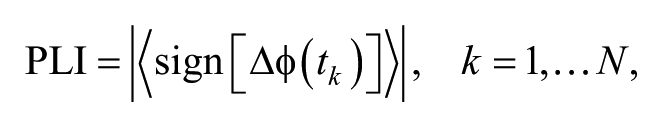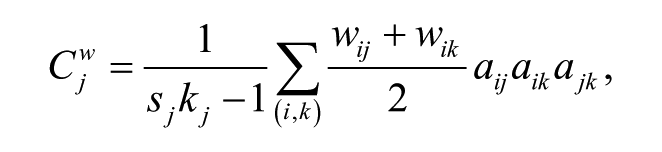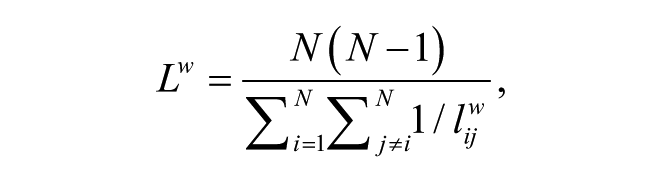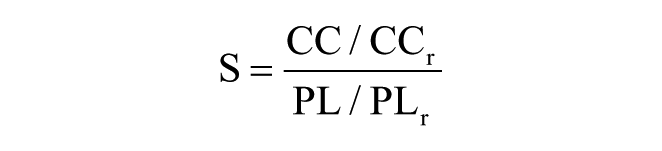Abstract
Background. Camera technique–based mirror visual feedback (MVF) is an optimal interface for mirror therapy. However, its efficiency for stroke rehabilitation and the underlying neural mechanisms remain unclear. Objective. To investigate the possible treatment benefits of camera-based MVF (camMVF) for priming prior to hand function exercise in subacute stroke patients, and to reveal topological reorganization of brain network in response to the intervention. Methods. Twenty subacute stroke patients were assigned randomly to the camMVF group (MG, N = 10) or a conventional group (CG, N = 10). Before, and after 2 and 4 weeks of intervention, the Fugl-Meyer Assessment Upper Limb subscale (FMA_UL), the Functional Independence Measure (FIM), the modified Ashworth Scale (MAS), manual muscle testing (MMT), and the Berg Balance Scale (BBS) were measured. Resting-state electroencephalography (EEG) signals were recorded before and after 4-week intervention. Results. The MG showed more improvements in the FMA_UL, the FMA_WH (wrist and hand), and the FIM than the CG. The clustering coefficient (CC) of the resting EEG network in the alpha band was increased globally in the MG after intervention but not in the CG. Nodal CC analyses revealed that the CC in the MG tended to increase in the ipsilesional occipital and temporal areas, and the bilateral central and parietal areas, suggesting improved local efficiency of communication in the visual, somatosensory, and motor areas. The changes of nodal CC at TP8 and PO8 were significantly positively correlated with the motor recovery. Conclusions. The camMVF-based priming could improve the motor recovery, daily function, and brain network segregation in subacute stroke patients.
Introduction
Mirror therapy (MT) is recognized as an effective adjunct treatment for improving the motor function of upper limb and abilities of daily life in stroke patients.1-4 A plane mirror usually used and vertically set in the sagittal plane of patients, while they are performing bilateral motor tasks.1,5,6 However, there are some technological limitations and disadvantages, such as posture pressure and lack of engagement, which hinder the treatment effects.7,8 To overcome these extrinsic limitations, camera technique–based mirror visual feedback (MVF) system has been proposed to provide novel interfaces.8-13 Feasibility and effectiveness of this approach have been demonstrated in both healthy populations and patients with peripheral nerve injury.8,9,11 However, high labor intensity and lack of self-discipline training still remain unsolved in these current novel interfaces.8,9 Furthermore, few studies have investigated the corresponding therapeutic effects in stroke patients.
MT is one of the priming techniques that is conducted prior to motor practice and preactivate the brain for subsequent exercises.3,14 Priming can enhance the effects of motor practice by promoting the activation of motor system and intercortical circuitries. 3 During MVF, stroke patients showed increased functional magnetic resonance imaging (fMRI) activities across widely distributed brain regions, such as the bilateral postcentral gyrus and precuneus, the ipsilesional primary motor cortex, and the contralesional supramarginal gyrus.15,16 MVF also presented the ability to normalize the interhemispheric asymmetry of movement-related beta desynchronization in stroke patients. 17 Besides the instant effects of MVF, MT training also showed evidences for brain activity reorganization in stroke patients. Chang et al 18 found that beta power (14-35 Hz) in stroke patients was changed for mirror observation after 4 weeks of MT. Bae et al 19 reported mu rhythm (8-13 Hz) suppression at C3, Cz, and C4 in subacute stroke patients after MT. fMRI studies revealed that the laterality index of the primary motor in stroke patients was improved after multiple MT sessions, presenting a shift in activation toward the ipsilesional hemisphere.20,21 MT also showed potentials for brain network reorganization. In healthy subjects, fMRI functional connectivity between premotor region and ipsilateral supplementary motor area was increased after 4-day MT training. 22 In stroke patients, Saleh et al16,23 found that MVF could modulate the functional/effective connectivity of the ipsilesional motor cortex. Given that the instantaneous effects could be consolidated through repeated rehearsal and therefore resulted in brain plasticity, 24 we hypothesized that the brain networks of stroke patients would be reorganized after sessions of MT training.
In the present study, a camera-based MVF (camMVF) that provided a systematic procedure was employed. Given that subacute patients achieved more from MT and a prominent recovery is within 6 months,2,25-27 we assumed that camMVF might be most suitable and effective in mediating neural reorganization in subacute patients. Thus, a randomized controlled study was designed to investigate the potential effects of camMVF based priming on subacute stroke rehabilitation.
We also aimed to test the effects of camMVF on brain network reorganization in stroke patients. Topological characteristics that reflect the efficiency of large-scale neural communication were compared before and after intervention. During spontaneous recovery after stroke, the motor execution network gradually shifted toward a random mode, involving a less optimized reorganization. 28 However, priming techniques, such as bilateral arm training with rhythmic auditory cueing (BACTRAC), have been found to be associated with increased small worldness and greater centrality, suggesting a more efficient network. 29 Therefore, we assumed that MT might also induce a more efficient topological reorganization, which might be related to the clinical outcomes.
Materials and Methods
Study Design and Participants
This study was an assessor-blinded, pretest-posttest, randomized, controlled study. All the clinical evaluations were conducted by an independent therapist before, and after 2 and 4 weeks of intervention. Electroencephalography (EEG) recordings were conducted by another independent researcher before and after the intervention. Eligible patients were stratified using motor deficit severity (according to the Fugl-Meyer Assessment Upper Limb score [FMA_UL], more impaired ≥35 and less impaired ≤34) 30 and side of lesion (left and right), 27 and assigned randomly to a camera-based MVF group (MG) or a conventional group (CG), after the baseline measurements (Figure 1). The allocation sequence was based on a computer-generated random number table. The randomization program and all the assignments were conducted by an independent researcher.
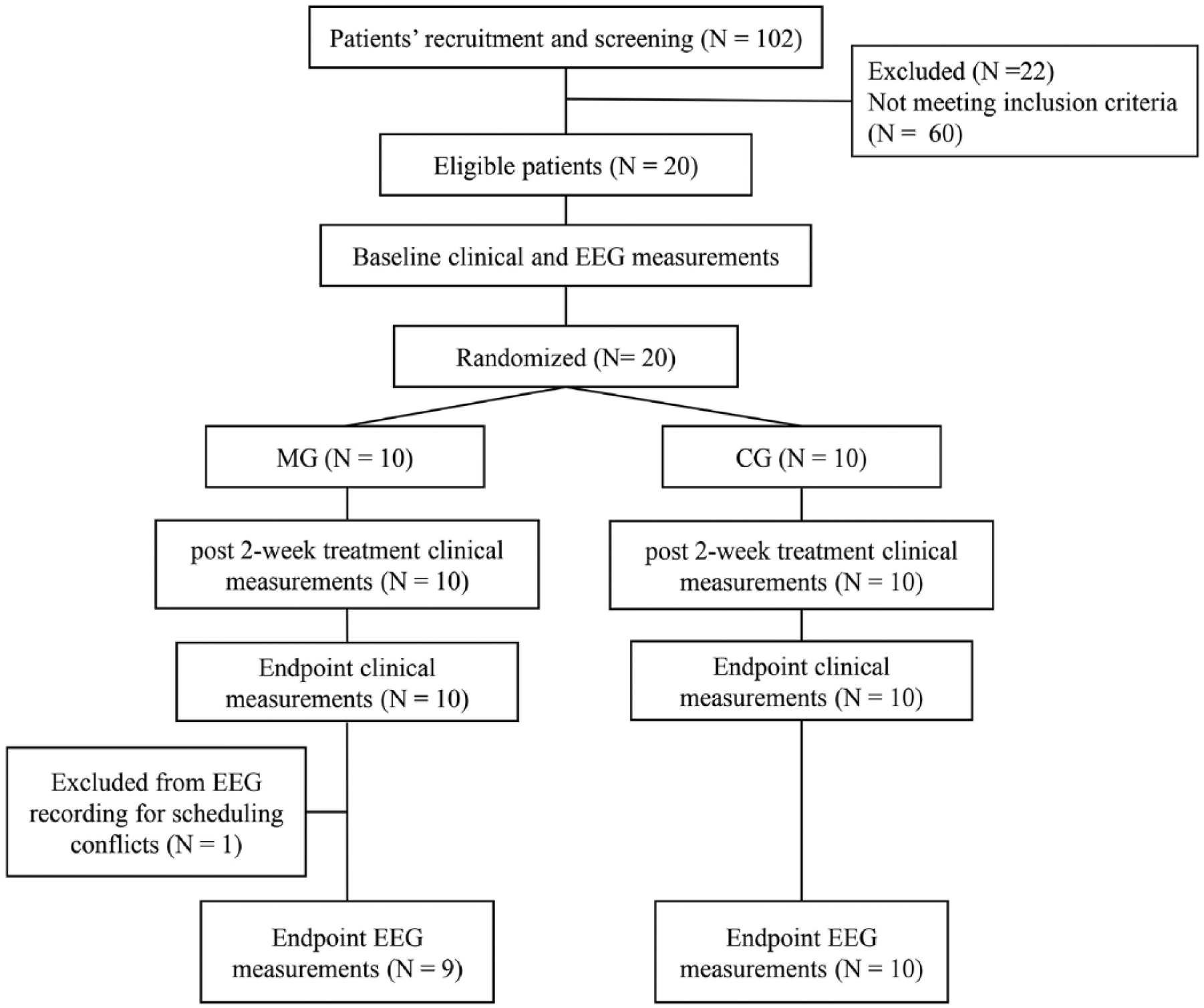
Flowchart of patients through the study. MG, camera-based mirror visual feedback group; CG, conventional groups.
Patients were recruited if they met the inclusion criteria: (1) computed tomography (CT) or MRI confirmed, unilateral ischemic stroke for the first time, (2) between 1 to 6 months after onset, (3) aged 25 to 75 years, and (4) the Brunnstrom stages ranging from I to V in the proximal and distal part. Patients with any of the following were excluded: severe cognitive disorder (Mini-Mental State Examination [MMSE] score <23), psychiatric disorder, aphasia, or severe spasticity in any joints of the affected upper limb (modified Ashworth scale [MAS] >2).
To provide an appropriate and reliable model, an estimation of sample size was carried out for this study. According to the previous studies in the field,5,6,20,27 the speculated effect size of approximately 0.45 for the main trial was anticipated. Thus, a sample size of 20 (10 per group) was estimated for the planned main trial with a power of 80% and type I error rate of 5%, using the stepped rules of thumb for pilot trials. 31 The study was approved by the Institutional Review Boards of Huashan Hospital (KY2017-230), and the trial was registered on the Chinese clinical trial registry (ChiCTR1800015674). All patients were informed of the nature of the study and signed informed consent forms prior to the participation.
Intervention
All patients received 1.5 hours of training per day beyond their routine rehabilitation in hospital, 5 days per week for 4 weeks.
camMVF Intervention
A customized camMVF was employed to present MVF instead of a real mirror (Supplementary Figure S1). It was a prism shape (1200 mm × 940 mm × 702 mm) with a height adjustable platform. A tilted 23.8-inch LED screen was fixed on one face of the prism to provide the visual feedback, and there was a rectangular opening at the bottom of the screen. Two cameras were mounted on the lateral edge of the prism to capture the movements of hands, wrists, and forearms. During the training, the pictures of unaffected hand and its mirror image were shown on the screen in front of the patients. It was shown previously, that the reflection equipment in coronally front of patients could reduce posture problems associated with MT, which typically are a shifting of weight and leaning toward the unaffected side, with possible disadvantageous effects on therapy efficacy. 7 We therefore expect the camMVF apparatus used in this study to likewise reduce cervical posture tension and weight shifting. To provide a standardized and systematic procedure for MT, the camMVF entailed 2 sections: (1) simple motor training, which contained gross and fine motor tasks, including grasping, finger opposition, pinching, wrist extension/flexion, forearm supination/pronation and so on, and (2) task-based motor training, where texture-varying objects like a glass, wooden blocks, tennis ball, and duster were used to provide task-based training, including specific reaching or placing tasks. Furthermore, video-guided motor training and verbal instruction were employed for systematic procedure.
As a visual input based priming strategy, we used the camMVF to preactivate the motor related cortex,3,32 and provided subsequent motor exercises. Thus, the patients in the MG received 1 h MT using the camMVF prior to 30-minute task-based training on hand function. An experienced therapist helped the patients to relax using stretching technique before training. Five out of 8 prespecified tasks were chosen by the therapist (6 simple motor tasks: forearm pronation/supination, wrist extension, thumb abduction, clenching and opening, opposition, and finger extension; 2 task-based motor tasks: grasping and releasing tennis balls and different dusters). Each task would repeat 30 times per session for two sessions. After priming, the therapist would administrate stretching and massage to help the patients relax again. Subsequently, task-based trainings on hand function, which varied depending on the chosen motor tasks, were provided by the therapist for half an hour.
Conventional Intervention
Patients in the CG received 1.5-hour dosage-equivalent (intensity and duration) rehabilitation therapies as the MG. Exercise prescription of the CG was in accord with the MG, including repetitive passive/active motor tasks and task-based motor training without MVF (1 hour, 5 out of 8 prespecified tasks), stretching/massage before and after the training, and subsequent task-based training on hand function (30 minutes).
Outcome Measurements
This study contained clinical measurements (including motor impairment, muscle status, daily function, and balance control) and EEG signal recordings.
Clinical Outcomes
As primary outcomes, the FMA_UL subscale was used to assess the motor impairment of the upper limb. The FMA_UL (maximum: 66) emphasized on movements and reflexes of the upper limb by using a 3-point ordinal scale from 0 to 2 (0 = cannot perform; 1 = can perform partially; 2 = can perform fully). Moreover, the score of wrist and hand of the FMA (FMA_WH, maximum: 24) was also calculated to evaluate the recovery of hand function. The MAS and manual muscle testing (MMT) were used to assess the muscle tone and strength of the wrist flexor and extensor respectively, where a higher score indicated more severe spasticity or stronger muscle. The Functional Independence Measure (FIM) was employed to measure improvements in daily function. The FIM (maximum: 126) measures independent functions by using a 7-point scale (1 = total assistance; 7 = complete independence), including 6 subscales (self-care, sphincter control, transfers, locomotion, communication, and social cognition). The Berg Balance Scale (BBS) is a 14-item scale that assesses the balance ability and control.
EEG Recording and Preprocessing
Resting-state EEG signals were recorded before and after 4-week intervention (before the training on the first day and 1 day after the training). Patients were sitting in a comfort chair and required to keep eyes closed. The EEG signals were continuously recorded for 5 minutes using a 64-channel Ag/AgCl electrode cap (EasyCap, Brain Products GmbH, Germany) connected to a BrainAmp MR Plus amplifier (Brain Products GmbH, Germany) at a sample rate of 1000 Hz. A ground electrode was placed at AFz. All electrodes were referenced to FCz, with impedance kept below 5 kohm.
BrainVison Analyzer software (v2.0, Brain Products GmbH, Germany) was employed to perform EEG data preprocessing. A notch filter at 50 Hz and an ocular artifact procedure based on the independent component analysis (ICA) was performed to mitigate the effect of the power line noise and unexpected eye blinks. For patients who had slight and infrequent eye blinks during the recording (N = 2, one in the CG before the intervention, and the other in the MG before the intervention), one independent component related to ocular movement was removed after ICA. Motion artifacts (amplitude value exceeding 200 μV or gradient value over 50 μV/ms) were detected by semi-automatically detecting. EEG signals were filtered into delta (1-4HZ), theta (4-8 Hz), alpha (8-13 Hz), and beta (13-30 Hz) bands. A continuous 30-second artifact-free segmentation at approximately middle of the recording period was selected. The EEG data from the stroke patients with left side lesion were flipped. Therefore, the right hemisphere was defined as the ipsilesional hemisphere, and the left was defined as the contralesional hemisphere.
Network Analysis
Construction of Resting-State EEG Network
The EEG network was defined by phase lag index (PLI) 33 using HERMES toolbox. 34 The PLI, which can measure statistical interdependence between 2 time series, is calculated as
where ∆ϕ(tk) is the phase difference at time tk. Compared with conventional methods like phase synchronization index, PLI is less affected by the volume conduction and active reference. 33 Moreover, it could construct stable brain networks from the EEG data at a length of ≥12 seconds. 35 Therefore, in this study, the EEG signals were not re-referenced and 30-second segments were selected to calculate PLIs. For each subject and time point (pre- and postintervention), PLIs at all bands between all possible pairs of EEG signals were calculated, and a correlation matrix was constructed for further network analysis. The nodes in the network represented EEG channels (n = 64), and the weight of the connection between 2 nodes was the PLI.
Network Topology
A sparsity thresholding method, which might reduce the false positives due to the uncertainty of weak link, 36 was applied to convert the correlation matrix to a weighted adjacent matrix. Sparsity is the ratio of edges preserved in the adjacent matrix. Median sparsity in brain network studies was typically located around 0.1 to 0.2.37-39 Therefore, in the following topological analysis, the networks with the sparsity from 0.1 to 0.2, at intervals of 0.02 (including 0.1, 0.12, 0.14, 0.16, 0.18, and 0.2) were studied. Clustering coefficient (CC; including global and nodal CC), characteristic path length (PL), and Small-World Index (S) were computed in this study to quantify the properties of a network. 28
The global CC quantifies the local efficiency of a network communication, which measures functional segregation.28,40 Nodal CC is the weighted CC of a single node j (
where sj and kj represent the strength and the degree of node j, wij is the weight between nodes i and j, and aij is the element in ith line and jth column of the adjacent matrix. The global CC was calculated by averaging the nodal CC of all nodes.
The PL (an index of functional integration) is the average minimum number of connections that link any two nodes of the network, which describes the global efficiency of a network.28,40 The characteristic PL (Lw) is defined as
where
The S can quantify the overall small-worldness of the network. 41 It is defined as
where CCr and PLr represent clustering coefficient and path length of equivalent random networks with same degree distribution.
Statistical Analyses
Statistical analyses were performed using SPSS (Version 24). The underlying model assumptions were thoroughly checked by the Shapiro-Wilk’s test for normality of distribution, and the Levene’s test for the homogeneity of variances. Baseline characteristics of the patients in the MG and the CG were compared by performing chi-square tests (for categorical variables, including gender and side of paralysis), one-way analysis of variance (for continuous variables with normal distribution, including ages and days after stroke onset) and Mann-Whitney U tests (for Brunnstrom stages as it did not follow a normal distribution). The differences of the outcomes, including the FMA-UL, the FMA-WH, the FIM, and the BBS, between the groups at each of the trial time points were analyzed using mixed-design 2-way repeated-measures analysis of variance (ANOVA), taking time (3 levels: preintervention, after 2 weeks and after 4 weeks of intervention) as the within-subject factor and group (2 levels: MG and CG) as the between-subject factor. The muscle status (the MMT and the MAS) at each of the trial time points was compared between the two groups with the Friedman test for the non-normal distribution and data were then further analyzed using the Wilcoxon signed-rank test. Mann-Whitney U test was used to compare the between-group differences of muscle status. Two-way repeated ANOVAs taking time (2 levels: pre- and postintervention) as the within-subject factor and group as the between-subject factor were performed on the network parameters (global/nodal CC, PL, and S). Potential correlations between the changes in network properties and improvements of motor function (the FMA_UL and FMA_WH) were examined using Pearson’s correlation. The Bonferroni correction was used to adjust p values for multiple testing of clinical outcomes. Results are presented as mean with standard deviations (SD). The significant level was set at P < .05 with a 2-sided test.
Results
Twenty stroke patients matching the criteria (17 male and 3 female; age 58.30 ± 12.86 years; days post stroke onset 72.45 ± 35.84 days) were recruited from Huashan Hospital Fudan University (N = 13) and the first Rehabilitation Hospital of Shanghai (N = 7), and assigned randomly to the MG (N = 10) or the CG (N = 10). All the patients completed the intervention successfully without adverse events. One patient in the MG was not able to complete the EEG data recording after the intervention due to the conflicting arrangement in the hospital. There were no significant differences between the 2 groups in the baseline characteristics (Table 1).
Baseline Characteristics of Patients.
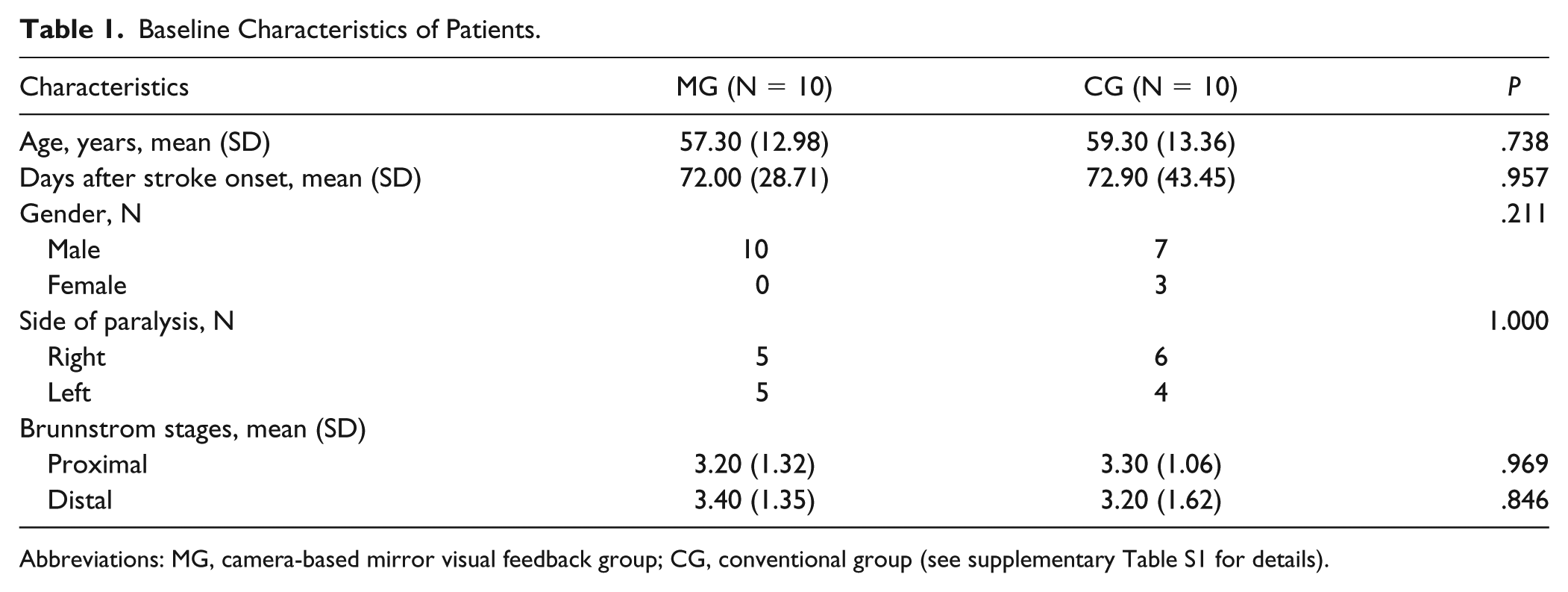
Abbreviations: MG, camera-based mirror visual feedback group; CG, conventional group (see supplementary Table S1 for details).
Clinical Outcomes
The statistical results of clinical scores on motor impairment, daily function, and balance control are shown in Table 2. Significant time × group interactions were found on the FMA_UL (F2, 36 = 4.045, P = .046) and FAM_WH (F2, 36 = 7.895, P = .007). Post hoc analyses (Bonferroni adjusted) indicated that the FMA_UL and FMA_WH scores were comparable between the 2 groups before intervention (P = .321 and P = .364, respectively), and the scores in the MG were significantly higher than the CG after 4 weeks intervention (P = .041 and P = .046, respectively); however, group differences were not significant after 2 weeks intervention (P = .095 and P = .150, respectively). On the other hand, constant significant improvements in motor function were observed at each assessment only in the MG but not in the CG, although both groups showed significant improvements in motor function, balance, and ability of daily life after 4 weeks of intervention compared with baseline (Supplementary Tables S2 and S3). The FMA_UL and the FMA_WH in the MG were improved significantly either in the first 2 weeks (Bonferroni adjusted, P = .001 and P = .005, respectively) or in the latter 2 weeks intervention (Bonferroni adjusted, P = .043 and P = .004, respectively). However, in the CG, the FMA_UL was only improved significantly in the latter 2 weeks (Bonferroni adjusted P = .015) and the FMA_WH was only improved significantly in the first 2 weeks (Bonferroni adjusted P = .014).
Descriptive and Inferential Statistics for Motor Impairment, Daily Function, and Balance Control.
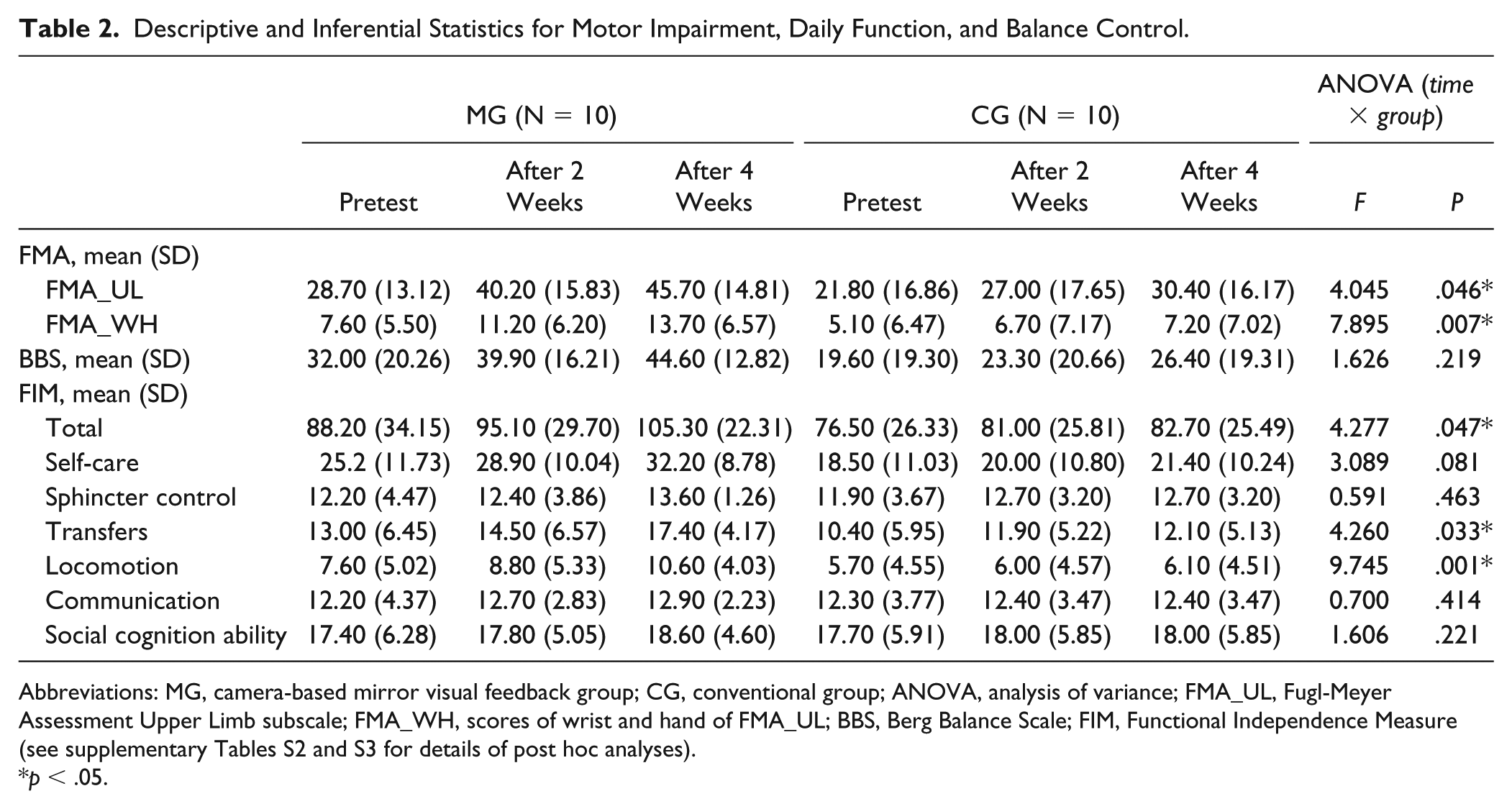
Abbreviations: MG, camera-based mirror visual feedback group; CG, conventional group; ANOVA, analysis of variance; FMA_UL, Fugl-Meyer Assessment Upper Limb subscale; FMA_WH, scores of wrist and hand of FMA_UL; BBS, Berg Balance Scale; FIM, Functional Independence Measure (see supplementary Tables S2 and S3 for details of post hoc analyses).
p < .05.
Significant time × group interactions were also observed on the total FIM (F2, 36 = 4.277, P = .047), and the transfer (F2, 36 = 4.260, P = .033) and the locomotion (F2, 36 = 9.745, P = .001) subscales. Further analyses (Bonferroni adjusted) suggested that the scores of the total FIM, the transfer, and the locomotion were higher in the MG than the CG after 4 weeks intervention (P = .049, P = .021, and P = .030, respectively), although they were comparable in the 2 groups before the intervention (P = .402, P = .361, and P = .387, respectively) and after 2 weeks of intervention (P = .272, P = .340, and P = .223, respectively). No significant effects of camMVF were found on balance control, muscle strength, or muscle tone (Supplementary Tables S4-S7).
Altered Network Properties by Intervention
The 2-way repeated-measures ANOVA on the global CC of resting EEG network demonstrated that the group × time interaction was consistently significant under different sparsity thresholds in the alpha band (P < .05 for sparsity from 0.12 to 0.2; marginally significant for 0.1, P = .055). Post hoc analyses suggested that the global CC was increased in the MG after intervention, which suggested improvements in brain network segregation after the camMVF priming (Figure 2A). However, no significant alterations were observed in the CG, and even an opposite trend (reduction of the global CC) was found (Figure 2A). No significant effects on global CC in other bands were obtained. Moreover, there were no significant differences on PL and S (Figure 2B and C).
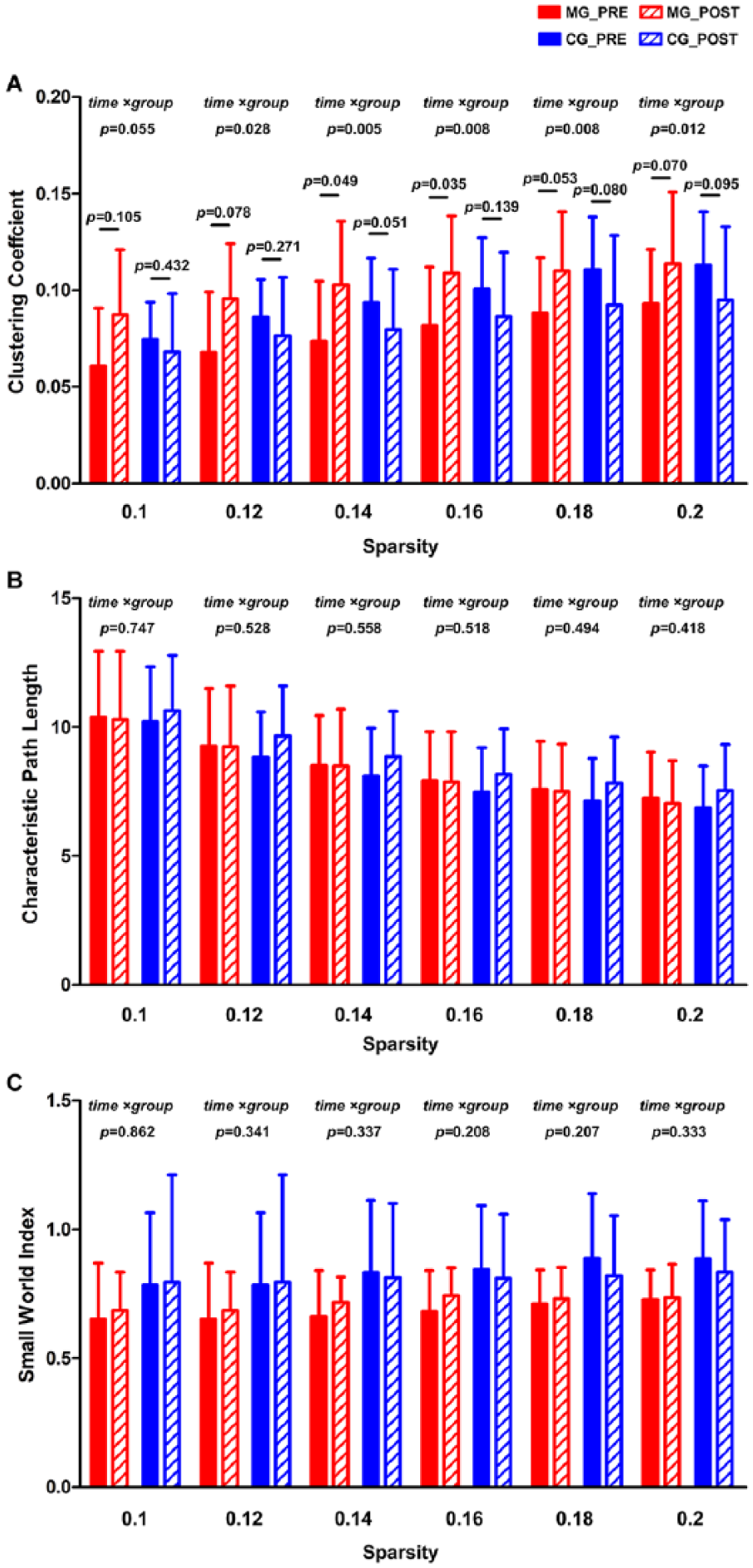
Comparison of the global clustering coefficient (A), characteristic path length (B), and Small-World Index (C) of the resting-state EEG network in the camera-based mirror visual feedback group (MG) and conventional group (CG) at pre- and postintervention.
To explore the brain regions with significant group × time interaction, analyses were conducted on the nodal CC of each electrode. We found that group × time interaction was significant mainly in the ipsilesional occipital and parietal (O2, PO4, PO8, P8), temporal (TP8, T8, FT8), and central (C2) areas, and the contralesional central (C1, C3, C5) and parietal (P3, P5, P7) areas (Figure 3). Consistently with the global CC, the nodal CC of these electrodes was increased significantly in the MG after intervention, while no increments were found in the CG (Figure 3).
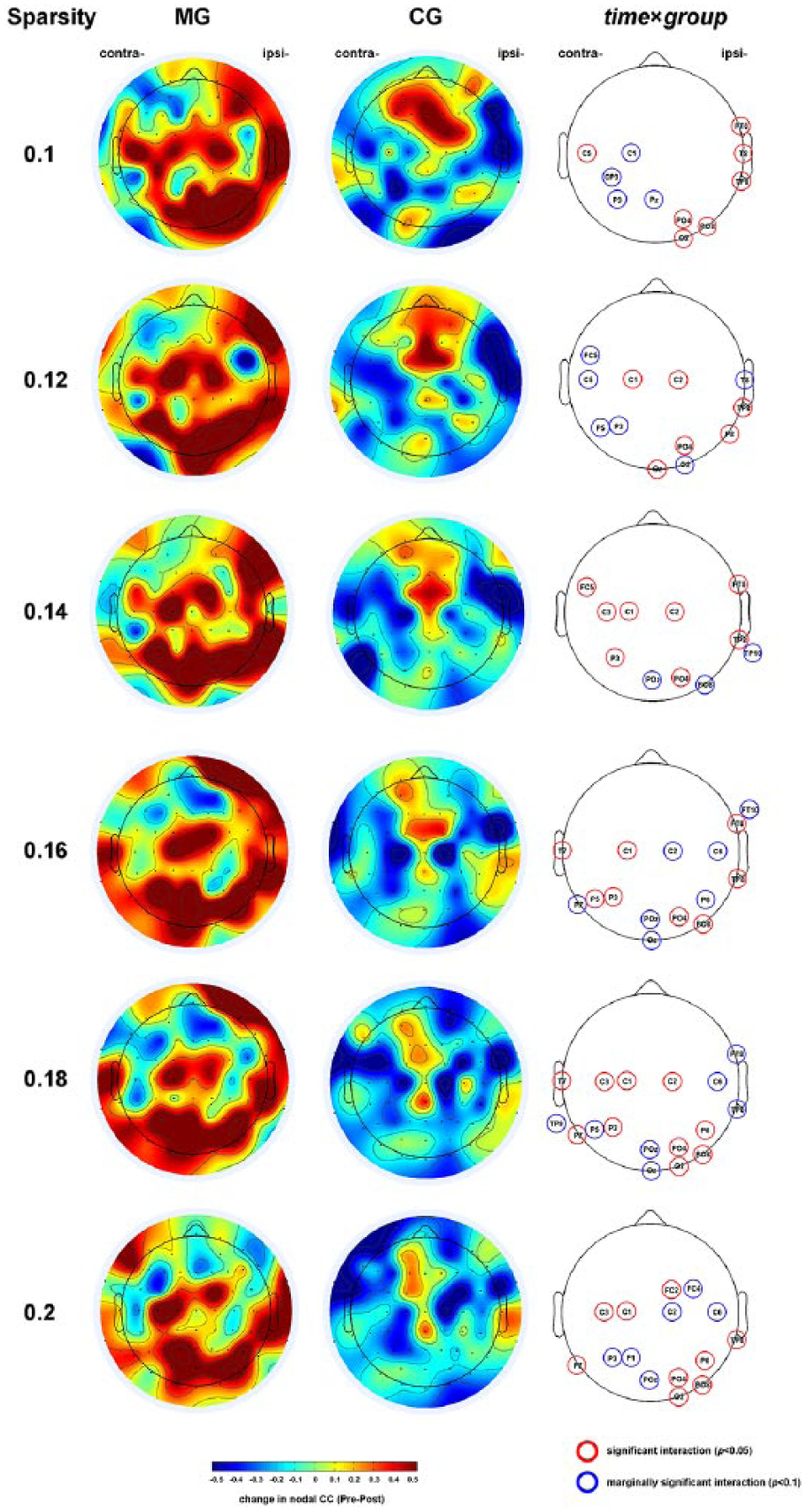
Alterations of the nodal clustering coefficient (CC) of the resting-state EEG network through intervention in the camera-based mirror visual feedback group (MG) and conventional group (CG). Topographic maps in the first 2 columns showed average differences of nodal CC before and after intervention within the MG and CG under different sparsity thresholds. Topographic maps on the right column highlight the electrodes with significant (P < .05, red circles) or marginally significant time × group interactions (P < .1, blue circles). The data from left-side stroke patients were flipped right. ipsi-, ipsilesional side; contra-, contralesional side.
Relationship Between Changes in Network and Motor Recovery
The relationships between the changes in CC and motor function were measured in global and nodal levels. In the nodal level, we only tested the nodes with significant time × group interactions (Figure 4). There were no significant correlations between the global CC and motor function (all P > .164). However, the changes in nodal CC at TP8 were significantly positively correlated with the improvement of FMA_UL and FMA_WH scores at sparsity thresholds from 0.14 to 0.2 (all r > 0.489, P < .03). In addition, a significant positive correlation was observed between the changes in nodal CC at PO8 and the improvement of FMA_WH score at the threshold 0.2 (r = 0.478, P = .039).
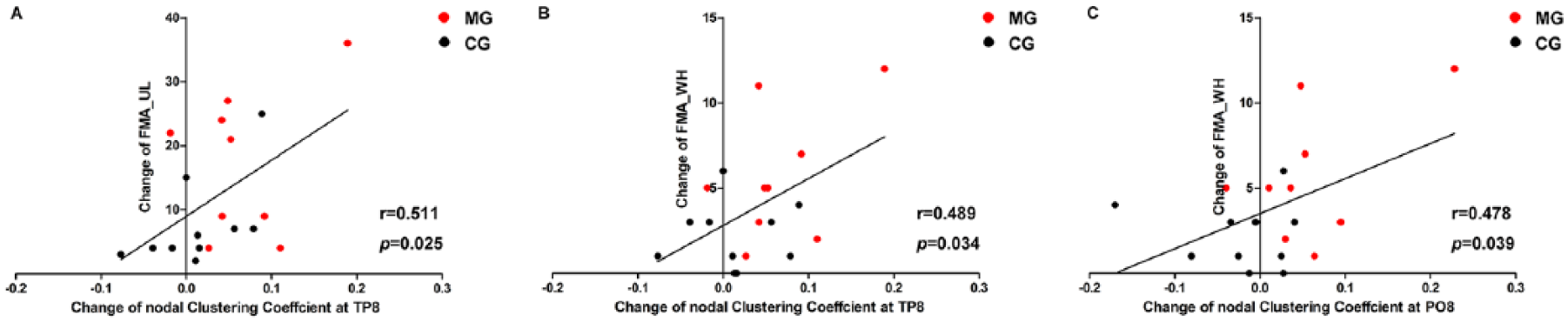
Relationships between the changes in nodal clustering coefficient (CC) and motor recovery under sparsity threshold of 0.2. (A) The change of nodal CC at TP8 was significantly positively correlated with the improvement of FMA_UL score. (B) The change of nodal CC at TP8 was significantly positively correlated with improvement of FMA_WH score. (C) The change of nodal CC at PO8 was significantly positively correlated with the improvement of FMA_WH score. FMA_UL, Fugl-Meyer Assessment Upper Limb subscale; FMA_WH, scores of wrist and hand of FMA_UL; MG, camera-based mirror visual feedback group; CG, conventional group. Supplementary Table S8 shows the results of correlation between the changes of nodal CC at TP8 and motor recovery under all the sparsity threshold.
Discussion
This study first provides tentative evidence that the visual input–based priming via camMVF prior to hand function exercise had superiority over the conventional therapy in reduction of upper limb motor impairment and improving daily function in subacute stroke patients. The camMVF also showed specific effects on brain network segregation. The CC of the resting EEG network in the alpha band was increased globally after intervention in the MG but not in the CG, and it tended to increase in the ipsilesional occipital and temporal areas, and bilateral central and parietal areas.
Improvements in Motor Recovery and Daily Function
As indicated by the FMA_UL and FMA_WH, the MG showed more improvements of upper limb and hand motor function comparing with the CG, which are in agreement with previous studies on stroke patients in subacute/acute stages.2,42,43 Moreover, the treatment effects of camMVF seemed constant, as suggested by the continuous improvements in motor recovery either at the first or the latter 2 weeks. One possible interpretation was that camMVF had a strong potential to enhance the perception of affected limb via combining visual input and motor imagery, which hindered the development of learned non-use in subacute stroke patients.2,4 In addition, camMVF could have similar abilities with MVF to promote and rebalance the cortical activation.15,17 Thus, we speculated that in our priming strategy, camMVF might preactivate the motor system, which enhanced the treatment effects of the following hand function exercise.
A significant improvement in daily function was demonstrated in the MG comparing with the CG, in line with previous studies. 4 Specifically, the MG obtained more benefits for the ability of transfer and locomotion than the CG. An explanation is that the improved motor function of upper limb might reduce the assistance during daily transfer and ambulation. Haptic stimulations also might play a role. Lin et al 5 reported that combination of MT and sensory stimulation could improve the ambulation function by eliciting an interlimb reflex response. Thus, the sensory input provided by texture-varying dusters during task-based motor training might contribute to the coordination between lower and upper limbs.5,44 However, we did not find significant improvements in the FIM subscale of self-care. It has been found that the self-care function could be benefited from MT only for the patients in chronic stage, 6 but not in subacute stage.2,43 This may be due to the short treatment duration and the fact that patients were in subacute stage, when assistance was provided by carers during self-care.
Topological Alterations of Brain Network
Brain functions depend on efficient neural communication within the brain networks. 45 Human brain functional networks usually have high CC (segregation) and short PL (integration) to promote efficiency of local and global information processing, respectively. 46 After stroke, the balance between the local segregation and global integration is disrupted, and a shift toward random network has been observed.28,40 This nonoptimal network might result from random development of new neural connections after stroke. 47 Although new neural connections may compensate for impaired motor pathways and contribute to stroke recovery, their random pattern might reduce the efficiency of network communication. 28 This is in line with our observation in the CG, which showed a trend of reduction in global CC, manifesting decreased functional segregation of brain network with conventional rehabilitation. However, the functional segregation of brain network could be improved by the camMVF as suggested by the significant increased global CC in the MG. The efficiency of camMVF in improving functional segregation of brain networks might be associated with the preactivation of the motor system prior to physical activities. 3 We speculated that the preactivated cortex could promote plastic reorganization and facilitate motor pathway in response to subsequent physical exercise, which might shift the brain network toward a high efficient pattern and enhance motor recovery in subacute stroke patients.3,32
Although EEG based topology analyses are not precise enough to investigate the space-varying connectivity, each node is ideally recognized as represent of brain region. 48 The nodal CC was found to increase in the ipsilesional occipital and temporal areas, and the bilateral parietal areas in the MG after intervention, suggesting a tendency that the local efficiency of communication was improved in visual and somatosensory regions. fMRI studies showed that the activity in the visual and somatosensory related regions, including visual cortex, posterior parietal cortex, and superior temporal gyrus, was upregulated during MVF.8,14,49,50 Therefore, it is possible that network topological changes in these areas observed in the current study might be accounted by long-term effects of camMVF priming. 24 Notably, visual cortex and temporal lobe are included in the ventral stream of visual system, which plays a crucial role in recognition and generating attention to limbs.50,51 We speculate that the ventral stream with projections from visual cortex to temporal lobe might be the structural basis for the long-term effects of camMVF priming. Thus, camMVF based priming might restore motor function via enhancing the awareness of affected limbs by improving the local information transmission efficiency in the ventral stream. Our hypothesis is supported by the significant positive correlation between the increment of nodal CC at PO8 and TP8 and motor recovery. MT has been demonstrated its capability in promoting the activation of the ipsilesional primary motor and/or premotor areas as instant or neuroplasticity effects, which reveals the benefits on motor recovery.10,11,20 We also observed increased tendency of nodal CC in the bilateral central regions in the MG, suggesting that local efficiency of communication within the motor network was improved after the camMVF based priming. This study suggested that the effects of camMVF on the motor network not only restrict in the ipsilesional hemisphere but also involve in the contralesional counterpart. The increased nodal CC in the contralesional motor regions induced by camMVF priming might be associated with the compensation from the contralesional hemisphere, which has been believed as a possible strategy for initial stage of recovery in subacute stroke patients.52,53
It should be noted that the effects of camMVF on network topology were significant only in the alpha band. Caliandro et al. suggested that the CC and small-worldness were affected by stroke not only in the alpha band but also in the delta and theta bands. 40 However, in our study, no significant differences were observed between the two groups in these two bands. Our results suggested that the alpha network in the resting state might be more sensitive to the camMVF-based priming. One possibility was that the alpha band was predominant during wakeful relaxation with closed eyes. 54 Besides, the alpha oscillations could be phase locked between widely separated cortical regions, which played an active role in network coordination and communication. 55
Limitations and Future Directions
There are some limitations in this study. First, although a sample size estimation was employed, the sample size is small and therefore hinders the power of statistical analyses. Second, no multiple testing corrections were applied for the analyses of nodal CC for the initial exploration. However, it should be noted that significant nodes (uncorrected P < .05, Figure 3) tend to form clusters which might not be accounted for random fluctuation. Cluster-based permutation test, 56 which is a nonparametric method to reduce the family-wise error rate, can further illustrate this observation (see Supplementary Material). Third, the study population was in subacute stage, when spontaneous recovery would be an interference and when patients had difficulties in cooperating with long-time EEG recordings. A long-term follow-up study and longer artifacts-free segments for network constructions are needed in the future for more valid inference in subacute stroke patients. Furthermore, in the present channel-based connectivity study, each node is assumed to be dominated by activation from the underlying brain region. Although source reconstruction allows for a reduction in the volume conduction bias present in scalp recordings, solution of the inverse problem relies on assumptions that potentially do not completely remove volume conduction bias.57,58 Future studies may adopt other imaging modalities, for example, fMRI, and appropriately designed motor tasks to justify the topological changes and show different motor networks after camMVF from more aspects. 59
Conclusion
In conclusion, to our knowledge, this study is the first to demonstrate the benefits of priming via camMVF combining task-based training on hand function for improving motor function of upper limb and hand, and daily function (especially for the ability of locomotion and transfer) in subacute stroke patients comparing with conventional therapy. In addition, an enhanced brain network segregation was reported after the intervention of the camMVF for the first time.
Supplemental Material
Revised_Supplementary – Supplemental material for Camera-Based Mirror Visual Input for Priming Promotes Motor Recovery, Daily Function, and Brain Network Segregation in Subacute Stroke Patients
Supplemental material, Revised_Supplementary for Camera-Based Mirror Visual Input for Priming Promotes Motor Recovery, Daily Function, and Brain Network Segregation in Subacute Stroke Patients by Li Ding, Xu Wang, Shugeng Chen, Hewei Wang, Jing Tian, Jifeng Rong, Peng Shao, Shanbao Tong, Xiaoli Guo and Jie Jia in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported in part by the National Key R&D Program of China (2018YFC2002300 and 2018YFC2002301) and the National Natural Science Foundation of China (No. 61771313).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.