Abstract
Background. Recent evidence suggested that Action Observation Therapy (AOT), based on observation of actions followed by immediate reproduction, could be a useful rehabilitative strategy for promoting functional recovery of children affected by unilateral cerebral palsy (UCP). AOT most likely exploits properties of the parieto-premotor mirror neuron system (MNS). This is more intensely activated when participants observe actions belonging to their own motor repertoire. Objective. The aim of the present study was to investigate the issue of whether MNS of UCP children is better activated by actions performed by a paretic hand rather than a healthy one. Methods. Using functional magnetic resonance imaging, we assessed brain activation in a homogeneous group of 10 right UCP children compared with that of 10 right-handed typically developing (TD) children, during observation of grasping actions performed by a healthy or a paretic hand. Results. The results revealed a significant activation within the MNS in both UCP and TD children, more lateralized to the left hemisphere in the TD group. Most important, region of interest (ROI) analysis on parietal and premotor regions showed that, in UCP, the MNS was more strongly activated by observation of actions performed by the paretic hand, a motor model more similar to the observer’s motor repertoire. Conclusions. This study shows that children affected by spastic UCP exhibit enhanced activation of the MNS during observation of goal-directed actions performed by a pathological model with respect to a healthy one.
Introduction
Cerebral palsy (CP) refers to a group of permanent disorders of the development of movement and posture, derived from nonprogressive disturbances occurring in the developing fetal or infant brain. 1 CP syndrome is the most common childhood motor disability ranging from 1.5 to 2.5 per 1.000 live births. 2 Children with unilateral cerebral palsy (UCP), in which mostly one side of the body is involved, represent 39% of the general CP population.3,4 UCP can be attributed to perinatal ischemic stroke or, in premature infants, to white-matter damage producing unilateral porencephalic cavities (or cysts).1,5 In UCP, the upper limb is generally more affected than the lower one, 6 and unimanual activities are normally performed by the less affected upper limb. Daily life activities, which are prevalently bimanual, could be severely impaired. 7
Several models of intervention are currently available to improve upper limb function in UCP patient, such as botulinum toxin A (BoNT-A), constraint-induced movement therapy (CIMT), hand-arm intensive bimanual training (HABIT), occupational therapy (OT), and neurodevelopmental treatment (NDT). A recent meta-analysis 8 highlights the higher efficacy of interventions based on motor skill learning theories requiring the child to develop a strategy to solve motor problems (eg, intensive activity-based, goal-directed, bimanual interventions). The elements playing the most important role appear to be the intensive structured task repetition, incremental level of difficulty and activity-based patient’s motivation.
A relatively new rehabilitative approach, quite different from those based on problem solving, exploits the systematic observation of actions followed by their imitation (Action Observation Therapy [AOT]). 9 In fact, this latter approach, differently from those described above, is driven by an external model. Although this intervention is not highly task demanding for UCP children, it has the advantage to rely on an automatic learning mechanism and can also be easily transferred to a home-based program.
The neural model for this therapy is the mirror neuron system (MNS), originally discovered in the monkey premotor and parietal cortex, formed by visuomotor neurons which fire both when a monkey performs a goal-directed motor act or when it simply observes the same or a similar motor act.10-14 A comparable MNS has been identified also in humans using several electrophysiological and neuroimaging techniques.15,16 Its 2 main nodes are the inferior parietal lobule (IPL) and the ventral premotor cortex (PMv), plus the caudal part of the inferior frontal gyrus (IFG). 17 Some studies also report additional involvement of other areas, forming the so-called Action Observation Network (AON), such as superior parietal lobule (SPL) and dorsal premotor cortex (PMd) for observation of reaching movements, 18 and primary and high order somatosensory areas, probably related to the sensory consequences of the observed actions. 19
It has been proposed that the MNS transforms visual representations of observed motor acts into the corresponding motor representations, allowing observers to immediately recognize motor acts belonging to their own motor repertoire, 20 including also motor skills resulting from long-lasting practice. Indeed, several studies21-24 indicate that the MNS is highly activated during observation of actions already embodied in the observers’ motor repertoire. In line with this idea, in the past 10 years an increasing number of AOT applications have been reported in adults and children with CP, using paradigms combining action observation with subsequent execution of observed actions.25-30
In AOT of UCP children it is possible that observation elicits, in the observer, a marked propensity to preserve task proficiency by selecting movements that enable attainment of action goal, regardless of kinematic resemblance with the observed model. This could be because the complexity of the model offered during observation of an action performed by a healthy hand is not appropriate for imitation, so that children decide to copy the final outcome of the action rather than the process used to achieve it. Thus, UCP children might have a reduced activation of the MNS during observation of actions performed by healthy subjects but increased activation during observation of the same actions performed by a child with comparable coping strategies. Hence, we performed a functional magnetic resonance imaging (fMRI) study on UCP and typically developing (TD) children, in order to investigate the neural correlates of observation of actions performed by agents with 2 different levels of motor skills, namely a UCP child with a moderate degree of hand impairment and an agent with a healthy hand. We hypothesized that in UCP children the observation of actions performed with a paretic hand should be more effective in producing MNS activation than the observation of a healthy hand.
Materials and Methods
Participants
Ten children (see Table 1) with unilateral brain lesion and spastic UCP (7 males and 3 females; range 9-14 years; M = 11.2; SD = 2.09) were selected starting from a large sample of patients (N > 100) in the hemiplegic children database of IRCCS S. Maria Nuova Hospital (Reggio Emilia, Italy) according to the following inclusion criteria: (1) confirmed diagnosis of right spastic UCP according to definition (MRI and clinical history); (2) age 9 to 14 years at time of recruitment; (3) mild or moderate upper limb disability, that is, active use of affected upper limb ranging from poor active assisted use to spontaneous use, according to House Functional Classification (HFC) 31 system with grades between 4 and 5 (corresponding to synergic hand of Ferrari and Cioni’s Kinematic Hand Classification 32 (see Supplementary Table 1, available online); (4) no cognitive, visual, or auditory impairments; (5) no history of seizures or seizures well controlled by therapy; and (6) sufficient cooperation in performing 45-minute fMRI session.
Demographic Data, Clinical Features, Radiological Findings, and Functional Classification in Cases Group.
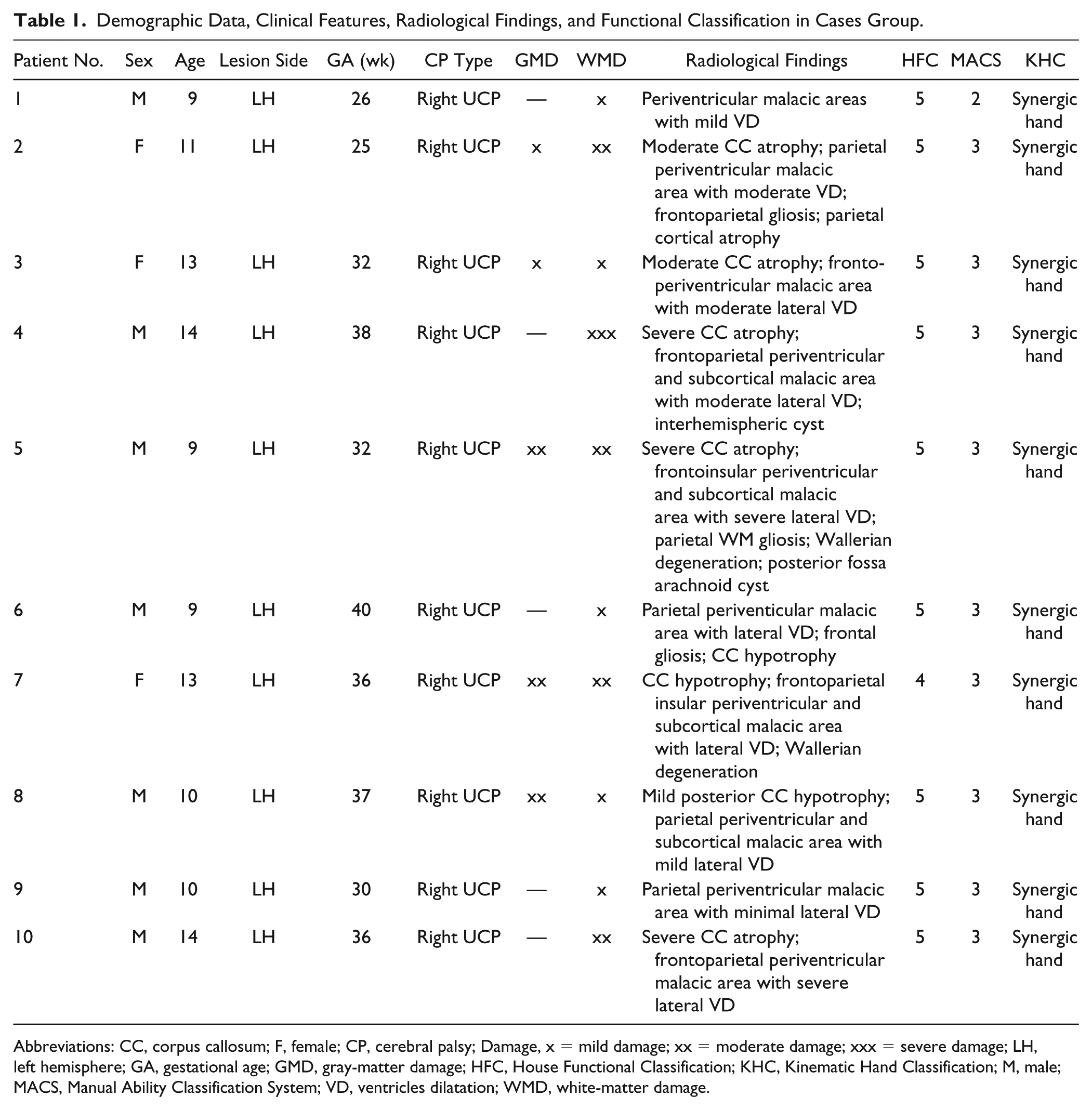
Abbreviations: CC, corpus callosum; F, female; CP, cerebral palsy; Damage, x = mild damage; xx = moderate damage; xxx = severe damage; LH, left hemisphere; GA, gestational age; GMD, gray-matter damage; HFC, House Functional Classification; KHC, Kinematic Hand Classification; M, male; MACS, Manual Ability Classification System; VD, ventricles dilatation; WMD, white-matter damage.
Children were excluded if they (1) received BoNT-A injections or had upper limb surgery within 6 months prior to testing; (2) were unsuitable for 3-T magnetic resonance system due to metal implants, shunts, and so on; (3) were ranked <4 according to HFC; and (4) had moderate or severe muscle spasticity and/or contracture 33 (Modified Ashworth Scale (MAS) score >2) so requiring spasticity management or orthoses. Ten right-handed TD children were enrolled as control group (5 males and 5 females; range 9-14 years; M = 11.1; SD = 2.05). We started from the assumption that the best model for the AOT in right UCP patients should be a right-hand model, therefore we used a paradigm based on observation of a pathological right-hand model and, for comparison, a healthy right-hand model. As a consequence, TD children group was chosen homogeneous for handedness lateralization, in order to avoid problems in interpreting the results obtained from a mixed group of TD children. All TD and UCP children attended regular public school and could consistently follow instructions. Children who met all inclusion criteria were invited to participate in this fMRI study and written informed consent was obtained from children and/or parents prior to recruitment, according to the Declaration of Helsinki. The experimental procedure was approved by the Local Ethics Committee (University of Parma).
Clinical Assessment
UCP children participating in this study were assessed using the HFC,31,34 Kinematic Hand Classification (KHC) system, 32 and Manual Ability Classification System (MACS). 35 HFC consists of 9 grades ranging from a hand that is not used at all (grade 0) to one that is used spontaneously and independently from the other hand (grade 8). The HFC has been reported to have an excellent interrater (intraclass correlation coefficient, ICC = 0.92) and intrarater reliability (ICC = 0.94). 34 Ferrari and Cioni’s Kinematic Classification describes five patterns of manipulation in hemiplegic children by analyzing hand kinematic profile and functional use. 32 According to it, all selected patients had a synergic hand (Supplementary Table 1). The MACS consists of 5 levels and classifies how children with CP aged 4 to 18 years use their hands when handling objects in daily activities. 35 Individual scores for these classification scales are reported in Table 1.
Experimental Design, Stimuli, and Tasks
Action Observation Task
A set of 48 videos showing grasping actions executed by the right hand, each lasting 4 seconds, was used in this study. These actions consisted of grasping a small object (a sphere, a cube, a cylinder) and putting it into 1 of 2 square boxes (size 6 cm × 6 cm), placed on the left and the right sides with respect to the object. An example of the stimuli used in the action observation task is shown in Figure 1A and Supplementary Videos 1 to 4. The object color was red, yellow, green, or blue. All videos were depicted in a first-person perspective, as if the observer was performing the action. A total number of 48 experimental video stimuli (3 objects × 4 colors × 2 object positionings × 2 conditions) were presented in the action observation task. There were 2 video conditions: (1) actions performed by an actor without any motor impairment (Action Observation Healthy Hand—AO Healthy Hand) and (2) actions performed by a UCP patient with a moderate level of hand impairment, corresponding to synergic hand of Kinematic Hand Classification (Action Observation Paretic Hand—AO Paretic Hand). All actions were performed with the same visual context, balanced for luminance, color, and amount of visual information.
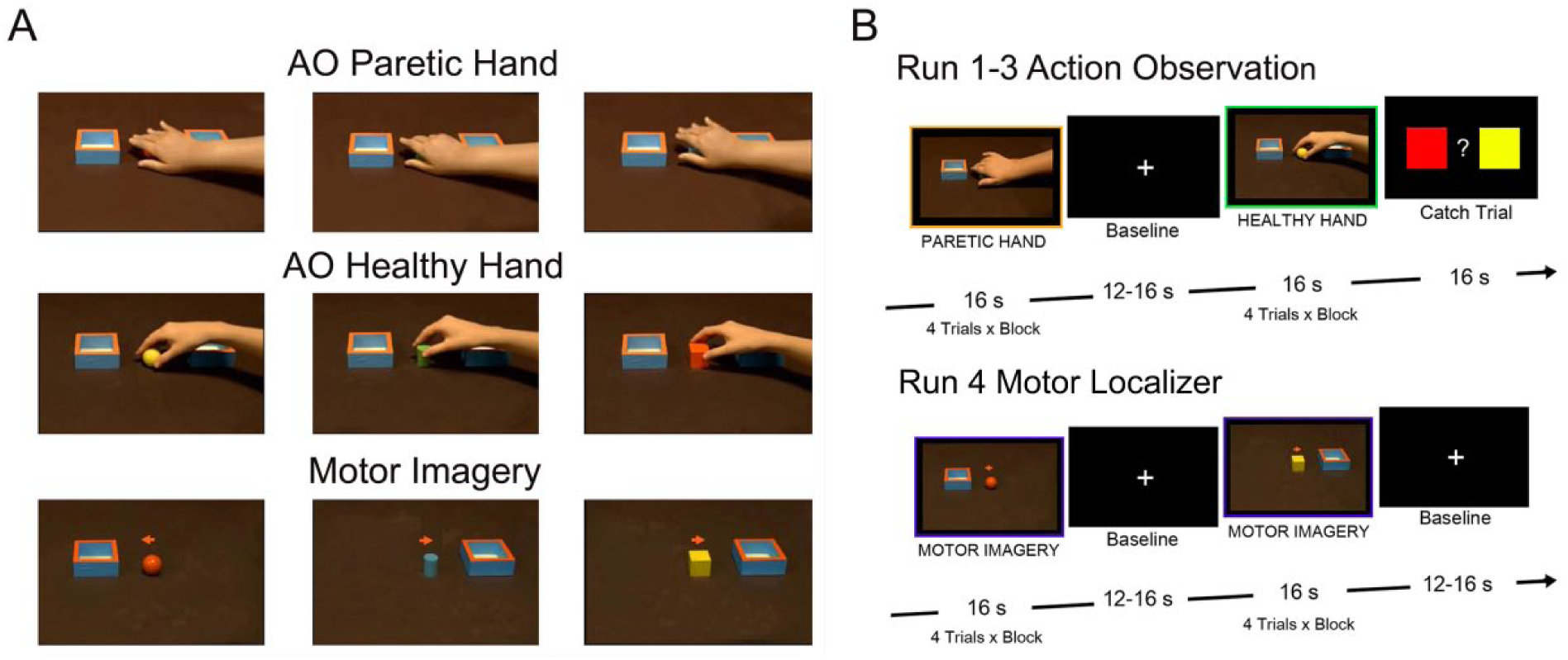
Experimental stimuli and task design. (A) In the action observation runs, participants observed videos showing actions performed by a paretic (AO Paretic Hand) or an healthy hand (AO Healthy Hand), from a first person perspective; actions consisted in reaching-grasping an object (a sphere, a cube, a cylinder) and putting it into 1 of 2 boxes. During the motor localizer, participants were asked to observe a video showing an object and a box on a table and simultaneously imagine themselves grasping the object with the right hand and placing it into the box. (B) Action observation task was presented in 3 functional runs, each made up of 16-second independent blocks, consisting of 4 randomly presented videos of the same condition. Each block was interleaved by a rest period (12-16 seconds). In 25% of action observation blocks, participants had to indicate the color of the presented object or the box (left or right) in which the object was placed. The motor localizer was composed by 8 blocks, each formed by 4 trials, lasting 4 seconds.
The action observation task was presented in 3 functional runs, in which both the experimental conditions (AO Paretic Hand, AO Healthy Hand) were presented to UCP and TD participants. Stimuli were presented in independent blocks (see Figure 1B), lasting 16 seconds each and constituted by 4 videos (4 seconds each) of the same condition, randomly selected from the initial set. Each run was arranged to include a total number of 12 blocks, 6 blocks for AO Paretic Hand and 6 blocks for AO Healthy Hand. Blocks of stimuli were alternated by a rest period lasting 12 to 16 seconds, during which no video was presented. The order of presentation of the blocks was random for each subject. A similar paradigm has been used in previous action observation studies in adults and children. 36 In order to control participant attention to the task, in 25% of the blocks they had to provide an explicit response (catch-trials) concerning color of the observed object, or position (left/right box) in which it was placed in the last video clip, response unrelated to the motor content of presented video. This response was provided by pressing a 2-choice button on a response pad placed inside the scanner by the less-affected hand.
Motor Localizer
In order to identify posterior parietal and premotor areas involved in action execution that usually largely overlap with those involved in action observation, participants performed a motor localizer at the end of the scanning session. Since executing a real action inside the scanner was not feasible for UCP patients, due to excessive head motion and difficulty in minimizing mirror movements in the less affected hand, during the localizer, participants were required to perform an explicit motor imagery task. 37 This paradigm was employed basing on previous evidence showing that motor imagery is able to activate neural structures involved in action execution,18,38 including also the main nodes of the MNS.
Participants were presented with videos showing an object (sphere, cube, or cylinder) and a box (6 cm × 6 cm) on a table (Figure 1B). They were instructed to observe the presented setting, then a cue (a little arrow appearing in the central part of the screen) asked them to imagine performing the action with their right hand (eg, imagine grasping the object and placing it in the box located in the position cued by the arrow). An example of the stimuli used in the localizer is shown in the Figure 1A. A total of 24 experimental video stimuli (3 objects × 4 colors × 2 box location) were presented in 8 blocks of the localizer. Each block was formed by 4 trials, each lasting 4 seconds.
Magnetic Resonance Imaging Data Acquisition
fMRI Data Processing and Statistical Analysis
Data analysis was performed with SPM12 (Wellcome Department of Imaging Neuroscience, University College, London, UK; http://www.fil.ion.ucl.ac.uk/spm). To assess the general activation pattern in UCP children and controls, and considering that for clinical fMRI studies the GLM group analysis is not always relevant due to the lesions extension, we computed consistency activation maps using a voxel-based overlap method19,39 (see also Supplementary Methods for more detailed description of contrasts used and statistical analysis).
In order to investigate possible different responses to actions performed with a paretic or healthy hand (motor experience effect), we performed a region of interest (ROI) analysis confined to 2 important MNS areas, premotor cortex (PMC) and posterior parietal cortex (PPC). Single-subject ROIs were localized using the local maxima of activated clusters in PMC and PPC regions during the performance of localizer. The averaged hemodynamic response has been analyzed using 2 different repeated-measures analyses of variance (rmANOVA). The first rmANOVA was performed on mean signal change in PPC ROIs, while the second one on PMC ROIs, using Group (UCP Children, TD Children) as between-subject factor and Condition (AO Paretic Hand, AO Healthy Hand) as repeated-measure factor. Post-hoc comparisons were computed by using paired-sample t tests with Bonferroni correction for multiple comparisons (alpha set to P < .01). See Supplementary Methods for details about preprocessing of MRI data and statistical analysis.
Lesion Analysis
Lesions were manually delineated on the T1-weighted images using MRIcro Toolbox (http://www.cabi.gatech.edu/mricro/mricro). Individual lesions were mapped by an expert neuroradiologist delineating the boundary of the lesion directly on the image for every single transverse slice. Both the MRI scan and the lesion shape were then mapped into stereotaxic space using the new normalization algorithm provided by SPM12.
Results
Lesion Anatomy
Most lesioned regions involved the periventricular area. The highest lesion overlap was found in subcortical white matter of the left hemisphere. Conversely, cortical involvement of regions outside the periventricular zone, that is, inferior frontal, dorsolateral frontal, inferior, and superior parietal regions, was much less frequently found (N = 3 subjects). Overall, lesion distribution was similar to previous CP lesion studies.40,41 Supplementary Figure 1 shows an overlap of the lesions of all UCP children and the 3D individual lesion reconstruction.
Behavioral Responses During fMRI
To test the response accuracy, we carried out an analysis based on the responses given by the participants during the scanning sessions when presented with the catch trials. Mean response accuracy recorded during catch trials was 85.6% (SD = 10.4%) for UCP children and 89.2% (SD = 8.4%) for TD children. No statistical differences (significance level set at P < .05) between TD and UCP children for the number of correct answers (t = −1.09; P = 0.28 nonsignificant) were observed.
Brain Activations During Action Observation
Figure 2 illustrates voxel-based overlap maps for the UCP and TD groups during action observation, based on the contrasts AO Paretic Hand > Rest and AO Healthy Hand > Rest. In UCP, activation patterns were largely symmetrical, although some clusters were only present in the right contralesional hemisphere, such as PMC activation, during observation of the healthy model. During observation of both paretic and healthy hand, consistent voxels across the 10 patients were detected in areas belonging to the MNS and the AON,15,19 including occipitotemporal, posterior parietal, and premotor cortices. Occipitotemporal activation reached a maximum near the middle temporal gyrus (MT/V5). This activation shows 2 rostrallydirected branches: a dorsal one extending to the posterior superior temporal gyrus (pSTG), and a ventral one extending into the posterior occipitotemporal sulcus (pOTS). Furthermore, there was an increased activation in the PPC, including both IPL and SPL. A large premotor activation, more lateralized to the right contralesional hemisphere, was observed during both action observation conditions. Figure 3 shows a further visualization of the individual activations in UCP, resulting from the direct contrast AO Paretic Hand versus AO Healthy Hand, together with the morphological images of individual lesions.
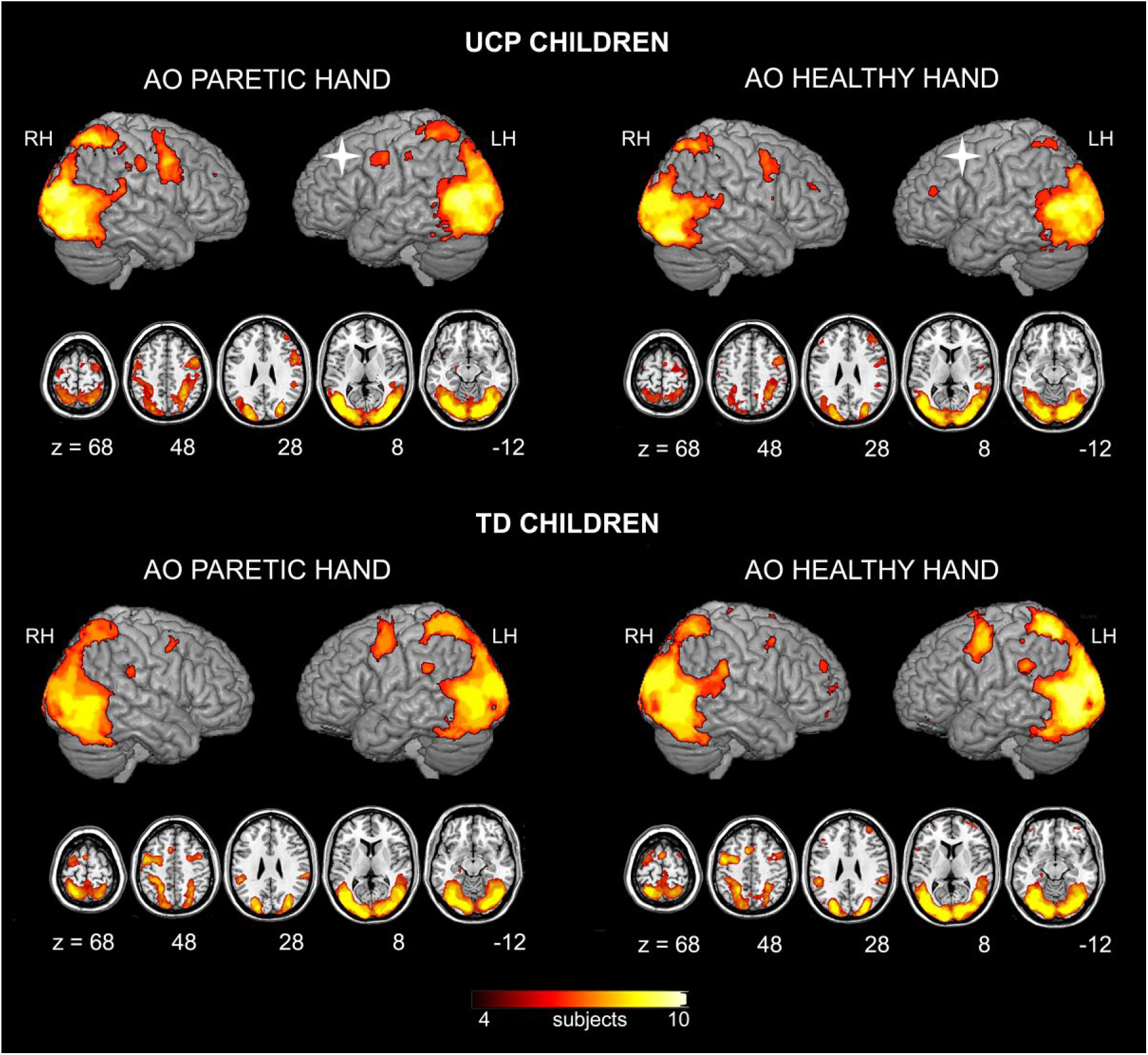
Voxel-based overlap maps of the mirror neuron system (MNS) in children with unilateral cerebral palsy (UCP) and typically developing (TD) children. Each panel shows a different contrast: AO Paretic Hand versus Rest in UCP (top, left), AO Healthy Hand versus Rest in UCP (top, right), AO Paretic Hand versus Rest in TD (bottom, left), AO Healthy Hand versus Rest in TD (bottom, right). Color bar indicates how consistently a given effect occurs across subjects, from 40% to 100% (meaning that the maximum value of the color scale was given by areas consistently activated in all ten subjects belonging to the same group). All activations are rendered into a standard Montreal Neurological Institute template and in 5 representative axial slices. The white cross indicates the lesioned hemisphere in UCP children. LH, left hemisphere; RH, right hemisphere.
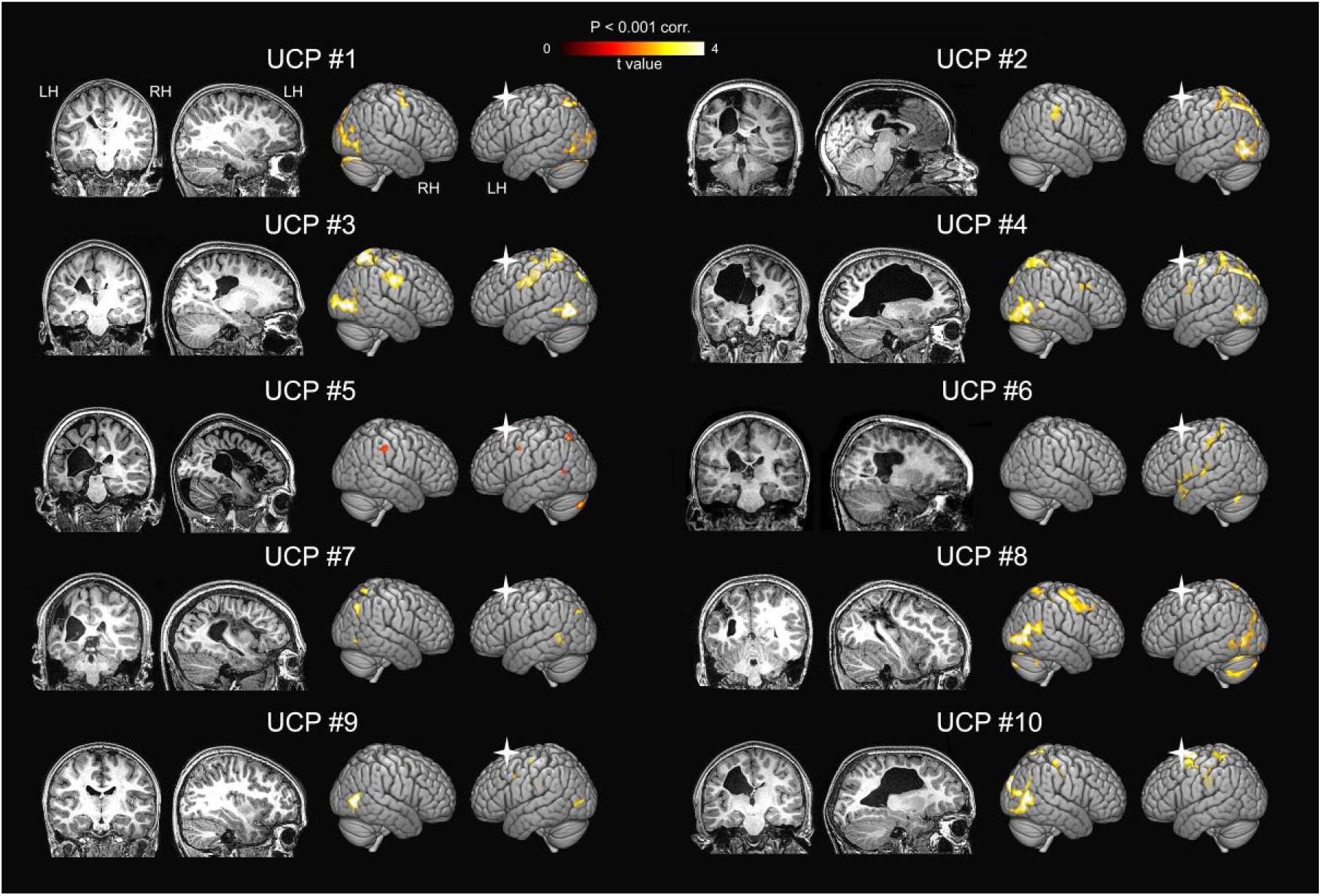
Individual brain activation maps in children with unilateral cerebral palsy (UCP) obtained by the contrast between observation of actions performed by paretic model and those performed by the healthy one (AO Paretic Hand vs AO Healthy Hand). All activations are rendered into a standard 3D Montreal Neurological Institute brain template (PFWE < .001 at cluster level). For each UCP patient: on the right side the 3D rendering of brain activations is presented in normalized space; on the left side, morphological images (T1 weighted) of individual lesions are displayed in one coronal and one parasagittal representative slice. The white cross indicates the lesioned hemisphere. LH, left hemisphere; RH, right hemisphere.
In TD children, in accordance with studies on action observation in adults, activation maps were more lateralized to the left hemisphere. Activation was observed in areas belonging to AON, such as the STG/MTG, anterior intraparietal sulcus (aIPS), IPL, SPL and PMC. All activations at single-subject level were thresholded at P < .001, after correction for multiple comparison (family-wise error [FWE]) at cluster level.
Brain Activations During Motor Localizer
In order to restrict our analyses to the main nodes of MNS, we identified, at a single-subject level, brain areas activated during an explicit motor imagery task (localizer).
TD Children
Similar to previous studies in adults, 37 imagining of reaching, grasping and placing an object activated, in TD children, the SPL and IPS, the precuneus, as well as the dorsal PMC (superior frontal gyrus/sulcus). Specifically, TD children’s activations included, in the frontal lobes, bilateral IFG (including the pars opercularis), PMC, middle frontal gyrus (MfG), and SMA. In the parietal lobes, bilateral SPL and rostral-left IPL were activated. Consistently activated subcortical regions included left putamen, pallidum and right thalamus. Finally, lobules VI (bilateral) of the cerebellum (CB) were found to be consistently activated (Supplementary Figure 2).
Children With UCP
Consistent activations were found in clusters similar to those of TD children (see below ROI analysis, Figure 4 and Supplementary Figure 2). In the parietal lobes, activations were consistently found in bilateral SPL, IPL, and precuneus, in addition to right postcentral gyrus (PocG). Consistent activations were also found in the CB bilaterally and in left putamen and right pallidum. However, a different functional pattern of activations was found during motor imagery among UCP children, in agreement with the action observation task (Supplementary Figure 2).
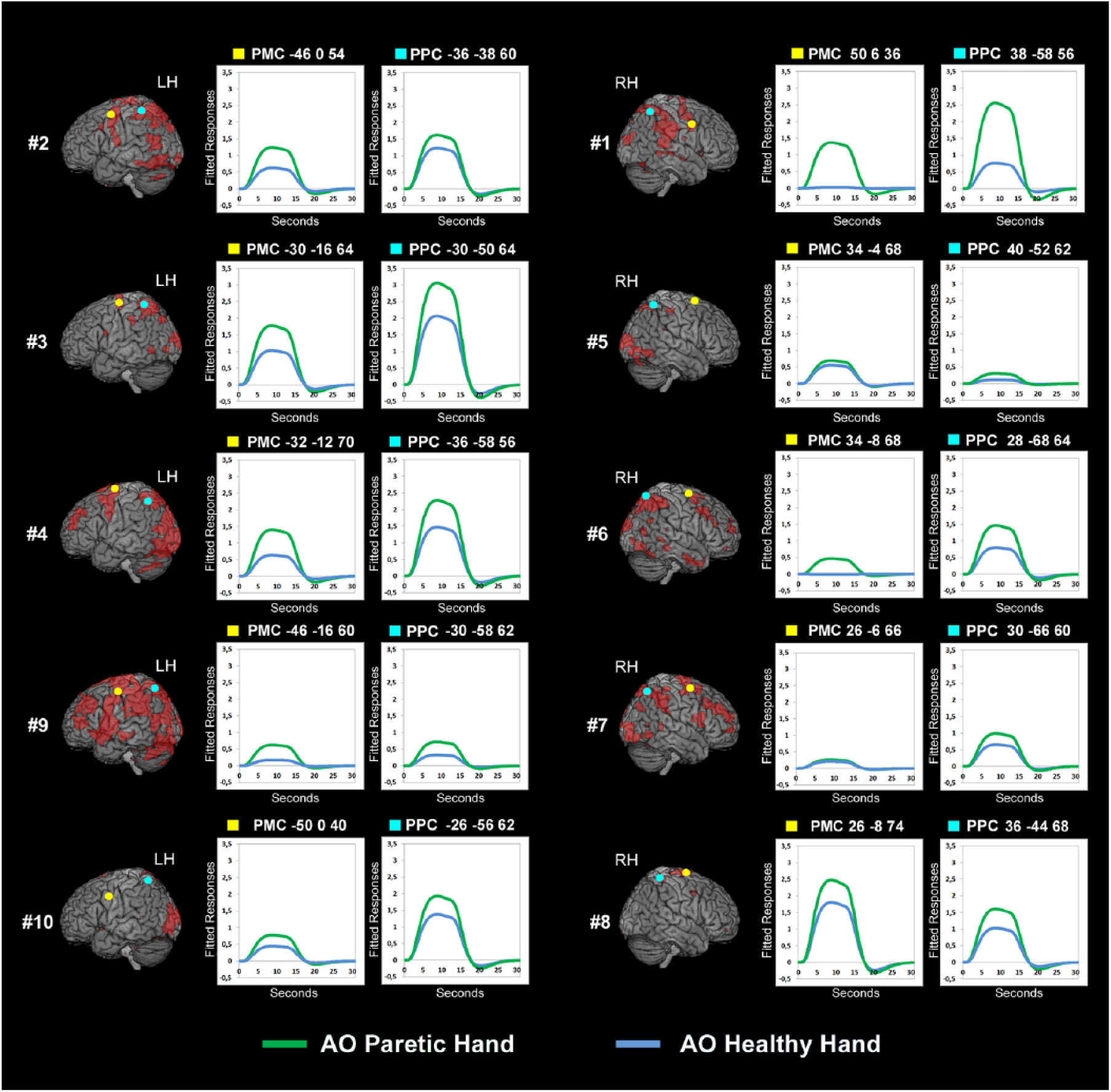
Individual anatomical location and fitted responses for premotor and parietal regions of interest (ROIs) in UCP patients. Premotor cortex (PMC) and posterior parietal cortex (PPC) ROIs are superimposed on the activation map (red color) resulting from the motor localizer based on motor imagery task, displayed into a standard Montreal Neurological Institute brain template (PFWE < .001 cluster level). Yellow points indicate peaks of activation within the PMC region, while light blue points indicate peaks in the PPC region, for each individual subject. Plots illustrate individual time course of fitted event-related responses from the conditions AO Paretic Hand versus Rest (green line) and AO Healthy Hand versus Rest (blue line). LH, left hemisphere; RH, right hemisphere.
ROI Analysis
ROI analysis allows testing of BOLD responses at single-subject level, with a high level of accuracy, also considering the lesioned tissue. On the basis of specific lesions and functional reorganization in the UCP sample, we reasoned that testing the effect of a mean entire ROI (averaged on 10 local maxima) might not be representative of the real effects occurring in a given region. PPC and PMC ROIs were centered at single-subject level on the local maxima at P < .001, after correction for multiple comparisons, in the left hemisphere for all TD participants and in the left hemisphere (subjects 2, 3, 4, 9, and 10) or in the right hemisphere of children presenting no ipsilesional activations (subjects 1, 5, 6, 7, and 8), as shown by the localizer. Individual Montreal Neurological Institute (MNI) coordinates of the local maxima for each ROI in PMC and PPC are reported in Figure 4 for the UCP group and in Supplementary Figure 3 for the TD group.
The multisubject ROI analysis performed in PPC using Condition as repeated-measure factor and Group as between-subject factor, revealed a significant main effect of Condition (F1,18 = 9.39, P = .007) (see Supplementary Table 2). Post hoc comparisons (Bonferroni) showed that in the PPC the observation of the paretic model produced a stronger activation as compared with the observation of the healthy one (P = .006). Most important, the rmANOVA performed in PPC ROIs revealed a significant interaction Condition × Group (F1,18 = 24.58, P < .001). In particular, UCP children activations within the PPC ROIs were higher during the observation of the pathological model (P = .0001) (Figure 5, A1, A2, A3) with regard to the healthy one. On the contrary, in the TD group, BOLD responses in PPC for observation of paretic versus healthy hand did not show any significant difference (P = 1, nonsignificant) (Figure 5, C1, C2, C3).
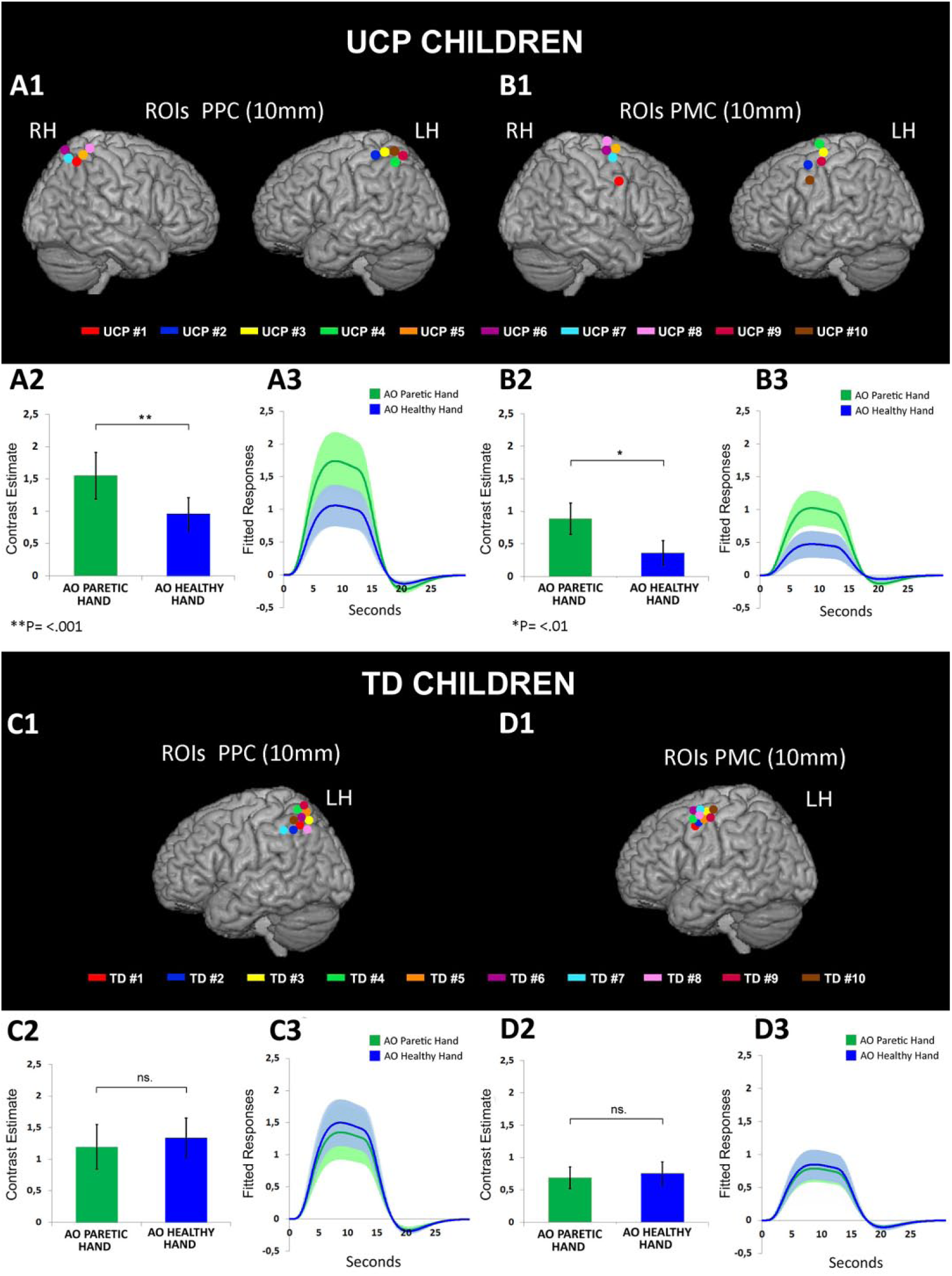
Multisubjects region of interest (ROI) analysis. Individual anatomical locations of posterior parietal cortex (PPC) and premotor cortex (PMC) ROIs displayed into a standard Montreal Neurological Institute (MNI) brain template, for both children with unilateral cerebral palsy (UCP) patients (A1, B1) and typically developing (TD) children (C1, D1). Histograms show the averaged magnitude of activation (parameter estimate) in each ROI for UCP (A2, B2) and TD group (C2, D2). Line graphs indicate event-related fitted response across peristimulus time in PPC and PMC ROIs for the experimental conditions AO Paretic Hand (green line) and AO Healthy Hand (blue line), in UCP cases (A3, B3) and TD group (C3, D3). Brackets above the columns indicate the statistical comparison among the conditions AO Paretic Hand and AO Healthy Hand. Bars and colored area indicate standard error of the mean (SEM). Asterisks indicate significant differences set at P < .01 (*) and P < .001 (**). LH, left hemisphere; RH, right hemisphere.
Similarly, the rmANOVA performed in PMC revealed a main effect of Condition (F1,18 = 11.52, P = .003). Post hoc pairwise comparisons showed a greater activation for the observation of the pathological model with regard to the healthy one (P = .003). In addition, a significant interaction Condition × Group was present also in PMC (F1,18 = 19.11, P < .001). Similarly to PPC, also in PMC the observation of the paretic model provoked in UCP a greater activation compared with the observation of the healthy model (P = .005) (Figure 5, B1, B2, B3), while no significant difference was found between the AO conditions in TD participants (P = 0.45, nonsignificant) (Figure 5, D1, D2, D3).
Discussion
This study suggests for the first time that children affected by UCP exhibit enhanced activation of the MNS during observation of goal-directed actions performed by a paretic hand, a model similar to their own motor repertoire, with regard to observation of a healthy hand. Using a ROI-based approach, we found, at both individual and multisubject level, that activation of the main nodes of MNS (PPC and PMC) in UCP depends on the specific model observed. On the contrary, TD children similarly activate MNS in both observation conditions.
Modulation of the MNS in Children With UCP: The Role of Motor Experience
Few studies have explored the neural basis of action observation in CP children using neuroimaging techniques.40,42 However, these studies were not carried out on homogeneous groups and did not take into account motor impairment level, lesion side, or specific hand kinematics. Furthermore, they did not use, as video stimuli, goal-directed hand actions. Our fMRI study was focused on observation of reaching-grasping actions in a specific cohort of UCP children, showing, both during observation of paretic and healthy hand, a bilateral pattern of activation of the MNS, although this activation was stronger in the right, contralesional hemisphere.
These findings could, in principle, be attributed to basic features of employed stimuli such as the effective duration of the observed hand movement, dissimilar between the 2 conditions. However, the absence of a differential activation in TD children indicates that this explanation can be excluded. Another possible confounding factor could be the type of grip used by the paretic and healthy hand model; however, in TD participants the activations in the two conditions were very similar. Finally, the present findings cannot be explained by visual familiarity with the observed model. In fact, the paretic model is unfamiliar to TD children, but activation within ROIs was not different between the two conditions, while children of UCP group have visual familiarity with both models, but brain activation was higher for the paretic hand condition.
The modulation of brain activity found in children with UCP appears to be better explained by the property of the MNS to reflect the individual’s own motor repertoire, in line with several neuroimaging studies on healthy participants.21-24 This suggests that, in children with UCP, cortical motor representation of the more-affected hand better matches the visual description of the paretic hand model. Indeed, the hand presented in the action observation conditions (either paretic or healthy) was always the right one in a first-person perspective, thus anatomically corresponding to the affected hand of the enrolled children with UCP.
In TD children, contrary to UCP ones, BOLD response in PPC and PMC regions of the left hemisphere during observation of paretic vs. healthy hand was very similar. A possible explanation for this result is that for TD participants the pathological model could be a simplification of their hand motor representation. Thus, when they observed a pathological model, their motor system matched the goal of the action more than the way in which it was executed. 43 Interestingly enough, after the experiment, some TD participants reported no evident differences between the 2 observed models. This result should not be surprising, considering that Aslamour et al 44 reported that observation of a paretic model in healthy children is effective in improving manual dexterity and upper limb velocity.
Another interesting aspect is that the use of the motor localizer allowed us to observe an activation of PPC and PMC also in the damaged hemisphere in 5 patients (UCP subjects 2, 3, 4, 9, and 10). Contrarily, in the remaining UCP patients (subjects 1, 5, 6, 7, and 8), activation clusters were found mainly in the right contralesional hemisphere. Thus, although the case group was relatively homogeneous in terms of degree of upper limb impairment and lesion side, patients showed different activation patterns. This suggests the possibility that the 2 groups of patients underwent a different type of brain reorganization, in agreement with that already described in the literature.45-48
Representation of Action Goals and Movement Kinematics in Children With UCP
The observation of grasp-to-place actions enhanced in UCP activation of posterior parietal and premotor areas, consistently with previous studies on action observation.15,16,36,43 However, in the majority of studies on action observation, it is difficult to disentangle the processing of action goal from action kinematic components. 43 Hamilton and Grafton49,50 suggested that action goal is encoded in the posterior parietal and premotor areas belonging to the MNS, while basic kinematic features of an action (eg, type of grip, trajectory) are processed by occipital, superior precentral and middle frontal areas. On the other hand, other studies indirectly showed that areas belonging to the parietofrontal MNS process not only the goal of the observed action but also the way to achieve it.21-23,51
Clinical reports 30 during AOT application indicate that observation of healthy models elicits, in children with UCP, a marked propensity, during movement reproduction, to preserve task proficiency, by selecting those movements which guarantee the final action outcome, likely because of the notable differences between UCP movement kinematics and observed healthy ones. On the contrary, when observed actions are performed by a hand kinematically similar and anatomically corresponding to their affected hand, children with UCP could tend to have a stronger activation of the MNS, which in turn could induce a motor reproduction more similar to the observed model. This interpretation is corroborated by previous studies on healthy individuals, showing that ventral premotor cortex 52 and left dorsofrontal and dorsal premotor cortex 53 can be sensitive also to biological kinematics when compared with impossible motor acts or acts violating the kinematic invariants of human movements. This supports the notion that the MNS is important for analyzing the goal of the observed action on the basis of its biologically possible kinematics. Thus, it is plausible that, also in children with UCP, the premotor and parietal cortices are sensitive to observed action kinematics. In situations in which the observed action is not within the personal motor repertoire, deviation in kinematics could be an important element for explaining the lower motor resonance with the observed model.
Limitation of the Study
First, no classical GLM group analysis was performed because children with UCP showed extensive cortico-subcortical lesions; therefore, we decided to use subject-specific functional localizer, that usually outperforms traditional group-based methods in both sensitivity and functional resolution. Admittedly, this could decrease the possibility to generalize the data. Second, sample size was low, because we used strict inclusion criteria and keep the sample homogeneous. Third, we recruited only UCP children with left damage, to limit possible confounding variables in the results interpretation. However, this does not allow us to directly extrapolate the results to children with UCP with right brain lesions (left UCP) or to other forms of CP. Future studies could investigate the effects of observation of pathological and healthy models in children with UCP presenting different types of brain lesions/sides of hemiplegia. Finally, the use of motor imagery as the localizer per se does not allow to make direct inference about the motor reorganization mechanisms in UCP. Thus, future studies could include other techniques, like transcranial magnetic stimulation, to assess motor reorganization and to correlate it to cortical activations during action observation, assessed by fMRI.
A Different Model of AOT as a Tool for Motor Rehabilitation
Current evidence suggests the implementation of goal-directed problem-solving strategies in the rehabilitation of UCP patients. In this respect, AOT is not requiring a strong cognitive effort to the child, because her/his performance is driven by the observation of an external model that automatically recruits, thanks to the MNS, the motor representations of the actions similar to those performed by the observed model. These representations are then reinforced by the subsequent action reproduction. 54 Based on this mechanism, AOT has been used in the rehabilitation of neurologic patients, both adults and children.9,25-30,54,55 However, a better understanding of the underlying mechanisms could lead to further improvements of the effects of observation-based therapies. Therefore, without denying the clinical relevance of the general functional achievement, the effects of AOT could be better highlighted by using scales that, beyond the goal achievement, also take into account the improvement of action kinematics features.
The present study suggests that observation followed by imitation of a model, whose motor skills are more closely matched to those of the observer, could lead to a greater motor improvement. In fact, if motor recovery in UCP patients were possible on the basis of imitation of a healthy model, they could simply copy their less affected hand. In addition, the use of different AOT models, based on the degree of motor impairment, could allow this therapy to be adapted to the upper limb clinical characteristics of each individual patient. To better define which patients would benefit most from observation therapy based on a pathological model, future studies should be designed as randomized controlled trials in order to evaluate, under homogeneous clinical conditions, efficacy of this type of AOT to improve upper limb activity in children with UCP.
Supplemental Material
Supplementary_Fig._1 – Supplemental material for Mirror Neuron System Activation in Children With Unilateral Cerebral Palsy During Observation of Actions Performed by a Pathological Model
Supplemental material, Supplementary_Fig._1 for Mirror Neuron System Activation in Children With Unilateral Cerebral Palsy During Observation of Actions Performed by a Pathological Model by Antonino Errante, Giuseppe Di Cesare, Chiara Pinardi, Fabrizio Fasano, Silvia Sghedoni, Stefania Costi, Adriano Ferrari and Leonardo Fogassi in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_Fig._2 – Supplemental material for Mirror Neuron System Activation in Children With Unilateral Cerebral Palsy During Observation of Actions Performed by a Pathological Model
Supplemental material, Supplementary_Fig._2 for Mirror Neuron System Activation in Children With Unilateral Cerebral Palsy During Observation of Actions Performed by a Pathological Model by Antonino Errante, Giuseppe Di Cesare, Chiara Pinardi, Fabrizio Fasano, Silvia Sghedoni, Stefania Costi, Adriano Ferrari and Leonardo Fogassi in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_Fig._3 – Supplemental material for Mirror Neuron System Activation in Children With Unilateral Cerebral Palsy During Observation of Actions Performed by a Pathological Model
Supplemental material, Supplementary_Fig._3 for Mirror Neuron System Activation in Children With Unilateral Cerebral Palsy During Observation of Actions Performed by a Pathological Model by Antonino Errante, Giuseppe Di Cesare, Chiara Pinardi, Fabrizio Fasano, Silvia Sghedoni, Stefania Costi, Adriano Ferrari and Leonardo Fogassi in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_Methods_rev – Supplemental material for Mirror Neuron System Activation in Children With Unilateral Cerebral Palsy During Observation of Actions Performed by a Pathological Model
Supplemental material, Supplementary_Methods_rev for Mirror Neuron System Activation in Children With Unilateral Cerebral Palsy During Observation of Actions Performed by a Pathological Model by Antonino Errante, Giuseppe Di Cesare, Chiara Pinardi, Fabrizio Fasano, Silvia Sghedoni, Stefania Costi, Adriano Ferrari and Leonardo Fogassi in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_Table_1 – Supplemental material for Mirror Neuron System Activation in Children With Unilateral Cerebral Palsy During Observation of Actions Performed by a Pathological Model
Supplemental material, Supplementary_Table_1 for Mirror Neuron System Activation in Children With Unilateral Cerebral Palsy During Observation of Actions Performed by a Pathological Model by Antonino Errante, Giuseppe Di Cesare, Chiara Pinardi, Fabrizio Fasano, Silvia Sghedoni, Stefania Costi, Adriano Ferrari and Leonardo Fogassi in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_Table_2 – Supplemental material for Mirror Neuron System Activation in Children With Unilateral Cerebral Palsy During Observation of Actions Performed by a Pathological Model
Supplemental material, Supplementary_Table_2 for Mirror Neuron System Activation in Children With Unilateral Cerebral Palsy During Observation of Actions Performed by a Pathological Model by Antonino Errante, Giuseppe Di Cesare, Chiara Pinardi, Fabrizio Fasano, Silvia Sghedoni, Stefania Costi, Adriano Ferrari and Leonardo Fogassi in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We wish to thank the participating children and their parents. We thank G. Crisi, F. Bozzetti and Silvia Piccinini for neuroradiological consulting. We are further grateful to G. Burani, E. Nasti, M. Rinaldi and C. Saccò (Parma) for their kind technical support during MRI acquisitions. We thank G. Cioni for his critical reading of a previous version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Chiesi Foundation (Parma, Italy).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
