Abstract
Background. Recent studies show that patients with Parkinson’s disease (PD) and freezing of gait (FOG) experience motor problems outside their gait freezing episodes. Because handwriting is also a sequential movement, it may be affected in PD patients with FOG relative to those without. Objective. The current study aimed to assess the quality of writing in PD patients with and without FOG in comparison to healthy controls (CTs) during various writing tasks. Methods. Handwriting was assessed by the writing of cursive loops on a touch-sensitive writing tablet and by means of the Systematic Screening of Handwriting Difficulties (SOS) test in 30 PD patients with and without freezing and 15 healthy age-matched CTs. The tablet tests were performed at 2 different sizes, either continuously or alternatingly, as indicated by visual target lines. Results. Patients with freezing showed decreased writing amplitudes and increased variability compared with CTs and patients without freezing on the writing tablet tests. Writing problems were present during both tests but were more pronounced during writing at alternating compared with writing at continuous size. Patients with freezing also had a higher total score on the SOS test than patients without freezing and CTs, reflecting more extensive handwriting problems, particularly with writing fluency. Conclusions. Writing is more severely affected in PD patients with FOG than in those without FOG. These results indicate that deficient movement sequencing and adaptation is a generic problem in patients with FOG.
Keywords
Introduction
Freezing of gait (FOG) can be defined as a brief, episodic absence or marked reduction of forward progression of the feet despite the intention to walk. 1 This episodic phenomenon was long considered a mere motor symptom of Parkinson’s disease (PD). Recent studies, however, suggest that patients with FOG also experience impairments in cognitive functioning, balance, and movements other than gait.2-5 Furthermore, studies indicate that the motor problems in patients with FOG are not merely episodic, but are also present outside the actual gait freezing episodes. 6 Patients with FOG were shown to have higher gait variability in-between freezing episodes, increased gait asymmetry, and shorter step lengths.7-9
Next to gait, upper-limb movement problems are common in PD as well, and despite their relevance to daily living, they are often overlooked. Patients with PD typically report difficulties in manipulating objects with appropriate speed and dexterity.10-12 Difficulties with manual tasks seem to be associated with problems in maintaining a constant force rather than in force development. 13 Up to now, only a limited number of studies have investigated the occurrence of upper-limb motor deficits in PD patients with FOG in comparison to those without. Vercruysse et al 14 and Barbe et al 7 showed motor blocks of the upper limb during finger tapping, referred to as freezing of the upper limb, with motor features similar to that of FOG episodes. Although the exact relationship between upper-limb freezing and FOG is still unclear, it has been hypothesized that they may be associated.7,14-17 The number of studies that investigated the kinematic movement trajectories outside the freezing episodes is, however, still limited, and most of these studies did not find a clear distinction in upper-extremity functioning between patients with and without FOG.7-9 Furthermore, most previous studies used tasks with limited relevance to daily living.14,15,17-19 In the current study, handwriting performance was examined because this is a relevant task for most patients. 20 Decreased handwriting size and legibility is often one of the first signs of the disease, and handwriting difficulties have been proposed to be potential biomarkers for early disease detection and subsequent disease progression.21,22 In this study, 2 types of assessments were used: tests on a touch-sensitive writing tablet allowing detailed kinematic analysis of the dynamic handwriting trace and a paper-and-pencil test that can easily be used in a clinical setting. The aim of the study was to investigate general differences in handwriting between patients with PD and healthy controls (CTs) as well as to compare handwriting performance in PD patients with and without FOG. Because patients with FOG were shown to experience scaling problems when generating repetitive movement sequences as well as difficulties in set switching,23,24 both writing at constant and variable size were investigated. We hypothesized that in analogy to gait, freezers would show greater problems with amplitude and increased variability of writing, irrespective of disease severity, during both continuous writing and writing with alternating size. This would offer further support for the hypothesis that freezing is not inextricably linked to gait but instead needs to be considered as a general deficit in motor functioning that can affect the whole body. Also, gaining insight into upper-limb movement problems is highly relevant to develop individualized treatment approaches for patients with different subtypes of PD.
Methods
Participants
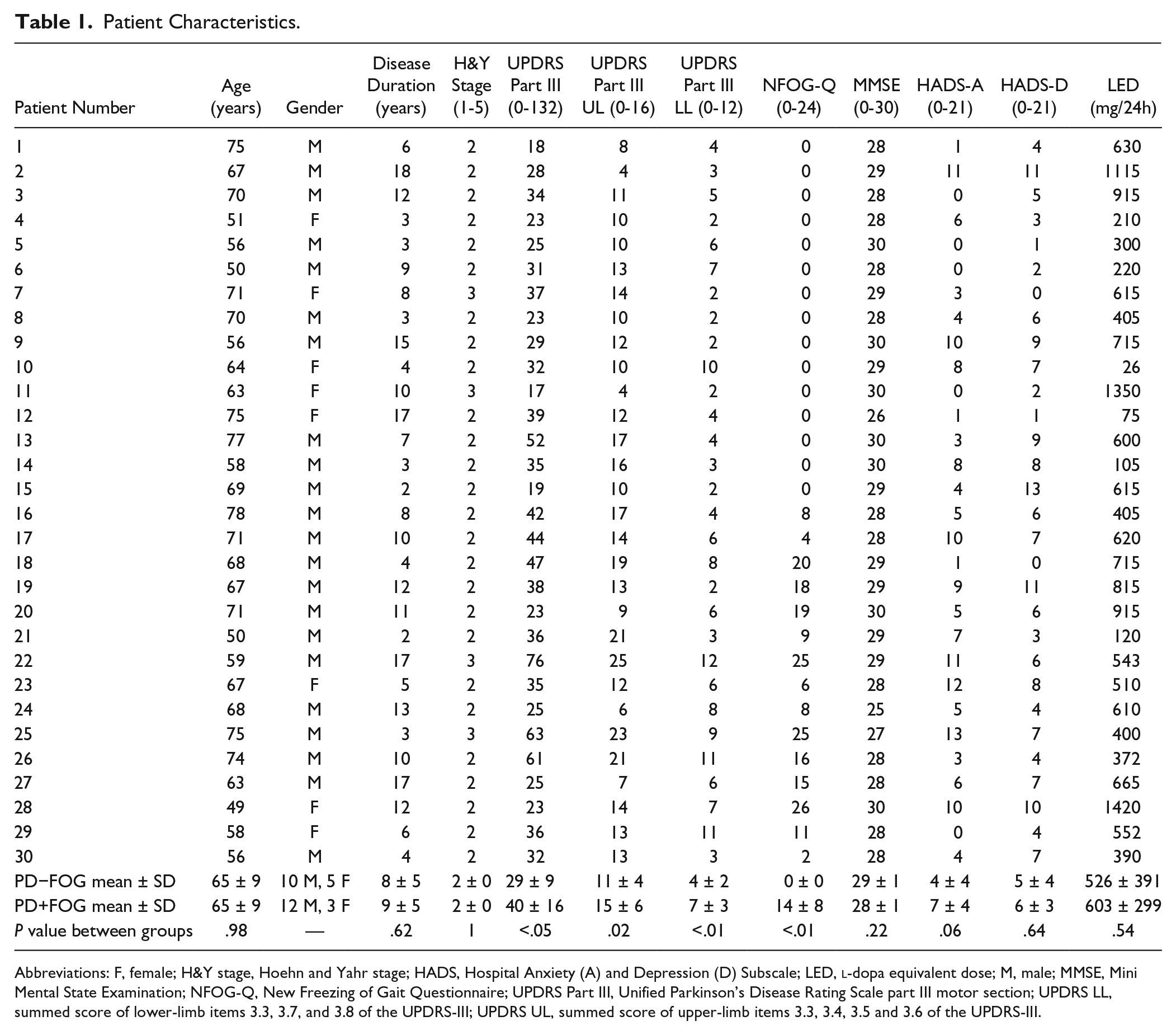
A total of 30 PD patients were grouped according to item 3 of the New Freezing of Gait Questionnaire (NFOG-Q) into freezers (PD+FOG; n = 15) and nonfreezers (PD−FOG; n = 15) and compared with 15 age-matched healthy CTs (64.3 ± 11.2 years; 5 males; Table 1). Freezers and nonfreezers were matched a priori by group according to age and disease severity as measured by the Hoehn and Yahr (H&Y) Scale. 25 Inclusion criteria for the patients were the following: (1) idiopathic PD, diagnosed according to the United Kingdom PD Society Brain Bank criteria 26 ; (2) H&Y stage I to III in the “on” phase of the medication cycle; (3) experiencing writing problems, as measured by the Movement Disorders Society–sponsored Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) part II item 2.7 (score > 1) 27 ; and (4) no history of depression or neurological disorders other than PD. Exclusion criteria were the following: (1) Mini Mental State Examination (MMSE) ≤24, 28 (2) surgery or diseases affecting the upper limb, (3) deep brain stimulation, and (4) vision problems hindering writing. The study design and protocol were approved by the local Ethics Committee of the KU Leuven and were in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki, 1967). After complete explanation of the study protocol, written informed consent was obtained from all participants prior to participation in the experiment.
Patient Characteristics.
Abbreviations: F, female; H&Y stage, Hoehn and Yahr stage; HADS, Hospital Anxiety (A) and Depression (D) Subscale; LED,
Experimental Procedure and Tasks
All test sessions were performed while patients were in the stable “on” phase of the medication cycle—that is, approximately 1 hour after medication intake. Before performing the writing tests, patients completed a clinical test-battery, including the (1) H&Y Scale, (2) MDS-UPDRS-III, 27 (3) NFOG-Q, 29 (4) MMSE, (5) Purdue Pegboard Test, 30 (6) Manual Ability Measure (MAM-16), 31 and (7) Hospital Anxiety and Depression Scale. 32 Per patient, laterality scores were calculated by making a summed score of the unilateral items of the MDS-UPDRS-III. In addition, a summed score of the upper-extremity part of MDS-UPDRS-III items 3.3, 3.4, 3.5, and 3.6 was determined to specifically assess upper-limb motor severity and of the lower-extremity part of items 3.3, 3.7, and 3.8 to assess motor severity of the lower limbs. Next, the 4 writing tests on a custom-made tablet and the Systematic Screening of Handwriting Difficulties (SOS) test 33 were offered in random order. During the SOS test, a standard text is copied on paper during 5 minutes while writing with a regular pen at normal speed. The other tests were performed on a writing tablet with a sampling frequency of 200 Hz and spatial resolution of 32.5 µm. 34 Tests were performed in a quiet room while sitting at a table on a height-adjustable chair. The tasks consisted of writing repetitive cursive loops of either continuously large- (1 cm) or small-sized (0.6 cm) letters—that is, “l” or “e”—or by alternating between both sizes—that is, letters “el” or “le.” The requested amplitude was indicated by colored target zones with a width of 2 mm. Participants were asked to make loops at comfortable speed for 27 s or until the end of the 13-cm reference line was reached. After providing 1 practice trial on paper and 1 on the tablet, the tasks were offered in 3 series, with 6-s breaks between trials. Each trial was preceded by a figure instructing the participant on the upcoming task and initiated with a starting tone.
Movement Analysis
Kinematic data of the writing tablet were filtered at 7 Hz using a fourth-order Butterworth filter and analyzed with custom-written Matlab R2011b software (Mathworks). 34 Data were visually checked for artifacts before kinematic analyses. Movement amplitude (percentage of target size) and velocity (cm/s) were determined by calculating the spatial and temporal differences between the local minima and maxima of each stroke for each size separately. Also, the coefficient of variation of the within-patient writing amplitude (COVampl)—that is, the ratio of SD to the mean expressed as percentage—was computed.
Data of the SOS test were assessed by a researcher blinded to group allocation, according to standardized criteria. 33 Subitems consisted of (1) fluency of letter formation, (2) fluency in connections between letters, (3) regularity of letter height, (4) space between words, and (5) straightness of the sentences. The scores of the subitems were combined to obtain a total score, with higher scores reflecting a lower writing quality. In addition, mean writing size (mm) and overall writing speed (number of letters/5 min) were measured.
Statistical Analysis
Data were analyzed using STATISTICA (Statistical Analysis Software, version 10). Normality and equality of variance were checked for all variables. If all assumptions for parametric testing were met, differences in writing amplitude, COVampl and writing velocity between PD+FOG and PD−FOG were assessed by means of an analysis of covariance (ANCOVA), with group (PD+FOG and PD−FOG) as a between-subjects factor. The MDS-UPDRS-III upper-limb score was included as a continuous predictor because this differed between groups. For the data of the writing tablet, task (continuous size, alternating size) and size (small, large) were added as within-subject factors. Because MDS-UPDRS scores were irrelevant for healthy CTs, the CT group was not included in this analysis. To investigate differences between patients and healthy CTs, an additional ANCOVA was performed with group (PD+FOG, PD−FOG, CTs) as a between-subjects factor and, for the data of the writing tablet tasks, task (continuous size, alternating size) and size (small, large) as within-subject factors. To control for the significant difference in gender between healthy CTs and PD+FOG, gender was added as a covariate. Tukey HSD (honest significant difference) post hoc tests were used in case of significant differences in both analyses. The partial η2 statistic (η2p) was reported to estimate the actual proportion of variation of the dependent variables attributable to each independent factor. Data that did not meet the assumptions of normality and homoscedasticity were analyzed by means of Kruskal-Wallis nonparametric tests, followed by Mann-Whitney U tests for between-group comparisons, with Bonferroni correction for multiple comparisons. Partial correlations were calculated to determine the relationship between the handwriting parameters, and clinical scales and scores on the NFOG-Q while controlling for the effect of MDS-UPDRS-III upper-limb score. For all analyses, α was set at .05.
Results
Subgroup matching revealed no significant differences in age, disease duration, H&Y score and
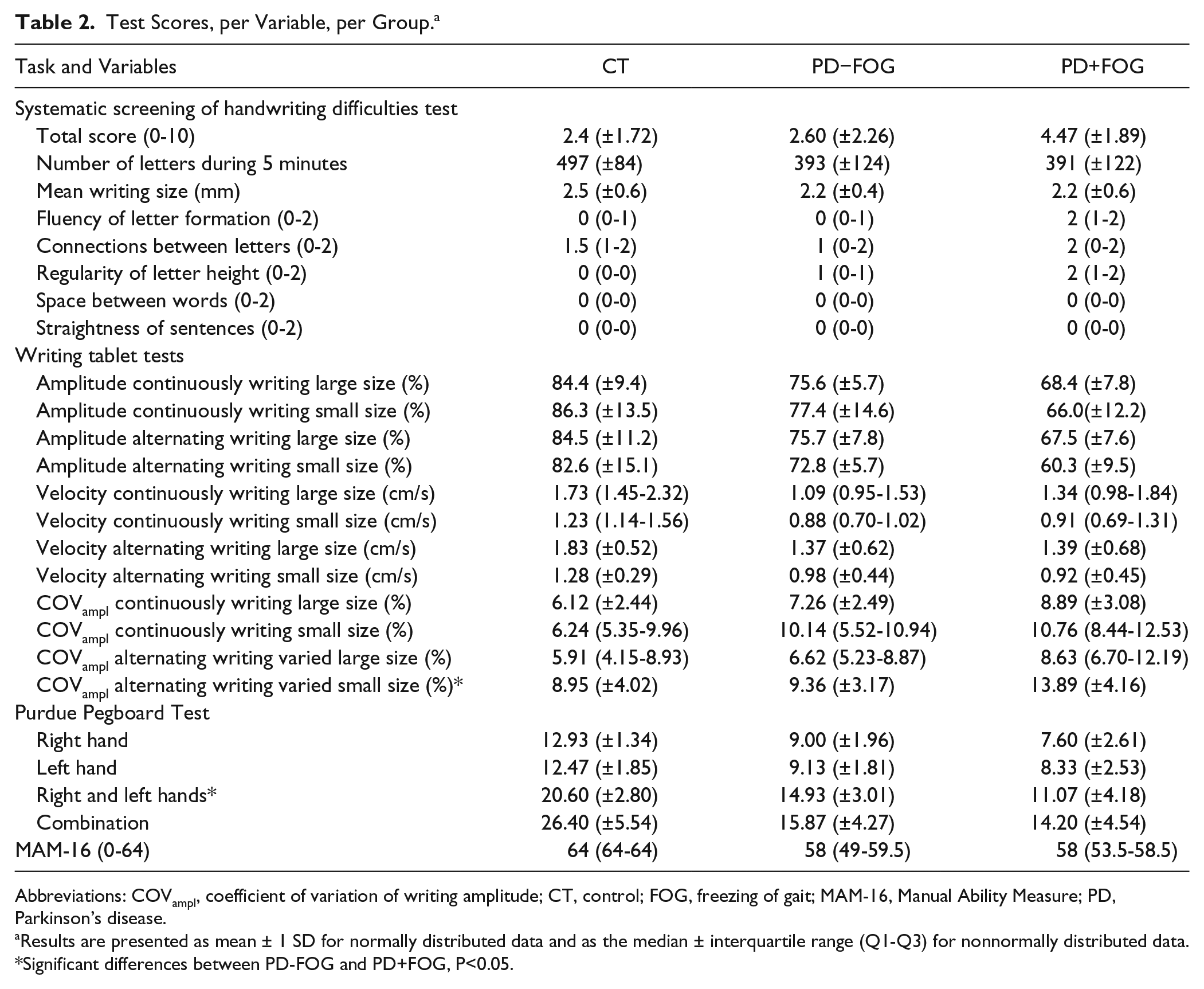
Test Scores, per Variable, per Group. a
Abbreviations: COVampl, coefficient of variation of writing amplitude; CT, control; FOG, freezing of gait; MAM-16, Manual Ability Measure; PD, Parkinson’s disease.
Results are presented as mean ± 1 SD for normally distributed data and as the median ± interquartile range (Q1-Q3) for nonnormally distributed data.
Significant differences between PD-FOG and PD+FOG, P<0.05.
Tests on the Writing Tablet
Writing Amplitude
For all tasks and sizes, PD+FOG was shown to have a significantly smaller writing size than PD−FOG: F(1, 27) = 6.96; P = .01, η2p = 0.20). The main effect for task—F(1, 27) = 4.95; P = .04, η2p = 0.15—showed that writing was smaller when writing size had to be alternated, compared with writing at continuous size for both patient groups. The ANCOVA including all 3 groups also showed a significant group effect: F(2, 41) = 9.99; P < .01, η2p = 0.33. During all conditions, writing was significantly smaller in both PD groups compared with CTs (PD−FOG: P = .02; PD+FOG: P < .01) and in freezers than in nonfreezers (P = .01; Figure 1). There were no significant effects of the covariates, MDS-UPDRS-III upper-limb score and gender.
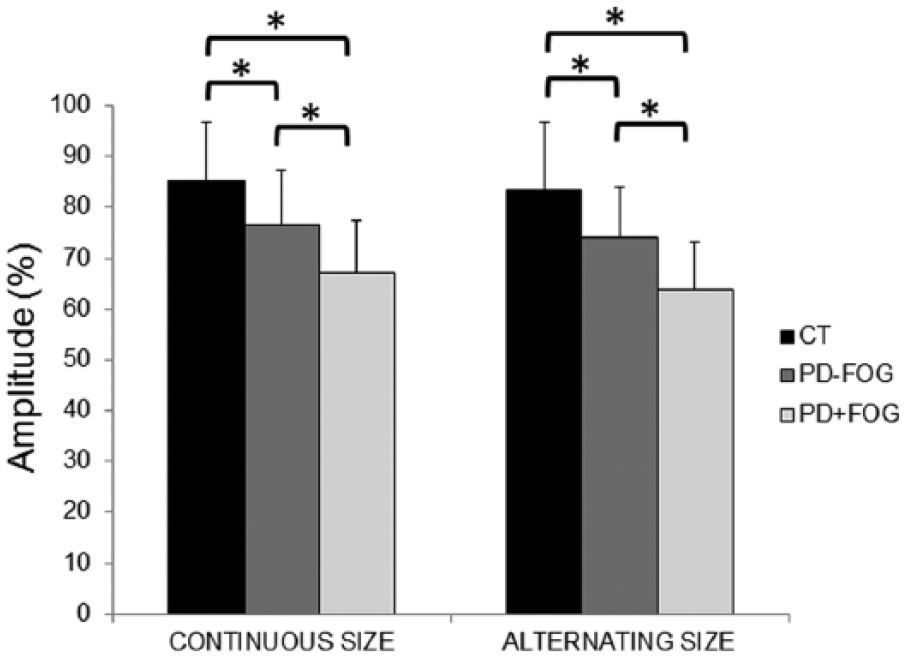
Amplitude on the tablet for writing at continuous and alternating sizes for all groups. Data are presented as group means and SDs. Asterisks indicate significant differences at α <.05.
Coefficient of Variation of the Writing Amplitude
A significant group effect was found when comparing both patient groups, showing that PD+FOG had a significantly higher COVampl than PD−FOG regardless of task and writing size: F(1, 27) = 7.60; P = .01; η2p = 0.22. The main effect for task indicated that COVampl was higher in both groups when alternating between writing sizes in comparison to writing at continuous size: F (1, 27) = 7.41; P = .01; η2p = 0.22. Similarly, a main effect of group was found when comparing all 3 groups: F(2, 41) = 5.36; P < .01; η2p = 0.21. Freezers showed a higher COVampl than CTs (P < .01) and nonfreezers (P = .02). No significant differences were found between PD−FOG and CTs. The main effect for size indicated that COVampl was higher during small-sized in comparison with large-sized writing: F(1, 41) = 12.00; P < .01; η2p = 0.23. No significant effects were found for both covariates.
Writing Velocity
Both ANCOVAs showed no differences in writing velocity between groups. The main effect for size indicated that writing velocity is higher when writing at large compared with small size: F(1, 27) = 8.24, P < .01, η2p = 0.23 for the comparison between both patient groups and F(1, 41) = 35.61, P < .01, η2p = 0.47 for the comparison between the 3 groups. No significant effects were found for both covariates.
Systematic Screening of Handwriting Difficulties Test
No significant differences in total score on the SOS test were found when only the patient groups were included in the analysis. In contrast, a significant group effect was found when comparing all 3 groups: F(2, 41) = 3.27; P = .04, η2p = 0.14. PD patients with FOG had a higher score than patients without freezing (P = .01) and CTs (P < .01), reflecting lower handwriting quality. No differences were found between PD−FOG and CTs. Bonferroni corrected nonparametric analyses of the subitems showed a significant group difference in fluency of letter formation—χ2(2, 41) = 15.20; P < .001—indicating more difficulties in freezers compared with both nonfreezers (P = .02) and CTs (P < .01). No significant difference was found between CTs and PD−FOG. Regularity of letter height also differed between groups—χ2(2, 41) = 8.23; P = .016—with more irregularity in freezers than CTs (P = .04) and no differences between the other groups.
Fine Motor Skills
Purdue Pegboard Test
The direct comparison between PD+FOG and PD−FOG did not show any group differences for any of the subtests of the Purdue Pegboard Test. The ANCOVA including all 3 groups, in contrast, showed a main effect for group on all 4 subtasks of the Purdue Pegboard Test: right-handed, F(2, 42) = 27.60, P < .001, η2p = 0.49; left-handed, F(2, 42) = 16.56, P < .001, η2p = 0.34; right- and left-handed, F(2, 42) = 30.08, P < .001, η2p = 0.51; and combination, F(2, 42) = 28.31, P < .001, η2p = 0.51. Both patients with and without FOG differed significantly from healthy CTs (P < .001 on all subtasks). PD+FOG performed worse than PD−FOG on the right- and left-handed subtask in which participants have to place the pins in the holes alternating between the right and left hand (P < .01). On the other 3 subtasks, no between-subgroup differences were found.
Manual Ability Measure
Nonparametric analysis showed a significant difference between groups: χ2(2, 41) = 29.92; P < .001. Post hoc tests revealed differences between both freezers and CTs (P < .001) and nonfreezers and CTs (P < .001). No differences were present between PD+FOG and PD−FOG.
Correlation Analyses
Partial correlations, controlling for the effect of MDS-UPDRS-III upper-limb score, revealed a significant correlation between writing amplitude of the large-sized loops when writing at alternating size (ρXY-Z = −0.40) and the NFOG-Q. NFOG-Q did not correlate significantly with the other handwriting parameters. No significant correlations were found between the NFOG-Q, Purdue Pegboard Test, and MAM-16.
Discussion
The present study revealed severe handwriting problems in patients with PD, which were more pronounced in patients with FOG than those without. These problems were expressed as an increased writing variability as well as difficulties in maintaining an appropriate movement amplitude and fluency. Because handwriting problems occurred both during sequential writing movements and during writing at alternating size, the current results provide evidence of both difficulties in continuous movement control as well as of a set switching deficit during handwriting in freezers. The extensive handwriting assessment that was performed in the current study consisted of a paper-and-pencil test and several tests on a touch-sensitive tablet. The tests on the tablet included colored zones to standardize performance. Because visual cues have been found to normalize handwriting amplitude in PD, the handwriting difficulties that were found in this study, particularly in freezers, are even more striking. The freezing-related handwriting problems were found while on medication and while subgroups were matched for age, disease duration,
Writing problems were most clearly apparent during writing on the tablet at alternating size, which could be perceived as more difficult for patients. Furthermore, a significant correlation was found between the NFOG-Q score and writing amplitude during the writing of the large loops when writing at alternating size. In contrast, the NFOG-Q score did not correlate significantly with handwriting variables during writing at continuous size. These impairments in handwriting are in accordance with the timing, scaling, and coordination deficits that were reported earlier in PD+FOG during gait outside the freezing episodes.35,36 The difficulties in flexibly changing between variable writing sizes may be interpreted as a set switching problem.23,24 Although PD patients with FOG are known to experience additional cognitive problems and PD has been associated with set switching difficulties in both the cognitive and motor domain, Smulders et al 37 found that difficulties in changing stepping direction in patients with FOG were mainly attributed to delayed motor switching.2,38 This reduced ability to flexibly adapt movement parameters when environmental conditions change has been ascribed to reduced structural connectivity between the pedunculopontine nucleus and the frontal/prefrontal cortices in PD+FOG. 39
During gait, patients with FOG often show a gradual step-to-step reduction in combination with an overall reduced step length, known as the “sequence effect.” 40 This effect is known to disappear with the use of external cues. 41 In the current study, because the tests on the tablet were all performed in the presence of visual cues, a true sequence effect during writing was not found. However, we did find a marked amplitude deregulation. A possible explanation for the difficulties in maintaining a constant movement amplitude during sequential movements in PD in general may be found in the fact that the basal ganglia provide internal cues to the supplementary motor area to enable submovements to be correctly linked together. 42 Moreover, recent studies in patients with FOG have shown specific impairments in the spatiotemporal dynamics between frontostriatal neural networks in this group.23,43 Freezing episodes during a virtual reality gait task were shown to be related to the degree of impairment of functional connectivity between the basal ganglia network and the bilateral cognitive control network, including the dorsolateral prefrontal cortex and the posterior parietal cortex. 43 In line, FOG has been associated with diffuse white matter damage in frontostriatal and other tracts as well as impaired executive network connectivity identified by means of resting state MRI.44,45 The above-mentioned neural changes that were shown specifically in patients with FOG may also lie at the base of difficulties in other sequential movements, including writing movements, in this group.
The results of the tablet test were only partially confirmed by the SOS test on paper. The SOS subitem identifying increased difficulties with movement fluency did discriminate between patient groups. Also, statistical analysis of the total SOS score comparing all 3 groups showed the worst handwriting quality in the PD+FOG. However, the direct comparison between PD+FOG and PD−FOG and between PD−FOG and healthy CTs did not reach significance. This may be a result of the limited sample size, resulting in a lack of power to detect more subtle differences between the groups. A study on a larger sample size is currently ongoing to investigate the sensitivity of the SOS test in different subgroups of patients with PD. An additional limitation of the study is that although patient groups were matched for H&Y scale, they were not matched for MDS-UPDRS-III scores. To compensate for the potential effect of differences in upper-limb disease severity, the MDS-UPDRS-III upper-limb score was added to the comparison between PD+FOG and PD−FOG as a covariate.
Unlike the writing test deficits, the differences between freezers and nonfreezers were nearly absent on the clinical scales for upper-limb functioning that were used in this study. No differences between PD+FOG and PD−FOG were found on the MAM-16, and when adding the MDS-UPDRS-III upper-limb score as a covariate, no differences remained present on the Purdue Pegboard Test. Furthermore, these tests did not correlate with scores on the NFOG-Q. This may indicate that freezing-related deficits are writing specific or that the measurement scales that are currently used to quantify upper-limb problems in PD are not sensitive enough, as also highlighted in a recent review by Proud et al. 46 The lack of sensitive screening instruments for upper-limb dysfunction in PD may partially explain why previous studies investigating finger tapping, arm swing, and asymmetry in repetitive hand movements did not identify differences in upper-limb motor control in patients with and without FOG.7-9 The current study, therefore, supports the addition of a thorough handwriting evaluation to PD patients’ clinical assessment, so as to be able to pick up the specific difficulties patients experience regarding upper-extremity functioning and adapt their treatment accordingly (eg, referral to physical therapy for upper-limb rehabilitation). The broad spectrum of handwriting aspects that were shown to be affected supports the claim that PD handwriting problems should be referred to as dysgraphia, a term covering a wide range of writing impairments, instead of micrographia, suggesting only a diminishment in writing size. Current work is being undertaken to develop a novel sensor system integrated with a digital pen to make the evaluation of handwriting in PD even more feasible and user-friendly in the future. 47
The current findings show that motor problems in freezers may be indicative of a wider systemic motor control deficit of sequential movements also reflected in other daily-life tasks. This is in line with previous studies showing additional problems in speech, lower-limb, and other upper-limb movements in patients with FOG.4,14,48 Several studies also reported an association between the nontremor dominant phenotype of PD and FOG.49,50 Our findings point to a new avenue for future studies investigating whether patients within the postural instability and gait disorder subtype of PD, who are known to develop FOG more quickly, 5 also show a tendency to have more severe upper-limb problems. Up to now, it is unknown whether FOG shares a common neural background with other motor problems reported in patients with freezing. FOG has been attributed to pathological processes within different areas, including brainstem structures such as the pedunculopontine nucleus but also frontostriatal pathways.51-53 Recent studies comparing patients with and without FOG revealed altered brain activation in the basal ganglia, frontoparietal regions, and the mesencephalic locomotor region in the brainstem.40,54 So far, the only study that investigated brain activity in freezers and nonfreezers during upper-limb tasks found a combination of hyperactivity in subcortical regions and cortical hypoactivity during the execution of finger tapping in PD+FOG. 55 Similar to finger tapping, handwriting is a sequential task that may highly depend on basal ganglia-cortical interaction because it requires the correct implementation and automatic running of movement amplitude and frequency. 4 As such, alterations in the neural circuitry similar to those reported by Vercruysse et al 56 may underpin the writing differences between patients with and without FOG that were presented here.
The current findings are highly relevant for the rehabilitation field and point to the importance of developing individually tailored intervention strategies for different subgroups of PD patients. Earlier work showed that patients with FOG respond differently to different types of cueing than nonfreezers.55,57 Similarly, the present study suggests that during retraining of writing skills, patients with FOG might need additional guidance to increase writing amplitude and movement stability as well as to improve movement fluency. Potentially, auditory cues might be used on top or instead of visual cues in this group.
A limitation of the current study is that handwriting was tested only while patients were in the “on” cycle of the medication. Although we believe that this condition best reflects upper-limb problems during daily living, future studies investigating patients in the on and off states of the medication can shed further light on the dopaminergic component of the deficits observed between patient subgroups. Furthermore, patients were only matched for H&Y stage and not for MDS-UPDRS-III scores. Because some of the differences between the patient groups were no longer present after adding the MDS-UPDRS-III upper-limb score as a covariate, it cannot be ruled out that disease severity may have affected our results.
In conclusion, we found that handwriting is more severely affected in PD patients with FOG than in those without. The current study supports the idea that more severe dysgraphia is a representation of faulty movement organization that affects PD patients with FOG at multiple domains.
Footnotes
Acknowledgements
We want to express our gratitude to all patients and controls who participated voluntarily in this study. We thank Prof Wim Vandenberghe of the University Hospital Leuven for his help with the recruitment of patients and Ir. Marc Beirinckx for developing the writing tablet and providing technical support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was funded by the Research Foundation—Flanders (FWO) (Project G.0906.11). E. Heremans is a Postdoctoral Researcher and E. Nackaerts a Research Assistant at the Research Foundation—Flanders (FWO).
