Abstract
Keywords
Introduction
Perinatal stroke is the leading cause of unilateral motor deficits in children. These early brain injuries damage the motor system, resulting in hemiparetic cerebral palsy (HCP) and lifelong disability. 1 Clinically, children with HCP may have comorbidities in sensory function, cognitive and behavioral difficulties, and epilepsy, complicating their physical disabilities. 2 Motor impairments last for decades and affect diverse aspects of function and quality of life for children and families. As focal, unilateral injuries of defined timing in an otherwise healthy brain, perinatal stroke is an ideal model of human developmental plasticity.3-6
Intensive upper extremity therapies have been shown to improve function in children with HCP in multiple randomized controlled trials (RCTs) and are recommended to improve function in a systematic review. 7 Two forms of intensive therapy have been developed based on motor learning principles. Unimanual intensive therapy focuses on mass practice of the affected hand while the less-affected hand is constrained (ie, constraint-induced movement therapy [CIMT]). 8 Bimanual intensive therapy focuses on enhancing bimanual coordination (i.e., hand-arm bimanual intensive therapy [HABIT]). 9 Both forms of therapy can produce long-term improvements in hand function in children with HCP10,11 though most studies have not been etiology specific.
Noninvasive neuromodulation, such as repetitive transcranial magnetic stimulation (rTMS) or transcranial direct current stimulation (tDCS) may enhance such therapies.12-14 We reported additive benefits of rTMS with CIMT when combined with intensive manual therapy in a large clinical trial. 15 Such therapies, however, are costly and time consuming (90-100 hours over 2-3 weeks). Additional evidence has shown that tDCS can enhance motor learning in children 16 and early clinical trials in HCP suggest possible efficacy.13,14 To improve such approaches, it is imperative to better understand the neurophysiological mechanisms that underlie positive responses. Investigating such biomarkers of functional improvement and neuroplasticity may advance individualized medicine for disabled children.
Probing motor cortex neurophysiology using transcranial magnetic stimulation (TMS) is an established method of examining such neuroplasticity in children.5,17 For example, children with a preserved ipsilateral corticospinal tract (CST) may have distinctive neurophysiology in the contralesional motor cortex (M1) compared to those without. 18 Similar approaches may also inform mechanisms of interventional neuroplasticity. In a study of 16 young people with HCP, Juenger et al 19 reported that TMS metrics of baseline ipsilateral CST status were associated with both clinical and TMS cortical excitability changes following intensive interventions. In an RCT, Friel et al 20 showed that changes in motor map size and motor-evoked potentials (MEPs) amplitudes differed depending on the type of therapy administered. No studies have yet explored the TMS neurophysiological changes associated with intensive therapy combined with neuromodulation in children with HCP.
Here, we aimed to investigate M1 neurophysiological changes associated with CIMT and/or brain stimulation within an intensive clinical trial. We hypothesized that changes in short-interval intracortical inhibition (SICI) would be associated with clinical responses and the type of therapy received.18,21 We also sought to examine potential relationships between bilateral TMS neurophysiology metrics and clinical outcomes.
Methods
This study analyzed TMS neurophysiological data obtained prospectively during a randomized, controlled, factorial clinical trial of rTMS and constraint therapy in children with perinatal stroke and HCP (PLASTIC CHAMPS, clinicaltrials.gov/NCT01189058). Complete methods and clinical results of the trial are described elsewhere. 15 Methods were approved by the institutional research ethics board.
Participants
Participants were recruited via the Alberta Perinatal Stroke Project, a population-based research cohort. 22 Inclusion criteria were the following: (1) symptomatic hemiparetic cerebral palsy (HCP), including Pediatric Stroke Outcome Measure >0.5 and Manual Ability Classification Scale (MACS) levels I to IV and both parent and child perceived functional limitations, (2) magnetic resonance imaging–confirmed unilateral perinatal arterial ischemic stroke in the middle cerebral artery (AIS-MCA) or periventricular venous infarction (PVI), (3) age 6 to 19 years, (4) term birth, and (5) written informed consent/assent. Exclusion criteria were the following: (1) additional neurological abnormality, (2) multifocal stroke, (3) severe hemiparesis (MACS V) or predominant dystonia, (4) developmental delay precluding compliance, (5) epilepsy, (6) TMS contraindications, or (7) CIMT within 6 months, upper extremity surgery or Botox within 12 months.
Interventions
Briefly, participants were engaged in goal-directed, age-appropriate, peer-supported intensive upper extremity training for a total dose of 80 hours over 10 weekdays (Figure 1A). Participants were randomized equally to receive rTMS or sham, and again to CIMT or not. Hence participants were randomized into four groups: rTMS, CIMT, both, or neither. Brain stimulation occurred at the Alberta Children’s Hospital Pediatric Non-invasive Neuromodulation Laboratory immediately before daily therapy. Using neuronavigation (Brainsight2, Rogue Research, Montreal, Canada), an air-cooled rTMS coil (70 mm, Airfilm, Magstim, Whitland, UK) was placed tangentially at 45 degrees over the contralesional M1 “hotspot” (details below). Parameters for rTMS were: intensity 90% resting motor threshold, frequency 1Hz for 20 minutes. Sham participants underwent identical procedures but the coil only produced comparable sensations without stimulation. CIMT methods were based on best available evidence. 15 Participants wore a cast for 90% of the waking hours during 10 days of intervention.
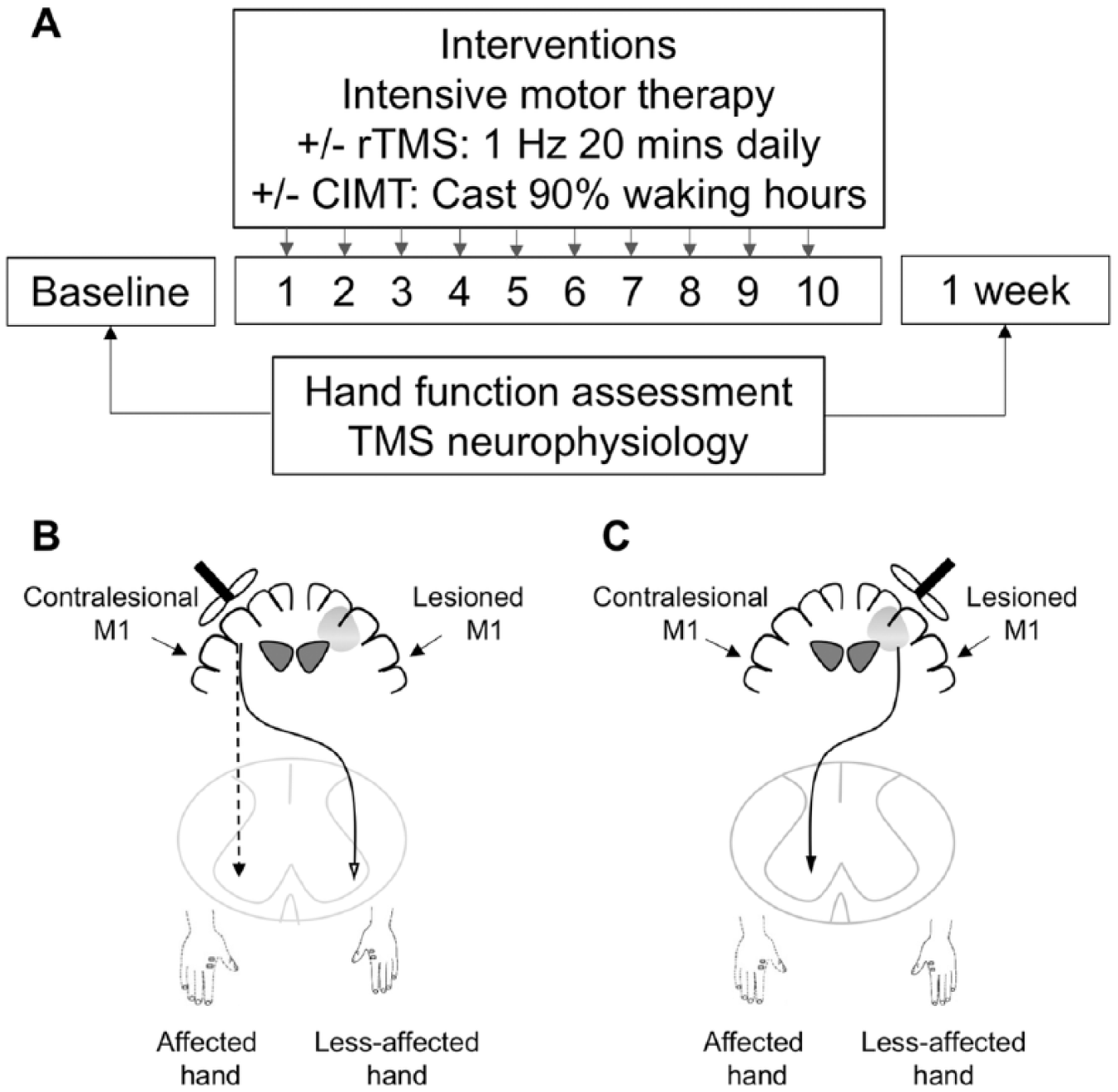
(A). Study design. Transcranial magnetic stimulation (TMS) neurophysiology was measured at baseline and within one-week after intervention. (B). Gray circle indicates lesion area. Single-pulse TMS was used to evaluate the contralesional motor cortex neurophysiology. Both the ipsilateral corticospinal pathway controlling the affected hand (dashed line) and the contralateral corticospinal pathway controlling the less-affected hand (solid line, cross over) are measured. (C). Single-pulse TMS was used to evaluate the lesioned motor cortex neurophysiology.
Neurophysiology Measures
TMS neurophysiology measurements were conducted within 2 weeks before starting (baseline) and 1 week after completing the intervention. Participants sat comfortably in a chair and watched a movie during times not requiring cooperation. A pair of Ag-AgCl electrodes (Kendall, Chicopee, MA) were used to record surface electromyogram (EMG) from bilateral first dorsal interosseous (FDI) muscles with a wrist ground band. EMG signals were amplified by 1000× and band-pass filtered from 20 to 2500 Hz and then digitized at a rate of 5000 Hz using CED 1401 hardware and Signal 6.0 software (Cambridge Electronic Design Limited, UK).
Single-Pulse TMS: Corticospinal Pathway Excitability
To assess corticospinal tract properties, we measured MEPs in the FDI muscles bilaterally with single-pulse TMS. A figure-of-eight coil (70 mm) connected to a Bistim stimulator (Magstim) induced posterior-anterior currents over M1 while recording FDI bilaterally. The M1 hotspot for the less-affected hand was used for all subsequent contralesional hemisphere stimulations (Figure 1B). Baseline neurophysiology of the contralesional hemisphere is described in detail elsewhere. 18 This location was landmarked for consistent coil placement and orientation on 3-dimensional anatomical T1 magnetic resonance imaging using neuronavigation (Brainsight2). The same procedures were carried out for the lesioned M1 where the FDI of the affected hand was targeted (Figure 1C). Resting motor threshold (RMT) was determined based on published standards as the minimum stimulator intensity (percentage of maximum stimulator output, %MSO) required to elicit FDI MEP >50 µV in 5/10 consecutive trials. Active motor threshold (AMT) was determined as the minimum stimulator intensity needed to elicit MEP >200 µV in 5/10 consecutive trials during 20% maximal voluntary contraction (MVC) of the FDI. Stimulus recruitment curves (SRCs) were then generated bilaterally according to published methodology23,24 at stimulation intensities of 10% RMT increments from 100% to 150% RMT (10 pulses/intensity in random order). The relative presence of the ipsilateral corticospinal tract (CST) was determined using previously published criteria. 18 When focusing on the contralesional M1, participants were classified as ipsilateral (IP) when stimulation of the contralesional M1 at 120% RMT resulted in >50 µV MEP in the affected hand in at least 3 out of 10 trials. Otherwise, children were characterized as nonipsilateral (NI). Additional classification of corticospinal tract organization derived from TMS measures of both hemispheres can be found in Supplementary Table 1 (available in the online version of the article).
Paired-Pulse Paradigm: Intracortical Inhibition and Facilitation
Two stimulators (Bistim and 200 Magstim) connected by an adaptor were used to conduct a standardized paired-pulse paradigm designed to examine intracortical M1 neurophysiology. This paradigm was performed bilaterally. Protocols evaluating short-interval intracortical inhibition (SICI) and intracortical facilitation (ICF) were in accordance with published evidence.25-28 Suprathreshold test stimuli (TS) at 120% RMT were preceded by subthreshold conditioning stimuli (CS) at 90% AMT. The CS and TS were separated by interstimulus intervals (ISIs) of 2 ms for SICI and 10 ms for ICF. Thirty pulses were delivered in a random order: 10 unconditioned (test-alone), 10 at 2 ms ISI, and 10 at 10 ms ISI. The average amplitude of the 10 conditioned MEP at both ISI was expressed as a ratio of the average of the 10 unconditioned test MEP (<1 suggesting inhibition, >1 suggesting facilitation).
TMS Data Analysis
EMG data were imported into MATLAB (Mathworks, Inc., Natick, MA) for blinded offline analysis. MEP files were visually inspected for artifacts (eg, facilitated trials with excessive baseline activity). Customized MATLAB scripts calculated peak-to-peak MEP amplitudes from unrectified EMG by setting a time window of 15 to 60 ms from the TMS pulse. SigmaPlot version 13 (Systat Software Inc, San Jose, CA) was used to fit individual SRC and plot box and whisker plots. GraphPad version 7.0b (La Jolla, CA) was used to plot group SRC.
Clinical Outcomes
Evidence-based motor outcomes were obtained in standardized procedures by certified pediatric occupational therapists blinded to all other data, including intervention type. Assessments were completed in a quiet environment and videotaped for validation. The Assisting Hand Assessment (AHA) was used to measure how well hemiparetic children use their affected hand in various bimanual activities in play. 29 The AHA carries the best evidence of inter- and intrarater reliability, test validity, and response sensitivity to detect change in upper extremity function in children with HCP. 30 The Canadian Occupational Performance Measure (COPM) was used to measure subjective impressions of performance and satisfaction of functional goal achievements. 31 Goals were established at baseline under the guidance of an occupational therapist and the family. The AHA and COPM clinical outcomes were assessed at baseline and again within 1 week following intervention.
Statistical Analysis
Analyses were performed using SPSS version 24 (IBM Corp, Armonk, NY). Neurophysiological variables were examined for normality using Shapiro-Wilk test. Paired
Results
Population
Participant characteristics, including demographics and baseline measures, are listed in Table 1. Of the 45 participants, 23 (51%) were classified as ipsilateral and 10 were nonipsilateral (22%). Ipsilateral status could not be classified in the remaining 12 participants (27%) where 5 did not have MEP responses at maximal stimulator output and 7 had high RMT (>82%) that precluded the ability to deliver suprathreshold stimuli (120% RMT). Only 25 participants (56%) had a measurable RMT in the affected hand when stimulating the lesioned M1. Details of sample size, corticospinal tract organization, and treatment group allocation for each experiment protocol can be found in Supplementary Table 1.
Demographic Characteristics, Baseline Neurophysiology, and Clinical Outcome.
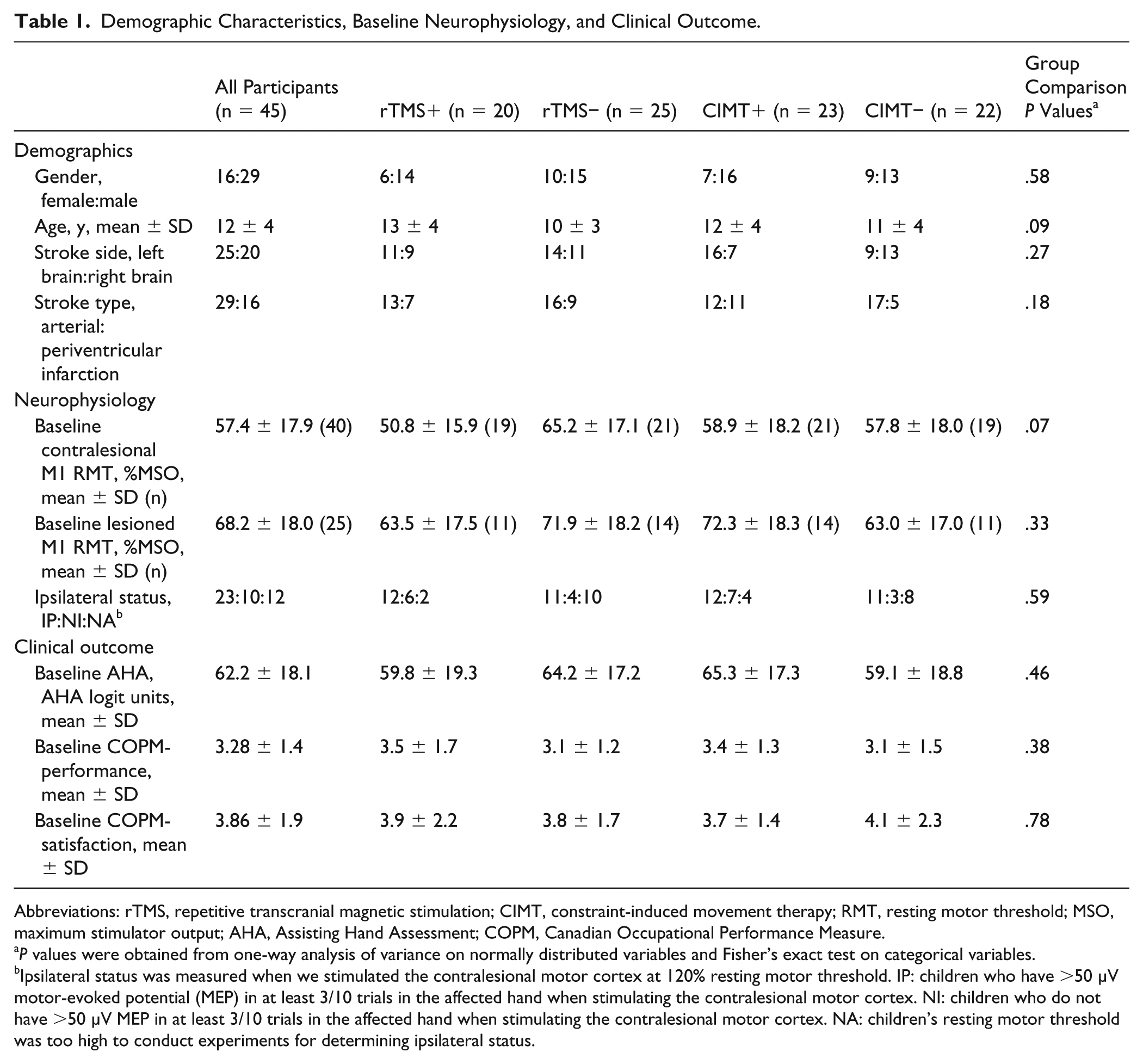
Abbreviations: rTMS, repetitive transcranial magnetic stimulation; CIMT, constraint-induced movement therapy; RMT, resting motor threshold; MSO, maximum stimulator output; AHA, Assisting Hand Assessment; COPM, Canadian Occupational Performance Measure.
Ipsilateral status was measured when we stimulated the contralesional motor cortex at 120% resting motor threshold. IP: children who have >50 µV motor-evoked potential (MEP) in at least 3/10 trials in the affected hand when stimulating the contralesional motor cortex. NI: children who do not have >50 µV MEP in at least 3/10 trials in the affected hand when stimulating the contralesional motor cortex. NA: children’s resting motor threshold was too high to conduct experiments for determining ipsilateral status.
Cortical Excitability Changes
MEP Amplitude: Contralesional M1
A complete dataset of 20 single-pulse suprathreshold (120% RMT) stimulations of the contralesional M1 for both pre and post sessions was available for 16 participants when MEP was measured in the less-affected hand; and a complete dataset was available for 14 participants when MEP was measured in the affected hand. An increase in excitability of the contralesional M1 after intervention was demonstrated as MEP sizes in the less-affected hand increased (Figure 2A,
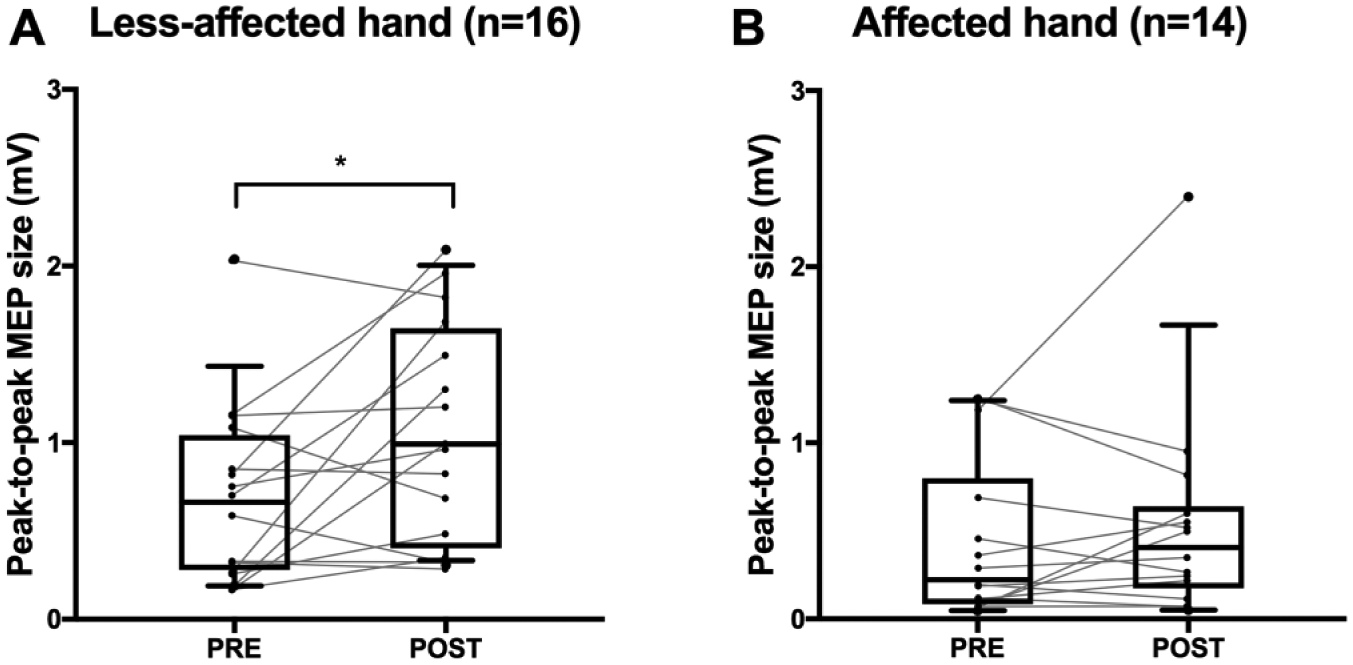
Cortical excitability of contralesional primary motor cortex (M1). For all figures, box edges represent 25th and 75th percentile; the solid line inside the box represents median; whiskers represent 10th and 90th percentiles. (A). Peak-to-peak motor-evoked potential (MEP) size measured in the less-affected hand from stimulating the contralesional M1 increased following intervention (n = 16). (B). Peak-to-peak MEP size measured in the affected hand from stimulating the contralesional M1 did not change (n = 14).
Similarly, MEP sizes in the less-affected hand appeared to increase as observed by a significant main effect when either CIMT+/− or rTMS+/− was used as a between-factor in the RM ANOVA (
There was an insufficient number of participants (n = 7) with complete MEP data from the lesioned M1 to compare pre-post changes.
SRC: Contralesional M1
Twenty-five participants had complete SRC data from the contralesional M1 to the less-affected hand; 19 participants had complete dataset of SRC experiment from the contralesional M1′ to the affected hand. A typical, sigmoidal-shaped stimulus response curve was fitted for group data where an upward trend was observed in the SRC across stimulation intensities for the less-affected, but not the affected hand (Figure 3A and B).
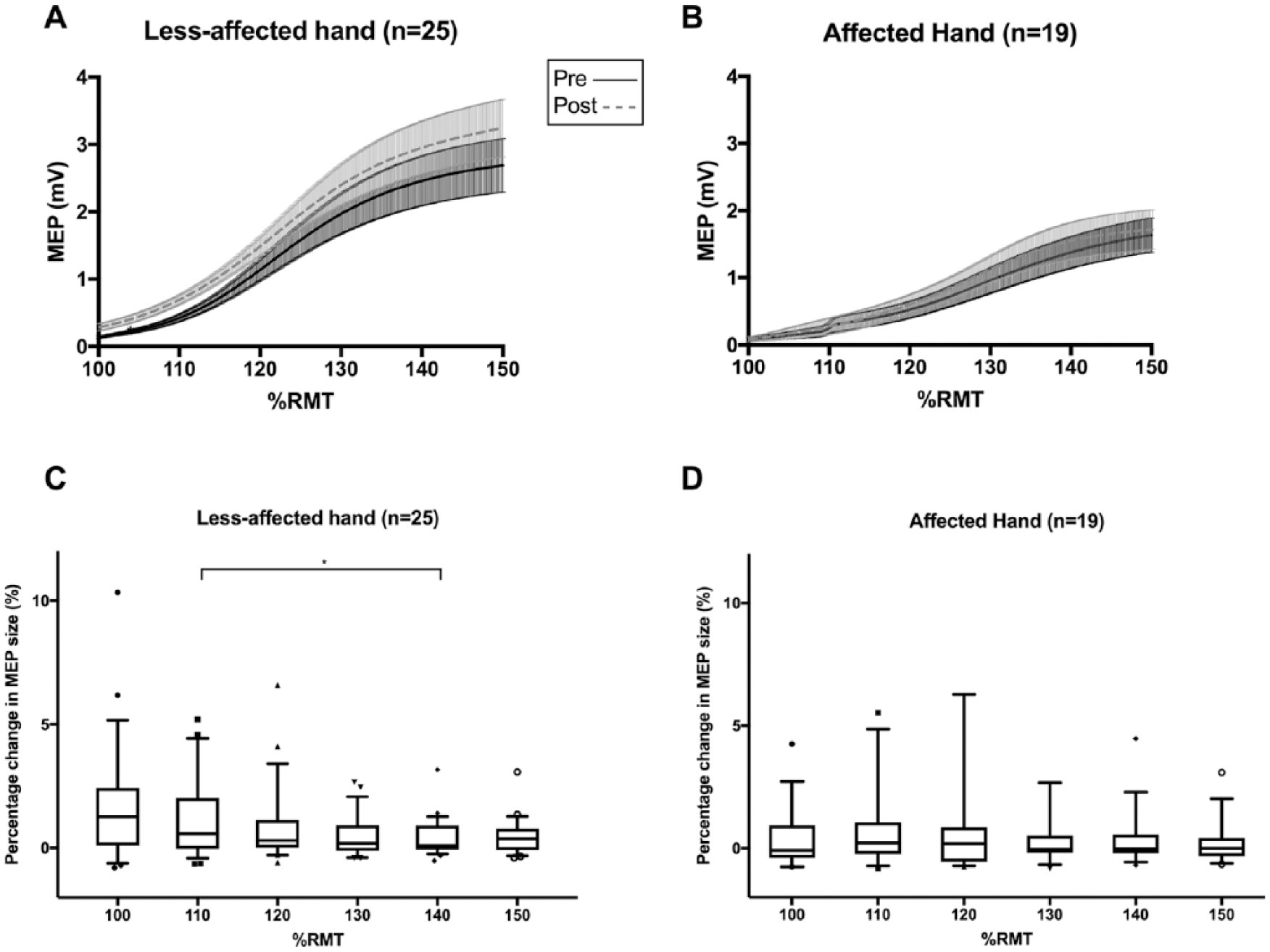
Averaged stimulus recruitment curves from the contralesional primary motor cortex (M1) (A, B). Solid line represents averaged curve values at baseline; dashed line represents averaged curve values at post-intervention. Shaded areas show ranges of standard errors of means (SEM). (C, D). Box edges represent 25th and 75th percentile; the solid line inside the box represents median; whiskers represent 10th and 90th percentiles.
Stimulation intensity in the SRC impacted the percentage change observed in the less-affected hand MEP from the contralesional M1 when either CIMT+/− or rTMS+/− was used as a between-factor in the RM ANOVA (Figure 3C, CIMT+/−,
Similarly, stimulation intensity appeared to influence the percentage change in the affected hand MEP size from the contralesional M1′ when we used CIMT+/− as a between-factor in the RM ANOVA (Figure 3D,
We then asked whether the effect of intensity on the percentage change would differ between the treatment groups by examining the interaction effects from the previous ANOVAs. There were no significant interaction effects from all RM ANOVAs, suggesting the effect of intensity on the MEP percentage change did not differ between groups (
Intracortical Neurophysiology Changes
SICI: Contralesional M1
Thirty-one participants had baseline and postintervention paired-pulse intracortical neurophysiological data from the contralesional M1 where MEP were recorded in the contralateral, less-affected hand. One participant was an outlier, therefore we excluded his data from analysis. Twenty-two participants had intracortical neurophysiological data from stimulating the contralesional M1′ when MEP was recorded in the ipsilateral, affected hand.
SICI of the contralesional M1 to the less-affected hand was evident on the group level at baseline with a mean reduction in conditioned MEP amplitude of 55% from neutral (Figure 4A, pre = 0.45 ± 0.29,
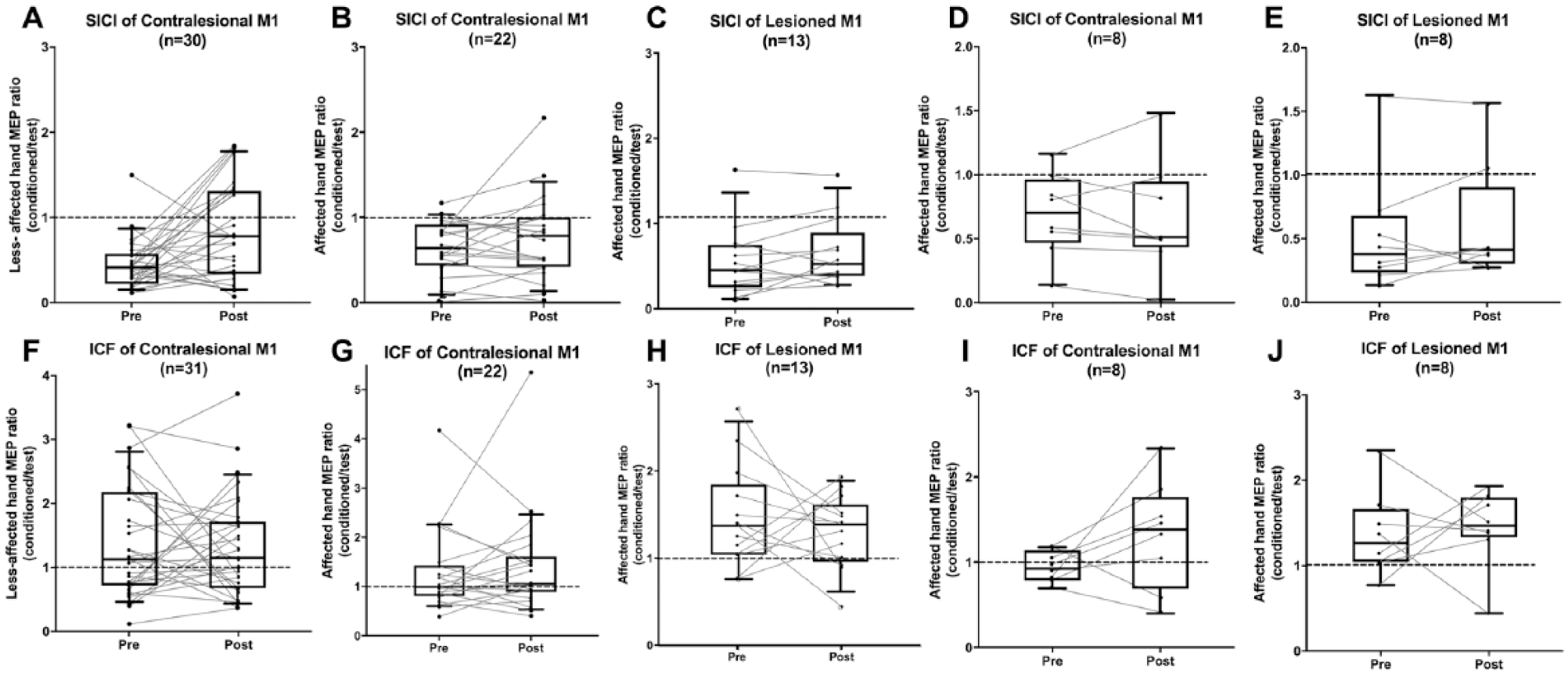
Intracortical neurophysiology. Box edges represent 25th and 75th percentile; the solid line inside the box represents median; whiskers represent the 10th and 90th percentiles. (A). Short-interval intracortical inhibition (SICI) of contralesional primary motor cortex (M1) controlling the less-affected hand (n = 30). A significant decrease was found (
We observed a significant decrease in the SICI of the contralesional M1 controlling the less-affected hand following intervention (Figure 4A, pre = 0.45 ± 0.29, post = 0.84 ± 0.56,
Treatment allocation did not affect the amount of decrease in SICI of the contralesional M1 to the less-affected hand as there were no significant interaction effects between intervention type and change in SICI (
SICI: Lesioned M1
Paired-pulse data from the lesioned M1 was available for 13 participants. At baseline, SICI was evident at the group level with a mean decrease in conditioned MEP of 45% from neutral (Figure 4C, pre = 0.55 ± 0.41,
We did not observe a significant decrease in SICI of the lesioned M1 controlling the contralateral, affected hand following intervention (Figure 4C, pre = 0.55 ± 0.41, post = 0.66 ± 0.39,
ICF: Contralesional M1
ICF of the contralesional M1 to the less-affected hand was evident on the group level at baseline with a mean increase in conditioned MEP of 39% from neutral (Figure 4F, pre = 1.39 ± 0.87,
No change was observed in the ICF of the contralesional M1 controlling the less-affected hand after intervention (Figure 4F, pre = 1.39 ± 0.87, post = 1.33 ± 0.79,
ICF: Lesioned M1
ICF was evident at baseline in the lesioned M1 at the group level with a mean increase in conditioned MEP of 47% from neutral (Figure 4H, pre = 1.47 ± 0.60,
Bilateral Neurophysiology Comparisons
Intracortical neurophysiological metrics in both the lesioned and contralesional M1′ to the affected hand were available for a subset of 8 participants (age range = 10-19 years, lesion hemisphere: left = 3, right = 5). In this subset, neither SICI nor ICF of either M1 appeared to change following intervention (Figure 4D and E, contralesional M1′: SICI,
Correlations Between Changes in Neurophysiological Measures and Clinical Outcome
The only neurophysiological outcome that consistently correlated with clinical improvement was ICF of the contralesional M1. Negative correlations of modest strength were observed between changes in ICF of the contralesional M1 to the less-affected hand and changes in the COPM-satisfaction from pre- to postintervention (Figure 5A, n = 31, Pearson
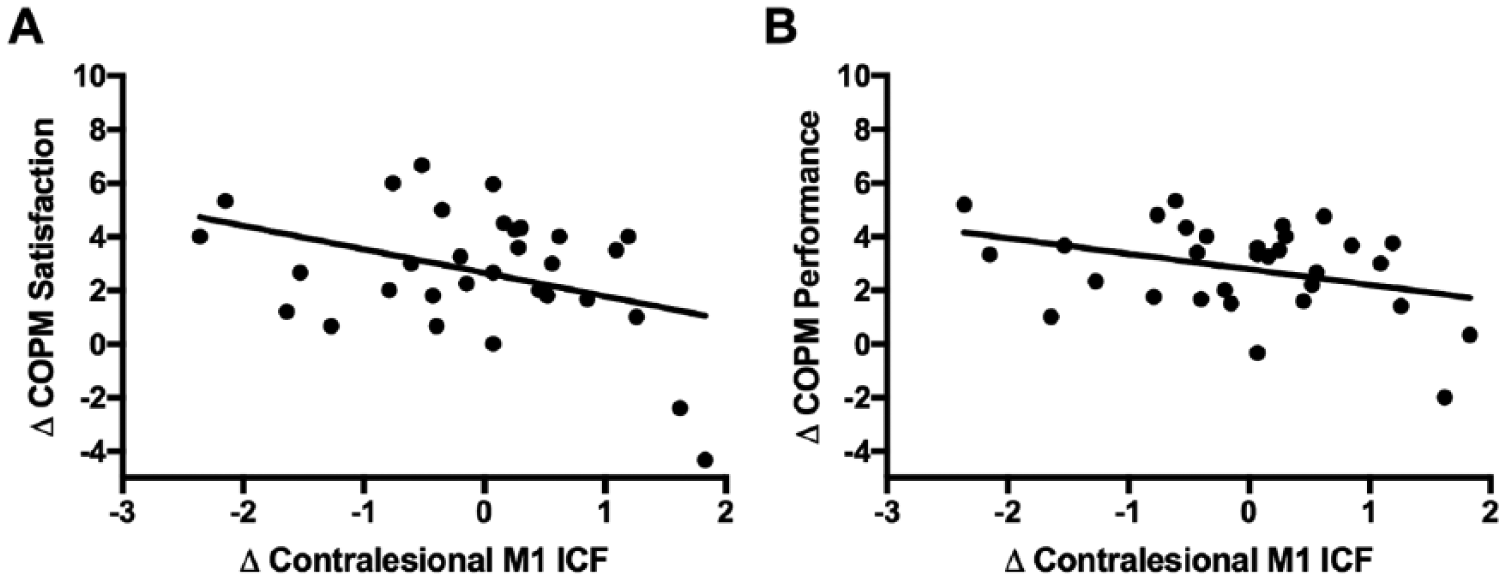
Correlations between clinical and neurophysiological changes. (A). Correlation between ΔICF and ΔCOPM-satisfaction. (B). Correlation between ΔICF and ΔCOPM performance. Change in ICF of the contralesional primary motor cortex (M1) to the less-affected hand was inversely associated with change in the COPM including both performance (A, n = 31,
Discussion
We investigated motor cortex neurophysiological changes using TMS before and after constraint and/or brain stimulation therapies in children with HCP. Our major finding is an overall increased excitability of the contralesional M1 controlling the less-affected hand reflected by increased MEP and SRC amplitudes as well as decreased SICI following intervention. We were unable to demonstrate specific effects of rTMS or CIMT while the relationship between clinical and neurophysiological changes require additional study. Comprehensive TMS neurophysiological evaluations are feasible and informative in children with perinatal stroke and HCP.
Based on 3 different neurophysiological outcomes, we found evidence that the excitability of the contralesional M1 appears to increase following intensive therapy in children with HCP. The term excitability is nonspecific and we would cautiously avoid oversimplifying to suggest that M1 physiology can be reflected by simple unidirectional changes. However, that we observed increases in both MEP amplitudes and SRC curves as well as a decrease in SICI supports this general impression. This finding is consistent with previous studies describing that increased cortical excitability is associated with motor training in HCP.19,20 Despite the trial being well-powered for clinical efficacy, we may have been under-powered for the neurophysiological measures to detect whether the interventions of rTMS and/or CIMT played more specific roles in this intervention-induced change. Nevertheless, the CIMT+ group appeared to show different neurophysiological changes as compared with the CIMT− group in 2 measures (affected hand MEP amplitude from the contralesional M1 and affected hand SICI from the lesioned M1). Though speculative, this observation could relate to the amount of mass practice of the affected hand or other factors specific to CIMT. Our finding may also indirectly alleviate previous concerns that constraint may deteriorate the function of the less-affected upper extremity and/or downregulate the excitability of the contralesional M1. 32 Similarly, theoretical concerns of direct, lasting inhibition by low-frequency rTMS are disputed by our findings.
As a paired-pulse paradigm linked to specific cortical neurotransmitter systems, SICI may represent the most specific of the neurophysiological measures we explored. In typical development, Mayston et al 33 showed that such intracortical neurophysiology is often variable in children. In fact, the SICI parameters used in our study were based on adult evidence 27 and remain to be specifically defined in children. Our finding that a decrease in SICI was associated with intervention is consistent with previous intervention-associated findings in healthy adults and adult stroke. For instance, Ziemann et al 34 showed that a decreased SICI may facilitate motor practice-dependent plasticity in adult healthy individuals. Fujiwara et al 35 found an association between improvements in upper extremity function and disinhibition of SICI of the lesioned M1 after a combined intervention of electrical stimulation of impaired finger extensors with wrist splint in adult stroke. Similarly, Fleming et al 36 found decreased SICI of the lesioned M1 after combined somatosensory stimulation with upper extremity training in adult chronic stroke. Although the models differ between adult and perinatal stroke, we add evidence here that SICI may also be an important biomarker of interventional motor cortex plasticity in children with HCP.
Alternative explanations may underlie the changes in contralesional M1 excitability. Alterations in the interhemispheric inhibition (IHI) whereby each motor cortex can inhibit its contralateral homologue via transcallosal pathways has played a central role in adult stroke hemiparesis models and neuromodulation trials. 37 However, only recently have IHI properties of children been defined using the same paired-pulse TMS methods as in adults. 38 Furthermore, a recent study in perinatal stroke children found that IHI is often altered but in a different manner than adults including that a loss of IHI (or even interhemispheric facilitation) in the lesioned-to-contralesional direction occurs, the degree of which correlates with better motor function. 39 While this same study did not show clear effects of intervention on IHI properties, the same directional effect would be expected to correlate with increased excitability of the contralesional motor cortex, the primary change we observed in the current study.
Our data add to previous studies highlighting the importance of the neurobiological features of the contralesional M1 in children with HCP.18,20 By focusing on perinatal stroke and excluding other causes of HCP, we used a higher degree of specificity to understand the neurophysiological changes that occur. While we are not able to fully separate the control of the affected and less-affected hands by portions of the contralesional M1, the effects we observed were most prominent when measured from the less-affected hand. This may in part reflect the better availability of measurements from the CST pathway between contralesional M1 and the less-affected hand as ipsilateral pathways are more variable. However, recent TMS studies in perinatal stroke have suggested that when two motor cortices are “coexisting” in the contralesional hemisphere, they appear to share many of the same neurophysiological properties, including those measured here. 18 This “common control theory” requires further study but suggests that different nodes in the motor cortex network of children with HCP should be considered both individually and collectively.
There are several important limitations that might have affected our results. First, the contralesional M1 control of the affected hand was measured when targeting the hotspot of the less-affected hand, rather than being mapped independently. As reported by Marneweck et al, 40 hotspots of the two hands likely reside in distinct locations within the contralesional M1 and future studies would benefit from defining these separately. Unfortunately, we did not have an adequate sample size to properly test several hypotheses including whether the relative preservation of the ipsilateral CST affected children’s responsiveness to intervention. A subanalysis comparing the neurophysiological changes showed that neither ∆SICI nor ∆ICF differed between children with and without preserved ipsilateral CST. Another challenge when examining the motor system in children with TMS is that they typically have high RMT. While our trial was large with a factorial design that optimized power for individual intervention effects, we were powered for the clinical outcomes and likely underpowered for the higher variance neurophysiological outcomes. As it is challenging to recruit participants to such trials, we recommend future studies consider such outcomes carefully in the context of trial design. 41 With incomplete data across four treatment groups, several of our analyses were likely underpowered to show significant interactions between treatment and neurophysiological metrics. Finally, the lack of significant correlation between clinical outcome and neurophysiological measures may be partially explained by the fact that most of our neurophysiological changes were significant when the MEP was measured in the less-affected hand, whereas the clinical outcome may capture changes more in the affected hand (such as in the AHA).
Conclusions
In conclusion, we demonstrate that complex TMS neurophysiology can be successfully measured before and after intensive interventions in children with HCP. Our primary observation of increased contralesional M1 excitability remains incompletely understood but provides new evidence of possible interventional plasticity mechanisms and biomarkers of responsiveness that might help advance neuromodulation interventions and inform personalized neurorehabilitation in disabled children.
Supplemental Material
Supplementary_Figure – Supplemental material for Intervention-Induced Motor Cortex Plasticity in Hemiparetic Children With Perinatal Stroke
Supplemental material, Supplementary_Figure for Intervention-Induced Motor Cortex Plasticity in Hemiparetic Children With Perinatal Stroke by Hsing-Ching Kuo, Ephrem Zewdie, Patrick Ciechanski, Omar Damji and Adam Kirton in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank families in this study and Dr Yi-An Ko for statistics consultations.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Heart and Stroke Foundation (HSF), Alberta Children’s Hospital Research Institute (ACHRI), and Canadian Partnership for Stroke Recovery (CPSR).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
