Abstract
Introduction
More than 50% of stroke survivors suffer from impaired motor function in the upper extremity. 1 Because of the huge impact, motor rehabilitation is the focus of clinical work in daily clinical practice as well as of neurorehabilitation research. The affected limb typically is weak, slow, and lacks coordination and dexterity with a loss of the ability to generate fractionated movements, and spasticity may be present.
Treatment paradigms for the affected limb may be classified into augmenting techniques, task-specific exercises, and priming techniques. 2 Priming techniques aim at increasing the excitability of the involved neural networks and promoting plastic reorganization. Mirror therapy is an example of a priming technique for improving motor function after stroke.3,4 In mirror therapy, a mirror is placed in the midsagittal plane, which reflects movements of the unaffected limb. The mirror reflection creates the illusion that the affected side moves with a normal movement pattern, creating a form of “virtual feedback.”
A number of trials on mirror therapy in stroke have been reported,5-7 and various mirror therapy protocols are composed and evaluated. Although many studies reported on a positive effect of mirror therapy on motor recovery after stroke,8-10 the exact working mechanisms are not fully understood. Selles et al 11 examined the effects of unimanual and bimanual mirror therapy protocols on motor learning after stroke. The largest intervention effects were found in the condition in which patients only trained the affected arm without a mirror and in the condition in which patients moved the unaffected arm in front of the mirror while observing the mirror reflection.
Several authors ascribed the effects of mirror therapy to neuronal activations of frontoparietal circuitries in the brain, involving mirror neurons.12,13 Mirror neurons discharge during both action execution and action observation (AO) of corresponding movements. 14 Previous functional magnetic resonance imaging studies showed that similar parts of the neuronal network are involved in AO as in mirror therapy.14,15
AO improves motor learning in healthy individuals16-18 and may be a promising tool in regaining motor function after stroke.19-21 A recent pilot study showed that self-directed video therapy improves motor recovery after stroke. 22 New motor skills become part of the patient’s motor repertoire, and observation activates the mirror neuron system. 23 The beneficial effects of mirror therapy may, at least partly, be explained by working mechanisms similar to that in AO. This study addresses the question of whether a mirror therapy–based AO protocol improves motor learning in the chronic stage after stroke by studying a simple upper-arm motor task.
Methods
Study Sample
Participants in the chronic stage after stroke were recruited from the outpatient clinic of Rijndam Rehabilitation Centre in Rotterdam, the Netherlands. The inclusion criteria were (1) a Brunnström score for upper-extremity function between III and VI, (2) a home dwelling status, and (3) at least 6 months poststroke. Patients with neglect (indicated by the star cancellation test and/or reported by the occupational therapist or psychologist), comorbidities that influence voluntary upper-extremity function, or who suffered from multiple strokes were excluded. All participants gave written informed consent, and the study was approved by the Medical Ethics committee.
Power Analysis
The sample size was calculated on the data from the study of Cirstea and Levin, 24 on which our experimental paradigm was based. We calculated that 12 participants in each group would be sufficient to have an 80% chance of detecting a statistically significant difference of 0.25 s between groups. To increase power, we aimed for a total of 20 participants in each group.
Clinical Assessment
Before starting the experiment, motor ability of the upper extremity was evaluated using the Fugl-Meyer assessment (FMA). 25 The upper-extremity part of the FMA examines voluntary upper-limb movements, tendon reflexes, range of motion, muscle tone, and the ability to execute proximal and distal movements inside and outside movement synergies. The FMA scores range from 0 to 66, with higher scores indicating better motor function. 26
Randomization
Participants were randomly allocated to the AO or control observation (CO) group. To minimize possible confounding effects of motor ability and age, we stratified participants into groups based on age (younger than 55 years of age or 55 years and older) and motor function (FM score <50 or a FM score of ≥50). In this way, we created 4 strata. Stratified randomization was achieved by performing a separate randomization procedure within each stratum. The randomization was based on a computer-generated random code using blocks of 20 patients and using sealed envelopes.
Motor Task
Participants performed a simple upper-arm reaching task. The task consisted of making a reaching movement from a starting position located next to the chair, toward a target located in front of them (Figure 1). A light indicated the start of each trial. After hitting the target, participants had to move their arm back toward the starting position and wait until the light turned on again for the next reaching movement. Participants were instructed to make the upper-arm reaching movement as fluently and fast as possible toward the target end point.
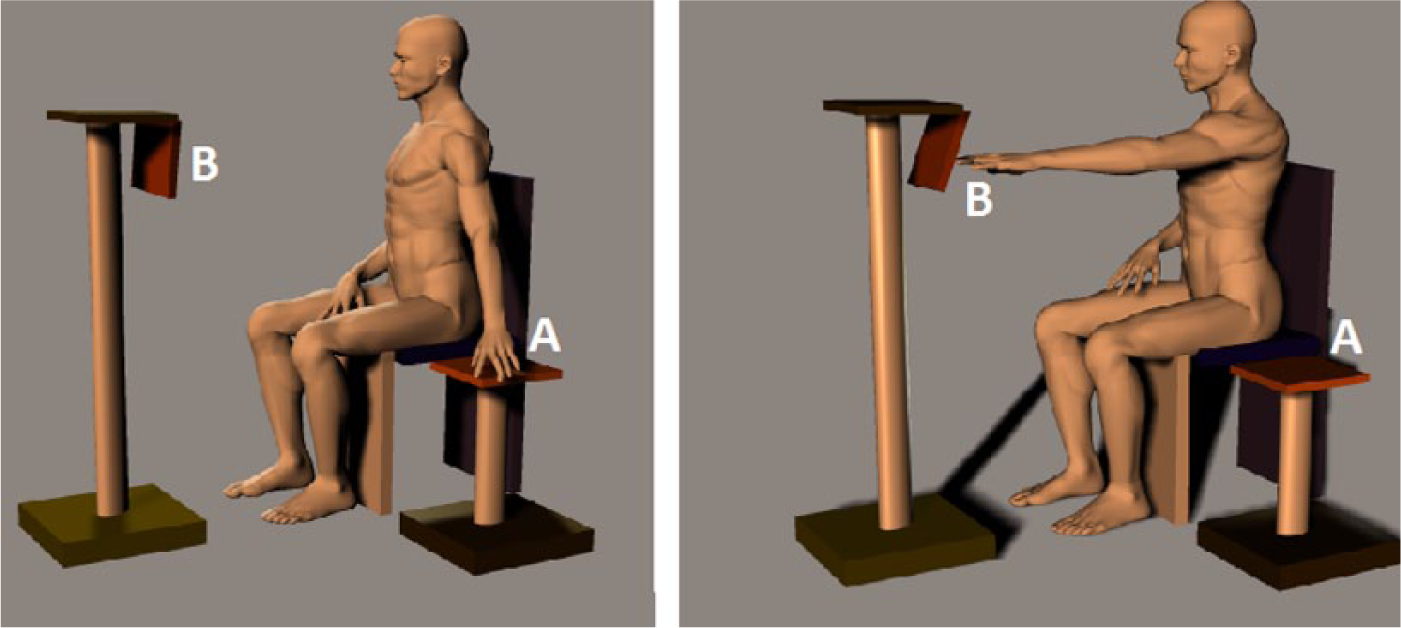
Side view of the experimental setup: Participants were instructed to move their arm from a position located next to the chair (A) toward a target located in front of them (B). The distance of the target was adjusted to the body dimensions of each participant and the participant’s ability to extend the arm, such that at the end of the reaching movement, the fingers would just hit the target.
Experimental Procedure
Participants start the experiment with a set of 10 upper-arm reaching movements with the affected arm, which served as baseline measurement (T0). Hereafter, participants performed a set of 10 trials with their unaffected arm, which was recorded with a digital camera and used for AO. After baseline measurements, participants start the training within the allocated condition. The training contained sets of affected-arm reaching movements, alternated with periods of observation (AO or CO). As in mirror therapy, the video screen was positioned in the midsagittal plane. After training, participants performed another set of 10 affected-arm reaching movements, which served as follow-up measurement (T1). Figure 2 shows the schematic representation of the experimental procedure.
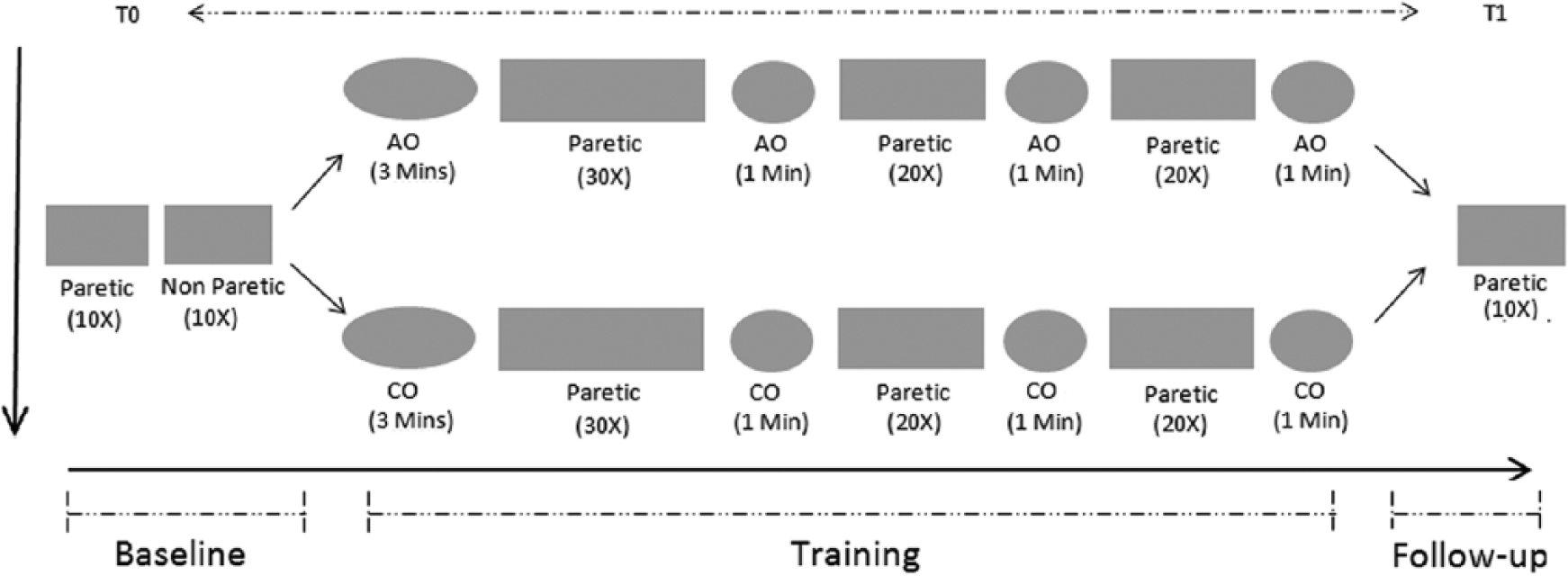
Schematic representation of the experimental setup. Baseline measurement: 10 affected arm movements (T0) followed by 10 unaffected arm movements, which were videotaped with a digital camera. Training: affected arm movements were alternated with either action observation (AO) or control observation (CO). Follow-up: 10 affected arm movements (T1). The numbers under each block represent the amount of physical repetitions, whereas the time in minutes indicates the duration of observation.
Experimental Conditions: AO and CO
In this study, AO is offered in such a way that the observation of the upper-arm reaching movements is almost similar to what patients would see in the mirror during mirror therapy. AO was displayed from a first-person perspective, so the observed movements correspond to the orientation of the participant. To provide participant-specific videos, reaching movements from the unaffected arm were videotaped and mirrored. Thereby, we tried to create maximal postural familiarity and the illusion that the affected arm performed the reaching movements in a normal movement pattern. Participants were instructed to focus on the arm reaching movements, which were displayed on the computer monitor (Figure 3). In addition, participants were instructed to observe the AO with the intention to reproduce the reaching task with the affected arm because this is proven to be a beneficial strategy in observational learning.26,27
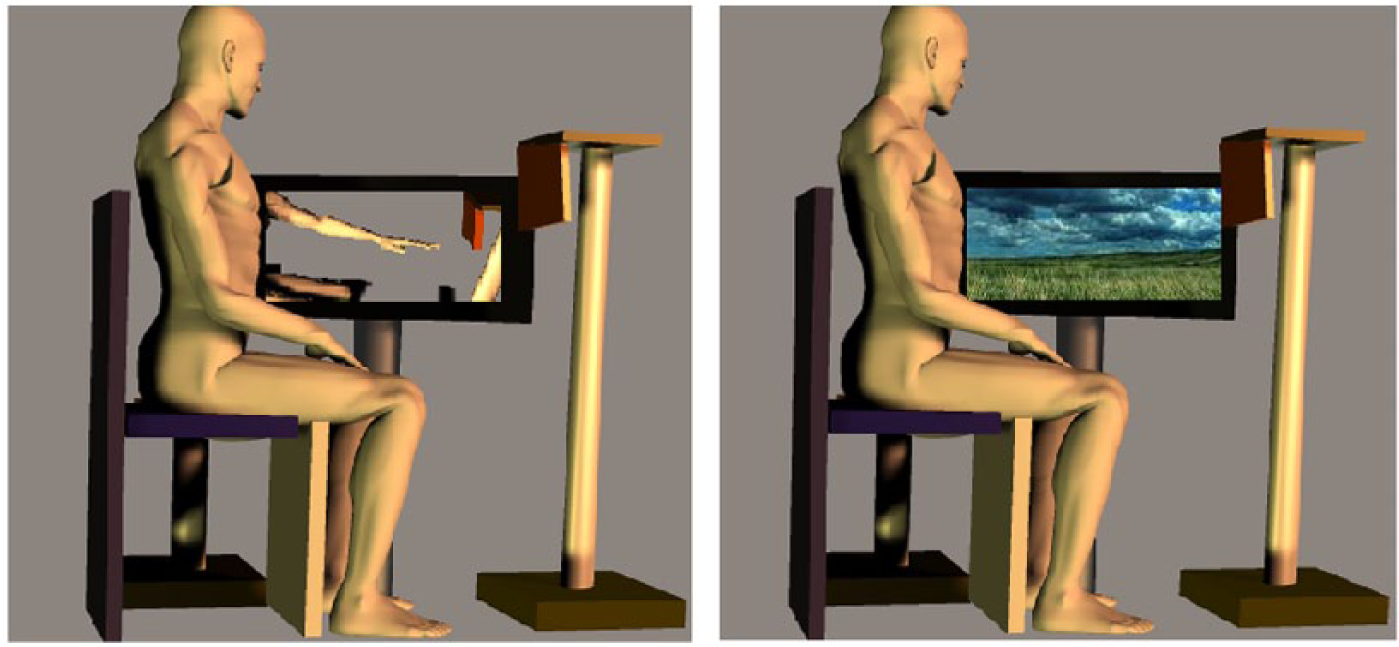
Side view of the experimental setup during action observation (AO) and control observation (CO): Participants in the AO group observed mirrored reaching arm movements of the videotaped unaffected arm, as can be seen on the left image. Participants in the CO group observed a slideshow with static photographs of landscapes, as can be seen on the right image.
Participants in the CO group observed a slideshow with static photographs of landscapes as control stimuli. The photographs contained no images of humans or animals and were selected such that they would not trigger the mirror neuron system; therefore, they were not likely to interfere with the goal in this study.
Data Acquisition and Analysis
Accelerometer data measured at the arm and target end point were recorded using a TEMEC Vitaport 3 digital recording system. A switch was used to turn on a light, which indicated the start of each reaching movement, and simultaneously left a marker signal in the recorded time series. Two 2D acceleration sensors (biaxial piezo-resistive accelerosensor, Temec Instruments, Netherlands) were placed on the ventral side of the wrist, measuring accelerations during the reaching movements in 3 dimensions. Another 2D acceleration sensor (bi-axial piezoresistive accelerosensor, Temec Instruments, Netherlands) was attached at the target end point and measured acceleration in 2 dimensions to indicate the end of each reaching movement. Accelerations were recorded at a sample frequency of 128 Hz.
Because movement time was found to be a predictive measure in learning a simple upper-arm motor task after stroke,11,24 it was selected as the primary outcome. Movement time was defined as the time between the start signal derived from the marker signal and hitting the target end point, derived from the accelerometer signal on the target end point. All data were analyzed using custom-made Matlab algorithms (7.4.0 r2007 a).
Statistical Analysis
To evaluate the experimental condition effect on motor performance, change in movement time between baseline (T0) and follow-up (T1) was measured and compared between groups. For each block of 10 movements, we calculated the median movement time of 9 trials (2-10), omitting the first reaching movement. Median values for movement time were used for group analysis. The assumption for normality was checked by visual inspections of the histogram and normal curve and by performing a Shapiro-Wilk test. Independent Student
We evaluated the effect of the following possible confounders on the experimental conditions: age, gender, time since stroke, stroke type (hemorrhagic or ischemic), FM score, affected side (dominant or nondominant), group (AO or CO), and movement time at baseline measurement (T0). To do so, first, we individually added each possible confounder to a multiple regression model, with experimental condition as the other dependent variable using the Enter-method, selecting each confounder that changed the β of the experimental condition effect by more than 10%. As a second step, we entered each selected confounder in a forward regression model, in order of confounding effect size. If the added confounder did not further change the β of the experimental condition effect by more than 10%, it was excluded from the final regression model.
All statistical analyses were 2-tailed and performed using IBM SPSS Statistics 20, and a
Results
A total of 134 participants were eligible for inclusion, of whom 37 participated in the present study (Figure 4). Table 1 shows group characteristics of participants allocated to the AO (n = 18) and CO group (n = 19). As can be seen, the poststroke time period was relatively long in both groups; most participants suffered from ischemic stroke and had a middle cerebral artery lesion. Based on the FMA, there was no baseline difference in upper-extremity function between both groups (
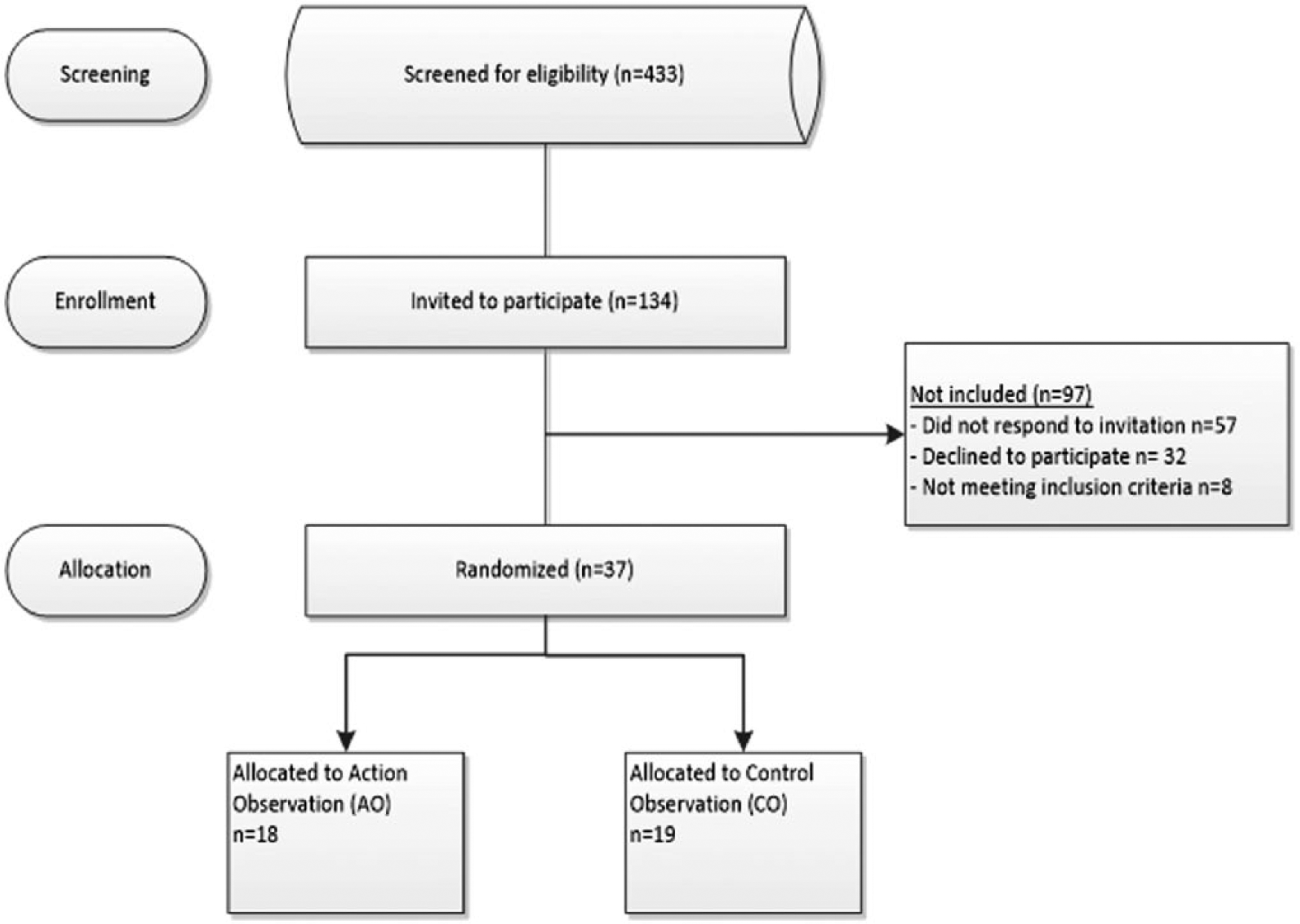
Flowchart describing the study sample and distribution of participants.
Patient Characteristics. a
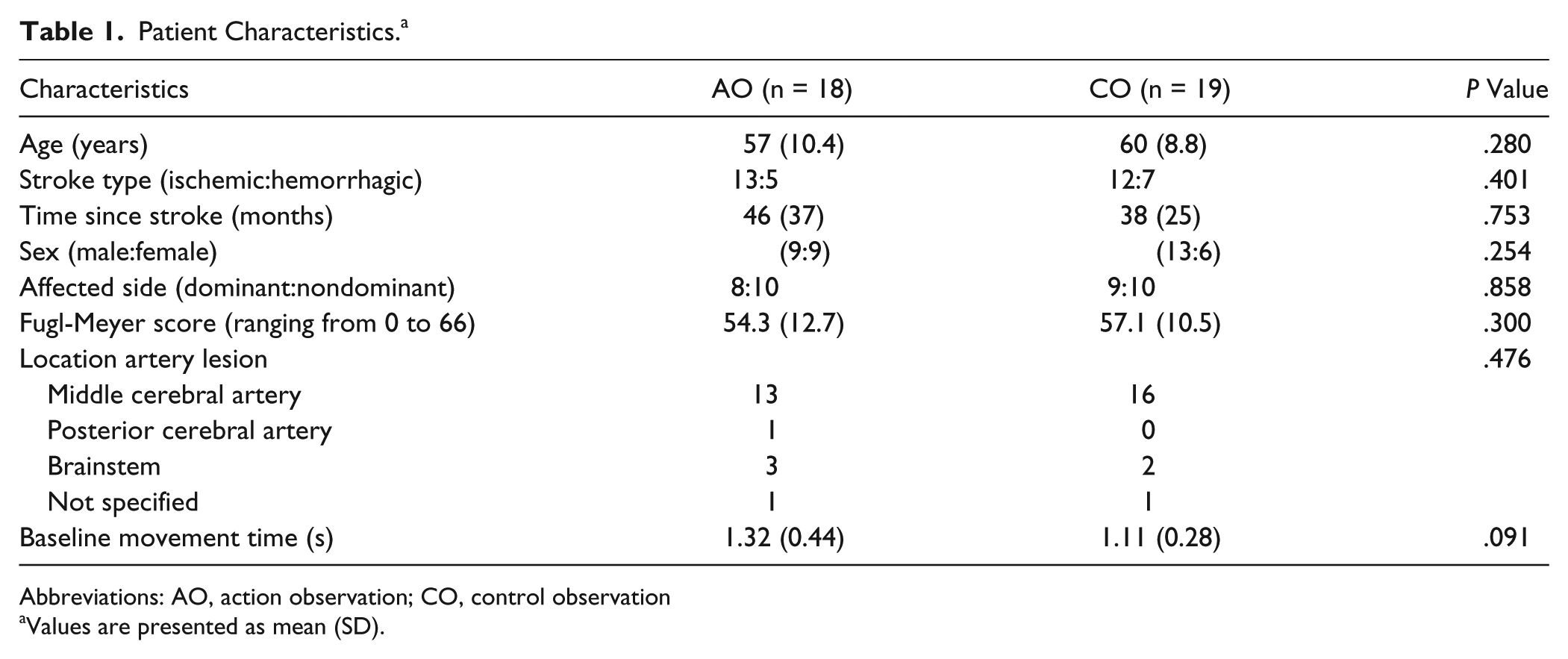
Abbreviations: AO, action observation; CO, control observation
Values are presented as mean (SD).
Movement time decreased significantly in both groups: 18.3% in the AO group (
The extended regression analyses indicated that baseline movement time (β = 0.001;
Discussion
To our knowledge, this is the first study ever to specifically investigate a mirror therapy–based AO protocol. Most studies on mirror therapy mainly focus on its effect on motor learning 10 and specific neuronal activations involved.8,13,15,28 Training without a mirror or with a nontransparent screen between both arms are commonly used control conditions, generally showing that mirror therapy is beneficial for increasing motor learning after stroke.8,9,11 The present study specifically investigated the effects of a mirror therapy–based AO protocol on motor learning in patients in the chronic stage after stroke. A significantly larger decrease in movement time was found in the AO compared with the CO group. The results indicate that a mirror therapy–based AO protocol contributes to motor learning after stroke and, at least partly, clarifies the beneficial effects of mirror therapy on motor learning.
The primary aim of this study was not to create a clinically meaningful change in arm function that would persist over longer time periods but merely to study the short-term impact of AO on learning a simple upper-arm motor task after stroke. This experimental approach is in line with previous studies that suggest that simple motor tasks can be selected to study the effects of different training paradigms on motor learning in an efficient way.11,15,28
Our findings indicate that AO increases motor learning when a simple upper-arm motor task was trained. Previous studies on AO in stroke evaluated the effects of several weeks of AO therapy, using functional outcome tests such as the Frenchay arm test, Wolf motor function test, or the Box and Block test.22,29 In the study of Ertelt et al, 22 patients in the chronic stage after stroke significantly improved motor function following a 4-week video therapy program compared with a control group. Patients entering the AO group observed sequences of daily arm and hand actions with increasing complexity, from 3 different perspectives on a television screen positioned in front of the patient. Beneficial effects of AO on motor learning were also found in the subacute phase after stroke by Franceschini et al, 29 who studied AO therapy as an add-on treatment to the standard rehabilitation of arm function. Patients observed 3 different motor sequences with increasing complexity from a first-person perspective, based on actions of daily living, which were performed by young nondisabled men and women. These studies provide evidence for an additional positive impact of AO on motor learning after stroke, which matches the findings of the present study. However, the AO setup and training paradigms selected in these studies do not allow transferring the results to the mirror therapy–based AO paradigm investigated in the present study.
In this experiment, a simple upper-arm motor task was selected to study the effects of AO on motor learning, which allowed us to examine change in motor performance over relatively short time periods. It should be noted that repetitive practice of sequentially simple motor tasks may lead to motor improvements by adaptation, 30 and short-term effects of AO on motor learning may not transfer to more complex motor and more clinically meaningful tasks, which are typically learned more slowly over multiple training sessions, such as hand dexterity or hand function in daily life.31,32
Despite randomized stratification, there were some baseline differences that might explain different intervention effects between groups. For example, patients in the CO group had, on average, a better Fugl-Meyer score and a faster baseline reaching movement. Therefore, we performed an extended regression analyses to control for possible confounding variables. Although we indeed found that baseline movement time and type of stroke were independent confounding variables of the condition effect, the condition effect was still significant after control for baseline values, indicating a Group × Time interaction effect.
Some limitations of the present study should be discussed. First, the mirror therapy–based AO slightly differed from the mirror reflection during mirror therapy. In contrast to the mirror reflection, the computer screen has limited resolution and depth perception. In addition, to gain a clear view, the computer screen was positioned close to the participant, and therefore differs slightly from the mirror position during mirror therapy. However, the presented AO corresponds to the mirror reflection and creates the illusion that the affected arm performed the reaching movements. Second, the relative heterogeneous sample should be mentioned, with participants suffering from either hemorrhagic or ischemic stroke. Including only participants with specific brain lesions was not feasible and would have decreased the external validity. A third limitation is that the number of participants in both groups was relatively small. The power analysis was based on findings of Cirstea and Levin, 24 who showed a 0.25-s improvement in movement time in the affected no mirror condition, which was similar to the improvement in the present study (0.24-s improvement). Therefore, there seems to be sufficient power to find a clear significant difference in the main outcome measure. A fourth possible limitation of the present study is that we did not further analyze movement trajectory, which may have provided additional information regarding motor learning after stroke. However, previous research on motor learning after stroke showed that movement time was a predictive measure when analyzing a simple goal-directed upper-arm reaching task.11,24
The present study showed that a mirror therapy–based AO protocol contributes to motor learning in patients in the chronic stage after stroke and, at least partly, clarifies the beneficial effects of mirror therapy on motor learning. However, many questions remain. For example, it is still not clear whether early priming techniques, applied in the subacute stage after stroke, may actually promote recovery beyond the level of spontaneous recovery. Additionally, it might be that the effects of mirror therapy or AO on motor learning can be maximized by combining it with other priming techniques, such as transcranial direct current stimulation. Answering these types of questions will help in changing the pragmatic, experience-based orientation of neurorehabilitation to an experimental and theory-based approach.
Conclusion
The present study showed that a mirror therapy–based AO protocol contributes to motor learning in patients after stroke.
Footnotes
Authors’ Note
Trial number: ISRCTN40128145.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
