Abstract
Background
Vagus nerve stimulation (VNS) combined with rehabilitation is a Food and Drug Administration approved intervention for moderate to severe upper extremity deficits in chronic ischemic stroke patients. Previous studies demonstrated that VNS improves upper extremity motor impairments, using the Fugl Meyer Assessment of Upper Extremity (FMA-UE); however, delineating where these improvements occur, and the role of VNS dosage parameters were not reported.
Objective
This study explored the relationship between dosing (time over which task repetitions were executed and number of VNS stimulations) and changes within proximal and distal components of the FMA-UE.
Methods
Participants underwent VNS implantation, with 1 group receiving VNS paired with rehabilitation (Active VNS) and the other group receiving rehabilitation with sham stimulation (Controls). Both groups received 6 weeks of in-clinic therapy followed by a 90-day at-home, self-rehabilitation program. Participants who completed at least 12 of 18 in-clinic sessions were included in the analyses (n = l06). Pearson correlations and analysis of covariance were used to investigate the relationship between dosing and FMA-UE outcome change along with the effect of covariates including baseline severity, time since stroke, age, and paretic side.
Results
Compared to Controls, active VNS favorably influenced distal function with sustained improvement after the home program. Significant improvements were observed in only distal components (FMdist) at both post day-1 (1.80 points, 95% Cl [0.85, 2.73], P < .001) and post-day 90 (1.62 points, 95% CI [0.45, 2.80], P < .007).
Conclusions
VNS paired with rehabilitation resulted in significant improvements in wrist and hand impairment compared to Controls, despite similar in-clinic dosing across both groups.
NCT03131960
Introduction
Upper limb motor impairment affects up to 80% of stroke survivors acutely and persists in 40% of survivors, resulting in disability with common activities such as reaching, picking up objects, and holding onto objects. 1 Traditional inpatient rehabilitation programs are approximately 2 to 3 weeks 2 and involve a coordinated, interdisciplinary program for 3 hours a day,3,4 focusing on but not limited to regaining upper extremity function as well as improving overall mobility. 5 Within the current traditional inpatient and outpatient rehabilitation programs, patients may not receive sufficient doses of task-specific practice to drive neural changes6,7or significant improvements in functional outcomes. 8 After the initial phase of rehabilitation, only 31% of patients continue outpatient rehabilitation programs, 9 with the rate of clinical recovery progressively slowing between 1- and 3-months post-infarct. 10
Vagus nerve stimulation (VNS) combined with task-specific practice (Active VNS) is a Food and Drug Administration (FDA) approved therapy for improving upper extremity impairment and function in chronic stroke survivors. 11 In an early rodent study, after Active VNS training, naive rats demonstrated significantly greater cortical reorganization in the primary motor cortex compared to rats that did not receive VNS during motor training. The region of reorganization was specific to the tasks that were paired with VNS (ie, proximal or distal). 12 In rats with ischemic stroke lesions, pairing VNS with repetitive forelimb movements restored limb function to pre-lesion levels more than provision of VNS uncoupled from rehabilitation or rehabilitative training in the absence of VNS. 13 Meyers et al, 14 also found that delivering VNS during task training to rats with ischemic stroke that emphasized forelimb supination, significantly improved performance on the task measuring forelimb strength compared to the Control group undergoing training without VNS.
Following these preclinical studies, pilot studies using Active VNS with functional movement tasks yielded favorable improvement in the impaired upper extremities of chronic stroke survivors.15,16 These improvements were sustained for at least 3 years. 17 Subsequently, a pivotal randomized placebo-controlled study (VNS-REHAB) with 108 chronic stroke participants having moderate to severe upper limb impairment was conducted across 19 clinical sites. 1 Participants receiving Active VNS intervention showed significant improvement in impairment (FMA-UE) and function (Wolf Motor Function Test) compared to Controls who received sham stimulation paired with a similar task-specific training protocol. 1 In fact, the collective findings from these studies provided the basis for FDA approval of paired VNS as a treatment to improve upper extremity impairment in chronic, ischemic moderate to severe stroke patients. 11
The primary outcome measure in the pivotal study was the FMA-UE, a multi-dimensional tool that evaluates both proximal and distal impairment. 18 Several studies utilizing FMA-UE assessment as an impairment outcome have analyzed its subcomponents (shoulder, elbow, wrist, and coordination/hand). For example, Hsu et al, 19 revealed task specific motor training (including reaching, grasping, and releasing an object), using mirror therapy resulted in statistically significant improvements in the FMA-UE wrist and coordination components. Another study 20 applied transcranial direct current stimulation and showed significant improvements in shoulder/elbow FMA-UE subscores. Additional work supports the value of applying VNS with occupational therapy. 21 The present study represents one of the first efforts to investigate proximal and distal change separately with the primary outcome measure being changes in the FMA-UE assessment before and after Active VNS.
Moreover, when considering future clinical application of VNS for stroke patients with chronic impairment, clinicians may also wish to know about patient attributes that produce a favorable response to VNS treatment. This concern is important for both the selection and individualization of a treatment plan. Some favorable predictors of chronic stroke recovery include: relative chronicity, non-dominant side affected, better proprioception, faster functional movements, lower grip strength, and genetic composition. 22 A recent post-hoc subgroup analysis of data accumulated in the VNS-REHAB trial, 23 revealed that overall improvements in impairment and function were not ascribed to specific participant attributes including severity, time since stroke, age, paretic side, and stroke location, suggesting the effects of Active VNS are likely to be consistent in a wide range of stroke survivors with moderate to severe upper extremity impairment. 16 The present study expands upon these observations by exploring whether in-clinic dosing parameters may be a predictor of changes in the proximal and distal components of the FMA-UE after 6 weeks of in-clinic therapy (post day-1) and 3 months thereafter (post day 90).
Methods
Study Design and Participants
All potential participants were screened to determine eligibility. 24 The pivotal study was a randomized, triple-blinded, sham-controlled clinical trial. Each participant received an implantable neurostimulator, lead, and electrode through a simple, outpatient procedure. A retrospective analysis was performed using data collected during this device trial completed by participants (n = 108) between October 2017 and September 2019. All participants' records were reviewed, including specific demographic information: age, sex, time since stroke, hand dominance, and paretic upper extremity. Participants who completed at least 12 of the 18 in-clinic sessions were included in this analysis (n = 106) with half of them receiving VNS plus rehabilitation (Active VNS group) and the other half receiving sham VNS plus rehabilitation (Control group). Dosing data from 2 participants were unavailable due to medical complications unrelated to the study and due to unavailability of a data file that was corrupted at the time of this analysis, respectively, and were excluded from this analysis. Baseline participant characteristics are shown in Table 1. Anonymized datasets will be available upon request.
Baseline Demographics and Characteristics of Participants.

Abbreviation: FM-UE, Fugl-Meyer Assessment-Upper Extremity.
For those individuals meeting these criteria and consenting to participate, baseline FMA-UE data were obtained. Immediately following in-clinic sessions, participants were re-evaluated (post-day 1). Following 90 days of home treatment (ie, end of the randomized phase of the study) and evaluation (post-day 90), those participants who were in the Control group were crossed over to receive the Active VNS stimulation protocol. Those participants who had been treated were followed for several months thereafter. The results beyond the post-day 90 assessment will be reported elsewhere.
In-Clinic and Home-Based Task Specific Therapy
The detailed protocol of the rehabilitation therapy has been previously published. 25 Participants received 6 weeks of in-clinic treatment (3 times/week) followed by a home exercise program. During each in-clinic session, participants practiced 6 to 7, high repetition, functional tasks. These tasks included reach and grasp, gross movement, object flipping, simulated eating tasks, inserting objects, opening containers, and patient-specific tasks. The first 6 tasks were standardized and performed sequentially across all participants, while the patient-specific task was individualized and was optional. 25 The patient specific task was not included in our analyses because of the lack of standardization and only 34% of patients (n = 36) performed the selected task for at least half of the total sessions.
In clinic, therapists triggered VNS by pressing a thumb switch connected to a laptop during task-specific movements. For some tasks requiring several phases to complete the movement, VNS was delivered for each change in directionality during these multi-phased movements (eg, opening and closing a jar). These multi-phased tasks included gross movement, simulated eating, and opening containers tasks, which were paired with 4, 3, and 2 stimulations, respectively. The Active VNS group received stimulations of 0.8 mA, 100 µs, 30 Hz pulses, each lasting 0.5 second. The Control group underwent similar task specific training but received sham stimulations with each sham pulse (0 mA) being recorded. 24 The timestamp for each stimulation was recorded within the implantable pulse generator and was downloaded at each assessment visit. After in-clinic therapy, participants self-activated real or sham stimulation for 1 or more 30-minute, therapist-prescribed home exercise sessions (1/2 second VNS duration every 10 seconds). The exercises involved 3 task categories: gross movement, hand/wrist, and finger dexterity. The number of stimulations was recorded and downloaded at each site at the end of the post-90 period. Therapists were blinded to group assignment so that they would believe that rehabilitation dosing was the same across all participants.
Dosage Determination
The stimulation timestamp records were used to calculate task duration and the number of stimulations delivered during each task. Total task duration was calculated by summing each of the task times for a given session (active or sham) and averaged over the total number of sessions. The time between starting a task (button selection) and first stimulation and the time between the last stimulation and ending the final task were excluded from the calculation. Number of stimulations for each of the tasks was also summed and averaged across all sessions. This approach provided the most accurate clinical indication for dosage in the absence of a wearable sensor that could trigger the stimulator with each phase of task requiring multi-directional kinematics.
FMA-UE Components
The FMA-UE total score ranges from 0 to 66 with 66 suggesting no discernable impairment. The score is categorized into 4 primary components each with specific point values: FM-A: shoulder/elbow/forearm, maximum = 36; FM-B: wrist, maximum = l0; FM-C: hand, maximum = 14; FM-D: coordination/speed: maximum = 6. FMA-UE components A (proximal upper extremity and reflex testing) and D (coordination and speed) were combined to represent a logical grouping for proximal impairment (FMprox, total proximal score 42) while components B (wrist) and C (hand) were combined to represent distal impairment (FMdist, total distal score 24). For this analysis, change in proximal and distal components of FMA-UE were the main outcome variables. To study predictors toward improvement for proximal or distal impairment, we explored the association between dosing parameters and change in these components at post-day 1 and post-day 90.
Statistical Analysis
Independent sample t-tests were used to compare between-group change in FMA-UE distal and proximal subscores at post-day-1 and post-day 90 with significance level of .05. Pearson’s correlation coefficient was used to determine the association between dosing parameters and change in FMA-UE proximal and distal scores. Analysis of covariance (ANCOVA) based on general linear models, with the proximal and distal FMA-UE change at post day-1 and at post day-90 as dependent variables, was used to determine the extent to which dosing, baseline severity, baseline demographics, or their interactions impacted outcome differences between groups.
Results
During in-clinic therapy sessions, participants in both the VNS and Control groups received comparable numbers of pulses (real or sham) per session (average stimulations: 406.5 ± 90.1 vs 404.1 ± 78.1 and comparable average time spent on practicing the tasks: 76.2 ± 12.10 vs 75.5 ± 13.0 minutes). The number of stimulations, average task time, and time for each of the 6 tasks was not significantly different between groups (Table 2). These results suggest that standardizing task-specific training across participants allowed for similarly delivered dosing parameters and were not significantly different between groups.
In-clinic Dosing Parameters Including Average Time in Minutes and Average Number of Stimulations for Each Task.
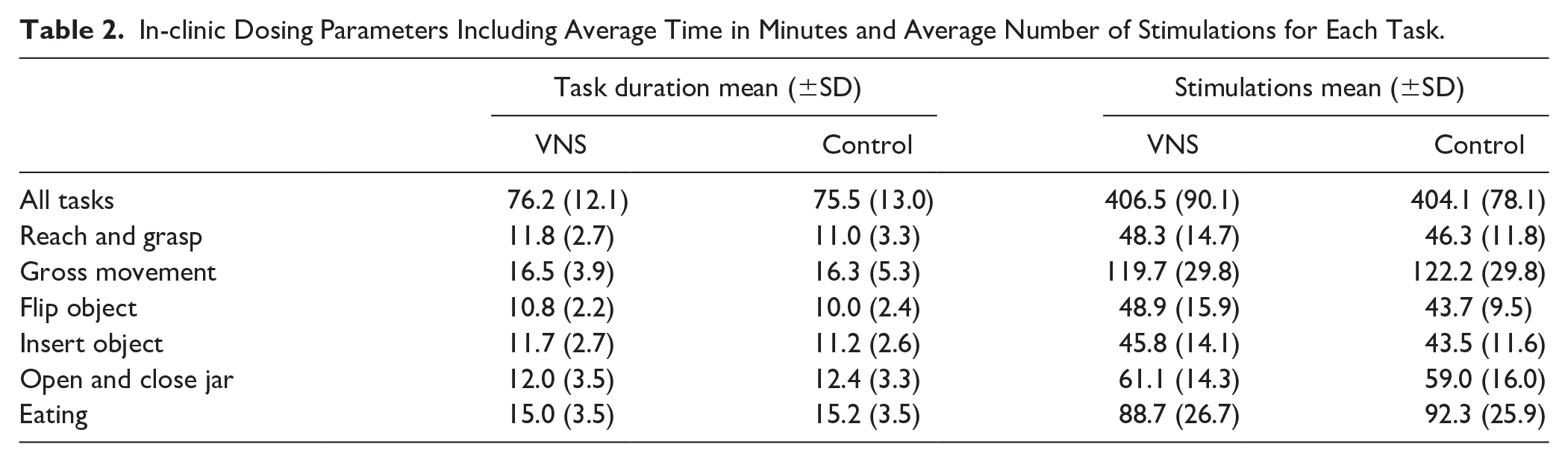
For in home sessions, the number of available days to undertake the therapy was not significantly different between the 2 groups (90.4 ± 6.4 days vs 88 ± 15.6 days, P = .31). Compliance was expressed as a percentage of device use per number of days. Participants used the device approximately every other day (ie, 50% of the time) and the difference was not significantly different between VNS and Controls (57.6 ± 28.2 vs 48.6 ± 34.1, P = 0.16).
As noted in the pivotal VNS-REHAB study, 24 the overall FMA-UE difference was significant between groups at post-1 (5.0 ± 4.4 vs 2.4 ± 3.8, P = .0014) and post-90 5.8 ± 6.0 vs 2.8 ± 5.2, P = .0077). However, neither differences for specific components of the FMA-UE nor the relationship of these changes to intensity of dosing, including dosing across specific tasks, were reported. After Active VNS, significant improvements were observed in distal components (FMdist) compared to Controls at both post day-1 (independent sample t-test, between group difference:1.80, 95% CI [0.85, 2.73], P = .001) and post-day 90 (1.62, 95% CI [0.45, 2.80], P = .007). There were no significant differences observed for the change in proximal component (FMprox; Table 3).
Changes in Proximal and Distal FMA-UE at Post Day-1 and Post Day-90.
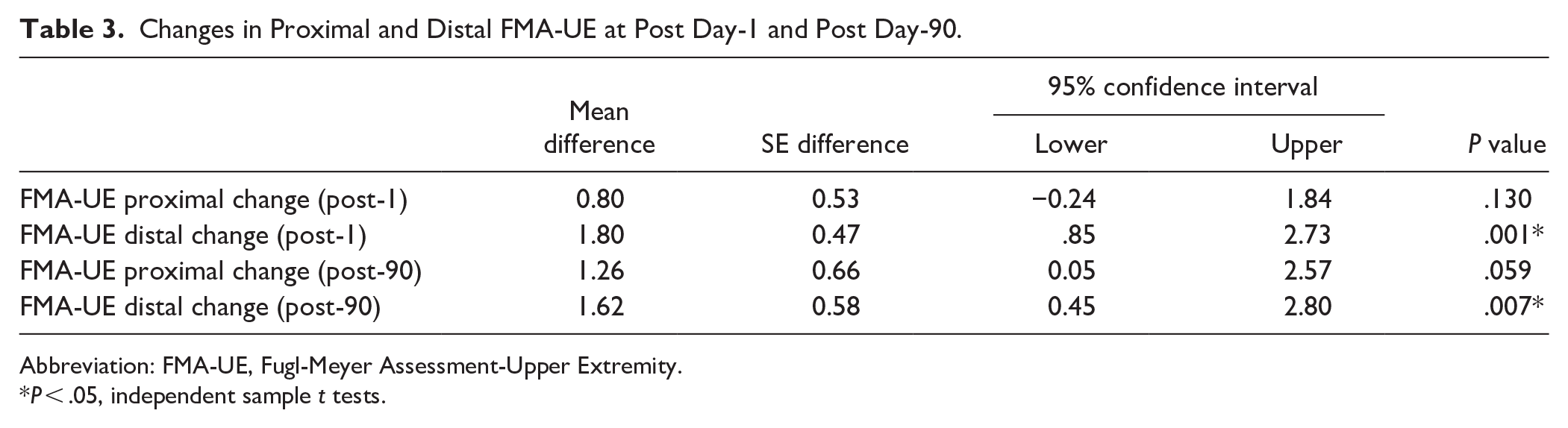
Abbreviation: FMA-UE, Fugl-Meyer Assessment-Upper Extremity.
P < .05, independent sample t tests.
At post-day 90, FMdist showed a weak but significant correlation with number of stimulations per session for the VNS group (r = .30, P = .02), but not for control participants (r = .17, P = .21; Figure 1). The correlation was not significant at post-day 1 for either group. Furthermore, there was no significant correlation between task time and change in proximal or distal FMA-UE scores for Active VNS or Control group.
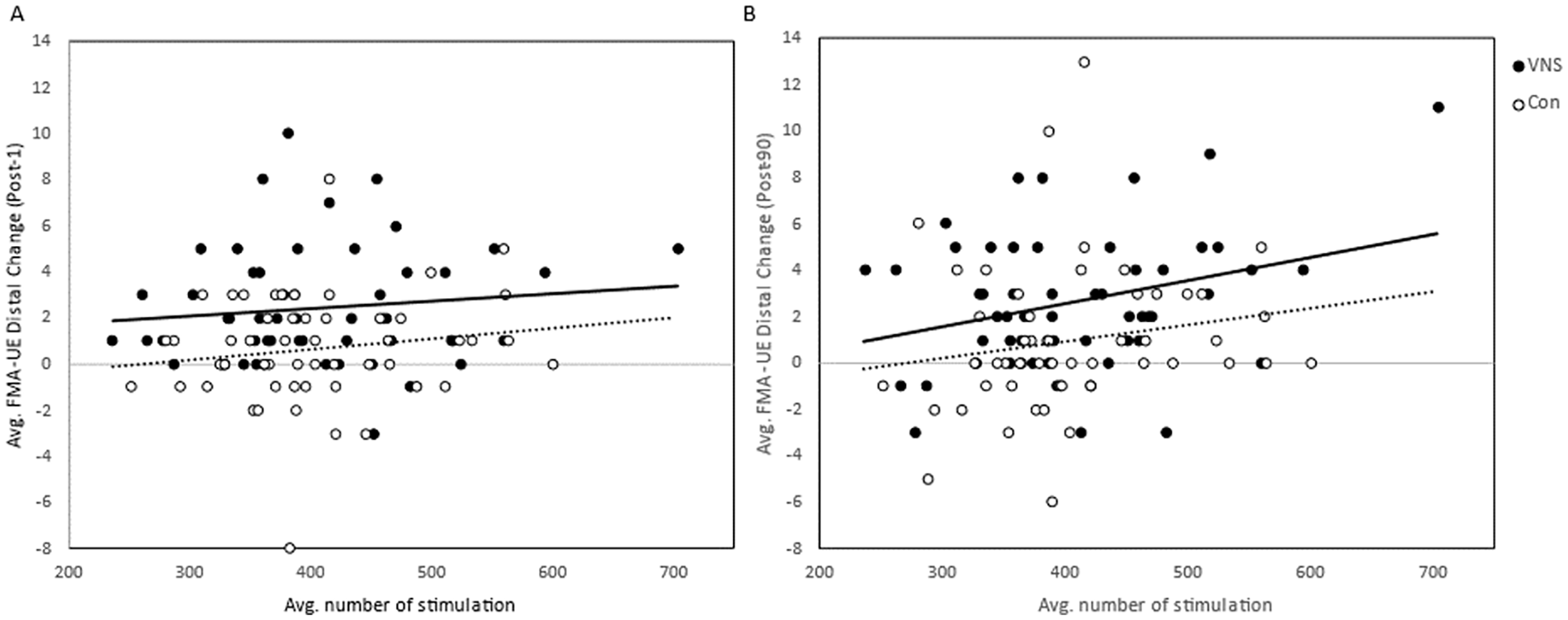
Pearson correlation showed a positive association between average number of stimulations per in-clinic session and change in FMA-UE distal score within VNS group (*P < .01, r = .36) at post-day 90. No correlation observed in the Control group.
We further determined if a significant association between number of stimulations for individual tasks and change in FMA-UE components was observed at post-day 1 and post-day 90. As seen in Table 4, there was a significant correlation between stimulations for the Reach and Grasp (RG) task and FMdist change scores at post day 1 in both groups (P < .05). The correlation continued to remain significant at post-day 90 for the VNS group (P < .001) but not Controls (P > .05). At post-day 90, both groups showed a significant correlation for flipping objects (P < .05) while the VNS group demonstrated a significant relationship for gross movements (P < .05). The control group showed this relationship for inserting objects at post-day 1 (P < .001) and at post-day 90 (P < .01).
Correlation Between Stimulations per Specific Task Versus Change in Distal FMA-UE Scores at Post Day1 and Post Day 90 for VNS and Control groups.
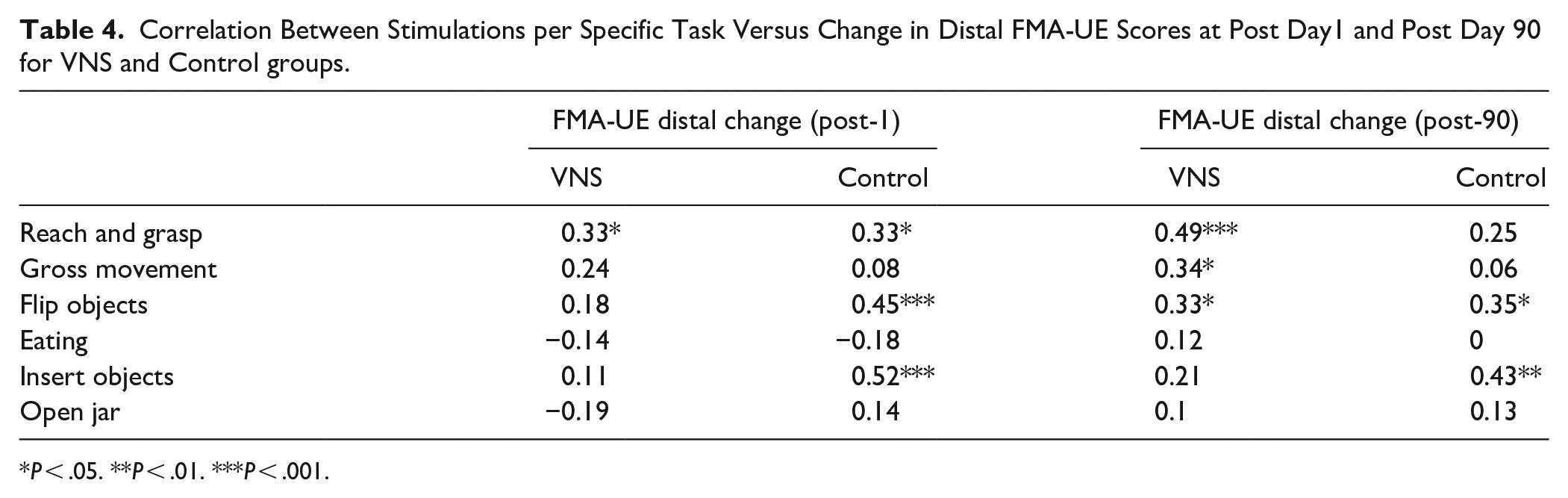
P < .05. **P < .01. ***P < .001.
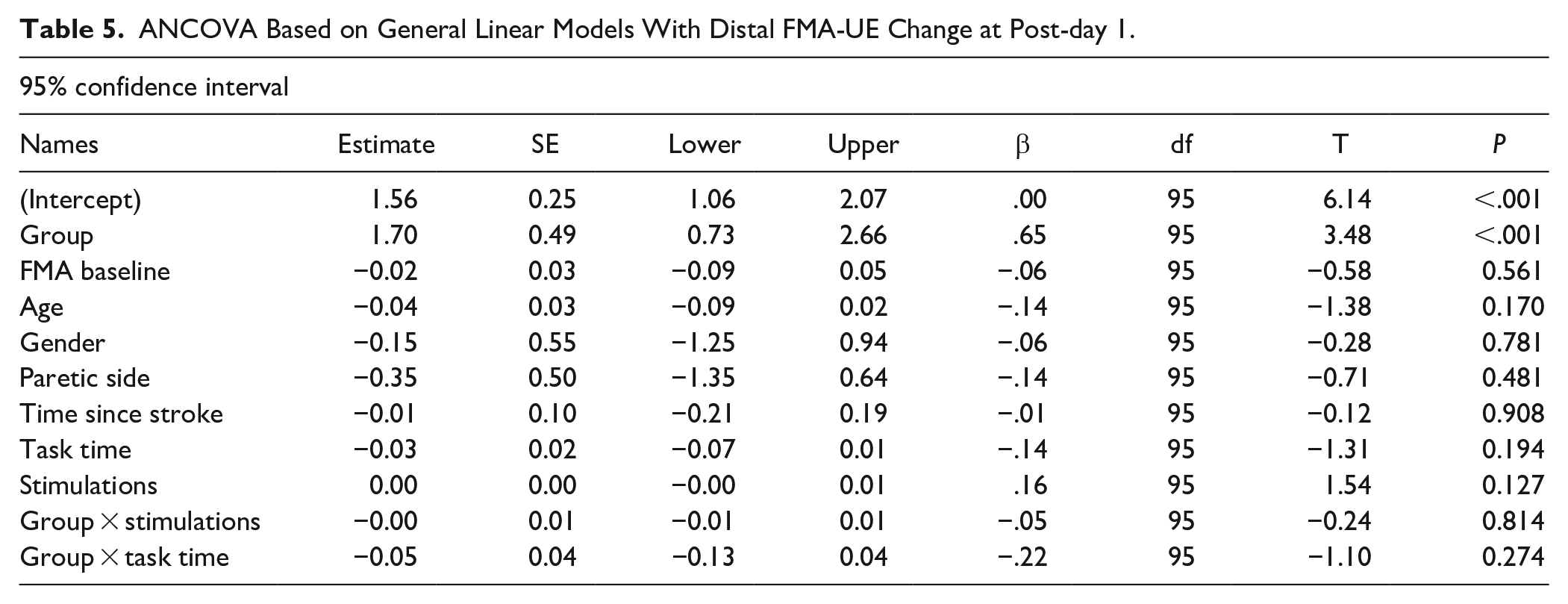
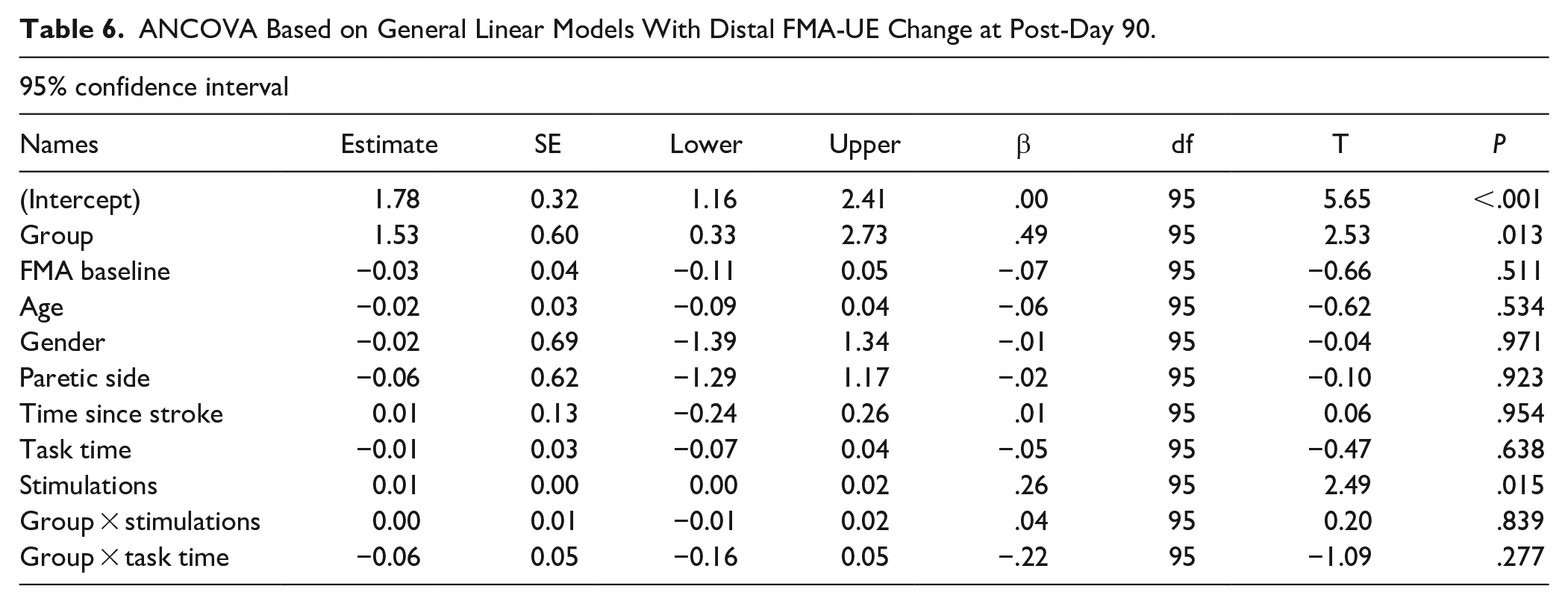
The number of stimulations, task time, age, baseline FMA-UE score, paretic side and time since stroke were not significant predictors of the change in FMA-UE score except for the number of stimulations at post-day 90 (β = .26, P = .015; Tables 5 and 6). However, there was no interaction between number of stimulations and group at this timepoint (P = .8). These results suggest that the significant differences in distal FMdist change between the active VNS and control groups were not explained by the number of stimulations, task time and covariates including demographics, paretic side, baseline severity, or time since stroke.
ANCOVA Based on General Linear Models With Distal FMA-UE Change at Post-day 1.
ANCOVA Based on General Linear Models With Distal FMA-UE Change at Post-Day 90.
Discussion
This retrospective analysis represents the first effort to examine the subsections of the FMA-UE when VNS was paired with rehabilitation for chronic stroke and the relationship of these subsections to dosing for specific tasks. The data suggest improvements were most prominent in distal UE impairment at post-day 90 in the VNS group and were correlated with higher in-clinic stimulation dosage. While there appears to be a main effect of higher dosage for both VNS and Control group at Post-day 90, no interaction between groups was apparent, thus reducing the probability of improved scores for the VNS group being primarily attributable to stimulation. The large variability within the data might partially explain this observation and invites the need for a larger sample size before a more definitive explanation can be discerned.
Improvement in distal UE impairment in both groups was significantly correlated with higher number of stimulations in certain tasks, including Reach and Grasp and Flipping Object, which could be expected, perhaps attributable to the high repetitions for these tasks in both groups. For Gross Movement, one would expect that this task would be correlated with FMprox because the task is more focused on proximal movement. Surprisingly, FMdist showed significant improvement with the Gross Movement task at both post-day 1 and post-day 90 after Active VNS. Perhaps the task paired with VNS might have favorably influenced both proximal and distal limb components, but more so for the latter. This notion is reasonable considering that gross tasks, such as reaching and grasping a hat to move it to a higher shelf level requires engagement of both proximal and distal upper extremity activation. These observations could possibly be accounted for by participants’ functional or anatomical deficits, 26 learning context, 27 saliency of tasks and the variability of skill acquisition among participants. 28 A more detailed analysis of imaging data, inclusion of kinematic assessments during execution of the individual task performances, or variations in behaviors including cognition and compliance, could provide greater clarity in subsequent studies. The Control group displayed a significant correlation between improvement in distal function and both Inserting Object and Flipping Object tasks; however, one cannot dismiss the possibility that this finding, as in other studies, might be influenced by outliers. The fact that these occasions were distributed across different participants, mitigates the importance of this observation.
Based on the original study protocol, there are several factors contributing to overall variability during in-clinic therapy including number of stimulation-movement pairings, number and length of sessions, degree of upper extremity impairment, patient goals, and functional deficits. 27 Our ability to delineate task characteristics that resulted in some tasks being more beneficial than others remains uncertain. Relative task difficulty compared to level of impairment, sequencing of tasks, or personalized task salience could contribute to this uncertainty. Further investigation is necessary to determine the degree to which these or other factors influence the beneficial effect of each task.
The Fugl-Meyer Assessment is a commonly used performance-based, predictor measure of initial motor capability after stroke. 29 However, in our analyses, baseline FMA-UE did not predict FM-UE total, proximal, and distal change scores at post-day 1 nor at post-day 90 in either group, indicating that baseline impairment by itself may not be an optimal predictor for characterizing responders to this Active VNS intervention. Participants with moderate-to severe arm impairment, defined as an FMA-UE score of 20 to 50, were included in this study. As with most scales, the FMA-UE when used as a measurement of recovery for patients with mild motor impairment, is limited by a ceiling effect. 30 Additionally, due to its ordinal nature, measures of improvement can vary significantly across the spectrum with the same number of point changes along the breadth of the FMA-UE representing different levels of clinical improvement.
Based on the current analysis, factors such as age, sex, stroke chronicity, and hand concordance/discordance do not appear to influence the impact of VNS on distal UE motor improvement at post-day 90. Results from rodent studies have shown that the magnitude of recovery observed in aged rats receiving VNS was comparable to that reported in previous studies using young rats receiving the same intervention.31,32 Our results support current findings that advanced age does not impede VNS-task training improvements in post-stroke recovery. 23 While the triple-blinded, pivotal study was well designed and rigorous, the study 24 posed some limitations, including disproportionate gender distribution, and constrained eligibility criteria. For example, individuals with very severe deficits (ie, severe sensory loss and severe spasticity) were not included in the study.
Preclinical and clinical studies have consistently demonstrated that changes in brain reorganization and improvement in motor outcomes operate across a narrow range of intensities around 0.8 mA. The extent to which optimal stimulation drives changes across all stroke participants for this study is unclear, especially when considering the variability in clinical attributes and other comorbidities among our participants. Additional factors that can directly impact neuromodulatory function including other neurological deficits or concomitant use of pharmacotherapeutic agents 33 require further consideration. Evaluation of the clinical effectiveness of Active VNS treatment in diverse stroke populations should be assessed, including the determination of optimal stimulation parameters (ie, frequency and amplitude) with respect to specific patient subgroups. Future studies may consider personalization of stimulation parameters in specific stroke populations. Preclinical studies31,34 suggest that the timing and intensity of stimulation parameters may influence the magnitude of VNS-dependent plasticity. These observations imply that manipulation of VNS timing may play a role in enhancing plasticity and recovery. 33 The relationship of lesion size and location to outcomes should also be considered.
Future endeavors may also address the possibility of utilizing paired VNS therapy during telehealth visits and assessing the efficacy of the combination of telehealth and home-based VNS program by interfacing biosensors to better define a criterion to initiate movement-triggered VNS delivery. This interface may improve the specificity of linking task initiation to VNS within the home while not requiring manual triggering by a therapist in the clinic, although whether precise timing to movement is necessary at behavioral timescales of motor learning it remains to be determined. The fact that, on average, participants continued to improve, particularly in FMdist, when either synchronous movement or any movement could not be determined, tracking movement using wearable sensors to detect when movement occurs relative to provision of stimulation pulse may provide a more comprehensive timestamp of movement overlap with VNS. These sensors could also trigger the stimulator based upon a predetermined magnitude of distal limb motion.
Footnotes
Acknowledgements
We would like to thank the MicroTransponder team for their invaluable assistance with data analyses, our participants for their contribution to this research project, and Susan Murphy for administrative assistance.
Author Contributions
Shiyu Lin: Data curation; Investigation; Writing—original draft. Chelsea Rodriguez: Investigation; Project administration; Writing—original draft. Steven Wolf: Conceptualization; Formal analysis; Investigation; Methodology; Project administration; Writing—original draft; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This narrative was generated by the Emory participating site under grant MT-ST-03 from MicroTransponder, Inc.
