Abstract
Background. While recent clinical trials involving robot-assisted therapy have failed to show clinically significant improvement versus conventional therapy, it is possible that a broader strategy of intensive therapy—to include robot-assisted rehabilitation—may yield clinically meaningful outcomes. Objective. To test the immediate and sustained effects of intensive therapy (robot-assisted therapy plus intensive conventional therapy) on outcomes in a chronic stroke population. Methods. A multivariate mixed-effects model adjusted for important covariates was established to measure the effect of intensive therapy versus usual care. A total of 127 chronic stroke patients from 4 Veterans Affairs medical centers were randomized to either robot-assisted therapy (n = 49), intensive comparison therapy (n = 50), or usual care (n = 28), in the VA-ROBOTICS randomized clinical trial. Patients were at least 6 months poststroke, of moderate-to-severe upper limb impairment. The primary outcome measure was the Fugl-Meyer Assessment at 12 and 36 weeks. Results. There was significant benefit of intensive therapy over usual care on the Fugl-Meyer Assessment at 12 weeks with a mean difference of 4.0 points (95% CI = 1.3-6.7); P = .005; however, by 36 weeks, the benefit was attenuated (mean difference 3.4; 95% CI = −0.02 to 6.9; P = .05). Subgroup analyses showed significant interactions between treatment and age, treatment and time since stroke. Conclusions. Motor benefits from intensive therapy compared with usual care were observed at 12 and 36 weeks posttherapy; however, this difference was attenuated at 36 weeks. Subgroups analysis showed that younger age, and a shorter time since stroke were associated with greater immediate and long-term improvement of motor function.
Keywords
Introduction
Given that many stroke survivors have persistent disability despite customary acute medical and rehabilitative care, there have been increasing clinical and research interest in understanding the potential of rehabilitative treatments for stroke survivors beyond the subacute period.1,2 One compelling and evolving approach is the incorporation of additional rehabilitative therapy in the chronic stage (often defined as 6 months poststroke), including intensive regimens delivered by a trained therapist as well as those delivered via a robot. However, whereas intensive rehabilitative therapy is generally recognized as qualitatively similar to conventional therapy, 3 studies testing intensive strategies typically exclude nontraditional strategies, that is, robot-assisted therapy. Nevertheless, regimen intensity is the focus of ongoing inquiry in robot-assisted therapy,4,5 and there is increasing interest in therapeutic efficacy in long-term follow-up.6,7 Given the increasing prevalence,8,9 cost-effectiveness, 10 and therapeutic equivalence in comparative effectiveness analyses,9,11 the distinction between robot-assisted therapy and therapy delivered by a therapist is becoming blurred. In this way, we propose that the integration of these to strategies into a single comparator arm is well supported.
In this article, we present a secondary analysis of the US Department of Veterans Affairs Robotic-Assisted Upper-Limb Neurorehabilitation in Stroke Patients (VA Robots in Chronic-Stroke [VA-ROBOTICS]). The primary objective of the VA-ROBOTICS study was to test for therapeutic benefit of robot-assisted therapy versus an active comparator (intensive comparison therapy) and versus usual care, in patients with upper limb deficit beyond 6 months from their index stroke. 8 Although no significant differences in improvement were observed between the 3 groups in most outcome measures in the primary analysis following 12 weeks of therapy, 11 there remains an unexplored opportunity to determine whether intensive rehabilitative therapy, as a general strategy utilizing either robotic assistance or facilitation by a human therapist, is of benefit for people with chronic stroke impairment. 12 Given that the robot-assisted and intensive comparison therapies in VA-ROBOTICS were designed to deliver a nearly identical dose of therapy (time and duration for sessions), we explore whether pooling the data of the 2 active treatment groups into a single group of “intensive therapy” could address a more generalized question about how the effects of intensive rehabilitation therapy compare to the effects from usual care. Specifically, is intensive therapy efficacious versus usual care in motor restoration immediately after therapy at 12 weeks, and at the 6-month follow-up time point? And are there particular subgroups who benefit more.
Methods
Study Population and Protocol
The VA-ROBOTICS multicenter, randomized, and controlled clinical trial was conducted among four VA Medical Centers between November 2006 and October 2008. 11 In this trial, 127 patients with moderate-to-severe upper limb impairment (Fugl-Meyer scores at baseline: 7 to 38 points) 6 months or more poststroke were enrolled and randomly assigned to 1 of 3 treatment groups: usual care (n = 28), robot-assisted therapy (n = 49), or intensive comparison therapy (n = 50). Enrollment in the usual care arm was terminated after 1 year per protocol; enrollment in the active treatment arms occurred over 2 years. A more comprehensive summary of the study design, including a detailed description of the training parameters in each treatment group (sessions frequency, duration, and intensity) is presented elsewhere; a summary is presented below. 13 The study was approved by the institutional review board at each medical center and all study participants provided written informed consent.
The robot-assisted therapy was delivered via In-Motion rehabilitation robots, designed for goal-directed, assisted upper extremity movement, with an interactive computer-generated video program providing visual and kinesthetic feedback to the patient. 14 The treatment was administered under the guidance of a trained research therapist, and the treatment protocol was structured into 4 consecutive blocks of 3 weeks (12 weeks total): shoulder-and-elbow therapy, antigravity and grasp therapy, wrist therapy, and integrated whole-arm therapy.
The intensive comparison therapy was supervised by the same trained research therapist who supervised the robot-assisted therapy at each site and included the use of conventional rehabilitation methods to match robot-assisted therapy in form, time and number of treatments. Intensive comparison therapy included four 15-minute sessions: (a) warm-up and assisted stretching, (b) active arm exercises, (c) goal-directed planar reaching, and (d) functionally based neurodevelopment techniques/Bobath arm training.
The usual care group received customary care available to them at their VA Medical Center, including secondary stroke risk management, and in some cases clinical rehabilitation services. Among the usual care group, 32% reported ongoing therapy for their affected upper extremity. In the interest of generalizability, activities of usual care were not dictated by the study protocol, and the activities of this group were not recorded. We note that participants in the robotic therapy and intensive comparison therapy arms were also allowed to continue with their usual care. Randomized study treatments were added on top of whatever care the participants were already receiving outside of the study. 13
The active study treatments are considered “high intensity” as the activity modules used separately and in combination to perform repetitive, task-oriented movements in high volume (up to 1024 per session on average). 11 We consider this “high dose” by the number of hours spent in training (36 hours), which is generally regarded as at or above the threshold of high dosage in similar contexts.15-17
The primary outcome measure for VA-ROBOTICS was the Fugl-Meyer Assessment of Sensorimotor Recovery at 12 weeks. The Fugl-Meyer Motor Function subdomain comprised 33 items, each scored on a 0, 1, 2 points ordinal scale. The range of Fugl-Meyer score Motor Function subdomain was from 0 (no function) to 66 (normal function). Secondary outcomes included the Wolf Motor Function Test (WMFT) and Stroke Impact Scale (SIS) scores. The WMFT consists of 17 items, of which 2 items measure strength and 15 measure time to perform functional tasks; strength is measured in pounds (for lifting or handgrip), and time is measured in seconds with an upper limit of 120 seconds to accomplish a specific task. The SIS is a 59-item self-reported questionnaire that asks persons with stroke to rate perceived problems in 8 domains using a 5-point scale: strength, hand function, mobility, activities of daily living, emotion, memory, communication and social participation. VA-ROBOTICS used only the specific subsections of hand function activities of daily living, and social participation (32 items); aggregate scores for questions within each domain are generated using an algorithm with a final scoring range from 0 to 100. On the Fugl-Meyer Assessment and SIS, higher scores indicate better functioning; for WMFT, lower scores indicate better functioning. All outcome assessments were performed by an independent trained therapist at each clinical site who was masked to the assigned treatment.
In the VA-ROBOTICS trial, a 3-point incremental change in the Fugl-Meyer Assessment was deemed to be the minimal clinically meaningful change, that is, the change that was of sufficient magnitude to differentiate patients on the basis of their disability score on the modified Rankin scale. 11
Statistical Analysis
Baseline comparisons among the three randomized treatment groups were previously conducted and showed that the characteristics of the three treatment groups were similar, except for the time from the index stroke to randomization (P = .04), which was significantly longer in the usual care arm. 11 Thus, baseline comparisons are only presented for the combined intensive therapy group versus usual care.
The effect of treatment on the primary and secondary outcomes at 12 and 36 weeks was analyzed using a mixed-effects model adjusted for the following covariates: study site, comorbidity disease index, and the baseline value of the outcome. Because randomization occurred over a 1-year period in the usual care arm and over a 2-year period in the 2 active arms, there is a possible nonrandom opportunity for imbalance in patient characteristics. To account for this possibility, an additional baseline comparison was made between the 2 recruitment periods (all participants recruited in year 1, versus all participants recruited in year 2); any baseline variable for which there was a significant difference between time periods was included as an adjustment covariate in the model to account for any temporal trends in the randomized patient population. Because the activities of usual care were not recorded, a focal analysis of the dose-response relationship in intensive therapy is not feasible; usual care was not prescribed in the protocol, but rather was intentionally broad to support a comparative effectiveness study between strategies.
The outcome variables for the primary and secondary outcomes were the change in the measure relative to baseline (12-week minus baseline score, and 36-week minus baseline). For the 12-week analysis, any missing 12-week assessment was replaced with the next available follow-up assessment; patients missing all the subsequent assessments were excluded from the analysis; if a participant was missing the 36-week measurement, they were still incorporated into the 12-week analysis. Treatment effects are summarized as least squares mean changes with corresponding 95% confidence limits. Analyses were conducted by combining robot-assisted therapy and intensive comparison therapy into a single intensive therapy group. Treatment effects were estimated in subgroups of patients by the mixed model. Tests of homogeneity were conducted to test for differences in subgroup effects. A P value of .05 (2-sided) was considered to be significant. Since analyses were considered exploratory, no adjustment for multiplicity was made. All statistical analyses were performed using SAS version 9.3 (SAS Institute, Inc, Cary, NC).
Results
Baseline Characteristics
We assessed the comparability of baseline characteristics between participants in the combined active treatment arm (intensive therapy) and usual care (Table 1). Generally, the distribution of baseline characteristics was comparable, except for time from index stroke to randomization, which was significantly longer for usual care (6.2 ± 5.0 years) versus intensive therapy (4.2 ± 4.0 years, P = .016); and prevalence of myocardial infarction, which was greater for the usual care (29% vs 17%, P = .037).
Baseline Characteristics of the Patients.
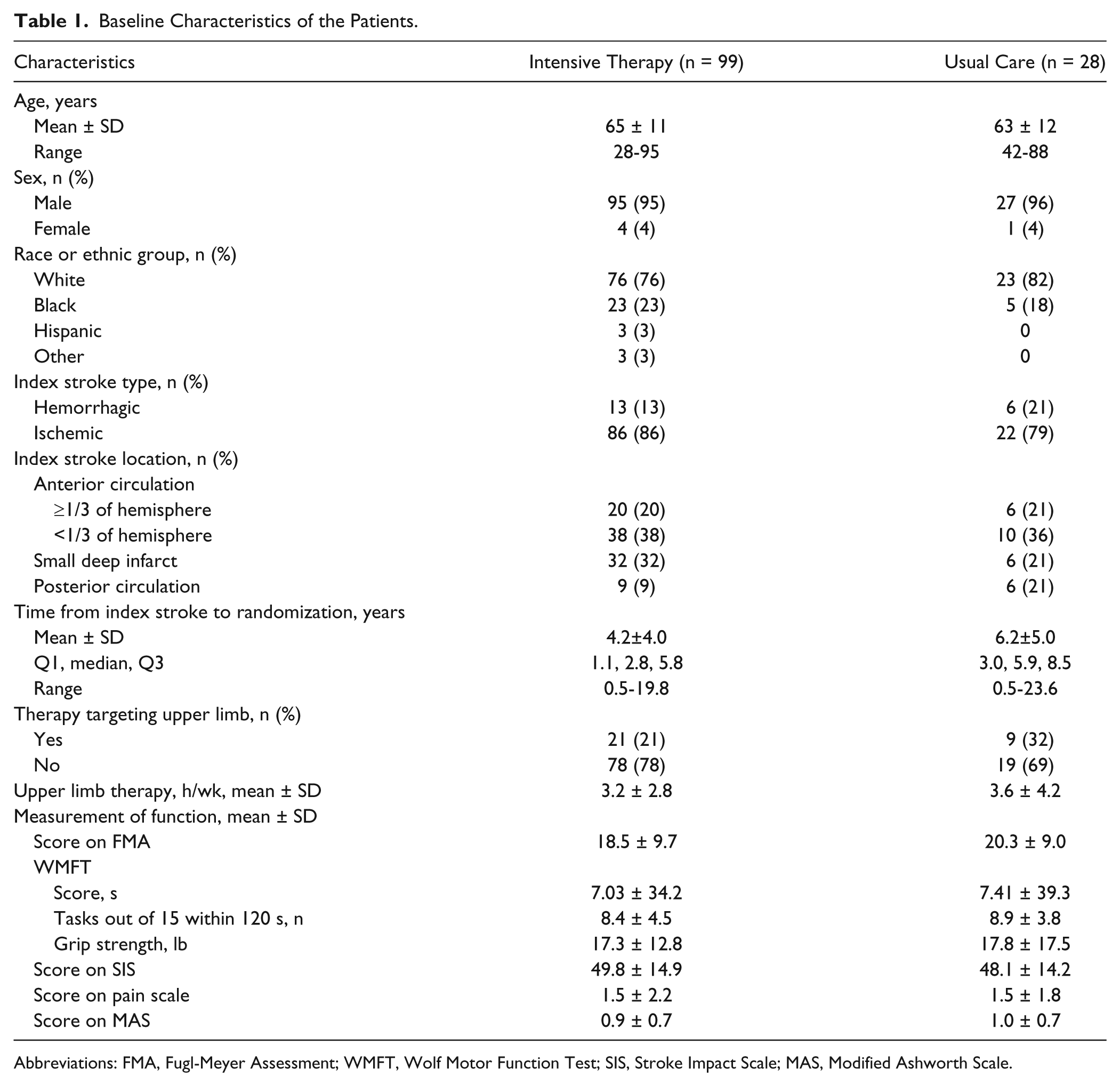
Abbreviations: FMA, Fugl-Meyer Assessment; WMFT, Wolf Motor Function Test; SIS, Stroke Impact Scale; MAS, Modified Ashworth Scale.
We also assessed the comparability of baseline characteristics between study enrollment periods, that is, year 1 versus year 2. The distribution of baseline characteristics was comparable between the 2 periods, except for baclofen administration—12 of 81 participants (14.8%) in period 1, versus 2 of 46 participants (4.3%) in period 2 (P = .031); and concomitant physical therapy—12 of 81 (21.0%) versus 2 of 46 (8.7%; P = .049). These 2 variables were included as adjustment covariates in the analysis of outcomes.
Outcomes for Intensive Therapy versus Usual Care
Results of intensive therapy versus usual care for the primary and secondary outcomes showed significant therapeutic benefit from intensive therapy at 12 weeks (Table 2). The mean improvement for intensive therapy compared with usual care was 4.0 points (95% CI, 1.3 to 6.7; P = .005) in the Fugl-Meyer score—more than the predefined minimal clinically meaningful change—and 7.4 points (95% CI, 2.7 to 12.0; P = .002) in the SIS; the treatment difference was marginal for the WMFT (−7.8, 95% CI, −15.7 to 0.08, P = .052). However, the treatment differences on the Fugl-Meyer and SIS were attenuated by 36 weeks, and was significant only for the WMFT (−0.2 ± 3.4 usual care vs −7.7 ± 2.9 intensive therapy, −7.5 mean difference, 95% CI, −13.9 to −1.1; P = .02). We note that a 3-point change in Fugl-Meyer Assessment corresponds to a 20-point change in SIS, 18 thus the observed treatment effect for SIS cannot be considered clinically meaningful.
Changes in Primary and Secondary Outcomes at 12 and 36 Weeks: Intensive Therapy Versus Usual Care. a
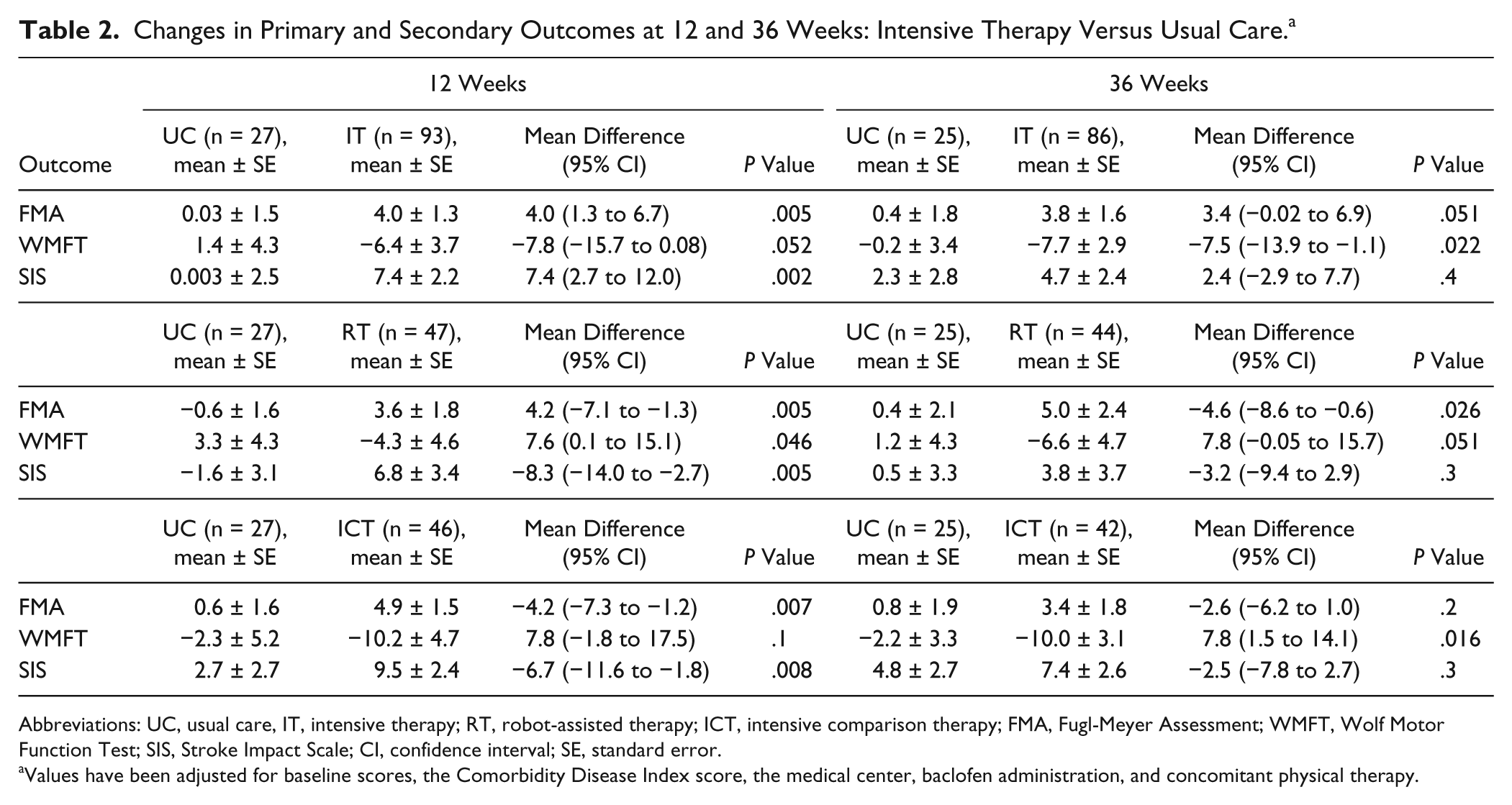
Abbreviations: UC, usual care, IT, intensive therapy; RT, robot-assisted therapy; ICT, intensive comparison therapy; FMA, Fugl-Meyer Assessment; WMFT, Wolf Motor Function Test; SIS, Stroke Impact Scale; CI, confidence interval; SE, standard error.
Values have been adjusted for baseline scores, the Comorbidity Disease Index score, the medical center, baclofen administration, and concomitant physical therapy.
Subgroup Analysis
Because beneficial effects of intensive therapy compared with usual care were found, treatment effects were examined in subgroups of patients according to their age, stroke type, time from stroke to randomization (months poststroke), presence of additional arm therapy, Fugl-Meyer score at baseline, and whether the dominant hand of the patient was affected by the stroke. Figure 1 displays forest plots in the selected subgroups at both 12 and 36 weeks on the Fugl-Meyer score for intensive therapy versus usual care. Significant interactions were detected for the subgroups defined by age (P = .023) and months poststroke (P = .011) at 12 weeks and these significant differences were maintained at 36 weeks (P = .01 and P = .03). That is, younger age and a shorter time since stroke were associated with greater immediate and long-term retention of motor function. Results were similar when age and months since stroke were treated as continuous measures (data not shown).
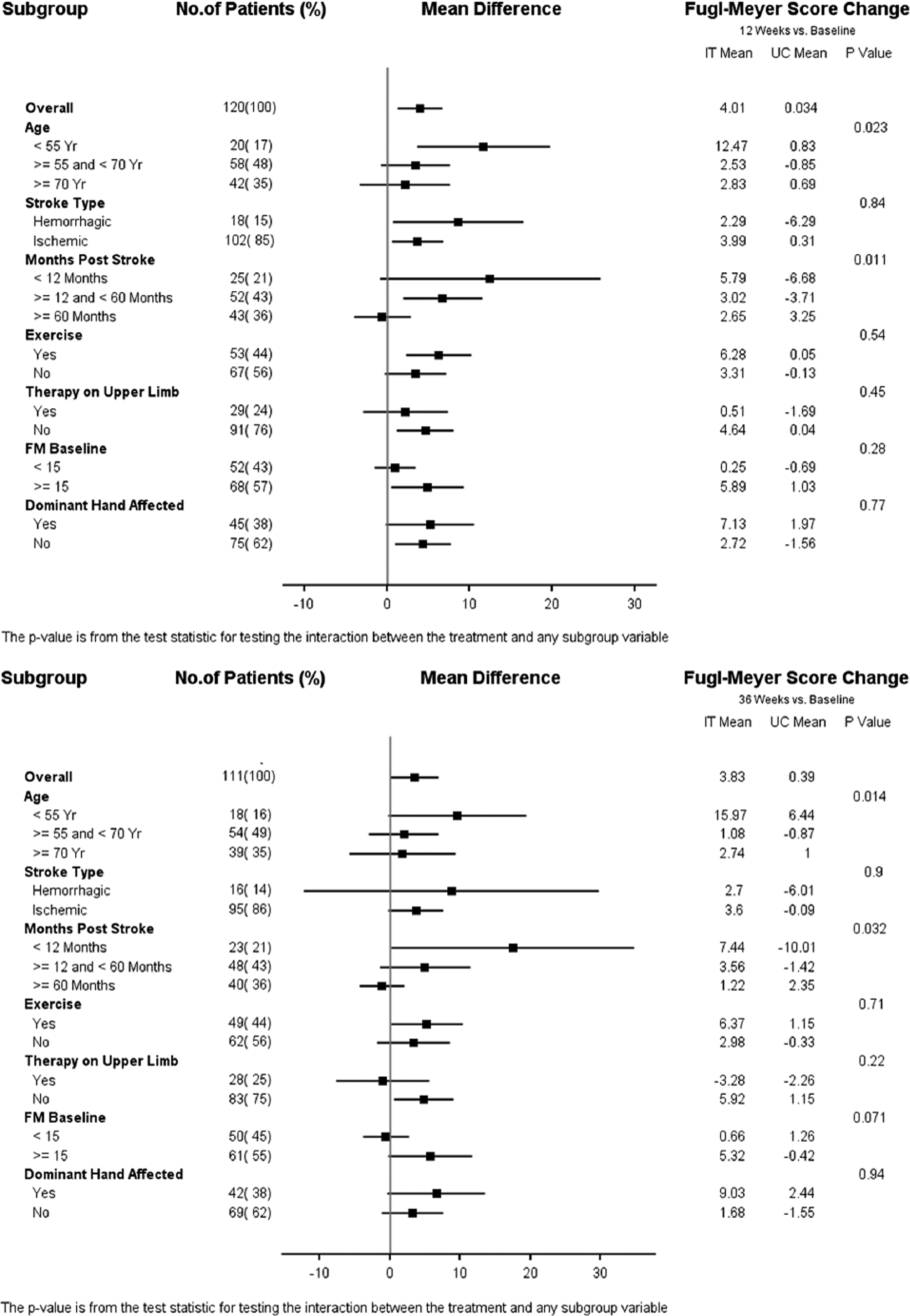
Forest plot of select subgroups: intensive therapy (IT) and usual care (UC) at 12 weeks (left) and 36 weeks (right).
Discussion
A fundamental concept applied to motor rehabilitation is the principle of repetitive practice based on the Hebbian learning rule for reinforcing neural networks. The VA-ROBOTICS trial tested this idea with a specific emphasis on having high-intensity rehabilitation delivered via robot rehabilitation. In this article, we explored the broader question of whether providing a high dose of additional rehabilitation therapy, in this case 36 hours of therapy, during the chronic phase of stroke (>6 months poststroke) made an immediate difference at the completion of therapy and a sustained difference 24 weeks after the completion of therapy, that is, 36 weeks after the start of therapy. Nevertheless, not much is known on the characteristics of those who are most likely to benefit from stroke rehabilitation. Where previous studies have compared robot-assisted therapy versus an intensity-matched comparison therapy led by trained therapist and have not found clear evidence of superiority,8,9,11,19 our study is the first to our knowledge to combined these treatment modalities into an “intensive therapy” group.
This study shows that analyzing all active intensive therapy together compared with usual care (which in this cohort actually included a moderate percentage of self-directed therapy) does show improvement on all motor and self-reported outcomes and significant differences immediately at the completion of therapy in 2 of the 3 outcome measures. However, while these motor benefits persisted at 24 weeks (6 months) after the completion of therapy, the differences between intensive therapy and usual care were attenuated on the Fugl-Meyer and SIS; whereas, the difference on the WMFT was significantly improved with intensive therapy compared with usual care at 36 weeks. Moreover, while both the Fugl-Meyer and SIS were statistically significant at 12 weeks, only the change in Fugl-Meyer score was actually clinically meaningful. The observation of moderate durable motor benefit at 6 to 9 months posttherapy of the upper extremity is consistent with data from prior studies that included either active groups 20 or a standard care group, 21 where although motor treatment effects were retained, they were not statistically different to the comparison group. The above studies were included in a Cochrane review of repetitive task-training after stroke, 22 and in the Cochrane summary statement about the lack of statistical significance for therapy post 6 months following a stroke. However, while appearing to be incongruent to the statement of retained durable effects, is actually consistent because the generalized statement refers to analysis of the difference between groups as is the case in this analysis and not the rendition of absolute benefit.
The results of the primary analysis of the VA-ROBOTICS study showed that although robot-assisted therapy did improve motor function over 12 weeks, the difference was not significant compared with usual care or intensive conventional therapy. However, the study also showed that robot-assisted therapy improved outcomes over 36 weeks compared with usual care but not compared with intensive comparison therapy. 11 Because the long-term results for robot-assisted therapy and intensive comparison therapy were comparable and not statistically different, the 2 groups were combined into an overall intensive therapy group for these analyses. We note the discrepancy between 12- and 36-week improvements as measured via the WMFT, in that the effect was marginally significant immediately following treatment (12 weeks), but significant at 36 weeks. Both groups (usual care, 1.4 ± 4.3 to −0.2 ± 3.4; intensive therapy, −6.4 ± 3.7 to −7.7 ± 2.9) showed improvement between week 12 and week 36; however, the change in mean difference (7.5 vs 7.8) was small. We attribute this outcome to a substantial reduction in within-group variability at week 36 (22% and 21% reduction in standard deviation in usual care and intensive therapy, respectively). On this basis, we do not necessarily infer a differential effect of intensive therapy versus usual care in terms of functional ability (measured by the WMFT), although this possibility exists and merits further exploration.
In the primary analysis, per protocol, the robot-assisted therapy versus usual care comparisons were restricted to participants enrolled during the same 1 year time period, which therefore excluded robot-assisted therapy data acquired during the second phase of the trial. In this article, we included all patients randomized to robot-assisted therapy and intensive comparison therapy from both periods in the combined intensive therapy group in the comparison against those randomized to usual care therapy. This provided us with additional statistical power to test whether an intensive dose of therapy after the acute stroke period (6 months poststroke) provides improvements in motor function and quality of life compared with usual care and to explore whether there were any sub groups of patients that particularly benefited.
While our inquiry into subgroup characteristics with particular responsiveness to intensive therapy is exploratory, the subgroups were based on biological plausibility and clinical relevance, to within the extent that sample size made for feasible comparisons. For instance, we found that younger patients (<55 years, n = 20) and those with an index stroke between 12 months and 5 years (n = 52) had significantly greater improvement with intensive therapy compared with usual care. Prior literature describing the prognosis of motor recovery as a function of time since stroke primarily has examined this issue in the setting of natural history and has shown that recovery is most dramatic, regardless of the initial motor severity, during the initial 90 days from stroke.23,24 Additionally, our finding that younger patients were more likely to respond to intensive therapy than older patients is consistent with the other reports in the literature showing age as a significant inverse predictor of functional improvement.25-27 In contrast, while many previous studies have shown significant differences in treatment outcomes in subgroups defined by baseline functional scores, we did not find any significant associations. We note that while the distribution of age and severity of stroke in our study is similar to these other studies, the association between training benefit and baseline severity is generally reported only for the subacute stage.28,29 In studies of chronic-stage patients, the relationship is more equivocal.30-32 It is possible that as time from index stroke increases beyond 6 months, the relationship between baseline functional status and differential benefit of therapy may become attenuated.
Very few rigorous studies have examined the potential of motor recovery during chronic stroke following a newly initiated rehabilitation perturbation, such as is the case of VA-ROBOTICS or the study by Klamroth-Marganska et al. 33 Some literature of metadata using multiple stroke rehabilitation studies to describe the effect size on therapeutic benefit of high or low dose have concluded that potential or additional recovery is not dependent on duration since stroke 12 ; however, those conclusions were based on studies only up to 5 years poststroke, whereas in the VA-ROBOTICS study, individuals were included who were up to approximately 20 years poststroke. Thus the VA-ROBOTICS provides evidence that initiating a protocol of intensive repetitive movement in patients more than 5 years poststroke, may not result in significant motor gains.
One notable limitation of this study is that receipt of parallel upper limb therapy was not controlled in any of the treatment groups. However, we note in Table 1 that the proportions of those receiving therapy targeting upper limb was comparable in intensive therapy (21%) and usual care (32%; P = .342). Given the baseline comparability between the groups, we believe that the group-wise differences in treatment effect are robust. Whereas the VA-ROBOTICS trial recorded only the presence of parallel therapy—and not the specific activities conducted therein—we cannot quantify the total level of therapy received by our study participants, and acknowledge it as an uncontrolled random effect. In conclusion, we found that in a chronic stroke population intensive therapy had significant benefit after 12 weeks of therapy and although the benefits persisted, the differences were slightly attenuated after 36 weeks compared with usual care. We also found that younger patients, particularly those younger than 55 years, and those with a shorter time since stroke, particularly those with a stroke <5 years ago, showed greater immediate and long-term improvement in motor function. We emphasize that these findings reflect secondary analyses of the VA-ROBOTICS trial and, thus, should be considered as exploratory, requiring confirmation in future studies.
Footnotes
Acknowledgements
We thank the study Planning Committee; Executive Committee; Data and Safety Monitoring Board; Human Rights Committee; Site Investigators, Coordinators, Therapists, and Evaluators; Study Consultants; Study Chair’s Office; the VA Cooperative Studies Program Coordinating Center staff; the VA Cooperative Studies Program Clinical Research Pharmacy Coordinating Center; The VA Cooperative Studies Program Health Economics Resource Center; The VA Office of Research and Development, Clinical Science Research and Development; and the VA Rehabilitation Research and Development Service, as listed in N Engl J Med 2010;362:1772-83, DOI: 10.1056/NEJMoa0911341.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was conducted by the Cooperative Studies Program, Department of Veterans Affairs, Office of Research and Development, with funding from the Department of Veterans Affairs. Additional support for this analysis was through the Veterans Affairs Connecticut Research and Education Foundation.
