health (ICF) in patients with chronic and acute/subacute stroke at the end of treatment (A) and after the follow-up period (B).
Immediately after the end of the intervention, an overall medium effect was found favoring the EMG-NMES group over the control group to improve the Body Structure and Function domain in stroke subjects (617 patients, SMD 0.47, P < .001, 95% CI 0.21 to 0.72, I2 = 50%; Figure 2A). Analyzing each patient subgroup separately, the SMD between the groups favoring EMG-NMES was significant for chronic patients (380 patients, SMD 0.52, P < .001, 95% CI 0.22 to 0.81, I2 = 37%; Figure 2A), but not for acute/subacute patients (237 patients, SMD 0.36, P = .15, 95% CI −0.13 to 0.86, I2 = 68%; Figure 2A). Seven studies24,25,34-37,41 that included follow-up assessments showed no evidence of an effect in favor of the EMG-NMES group (231 patients, SMD 0.22, P = .20, 95% CI −0.12 to 0.55, I2 = 27%; Figure 2B). In the sensitivity analysis, when the fair- and poor-quality trials were removed, the beneficial effect of EMG-NMES over the control group remained at the end of treatment (425 patients, SMD 0.33, P = .04, 95% CI 0.01 to 0.64, I2 = 55%). The sensitivity analyses did not notably affect the results at follow-up (195 patients, SMD 0.28, P = .23, 95% CI −0.18 to 0.74, I2 = 51%).
In the subanalyses, when comparing EMG-NMES to cyclic NMES24,25,31,33,37 at the end of treatment, no evidence was found in favor of either group (165 patients, SMD 0.15, 95% CI −0.16 to 0.46, P = .34, I2 = 0%). Overall effects favored both EMG-NMES alone (127 patients, SMD 0.66, P = .001, 95% CI 0.26 to 1.06, I2 = 0%) and EMG-NMES with concomitant therapies (490 patients, SMD 0.42, P = .0006, 95% CI 0.12 to 0.73, I2 = 57%) compared to control. Due to an insufficient number of trials, pooling for these comparisons at follow-up was not possible.
Effects of EMG-NMES on the ICF Activity Domain
Twenty-four studies (735 patients) investigated the effect of EMG-NMES on Activity, of which 6 trials involved acute/subacute patients (214 patients)16,24,29,34,36,41 and 18 included chronic patients (521 patients).17-23,25-28,30-33,35,37,38 After excluding 5 studies16,19,20,26,30 due to missing data, 19 trials were included in the meta-analysis (Figure 3). The following outcome measures were used for statistical pooling: (1) Box and Blocks Test (BBT),17,18,21,23,27,28,35 (2) Action Research Arm Test (ARAT),24,25,29,32,36,38,41 (3) Arm Motor Abilities Test (AMAT), 37 Functional Independent Measure (FIM), 31 modified Barthel Index (mBI), 33 Motor Assessment Scale (MoAS), 34 and (4) Nine-Hole Peg Test (NHPT). 22
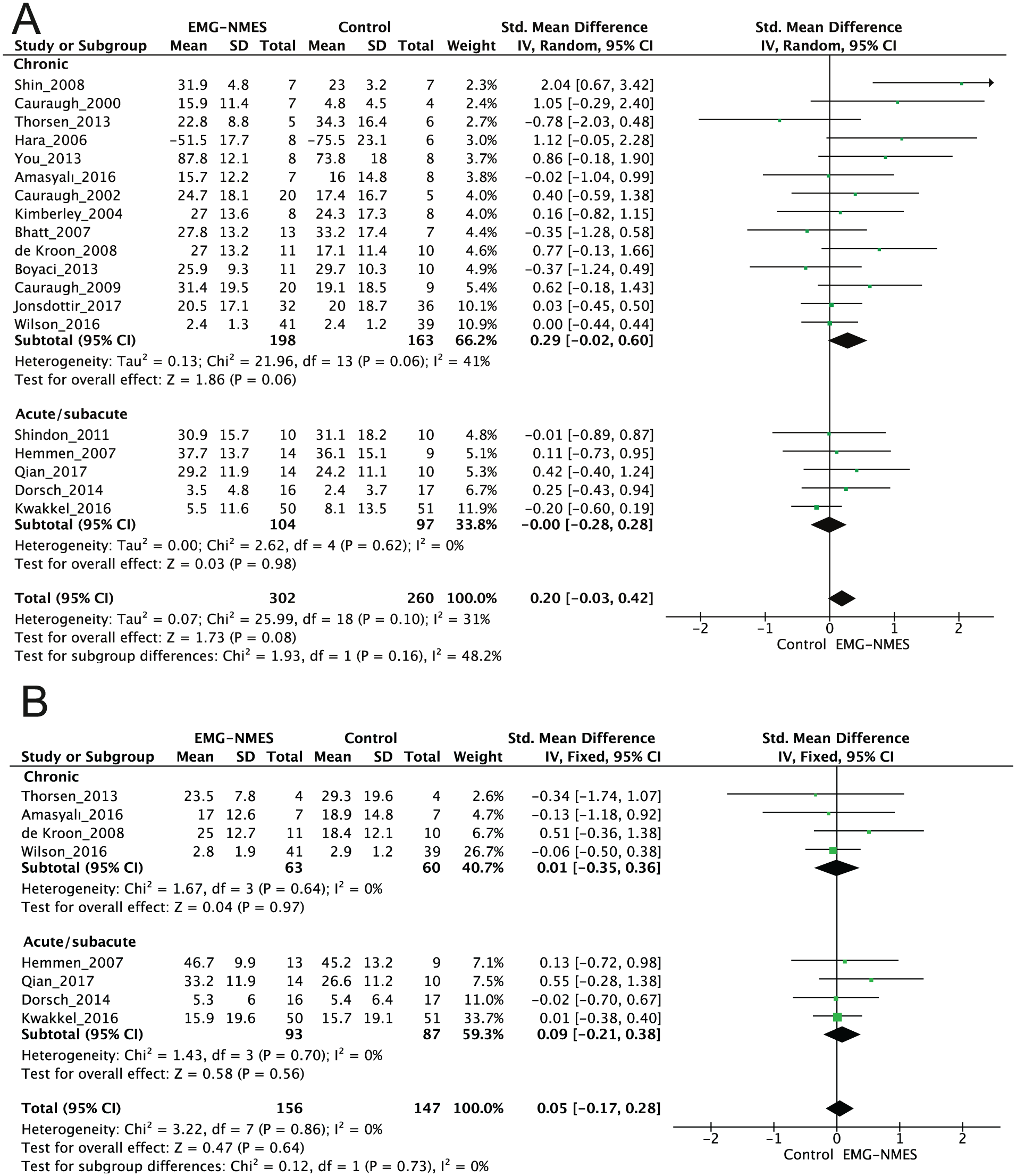
Meta-analyses of the effect of EMG-NMES versus control interventions on the Activity domain of the International Classification of Functionality, Disability, and Health (ICF) at the end of treatment (A) and after the follow-up period (B).
The meta-analysis revealed no differences for the effect of EMG-NMES compared to a control intervention on Activity at the short-term (562 patients, SMD 0.20, P = .08, 95% CI −0.03 to 0.42, I2 = 31%; Figure 3A) or the longer term follow-up time point (303 patients, SMD 0.05, P = .64, 95% CI −0.17 to 0.28, I2 = 0%; Figure 3B). Subgroup analyses revealed evidence of an effect on Activity favoring the EMG-NMES groups for chronic (361 patients, SMD 0.29, P = .06, 95% CI −0.02 to 0.60, I2 = 41%; Figure 3A), but not for acute/subacute patients (201 patients, SMD 0.00, P = .98, 95% CI −0.28 to 0.28, I2 = 0%; Figure 3A). There was no between-group difference found at the end of treatment based on sensitivity analysis (391 patients, SMD 0.05, P = .65, 95% CI −0.17 to 0.27, I2 = 12%). No studies were removed based on quality review from the follow-up meta-analysis.
Effects of EMG-NMES on the ICF Participation Domain
Seven studies involving 232 patients investigated the effect of EMG-NMES on Participation: 3 studies in acute/subacute patients (153 patients)29,36,40 and 4 studies in chronic patients (79 patients).21,30,31,33 Six studies21,29,31,33,36,40 were included in the meta-analysis, all using Motor Activity Log-Amount of Use (MAL-AOU) as the outcome measure (Figure 4).
Pooled results suggested that the EMG-NMES group effect on Participation did not differ from the control group at the end of the intervention (208 patients, SMD 0.44, P = .10, 95% CI −0.08 to 0.96, I2 = 64%). Subgroup analyses revealed a large mean effect size in chronic patients (55 patients, SMD 0.63, P = .02, 95% CI 0.08 to 1.17, I2 = 0%), but not for acute/subacute patients (153 patients, SMD 0.32, P = .47, 95% CI −0.55 to 1.19, I2 = 81%). The sensitivity analyses did not affect this result (156 patients, SMD 0.29, P = .35, 95% CI −0.32 to 0.90, I2 = 60%). Pooling at follow-up was not possible due to an insufficient number of trials.
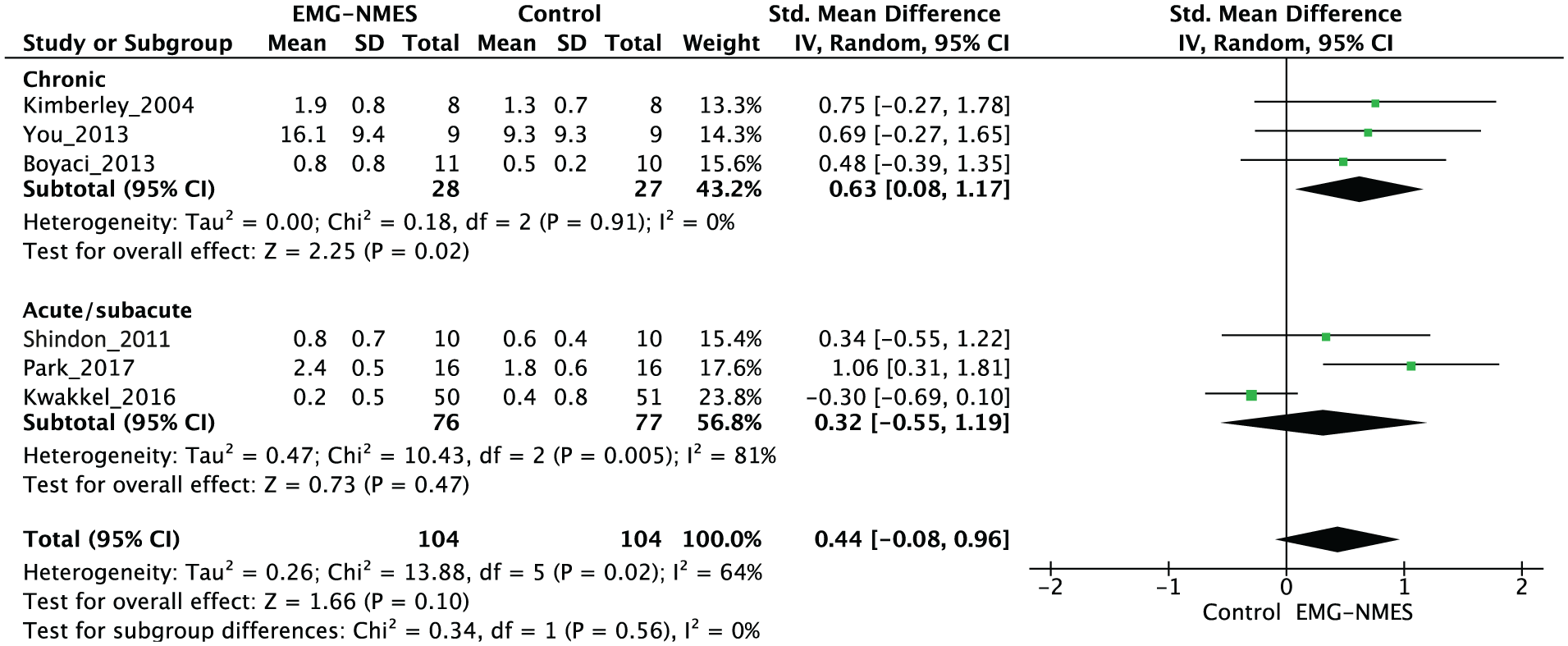
Meta-analyses of the effect of EMG-NMES versus control interventions on the Participation domain of the International Classification of Functionality, Disability, and Health (ICF) at the end of treatment.
Discussion
This systematic review and meta-analysis demonstrated that EMG-NMES is a good option to improve UL motor impairment in patients with severe or moderate stroke in the chronic phase of recovery. Based on a larger number of participants (n = 617) than previous reviews (n = 86, 5 n = 103, 6 n = 157 7 ), we found that EMG-NMES leads to greater short-term improvements in the ICF Body Structure and Function domain than other dose-matched therapies or no treatment. However, these effects were not retained over long-term follow-up periods.
This is in contrast to the conclusions of 2 previous systematic reviews conducted in 2004 6 and in 2008, 7 Van Peppen et al 6 showed no evidence to support the use of EMG-NMES based on a qualitative best-evidence synthesis. The lack of meta-analysis limits the precision with which we can compare their results to ours. No evidence of a favorable effect of EMG-NMES was reported in a meta-analysis of 8 studies by Meilink et al. 7 It is important to note that new and higher quality studies have been published since the 2008 systematic review, resulting in larger sample sizes in the present review, which can account for the different conclusions.
Quality of Evidence
Half of the studies (50%) achieved a high-quality score. None of the studies clearly satisfied the criteria for therapist blinding and only a small number had patient blinding.21,31,32 These biases could not be avoided because of known difficulties in blinding investigators and participants for rehabilitation intervention groups. Additionally, there was a lack of data on allocation concealment16,18-28,31-33,35,39-41 and intention-to-treat analysis16,18-28,31-33,35,39-41 in more than half the studies. Bias due to these methodological limitations tends to overestimate treatment effects and may consequently lead to higher effect sizes.42,43
Impact of EMG-NMES Treatment on ICF Domains
There was a moderate overall effect size in favor of EMG-NMES at the Body Structure and Function domain, suggesting EMG-NMES is effective for the short-term improvement of UL impairment. There was a significant benefit of EMG-NMES for the combination of 4 outcomes, FMA-UE, reaction time, force, and ROM, although larger effects were noted for combinations that included force 27 and ROM.22,26 The effect of EMG-NMES at the Activity domains was nonsignificant. The reported differences in outcomes at the 2 domains may be related to EMG-NMES interventions focusing on remediating impairments only, while outcomes are measured at both Body Structure and Function and Activity domains. Based on the motor learning principle of task specificity, 44 it is not entirely surprising that improvements in impairment are not good predictors of limb use in the execution of tasks or actions. In addition, training gains are noted when transfer conditions match those that were practiced during the training session. 44
Despite the large mean effect size for the Participation domain in chronic patients, this effect should be interpreted with caution because of the large potential of effect bias. First, there was no significant overall effect size for the whole group (acute/subacute and chronic). Second, only 6 heterogeneous studies investigated EMG-NMES in the Participation domain, preventing any conclusions to be made regarding benefits at this domain.
Impact of EMG-NMES Treatment in Acute/Subacute Versus Chronic Stroke
According to the subgroup analysis, EMG-NMES was more beneficial to patients with chronic versus acute/subacute stroke. However, this effect should be interpreted with caution because of the large variability of chronicity and initial impairment level in the acute/subacute group, but not in the chronic group. For example, chronicity in the acute/subacute groups ranged from 9 days 36 to 1.8 months 24 poststroke and patients had a large baseline UL impairment variability (initial FMA ranged from severe—6.6 points 36 to moderate—44.2 points 24 ).
The lack of positive effects in the acute/subacute group was surprising since it was expected that acute patients would have better improvement due to concomitant spontaneous recovery. The amount of sensorimotor improvement due the interventions such as EMG-NMES may be related to the initial level of impairment severity. 45 Indeed, in a large multicenter study, Kwakkel et al 36 found no evidence that EMG-NMES improves UL function in patients without some voluntary control of finger extensors at baseline. Therefore, the heterogeneity of baseline impairment among studies that involved acute/subacute patients might have limited trial comparability.
Comparison of EMG-NMES to Stimulation Alone
Learning is enhanced when motivation and attention are engaged during training. 44 In contrast to cyclic NMES, EMG-NMES requires active patient participation due to the voluntary effort needed to direct attention to the motor training. Therefore, we expected that EMG-NMES would be more effective than cyclic NMES. Indeed, previous evidence showed that combining EMG signal detection of intended movements with electrical stimulation promoted more brain plasticity than cyclic NMES as demonstrated by increased amplitudes of motor evoked potentials. 46 However, our pooled data showed that EMG-NMES was not more effective than cyclic EMG. The small number of studies (n = 5) may have resulted in a lack of power to detect between-group differences.
Long-Term Effects
There were no differences between groups at follow-up, indicating the lack of beneficial effects of EMG-NMES beyond the stimulation period. This may be related to the variability in the timing of follow-up measurements among studies that ranged from 5 weeks 38 to 1 year. 24 Examination of effects at specific follow-up time points may provide more information about dosing effects and carryover in future studies. However, for the present time, the short-term benefit of EMG-NMES should be balanced against the lack of long-term evidence currently available.
Limitations
Differences in stimulation parameters may be crucial to treatment effects. 45 However, the variability and lack of details regarding stimulation parameters limited our ability to determine an optimal EMG-NMES protocol. No distinction was made between different types of control interventions, which may influence results significance. According to the ICF, 8 Body Structure is defined as “anatomical parts of the body such as organs, limbs and their components” and Body Function as “the physiological functions of body systems.” Pooling of both concepts in a single domain could have limited results interpretation and resulted in the loss of useful information since the various impairments after stroke may have different impacts on Activity limitations. Finally, this systematic review focused on the general effects of EMG-NMES and ignored the impact of different types of NMES applications (ie, EMG-triggered, controlled, ipsilateral, contralateral) due to the small number of studies for each type. However, absence of high heterogeneity in most analyses suggest that this was not an important source of bias.
Clinical Implications
Generalizing quantitative effects using systematic reviews or meta-analyses related to clear clinical targets such as those described in the ICF can help clinicians make decisions about clinical practice. Considering the high quality of half of the included studies and the moderate effect sizes found, the present meta-analysis suggests that EMG-NMES may be included in clinical practice to achieve greater gains on poststroke UL impairment. EMG-NMES may be readily integrated into the clinic since it inexpensive and can be applied at home. Our findings support previous recommendations that patients with chronic impairment may be the best candidates for such therapies. 47 It is important to highlight that the EMG-NMES should be proposed as a supplemental therapy and not as a stand-alone therapy since most of the included studies used it as additional therapy.
Conclusion
We found that adding EMG-NMES to conventional therapy programs improves short-term UL impairment, but not task performance or participation in individuals who are more than 3 months poststroke. Although our results are encouraging, more research is needed to extend our knowledge about the benefits of EMG-NMES. Thus, future studies should address the following issues: (1) the benefits of EMG-NMES for patients within the first 3 months poststroke, (2) the comparison of the effects of different EMG-NMES modalities, and (3) the optimal EMG-NMES protocol.
Supplemental Material
Supplementary_Table – Supplemental material for Electromyogram-Related Neuromuscular Electrical Stimulation for Restoring Wrist and Hand Movement in Poststroke Hemiplegia: A Systematic Review and Meta-Analysis
Supplemental material, Supplementary_Table for Electromyogram-Related Neuromuscular Electrical Stimulation for Restoring Wrist and Hand Movement in Poststroke Hemiplegia: A Systematic Review and Meta-Analysis by Katia Monte-Silva, Daniele Piscitelli, Nahid Norouzi-Gheidari, Marc Aureli Pique Batalla, Philippe Archambault and Mindy F. Levin in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: KMS was supported by Capes, Coordination of Superior Level Staff Improvement—Brazil (Grant No. 88881.120194/2016-01).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
