Abstract
Keywords
Introduction
Hemiparesis is one of the most frequent motor disabilities in children and is mainly a consequence of cerebral palsy (CP), that is, due to acute lesions in the developing brain acquired during the prenatal and/or perinatal periods. The neonatal prevalence of unilateral spastic CP is estimated between 0.6 and 0.9 per 1000 live births.1,2 Hemiparesis more infrequently results from lesions in later childhood, for example, after traumatic brain injury, stroke, or brain tumors.
Corticospinal projections are established at the beginning of the third trimester of pregnancy. Initially these projections are bilateral. During normal development, ipsilateral projections are withdrawn. 3 In pre- or perinatal brain lesions corticospinal projections to the affected hand can arise from the contralateral lesioned hemisphere (contralateral reorganization), or if contralateral projections are severely disrupted, ipsilateral projections from the unaffected hemisphere persist and take over the control of the paretic hand (ipsilateral reorganization). 3
Mirror therapy (MT) is a rehabilitation strategy based on the repeated use of the mirror illusion (MI). Patients train by looking into a mirror placed along their midline and hiding their defective limb. The observed reflection of the unimpaired limb superimposes itself on the defective one, thus generating the visual illusion of a functional limb. MT was initially devised as a strategy to alleviate phantom limb pain in amputees, before being applied as a neurorehabilitation approach for hemiparetic adults after stroke. 4
There is increasing evidence from randomized controlled trials regarding the effectiveness of MT for improving upper extremity motor function, activities of daily living and pain, at least as an adjunct to normal rehabilitation for adults after stroke. 5 In children with hemiparesis, a single pilot clinical trial demonstrated that MT may increase the strength and improve the dynamic function of the paretic arm. 6 The mechanism believed to underlie MT is its effect on “learned paralysis,” in which conflicts between motor efferences and reafferent sensory feedbacks impede motor function. 4 Every time a motor command is sent to the paretic limb the returning visual and proprioceptive signals inform the brain that the arm is not moving as expected. The aim of mirror visual feedback is to restore the congruity between motor efferences and visual afferences, allowing the subject to unlearn the “learned paralysis.”
The neural correlates of the MI have essentially been studied in healthy adults and adults with stroke by using functional magnetic resonance imaging (fMRI) and transcranial magnetic stimulation (TMS), as well as by positron emission tomography, magnetoencephalography, electroencephalography and near infrared spectrometry in healthy individuals only. 7
TMS is a neurophysiological method that measures the excitability of the motor cortex noninvasively. TMS has been repeatedly used to show how the excitability of the primary motor cortex (M1) is influenced by MI. A vast majority of TMS studies show that MI increases the excitability of the M1 projecting to the active hand, suggesting an instantaneous neuromodulatory effect of the visual feedback. 7 With increasing interest in implementing MT in the field of child neurorehabilitation, and despite growing evidence from the adult literature regarding neuroplastic changes, there is only scarce evidence of the potential underlying mechanisms of action in children. No studies of corticospinal activation in response to the MI have been performed in children and adolescents. We hypothesized that the MI would modulate corticospinal excitability in hemiparetic children with ipsilateral and contralateral reorganizations, as well as in typically developing (TD) subjects. This was tested by measuring the motor-evoked potentials (MEPs) elicited by the MI in children with hemiparesis and in TD peers. For this purpose, we used single-pulse TMS, and the amplitude and latency of MEPs were derived from the target muscle.
To determine which factors influenced motor cortex excitability, we compared the effect of the MI during active movement and when subjects only looked at the mirror without moving their hand. We measured MEPs in two muscles, each involved in hand opening and closing. We looked for effects of the MI in a unimanual and a bimanual motor task and we examined whether the kind of cortical reorganization influenced the effect of the MI on motor cortex excitability. We assessed whether the effects of the MI were influenced by the amount of hand function and how the amplitude of MEPs was affected by the measured muscle.
Methods
Subjects
Twelve participants with hemiparesis between 8 and 20 years and 8 TD controls were included. Exclusion criteria were: upper limb surgery <6 months preceding inclusion; functional impairment or comorbidities inhibiting active hand-opening; severe nonrefractive visual disorders (eg, hemianopia); mental age <8 years; behavioral comorbidities impeding participation; active epilepsy; pacemaker (for deep brain stimulation or heart disease); history of brain surgery/brain injuries (possible foreign bodies); eye surgery/injury (possible foreign bodies). The study was approved by the local ethics committee. All participants and parents gave written informed consent.
Functional Assessments
Stimulation and Recording
Subjects were seated with hands in neutral position, hands and forearms resting on a table surface. We recorded electromyographic (EMG) signals simultaneously from both forearms. We used silver–silver chloride surface electrodes (ALPINE, bioMed) attached in a tendon-belly arrangement over the musculus abductor pollicis brevis (APB) and the musculus flexor digitorum superficialis (FDS). 10 A Neurodata Amplifier System connected to an IPS230 Isolated Power System (Grass-Telefactor, Braintree, MA, USA) was used to preamplify 1000×) and bandpass filter (10-1000 Hz) EMG signals. Input was fed into a computerized data acquisition system (sampling rate 5 kHz). 11 EMG signals amplitudes were calculated for all recorded muscles in a time window of 65 ms. Latencies were automatically measured in every trial. We delivered monophasic TMS pulses at a frequency of 0.2 Hz on the M1 through a custom figure-of-8-shaped coil (diameter 5 cm) connected to a Magstim 200 (Magstim Co Ltd, Whitland, Wales, UK). Using this coil, with the Magstim set at 100%, the maximal field strength is 2.89 T, maximum rate of change is 129 A/µs, the peak current 7.2 kA, and pulse duration 95 µs. The coil was tangentially placed to the scalp (intersection of both wings at a 45° angle with the midline). 12
Both hemispheres were searched for stimulation points eliciting contralateral and/or ipsilateral MEPs. 13 The APB served as the target muscle for determining the corticospinal organization pattern. The nasion-inion line and the interaural line were used as reference markers for determination of the vertex. Marks were made on the vertex and 5 cm aside on the interaural line. 14
In participants with hemiparesis the hemisphere contralateral to the unaffected hand was stimulated first. The experiment started with identification of the “hot spot” (HS). 9 The coil was moved relatively to the starting point in anteroposterior and mediolateral directions. Starting stimulation intensity was set to 50% of maximal stimulator output and increased respectively decreased if applicable. Once the HS was located the resting motor threshold (rMT) of the unaffected APB was assessed (defined as the minimum stimulation intensity that produces 5 MEPs exceeding 50 µV amplitudes in 10 consecutive trials at rest 15 ). To measure the contralateral motor response 20 stimulations were applied at an intensity of 120% rMT. Correspondent MEPs were recorded for offline analysis. If stimulation of the HS elicited ipsilateral responses in the affected hand the rMT of the ipsilateral affected APB was measured separately. Again 20 stimulations were applied and ipsilateral MEPs recorded at 120% rMT. Hemisphere was then switched. Possible contralateral responses of the affected APB were measured using the same procedure. Target muscles were first tested in a relaxed state. In the absence of MEPs at rest, MEPs were examined under facilitation (gently squeezing a ball of foam at approximately 10% of maximal isometric contraction). The absence of ipsilateral/contralateral responses in the precontracted APB was determined by stimulation intensity up to 100% stimulator output (or as high as the participant did not experience the stimulation as too uncomfortable).
In TD subjects, both hemispheres were assessed, contralateral amplitudes and latencies recorded, and rMT of the corresponding contralateral APBs defined.
Experimental Paradigm
To assess the effect of the MI, the following experiment was performed. 16 Two separate conditions (with mirror and without mirror) each consisting of a unimanual and a bimanual task (Figure 1) were completed. During the mirror condition the affected/nondominant hand was placed within a mirror box (Reflex Pain Management Ltd). The movement task consisted in a paced hand opening and closing. Participants either moved the unaffected/dominant hand (unimanual task, Figure 1c) or both hands simultaneously (bimanual task, Figure 1d). In the mirror condition, the mirror reflection of the unaffected/dominant hand was observed. In the nonmirror condition, the unaffected/dominant hand (unimanual task, Figure 1a) or the affected/nondominant hand (bimanual task, Figure 1b) were observed. The 4 parts were performed in a random sequence. They were performed using a block design (alternating 30-second periods of active and inactive blocks). During active blocks participants opened and closed their hands whereas during inactive blocks participants held their hands still. An auditory metronome (1 Hz cadence, 500 ms duration; 880 Hz; Audacity 2.0.0, Linux) was used to pace movements. Subjects were instructed to pace one complete open-close cycle of the hand to each beat of the metronome. During inactive blocks, a different tone was provided by the auditory metronome (440 Hz). The HS of the motor cortex of the hemisphere projecting to the affected (nondominant) hand was stimulated with an intensity of 120% of rMT. TMS pulses were delivered every 5 seconds. Consequently, 6 stimulations were applied for each block, which amounted to 24 stimulations for each of the 4 parts. Opening and closing of the hand influences EMG activity. Therefore, TMS pulses were triggered by the movements. The Labview program “conditional triggering” 11 feature was used to deliver TMS stimuli only when the APB showed appropriate activation. The activation of the APB was defined as the root mean square (time window = 100 ms) of EMG activity between 0.025 and 0.3 mV at baseline. The onset of the rest and active blocks were announced verbally and the examiner controlled task performance visually.
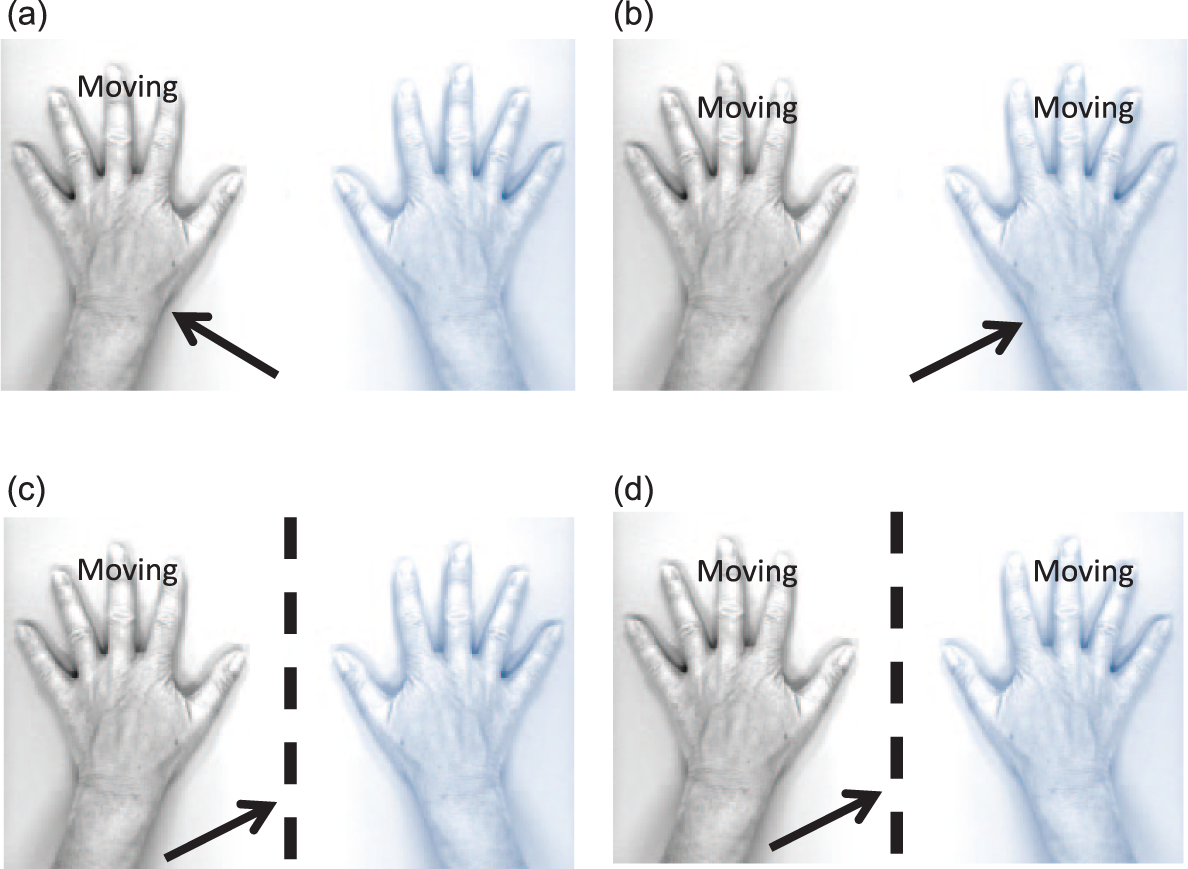
Illustration of the mirror paradigm used in the study. The affected/nondominant hand is illustrated in blue. The arrow illustrates the direction of gaze. The dotted line represents the mirror. The movement task consisted in a paced hand opening and closing. Participants either moved the unaffected/dominant hand (unimanual task, Figures 1c and 1a) or both hands simultaneously (bimanual task, Figures 1b and 1d). In the nonmirror condition, the unaffected/dominant hand (unimanual task, Figure 1a) or the affected/nondominant hand (bimanual task, Figure 1b) were observed. In the mirror condition, the mirror reflection of the unaffected/dominant hand was observed (Figures 1c and 1d).
Data Processing and Statistical Analysis
EMG signals were stored for offline-analysis. A “playback” Labview program 11 was used to review waveforms of all measurements. EMG signals were visually examined. We excluded high misleading amplitudes due to technical artifacts. Mean MEP amplitudes, mean MEP latencies and rMT were used as descriptive data. Since individual values of MEP amplitudes were not normally distributed, they were transformed into the natural logarithm. 17 Mean MEP amplitude of the transformed amplitudes (log MEP amplitude) was calculated for each participant in order to normalize the distribution. 18
In order to assess how Log MEP amplitudes were influenced by covariants they were compared using repeated-measures analysis of variance (ANOVA) with 3 within-subject factors: “muscle” (APB or FDS), “task” (unimanual or bimanual), and “condition” (mirror or nonmirror). Functional assessments provided descriptive data regarding hand function and activity level. The results of each functional assessment were correlated with an individual ratio (MEP amplitude in mirror condition)/(MEP amplitude in nonmirror condition). A 2-tailed Spearman correlation was used to correlate the functional data with the effect of mirror visual feedback in the unimanual as well as the bimanual task. Statistical analysis was performed using SPSS version 19 (IBM Corp, Armonk, NY, USA).
Results
Participants
Descriptive data on baseline characteristics and functional measurements are provided in Table 1. Twenty subjects participated in the study (9 girls and 11 boys, mean age 13.05 years, range 8-20 years). Twelve subjects were diagnosed with hemiparesis and 8 were TD controls. Except for 1 subject (subject 9, who suffered from an arterial ischemic stroke within the vascular territory of the medial cerebral artery at 5 years of age) all patients were diagnosed with congenital hemiparesis.
Characteristics of Study Participants.
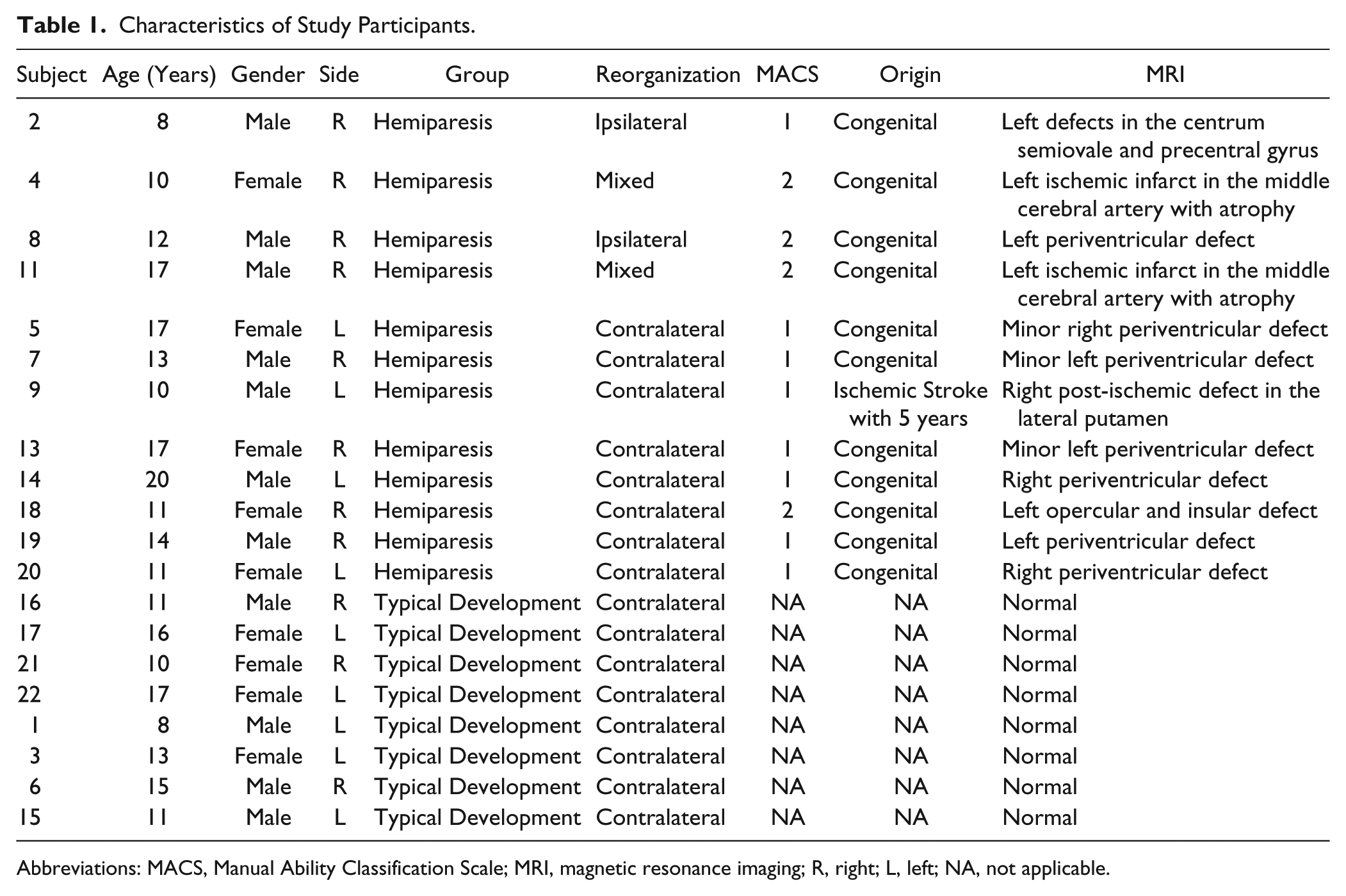
Abbreviations: MACS, Manual Ability Classification Scale; MRI, magnetic resonance imaging; R, right; L, left; NA, not applicable.
Functional Assessments
There was a significant difference in the quality of unilateral hand movements in the MA (
Cortical Reorganization and Effect of Covariants
Based on TMS, 2 subjects with hemiparesis showed ipsilateral corticospinal reorganization (subjects 2 and 8), 2 subjects showed mixed corticospinal reorganization (subjects 4 and 11) and 8 subjects with hemiparesis showed contralateral reorganization (subjects 5, 7, 9, 13, 14, 18, 19, 20). As expected TD subjects showed contralateral cortical reorganization.
The effects of the factors “task,” “condition,” and “muscle” did not differ between patients with contralateral reorganization and TD controls in the active or inactive blocks. Therefore the repeated-measures ANOVA were performed for 3 distinct groups: (1) all participants, (2) participants with ipsilateral/mixed reorganization, and (3) participants with contralateral reorganization (patients with hemiparesis and TD controls). Active and inactive blocks were analyzed separately for each covariant. Table 2 provides an overview of the statistical analysis and Figure 2 illustrates the averaged logMEP amplitudes in each experimental condition. A detailed overview of the logMEP amplitudes in the different conditions is presented in the Supplemental Material.
Effects of Motor Task, Mirror Condition, and Muscle on log MEP Amplitudes.
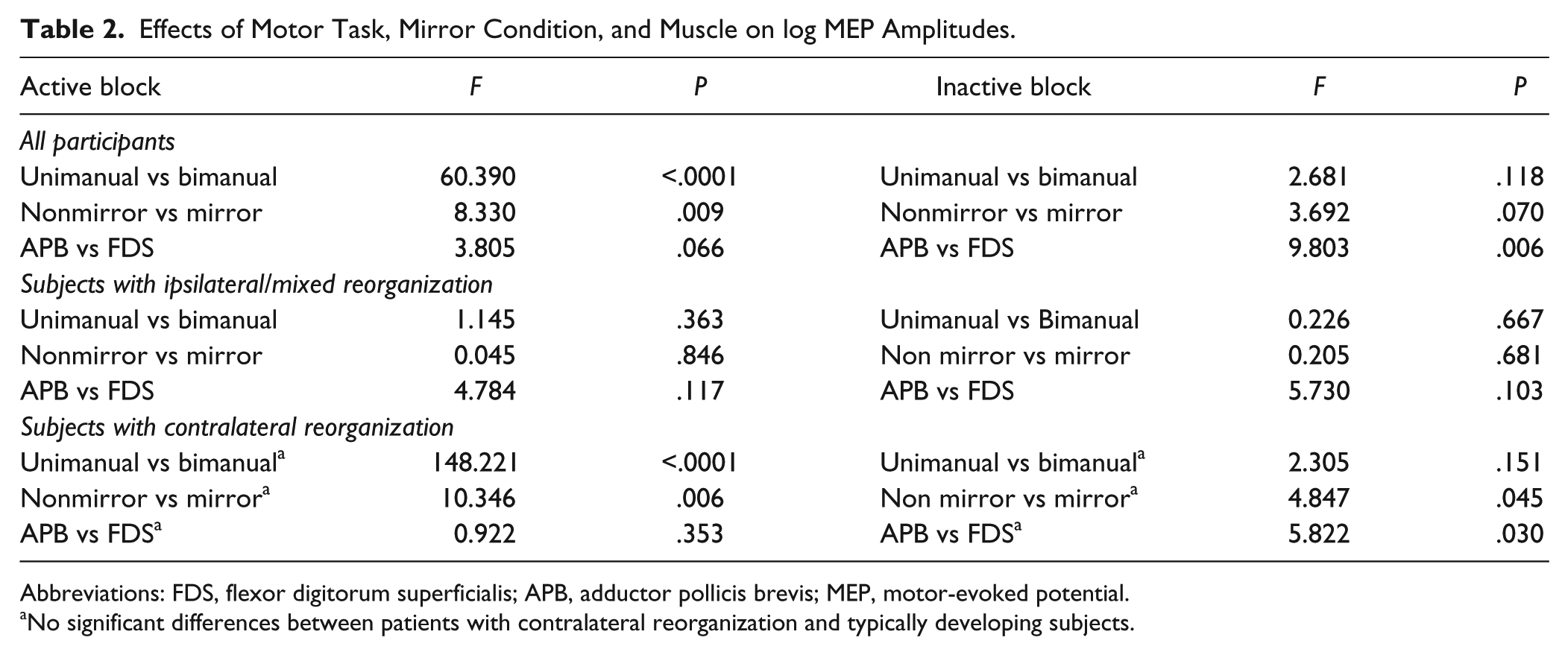
Abbreviations: FDS, flexor digitorum superficialis; APB, adductor pollicis brevis; MEP, motor-evoked potential.
No significant differences between patients with contralateral reorganization and typically developing subjects.
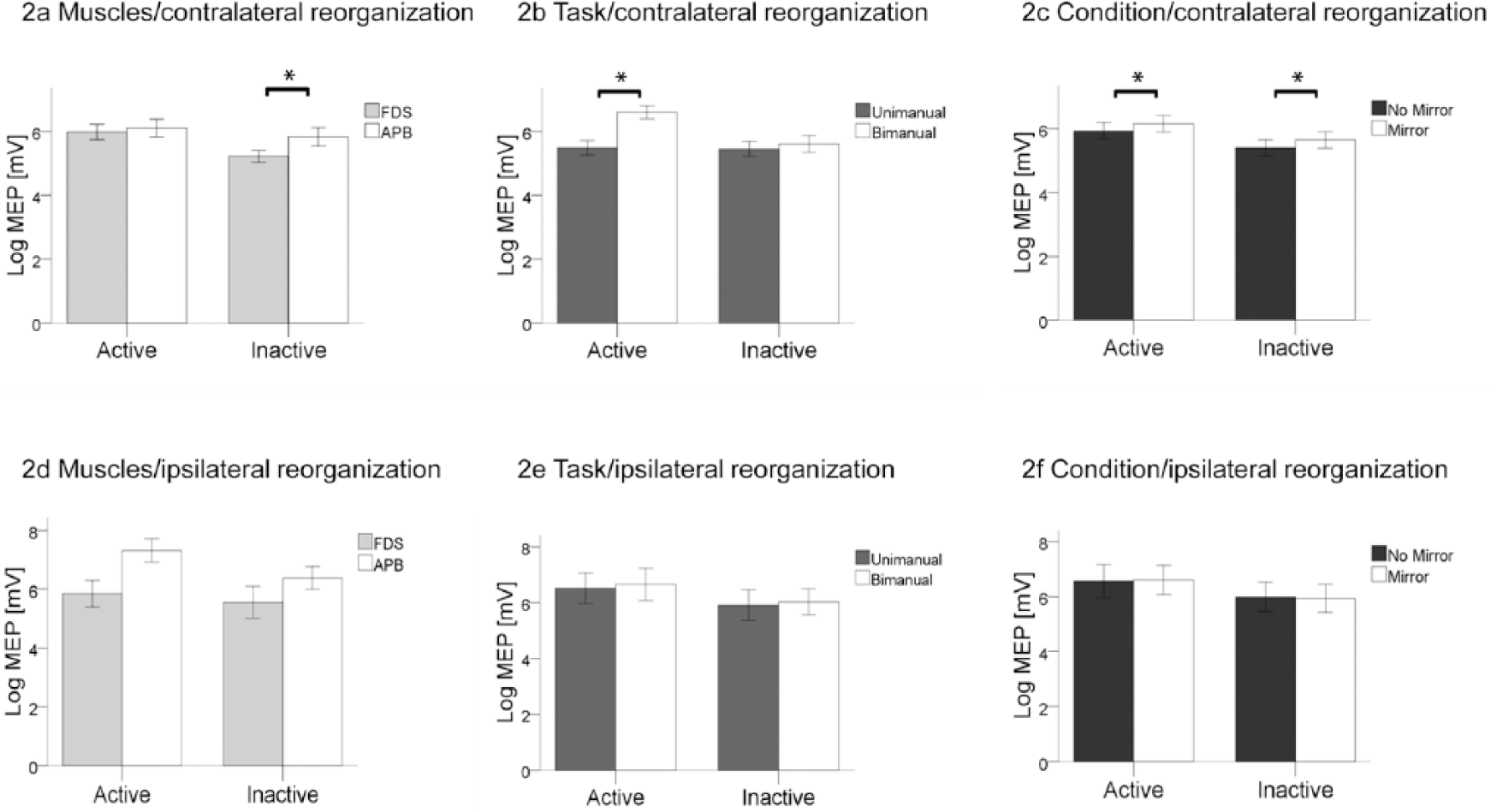
Averaged log MEP amplitudes according to covariants during active and inactive blocks in subjects with contralateral reorganization and subjects with ipsilateral/mixed reorganization. *
When
In participants with
In participants with
Figure 3 illustrates the effect of the mirror in TD subjects, patients with hemiparesis and contralateral reorganization, and patients with hemiparesis and ipsilateral/mixed reorganization in the active condition. Individual changes of log MEP amplitudes due to the MI are available in the Supplemental Material.
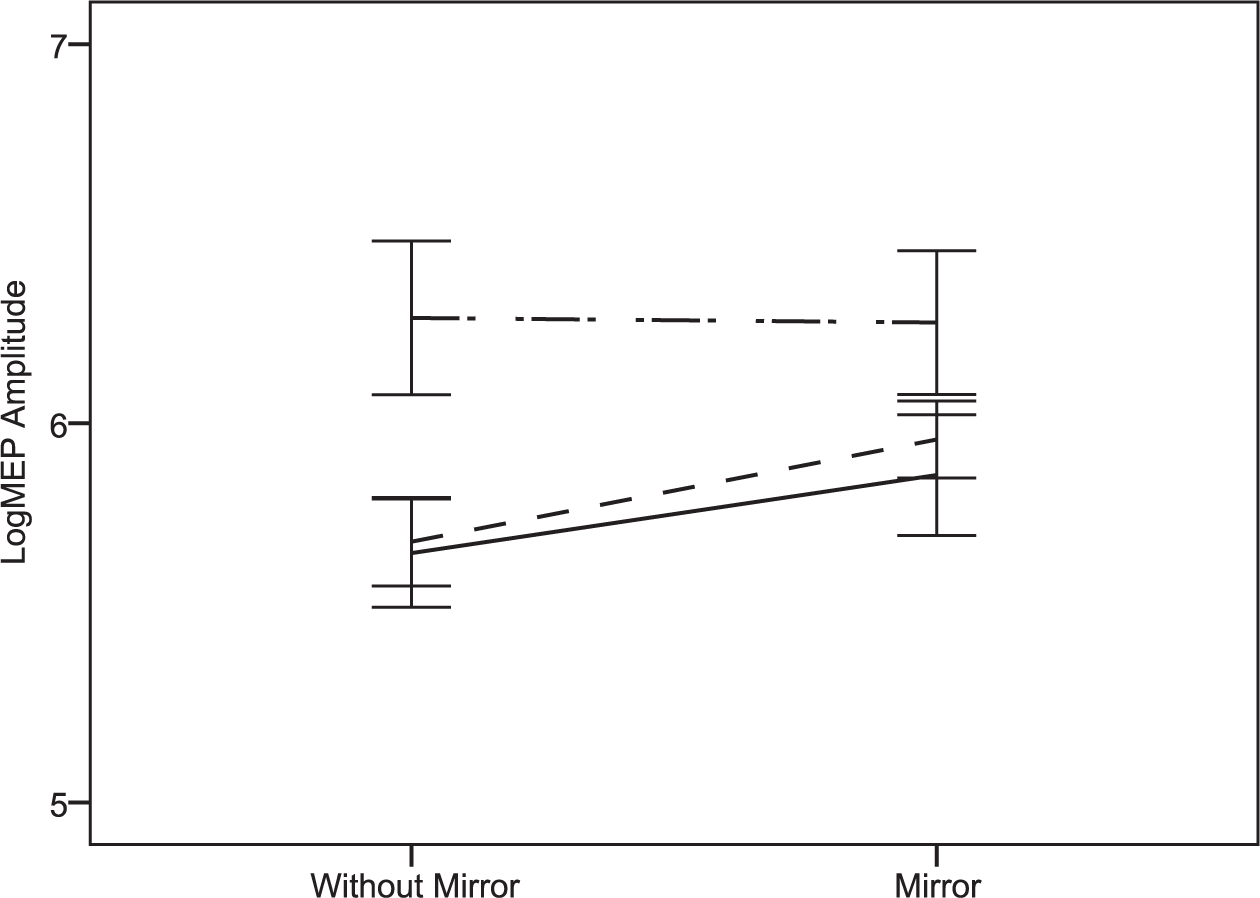
Figure 3 shows the effect of mirror illusion in typically developing (TD) subjects (n = 8), patients with hemiparesis and contralateral reorganization (n = 8) and patients with hemiparesis and ipsilateral/mixed reorganization (n = 4) during active blocks. The black line represents patients with contralateral reorganization, the dashed line represents TD subjects with contralateral reorganization and the dashed/dotted line represents patients with hemiparesis and ipsilateral reorganization. The bars represent 1 standard error.
Correlations
In the active blocks, there were no significant correlations between functional measures and MEP ratios of the FDS and APB during unimanual or bimanual tasks. In inactive blocks, there was a significant correlation between the FDS ratio during unimanual tasks and palmar grip strength (ρ = 0.047,
Discussion
During active hand movements, M1 excitability in the hemisphere projecting to the paretic hand (nondominant hand in TD subjects) increases with MI in children. Various studies with adult subjects have been performed previously and studied the direct effect of the MI using TMS.7,18-24 Similarly to the present study, most previous studies showed that MI has a direct effect on M1 excitability in the hemisphere projecting to the paretic hand, despite considerable differences in methods (tasks, viewing conditions) and participants.7,18-23 Compared with other studies22,23,25 the observed effect on MEP amplitudes in our study was rather small, albeit significant, under certain conditions. Since certain of our participants had very limited hand function, we chose an easy hand opening and closing task, whereas in other studies the participants performed flexion-extension of wrist19,20,22 as well as isolated finger/thumb movements.21,23,24 Adult studies included healthy subjects7,19-21,24 and patients diagnosed with hemiparesis due to stroke.22,23 Two studies showed that the effect of the MI was more prominent when it was combined with motor imagery.23,25 Only one study performed with healthy adults showed no effect of the MI on M1 excitability. 25
We investigated whether the effect of the MI on cortical excitability differed in hemiparetic patients with different reorganization patterns. When subjects with ipsilateral/mixed cortical reorganization were studied separately, we did not detect an increase in M1 excitability due to the mirror. Hemiparetic children with differing corticospinal tract organizations respond differently to treatments that improve hand function. 26 Regardless of corticospinal tract organization sensory tracts are contralaterally organized in hemiparetic children. 27 This might explain why TD participants and participants with contralateral corticospinal tract organization (sensory and motor tracts in the same hemisphere) showed comparable responses to MI, while participants with ipsilateral/mixed reorganizations did not (sensory and motor tracts in different hemispheres). This may lead to differences in sensorimotor integration, especially when induced by mismatches associated with MI. We are not aware of any other study that assessed the effect of the MI in patients with different types of cortical reorganization as seen following unilateral brain lesions during early brain development.
In most previous TMS studies on the effect of MI in adults, participants performed unimanual movements only, whereas in the present study the participants performed unimanual and bimanual movements. We demonstrated that the type of motor task (unimanual vs bimanual) had a large influence on cortical excitability, which had not been addressed previously in TMS studies. One fMRI study in adults with stroke, using the same experimental paradigm as ours, found no significant activation induced by the MI for the unimanual task, while significant activations in the precuneus and posterior cingulate cortex were found for the bimanual task. 16 These results differ from a study of MT in adults with stroke, reporting bimanual movements as less effective. 28 The effect of differing motor tasks during MT deserves more thorough exploration.
MI has been suggested to act by dampening the interhemispheric inhibition from the healthy to the damaged hemisphere. Only one study assessed interhemispheric inhibition before and after MT in adults with stroke. There was no association between M1 excitability and the amount of interhemispheric inhibition. 29 The study did not use TMS during MI, so the direct effect of MI on interhemispheric inhibition remains unknown. MI has been postulated to “delude” the brain by manipulating visual feedback. Since sensory afferences (including visual afferences) are known to influence motor pathways, manipulating sensory feedback is suspected to modify motor efferences. In our study effects of MI on M1 excitability were stronger when participants performed a motor task. To understand the interactions between movement and observation, plain observation conditions are necessary. We did not include a condition where subjects looked at a moving limb without moving their own hand. A previous study using an action-observation–based approach to investigate the effect of movement observation on motor performance, suggested that MT-based action-observation contributes to motor learning. 30
Our study was not designed to investigate the therapeutic effect of MT. It also does not allow us to infer whether the increase of M1 excitability induced by MI is therapeutically relevant. Adding to the studies investigating the direct effect of the MI on cortical excitability 2 studies examined the effect of MT (the repeated use of the MI for therapeutic purposes) on the cortical excitability of M1 in adults with stroke.7,31 It was shown that the training enhanced the excitatory function of the corticospinal pathway projecting to the paretic hand. MT increased activation of the affected side and reestablished the hemispheric balance. The relation between the effect of MI on M1 excitability and motor learning due to MT has been poorly investigated, since no MT trials have included measurements of the effect of MI on M1 excitability.
We are not aware of any study that has examined the effect of MT on neuronal activity in children. If no similar evidence has been reported up till now for the childhood population, there is indirect substantiation that similar mechanisms may be at play. In the pilot study of MT in hemiparetic children, strength testing performed on the paretic hand behind the mirror during a bimanual grasp task significantly increased grasp strength by 15%, suggesting an immediate effect of the MI on motor cortical recruitment. 5 In a study that examined the effects of MI on muscular activation in children with spastic cerebral palsy during bilateral symmetric upper limb movements, the mirror lead to shorter eccentric and concentric activity in the muscles of the impaired arm, suggesting improved motor control during interlimb coupling. 31 The authors suggested improved ipsilateral M1 excitability as a potential mediator to the amelioration in motor control.
In the present study, we did not find a correlation between the effect of the mirror on log MEP amplitudes and hand function. We therefore hypothesize that functional factors and residual deficits do not significantly influence the effect of mirror visual feedback on M1 excitability if the physiological contralateral corticospinal organization pattern is preserved. With regard to MT this suggests that MT can influence cortical excitability and might lead to improved hand function irrespective of the severity of motor impairment.
Our study reproduced findings from adult studies showing a positive effect of MI on M1 excitability in a pediatric population. Various questions regarding the neurophysiologic effects of MI and MT in children require further exploration. Future studies should investigate the direct effects of MI on interhemispheric inhibition and M1 excitability using paired pulsed TMS, explore the mechanisms underlying MI and M1 excitability using action-observation–based tasks compared with motor tasks, and study the relation between effects of MI on M1 excitability and motor learning in trials using direct measurements of M1 excitability during MI.
Limitations
Generalization of our results should be taken with caution due to limitations. The major limitation was the size of the study population and the heterogeneity of motor function, lesion type and location in subjects with hemiparesis. Incomplete age matching limits the comparison between TD participants and participants with hemiparesis. Especially the size of the subgroup with ipsilateral/mixed reorganization is small. Because of the small sample size, participants with ipsilateral and mixed cortical reorganization were summarized in one group. However, participants with mixed projections to the affected hand are not identical to children with ipsilateral projections. Mixed projections could result in one, the other, or both patterns depending on the strength of these projections. Therefore, no definite conclusions should be drawn regarding the effect of MI on cortical excitability in patients with ipsilateral and mixed reorganization. As in the present study we used single pulse TMS and interhemispheric inhibition was not assessed, we were not able to assess the role of interhemispheric inhibition on cortical excitability modulation due to the MI. The present study was designed to determine the neurophysiological changes underlying MI. It does not allow any conclusions on the behavioral benefits of MI.
Conclusions
The MI increases cortical excitability in M1 on the hemisphere projecting to the moving hand in children and adolescents with contralateral cortical reorganization but only during active movement. The effect of the MI can be observed in patients with hemiparesis and in TD subjects. No definite conclusion can be drawn regarding the effect of the MI on cortical excitability in patients with hemiparesis and ipsilateral cortical reorganization even though our results suggest that effects of MT depends on the type of corticospinal organization.
The cortical modulation obtained through the MI suggests that MT may prove an efficient approach in children with hemiparesis, or at least for the majority who present a contralateral motor organization. In order to assess the effect of MT per se on cortical excitability in children with hemiparesis larger studies are required to assess patients pre- and postintervention using TMS.
Footnotes
Acknowledgements
We are thankful to all participating children and their parents.
Authors’ Note
Sebastian Grunt and Christopher J. Newman made equal contributions to the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Johanna Durmüller Foundation, Switzerland, the Foundation Vinetum, Switzerland, and the Foundation Batzebär.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
