Abstract
Background. In healthy adults, hand movements are controlled largely by the contralateral primary motor cortex. Following amputation, however, movements of the intact hand are accompanied by increased activity in the sensorimotor cortices of both cerebral hemispheres. Objective. The authors tested whether use of the intact hand reactivates the cortical territory formerly devoted to the now missing hand and whether these effects can be augmented by motor imagery (MI) and/or exposure to illusory visual “feedback” (VF) of the absent hand created with a mirror. Methods. Functional magnetic resonance imaging (fMRI) was used to delineate the boundaries of normative sensorimotor hand representations in healthy controls. Brain activity from 11 unilateral hand amputees was recorded while they performed aurally paced thumb–finger sequencing movements with their intact hands under 4 conditions: (1) motor execution of the intact hand alone (ME), (2) ME with corresponding MI of the amputated hand, (3) ME with VF of the amputated hand, and (4) ME with MI and VF. Results. Intact hand movements increased activity specifically within the former sensorimotor hand territory during all conditions, an effect that may be attributable to decreased levels of interhemispheric inhibition and/or use-dependent functional reorganization following amputation. This effect was not significantly increased by the addition of VF and/or MI of the amputated hand. However, in amputees, MI was associated with an expansion of this ipsilateral response into parietal, premotor, and presupplementary motor areas. Conclusion. Active engagement of the intact hand may be critical for therapies seeking to stimulate the former hand territory.
Keywords
Introduction
There is considerable evidence that the functional organization of cortical sensory and motor representations is activity dependent.1,2 Studies of nonhuman primates reveal training-related expansions in primary sensory 3 and motor 4 maps located contralateral to the involved effector. Conversely, lesions of the peripheral nervous system that disrupt the flow of afferent and efferent signals, including those accompanying limb amputation, lead to extensive reorganizational changes in the contralateral sensory5,6 and motor cortex.7,8 The result is that some primary sensory neurons in the now deafferented territory become responsive to tactile stimuli applied to somatotopically adjacent regions of the residual forelimb or face. 9 Years after amputation, microstimulation of motor neurons in areas that formerly targeted amputated hand muscles evoke movements of the residual forelimb or shoulder but not the face.8,10
Evidence from noninvasive functional neuroimaging studies suggests that similar reorganizational changes occur in humans following hand amputation. Postinjury, the sensory representation of body surfaces proximal to the injury, 11 and/or the face, 12 may intrude into the former sensory hand territory. Likewise, evidence suggests that amputees show an expanded motor representation of muscles of the residual limb 13 and a medial shift in the representation of facial muscles. 14 The interpretation of this evidence has, however, recently been questioned. 15 Amputees also exhibit a larger contralateral sensory representation of the intact hand that may result from increased use. 16
Amputation also affects the ipsilateral hemisphere. Whereas healthy adults sometimes show small increases in ipsilateral premotor cortex activity during hand movements, 17 amputees manifest large ipsilateral increases in sensorimotor cortex activity when using the intact hand.13,18,19 These changes may reflect an immediate reduction in normal levels of interhemispheric inhibition (IHI) following unilateral deafferentation 20 and/or experience-dependent changes in the representation of the intact hand associated with increased use. 21 It is important to note that it remains unknown whether these increases are localized to the portion of the sensorimotor cortex formerly devoted to the now absent hand (ie, the former hand territory).
Despite considerable evidence for activity dependence, other findings suggest persistence of a cortical representation of the amputated hand. 22 Attempts to move amputated segments of the limb volitionally are associated with selective increases in activity of severed nerves previously targeting those specific muscles of the missing hand23,24 and are accompanied by distinct patterns of electromyographic activity. 25 Transcranial magnetic stimulation of the motor cortex contralateral to the amputation is known to evoke “phantom” sensations of movement in the absent limb.25-29 Phantom sensations can also be elicited in some patients by exposing them to an illusion of the missing limb30-32 created by reflecting movements of the intact hand in a mirror. 33 This illusory visual feedback (VF) 34 and motor imagery (MI) of the amputated hand30-32 are reported to increase contralateral sensorimotor activity. However, it remains unknown whether these effects occur precisely within the former sensorimotor hand territory. This is relevant because a growing number of reports indicate that mirror therapy reduces pain in unilateral amputees 35 and complex regional pain syndrome patients 36 and increases function after stroke,37-39 hand, 40 or brain surgery. 41 Yet an understanding of the neural mechanisms underlying these effects is presently lacking (see the review by Moseley et al 42 ).
We tested whether former hand territory (defined functionally on the basis of independent control data) activity is modulated by movements of the intact hand performed alone or with accompanying MI and/or illusory VF of the absent hand. A positive result would indicate a means of effectively stimulating cortical representations even in the absence of contralateral hand movements. Furthermore, it would suggest that experience-dependent changes in primary sensorimotor representations might account for the reported therapeutic benefits of mirror therapy.
Material and Methods
Participants
A total of 11 adult unilateral upper-limb amputees (3 women), averaging 50 years of age (range, 25 to 70 years), and 11 healthy yoked controls consented to participate in the approved protocol (Table 1). Amputations occurred at an average of 25 years before testing (range, 5 to 42 years). Each control performed movements with the same hand (left, right) as his or her yoked patient.
Selected Demographic Information
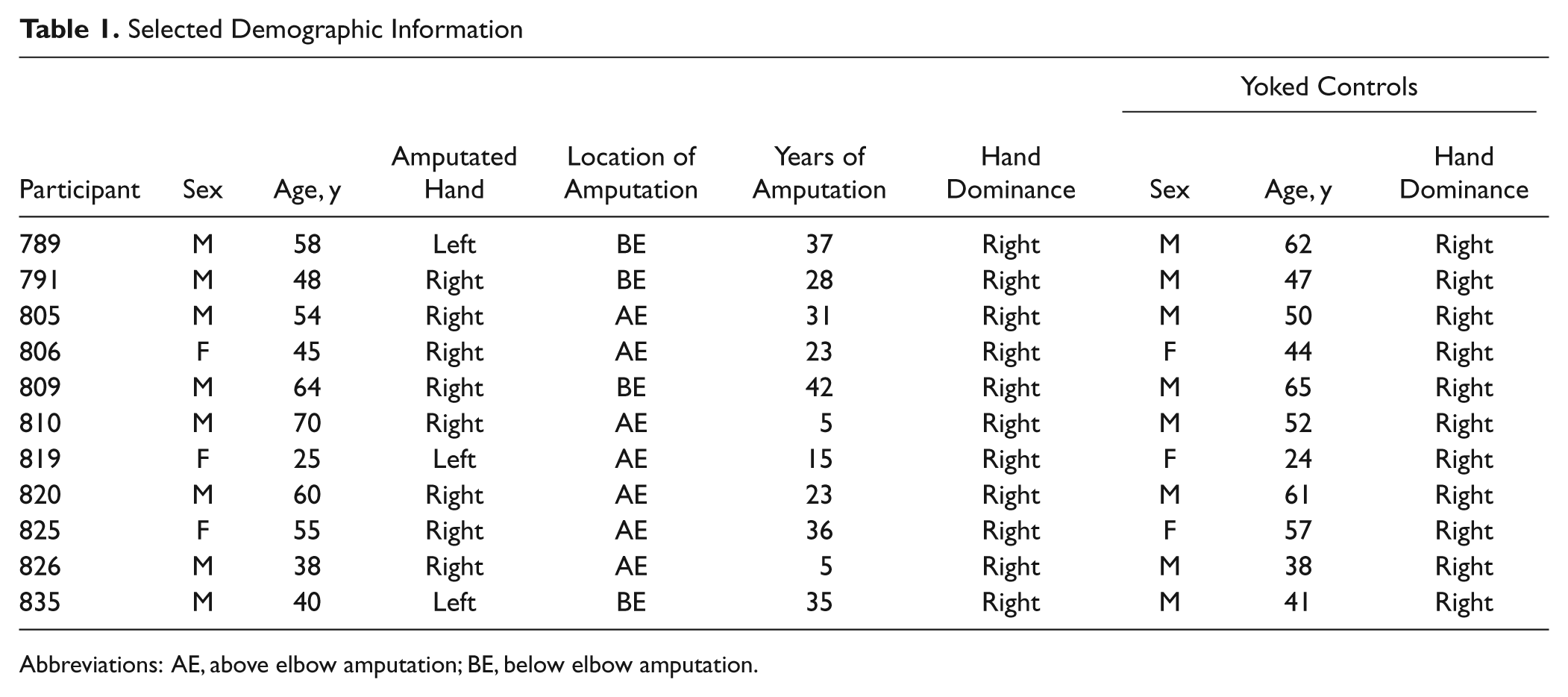
Abbreviations: AE, above elbow amputation; BE, below elbow amputation.
Apparatus
A midsagittal mirror was placed on the participant’s torso so that it reflected movements of 1 hand, creating an illusion of bimanual coupled movements. Stiff black boxboard was used to cover the mirror in conditions without mirror feedback. Dark cloth covered the area surrounding a participant’s moving hand to create a homogeneous background. Participants removed any rings prior to the experiment.
A lateral free-standing mirror, placed on either the left or the right side of the MRI bed, reflected the image of the participant’s moving hand from the midsagittal mirror. A remotely controlled, MRI-compatible camera captured the image from this lateral mirror. The camera was aligned with the magnet table and located approximately 1 m above the end of the bed. Live video stimuli were projected through a waveguide and onto a back-projection screen located at the rear of the magnet’s bore. Participants viewed this image in a small mirror attached to the birdcage head coil. Prior to testing, adjustments were made to create a compelling and uniformly sized bimanual illusion.
An experimenter was present in the room and controlled the illusion by manually covering or uncovering the midsagittal mirror during rest periods when the participant’s eyes were closed. The experimenter was cued by instructions on a video monitor.
Functional Magnetic Resonance Imaging (fMRI) Task
Every participant completed 2 functional runs, each lasting 720 s. A run consisted of 4 experimental conditions (detailed below), each presented 5 times. All the 20 experimental blocks were presented in a pseudorandomized order. Each block was cued by a 3-s auditory command that signaled participants to prepare for the upcoming task. Immediately following the auditory cue, a tone signaled the beginning of a trial and was repeated at 1 Hz for the trial’s 21-s duration. Participants were required to open their eyes and perform a unimanual thumb–finger sequencing task (TFST) in synchrony with this pacing tone. The TFST involved sequentially touching the index, middle, ring, and little finger to the thumb. The word “stop” cued participants to end their movements and close their eyes for the 12-s rest period. Each block thus lasted for a total of 36 s.
Although all 4 experimental conditions involved performing the TFST task, they differed in the following ways:
Motor execution (ME). The mirror was covered, and participants saw only their moving hand. “Move right” or “Move left” instructed the participants to start TFST with the intact (amputees) or matched (controls) hand.
Motor execution with motor imagery (ME+MI). The mirror was covered and participants saw only their moving hand. “Move right, imagine left” or “Move left, imagine right” instructed them to imagine moving their amputated hand (or the matched hand in the case of controls) in synchrony.
Motor execution with visual feedback (ME+VF). This was identical to the ME condition except that the mirror was uncovered.
Motor execution with motor imagery and visual feedback (ME+MI+VF). This was identical to the ME+MI condition, except that the mirror was uncovered.
fMRI Data Acquisition
All scans were performed on a Siemens 3-T Allegra MRI system. Whole-brain BOLD echoplanar images (EPIs) were collected using a standard birdcage radio-frequency coil and the following parameters: Repetition time (TR) = 2000 ms; echo time (TE) = 30 ms; flip angle = 80°; 64 × 64 voxel matrix; field of view (FoV) = 200 mm; 33 contiguous axial slices; slice thickness = 3.0 mm. A double echo gradient echo sequence was used to acquire a field map that was used to correct for EPI distortions. A high-resolution T1-weighted structural image was acquired using the 3D MP-RAGE pulse sequence: TR = 2500 ms; TE = 4.38 ms; TI = 1100 ms; flip angle = 8.0°; 512 × 512 × 176 voxel matrix; FoV = 256 × 256 mm2; slab thickness = 176 mm. Siemens’ Auto Align Scout and True FISP sequences were executed prior to the start of each functional run to ensure that slices were prescribed in exactly the same positions across runs. DICOM image files were converted to NIfTI format using MRIConvert software (http://lcni.uoregon.edu/~jolinda/MRIConvert/).
fMRI Data Analysis
Data were analyzed using the FSL software tools version 4.1 43 : http://www.fmrib.ox.ac.uk/fsl/. Each fMRI run for a given participant was modeled separately at the first level. Prior to statistical estimation, the following preprocessing steps were undertaken: EPI dewarping using FUGUE (unwarps geometric distortion in EPI images using B0 field maps), motion correction using MCFLIRT (corrects intramodal motion), nonbrain removal using BET (segments the brain from nonbrain structures), spatial smoothing using a Gaussian kernel of 5 mm (full width at half maximum), mean-based intensity normalization of all volumes by the same factor, and high-pass temporal filtering (σ = 100 s). Estimates of the degrees of freedom in the statistical model were corrected for autocorrelation in the data by using the FSL prewhitening technique. Time-series statistical analysis was carried out using FILM (robustly estimates first-level general linear model using prewhitening) with local autocorrelation correction. Delays and undershoots in the hemodynamic response were accounted for by convolving the model with a double-gamma basis function. Registration to high resolution and standard images (MNI template) was implemented using FLIRT (linear registration) and FNIRT (nonlinear registration). 44 Intersession (level 2) and interparticipant (level 3) levels of analysis were carried out using a fixed-effects and mixed-effects model in FLAME (models and estimates the random-effects component of the measured intersession mixed-effects variance for group statistics), respectively.45,46 Z (Gaussianized t) statistic images were thresholded using clusters determined by z > 2.3 unless otherwise indicated and a corrected cluster significance threshold of P < .05.
To enable group analysis, data from the 3 left-hand amputees and their yoked controls were left–right flipped prior to image preprocessing. As a consequence, the left hemisphere was always contralateral to the amputated hand and ipsilateral to the intact moving hand. Participants’ group statistical parametric maps were overlaid on a 3D flattened rendering of the cortical surface of the standard brain created with version 5.61 of the CARET software (http://brainvis.wustl.edu/wiki/index.php/Caret:About). To account for individual variation in cortical topography, average data of the amputeetic amputee and control groups were registered to the population, landmark, and surface-based atlas (PALS B12) using the multifiducial mapping procedure. 47
Functionally Defined Normative Sensorimotor Hand Representation
Data from a separate group of 17 participants (10 female) were used to functionally define the normative sensorimotor hand representations. This approach allows us to establish the boundaries of the normative hand representation probabilistically and is well suited for population-level inference. 48 The group’s mean age was 28.2 ± 7.7 years; 8 participants were right- and 9 left-handed as verified by the Edinburgh handedness inventory. 49 Data from 1 left-handed female participant were discarded from this sample prior to analysis because of her noncompliance with the experimental instructions.
Participants performed the TFST paced by 1-Hz auditory tones with their eyes closed. Each participant completed 2 functional runs composed of 3 conditions presented in counterbalanced order: (1) right hand moving, (2) left hand moving, and (3) both hands moving. Each 24-s movement condition was separated by a 12-s period of rest. Because our objective in the main study was to determine whether sensorimotor activity during unimanual movements performed under various manipulations would approximate activity detected during bimanual movements, data from condition 3 were used to define the normative sensorimotor maps.
Because no cortical areas showed a significant effect of hand dominance, the group statistical parametric maps were obtained by pooling the data from both left- and right-handed participants. All voxels located within the neuroanatomically restricted primary sensorimotor cortices contralateral to the moving hands, and surviving a statistical threshold of z > 2.3 with clusterwise correction of P < .05, were defined as being within the normal left or right sensorimotor hand territory (Figure 1).
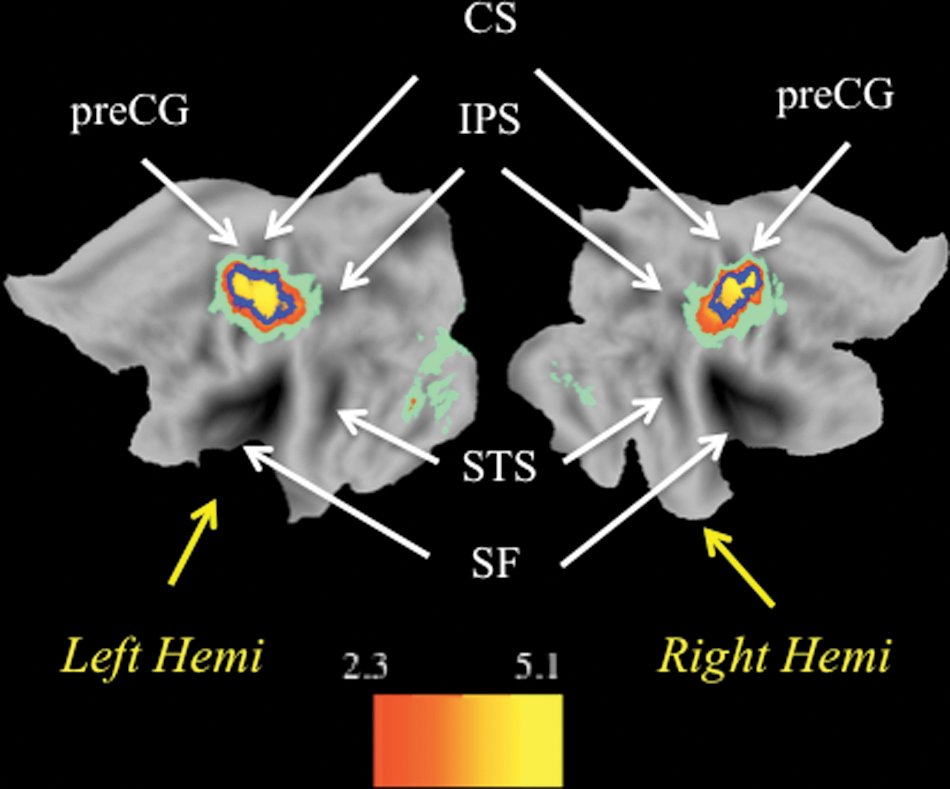
Normative maps of the sensorimotor hand representations derived functionally from a separate group of healthy controls executing thumb–finger sequencing task movements with both hands simultaneously, without visual feedback. Here and in subsequent figures, group average data are overlaid onto the flattened PALs atlas in Caret 5.61 using the multifiducial mapping procedure. 47 Areas showing significant activation after multifiduci mapping are represented in warm hues. Pale green areas were significantly activated in the group analysis but did not survive the multifiducial correction for interparticipant anatomic variability. The blue outline represents the boundaries resulting from thresholding at z > 4.0, clusterwise corrected P < .05. The left hemisphere primary sensorimotor activation spans the central sulcus (CS) and extends into precentral gyri (preCG) and postcentral gyri. It includes 1092 voxels with peak activation at (−38 mm, −22 mm, 54 mm). The right hemisphere primary sensorimotor activation includes 769 voxels, with peak at (38 mm, −22 mm, 52 mm).
Region of Interest Analyses
Mean percentage signal change relative to resting baseline was computed separately for every participant and each condition of the main experiment within the respective normative hand representations contralateral and ipsilateral to the moving hand. Mean percentage signal change values were then submitted to a 2 (Group: amputee, control) × 4 (Condition: ME, ME+MI, ME+VF, ME+MI+VF) repeated-measures ANOVA.
Results
Equivalent Contralateral Hand Area Responses for Both Groups
Relative to resting baseline, increased activity was detected within a widely distributed network of cortical regions during the performance of all 4 experimental conditions for both amputees and healthy controls (Figure 2). In addition to regions within and along the central sulcus (CS) contralateral to the moving hand (visualized here as the right hemisphere), these cortical areas included the supplementary motor area (SMA), ventral and dorsal premotor areas, posterior parietal area, rostral middle frontal gyrus, and occipital and temporal cortices. Subcortical increases were also detected in both groups within the thalamus, basal ganglia, and lateral cerebellum. Critically, as elaborated below, only amputees showed evidence of significant increases along the CS in the hemisphere ipsilateral to the moving hand (visualized here as the left hemisphere).
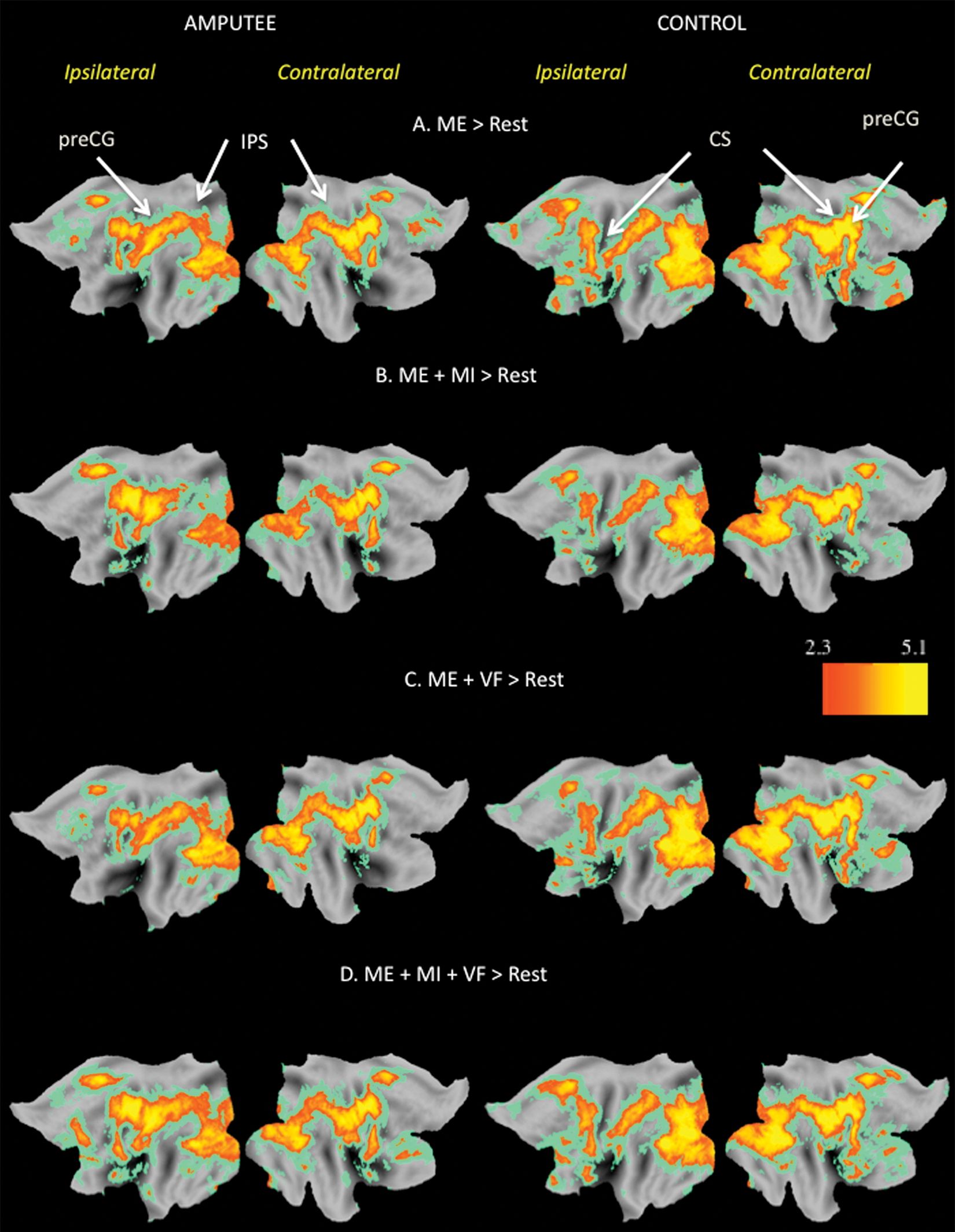
Statistical parametric maps representing areas of significantly increased activity (z > 2.3, clusterwise corrected P < .05) associated with comparison of experimental conditions versus rest for both amputee and control groups. Here and in the subsequent figures, contralateral refers to the hemisphere opposite to the moving hand, whereas ipsilateral denotes the hemisphere located on the same side as the moving hand (ie, contralateral to the amputated hand in patients). Motor execution (ME; panel A) activates the ipsilateral sensorimotor cortex in amputees but not in controls. Both groups do show increased activity in bilateral dorsal and ventral premotor cortices and posterior parietal areas within and along the intraparietal sulcus (IPS) and contralateral to the moving hand along the precentral gyrus (preCG), central sulcus (CS), and postcentral gyrus. This same network of areas is engaged when undertaking motor imagery (ME+MI) (panel B), visual feedback (ME+VF; panel C), or both (ME+MI+VF; panel D).
Increased Ipsilateral Hand Area Responses for Amputees
When compared directly with controls, amputees showed focal increases in activity during all 4 conditions along the CS, extending rostrally into the precentral gyrus and caudally into the postcentral gyrus (Figure 3). The high degree of overlap between this cluster and the functionally defined normative hand representation (blue outline) indicates that it likely includes the former hand territory, a point to which we will return shortly. It is also worth noting that the spatial extent of this cluster (at a fixed statistical threshold of z > 4, with clusterwise correction of P < .05) was similar for both the ME and ME+VF conditions (see panels E and G, Figure 3). Relative to these conditions, however, the addition of MI in both the ME+MI and ME+MI+VF conditions was associated with an expansion of this cluster in the medial direction (see panels E and G vs F and H in Figure 3). The similarity of clusters for the ME+MI and ME+MI+VF conditions suggests that combining MI and mirror feedback did not further influence the spatial extent of this activation (see panels F and H, Figure 3).
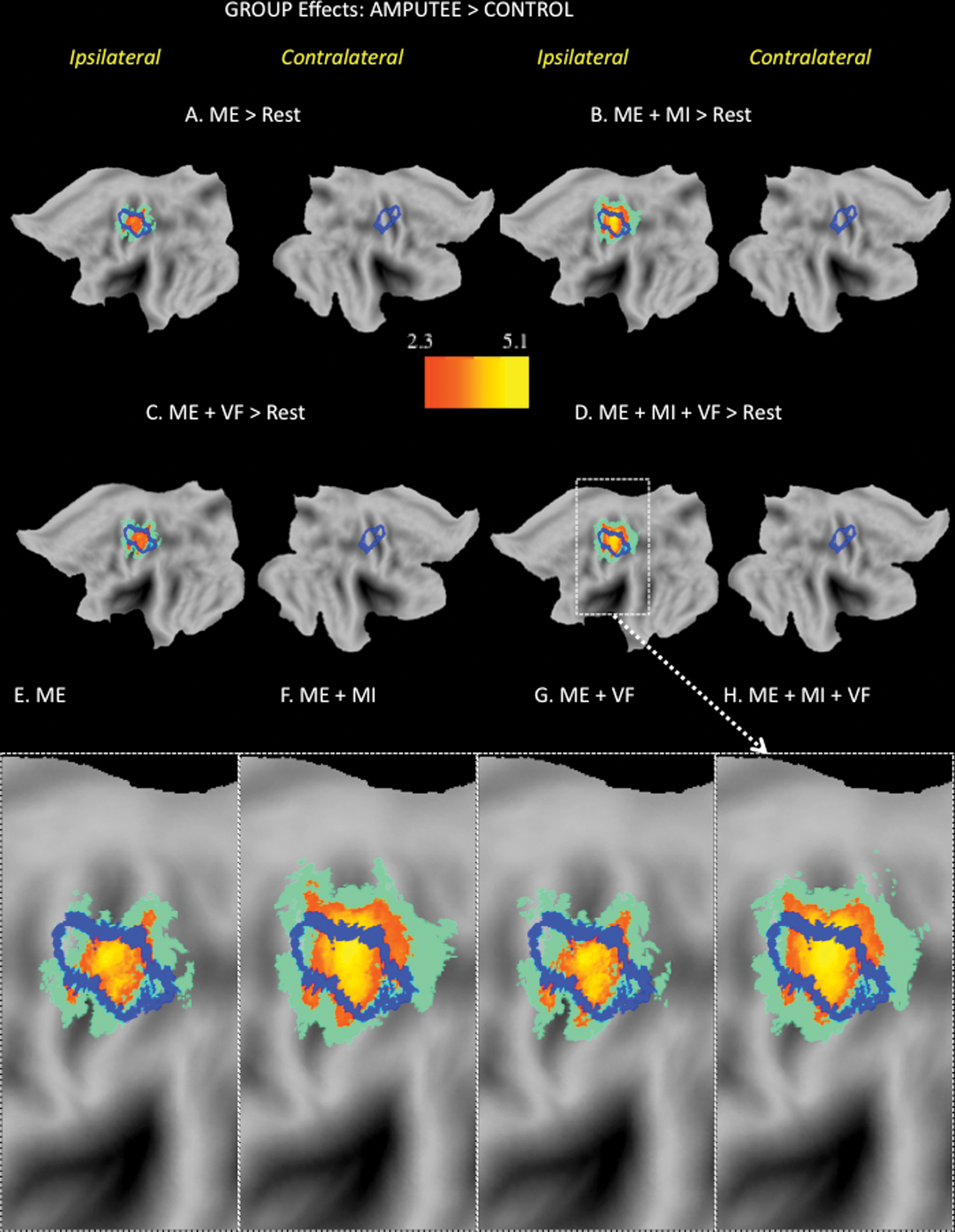
Amputees show significant increases in the ipsilateral sensorimotor cortex in all conditions relative to controls. Increased activations in the ipsilateral sensorimotor cortex were present only in the amputee group. The normative hand representations from the localizer are overlaid as blue contours on the group results (A-D). The zoomed views of the ipsilateral sensorimotor data from panels A to D (E-H). The cluster size associated with the motor execution (ME) task alone (630 voxels in panel E) slightly increases with the addition of visual feedback (ME+VF; 682 voxels, panel G). The addition of motor imagery (MI) is associated with substantial medial expansion of the ipsilateral sensorimotor cluster (ME+MI: 1522 voxels, panel F; ME+MI+VF: 1372 voxels, panel H).
Effects of MI and Mirror VF Relative to ME With the Intact Hand
Whole-brain contrasts were run to identify regions modulated by either MI or VF when activity associated with ME of the intact hand was subtracted. For amputees, but not controls, MI was associated with significantly increased activity in the former hand territory (blue outline, left hemisphere), extending into more medial regions of the precentral and postcentral gyri and rostrally into the premotor area and the pre-SMA (Figure 4A). In contrast, in the VF condition, both amputees and controls exhibited increased activity within areas of the occipital, posterior temporal, and parietal cortices but not in sensorimotor hand representations or other regions of the frontal cortex implicated in motor representation (Figure 4B). This is consistent with recent findings with controls where only effects of VF in visual areas were detected. 50
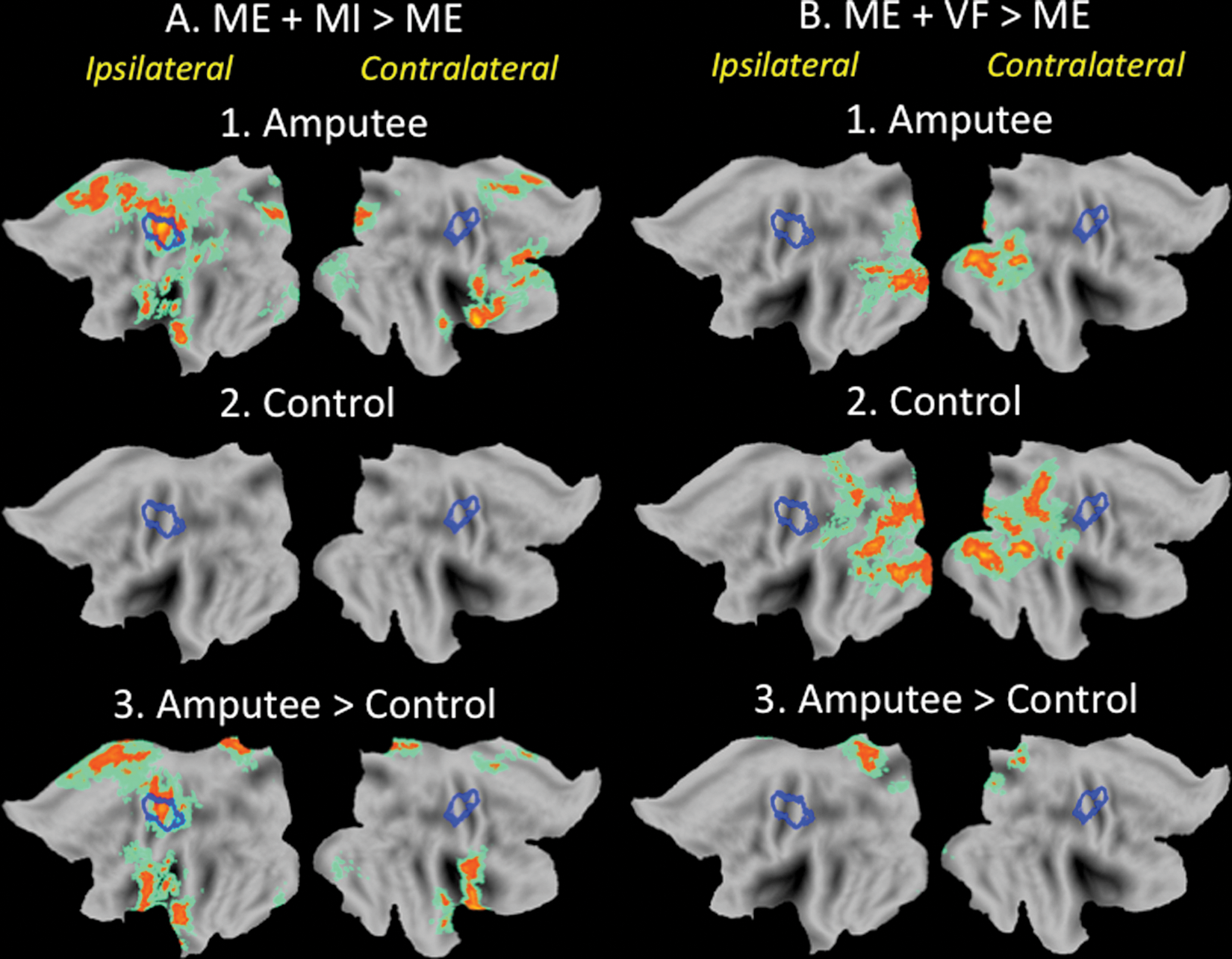
Effects of motor imagery (MI) on activity in the distributed sensorimotor network exceed those associated with intact hand movements in amputees. A. Relative to motor execution (ME) alone, the addition of MI (ie, ME + MI) resulted in increased activity in the former sensorimotor hand territory (blue outline, left hemisphere). Increases were also detected in more medial precentral and postcentral gyri and in the premotor and pre–supplementary motor area (SMA) of amputees (1) but not controls (2). These effects were significantly greater than those detected in controls (3). B. In contrast, the addition of mirror visual feedback (ME + VF) resulted in increased activity in occipital, posterior temporal, and parietal areas in both groups (1 and 2), most likely because of increased visual stimulation. For reasons that are uncertain, amputees showed larger increases in the medial parietal cortex (3).
Region-of-Interest Analyses: Activity in the Normative Sensorimotor Hand Area Contralateral to the Moving Hand
Increased activity relative to baseline was detected in the functionally defined primary sensorimotor hand representation contralateral to the moving hand during all experimental conditions for both groups; P < .001 in all cases. Differences between groups or conditions were nonsignificant; P > .10 in both cases (Figure 5).
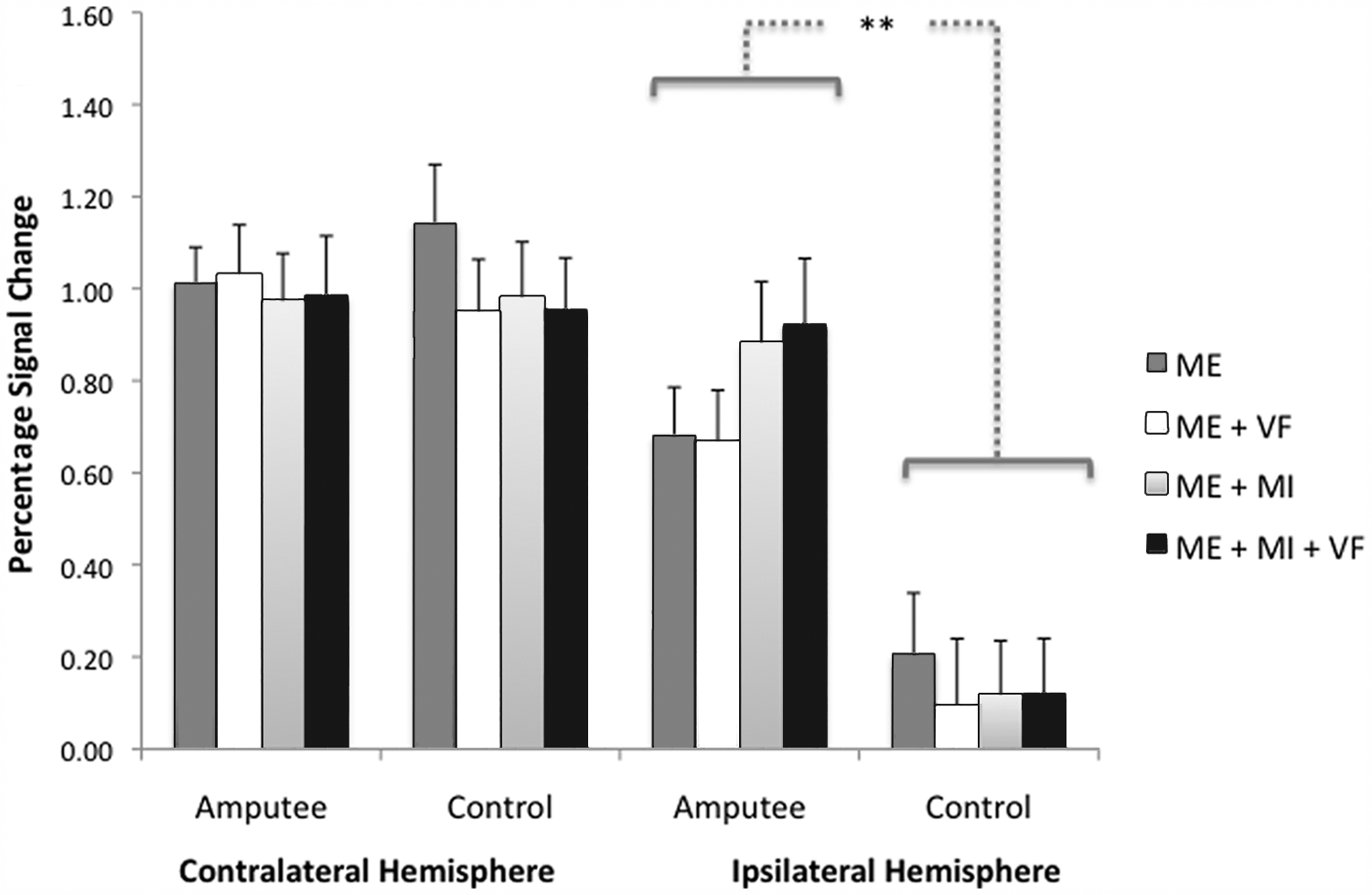
Group mean percentage signal change (PSC) within functionally defined normative sensorimotor hand representations during the 4 experimental conditions. Error bars represent standard errors; **P < .001. Hemisphere contralateral to the moving hand: the magnitude of mean group PSC did not differ significantly between amputee and control groups. Hemisphere ipsilateral to the moving hand: controls showed no increases above baseline. For amputees, imagining moving the absent hand while moving the intact hand (motor execution + motor imagery [ME+MI]) was associated with a nonsignificant trend toward increased activity relative to moving the intact hand alone (ME). The addition of illusory visual feedback (VF) to motor execution (ME+VF) failed to increase activity beyond what was observed in the ME condition. The MI and VF condition (ME+MI+VF) also resulted in a nonsignificant trend toward increased activity relative to the ME condition. See text for details.
Region-of-Interest Analyses: Activity in the Normative Sensorimotor Hand Area Ipsilateral to the Moving Hand
All conditions were associated with increased ipsilateral activity above what was detected during resting baseline for the amputees; P < .001 in all cases (Figure 5). In contrast, none of the conditions was associated with significant increases in activity for controls; P > .05 in all cases. The magnitude of the signal change differed significantly between amputee and control groups: F(1, 20) = 14.8, P = .001. The main effect of Condition was not significant: F(3, 60) = 2.12, P = .11. The interaction effect between Group and Condition was significant, with amputees showing a greater response than controls particularly in conditions involving MI: F(3, 60) = 3.23; P = .03.
For amputees, imagining moving the absent hand in sync with moving the intact hand (ME+MI) compared with moving the intact hand alone (ME) was associated with a nonsignificant trend toward increased activity within the former hand territory: t(10) = 1.83; Bonferroni corrected P > .05. The addition of illusory VF to ME (ME+VF) failed to increase activity beyond what was observed in the ME condition: t(10) < 1.0. Combined MI and VF (ME+MI+VF) also resulted in a nonsignificant trend toward increased activity relative to the ME condition: 1-tailed paired t(10) = 2.0; Bonferroni corrected P > .05. This was no greater than what was observed in the ME+MI condition: t(10) < 1.0. The difference between conditions involving (ME+MI and ME+MI+VF) versus those not involving MI (ME and ME+VF) approached, but failed to achieve, significance: t(10) = 2.04; Bonferroni corrected P > .05.
Discussion
The results of this experiment demonstrate that chronic amputees reactivate the former sensorimotor hand territory when moving their intact hands. There is a nonsignificant tendency for these effects to be enhanced when simultaneously imagining movements of the missing hand but not when viewing illusory VF created with a mirror. We now consider each of these findings, and their implications, in detail.
Increased Former Hand Area Activity Accompanying Movements of the Intact Hand
Compared with matched controls, unilateral amputees showed significantly increased activity in the sensorimotor cortex ipsilateral to the moving intact hand (ie, contralateral to the amputated hand). This was true for all conditions, including simply moving the intact hand under visual guidance (Figure 3). Whereas increased ipsilateral sensorimotor activity has been reported in previous functional neuroimaging studies of unilateral amputees,18,19,51,52 we show that these effects are located specifically within the former hand territory as defined functionally on the basis of data from matched controls. This is an important difference from the modest ipsilateral increases reported in healthy adults during hand movements, which are located rostrally in the precentral cortex 17 (Figure 5).
Increases in the former hand territory associated with use of the intact hand may be attributable to a reduction in IHI following unilateral deafferentation. 53 In healthy adults, transcranial magnetic stimulation of the ipsilateral primary motor cortex exerts a time-dependent inhibitory influence on the contralateral motor cortex. 54 Acute deafferentation reduces IHI, leading to increased excitability in the primary motor cortex ipsilateral to the affected limb. 55 These changes are likely mediated by GABAergic transcallosal pathways and are specific to primary motor areas representing muscles homotopic to those that underwent deafferentation. 20 It is interesting to note that these inhibitory effects are reported to be moderate in unilateral amputees, suggesting a possible rebalancing of interhemispheric interactions following chronic deafferentation. 20 Our fMRI findings, however, indicate persistence of these ipsilateral increases in patients tested an average of 25 years postamputation (range, 5-42 years). This raises the interesting question of whether long-term increased use of the intact hand contributes to the ipsilateral effects found here.
MI and Activity in the Former Hand Territory
Recent behavioral findings suggest that chronic amputees maintain the ability to imagine certain movements of their missing limbs accurately. 56 MI was associated with greater activity in the hand territory ipsilateral to the moving hand (former hand territory) for amputees versus controls. However, when the effects of intact hand movement were controlled, trends toward increased activity during MI for amputees did not survive corrections for multiple comparisons (Figure 5). For amputees, MI was associated with significant increases in activity that extended beyond the former hand territory into more medial portions of the precentral and postcentral gyri and rostrally into premotor areas and the pre-SMA (Figure 4). Increased activity in these latter 2 regions is commonly detected in studies of MI. In short, activity associated with imagining movements of an amputated hand differs substantially from what is observed during MI of an intact hand by controls, with the most pronounced effects occurring in regions outside the former hand territory. As a result of extended training, these responses could come to affect experience-dependent changes in the former hand territory.
Failure to Detect Effects of Mirror VF on Sensorimotor Activity
In the absence of MI, illusory VF (ME+VF) failed to increase activity in the sensorimotor cortex beyond what was observed during movements of the intact hand alone (ME; Figures 4 and 5). Consistent with earlier results in healthy adults, 50 effects of the mirror in both groups were detected exclusively in occipital, posterior temporal, and parietal cortices and are most likely associated with increased visual stimulation (ie, observing movements of both hands vs one). A recent article reports increases in the primary sensorimotor cortex ipsilateral to the moving hand when healthy adults or amputees without phantom limb pain perform visually guided unilateral hand movements but not when performed by patients with pain. 34 These ipsilateral movement-related effects increased in the presence of the mirror illusion (ie, our ME+VF condition) and were also detected when these 2 groups imagined movements of the affected side while the intact hand remained still. Because of the absence of a functional localizer, however, it is unclear that these responses are specific to the former hand territory. Compared with the normative hand representations used in the current study (left primary motor cortex: −38 mm, −22 mm, 54 mm; see Figure 2), peaks of these ipsilateral responses are quite rostral (refer to Table 2 of ref 34, right primary motor cortex: 39 mm, −9 mm, 54 mm in healthy controls and 42 mm, −6 mm, 57 mm in amputees without phantom pain). According to an established histological atlas, 57 these loci have a high probability of being in the premotor cortex (Brodmann area 6) and a relatively low likelihood of falling in the primary motor cortex (Brodmann areas 4/4a). We too find strong bilateral increases in the premotor cortex for both patients and controls in all experimental conditions relative to resting baseline (Figure 2) but not in the ipsilateral hand area for controls (Figures 3 and 5). When the effects of intact hand movement are controlled, these ipsilateral premotor activations are increased only for the amputees and in conditions involving MI but not for mirror feedback alone (Figure 4). This is true despite the fact that only 4 out of 11 participants in the current work indicated no current or recent pain.
Conclusions
It is possible that the effects of the mirror feedback on activity in the former hand territory may evolve with practice or that responses may differ in acute amputees. That said, our failure to detect increases in association with the addition of mirror feedback may explain why reductions in pain for amputees assigned to such an intervention failed to exceed those experienced in a control condition where the same exercises were performed with the intact limb while the mirror was covered. 58 At first glance, our findings appear less easily reconciled with those of another trial demonstrating a substantial reduction in pain in all 6 lower-extremity amputees who undertook a month-long course of 15 minutes of daily mirror training. None of the 6 individuals assigned to the MI control group experienced a significant reduction in phantom limb pain until crossing over to the mirror intervention. 35 However, participants only performed simultaneous movements of the intact limb during the mirror treatment and not the imagery condition. At least for upper-extremity amputees, movements of the intact limb are a highly effective stimulus for the portion of the sensorimotor cortex that would have previously represented the amputated limb. With practice, the effects of MI on the adjacent cortex might enhance these responses.
Footnotes
Acknowledgements
The authors thank Scott Watrous for his technical assistance. SHF designed the research. JS developed the hardware and software. SB, JS, and SHF collected the data. SB and SHF analyzed the data, and SHF and SB prepared the manuscript. We thank Michael Posner, Leonardo Cohen, and anonymous reviewers for their helpful feedback on earlier drafts.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by grants from the MJ Murdock Charitable Trust and USAMRAA (W81XWH-09-2-0114) to SHF.
