Abstract
Introduction
Stroke is a leading cause of adult disability and usually occurs in the elderly. Persons after stroke require long-term rehabilitation treatment to release the uncomfortable symptoms and partially restore motor functions for a more independent life. Device-assisted rehabilitation programs have been proposed to assist human physical therapists in long-term service during the past decades. Among them, rehabilitation robot and neuromuscular electrical stimulation (NMES) are 2 main techniques that have been widely integrated into the rehabilitation practices.1,2
The extent of the motor restoration after stroke is highly associated with the intensive practice of the affected limbs by the neuromuscular efforts from the paretic side with desired muscle patterns.3-5 The practice with the voluntary effort of the affected limb could result in better motor outcome and more sustainable improvement than treatments with continuous passive motions (CPM).6,7 Physical limb practices with less compensatory motions could minimize the ‘learned disuse’ that stroke survivors are gradually accustomed to managing their daily activities without using certain muscles. It has been considered as a significant barrier to maximizing the recovery of poststroke motor function. 8 Robots with motors can provide external assistance to the paralyzed limb in poststroke physical training with intensive and repeated movements. Various robotic systems have been proposed for joint rehabilitation after stroke, for example, the MIT MANUS and its diversities,9,10 ARMin systems with multiple degrees of freedom for the upper limb training, 11 HWARD, 12 and HapticKnob 13 systems for hand rehabilitation. However, a difficulty in designing rehabilitation devices is to involve and maximize voluntary effort from the affected limb of a user during the training.6,7 Robot alone also cannot directly activate the desired muscle groups for a limb motion, although it can assist, or even can be dominant (eg, CPM), in the limb movement. Different from the external assistance provided by the robot with motors, a mechanism of the therapeutic effects of NMES is that a cyclic electrical stimulation on muscles would produce repetitive motosensory experiences. These experiences not only improve the paretic muscle force but also would either trigger one or all of the following pathways associated with neuroplasticity—functional reorganization, where intact areas assume function of damaged area, or activation of alternate descending pathways and structures in injured side.14,15 Poststroke rehabilitation assisted with NMES has been found to effectively prevent muscle atrophy and improve muscle strength. 15 NMES also limits the problem of “learned disuse” of stroke survivors by directly stimulating the target muscles. 8 However, it is difficult for NMES alone to control the movement trajectories 16 as in the robot-assisted training; and the early appearance of fatigue 17 has been commonly observed in NMES-assisted rehabilitation. It is mainly because of the reversed motor unit recruitment sequence during the artificial stimulation and the poor stimulation accuracy on the muscle fibers in comparison with the voluntary muscle contractions. 18 It has been found that the limb experience with desired kinematic accuracy contributed to motor relearning after stroke. 19 Therefore, as pointed out in the studies by McCabe et al 20 and del-Ama et al 21 for restoration of gait functions in respective stroke 20 and spinal cord injury 21 by a combination of robot and NMES, the difficulty was how to combine the advantages of the 2 unique techniques, while minimize their disadvantages; and then, to prove its effectiveness in the rehabilitation.
It is already known that involvement of voluntary efforts in stroke rehabilitation could achieve better motor outcomes than CPM3-5,22; however, less work has been done to combine the NMES and robot under the control of a user’s voluntary motor intention. Preliminary attempts have been carried out on poststroke gait training by using the NMES with robotic gait trainer together for lower limb rehabilitation by Tong and coworkers23,24 and McCabe et al, 20 these 2 studies only adopted the CPM mode for both robot and NMES, that is, there was no voluntary effort involved during the training. In the studies by Tong et al23,24 on gait training, there was no difference found when using robot alone and when using robot and NMES together, whereas the 2 training programs achieved better motor outcome than the conventional overground gait training. In the study by McCabe et al, 20 the feasibility of combining the implanted NMES with gait trainer was investigated; however, there is a lack of the report on the training effects and comparison with either technique. 20 In our previous study, a NMES robot driven by voluntary electromyography (EMG) was developed for wrist training on chronic stroke. 25 In this system, both NMES and robot were under the control of a user’s voluntary intention, represented by the EMG signals detected from the paretic limb. The NMES and the robot parts in the system can provide assistance to a paretic limb at the same time, that is, the robot assists the kinematic movement of the wrist; meanwhile the NMES stimulates the agonist muscles during the wrist movement. For stroke subjects without any learning experience, the movement accuracy of the wrist in a target tracking task could be immediately improved, and a reduction of the compensatory movement from the proximal was also observed when assisted with the NMES robot system. 25 To further investigate the long-term rehabilitation effects of the NMES robot–assisted training, we compared the poststroke training effects of EMG-driven robot and the combined training effects from the EMG-driven NMES robot on the wrist joint by a randomized clinical trial with a 3-month follow-up test, based on the developed EMG-driven NMES robot system in this work.
Methods
After obtaining approval from the Human Subjects Ethics Subcommittee of the university, we screened persons after stroke coming from local districts. Subjects recruited in this study satisfied the following inclusion criteria: (1) had unilateral ischemic brain injury or intracerebral hemorrhage at least 6 months after the onset of single stroke without other diagnosed neurological deficits; (2) had moderate level of motor impairment in the affected upper limb, assessed by Fugl-Meyer Assessment (FMA) 26 (9 < shoulder/elbow < 27; 6 < wrist/hand < 18); (3) had enough cognition to be able to follow the training protocol as assessed by the Mini-Metal State Examination (MMSE > 21) 27 ; and (4) had detectable EMG signals (3 times of the standard deviation above the baseline) from the flexor carpi radialis (FCR) and extensor carpi radialis (ECR). The study design was a single-blinded randomized controlled trial with a 3-month follow-up for comparing the motor improvements on the upper limb with the EMG-driven NMES robot (NMES robot group) and the EMG-driven robot (robot group). During the randomization, each subject was randomly assigned to a number of “1” (NMES robot group) or “2” (robot group) with equal probability of .5 (Figure 1). The study used a multiple baseline design, assessed by the FMA (shoulder/elbow and wrist/hand), Modified Ashworth Score (MAS, elbow and wrist), 28 and the action research arm test (ARAT). 29 Each outcome was measured 3 times in 2 weeks before the training as the baseline with a minimal 2 days of separation between the assessments. The clinical assessments were conducted by a blinded assessor.
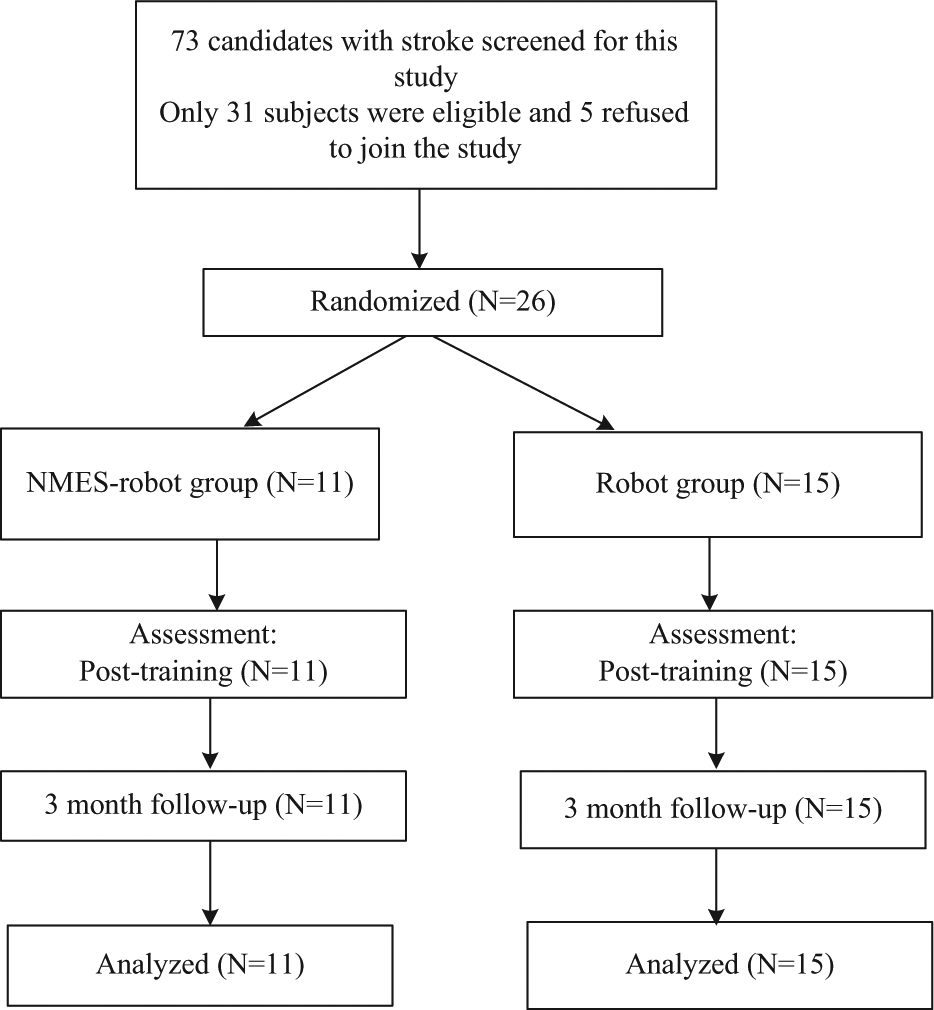
CONSORT (Consolidated Standards of Reporting Trials) flowchart of the training program.
Interventions
For both groups, each recruited subject received the wrist training with an intensity of 3 to 5 sessions/week for 20 sessions, finished within 7 consecutive weeks. The setup of the training for both groups is shown in Figure 2. In a training session, a subject was seated in front of a computer screen with the paretic arm attached on the robotic system, the shoulder abducted at around 80° and extended 0°, and the elbow flexed at 90°. The wrist joint was in line with the rotation center of the motor system, and the palm was mounted on a manipulandum which could rotate with the motor. The shoulder and the forearm were fixed by belts during the experiment. The subject was instructed to conduct tracking tasks by wrist flexion/extension when following a moving cursor on the screen with a constant angular velocity of 10°/s at the wrist joint, with a target to minimize the difference between the target and the actual wrist positions indicated by cursors as much as possible. The working principles of the EMG-driven robot and EMG-driven NMES robot have been described in details in our previous work.22,25,30 For the EMG-driven robot, a subject received the interactive assistance from the motor, where the torque provided to the wrist was proportional to the real-time voluntary EMG amplitude of the agonist muscles, that is, the EMG of FCR during the wrist flexion phase and the EMG of the ECR during the wrist extension phase. 30 It was also equivalent to the case of when robot gave 100% support, while 0% support from NMES (R100F0 25 ). In the scheme of R100F0, the maximum assistive torque a subject could obtain was the torque value during isometric maximum voluntary contractions in a training session, and usually would not exceed 8 Nm for chronic stroke subjects in both flexion/extension with the wrist joint angle at 0°.22,25,30 For the NMES robot group, the subject received the interactive assistance from both the motor and the NMES parts at the same time during the tracking. In the case when robot gave 50% support, and NMES provided 50% assistance (R50F50), the assistance from the motor was the half value as for the robot group; The assistance from the NMES was electrical stimulation on the agonist muscle with the intensity proportional to the voluntary EMG amplitude of the muscle 25 ; The maximum assistance from the NMES was the half value of the threshold to evoke maximal wrist flexion and extension when the forearm was put horizontally on a table with the wrist joint starts at its neutral position. 25 The assistive schemes of R100F0 and R50F50 could help chronic stroke subjects who did not have experiences of using the system achieve comparable and better tracking accuracies with a coverage of most range of motion of the wrist during the tracking than the other assistive combinations between the robot and the NMES. 25 Therefore, we selected the 2 assistive schemes in this work to compare their rehabilitation effects. For the EMG-driven robot–assisted training, the NMES part was turned off. In a training session, the subject was first required to conduct maximum isometric voluntary flexion and maximum isometric voluntary extension at 0° of the wrist angle with a repetition of 3 times before the training. Each maximum isometric contraction lasted for 5 seconds. Between 2 consecutive contractions, there was a break of 5 minutes for rest to avoid muscle fatigue. After that, the subject was instructed to conduct the wrist tracking tasks. In each session, there were totally 14 trials, and each trial contained 5 cycles of wrist extension and flexion. In the first and the last tracking trials, there was neither interactive assistance nor resistance from the training device for both groups, and they were used for evaluation. Between 2 consecutive trials, there was a break of 2 minutes for rest in each session.
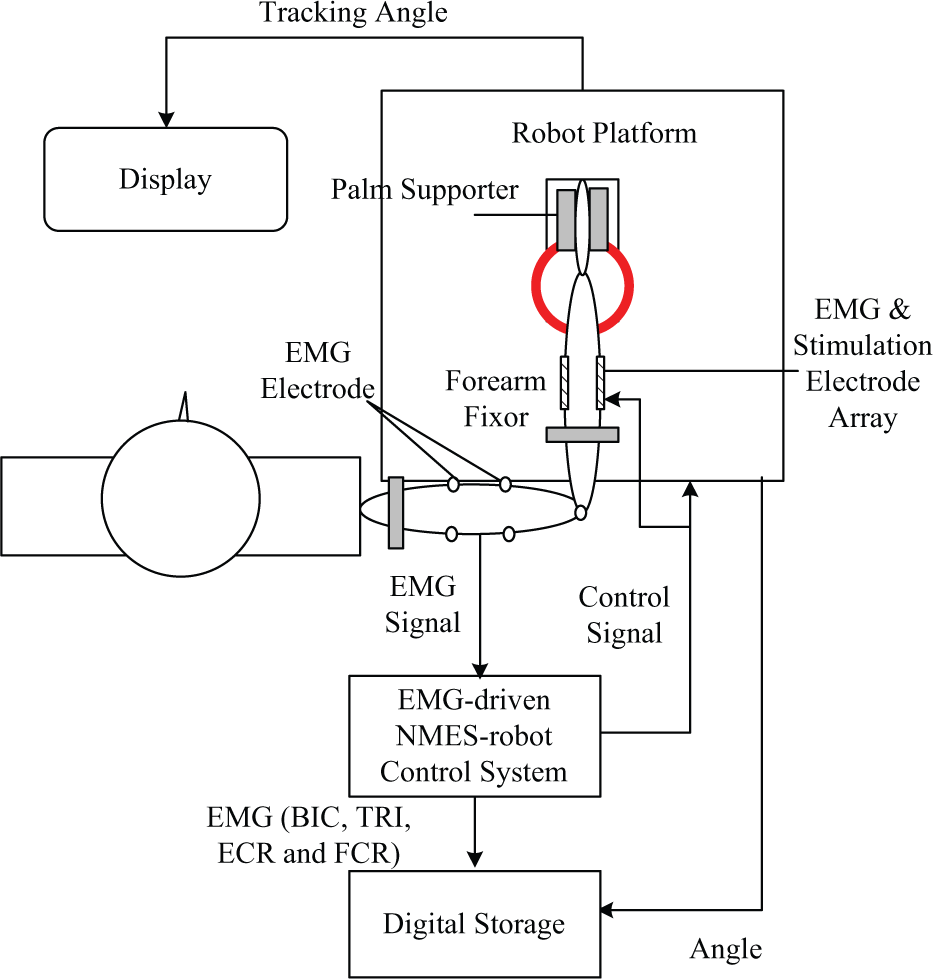
The experimental setup for the electromyography (EMG)-driven robot-assisted and EMG-driven neuromuscular electrical stimulation (NMES) robot–assisted wrist training.
Evaluation on the Training Effects
Clinical Scores
The clinical scores of FMA (the shoulder/elbow part ranging from 0 to 42, and the wrist/hand part ranging from 0 to 24), ARAT, and MAS in the pretraining assessments were applied for comparison with those immediately after the training and in the 3-month follow-up.
Electromyography Parameters
For both groups, EMG signals were recorded from the biceps brachii (BIC), triceps brachii (TRI, lateral head), FCR, and ECR muscles during the evaluation tracking trials in each session. The coactivation patterns among muscle pairs during the tracking were studied by the co-contraction index (
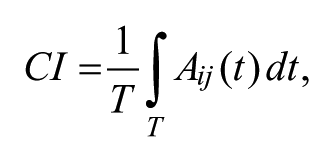
where
Statistical Analysis
Power calculations to determine subject numbers in each group were assessed according to the expected change in the total FMA score after the training, based on the method introduced in the work by Khedr et al. 32 The reason for the selection of FMA to estimate the subject number was that the score was a widely used and effective indicator for monitoring the voluntary motor improvement in poststroke rehabilitation. 33 In our previous work on EMG-driven pure robot-assisted wrist training on chronic stroke, 30 the mean expected improvement in FMA of the upper extremity was around 8 points after the training with a standard deviation of 6 points. A clinically significant difference in FMA between groups was estimated to be 7 points in the mean in the work by Boyaci et al 34 on NMES-assisted stroke rehabilitation with a similar group sample size (around 10 subjects in a group). A power calculation with P=0.05 and 80% power suggest that it would require at least 9 subjects per group to detect whether such a difference was produced between the robot and the NMES robot groups.
The analyses of variance (ANOVAs) were used to investigate the effects from the group difference and that across the training sessions (or different time points of the clinical assessments). Two-way ANOVA was first used to tell the difference with respect to the 2 factors, that is, the group and the training/assessment session. Then, 1-way ANOVA was used to investigate the variation across the different sessions with Bonferroni correction in the post hoc tests on the multiple means. Independent
Results
During the subject recruitment, a total of 73 subjects were screened for the training (Figure 1). Twenty-six of them met the selection criteria and were recruited. The demographic data of the subject after the randomization are shown in Table 1. The minimum time after stroke was 8 months and the maximum was 12 years at the time of recruitment.
Demographic Information of the Subjects After the Randomization.
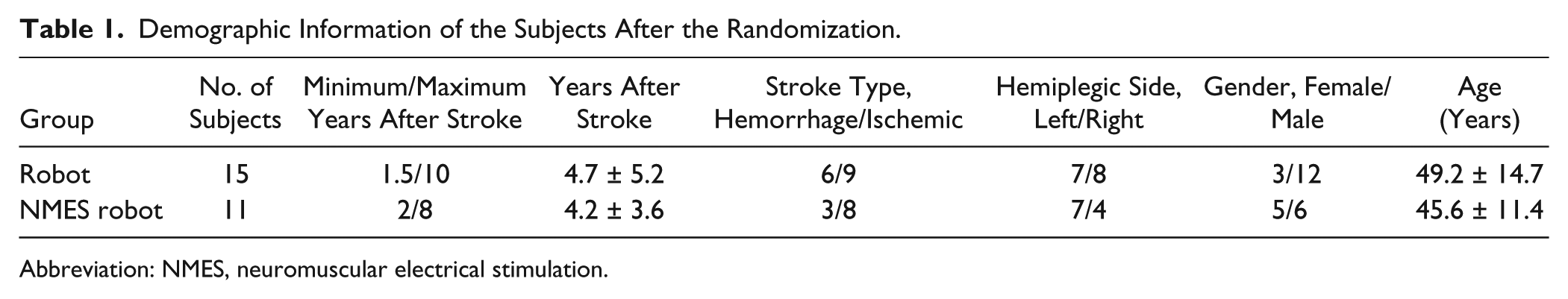
Abbreviation: NMES, neuromuscular electrical stimulation.
Scores of Clinical Assessments
Figure 3 shows the clinical scores of FMA, MAS, and ARAT in the 3 pretraining baseline tests, in the posttraining assessment, and in the 3 months follow-up test for the 2 groups. Table 2 lists the means and the standard deviations for each clinical outcome in Figure 3, together with the ANOVA results on the intergroup and intragroup differences for each outcome. There was no significant intra- or intergroup difference observed in the baseline tests for all clinical scores. Significant improvements in the FMA shoulder/elbow after the training were obtained in the two groups, and the improvements could be maintained for 3 months later (
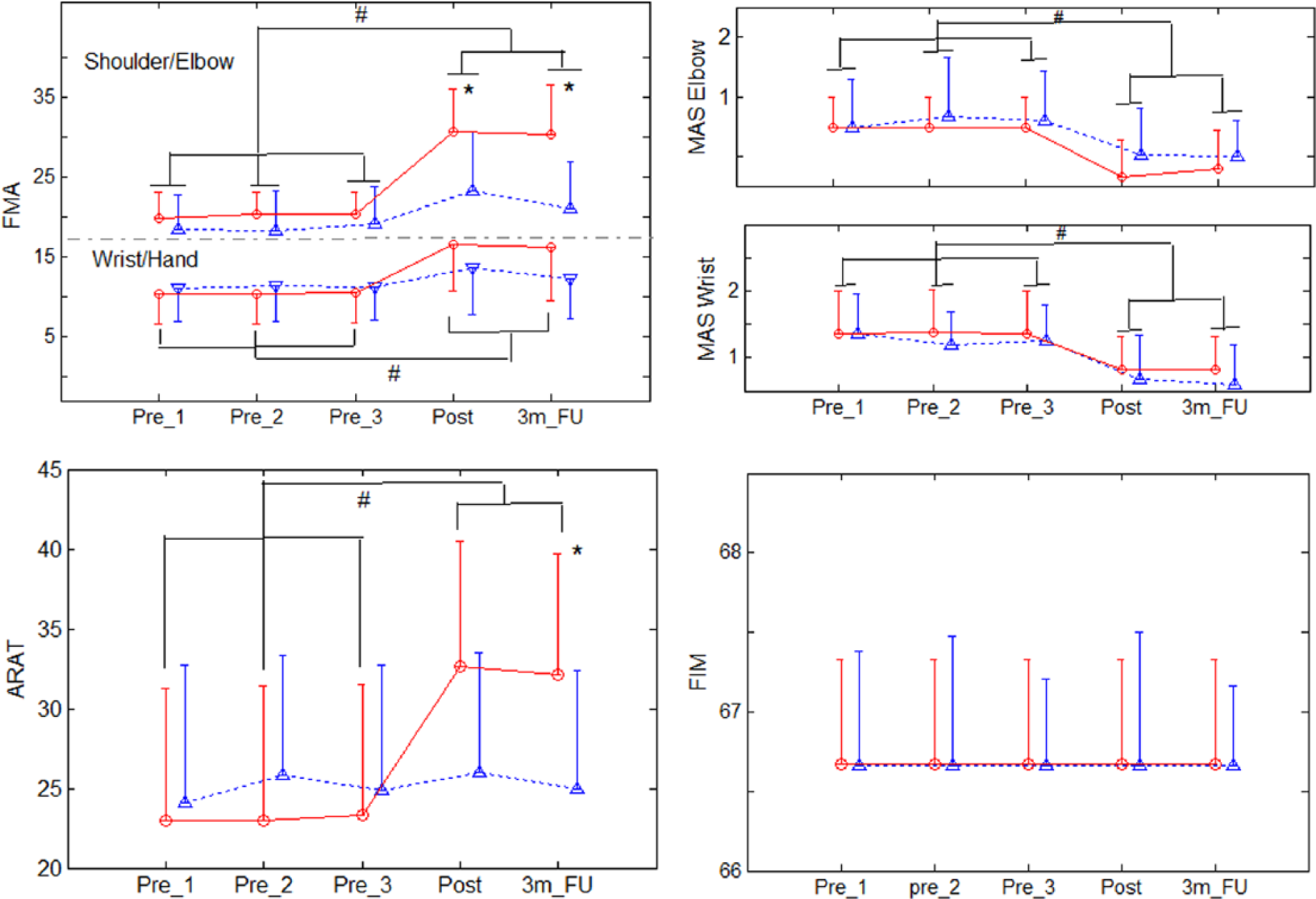
The variation of the clinical scores measured before and after the training, as well as after 3 months. The solid lines with circles are for the neuromuscular electrical stimulation (NMES) robot group, and the dotted lines with deltas are for the robot group. The significant intergroup difference is indicated by “*”(
The Means and Standard Deviations for Each Measurement of the Clinical Assessments. a
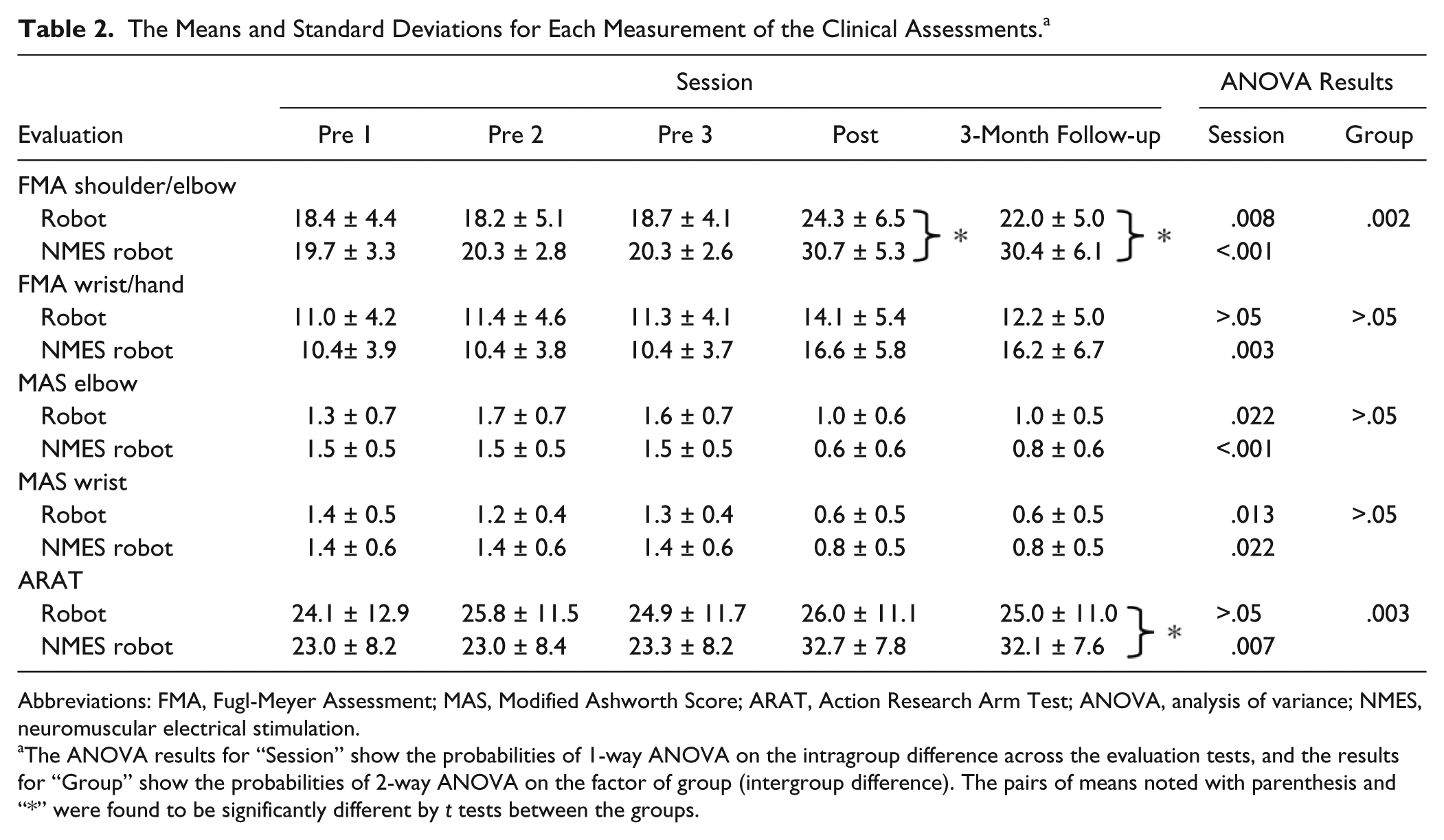
Abbreviations: FMA, Fugl-Meyer Assessment; MAS, Modified Ashworth Score; ARAT, Action Research Arm Test; ANOVA, analysis of variance; NMES, neuromuscular electrical stimulation.
The ANOVA results for “Session” show the probabilities of 1-way ANOVA on the intragroup difference across the evaluation tests, and the results for “Group” show the probabilities of 2-way ANOVA on the factor of group (intergroup difference). The pairs of means noted with parenthesis and “*” were found to be significantly different by
Electromyography Parameters
Electromyography parameters mainly reveal the muscle activation patterns during the training. Figure 4 shows the comparison of the
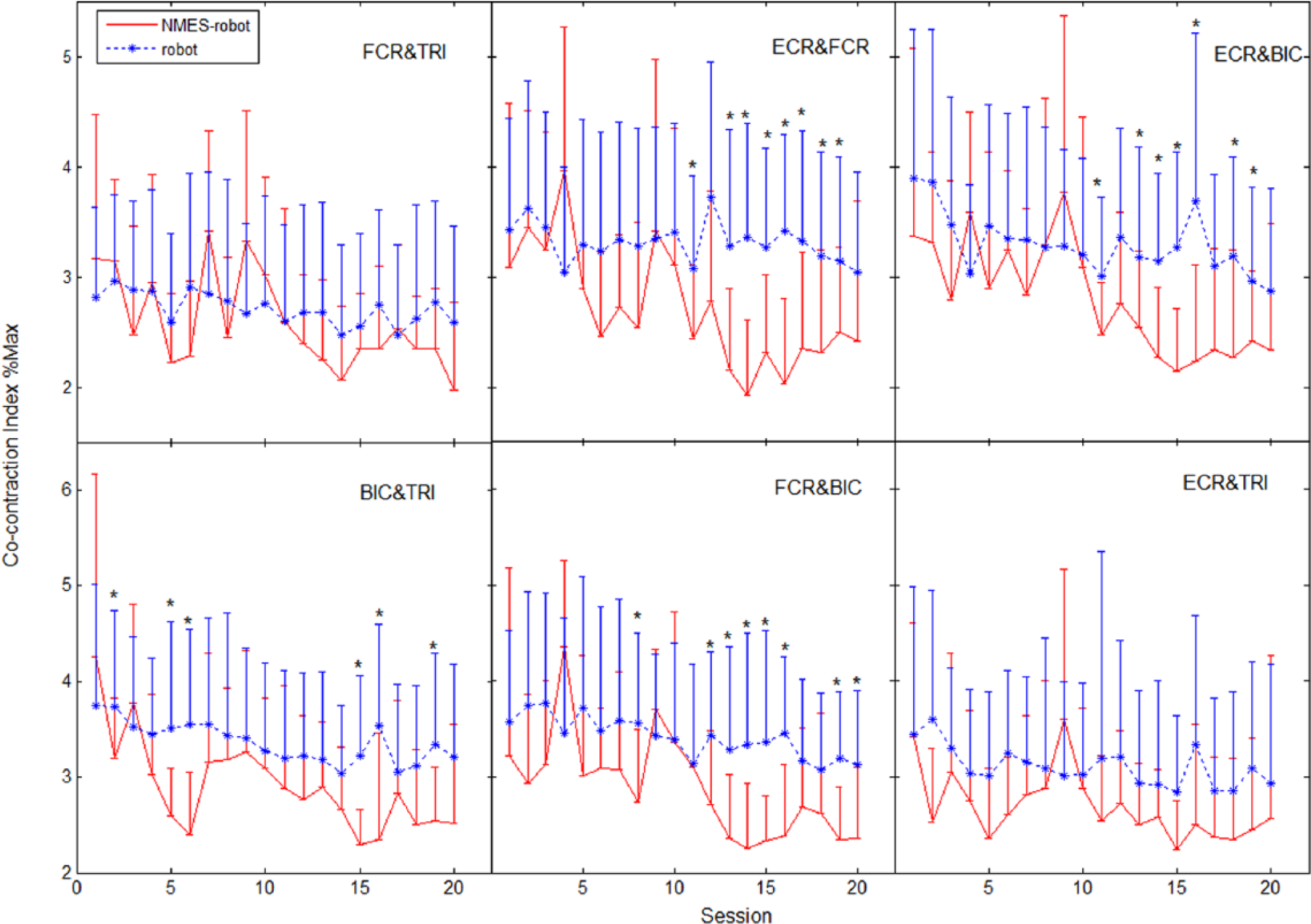
The variation in co-contraction indexes. The co-contraction indexes across the sessions are represented by mean and standard deviation of the muscle pairs, FCR and TRI, ECR and FCR, ECR and BIC, BIC and TRI, FCR and BIC, and ECR and TRI. The intergroup difference is indicated by “*” on top of the error bar (
Discussion
Motor Outcomes Evaluated by Clinical Scores
The FMA results in Figure 3 suggested that after the EMG-driven NMES robot–assisted wrist training, the subjects obtained significant motor improvements on both the shoulder/elbow and wrist/hand functions; and these improvements could be maintained after 3 months. However, after the EMG-driven robot–assisted training, the subjects only gained significant motor improvement on the shoulder/elbow part, which also could be kept for 3 months. No significant motor improvement achieved in the wrist/hand after the EMG-driven robot training. The NMES robot group achieved better improvements in FMA than the robot group. In comparison with the rehabilitation of the proximal joints, motor recovery of the distal joints after stroke is more difficult. It is common to observe in the traditional rehabilitation that reasonable recovery could be obtained at the proximal joints (eg, the shoulder and elbow); however, the recovery in the distal (eg, the wrist and fingers) was poor. In this work, we also observed that although the direct physical training was applied on the wrist joint and the upper arm was fixed to be still during the training, the better improvement was obtained at the proximal (ie, shoulder/elbow part), especially for the robot group. Better improvements in the proximal joints when practiced in the distal in robot assisted training have been observed in our previous studies22,30,35 and others.36,37 The possible reasons could be the compensatory muscular activities from the proximal when the subjects performing distal limb tasks and the competitive interaction between the proximal and the distal joints. 38 For chronic stroke, most of them have better motor function in the proximal than the distal of the upper limb mainly because of more intensive training on the proximal joints than the distal in early stage after stroke. They are used to exert more muscular effort in their shoulder and elbow when required to conduct wrist or hand movements, which would develop new compensatory muscular activation patterns differed from the unimpaired.5,14 When the muscles related to both the proximal and the distal joints practiced in the training, competitions in the motor recovery would occur, for example, more the practice, the better the recovery. Therefore, in some studies related to distal joint rehabilitation, temporary paralysis of the muscles of proximal joints (eg, botulinum toxin injection) was applied to minimize the competition between the proximal and the distal 38 . In this work, it was found that although we did not paralyze the muscles of the proximal joints, for the NMES robot group, the significant motor improvements in the distal joints were also achieved, together with the improvements at the proximal. It implied that the introduction of NMES into robot-assisted training in this work could facilitate better motor coordination between the proximal and the distal during the rehabilitation, which was related to the improved muscle coordination in the elbow and the wrist to be discussed later by the EMG parameters. The significant increases in ARAT for the NMES robot group suggested that the treatment improved the distal finger functions, since the 16 out of 19 items in the ARAT assessment are related to finger tasks. However, no improvement in ARAT was obtained for the robot group. In this work, the selection of the training intensity was set to be slightly flexible from 3 to 5 sessions/week in a 20-session program. It was based on the balance between the availability of the training system and the commitment to the training program, which could be achieved by the subjects from the local districts. This arrangement has been applied in our previous studies on chronic stroke.22,30,35 There was no clear correlation between the number of training sessions/week and the motor outcome after the training.
The MAS scores in Figure 3 suggested that both EMG-driven robot and NMES robot assisted training could release the muscle spasticity at the wrist and the elbow joints effectively, and the effect could be maintained till 3 months later. It possibly suggested that the release of muscle spasticity was mainly because of the robot-assisted training, while the additional practice with NMES contributed little to the reduction of muscle tone. The results in Figure 3 also indicated that NMES robot–assisted wrist training was more effective in improving the voluntary motor function of the whole upper limb, especially in the distal joints.
Motor Improvement Evaluated by Electromyography
For the EMG parameter measured by
The Selection of Assistive Schemes
In general, the control schemes used in both EMG-driven robot and EMG-driven NMES robot were to facilitate the subjects to learn the control of generating voluntary residual EMG signals in association with the output of joint torques/muscle forces as the rewards in the biofeedback. The larger EMG could generate a larger torque/muscle force, which is similar to the musculoskeletal control in the unimpaired and easy to be understood during the motor relearning in poststroke rehabilitation. The control schemes used in both EMG-driven robot (R100F0) and EMG-driven NMES robot (R50F50) in this work also could help the stroke subject achieve the optimal tracking accuracies in comparison with other assistive combinations between the robot and the NMES in the designed system. 25 Under these 2 schemes, the subjects also needed to use their voluntary effort (ie, the detectable EMG) continuously to acquire assistance, with the purpose of encouraging them actively to participate in the training. Furthermore, the subjects recruited in the study were able to generate EMG signals from the target muscles, although the efforts might not be related to a correct limb movement. In this work, we only compared the training effects between the EMG-driven robot–assisted and NMES robot–assisted rehabilitation; however, we did not include the EMG-driven NMES-assisted training. The main reason is that the tracking performance by stroke subjects with EMG-driven NMES was poor when compared with those for the EMG-driven robot and NMES robot. 25 When only assisted with NMES, most of the subjects could not follow the moving target on the screen with large root mean square errors between the target and the actual wrist positions and lowered range of motions in the tracking tasks. 25 The rehabilitation effects would be affected by the self-perceived low performance by the subjects during the training, 19 if they always could not achieve the tasks as required. Therefore, in this work we only select the assistive configurations (ie, R100F0 and R50F50) with the optimal and comparable tracking accuracies. It was also noticed that in some early reports on robotic gait trainer in lower limb rehabilitation after stroke,23,41 no significant advantage was observed after introducing additional NMES into the robot, when compared with the pure robotic training. One of the main reasons was that the control in their system was only continuous passive motions,23,41 and there was no voluntary effort required from stroke subjects. However, in this work, the subjects needed to exert voluntary EMG to obtain the proportional assistances from the training devices continuously. Intensive and repeated limb practice with voluntary effort has been identified to be necessary for effective rehabilitation after stroke.6,7
In this study, we introduced a combined rehabilitation method for poststroke wrist training by integrating NMES and robot together under the voluntary motor intention control by EMG. We compared the training effects of the EMG-driven NMES robot with those by the EMG-driven pure robot on chronic stroke. The combined treatment with NMES and robot demonstrated better improvements in upper limb functions, especially in the wrist/finger functions, in comparison with the pure robot assisted training. NMES could facilitate the additional neuromuscular effort and attention on the target muscles, which resulted in a faster release in muscle co-contractions than the pure robot group between the wrist muscle pair of ECR and FCR, and between the muscles in neighboring joints (the wrist and the elbow), that is, ECR and BIC and FCR and BIC pairs. The separation in the muscle activities between the wrist and the elbow, as well as the improved distal muscle coordination (ECR and FCR), also led to a better motor functional recovery in the whole upper limb than the pure robot training, that is, the shoulder/elbow and the wrist/hand functions measured by the clinical scores. The setup and training time for the NMES robot training is same as the robot training. There is a potential to apply the NMES robot in the clinical setting in the future to facilitate better motor recovery in the upper limb function.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was financially supported by a GRF grant (PolyU 5318/09E) from the Research Grants Council and an ITF grant (ITS/033/12) from the Innovation and Technology Commission of the Hong Kong Special Administrative Region.
