Abstract
Objectives. To determine retention of treatment outcomes at 52 weeks following a matched-pairs randomized comparison trial of constraint-induced movement therapy (CIMT) and bimanual training (BIM). Methods. Sixty-four children (mean age = 10.2 ± 2.7 years, 52% male) were included. The Melbourne Assessment of Unilateral Upper Limb Function (MUUL), Assisting Hand Assessment (AHA), and Canadian Occupational Performance Measure (COPM) were the primary outcome measures. Evaluations were at baseline and at 26 and 52 weeks. Results. There were no baseline differences between groups on any measure. No significant differences were found between groups on primary outcomes at 52 weeks. Both groups retained the significant gains made from baseline to 26 weeks at the 1-year follow-up assessment for unimanual capacity on the MUUL, for bimanual performance on the AHA, and on the COPM. Conclusion. Intensive unimanual and bimanual training can both lead to long-term significant improvements in unimanual capacity, bimanual performance, and individualized outcomes. Gains established at 26 weeks were maintained at 12 months postintervention despite most children receiving no direct therapy during that time.
Cerebral palsy (CP) is the leading cause of childhood physical disability with an incidence of 1 in 500 live births. 1 Hemiplegia (unilateral CP) accounts for 35% of these children, and the upper limb (UL) is usually more affected than the leg. 2 Intensive UL training approaches such as constraint-induced movement therapy (CIMT) and bimanual training (BIM) have received increasing attention as methods of addressing UL dysfunction for children with unilateral CP. 3 Each approach involves intensive practice of UL skills. CIMT constrains the unimpaired hand and targets unimanual task-related practice with shaping movements and repetition. BIM focuses on bimanual skills through functional and developmentally appropriate fine and gross motor activities requiring use of 2 hands 4 to improve bimanual coordination.
A systematic review and meta-analysis of all UL nonsurgical therapy interventions identified a small number of randomized controlled trials of CIMT 5,6 that demonstrated immediate gains in the amount of use (effect size [ES] = 0.54) and slightly improved movement efficiency (ES = 0.10) of the impaired UL when compared with a regular care/control group. 3 A controlled clinical trial comparing CIMT with conventional care demonstrated improved bimanual performance when employing the affected UL (ES = 1.16). 7 One small trial of bimanual training (Hand Arm Bimanual Intensive Training [HABIT]) demonstrated small immediate gains in bimanual performance (ES = 0.16) when compared with conventional therapy. 8 A combined model of CIMT and bimanual task-specific training (mCIMT-BiT) was compared with usual care of the same duration, and the study found that mCIMT-BiT demonstrated improved bimanual performance (ES = 0.43), manual skills (ES = 1.01), perceived occupational performance (ES = 1.32), and quality of movement of the impaired UL (ES = 0.40). 9 The total dosage of intervention has varied between studies ranging from 60 to 120 hours, delivered either intensively for up to 6 hours a day for 10 to 21 days 5,6,8 or using a more distributed model of 2 to 3 hours a day over 2 months. 7,9 However, for CIMT, BIM training, and mCIMT-BiT, it was unclear whether immediate gains were retained as only 3 studies assessed outcomes to 6 months. 5-7 No study has investigated outcomes beyond 6 months for retention of treatment effects.
The INCITE trial (National Health and Medical Research Council of Australia) was a single-blind matched-pairs randomized trial that directly compared equal dosages of CIMT and BIM training for 64 children with unilateral CP. On primary outcome measures (Melbourne Assessment of Unilateral Upper Limb Function [MUUL] and Assisting Hand Assessment [AHA]), there were no differences between groups immediately following intervention of 60 hours over 2 weeks. 10,11 Overall, the results for each group reflected the mode of training. The CIMT group demonstrated significantly improved unimanual capacity (ie, how the impaired hand/arm is used when asked) for both quality (MUUL) and efficiency (Jebsen Taylor Test of Hand Function [JTTHF]) of movement. The BIM group demonstrated improved bimanual performance (AHA) but no change in quality of UL movement (MUUL). Both groups had equal gains in occupational performance on the Canadian Occupational Performance Measure (COPM) and in personal care on the Assessment of Life Habits (LIFE-H). 10,11 We now evaluate for subsequent retention of treatment effects at 52 weeks postintervention.
Method
Participants
Methods used in the INCITE trial were reported in the study protocol. 12 The study was approved by the Ethics in Human Research Committees at The Royal Children’s Hospital, Melbourne; La Trobe University; The Royal Children’s Hospital and Health Services District, Brisbane; and The University of Queensland.
Children were included if they had unilateral CP, were aged 5 to 16 years, and had a Modified Ashworth Scale (MAS) ≤ 3 at the distal UL. Ineligibility criteria included dystonia, contracture (MAS > 3), previous UL orthopedic surgery, or either serial casting or botulinum toxin injections in the UL within 6 months.
Design
The INCITE trial used a matched-pairs design with children matched according to age (12-month age bands), gender, side of hemiplegia, and function based on unimanual capacity of the impaired UL (MUUL scores within 10%) to minimize baseline differences that have been problematic in previous UL studies. 8 Once matched, children were randomized within pairs using a computer-generated list of random numbers in concealed envelopes opened by nonstudy personnel.
Sample size of 26 participants in each group (total sample of 52) was calculated to have at least 80% power to detect a 7-unit difference (10% of the anticipated control group mean at baseline) in MUUL score between groups (2-sided test at .05 significance). 12
Interventions
Each intervention was delivered in groups of 9 to 13 children for 6 hours per day for 2 weeks (10 days) using a day camp model. In total, 6 camps were run in Melbourne and Brisbane, Australia. Each pair of camps (BIM training and CIMT) were age grouped to ensure the programs were developmentally tailored. Camps were community based and used a novel circus theme to optimize children’s motivation and engagement. A goal directed, activity-based framework was adopted for both interventions using principles of motor learning in specific task practice by fostering problem solving and modification of the environment to achieve goal attainment. The daily program involved 1 hour of fine motor training, training on 3 functional goals identified by children/caregivers, 2 hours of circus training, gross UL games, and 30 minutes of group debriefing. The BIM training group was conducted first, and then tasks were modified for the CIMT group to accommodate the unimanual nature of the intervention. Each group received the same dosage and overall content of intervention, delivered in the same environment. Participants in the CIMT group wore a tailor-made glove on their unimpaired limb while attending the day camp, which was only removed for toileting for no longer than 15 minutes of each daily session. For the BIM training group, explicit instructions on how each hand should be used in each activity were provided, similar to the strategy adopted by HABIT. 8 For both CIMT and BIM training, the focus was on the completion of whole tasks.
The primary activity outcomes, the MUUL to measure quality of movement of the impaired UL and the AHA to measure bimanual performance, are valid and reliable assessments for children with unilateral CP and are described in detail elsewhere. 13 The MUUL was reported as a percentage, with higher scores reflecting greater quality of UL movement. Previous reliability studies have found a change of 12% for intrarater reliability and 14% for interrater reliability, representing a clinically significant effect. 14 Subsequent reliability studies have yielded smaller standard error of measurement, suggesting the smallest detectible difference may be lower than originally reported. 15,16 However, all studies differed in terms of the study populations, necessitating establishment of intrarater reliability for the current study. Intrarater reliability established for the INCITE trial was high (intraclass correlation coefficient = .93), with a standard error of measurement of 2.7% and smallest detectible difference of 7.4%. 10 Therefore, a change of greater than 7.4% on the MUUL can be considered to represent true change beyond measurement error. The AHA was reported in logits (log odds probability units) using Winsteps version 3.65.0 (copyright 2006 John M. Linacre) with item calibrations anchored according to their difficulty level. 17 The logit scale was transformed by test developers to a more easily interpretable 1 to 100 logit scale, with higher scores reflecting greater ability to use the impaired UL in bimanual activities. A change of 4 raw score points is considered clinically significant. 18 All assessments were performed by experienced and trained occupational therapists and physiotherapists who were aware of group allocation. However, the primary assessments (AHA and MUUL) were videoed and scored in random order by trained occupational therapists masked to group allocation. A secondary activity outcome measure, the JTTHF measured movement efficiency of the impaired UL on 6 timed tasks. The JTTHF measures a different aspect of unimanual capacity (movement efficiency), and it was chosen as a secondary measure because of the lack of evidence of validity and reliability for children with unilateral CP. 13 It is unclear as to what degree of change could be considered to be clinically meaningful.
Results of Baseline and Follow-Up Assessments for CIMT and BIM Groups
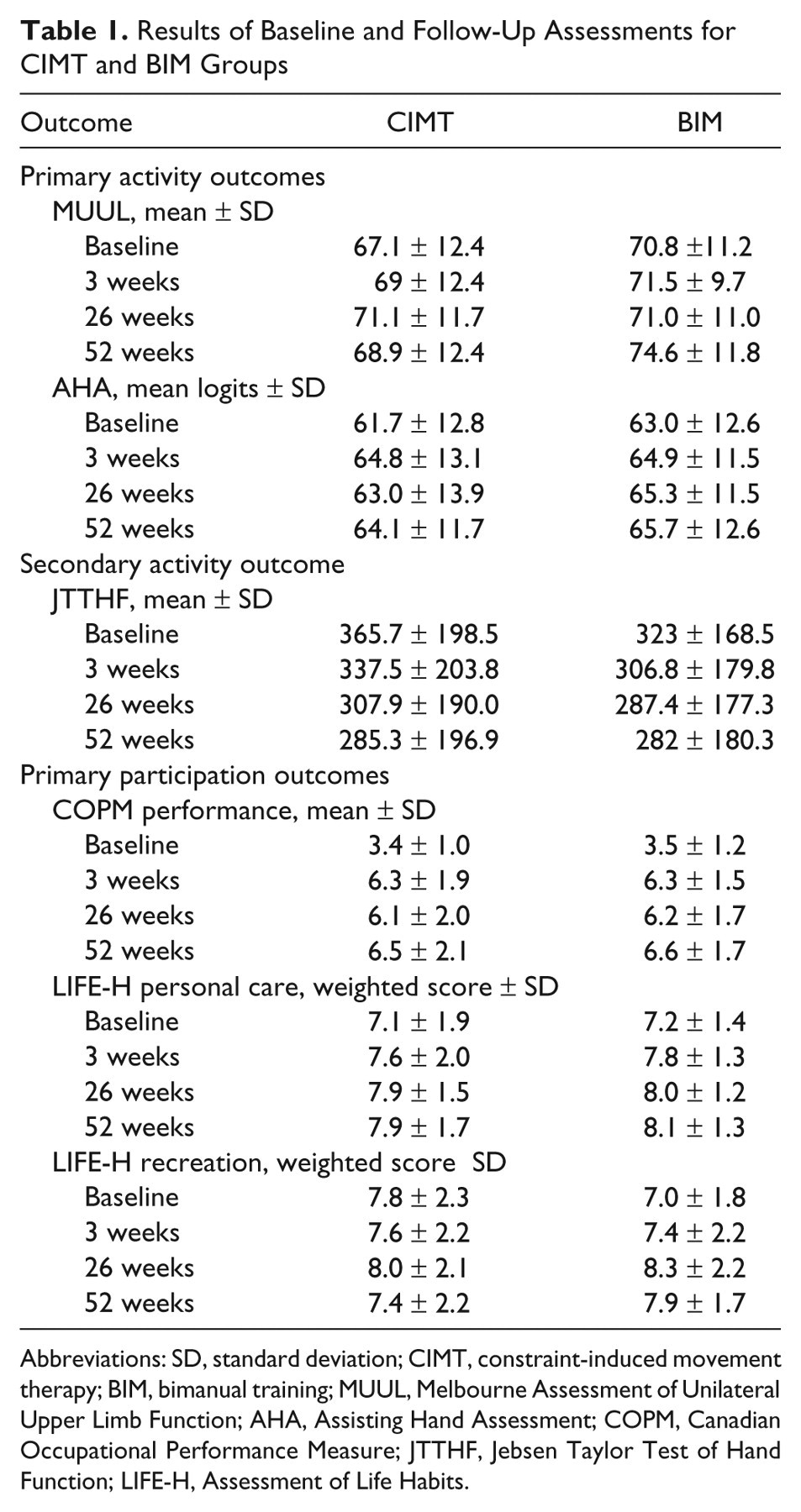
Abbreviations: SD, standard deviation; CIMT, constraint-induced movement therapy; BIM, bimanual training; MUUL, Melbourne Assessment of Unilateral Upper Limb Function; AHA, Assisting Hand Assessment; COPM, Canadian Occupational Performance Measure; JTTHF, Jebsen Taylor Test of Hand Function; LIFE-H, Assessment of Life Habits.
The COPM was a secondary outcome measure for individualized outcomes and is a standardized, individualized, client-centered assessment evaluating self-perception of occupational performance of identified goals. The COPM has good evidence of construct, content, and criterion validity and demonstrated responsiveness to change in pediatric clinical trials. 6,8 A change of 2 points is considered clinically significant. 19 Children identified 3 to 5 goals and rated their perceived level of performance and satisfaction on a 10-point scale. Two domains of the LIFE-H (personal care and recreation), which demonstrated change at 3 or 26 weeks, were included in the present analysis. Further details are reported in the primary outcome articles. 10,11
Concurrent therapies (eg, occupational therapy or physiotherapy) with or without spasticity management (eg, intramuscular BoNT-A injections) of the upper or lower limbs received as standard care over the duration of the intervention and follow-up period were recorded.
Statistical Analysis
Analyses were on an intention-to-treat basis according to CONSORT guidelines using STATA 10. 20 A significance level of .05 was used for the primary outcomes (MUUL and AHA). The significance level was adjusted to .01 to account for multiple comparisons of secondary outcomes (JTTHF, COPM, and LIFE-H). Continuous data were compared between and within groups by fitting a regression model using generalized estimating equations (GEEs) to baseline, 26- and 52-week measurements with an interaction term between intervention group, and 3-level factor indicating time of measurement. Matching characteristics of age, gender, and side of hemiplegia were entered as covariates. An exchangeable working correlation matrix for the repeat measurements on participants was used. 21 Comparison of estimated mean difference (EMD) at 26 and 52 weeks using GEEs determined retention of treatment effects, with a reduction in the EMD indicating no retention, and maintenance or further increases in gains regarded as retention of effects.
Results
Sixty-four children (32 matched pairs) were randomized into 2 treatment groups delivered over 6 camps (see Figure 1 for trial profile). Sixty-two children received their allocated intervention. There was 1 dropout on day 2 of the camp from the BIM group, due to preexisting emotional and behavioral difficulties; thus, there was 100% compliance in the CIMT group and 97% in the BIM group. Ninety-four percent of the sample (n = 3 CIMT; n = 1 BIM lost to follow-up) were retained at 6 months and 92% (n = 2 CIMT; n = 3 BIM lost to follow-up) at 12 months. Six children (CIMT n = 4; BIM n = 2) received BoNT-A injections to the UL and concurrent therapy between 26 and 52 weeks as part of their standard care. No other variations from preintervention therapy levels were recorded. There were no adverse events for either group to report.
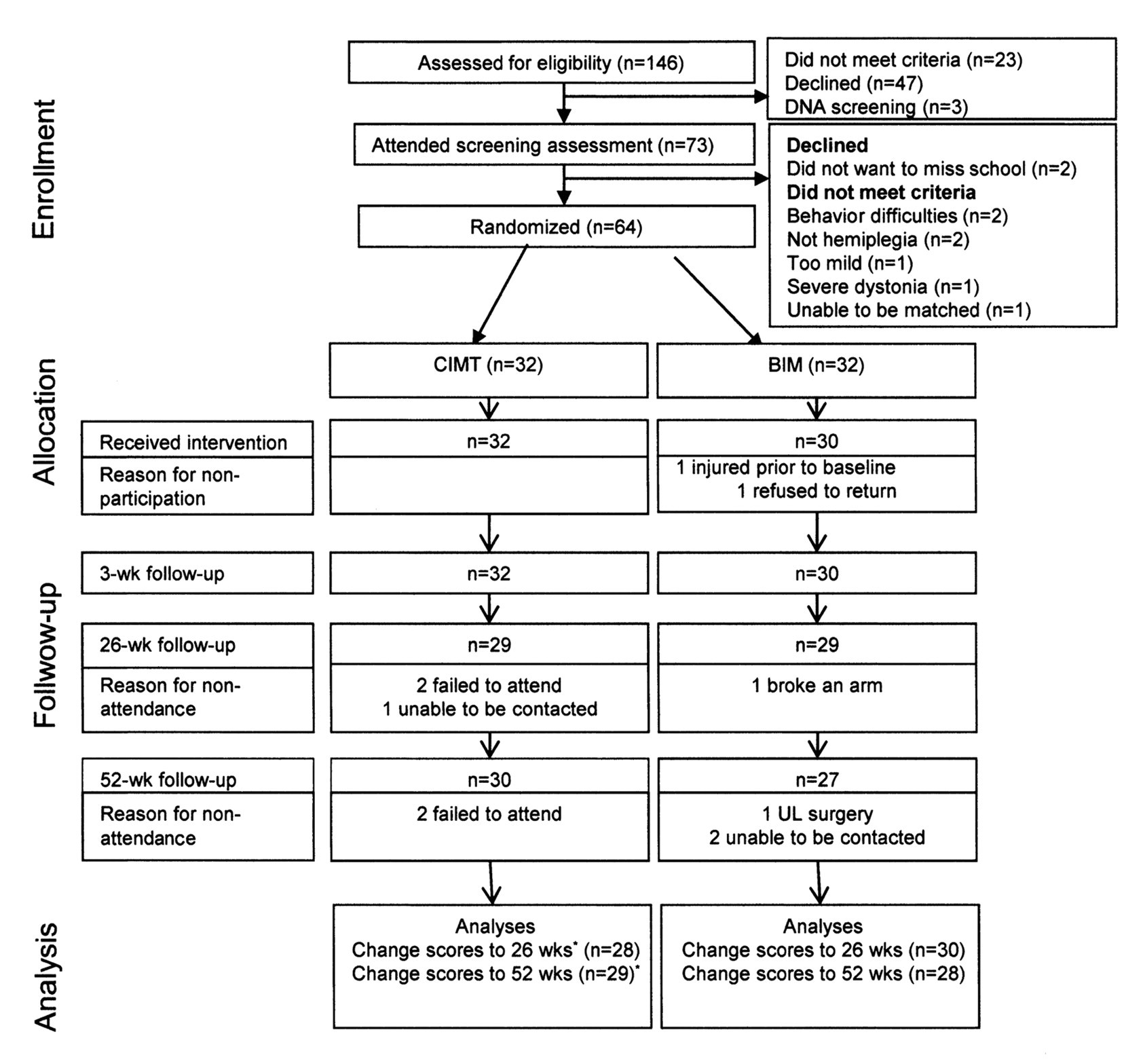
Trial profile following CONSORT guidelines. Abbreviations: UL, upper limb; wk, week; CIMT, constraint-induced movement therapy; BIM, bimanual training.
Baseline and follow-up scores are reported in Table 1. There were no between-group differences except for unimanual capacity (MUUL) at 26 weeks favoring CIMT (Table 2; EMD = 4.1, 95% confidence interval [CI] = 1.3-7.0, P = .005); however, this between group difference was not maintained at 52 weeks (EMD = −1.9, 95% CI = −4.0-0.1; P = .06). The CIMT group demonstrated significant gains in quality of movement (MUUL) and movement efficiency (JTTHF) of the impaired UL from baseline to 26 weeks (Figure 2). At 52 weeks, there was retention of treatment effects with a trend to continued improvement in movement efficiency (EMD = 76.9, 95% CI = 53.9-100.0; P < .001). The BIM group continued to demonstrate gains in movement efficiency (JTTHF) from 26 to 52 weeks and was significantly different from baseline (EMD = 40.3, 95% CI = 17.4-63.3; P = .001). Improved bimanual performance (AHA) for the BIM group peaked at 26 weeks, and while there was a small but nonsignificant decline at 52 weeks, change from baseline remained significant (EMD = 2.0, 95% CI = 0.2-3.9; P = .03). The CIMT group demonstrated a trend to improved bimanual performance from 26 to 52 weeks. Both intervention groups retained gains in perceived occupational performance (COPM) at 52 weeks with clinically significant change from baseline (EMD = 3.1, 95% CI = 2.5-3.7, P < .0001, for CIMT and EMD = 3.2, 95% CI = 2.6-3.8, P < .0001, for BIM). Both groups maintained gains in LIFE-H personal care at 52 weeks (EMD = 0.8, 95% CI = 0.3-1.3, P = .002, for CIMT and EMD = 1.0, 95% CI = 0.5-1.5, P < .001, for BIM).
Between-Group and Within-Group Differences at 26 and 52 Weeks and Retention of Effects at 52 Weeks for CIMT and BIM Training Groups
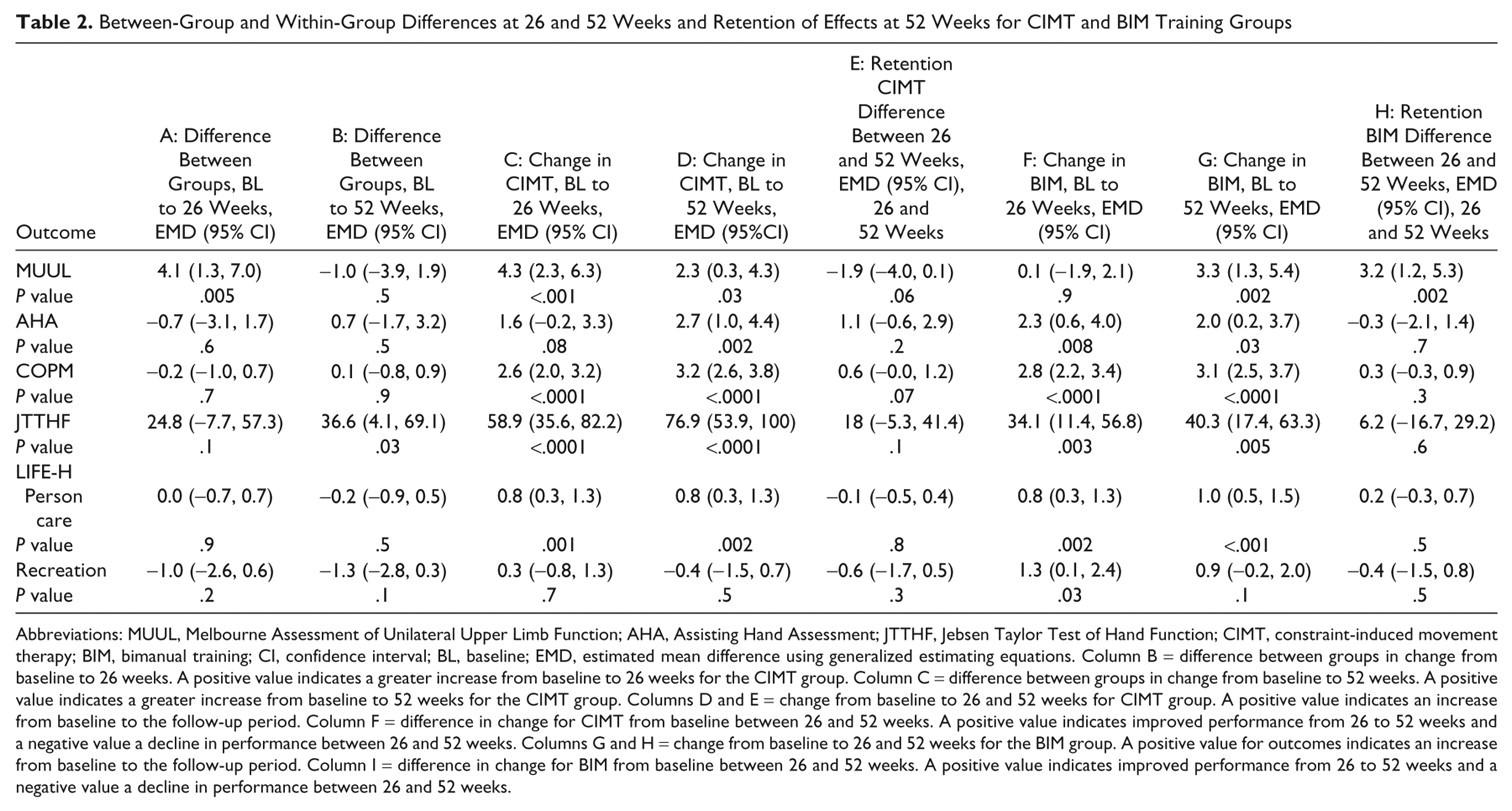
Abbreviations: MUUL, Melbourne Assessment of Unilateral Upper Limb Function; AHA, Assisting Hand Assessment; JTTHF, Jebsen Taylor Test of Hand Function; CIMT, constraint-induced movement therapy; BIM, bimanual training; CI, confidence interval; BL, baseline; EMD, estimated mean difference using generalized estimating equations. Column B = difference between groups in change from baseline to 26 weeks. A positive value indicates a greater increase from baseline to 26 weeks for the CIMT group. Column C = difference between groups in change from baseline to 52 weeks. A positive value indicates a greater increase from baseline to 52 weeks for the CIMT group. Columns D and E = change from baseline to 26 and 52 weeks for CIMT group. A positive value indicates an increase from baseline to the follow-up period. Column F = difference in change for CIMT from baseline between 26 and 52 weeks. A positive value indicates improved performance from 26 to 52 weeks and a negative value a decline in performance between 26 and 52 weeks. Columns G and H = change from baseline to 26 and 52 weeks for the BIM group. A positive value for outcomes indicates an increase from baseline to the follow-up period. Column I = difference in change for BIM from baseline between 26 and 52 weeks. A positive value indicates improved performance from 26 to 52 weeks and a negative value a decline in performance between 26 and 52 weeks.
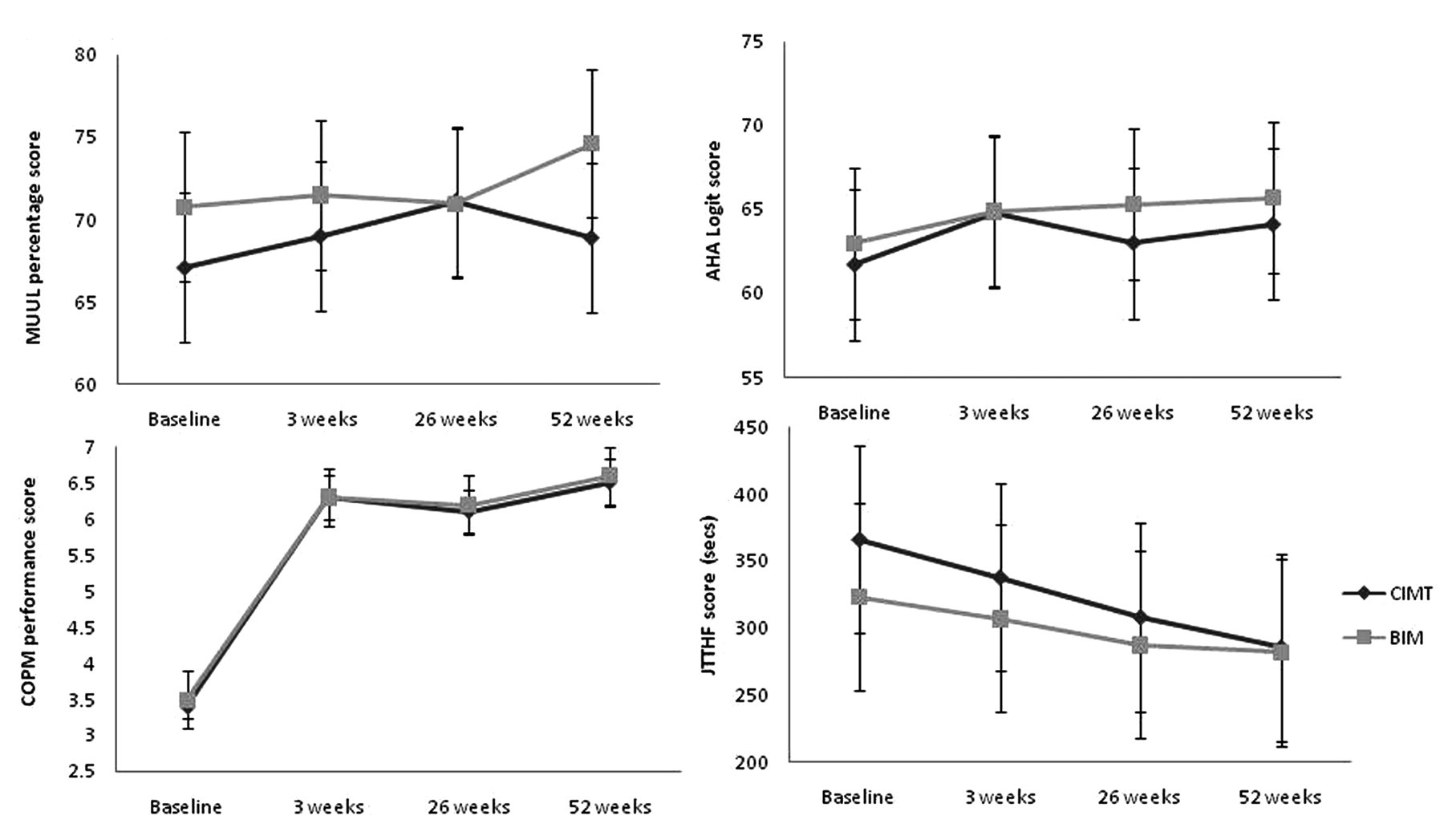
Results for unimanual capacity, bimanual performance, and individualized outcomes for CIMT and BIM training groups. Abbreviations: CIMT, constraint-induced movement therapy; BIM, bimanual training; MUUL, Melbourne Assessment of Unilateral Upper Limb Function; AHA, assisting hand movement; COPM, Canadian Occupational Performance Measure; JTTHF, Jebsen Taylor Test of Hand Function.
Discussion
This study demonstrated retention of treatment effects at 52 weeks for activity and individualized and participation outcomes in a randomized trial comparing CIMT and BIM. Results reported from the primary analysis of the INCITE trial suggested that the full impact of each intervention on activity and individualized and participation outcomes was not immediately apparent, recommending that longer term follow-up of efficacy was necessary. 10,11 This study highlighted that long-term gains in quality and efficiency of movement of the impaired UL, the improved use of the impaired UL as an assisting hand in bimanual activities, and achievement of individualized outcomes can be achieved following a 2-week, 60-hour block of intensive unimanual or bimanual intervention.
Gains made for both quality of UL movement (MUUL) and bimanual performance (AHA), while statistically significant, failed to exceed differences that were determined to be of clinical significance. The MUUL has only been used as an outcome in 1 other randomized trial of combined CIMT and BIM (mCIMT-BiT). 9 Similar changes in MUUL percentage scores following intervention were demonstrated by both mCIMT-BiT and the current study. This might suggest that the MUUL is not sensitive to capturing change in quality of UL movement following intervention as has been suggested in other UL rehabilitation studies, 22 the dosage of intervention was not sufficient, or the intervention effect was not large enough. The MUUL assessment has recently undergone Rasch analysis, with the revised version comprising 4 unidimensional subscales that measure different elements of UL quality of movement (range, accuracy, fluency, and dexterity), 23 which may better detect clinically important changes.
Overall group estimated mean change in bimanual performance on the AHA was modest following either CIMT or BIM. The effect size (ES = 0.22) was greater than the smaller trial of HABIT (ES = 0.15), 3,8 but smaller than the studies of mCIMT-BiT (ES = 0.43) 9 and CIMT (ES = 1.12). 7,24 These differences may reflect the population of children recruited for each study. The studies by Eliasson et al 7 and Aarts et al 9 targeted younger children aged between 18 months and 4 years and between 2.5 and 8 years, respectively, whereas HABIT 8 and the current study had a broader age range up to 16 years. This might suggest that younger rather than older children may achieve greater changes in bimanual performance following intensive UL training. Our secondary analysis of characteristics of best responders in the INCITE trial found that overall 47% of children achieved a clinically significant response for bimanual performance on the AHA. 25 However, age was not found to predict a clinically significant response. These results highlight the heterogeneous nature of the population of children with unilateral CP who exhibit variable responses to intervention, and as yet, definitive predictors of clinically significant outcomes are not clearly understood.
Importantly, for both interventions, any significant gains made by 26 weeks were retained in the longer term at 52 weeks, indicating that intensive unimanual or bimanual UL training programs effect lasting changes in activity and individualized and participation outcomes. It remains unclear whether gains made following CIMT or BIM using a more distributed model of practice would also lead to longer term retention of effects or similar gains. 7,26
One limitation of the current study is the lack of a true control group. This was considered; however, the heterogeneous population of children with unilateral CP and the complications of matching children into 3 groups precluded this option. Furthermore, the primary aim of the study was to determine whether an equal dose of CIMT was superior to BIM. However, given the limited data regarding the natural history of UL development in children with unilateral CP, 27 a control group may have shed additional light on the magnitude of clinically important change following the interventions.
For a 1-year follow-up, we have to consider the natural history of UL development in order to understand the significance of the late findings. Studies suggest that most gains in skill acquisition will occur prior to 7 years, and ongoing improvement or decline beyond that age may be dependent on both severity of impairment and access to ongoing intervention. 27-29 Aspects of cognitive, social, and emotional maturation may have contributed to some of the long-term changes in hand function, but age was not found to be a significant predictor in the regression modeling for any of the motor outcomes.
A combination of CIMT and BIM may yield greater response than either approach alone. 30,31 Rapid immediate improvements in unimanual capacity following CIMT might be further enhanced by subsequent bimanual training. This appears to be supported by the mCIMT-BiT study of combined CIMT and BIM compared with standard care of equal dosage. 9 The optimum combination of approaches, determination of key critical periods to best intervene, and identification of participant characteristics that contribute to greater treatment response need to be further investigated. Either intensive approach will lead to relatively good, long-lasting gains in unimanual capacity, bimanual performance, and individualized outcomes for children with unilateral CP.
Conclusion
In a large single-blind matched-pairs randomized trial, findings suggest that significant gains in unimanual capacity, bimanual performance, and individualized outcomes achieved by 26 weeks’ postintervention are retained at 52 weeks with CIMT and BIM for children with unilateral CP.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: The research was funded by the National Health and Medical Research Council of Australia (NHMRC) for a project grant (INCITE: A randomised trial of novel upper limb rehabilitation in children with congenital hemiplegia NHMRC no. 368550), Dora Lush Postgraduate Scholarship (NHMRC no. 384488) and Career Development Grant (NHMRC no. 473860).
