Abstract
Objective. A randomized controlled observer-blind trial was designed to evaluate the effectiveness of action observation as an add-on treatment to the standard rehabilitation of upper-limb function, early after stroke. Methods. Stroke survivors (N = 102) were consecutively recruited from 13 centers 30 days (±7) after a first-ever stroke and randomly assigned to the experimental (EG) or control group (CG). EG participants watched video footage of daily routine tasks (actions) carried out with the upper limb in order to prepare to imitate the presented action. At the end of each sequence, a therapist prompted the patient to perform the same movement for 2 minutes, providing help when needed. Static images without animals or human beings were shown to the CG. At the end of each sequence, the CG executed movements that simulated the shoulder and elbow joint mobilization activities performed by the EG. Results for the Fugl-Meyer test, Frenchay Arm test, Box and Block test (BBT), Modified Ashworth Scale, and Functional Independence Measure Motor items were recorded before treatment (T0), after 4 weeks of treatment (T1), and at the follow-up visit 4 to 5 months after the conclusion of treatment (T2). Results. An improvement over time was appreciated on all measures of impairment and functional ability with both treatment programs. A Time × Treatment interaction emerged from the generalized estimating equations analysis of BBT, showing significant T0–T1 and T0–T2 differences in favor of EG. Conclusion. This multicenter trial endorses the use of action observation in upper-extremity rehabilitation, along with a role for the mirror neuron system in poststroke recovery.
Introduction
Up to 60% of patients with stroke have poor dexterity in the upper extremity late after onset. 1 Numerous studies showed that ascertaining the effectiveness of rehabilitative interventions on conditions leading to long-term disability, such as stroke, is a complex task because the outcome depends on many interacting factors. 2 Several studies underline the importance of brain plasticity and its therapeutic potential in neurological disorders. Accredited theories of cortical reorganization after brain lesion endorse the use of early, intensive, repetitive, and context-related exercise as optimal strategies to promote motor relearning and minimize motor deficit.3-5 Wang et al 6 described the dynamic topological changes in the motor execution network, suggesting that cortical reorganization may contribute to motor function restoration following stroke. 7 This recovery has been commonly attributed to cortical reorganization, as confirmed by findings from functional neuroimaging studies, which also indicate the increased recruitment of contralesional motor areas. 8 Emphasis on the relevance of sensory feedback as a facilitating stimulus to the motor cortex is also present in the literature. 9 The mirror neuron system (MNS) is an observation execution matching system activated during action observation, motor learning, and imitation of action. In healthy humans, action observation increases cortical excitability of the primary motor cortex, thus potentially enhancing motor learning. 10 A few trials have been performed on stroke survivors, where action observation was used to facilitate paretic hand movements either through mental practice 11 or visual illusions created by mirrors. In the past year, several works have been published about action observation and the neuron mirror system. 12
Through a functional magnetic resonance imaging with an experimental paradigm in which motor acts had to be observed and/or imagined from a first-person perspective, Nedelko et al 13 showed that activity within the MNS is not age dependent and provides a neural basis for therapeutic interventions and novel rehabilitation treatments such as video therapy.
Small et al 14 suggest in their review that an MNS devoted to hand, mouth, and foot actions might also be present in humans and showed evidence regarding the role of the MNS in action understanding and imitation, both in hand motor function and speech. They also suggest the use of action observation and imitation as an approach for systematic training in the rehabilitation of patients with motor impairment of the upper limb and aphasia following stroke.
We tested the hypothesis that congruent action observation combined with physical training is more effective than static image observation and physical training in the recovery of upper-limb motor functions in the early phase of stroke.
Methods
Eligible hemiparetic stroke survivors from 13 rehabilitation centers were consecutively recruited from July 1, 2007, to December 31, 2009. The local ethics committees of the involved rehabilitation facilities approved the study. All patients gave informed consent to the investigation. The study included only patients with first-ever stroke, enrolled 30 days (±7) after the event onset with ischemia or primary hemorrhage (Figure 1). All patients were right handed prior to stroke. They had unilateral lesions with 45% in the right hemisphere. Exclusion criteria were (1) posterior circulation infarction, (2) subarachnoid hemorrhage, (3) severe forms of neglect and anosognosia (number of errors in Bell Barrage test ≥15), 15 (4) impaired comprehension (Token test score ≤ 17), 16 (5) history of endogenous depression or serious psychiatric disorders, and (6) severe visual deficits (restricting the access to visual stimuli). Diagnosis was confirmed by CT or MRI scan.
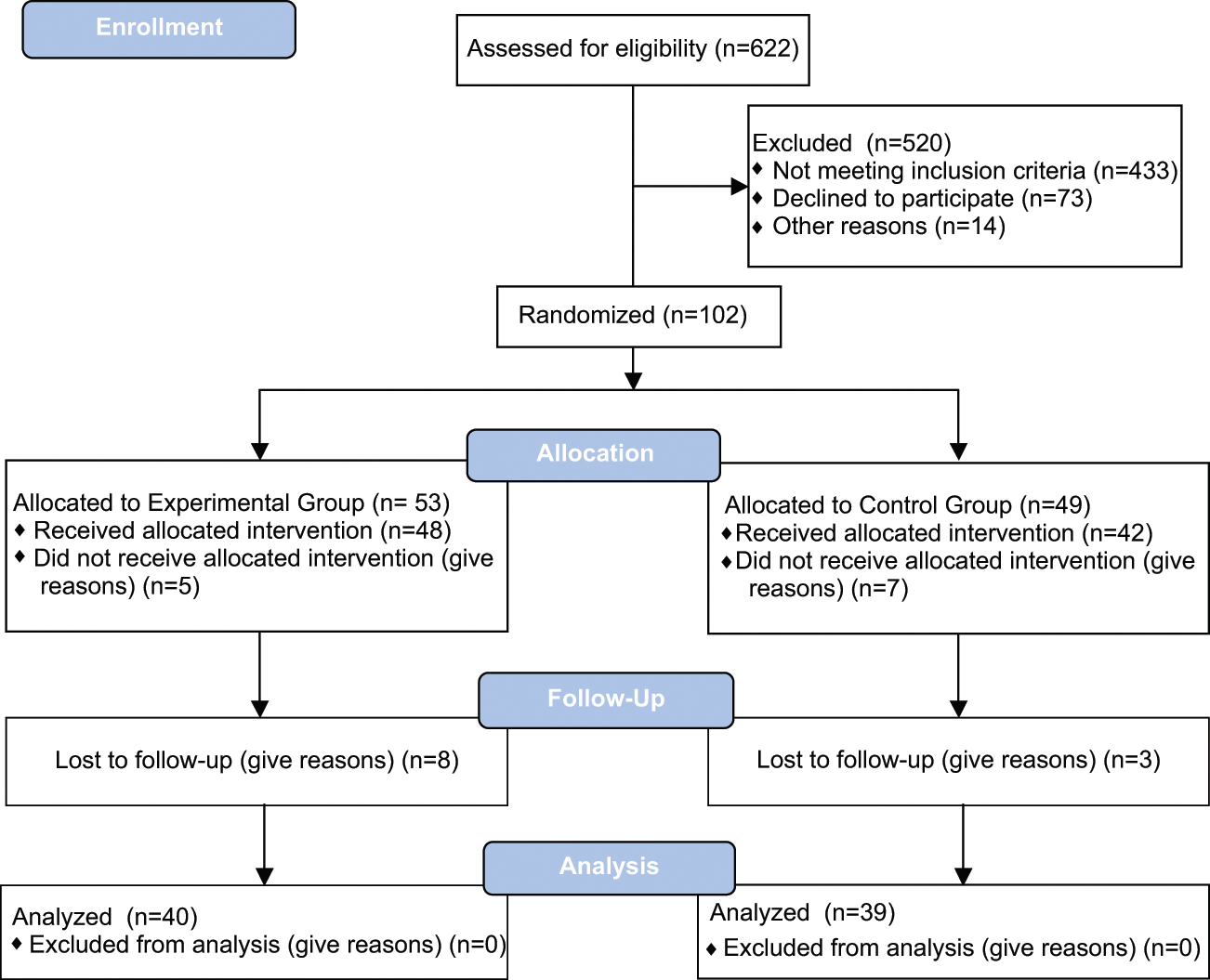
Study consort
The random allocation to treatment was concealed and based on a custom computerized system. Each participating center asked the server for the group allocation after typing in age, gender, and brain lesion side. The server was accessed through the Client software and an HTTPS Internet protocol. It applied the Lehmer algorithm to allow a balanced participant allocation into the EG and CG groups. Within each center, occupational therapists (OTs) were randomly assigned to patients, following the same procedure.
The baseline assessment included the Canadian Neurological Scale, the Mini-Mental State Examination (MMSE), 17 the Bell Barrage test, and the ideomotor apraxia test (Spinnler-Rognoni). 18 The following impairment and functional evaluations were performed at entry (T0), at the end of the treatment at 4 weeks (T1), and at follow-up 4 to 5 months after treatment ended (T2): Fugl-Meyer test (FM) (of the upper limb), 19 Frenchay Arm test (FAT), Box and Block test (BBT), Modified Ashworth Scale (AS), 20 and Functional Independence Measure Motor items (FIMM). All assessments were performed by trained professionals not involved in the research treatment and blind to group allocation. These raters came from the 13 centers and were trained to watch footage of stroke patients and independently provide their ratings of the FM, BBT, and FAT. Interrater reliability was assessed through κ statistics and proved to be high (Cohen’s κ = 0.905; 95% confidence interval = 0.850-0.959).
Interventions
All participants underwent inpatient rehabilitation with at least 3 h/d of physiotherapy, including both dexterity and gait training, according to individually tailored exercises. In addition to standard rehabilitation, eligible patients also received two 15-minute daily sessions of either experimental (EG) or control treatment (CG).
Experimental treatment
Every day, before starting physical training, the patient was asked to carefully watch video footage showing 20 different daily routine tasks (actions) carried out with the upper limb 12 (see Supplementary Table 1). The patient was presented only 1 task per day, starting from the easiest and ending with the most complex action throughout 20 sessions, the whole treatment period lasting 4 weeks (5 sessions/wk). Each action consisted of 3 different motor sequences displayed in order of ascending difficulty and lasting 3 minutes each. Tasks were based on some relevant activities of daily living such as drinking from a glass, combing hair, opening a box, or eating an apple. Tasks were unimanual and bimanual. The actions were observed from a first-person perspective. Actors in the videos were young nondisabled men or women and differed from video to video. All actions were object and goal directed. For example, take and drink a cup of coffee was divided into 3 acts: (1) Reach and grasp the handle of the cup with the affected arm and return to the starting point; (2) reach and grasp the handle of the cup with the affected arm; raise the cup toward the mouth; return to the starting point; and (3) reach and grasp the handle of the cup with the affected arm; raise the cup toward the mouth and drink; and then return to the starting point.
Distribution of the Study Participants by Age, Gender, and Main Clinical Characteristics
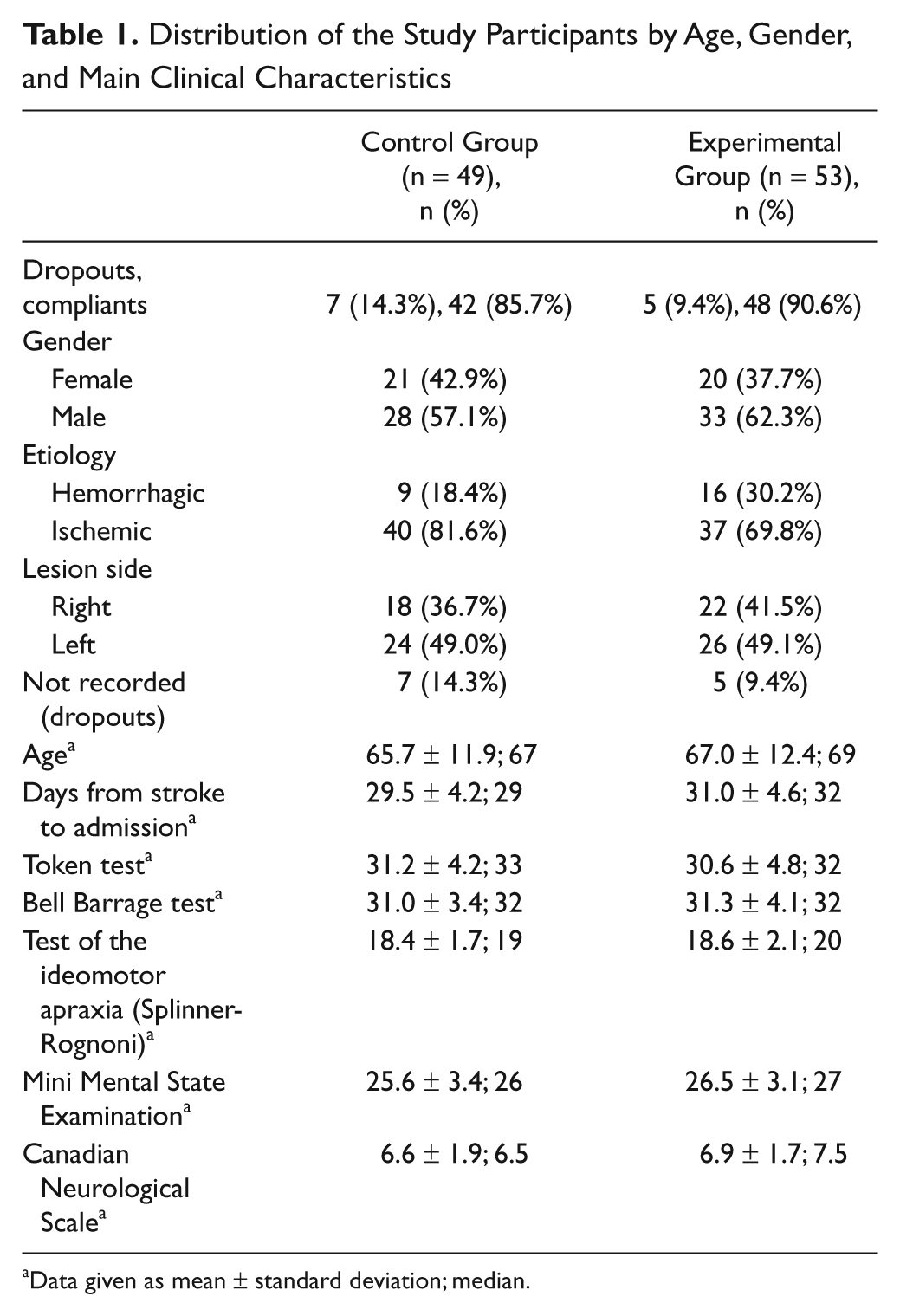
Data given as mean ± standard deviation; median.
During each daily session, the patient had to watch the video under OT supervision. Participants were asked to prepare to imitate the presented action. The OT maintained the patient’s attention with verbal feedback. At the end of each sequence, the OT prompted the patient to perform the same movement over 2 minutes, providing help when needed. The patients were asked to perform the observed action with the paretic upper limb as many times as they could. They received verbal instructions by the OT such as the following: “Slowly put the hand of your affected arm to the top of your head. You may use your unaffected arm to help guide if needed” or “Extend your affected arm to the wall in front of you. You may use your unaffected arm to help guide if needed.” The OT judged whether patients accomplished the task themselves or should be assisted in the task of imitating the observed action. If the latter, the OT provided patients with physical help such as limb support or passive mobilization but did not interact with the object. Participants had only to imitate the motor sequence they had observed.
Each session had to last about 15 minutes (3-minute sequence observations and 2-minute action performances for 3 motor sequences) and was repeated twice per day in 2 separate sessions, at least 60 minutes apart. In between, the patient was requested to rest.
Control treatment
We used “sham” action observation. Participants were shown 5 static images displaying objects—none were animals or people—for 3 minutes. 12 A cognitive task was required to keep the patient’s attention high. For a 3-minute sequence, images were separately displayed, each for 30 s, and then together during the last 30 s, with an intrusive image (interloper) that the patient was asked to identify so that his or her attention span could be checked in real time by the OT. Participants were then asked to perform limb movements as well as feasible for 2 minutes according to a standard sequence, simulating those performed by the EG, involving shoulder and elbow joint mobilization. They received verbal instruction by the OT, such as the following: “Starting with your elbow flexed at an angle of 90° and your shoulder adducted, please abduct your shoulder and extend your elbow as far as you can. You may use your unaffected arm to help guide if needed.” They did not interact with objects or see the OT perform the movement. The OT provided limb support or passive mobilization to help them accomplish the motor sequence.
Every missed session was made up. Those who missed more than 5 consecutive days were excluded from the study.
Statistical Methods
The BBT is the primary outcome. Given the multiple end points measured in the study, the sample size was calculated according to the parameter expected to benefit the most from the experimental treatment—that is, the BBT. From a systematic reevaluation of the patients, performances, as reported in the clinical records of the Department of Neurorehabilitation at the IRCCS San Raffaele Pisana, an average reference score of 20 ± 20 points was identified and set to be compared with a final score of 30 ± 20 points in the EG. Assuming a type I error rate of 5% and an allocation ratio to the 2 treatment groups of 1:1, it was estimated that a study with a power of 80% required the recruitment of 100 early-phase first-stroke patients. The presence of heterogeneity between the EG and CG with respect to the distribution of demographics and clinical features, including stroke etiology and lesion side, was tested by means of univariate statistics. Pearson’s χ2 statistics, or Fisher’s exact test where appropriate, were applied to categorical outcomes. A conservative nonparametric approach (Kruskal-Wallis test) was applied to investigate for the presence of a trend over time within each of the 2 treatment groups for all assessments of functional ability. The difference between the 2 treatment groups at each visit was tested with the Mann-Whitney U test. The departure from normality for the FIMM, FM, and the BBT was minimal, so it was possible to further explore these variables by means of the generalized estimating equation (GEE). This methodology takes into account the autocorrelation intrinsic in the nature of repeated measures over time. The functional recovery of patients who experienced a hemiparetic stroke is a learning process; therefore, the performance of an individual at T1 and T2 is likely to be dependent on previous performances. For this reason, the order 1 autoregressive correlation structure seemed the most appropriate for our study and was assumed in the model fitting. Gaussian family with identity link function was also specified. The following terms were included as covariates/cofactors: baseline (FIMM, FM, or BBT) scores, time, treatment and time × treatment interaction, age, etiology, lateralization of the event, and sex. To take into consideration the high drop-out rate of patients at T2, a GEE model was fitted to the subset of patients with measurements at all time points (T0, T1, and T2). The problem of missing data was also addressed by adopting the last observation carried forward approach. This approach artificially builds a worst-case scenario where missing observations for each study participant are substituted with his or her last observed value. A new GEE analysis was performed, including baseline scores, time, treatment, time × treatment interaction, age, whether a dropout/nondropout, sex, and etiology as covariates. The side of the stroke for the dropouts was unknown; therefore, this term could not be fitted as a cofactor in the model. The critical limit for significance was set at the 5% level. All analyses were intended as explorative and were carried out in STATA/SE Release 10.0 (StataCorp, College Station, TX).
Results
Out of 102 participants (Figure 1), randomly assigned to either the EG (53 cases) or CG (49 cases), 12 did not fulfill the treatment protocol (5 from EG and 7 from CG). Also, 11 more were lost at follow-up (8 from EG and 3 from CG), limiting the number of participants with a complete outcome assessment to 79 (40 on EG and 39 on CG).
The distribution of the study participants (N = 102) by age, gender, and main clinical characteristics did not significantly differ between the EG and the CG (Table 1).
Within each treatment group, the dropouts (n = 12) were compared with those fulfilling the protocol (compliant participants, n = 90) for baseline demographic and clinical parameters, including those concerning impairment and functionality. The Mann-Whitney test was used to compare median scores, and the Pearson’s χ2 test or Fisher’s exact test was used for frequencies. No significant differences emerged except for the Canadian Neurological Scale in the EG (P = .03), which showed a 2-point higher median score in the compliant participants.
Table 2 summarizes the observed means ± standard deviations of all tests (FAT, FM, BBT, AS-Shoulder, AS-Elbow, AS-Wrist, FIMM), as measured for the compliant participants at baseline (n = 90), T1 (n = 90), and T2 (n = 79). From the Mann-Whitney U test, it was seen that the degree of impairment and functional ability between the 2 treatment groups did not differ significantly at baseline, T1, and T2. Improvement over time was demonstrated by the Kruskal-Wallis test for the FAT (CG: P = .03; EG: P = .0005), FM (CG: P = .01; EG: P = .0008), BBT (CG: P = .03; EG: P = .0001), and FIMM (CG: P = .0001; EG: P = .0001) in both treatment groups. No evidence of a change in the spasticity of the shoulder, elbow, and wrist could be detected in either treatment group.
Observed Means ± Standard Deviations of All Tests and Results from Generalized Estimating Equation Analysis on the Effect of Action Observation Versus “Sham” Observation
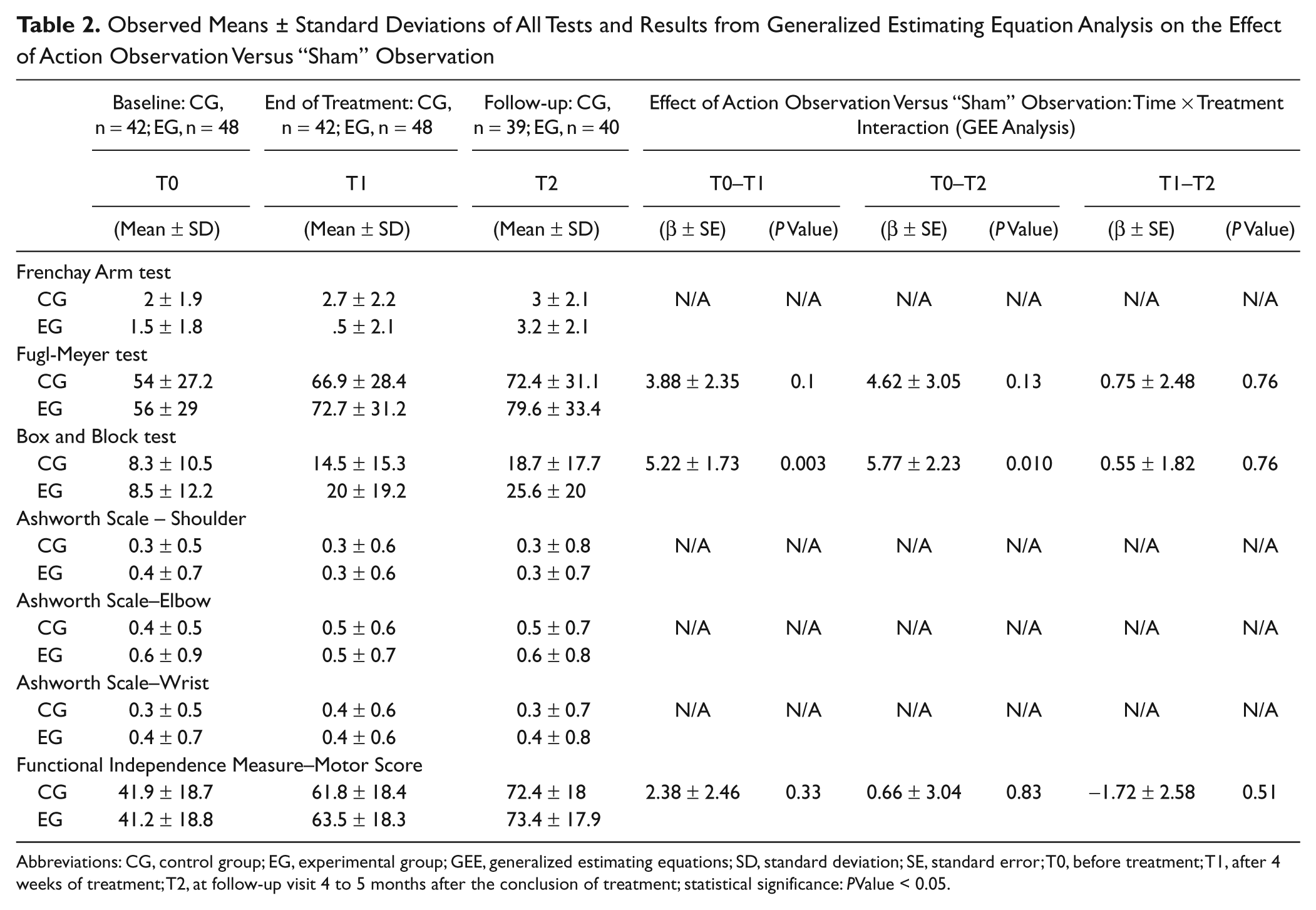
Abbreviations: CG, control group; EG, experimental group; GEE, generalized estimating equations; SD, standard deviation; SE, standard error; T0, before treatment; T1, after 4 weeks of treatment; T2, at follow-up visit 4 to 5 months after the conclusion of treatment; statistical significance: PValue < 0.05.
The GEE analysis, which included all participants at the 3 time points as specified for Table 2, confirmed the significance of the improvement over time of the FIMM, FM, and BBT scores. Evidence for a Time × Treatment interaction was only found with regard to the BBT, which was significantly higher in the EG. In particular, differences between the 2 groups were found from T0 to T1 (5.22 ± 1.73 points; P = .003) and from T0 to T2 (5.77 ± 2.23 points; P = .010). However, no difference was found on their change in BBT performance from T1 to T2 (0.55 ± 1.82 points; P = .76). Figure 2 shows the BBT trajectory. No evidence for a significant effect of etiology, sex, and age was found. The reported GEE analysis results are mean estimates ± standard errors. No significant difference between the study groups was found with regard to the FIMM and FM performance.
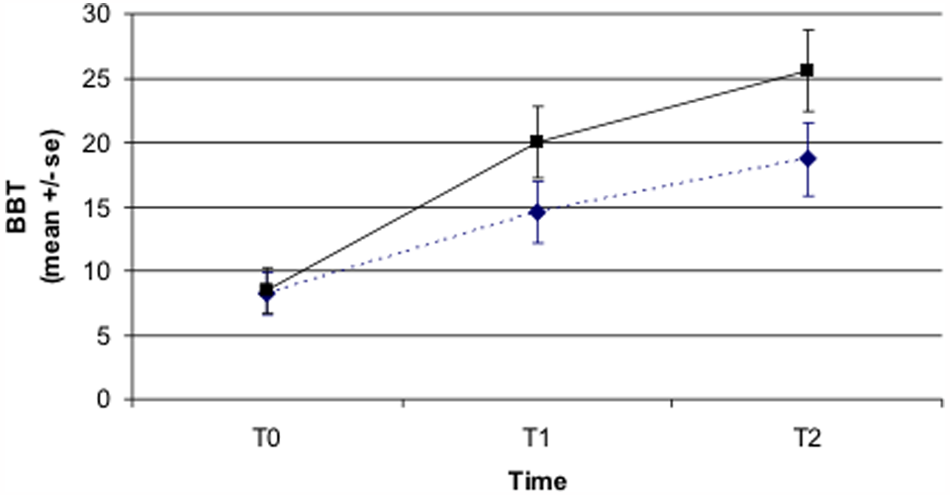
Trends over time for the BBT score in the control (diamond) and experimental (square) groups: statistically significant Treatment × Time interactions (GEE analysis) were found: T0–T1 (P = .003); T0–T2 (P = .01); T1–T2 (P = .76). Statistical significance set at P < .05 (in bold)
An additional analysis of the subset of patients with valid measurements at all time points and the last observation carried forward were performed to take into account the high rate of dropout. The results (not shown) agreed with the models fitted to the dataset of observed data.
Discussion
The significant Time × Treatment interaction that emerged from the GEE analysis of the BBT performance suggests a positive effect of the action observation approach on the functional dexterity of subacute stroke patients and demonstrates the potential of this treatment to implement traditional rehabilitation procedures.
Previous studies have shown that action observation appears to activate the motor system similarly to execution by generating an internal representation of action that may be a target for motor relearning.21,22 Experimental investigations in monkeys and humans have shed light on what the parietofrontal cortical circuit encodes and its possible functional relevance. According to the mirror neuron paradigm, the physiological mechanism aims at unifying perception and action and at transforming sensory representations of others’ behavior into motor representations of the same behavior within the observer’s brain (mirror mechanism). 23 The key rationale for action observation in stroke rehabilitation is to provide a means to recover lost function within the damaged motor network; so the critical condition is observation with intent to imitate the observed action. 24
A recent functional MRI–based study by Cantarero et al 25 further supports the hypothesis that action observation coupled with physical practice may enhance use-dependent plasticity both in healthy and stroke patients. In contrast with previous small studies of patients with chronic stroke,26,27 the present investigation was conducted on a relatively large population of subacute patients with action observation added to standard rehabilitation.
Multiple strategies were applied to reduce bias. Possible limitations include the choice of treating a relatively heterogeneous sample and including hemorrhagic and ischemic lesions. However, exclusion of hemorrhagic stroke would have probably reduced the external validity of the results and the clinical relevance of the research. The attrition rate was higher than expected but similar in the 2 groups, suggesting good feasibility of the experimental treatment protocol. Dropouts reduced the statistical power of the study. Nevertheless, the calculation of the required sample size was based on the expected group differences in the performance at the BBT, and a significant treatment difference was found. The choice of the BBT as principal outcome is related to the need for an activity-based instead of impairment-based (FM) measure. Lin et al 28 showed how the BBT could be considered more appropriate for evaluating the responsiveness and validity of 3 dexterous pretreatment and posttreatment functional measures after stroke. The BBT is also designed to show improvements in accuracy and speed of a single pattern of movement, whereas the FM reflects the ability to perform movements of various kinds, some of which are not likely to be used in daily routine and were not specifically trained in our patients.
A trend toward higher upper-limb function in the EG than the CG was also found 4 to 5 months after treatment ended. The improvement experienced during the intensive rehabilitation phase presumably encouraged upper-limb use, thus prompting further recovery. It is unlikely that the gap arises from later management. Both groups underwent a maximum of 90 days of inpatient rehabilitation, followed by outpatient treatment with 3 to 5 sessions lasting 1 hour in a week for up to 6 months. During the study time frame, none received innovative treatments such as robot-assisted rehabilitation.
The transfer of our findings from basic sciences (MNSs and action observation) to clinical rehabilitation makes our approach innovative. The focus on the very early phase of stroke recovery makes this research relevant to clinical practice, although spontaneous changes could account for some gains. On the whole, the simplicity of treatment, the lack of adverse effects, and the positive preliminary results support the use of this treatment in association with physiotherapy in clinical practice and perhaps for telerehabilitation. 29
Footnotes
Acknowledgements
We thank the following people for helping with data collection: Giorgetti Simonetta, Silvia Baronci, and Patrizia Nati (Rehabilitation Department ASL 3 Umbria); Michela Coccia, MD (Department of Neuroscience, Polytechnic University of Marche, Ancona); Federica Manghi and Chiara Iotti (Rehabilitation Department Universitari Hospital Parma); Sciarrini Francesco, MD, Saltalippi Milena, MD, Rellini Marusca, MD, and Bacuccoli Tatiana, MD (Rehabilitation Department ASL 2 Umbria); Faenza Manuela (Baggiovara Hospital AUSL Modena); Massimo Vallasciani, MD, Alessandra Giusti, MD (Stefano Rehabilitation Center Porto Potenza Picena, MC); Aliboni Sara, MD (Rehabilitation Unit Versilia Hospital, Lucca); Roberto Antenucci, Giuseppe Buonanno, Giuseppe Sacco, Anna Cassio, Rossella Raggi, Pellegrino Nicoletta, Dellabianca Silvia, and Santin Monica (Rehabilitation Unit Borgonovo Val Tidone Hospital Piacenza); Daniele Malgrati, MD, Alessandro Cattaneo, Erica Palazzini, Katia Franchina (Rehabilitation Unit, Casa di Cura S. Francesco, Bergamo); Donatella Bonaiuti (Neurorehabilitation Department, S. Gerardo Hospital, Monza); Francesco Ferraro, MD (Rehabilitation Department, A O “Carlo Poma” di Mantova-PRM di Bozzolo); Michele Scarazzato, MD, Barbara Decchi, MD, Luciano Bissolotti, MD, Cristiano Bara, Elena Castelnovo (Neurological Rehabilitation Unit Brescia); Mauro Mancuso, MD (Neurorehabilitation Unit, Misericordia General Hospital, Grosseto).
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
