Abstract
Background. Robot-assisted therapy provides high-intensity arm rehabilitation that can significantly reduce stroke-related upper extremity (UE) deficits. Motor improvement has been shown at the joints trained, but generalization to real-world function has not been profound. Objective. To investigate the efficacy of robot-assisted therapy combined with therapist-assisted task training versus robot-assisted therapy alone on motor outcomes and use in participants with moderate to severe chronic stroke-related arm disability. Methods. This was a single-blind randomized controlled trial of two 12-week robot-assisted interventions; 45 participants were stratified by Fugl-Meyer (FMA) impairment (mean 21 ± 1.36) to 60 minutes of robot therapy (RT; n = 22) or 45 minutes of RT combined with 15 minutes therapist-assisted transition-to-task training (TTT; n = 23). The primary outcome was the mean FMA change at week 12 using a linear mixed-model analysis. A subanalysis included the Wolf Motor Function Test (WMFT) and Stroke Impact Scale (SIS), with significance P <.05. Results. There was no significant 12-week difference in FMA change between groups, and mean FMA gains were 2.87 ± 0.70 and 4.81 ± 0.68 for RT and TTT, respectively. TTT had greater 12-week secondary outcome improvements in the log WMFT (−0.52 ± 0.06 vs −0.18 ± 0.06; P = .01) and SIS hand (20.52 ± 2.94 vs 8.27 ± 3.03; P = .03). Conclusion. Chronic UE motor deficits are responsive to intensive robot-assisted therapy of 45 or 60 minutes per session duration. The replacement of part of the robotic training with nonrobotic tasks did not reduce treatment effect and may benefit stroke-affected hand use and motor task performance.
Introduction
Stroke is the leading cause of long-term adult disability and has a prevalence of 25.7 million people worldwide. 1 Chronic stroke affects 7.2 million Americans, 1 and upper extremity (UE) impairment persists in 30% to 66% of individuals 6 months or more postonset.2-6 The degree of initial paresis affects recovery, and only 9% to 20% of people with severe paresis regain full arm use.3,7 Chronic dysfunction has lasting negative consequences on independence and quality of life8,9 and is correlated with the degree of UE impairment. Innovative motor learning strategies using intensive repetitive task-specific practice show associated activity-dependent neuroplasticity and functional improvement in all phases poststroke10,11; however, severe paresis impedes participation in evidence-based therapies such as constraint-induced therapy 12 or repetitive task training. 13 Instead, rehabilitation has focused on teaching compensatory techniques to substitute for functional loss. 14 As a result, use-dependent experiences are limited, and the response to intensive task-specific training in individuals with moderate to severe chronic impairment has not been fully explored.
Robot-assisted arm rehabilitation offers a cost-effective method to provide quantifiable, intense, and reproducible therapy to individuals with significant disability.15-19 Studies have shown durable robot-mediated benefits for moderate-to-severe chronic arm dysfunction in single18,20,21 and multisite trials. 22 The clinical impact of robot rehabilitation, viewed within the context of the World Health Organization International Classification of Functioning, Disability and Health, has largely occurred at the body function level (range of motion, strength, spasticity) specific to the proximal joints trained 23 and not at the activity or participation level. Evidence for improvement in activities of daily living (ADLs) and increased real-world arm use is insufficient and inconclusive. 24 Moreover, attempts to make robot training functionally relevant have not had a compelling effect. A multijoint, multiplanar, functional arm robot paradigm using an exoskeletal (BONES) design did not prove superior to single-joint robot training. 25 Likewise, a comparison of functional-based robot training to impairment-based robot training did not suggest additional benefit. 26
A large multisite robot therapy (RT) trial by Lo et al 22 found no difference between robot training and intensity-matched therapist-assisted training. 27 In this study, the quantity of therapy or dose per session for the 2 interventions was comparable. The therapeutic delivery and training, however, were very different, and the significance of treatment content or type is an important question. An investigation of robotic versus nonrobotic motor learning interventions 28 suggested intensity and not treatment type as a potential key factor. In contrast, nonrobotic task-specific training added to robot-assisted wrist training showed increased motor and quality-of-life benefits in individuals with chronic deficits compared with active control therapy or a combination of robot and impairment-based therapy.29,30 There is substantial evidence supporting a combined treatment approach in constraint-induced therapy. Key elements of this approach are constraint of the less-affected arm, intensive repetitive task training, and a behavioral or “transfer package” promoting use outside the clinical setting. 31 Whereas, RT has demonstrated benefits via high-intensity task training, 32 an integrated treatment approach transitioning robot-mediated improvement to functional use has yet to be developed.
In this clinical trial, “hands on” therapist-assisted task-specific training named transition-to-task training (TTT) was added to RT to determine if this combination improved arm use. This 12-week single-blind randomized trial compared the effectiveness of 45 minutes of robotic therapy combined with 15 minutes of TTT to 60 minutes of robotic therapy alone (RT) for participants with chronic moderate to severe hemiparesis. We hypothesized that impairment-based robot training combined with TTT would decrease impairment and promote a transition to real-world use. By keeping intervention time constant across groups, we were also able to examine whether robot-mediated gains were achieved with less time.
Methods
This was a 12-week prospective, randomized, single-blind, controlled trial. The Veterans Affairs Maryland Health Care System (VAMHCS) Research Safety Committee, University of Maryland Institutional Review Board (IRB), and the Massachusetts Institute of Technology Committee on the Use of Humans as Experimental Subjects approved the study; written informed consent was obtained; and all procedures followed protocol in accordance with the ethical standards of the IRBs. Participants with chronic moderate-to-severe UE deficits were recruited from VAMHCS clinics and the community. Inclusion criteria were (1) adults with clinically defined, unilateral, hemiparetic ischemic stroke onset ≥6 months or hemorrhagic stroke onset ≥1 year; (2) Fugl-Meyer (FMA) score 7 to 38 (range, 0-66). Exclusion criteria were (1) inability to give informed consent; (2) medical, orthopedic, or visual conditions precluding or limiting robot use; (3) botulinum toxin injection to the affected arm within 3 months of enrollment; and (4) inability to commit time or lack of transportation to the trial.
Participants
There were 127 candidates screened, and 45 met enrollment criteria. The most common reasons for exclusion were scoring outside the required FMA range, inability to commit time, or lack of transportation to the trial. Participants were randomly assigned to 1 of 2 RT groups: 1-hour RT alone (n = 22) or a combination of RT (45 minutes) combined with 15 minutes of therapist-assisted TTT (n = 23; Figure 1). Randomization was stratified by baseline FMA scores, where moderate and severe impairments were defined as a range between 26 and 38, and 7 and 25, respectively. This range was based on guidelines from Luft et al, 33 where severely impaired patients scored below 25 and moderately impaired patients scored between 26-to-50 on the FMA. Three baseline scores, taken 1 week apart, defined motor stability. A difference ≤2 points, representative of a ≤3% change in the maximum score of 66, was required to proceed to randomization. If 1 of the 3 evaluations was outside this value, a fourth baseline was completed. Four participants completed a fourth baseline, and no one was disqualified based on this criterion. A total of 12 participants presented with moderate impairment (baseline FMA scores ≥ 26) and were equally distributed in the randomized groups: 6 in RT (mean FMA = 32.6 ± 1.92) and 6 in TTT (mean FMA = 32.9 ± 1.55). In all, 29 participants presented with severe impairment (baseline FMA scores ≤ 25), with 13 randomized to RT (mean FMA = 17.8 ± 1.61) and 16 to TTT (mean FMA = 15.9 ± 1.05).
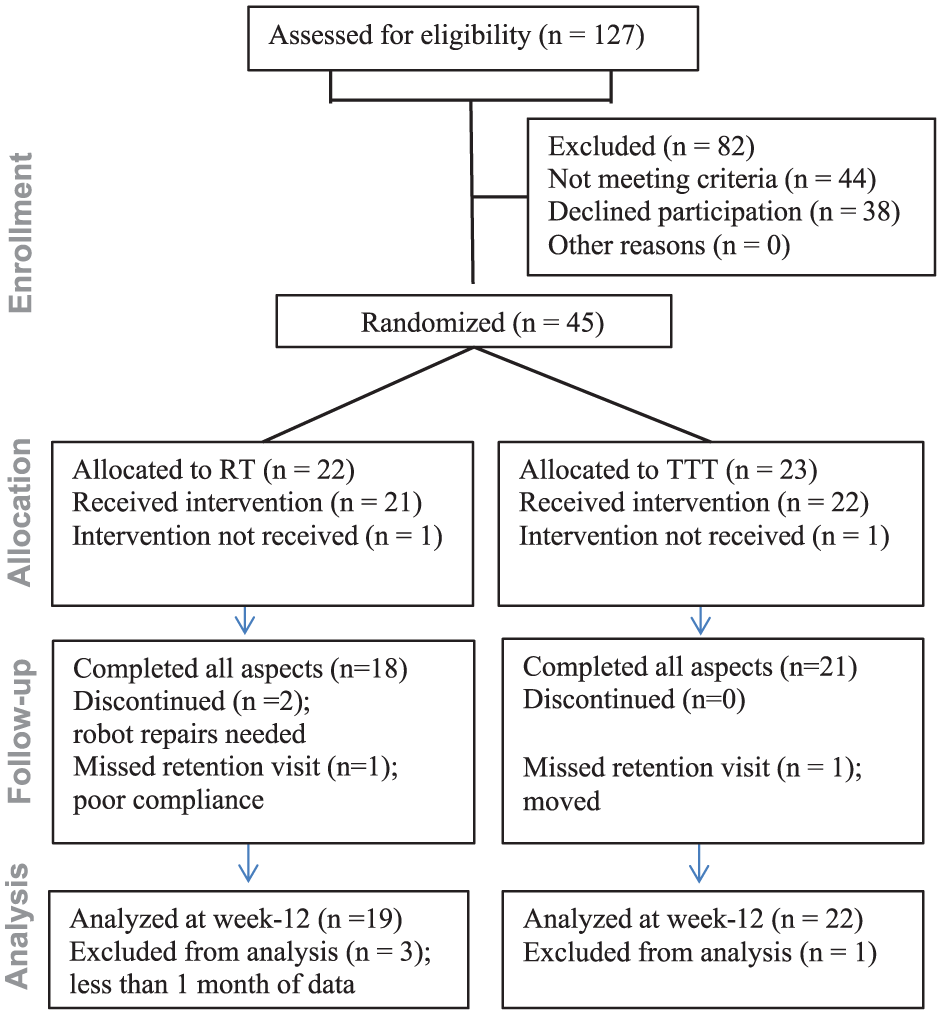
CONSORT study flow diagram.
Intervention
The study intervention was administered for 1 hour, 3 times a week, over a 12-week period for a maximum of 36 visits. The robot component was identical, with the exception of time allotment. Participants randomized to RT received 60 minutes of robot training, and participants randomized to TTT received 45 minutes of robot training followed by 15 minutes of TTT. All participants trained on 2 distinct robots in a set progression of 4-week training blocks. Month 1 (weeks 1-4) focused on distal arm training using a 3-degree-of-freedom (DOF) wrist robot. Month 2 (weeks 5-8) focused on proximal arm training using a 2-DOF shoulder-elbow robot. Month 3 (weeks 9-12) alternated distal and proximal training by intervention day using the wrist or shoulder-elbow robot, respectively. This training sequence was based on findings by Krebs et al, 34 where distal robot training preceding proximal suggested better outcomes. The reverse sequence of proximal robot training immediately followed by distal robot training did not show a benefit. 35
The performance-based directional impedance control system34,36 common to both robots automatically adjusted stiffness and damping for “assistance as needed” in response to movement metrics monitored during multiple games per session of a visually guided 320-target point-to-point game. A compliant interface ensured safety, with robot assistance yielding to unexpected movements or hypertonicity. Performance-based adjustments reset at the start of each training day. Therefore, assistance or challenge adapted within each session but was not progressive across the intervention. Scores representing robot-derived metrics for initiation, smoothness, accuracy, distance, and power displayed at a set interval after every 80 targets. A therapist supervised each session to keep environmental distractions to a minimum and provided encouragement and feedback during the robot scores. An unassisted trial of 16 targets preceded and succeeded each robot-assisted game to gauge active movement and promote active participation. The robot-mediated adaptive training, set intervals of feedback, and unassisted reaching trials repeated for 60 or 45 minutes respectively, based on group assignment.
Wrist Robot–Assisted Training
The first block of training (weeks 1-4) targeted distal movements using a 3-DOF wrist robot 34 (InMotion 3.0; Bionik Labs, Watertown, USA). Participants sat in the robot chair with a 4-point seatbelt to minimize compensatory trunk movements. The arm was positioned in 20° of shoulder abduction, 30° of shoulder flexion, and approximately 80° of elbow flexion supported by the robot. Straps secured the forearm in neutral, and the hand grasped or was secured to the robot handle with a glove. Multiple available planes of motion allowed for wrist flexion/extension, radial/ulnar deviation, and forearm pronation/supination. Wrist motions, trained separately from forearm motions, consisted of visually guided point-to-point reaching from the center of a circle out to 1 of 8 peripheral targets. The targets, distributed around the circle, corresponded to an ellipse with a major axis of 60° (30° for flexion/extension each) and a minor axis of 30° (15° for radial/ulnar deviation each). Participants flexed, extended, or radially or ulnarly deviated their wrist to reach a pseudo-random presentation of 320 targets. The wrist-specific training changed to a forearm task after two 320-target performance-based games. The forearm training required pronation and supination to reach targets along a horizontal line.
Robot-Assisted Planar Shoulder-Elbow Reaching
The second block of training (weeks 5-8) targeted proximal movements using the shoulder- elbow robot (InMotion2; Bionik Labs, Watertown, USA). This robot, described in detail elsewhere, 37 afforded 2-DOF reaching in the horizontal plane. The point-to-point reaching task required coordinated shoulder and elbow movements to direct the robot handle to targets distributed around a 14-cm radius circle.
Multisegment Alternating Robot Training
The third block (weeks 9-12) was an integration of distal and proximal training and alternated between the wrist or shoulder-elbow robot on each training day. The alternating training sessions were separated by 24 hours or more to minimize potential motor learning interference related to exposure of 2 distinct tasks in close temporal proximity. 34 In this manner, multisegment training for the whole arm (distal and proximal) was completed each week.
Transition to Task Training
Participants randomized to TTT received 45 minutes of RT followed by 15 minutes of therapist-assisted TTT activities. These activities were progressive in nature utilizing everyday items for contextual multijoint tasks (Figure 2). The TTT activities complemented the distal (wrist) or proximal (shoulder-elbow) robot training. For example, robot-trained wrist extension and radial deviation was followed by tasks integrating these movements to turn on a light switch or lift a cup. Robot-assisted forearm training was followed by tasks to open a door handle or turn a water bottle upright. Shoulder-elbow robot training was followed by tasks combining elbow and shoulder movements to slide objects to varying locations across a tabletop. The final month alternated between the 2 robots by intervention day; therefore, the TTT activities included complex arm movements. Some examples were simulated drinking from a cup, reaching for an object on the floor, and feeding tasks.

Transition-to-task training activity examples.
In summary, wrist movement and object manipulation were trained in block 1, arm transport and horizontal reaching in block 2, and multijoint combinations of wrist and shoulder-elbow movement in block 3. An experienced therapist selected 2 new goal-directed tasks every 2 weeks based on each participant’s abilities and interests. Tasks were completed in a seated position, emphasizing unilateral arm use. A stopwatch timed each of the prescribed 7.5-minute tasks, and resting, once the task started, was discouraged. The number of completed repetitions in each 7.5-minute interval was recorded using a simple counter. Modeling on the proof-of-concept task-specific training study by Birkenmeier et al, 38 objective measures of reaching distance, repetitions completed, and task independence guided grading and progression. Parameters such as movement amplitude and demand (resistance, speed, and/or precision) were adjusted for challenge and generalization to real life. For example, resistance was altered by adding weight to a container (water or dried beans), adjusting surface friction while reaching, or lifting the arm against gravity. Functional stretching and weight bearing were integrated within the activities, and manual assistance was provided, when necessary, to limit abnormal or compensatory strategies.
Outcome Measures
The primary outcome was the change in the UE FMA from baseline (mean of the 3 baseline measures) to final training at week 12. The FMA has established reliability and validity as a stroke-specific motor impairment measure 39 and is commonly used in robot rehabilitation trials. 40 Participants were evaluated by an experienced occupational therapist blinded to group assignment. Evaluations were conducted according to written protocol and training videos. A coinvestigator experienced in all measures verified training. Interrater reliability between the 2 was established using videotaped evaluations administered on a volunteer stroke patient. All discrepancies were discussed and clarified until agreement reached 100%. This same evaluator assessed all participants. Evaluations were conducted at baseline; completion of block 1 (week 4), block 2 (week 8), and block 3 (week 12); and at a 12-week retention visit. The Stroke Impact Scale (SIS) was completed at baseline, final training, and retention.
Secondary Outcome Measures
The UE FMA proximal (shoulder/elbow/forearm; maximum = 42) and distal (wrist/hand; maximum = 24) subscores were calculated to examine treatment effect on each UE segment. Arm function and perceived participation in daily real-world activities were examined using the Wolf Motor Function Test (WMFT) and SIS, respectively. The WMFT assessed training related performance change in 15 specific timed tasks and 2 strength activities. It has high interrater reliability, internal consistency, and test-retest reliability. 41 The SIS is a reliable and valid structured poststroke interview of perceived ability shown to be sensitive to quality-of-life changes. 42 The hand section was selected based on the hand’s importance in goal-directed tasks.
Data Analysis
Participant characteristics were compared by intervention group for all individuals randomized using a t-test for approximating normally distributed variables, the Wilcoxon rank-sum test for variables with skewed distributions, and the χ2 test or Fisher exact test for comparing categorical variables. Performance between groups was compared separately on the FMA, log-transformed WMFT, and SIS hand at week 12 (posttraining) and week 24 (retention), based on linear mixed models. Any missing data were excluded from analysis. One participant in each group did not complete all aspects of the study because of missing the follow-up retention visit. The missing data were excluded from the 24-week retention analysis. The primary 12-week analysis was based on linear mixed models using an unstructured covariance model. The response variables were FMA change score, log-transformed WMFT change score, and SIS hand change score, in separate models. The independent variables included the indicator for the intervention group, indicators for time, baseline FMA stratum (FMA ≥26 vs FMA ≤25), and the 2-way and 3-way interactions among them. Subgroup comparisons were performed based on the linear mixed models. Adjusted P values were computed using Tukey-Kramer’s method. Group mean ± standard error were reported. Statistical significance was declared when P <.05. Analyses were conducted using SAS 9.3 (SAS Institute Inc, Cary, NC).
Results
No significant differences in age, sex, stroke characteristics, or baseline FMA scores were seen in the 2 groups (Table 1).
Demographics and Stroke Characteristics.
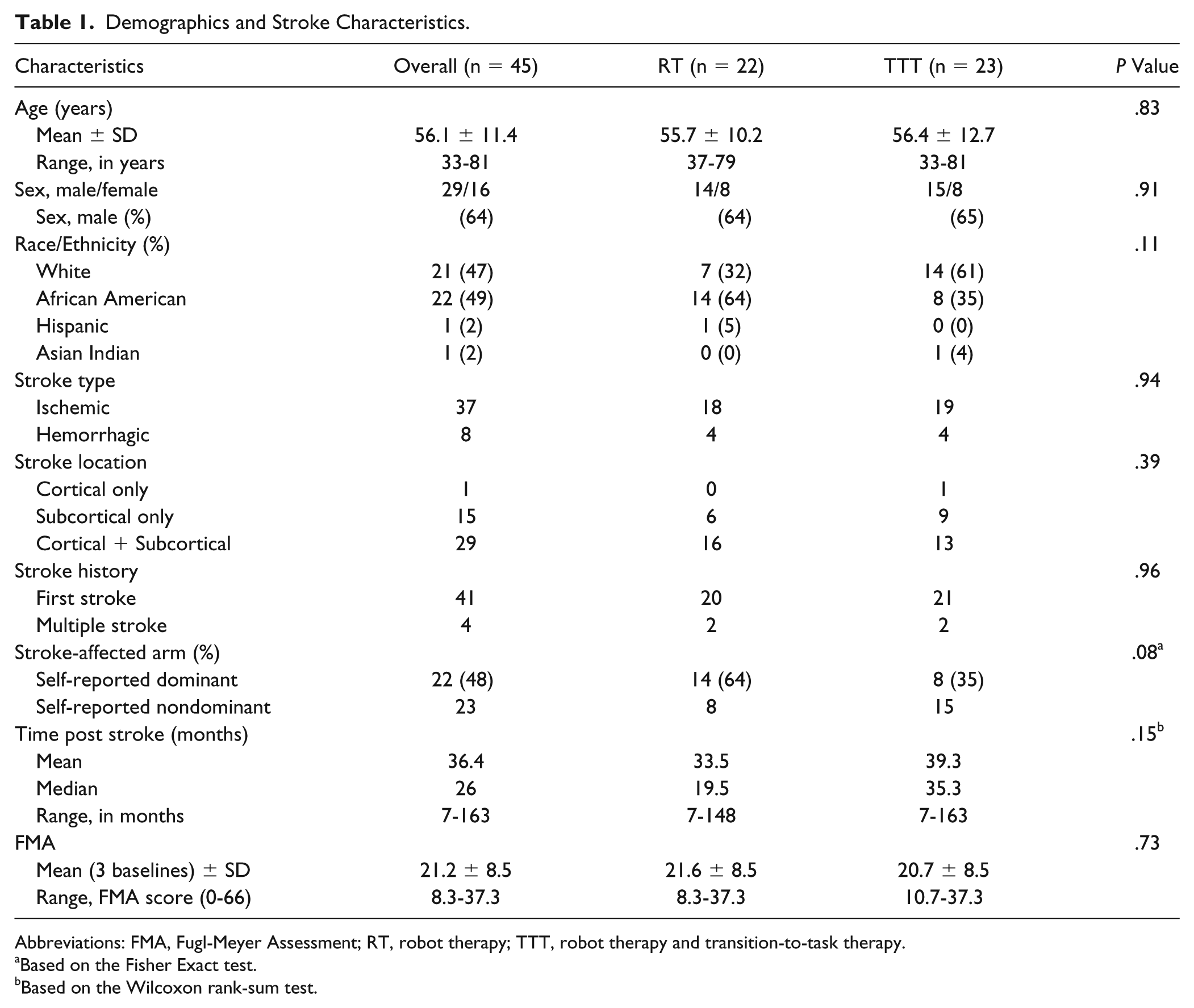
Abbreviations: FMA, Fugl-Meyer Assessment; RT, robot therapy; TTT, robot therapy and transition-to-task therapy.
Based on the Fisher Exact test.
Based on the Wilcoxon rank-sum test.
The primary outcome measure, FMA change posttraining (week 12), did not differentiate the 2 robot therapies, with similar impairment gains (P = .5) seen for robot-assisted therapy (RT) and for robot-assisted therapy combined with therapist-assisted TTT. A significant within-group change occurred at week 12, and gains were 2.87 ± 0.70 and 4.81 ± 0.68, respectively, for RT and TTT. The TTT group had a larger FMA distal (wrist-hand) subscale gain, but the amount of change did not differentiate the 2 interventions (Table 2). Overall, FMA improvement was comparable and sustained at retention (week 24) for RT (4.35 ± 0.93; P < .01) and TTT (4.71 ± 0.90, P < .01; Table 3).
Motor Function Changes in Primary and Secondary Measures: Week 12 (Final Training). a
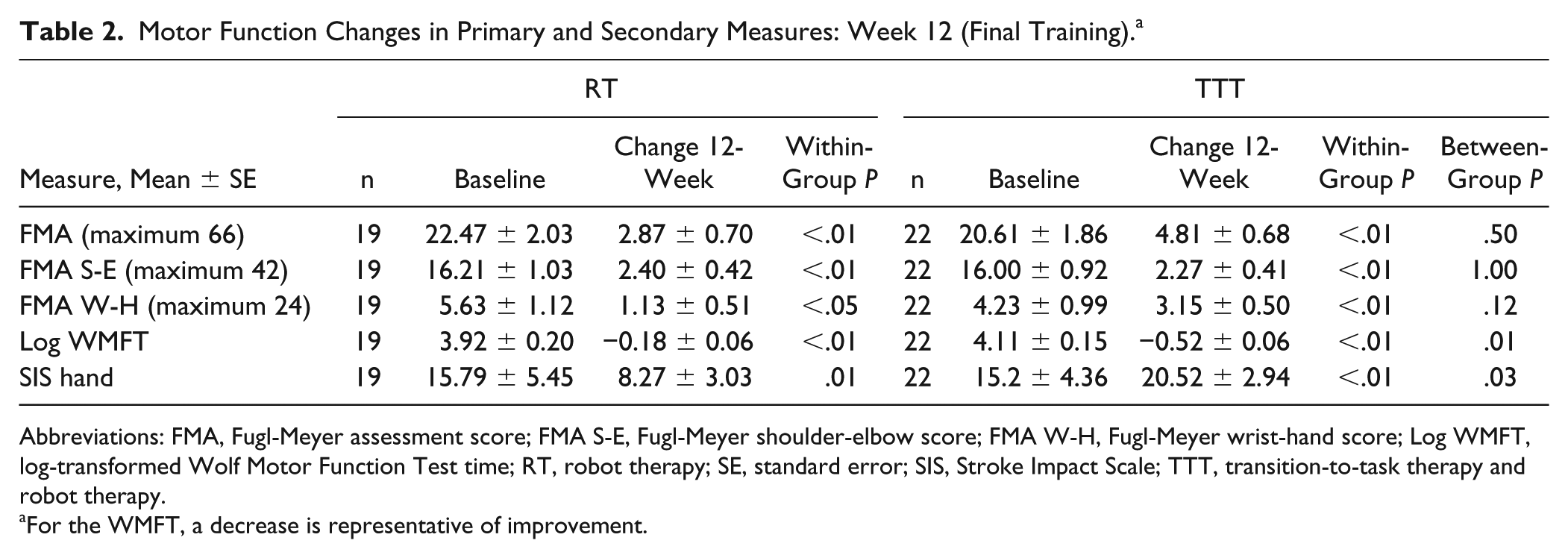
Abbreviations: FMA, Fugl-Meyer assessment score; FMA S-E, Fugl-Meyer shoulder-elbow score; FMA W-H, Fugl-Meyer wrist-hand score; Log WMFT, log-transformed Wolf Motor Function Test time; RT, robot therapy; SE, standard error; SIS, Stroke Impact Scale; TTT, transition-to-task therapy and robot therapy.
For the WMFT, a decrease is representative of improvement.
Motor Function Changes in Primary and Secondary Measures: Week 24 (Retention). a
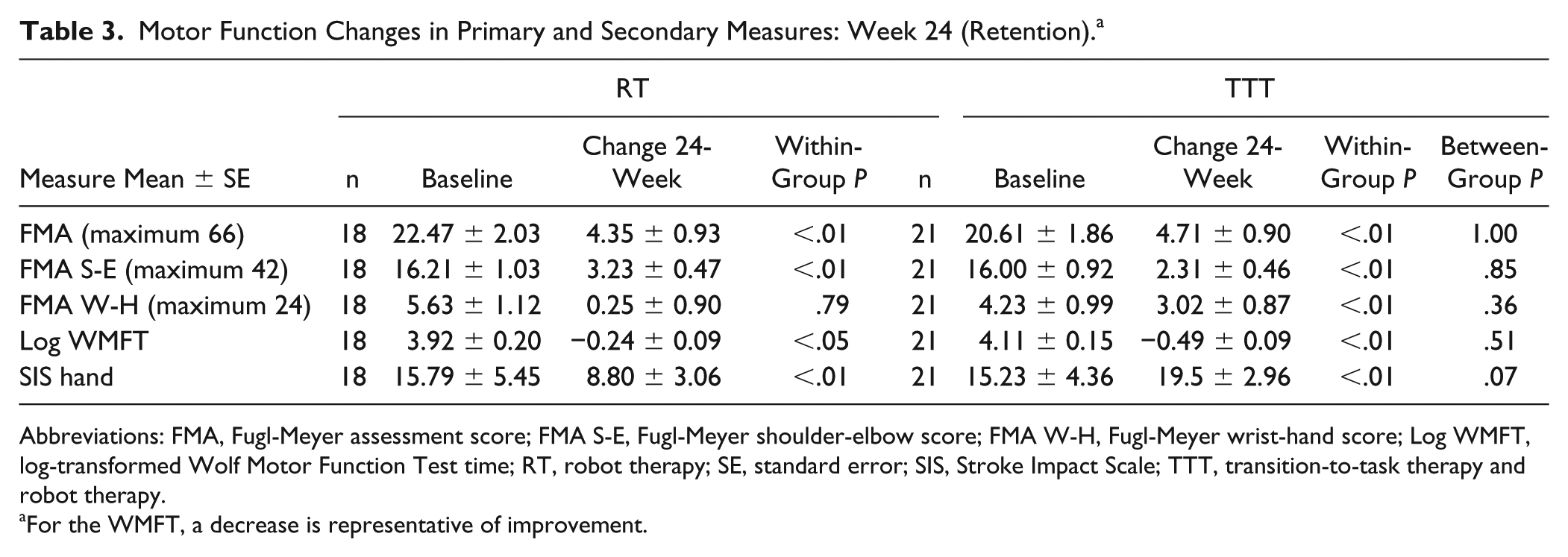
Abbreviations: FMA, Fugl-Meyer assessment score; FMA S-E, Fugl-Meyer shoulder-elbow score; FMA W-H, Fugl-Meyer wrist-hand score; Log WMFT, log-transformed Wolf Motor Function Test time; RT, robot therapy; SE, standard error; SIS, Stroke Impact Scale; TTT, transition-to-task therapy and robot therapy.
For the WMFT, a decrease is representative of improvement.
Secondary Outcome Measures
Significant between-group differences were identified at week 12 on the WMFT and SIS hand (Table 2). The TTT group had a greater 12-week log-transformed WMFT time score change (−0.52 ± 0.06) compared with the RT group (−0.18 ± 0.06); P = .01. This larger negative value represents greater improvement in the WMFT task time. A significant between-group difference was also seen in the 12-week SIS hand score (P = .03). The TTT group SIS hand change was 20.52 ± 2.94 compared with 8.27 ± 3.03 for RT at week 12. A group differentiation was not seen at the 24-week retention visit, although modest significant within-group gains were retained (Table 3).
Subgroup Analysis
A subgroup analysis of between-group differences in FMA change in each baseline FMA stratum was carried out. Between-group differences in FMA change comparing baseline FMA ≥26 and FMA ≤25 were not significant (P = .41 and P = .98, respectively). Longitudinal within-group FMA total and shoulder-elbow changes were similar regardless of baseline stratum or intervention (Figures 3A and 3B). TTT participants with baseline FM ≥26 (n = 6) had significant within-group wrist-hand improvement at week 12 (5.66 ± 1.28) and week 24 (5.50 ± 1.54; Figure 3C).
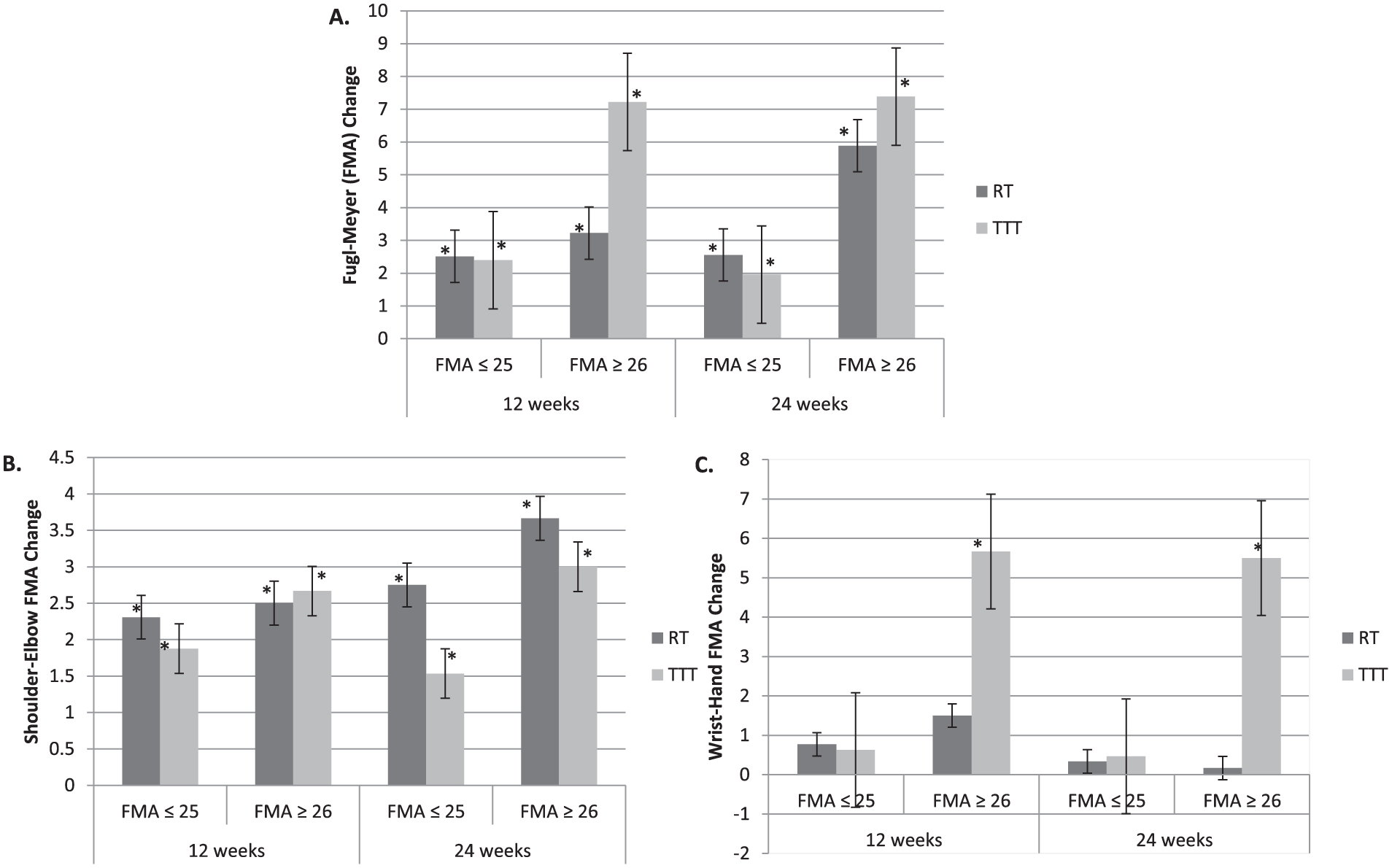
FMA change by baseline FMA total score (A), shoulder-elbow score (B), and wrist-hand score (C). The less impaired (FMA ≥26) had greater overall change, but between-group differences were not significant within the baseline FMA strata. Within-group total and shoulder-elbow improvement occurred regardless of intervention or baseline impairment, and TTT participants with higher baseline FMA scores (n = 6) had a significant within-group wrist-hand score change.a
Training Repetitions
The robot interventions provided a large therapy dosage, and the number of repetitions varied individually per group because time, not repetitions, was held constant. The resulting mean number of robot repetitions per 1-hour training session was greatest for the RT group. Per session, the RT group completed 1132 repetitions in block 1 (wrist robot training), 1539 repetitions in block 2 (shoulder-elbow training), and 1443 repetitions in block 3 (alternating robots). For TTT, per-session repetitions were 724, 1118, and 958 in the same training blocks. We expected to see 25% fewer robot repetitions based on the 15-minute robot training difference; however, the TTT group completed 36% fewer repetitions in block 1, 27% fewer in block 2, and 33% less in block 3. The average number of therapist-assisted task repetitions per session in the TTT group was 41 (across the 2 tasks), which was achieved in a 15-minute window.
Discussion
This clinical trial compared the partial replacement of robotic therapy with task-specific training (TTT) to RT alone in individuals with moderate to severe chronic stroke-related arm disability. We hypothesized that the TTT group’s task training would be more efficacious than RT alone. The FMA results found no significant difference between the 2 robot therapies and did not support the hypothesis. The modest FMA posttraining group gains, however, were consistent with previous chronic stroke robot rehabilitation studies.22,43,44
The premise in human motor recovery that more practice is better45,46 has been called into question by recent clinical stroke trials. Additionally, the key dosage and timing of rehabilitation for a meaningful difference has yet to be defined.47-49 An investigation of early intensive arm therapy by Dromerick et al 50 found an inverse relationship of dose to improvement, and a large multisite subacute stroke trial found no associated benefit of high-dose intensive arm therapy on motor ability. 47 Additionally, no distinct difference was found for varying levels of high-repetition task-specific training in individuals with chronic (≥6 months) UE paresis. 48 This latter study investigated dose-response in individuals with chronic mild to moderate motor disability, where higher doses of arm exercise did not result in better outcomes. Our results extend these findings to individuals with chronic moderate to severe motor disability because we did not find a substantial difference between impairment outcomes regardless of number of robot-assisted therapy repetitions. Our RT treatment intensities greatly exceeded the 300 repetitions per practice session proposed by Birkenmeier et al 38 to improve arm function. However, dosage alone may not be the sole stimulus for recovery because 60 minutes of robot training was no different than 45 minutes.
A strength of our study was the ability to examine robot dosage in more detail based on repetitions within the treatments. The expected 25% reduction in TTT group robot repetitions was larger for all training blocks. Considerably fewer repetitions occurred when training included the wrist robot (blocks 1 and 3). It is not clear why this happened. Group demographics and stroke characteristics were not statistically different; however, 63% of RT participants were dominant hand affected (per self-report) compared with 36% for TTT. The RT group may have had more successful and/or faster wrist movements toward the targets because of past dominant hand use for reaching and fine motor skills. Notably, the reduced RT dosage, both in minutes and in repetitions, did not result in a substantial difference in impairment outcomes.
Currently, there is no consensus on the benefit of RT for real-world arm use. The Cochrane review by Mehrholz et al 24 reported RT benefits for ADLs, whereas a systematic review and meta-analysis by Veerbeek et al 32 suggested no difference in improvement in ADLs compared with usual care or no treatment. The field of UE robotics, however, is rapidly developing, and an increasing number of studies using different robot designs—for example, bilateral, distal, and hybrid training—have shown functional improvements.29,30,51,52 Our novel robot and task training combination resulted in significant motor performance (WMFT) and self-reported hand use (SIS hand) improvement. This suggests a functional benefit for substituting 15 minutes of RT with 15 minutes of task-specific training. A plausible contributor to the benefit may be that the therapist-guided progression was more aggressive than the robot algorithm. Also, the TTT therapist interaction time was not tightly controlled, allowing more opportunities for encouragement and a potential influence on motivation. The robot training was goal directed, random, and included positive feedback, but it did not provide a contextual reaching task. This noncontextual reaching, along with decreased patient-therapist interaction, may have limited improvement to the specific muscle and joints trained. A randomized cross-over trial by Brokaw et al 51 using an exoskeletal robot found distinct robot-mediated improvements and greater gains when RT preceded conventional therapy. The authors proposed that RT stimulated motor and joint range of motion gains, allowing more productive practice during the subsequent conventional therapy.
The intensive and progressive segmental robot-assisted training within our protocol may have acted similarly as a catalyst, priming the motor system for transfer of impairment gains to active use during the therapist-assisted task training. Given the nature of the training, it is not surprising that the SIS hand score favored TTT and the positive impact of a short 15-minute training substitution has clinical implications. Not only was the average of 41 task repetitions greater than the 32 recorded by Lang et al 53 over an hour-long period in conventional therapy, but the TTT activities emphasized and complemented movement patterns delivered by the robot. The TTT group SIS hand gain of 20.5 points reached significance and exceeded the minimal clinically important difference of 17.8 points defined by Lang et al 54 for a meaningful perceived functional benefit. This gain was comparable to the wrist robot and task-specific training by Hung et al, 30 which had a greater distal extremity emphasis and higher intensity (90-100 minutes, 5 d/wk), but an overall shorter duration (4 weeks). The WMFT, also, showed a significant improvement for TTT compared with RT. Additionally, the average WMFT time change (9.4 s) for TTT at week 12 represented a clinically important difference for chronic stroke deficits. 55 This magnitude of change was similar to results from the EXCITE constraint-induced movement trial 12 and the task-oriented multisite ICARE rehabilitation trial. 47 This is an exciting finding because it occurred in a study population that, on average, presented with greater and more chronic impairment. Unlike many other stroke rehabilitation studies, our inclusion criteria did not have requirements for hand movement,12,47,48 and overall, our participants presented with long-standing limited or absent hand function. The robot-assisted protocol provided a high dose of wrist training but did not directly address the hand. Therefore, our results were plausibly limited by the severity of hand disability because participants may have had less motivation and opportunity to explore arm use on their own.
This study provided insight into the delivery of robotic therapy and the extent to which delivery time and content can be altered to have a treatment effect. Although we do not yet know if the 75% robot, 25% task training session divide is the ideal, future research should explore the benefit of technologically generated quantitative measures and different percentages of combined robot-assisted impairment and functional training. Robot kinematics or arm accelerometer measures may provide insight into subtle progressive changes not detected by our current therapist-based measures. In the clinic, we suggest a renewed emphasis in the chronic recovery phase on intensive impairment training combined with therapist-supported task-specific training.
Limitations
Interpretation of our results is specific to the chronic stroke population and outcomes examined. For example, we cannot generalize our findings to the mild or mild-moderate segment of the stroke population or to the subacute stage. The lack of statistical difference across the RT and TTT groups for our primary outcome may be partly attributed to heterogeneity of the stroke deficits. We tried to control for this by stratifying groups based on FMA-defined arm impairment; however, this measure defines the general characteristics of arm movement and is limited in accounting for the variability in task performance between patients. 56 This, in combination with the number of participants per group, lowered the statistical power.
Conclusion
Individuals with stroke who have moderate to severe levels of arm disability can benefit from high-intensity robot-mediated repetitive task practice with or without real-world task-specific training. However, the replacement of part of the robotic training session with nonrobotic therapy had an additional benefit on motor performance (WMFT) and self-reported stroke-affected hand use (SIS) without loss of benefit from less time on the robot.
Footnotes
Acknowledgements
The authors would like to thank our study participants and Patti McCarthy, OTR, and Jaime Lush, BS. This work was supported by Merit Award B6935R from the US Department of Veterans Affairs Rehabilitation Research and Development Service with additional support from the Maryland Exercise and Robotics Center of Excellence, Baltimore VA Medical Center. The contents do not represent the views of the US Department of Veterans Affairs or the US Government.
Authors’ Note
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HIK is a coinventor in several MIT-held patents for robotic therapy. He holds equity positions in 4Motion Robotics, the company that manufactures this type of technology.
Funding
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This work was supported by VA Merit Award B6935R from the United States Department of Veterans Affairs Rehabilitation Research and Development Service.
