Abstract
Background. Little is known about the induction of functional and brain structural reorganization in hemiplegic cerebral palsy (HCP) by constraint-induced movement therapy (CIMT). Objective. We aimed to explore the specific molecular mechanism of functional and structural plasticity related to CIMT in HCP. Methods. The mice were divided into a control group and HCP groups with different interventions (unconstraint-induced movement therapy [UNCIMT], CIMT or siRNA-Nogo-A [SN] treatment): the HCP, HCP+UNCIMT, HCP+CIMT, HCP+SN, and HCP+SN+CIMT groups. Rotarod and front-limb suspension tests, immunohistochemistry, Golgi-Cox staining, transmission electron microscopy, and Western blot analyses were applied to measure motor function, neurons and neurofilament density, dendrites/axon areas, myelin integrity, and Nogo-A/NgR/RhoA/ROCK expression in the motor cortex. Results. The mice in the HCP+CIMT group had better motor function, greater neurons and neurofilament density, dendrites/axon areas, myelin integrity, and lower Nogo-A/NgR/RhoA/ROCK expression in the motor cortex than the HCP and HCP+UNCIMT groups (P < .05). Moreover, the expression of Nogo-A/NgR/RhoA/ROCK, the improvement of neural remodeling and motor function of mice in the HCP+SN group were similar to those in the HCP+CIMT group (P > .05). The neural remodeling and motor function of the HCP+SN+CIMT group were significantly greater than those in the HCP+SN and HCP+CIMT groups (P < .05). Motor function were positively correlated with the density of neurons (r = 0.450 and 0.309, respectively; P < .05) and neurofilament (r = 0.717 and 0.567, respectively; P < .05). Conclusions. CIMT might promote the remodeling of neurons, neurofilament, dendrites/axon areas, and myelin in the motor cortex by partially inhibiting the Nogo-A/NgR/RhoA/ROCK pathway, thereby promoting the improvement of motor function in HCP mice.
Keywords
Introduction
Hemiplegic cerebral palsy (HCP) is the most common subtype of cerebral palsy (36%). 1 Children with HCP frequently exhibit unilateral dyskinesia, which commonly results in developmental neglect or disuse, and abnormal development after injury due to abnormal plasticity.1-3 Therefore, HCP can gradually change musculoskeletal biomechanics and cause contractures, severely reducing the quality of life among children. 4 So far, there are various rehabilitation techniques for HCP, for instance, constraint-induced movement therapy (CIMT), bimanual therapy, occupational therapy, and botulinum toxin injection.2,5 CIMT may improve hand function and overcome the phenomenon of developmental neglect in HCP. 6 Rha et al investigated the effects of CIMT on neurogenesis and functional improvement in HCP mice, but the endpoints were limited to increases in the numbers of neurons, and unable to produce any evidence relating increased neurogenesis to functional recovery. 7 Little is known about the microscopic variations in brain tissue induced by CIMT, and studies on CIMT-mediated functional changes and brain structure reorganization in HCP are still limited. 8 Therefore, we sought to further explore the specific molecular mechanisms by which CIMT affects functional and structural plasticity.
Nogo-A is widely recognized as a suppressor of neurite growth in the central nervous system (CNS). 9 Two domains of Nogo-A have been reported to regulate the same intracellular signaling pathway; in this pathway, Nogo-66’s neuroreceptor NgR1 10 and Nogo-A-Δ20 specific receptors together activate the small GTPase RhoA, which in turn binds to and activates Rho-related protein kinase (ROCK) to act on neural cell effectors, eventually resulting in cytoskeletal rearrangement, growth cone collapse, and axonal growth inhibition.10,11 Inhibition of the expression of Nogo-A in dorsal root ganglion neurons of neonatal mice increases growth cone areas and axonal growth and markedly enhances motor ability. 12 Moreover, Nogo-A exerts a sustained inhibitory effect on the growth and sprouting of axons until a relatively late stage of development. 13
CIMT promotes axon sprouting and motor recovery after stroke in rats by inhibiting the Nogo-A/RhoA/ROCK signaling pathway. 14 Nogo-A, a stabilizer and modifier of neural networks, has been confirmed to be related to a series of CNS disorders. 11 Therefore, we speculated that CIMT might promote neural remodeling by partially inhibiting the Nogo-A/NgR/RhoA/ROCK pathway signaling, thereby significantly improving the motor function of HCP mice.
Methods
Data Availability Statement
The experimental data generated and analyzed in this study will be made available and shared on request by any qualified researcher after approval of the ethics committee.
The timeline and mice assignment of the whole experiment is depicted in Figure 1.
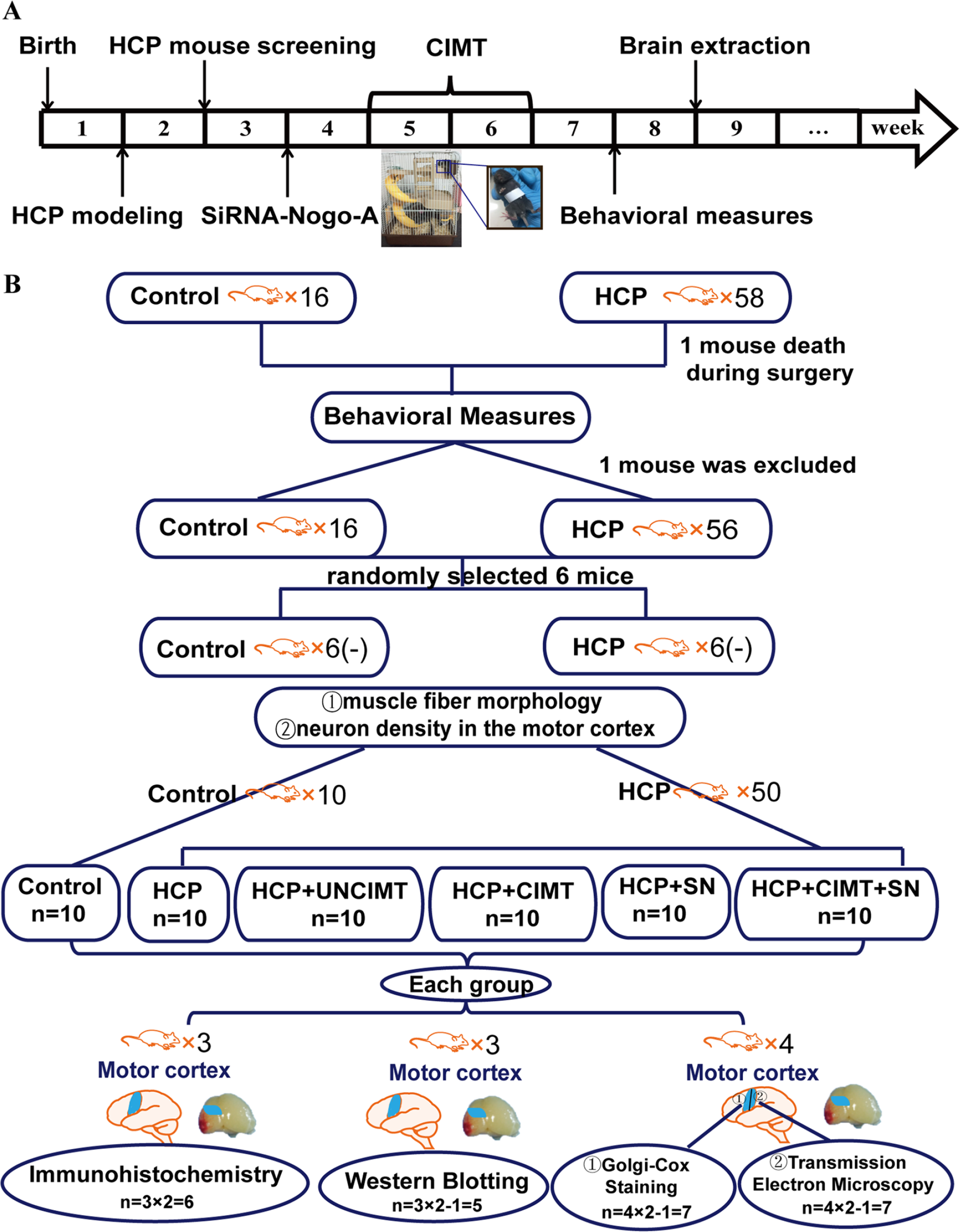
The timeline and mice assignment of the whole experiment. (A) The timeline of this study. In the whole experiment, treated mice were assessed prior to control mice. (B) Mice assignment of the whole experiment. HCP, hemiplegic cerebral palsy; CIMT, constraint-induced movement therapy; UNCIMT, unconstraint-induced movement therapy; SN, siRNA-Nogo-A.
HCP Model
This study received authorization from the institutional review board of Guangzhou Women and Children’s Medical Center, China (Protocol 2016021619). Pregnant female C57BL/6 mice (n = 12, 20-24 g, SCXK Yue2016-0041 [year 2016]) purchased from the Guangdong Medical Experimental Animal Center (Guangzhou, China) were used. The mice were raised in specific pathogen-free laboratory that approved by the laboratory animal ethics review committee.
On postnatal day 7 (equivalent to human fetuses between 32 and 34 weeks of age), 15 neonatal mice (n = 58) were selected to undergo left common carotid artery ligation after inducing anesthesia with a mixture of ketamine (80 mg/kg) and xylazine (16 mg/kg). After the operation, the mice were placed in a 37 °C incubator in which the oxygen concentration was controlled at 8% and the nitrogen concentration was controlled at 92% for 2 hours of hypoxia. 7 One mouse was excluded from the study owing to death during surgery.
The mice were selected based on motor deficits, then histological analysis of neuron density 16 and muscle fiber morphology17,18 as terminal procedure were used for screening. One week after the operation, a beam-walking test was carried out to analyze motor function of the 57 mice. The 56 mice with obvious unilateral limb motor dysfunction were selected for subsequent experiment. In order to assess whether the 56 selected mice had successfully modeled HCP, 6 mice of these mice were chosen at random and neuron density in the motor cortex measured, and biceps brachialis muscle fiber morphology examined. The neurons density in the motor cortex region was observed by hematoxylin and eosin (HE) staining (Figure 2A and B) and the morphology and arrangement of the muscle fibers of the right upper limb biceps brachialis of the mice was observed by electron microscope (Fei, USA, Tecnai G2 [year 2013]; Figure 2C). In these 6 mice, motor cortex neurons density was significantly reduced and muscle fiber morphology appeared abnormal (Figure 2B and D), indicating that HCP mice were successful in modeling.
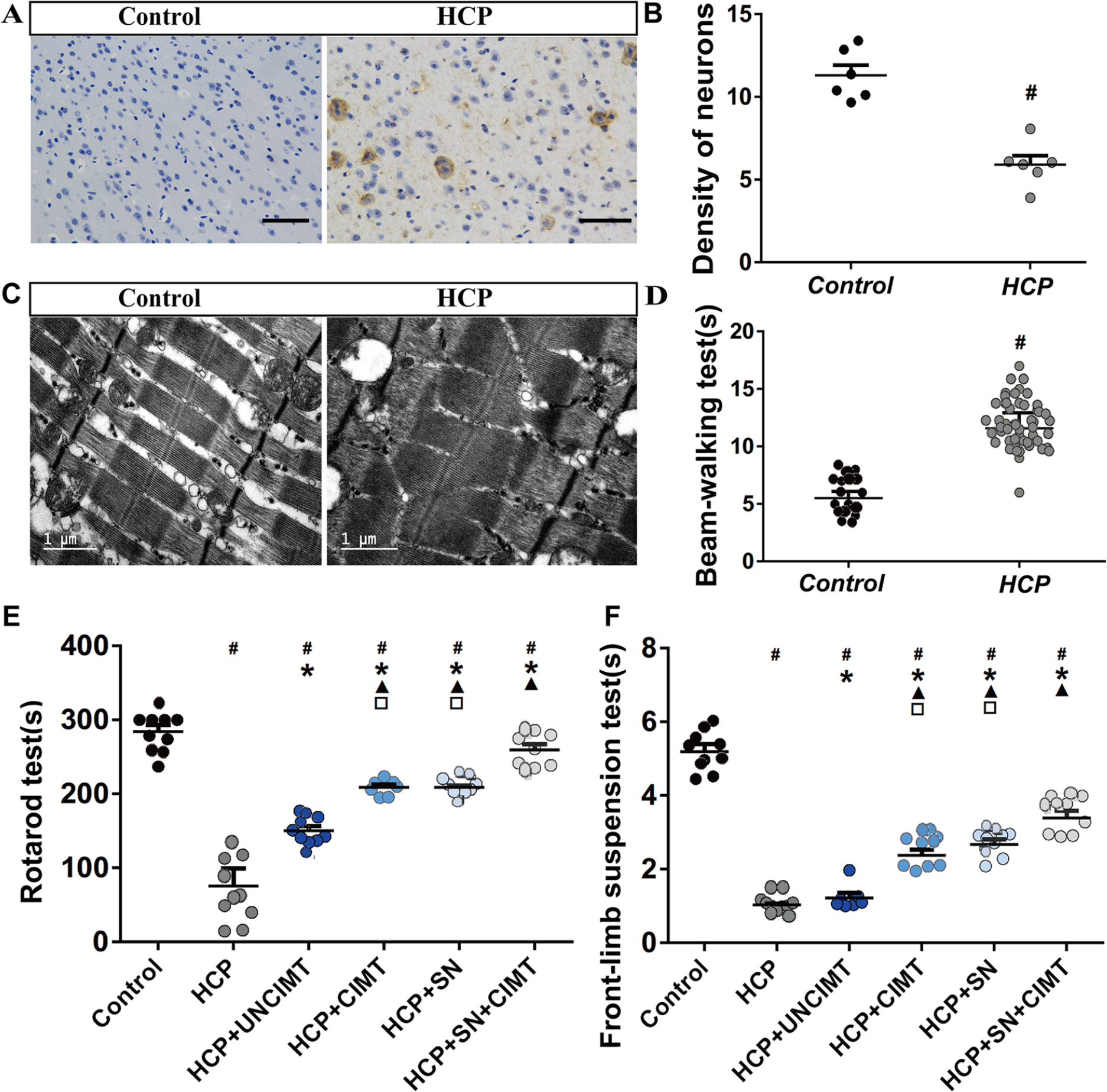
HCP model and behavioral test results. (A) The density of neurons in the motor cortex were observed by HE staining (n = 6). (B) The quantitative results of neurons density (n = 6). (C) The morphology of biceps brachialis muscle fibers were observed by electron microscopy (n = 6). (D) The beam-walking test results (HCP group, n = 57; control group, n = 16). (E) Comparison of the rotarod test results among the different groups (n = 10). (F) Comparison of the front-limb suspension test results among the different groups (n = 10). The data are shown as the mean ± SEMs. HCP, hemiplegic cerebral palsy; CIMT, constraint-induced movement therapy; UNCIMT, unconstraint-induced movement therapy; SN, siRNA-Nogo-A; scale bar = 100 µm (
Based on the above-mentioned results and the motor dysfunction of the remaining 50 mice, 19 the 50 HCP mice and 10 normal mice were ultimately selected for the study. The power analysis was used to calculate a reasonable sample size. 20 Aside from those in the control group (n = 10), a randomized 5-mice block allocation was employed to assign to 1 of 5 experimental groups (Figure 1B): an HCP group (n = 10); an HCP+UNCIMT (unconstraint-induced movement therapy [UNCIMT]) group (the HCP+UNCIMT group; n = 10); an HCP+CIMT group (the HCP+CIMT group; n = 10); an HCP group that received intracerebroventricular injection of siRNA-Nogo-A in the left brain (the HCP+SN group; n = 10); and an HCP+CIMT group that received intracerebroventricular injection of siRNA-Nogo-A in the left brain (the HCP+SN+CIMT group; n = 10). The specific randomization method was to number HCP mice as 1 to 50, so that for every 5 consecutive numbers, a mouse was assigned to 1 of the 5 groups.
Intervention
The interventions included CIMT, UNCIMT, and siRNA-Nogo-A treatment. The CIMT emphasized restraint of unaffected forelimbs and intensive structural training,6,21-23 which was meant to strengthen the training of the affected forelimbs, and structurally adjust the overall arrangement of the training environment according to the motor proficiency of the mice. The unaffected forelimbs of the mice in the HCP+CIMT and HCP+SN+CIMT groups were fixed onto the body with soft adhesive tape for 8 hours every day to induce activity on the affected side (4 hours in the morning and 4 hours in the afternoon). During the intervention time, the researchers would continuously check the condition of the mice, and once restricted forelimb was found to be broken away, they would immediately restore the restriction. The mice were placed in standard cages with abundant toys, and the types or positions of the devices were changed once a week to provide the mice with rich and novel play environments.
24
In order to achieve the effect of intensive training, mice need to continuously pass through the game facilities in the cages to obtain food and water. The treatment cycle lasted for 2 weeks.
7
The UNCIMT mainly included intensive structural training that did not constrain the unaffected forelimbs. Except that the unaffected forelimbs were not restrained, the intervention methods and feeding environment of the mice in the UNCIMT group were consistent with those in the CIMT groups. We performed intracerebroventricular injection of siRNA-Nogo-A into the left brain to silence Nogo-A expression in the HCP+SN and HCP+SN+CIMT groups between 8 and 12
Behavioral Measures
Rotarod and front-limb suspension tests were performed between 8 and 10
Rotarod Test
The rotarod test was implemented as previously described with some revisions using a 6-station rotarod treadmill (Panlab, LE8205 [year 2016]). 26 Mice were placed on the rotarod treadmill, and the time of stay on the rotarod was recorded. During the formal test, the speed was set to increase from 4 rpm to 80 rpm within 5 minutes, and the total test time was limited to 300 seconds. Each test finished when a mouse dropped off, made one full backwards revolution while hanging, or reached 300 seconds. The average duration of 3 tests was analyzed for each mouse. 27
Front-Limb Suspension Test
The test was adapted from previous research. 19 The experimental setup consisted of a steel wire tied to a stable object and a thick cushion in the drop area. The experimenter first grasped a mouse so that its 2 forelimbs could grasp the steel wire; then, the mouse was released, a stopwatch was used to record the total time taken to fall, and limb weakness was observed. The average value of 3 drop times was analyzed.
Neurogenesis Detection and Protein Determination
Immunohistochemistry
Brain samples from mice in each group were taken out after inducing anesthesia (dose as described above) and processed according to routine sampling and fixation methods for immunohistochemistry. 28 The tissues were sliced into 15-µm-thick coronal frozen sections on a microtome-cryostat (Leica, CM1860 [year 2015]) for immunostaining of neurons, neurofilament, and Nogo-A/NgR/RhoA/ROCK pathway members. The labeled antibodies included mouse anti-NF160 (1:1000, DSHB, 3409S [year 2017]), rabbit anti-NeuN (1:1000, Abcam, ab177487 [year 2017]), rabbit anti-Nogo-A (1:400, Abcam, 13401S [year 2017]), rabbit anti-NgR (1:400, Abcam, ab184556 [year 2017]), rabbit anti-RhoA (1:500, Abcam, 2117S [year 2017]), and rabbit anti-ROCK (1:1000, Abcam, 9029S [year 2017]) antibodies. Goat anti-mouse antiserum (1:200, Abcam, Alexa Fluor 488, ab150117 [year 2017]) and goat anti-rabbit antiserum (1:200, Abcam, Alexa Fluor 594, ab150080 [year 2017]) were used for signal detection. The morphology and density of neurons, neurofilament, and Nogo-A/NgR/RhoA/ROCK signal in the motor cortex were observed under a 400× light microscope (Leica, TCS SP8 STED 3X [year 2014]) with a laser confocal imaging system. We loaded the confocal imaging results into Image J to statistically analyze the positive neurons, neurofilament, and Nogo-A/NgR/RhoA/ROCK signal expression in the motor cortex by measuring the average intensity of immunofluorescence within a given area to observe differences among groups. 29
Golgi-Cox Staining
The mice were humanely killed after inducing anesthesia (dose as described above), and their brain tissues were removed directly and washed with normal saline. The brain tissues were then processed according to the instructions of the commercially procurable FD Rapid Golgi Stain Kit (Neurotechnologies, PK401A [year 2017]). Subsequently, the coronal surfaces of the agarose-embedded motor cortex tissues were cut into 150-µm-thick sections with a vibrating microtome (Leica, VT1200S [year 2013]). The sections were mounted, dehydrated, and sealed. An Olympus IX81 microscope (Olympus, IX81 [year 2014]) equipped with an F-View II (sw) camera was used for imaging. The reconstruction of single neuron was realized and the total surface area of dendrites and axon on a single neuron was counted with the Imaris 20.0 software. 30 All pictures were processed and analyzed with Adobe Photoshop CS6 13.0×64 and Image J software. 31
Transmission Electron Microscopy
After inducing anesthesia (dose as described above), mice were transcardially perfused with normal saline. Then, brain tissue samples with a size of 1 mm × 1 mm × 1 mm were taken from the motor cortex and fixed in phosphate buffer containing 4% PFA and 2.5% glutaraldehyde for 3 to 4 hours. Routine preparation procedures for electron microscopy samples were used to process the tissues. Ultrathin sections of the processed tissues were subjected to uranyl acetate and lead citrate staining. The myelin morphology and ultrastructure in pyramidal cell layer of motor cortex were observed and photographed under a 135 000× transmission electron microscope (Fei, Tecnai G2 [year 2013]). 32 All images were processed and analyzed with Image J software.
Western Blot Analysis
The cerebral cortices were quickly excised from the brain tissues of mice from each group after inducing anesthesia (dose as described above), dissected and homogenized in lysis buffer containing a complete protease inhibitor mixture. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis-separated proteins were transferred to a polyvinylidene fluoride membrane, which was then incubated with specific rabbit anti-Nogo-A (1:1500, Abcam, 13401S [year 2017]), rabbit anti-NgR (1:600, Abcam, ab184556 [year 2017]), rabbit anti-RhoA (1:1000, Abcam, 2117S [year 2017]), and rabbit anti-ROCK (1:1000, Abcam, 9029S [year 2017]) antibodies. The immunoreactivity was observed by enhanced chemiluminescence, and the protein bands were detected with a ChemiDoc XRS+ system (Bio-rad, 1708265 [year 2010]). 33 β-Actin were the internal references for quantitative analysis of the protein expression levels. The densitometric analysis and quantification of protein bands were performed using Image J software. 34
Statistics
Statistical analysis was conducted using SPSS software (version 20.0). The Shapiro-Wilk and Levene tests were used to evaluate data normality and to confirm the homogeneity of the variance, respectively. One-way analysis of variance was used to analyze the differences in behavioral results, neural remodeling indexes and Nogo-A/NgR/RhoA/ROCK expression among groups, and the results are shown as the means ± SEMs. The neural remodeling indexes and Nogo-A/NgR/RhoA/ROCK expression 2 weeks after CIMT was defined as the primary endpoint of this study. Pearson correlation analysis was applied to analyze the correlations between the neural morphology and behavioral results. In addition, the analysis and experimental group assignment were performed by a different person than the experimenters. The statistical significance threshold was set at P < .05.
Results
Behavioral Measures
The results of the rotarod and front-limb suspension tests are shown in Figure 2E and F. The residence times on the rotarod and wire of the control group were significantly longer than those of the other groups (P < .05, Figure 2E and F). The residence time of the HCP group was significantly shorter than those of the HCP+UNCIMT, HCP+CIMT, HCP+SN, and HCP+SN+CIMT groups (P < .05, Figure 2E and F). The residence time of the HCP+UNCIMT group was significantly shorter than those of the HCP+CIMT, HCP+SN, and HCP+SN+CIMT groups (P < .05, Figure 2E and F). There was no significant difference between HCP+CIMT and HCP+SN groups (P > .05, Figure 2E and F). In addition, the HCP+CIMT and HCP+SN groups showed shorter times than the HCP+SN+CIMT group (P < .05, Figure 2E and F).
Neurogenesis Detection and Protein Determination
Neurons and Neurofilament Remodeling
The intensity of Neun immunohistochemistry labelling of neurons in the motor cortex samples were determined for each group (Figure 3A and B). The differences between the control group and the other groups were statistically significant (P < .05, Figure 3B). And the intensity of Neun immunohistochemistry labelling of neurons in the HCP and HCP+UNCIMT groups exhibited fewer than the HCP+CIMT, HCP+SN, and HCP+SN+CIMT groups (P < .05, Figure 3B).
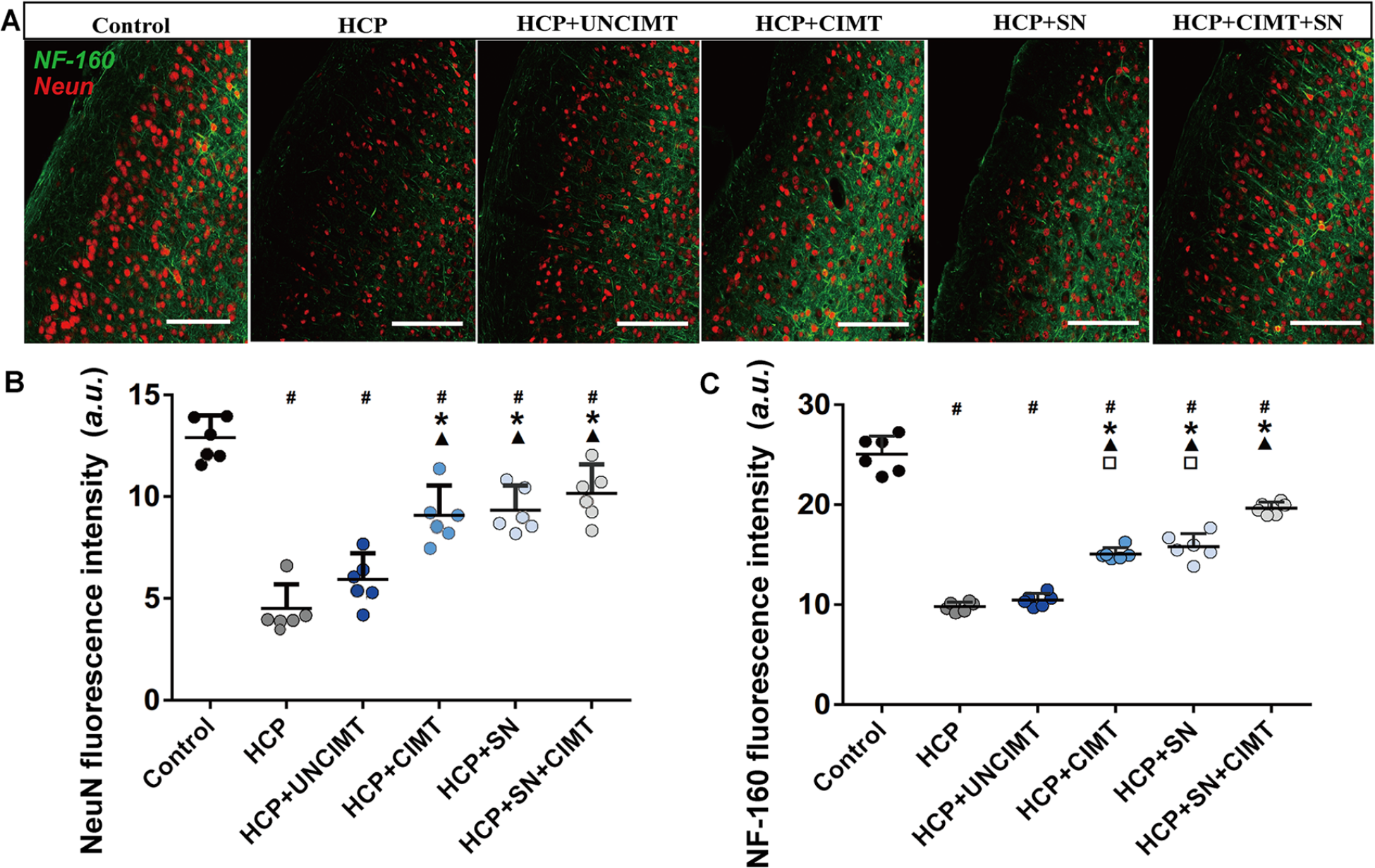
Neurons and neurofilament remodeling. (A) Neurons and neurofilament in the motor cortex of each group were observed under a 400× laser confocal microscope (red represents neurons, and green represents neurofilament). (B) The intensity of Neun immunohistochemistry labelling of neurons was measured in the motor cortex among the different groups. (C) The intensity of NF-160 immunohistochemistry labelling of neurofilament was measured in the motor cortex among the different groups. The data are shown as the mean ± SEMs. Neun, neuron; NF-160, neurofilament; a.u., arbitrary unit; HCP, hemiplegic cerebral palsy; CIMT, constraint-induced movement therapy; UNCIMT, unconstraint-induced movement therapy; SN, siRNA-Nogo-A; scale bar = 100 µm; n = 3 per group (2 sections from each mice, 3 × 2 = 6) (
The intensity of NF-160 immunohistochemistry labelling of neurofilament in the control group was significantly higher than those in the other groups (P < .05, Figure 3C). The HCP and HCP+UNCIMT groups showed lower intensity of NF-160 immunohistochemistry labelling of neurofilament than the HCP+CIMT, HCP+SN, and HCP+SN+CIMT groups (P < .05, Figure 3C). However, the intensity of NF-160 immunohistochemistry labelling of neurofilament in the HCP+CIMT group and HCP+SN group had no significant difference (P > .05, Figure 3C). The HCP+SN+CIMT group showed a higher intensity of NF-160 immunohistochemistry labelling of neurofilament than the HCP+CIMT and HCP+SN groups (P < .05, Figure 3C).
The results of the rotarod and front-limb suspension tests on HCP mice were positively correlated with the intensity of Neun immunohistochemistry labelling of neurons (r = 0.450 and 0.309, respectively; P < .05) and intensity of NF-160 immunohistochemistry labelling of neurofilament (r = 0.717 and 0.567, respectively; P < .05), as determined by Pearson analysis.
Dendrite and Axon Remodeling
On the basis of the single-neuron reconstruction (Figure 4C), the total surface area of dendrites and axon on single neuron in the motor cortex were quantified (Figure 4D). The differences between the control group and the other groups were all statistically significant (P < .05, Figure 4D). The areas of the HCP and HCP+UNCIMT groups were significantly smaller than those of the HCP+CIMT, HCP+SN, and HCP+SN+CIMT groups (P < .05, Figure 4D). There were no significant differences between HCP+CIMT and HCP+SN groups (P > .05, Figure 4D). Moreover, the HCP+SN+CIMT group showed a greater area than the HCP+CIMT and HCP+SN groups (P < .05, Figure 4D).
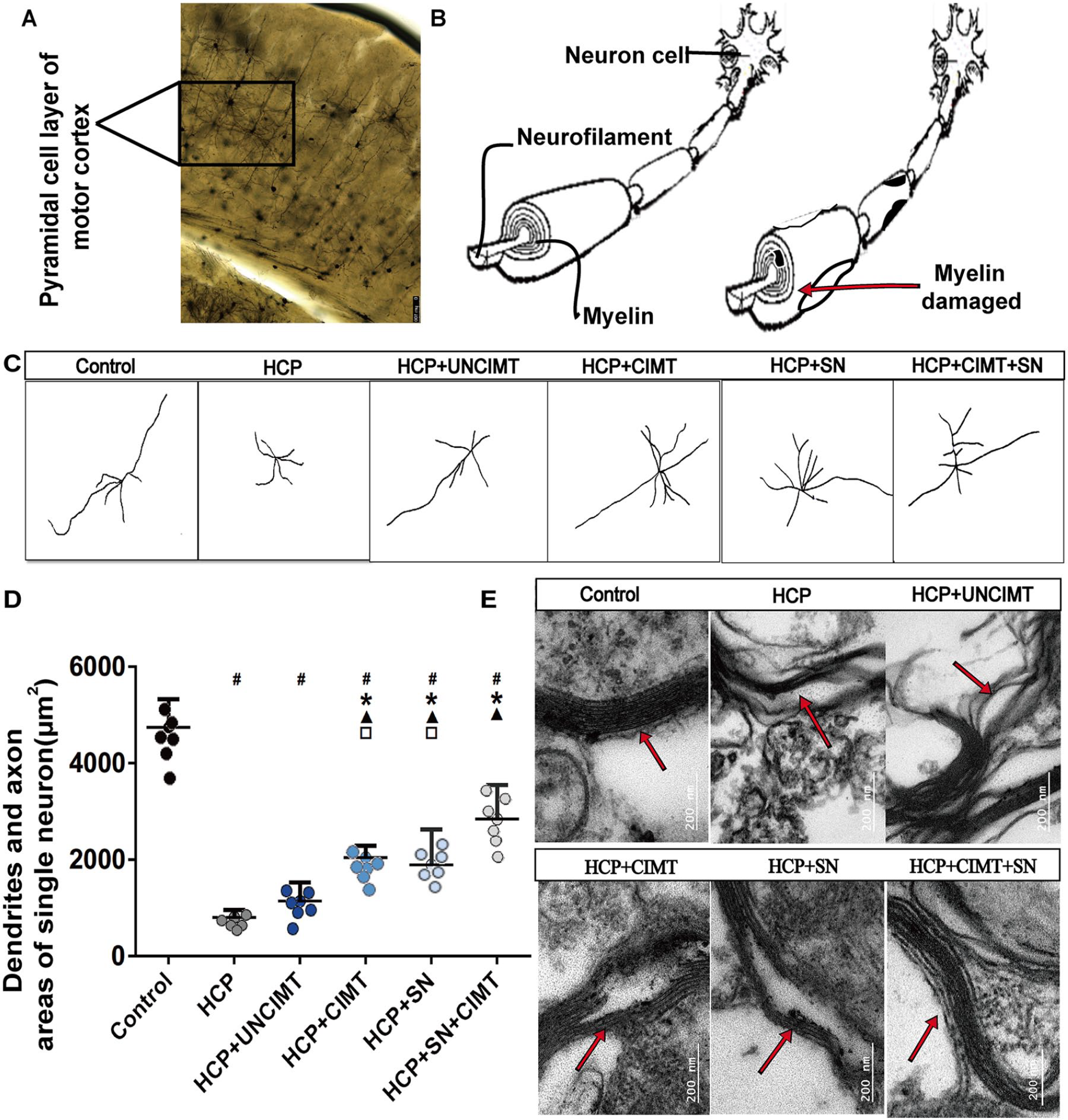
Dendrite/axon and myelin remodeling. (A) The morphology of neurons and neurofilament in the pyramidal cell layer of motor cortex under Golgi staining. (B) The schematic diagram of neurons and neurofilament. (C) Individual neurons in the motor cortex in each group were reconstructed (7 neurons from each group were selected for reconstruction). (D) Comparison of the dendrites and axon areas of single neuron in the pyramidal cell layer of motor cortex among the different groups. (E) The ultrastructure of myelin in the pyramidal cell layer of motor cortex was observed under a 135 000× transmission electron microscope. The data are shown as the mean ± SEMs. HCP, hemiplegic cerebral palsy; CIMT, constraint-induced movement therapy; UNCIMT, unconstraint-induced movement therapy; SN, siRNA-Nogo-A; scale bar = 200 µm; n = 4 per group (2 sections from each mice, with 1 section was damaged during processing, 4 × 2 − 1 = 7) (
Myelin Remodeling
Observation of the morphology of myelin on the outer layer of nerve axon under transmission electron microscopy (Figure 4E). It was found that the myelin in the control group were arranged clearly and orderly, and the boundary was clearly defined (Figure 4E). The myelin in the HCP group showed a loose and irregular state. A large number of vacuoles and dense materials accumulated around the myelin, the continuity of which was interrupted, and severe axon demyelination occurred (Figure 4E). The continuity of the myelin in the HCP+UNCIMT group was still obviously interrupted, but the accumulation of dense matter and vacuoles around the myelin were significantly reduced (Figure 4E). In the HCP+CIMT group, the myelin was significantly remodeled, which was shown in arrangement level and the continuity of myelin (Figure 4E). In the HCP+SN group, the arrangement and the continuity of the myelin was relatively improved, the accumulation of vacuoles and dense matter in the myelin was significantly reduced (Figure 4E). In the HCP+SN+CIMT group, the laminar structure of the myelin was neatly arranged, the myelin structure was dense, and the continuity was complete (Figure 4E).
Expression of Nogo-A/NgR/RhoA/ROCK
According to the results of immunohistochemistry and Western blot analysis (Figures 5 and 6). The Nogo-A/NgR/RhoA/ROCK expression levels in the HCP and HCP+UNCIMT groups were significantly higher than those in the control group (P < .05, Figures 5B and 6B). However, there was no significant difference among the control, HCP+CIMT, HCP+SN, and HCP+SN+CIMT groups (P > .05, Figures 5B and 6B). The HCP+CIMT, HCP+SN, and HCP+SN+CIMT groups showed lower expression of Nogo-A/NgR/RhoA/ROCK than the HCP and HCP+UNCIMT groups (P < .05, Figures 5B and 6B).
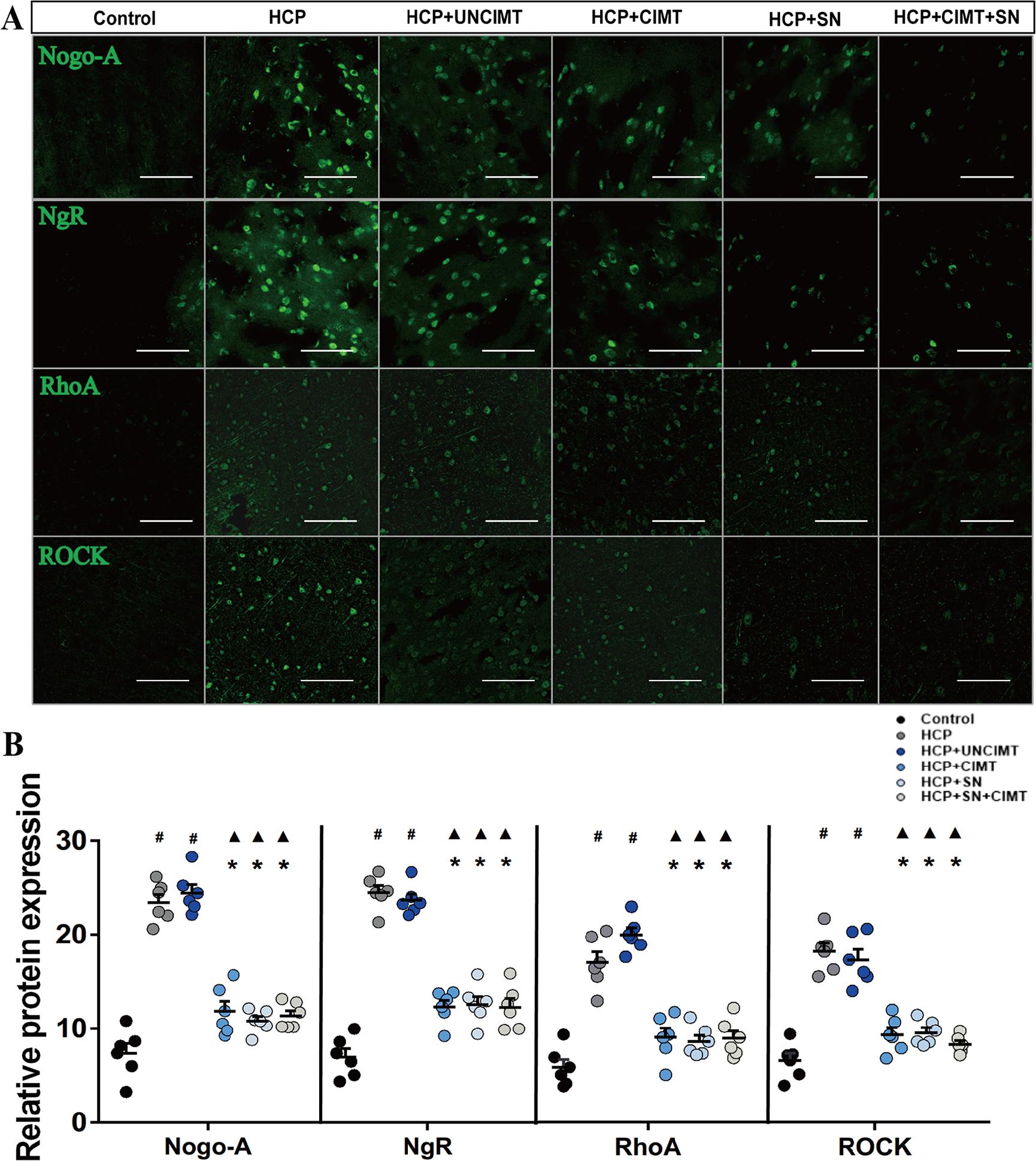
Expression of Nogo-A/NgR/RhoA/ROCK in the motor cortex by immunohistochemistry. (A) Expression of Nogo-A/NgR/RhoA/ROCK in the motor cortex in each group was assessed by immunohistochemistry. (B) Comparison of the expression of Nogo-A/NgR/RhoA/ROCK in the motor cortex among the groups based on immunohistochemistry. The data are shown as the mean ± SEMs. HCP, hemiplegic cerebral palsy; CIMT, constraint-induced movement therapy; UNCIMT, unconstraint-induced movement therapy; SN, siRNA-Nogo-A; scale bar = 100 µm; n = 3 per group (2 sections from each mice, 3 × 2 = 6) (
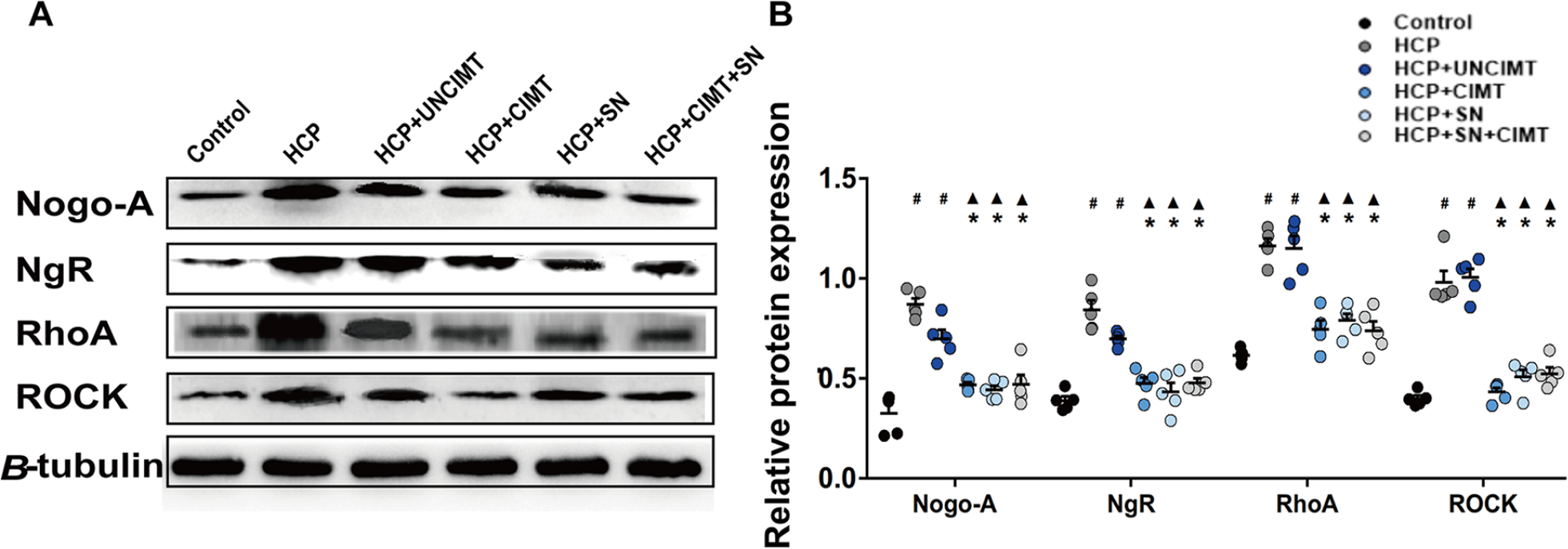
Expression of Nogo-A/NgR/RhoA/ROCK in the motor cortex by Western blotting. (A) Expressions of Nogo-A/NgR/RhoA/ROCK were analyzed with Western blotting. (B) Comparison of the expression of Nogo-A/NgR/RhoA/ROCK in the motor cortex among the groups. The data are shown as the mean ± SEMs. HCP, hemiplegic cerebral palsy; CIMT, constraint-induced movement therapy; UNCIMT, unconstraint-induced movement therapy; SN, siRNA-Nogo-A; n = 3 per group (2 sections from each mice, with 1 section was damaged during processing, 3 × 2 − 1 = 5) (
Discussion
In this study, a model of immature brain injury was used to recapitulate the brain injury observed in children with HCP. 35 This model has been widely accepted and applied in animal studies on perinatal hypoxic-ischemic brain injury.35,36 This was the first study based on pros and cons, to indicate that CIMT might promote the remodeling of neurons, neurofilament, dendrites/axon, and myelin in the motor cortex of mice by partially inhibiting the Nogo-A/NgR/RhoA/ROCK pathway and to suggest that brain structural remodeling drives functional reorganization which substantially improves the motor function of HCP mice. Studying the beneficial mechanism of CIMT on HCP mice might provide a promising target for drug intervention and optimize the clinical application of CIMT.
The rotarod and front-limb suspension tests are known to reveal an animal’s balance coordination and forelimb strength, respectively.19,27 Perinatal hypoxic-ischemic injury damages the motor cortex, periventricular white matter and striatum 37 and thus greatly reduced the balance coordination and forelimb strength of HCP mice in this study. Our research demonstrated that CIMT significantly improved the balance coordination and forelimb strength of HCP mice, and the improvement degree of HCP+CIMT group was significantly higher than that of the HCP+UNCIMT group. These findings are identical to those of previous studies in which CIMT has improved affected limb use and movement quality in perinatal hypoxic-ischemic brain injury and stroke models.7,14 And our results further revealed that the efficacy of CIMT was greater than that of equivalent amounts of unrestrained therapy.
CIMT has been widely used in the rehabilitation of hand dysfunction caused by a series of neurological diseases, such as cerebral palsy and stroke.6,38 Sterling et al found that CIMT increases gray matter volume in the developing CNS of HCP children. 39 Rha et al revealed that CIMT significantly increased of BrdU+ cells in the subventricular zone and BrdU+⁄βIII-tubulin+ cells in both the subventricular zone and striatum treated in HCP mice. 7 Our results not only confirmed the findings of previous studies but also revealed additional in-depth structural remodeling of the brain, showing that CIMT could preserve the neurons and neurofilament density, dendrites/axon areas on single neuron, and myelin integrity in the motor cortex region in HCP mice. Current research has indicated that dyskinesia in children with HCP is mainly due to impaired sensorimotor pathways and impaired motor planning. 1 As the most advanced regulatory center for body movement, the motor cortex receives and integrates information from different regions and then outputs the processed motor signals to control the movement of the entire body. 40 The human cerebral cortex contains about 16 billion neurons. 41 Neurons in the connected brain regions of motor cortex begin planning for specific movements before they occur. 42 Each neuron receives information through dendrites and then transmits the processed information to the designated targets through axons, and their interconnections form functional neural networks based on behavior.43,44 Preservation of neurons and neurofilament density, dendrites/axon areas likely play significant roles in the processing of motor loop information and afferent output. Previous research has indicated that impairment of myelin continuity changes the passive “cable” characteristics of axons, thus significantly reducing the conduction velocity and peak arrival times of signals in the CNS. 45 The improvement of myelin integrity in HCP mice likely markedly increased the pulse transmission speed of the neural circuit and improved the efficiency of information transmission. Functional outcomes deteriorate with reduced neurogenesis, indicating the important role of neural remodeling and the involvement of this process in mediating functional recovery. 46 In addition, our correlation analysis showed that the density of neurons and neurofilament were both significantly positively correlated with motor function in mice. These results showed that CIMT prominently enhanced the balance coordination and forelimb strength of HCP mice by promoting neural remodeling (of neurons, neurofilament, dendrites/axon areas, and myelin).
However, the extent of neural remodeling was regulated to some extent by endogenous growth inhibitory molecules or neurotrophic factors. 47 Therefore, aiming to counteract or block inhibitory factors could improve brain remodeling and functional outcomes in HCP mice. Nogo-A, an inherent inhibitor of myelin-associated axon growth, which obstructs the regeneration of axons and the myelinization of newly formed fibers, restricts synaptic plasticity and thus prevents the establishment of new functional circuits. 9 Our results demonstrated that the expression of Nogo-A/NgR/RhoA/ROCK in HCP mice was significantly higher than that in normal mice, consistent with the findings of previous studies. Previous studies have revealed that neurological diseases such as stroke cause marked increases in Nogo-A/NgR and RhoA/ROCK levels in the cortex surrounding areas of cerebral infarction. 14 The transient promotion of Nogo-A is consistent with the failure of spontaneous axonal sprouting after stroke.48,49 In addition, our results showed that CIMT significantly reduced the expression of Nogo-A/NgR/RhoA/ROCK in the motor cortex in HCP mice and that the levels of Nogo-A/NgR/RhoA/ROCK after intervention were closer than those before intervention to the levels in typically developing mice. Deletion of the Nogo-A gene leads to larger and more complex dendritic structures and larger and stronger parallel fiber synapses. 50 Consistent with this conclusion, we found that the expression of Nogo-A/NgR/RhoA/ROCK, neural remodeling, and motor function levels in HCP+SN group were comparable to those in the HCP+CIMT group. Based on the above positive and negative arguments, our study demonstrated that CIMT might partially inhibit Nogo-A/NgR/RhoA/ROCK signal pathway to offset the inherent myelin-associated neurite outgrowth inhibition, thus significantly promoting neural remodeling and improving the motor function of HCP mice.
Nogo-A is expressed in the developing nervous system, especially in neurons after mitosis and neurons related to axon growth,12,51 and it participates in the competition of developing oligodendrocytes for axonal myelination and myelin space distribution. 52 However, the specific mechanism of Nogo-A and its related effectors on the developing brain is still unclear. Previous studies have suggested that the 2 main functional domains of Nogo-A, Nogo-A-Δ20, and Nogo-66 could activate small GTPase RhoA and Rho related protein containing kinase (ROCK), leading to the instability of growth machinery.53,54 Downstream cytoskeleton effectors of RhoA/ROCK pathway include myosin light chain II and cofilin, which induce contraction of growth cones by promoting contractile activity of myosin II or depolymerization of F-actin, respectively.55,56 In addition, Nogo-A may significantly inhibit the plasticity of the adult CNS by downregulating the expression of growth-related genes.13,57 Combined with our research results, we speculate that CIMT might have a significant regulatory effect on the neural remodeling of the HCP and even the developing brain by inhibiting the growth cone skeletal mechanism and the regulation mechanism of growth-related transcription factors of Nogo-A and its related effectors.
Rehabilitation methods that force the use of limbs after rodent brain injury might induce neurotrophic factors or other neural growth factors. 58 We found that neurofilament density, dendrites/axon areas, and myelin integrity in the HCP+SN+CIMT group were significantly greater than those in the HCP+SN group and HCP+CIMT group. However, there was no significant difference in the expression of Nogo-A/NgR/RhoA/ROCK among the HCP+CIMT, HCP+SN, and HCP+SN+CIMT groups. Therefore, in addition to the influence of the endogenous neural growth inhibitory factors Nogo-A and its downstream effector molecules, we could not exclude that the neural growth factors in the cortex around the infarct were also partly involved in the process of CIMT-induced neural remodeling in HCP mice. Zhai et al proclaimed that CIMT might improve behavioral outcomes in stroke rats through the combined effects of 3 mechanisms, including promotion of endogenous neurogenesis, promotion of angiogenesis in the area of ischemic infarction, and repair of the blood-brain barrier. 59 Some inherent myelin-related axonal growth inhibitors existed in the brain, including Nogo-A, myelin-associated glycoproteins, and oligodendrocyte glial glycoproteins; these factors might limit axonal growth and plasticity simultaneously. 60 The Nogo-A/NgR/RhoA/ROCK pathway is only one of the endogenous inhibitory pathways of neural regeneration and remodeling in HCP mice.
Notably, some limitations existed in our study that needed to be improved in further researches. First, the specific mechanisms by which silence of Nogo-A and its receptor promoted brain structural remodeling in HCP were still unclear. Furthermore, the etiology of HCP was complex and diverse, and our model of perinatal hypoxic-ischemic brain injury could not fully replicate the brain damage observed in children with HCP. Finally, our results needed to be confirmed in a larger sample size.
Conclusions
In summary, CIMT might promote the remodeling of neurons, neurofilament, dendrites/axon areas, and myelin in the motor cortex by partially inhibiting the Nogo-A/NgR/RhoA/ROCK pathway in the developing brain, thereby promoting the improvement of motor function in HCP mice.
Footnotes
Acknowledgements
We are grateful to Dr Libing Zhou (Jinan University) for providing technical assistance and a conducive environment.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant Numbers 81902309, 81672253); the Natural Science Foundation of Guangdong Province (Grant Number 2019A1515010420); the Science Technology and Innovation Development Foundation of Guangzhou (Grant Number GWCMC2020LH-1-004); Featured Clinical Technique Foundation of Guangzhou (Grant Number 2019TS55); and the Guangzhou Institute of Pediatrics from Guangzhou Women and Children’s Medical Center (Grant Numbers YIP-2019-015, GCP-2018-007, YIP-2018-014).
