Abstract
Background. Poststroke dysphagia is associated with considerable morbidity and has high health care cost implications. Objective. To evaluate whether anodal transcranial direct current stimulation (tDCS) over the lesioned hemisphere and cathodal tDCS to the contralateral one during the early stage of rehabilitation can improve poststroke dysphagia. Methods. A total of 40 patients referred to our neurorehabilitation department were randomized to receive anodal tDCS over the damaged hemisphere plus cathodal stimulation over the contralateral one versus sham stimulation during swallowing maneuvers over the course of 10 sessions of treatment. Swallowing function was evaluated before and after stimulation using the Dysphagia Outcome and Severity Scale (DOSS). Results. The percentage of patients who reached various thresholds of improvement was higher in the tDCS group than in the sham group, but the differences were not significant (eg, DOSS score ≥ 20% increase from baseline: 55% in the tDCS group vs 40% in the sham group; P = .53). Among all variables recorded at baseline, only a subgroup of patients without nasogastric tube showed a significantly higher improvement with tDCS treatment versus sham (DOSS score ≥10% and ≥20% from baseline: 64.29% vs 0%, P = .01). Conclusions. In patients with poststroke dysphagia, treatment with dual tDCS in the early phase of rehabilitation does not significantly increase the probability of recovery compared with sham stimulation.
Keywords
Introduction
Dysphagia is a common complication of stroke and is associated with malnutrition, dehydration, and increased duration of hospitalization 1 as well as an increased risk of mortality in the acute and subacute phases.2,3 More than 50% of stroke survivors will experience swallowing dysfunction acutely. 4 Although the majority of them recover swallowing function within 7 days, 5 11% to 13% continue to have dysphagia at 6 months. 6
Early interventions that improve swallowing may have a major impact on clinical outcome, mortality, and institutionalization of patients. However, treatment of poststroke dysphagia remains limited and not always effective. Transcranial direct current stimulation (tDCS) has been reported to enhance recovery from poststroke dysphagia,7-9 but hemispheric targets and the modality and duration of stimulation differ among the published studies, and the variability of response among individual patients remains a matter of debate. More important, no study has assessed the characteristics of responders versus nonresponders.
Swallowing is a bilaterally innervated process with lateralization to a dominant hemisphere.10-13 After a stroke, compensatory reorganization of the contralesional hemispheric projections helps the recovery of swallowing.11,12,14 Some studies have investigated whether anodal stimulation of the damaged hemisphere can restore output from the lesioned side or limit hemispheric inhibition from the contralesional hemisphere, which is believed to be hyperactive after a stroke.15,16 In other studies, the contralesional hemisphere was stimulated to improve naturally compensatory excitability and plasticity. 17 Few studies have sought to inhibit the hyperactivity of the unaffected hemisphere and decrease the transcallosal inhibition as a means to treat dysphagia.18,19 The theory behind this approach is that transcallosal inhibition increases after a stroke, and decreasing this inhibition could help restore swallowing. 9 A recent systematic review and a meta-analysis of noninvasive brain stimulation (NIBS) for the management of poststroke dysphagia (Yang et al 7 published in 2015 and Pisegna et al 9 published in 2016, respectively) included 3 randomized controlled trials using tDCS (for a total of 50 patients).
The meta-analysis showed that NIBS has a significant effect on the severity of poststroke dysphagia, but at present, no conclusions can be drawn regarding which site of stimulation is more beneficial, although there may be a stronger argument for focussing stimulation on the unaffected hemisphere. 20
Dual stimulation with anodal tDCS over the damaged hemisphere plus cathodal stimulation over the contralesional one has been investigated in the recovery of motor skills 21 and, more often, in hemispatial neglect rehabilitation. 22 However, the numbers of patients in each of the above-mentioned studies on dysphagia were small. In this study, in a large group of patients admitted to a neurorehabilitation department with poststroke dysphagia undergoing conventional swallowing rehabilitation therapy, we investigated whether anodal tDCS over the lesioned hemisphere and cathodal stimulation of the contralateral one could improve swallowing function compared with sham stimulation and whether there were any clinical features predictive of the effect of tDCS in individual patients.
Methods
We recruited patients with poststroke dysphagia referred to the Neurorehabilitation Department of the ICS Maugeri IRCCS between 2014 and 2015. Inclusion criteria were the following: (1) occurrence of unilateral stroke in the 4 weeks prior to enrollment; (2) age >18 years; (3) no other muscular and neurological disease or severe disorder of consciousness; (4) mild to severe dysphagia, with a Dysphagia Outcome Severity Scale (DOSS) score <5; and (5) a National Institutes of Health Stroke Scale (NIHSS) score at enrollment <22. We excluded patients with a prior history of dysphagia, NIHSS score at enrollment >22, other severe clinical conditions (eg, severe infections), or potential contraindications to tDCS (electrically activated implants; intracerebral derivations, stimulators, clips or brain metastases; metal in any part of the body; history of epilepsy). The average duration of hospitalization was 2 months.
For each patient, we recorded age, sex, type and site of stroke, use of any drugs acting on the nervous system (antidepressants, antipsychotics, antiepileptic drugs, benzodiazepines), the presence of devices (nasogastric tube, tracheostomy tube), and other neurological symptoms influencing dysphagia (aphasia, apraxia, cranial nerve deficit). We used NIHSS score before and after stimulation as a measure of stroke disability and the Functional Inde-pendence Measure (FIM), 23 which consists of 13 items with a motor and a cognitive assessment, to evaluate functional dependence.
Swallowing was evaluated clinically by rehabilitation doctors and speech-language therapists specialized in dysphagia. The DOSS score was used as an outcome measure; this validated dysphagia scale rates decreasing severity from 1 to 7 and was developed to systematically rate the functional severity of dysphagia based on objective assessment and make recommendations for diet level, independence level, and type of nutrition. 24 On the same day as the DOSS assessment, a video-fluoroscopy study (VFSS) with a 10 mL bolus of liquid and semiliquid, and solid gastromiro-containing foods was performed. During the examination, we evaluated the risk of aspiration using the Penetration Aspiration Scale (PAS), a validated 8-point ordinal scale of increasing severity that quantifies penetration and aspiration events observed during VFSS. 25 Dysphagia was assessed 1 week before and 1 week after completion of the treatment protocol. In all, 20 patients with a risk of aspiration had a nasogastric tube.
Patients were allocated to 2 groups based on random extractions from a Bernoulli distribution with parameter 0.5. One group received 2 mA of anodal tDCS over the lesioned hemisphere and cathodal stimulation to the contralesional one; the other group received sham stimulation by means of a battery-driven constant current stimulator (HDCkit Newronika, Italy) over 10 days of treatment during conventional swallowing rehabilitation therapy. In the sham treatment group, the same protocol was applied, except that the 2-mA current was delivered for only 30 s through 2 electrodes, producing an initial tingling sensation but no significant changes in cortical excitability. 26
In this single-blind study, the patients were unaware of whether they were receiving the active treatment or the sham treatment. The swallowing evaluation and training as well as the (real or sham) stimulation were performed by an investigator who did not participate in outcome measurements or data analysis. The targeted cortical area was the pharyngeal motor cortex. The 25-cm2 rectangular surface electrodes, in saline-soaked synthetic sponges, were applied between C3/T3 on the left and C4/T4 on the right, according to the international 10-20 EEG electrode system. This montage was expected to generate the maximum current density over the motor and premotor areas, which, as shown by previous studies,11,13,17,27 play a prominent role in reorganization of the swallowing motor cortex in poststroke dysphagia. The stimulation was applied during the swallowing rehabilitation therapy. The duration of each swallowing rehabilitation session was 30 minutes, which is the same period as the tDCS stimulation.
Swallowing training consisted of direct therapies (including compensatory methods, behavioral maneuvers, supraglottic and effortful swallowing) and indirect approaches (physical maneuvers, thermal tactile stimulation). Patients who were at risk of aspiration were tube fed and underwent only indirect therapy. Long-term follow-up outcomes were assessed by telephonic interview, recording changes of diet or occurrence of lung infections 2 months after the patient’s discharge from the rehabilitation unit.
This randomized controlled trial, which complied with CONSORT guidelines, 28 was approved by the institutional review board of ICS Maugeri IRCCS of Pavia (CEC 1045) and was carried out according to the principles of the Declaration of Helsinki. Written informed consent was obtained from the patients or legal representative before enrollment.
Statistical Analysis
Variations from baseline (Δ) were estimated as the difference between measurements at t1 and t0 (ie, t1 − t0). We defined a clinically relevant variation as being an improvement of ≥2 points in the DOSS score, as in previous studies. 17 Percentage variations from baseline (Δ%) were estimated as [(t1 − t0)/t0] × 100. Given a specific threshold x (with x increasing iteratively from 10% to 90% in steps of 10%), improvements from baseline conditions were defined for DOSS and PAS scores as follows:
DOSS score: patients with Δ% from baseline ≥(+x)
PAS score: patients with Δ% from baseline ≤(−x)
Deviations from normality assumptions of quantitative variables were assessed by Shapiro’s test. The correlation between normally distributed quantitative variables (Shapiro’s P > .05) was assessed by the Pearson test or Spearman test: correlation coefficients >|0.5| were considered clinically significant. The presence of statistically significant differences in normally distributed quantitative variables between binary subgroups was tested by Student’s t-test or the Wilcoxon rank-sum test. The Wilcoxon signed rank test was applied to test for the presence of significant variations in DOSS and PAS scores from baseline measurements. The presence of statistically significant differences in categorical variables by subgroups was assessed by Fisher’s exact test. The distribution of categorical variables was described by count (frequency, percentage), whereas that of quantitative variables was described by the median value (25th, 75th percentiles, interquartile range [IQR]). The threshold for identifying statistically significant associations was set at a P value <.05. Statistical procedures were performed using the R statistical software, version 3.2.1 (http://www.r-project.org).
Results
A total of 53 patients were assessed for eligibility, of whom 13 were excluded because they did not meet the inclusion criteria (respiratory infection, epileptic seizures). The remaining 40 patients (median age = 66 years; IQR = 55.75-73.25; 20 men and 20 women) were randomly allocated to 2 groups: 20 received the anodal/cathodal current, and 20 were given sham treatment over the lesioned hemisphere and the contralateral one. Figure 1 shows the study flowchart.
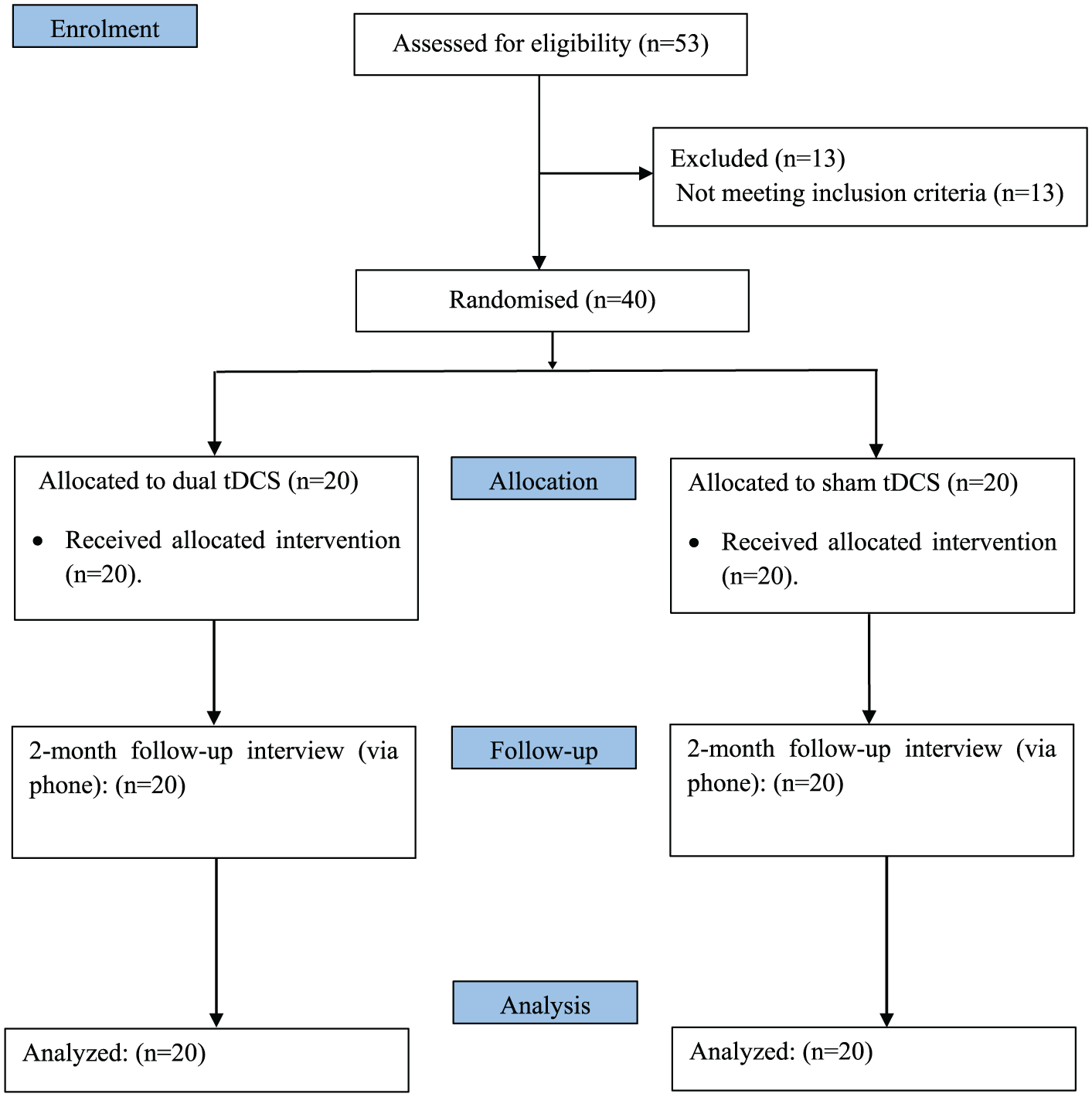
Trial design flowchart: 53 patients were assessed for eligibility; 13 were excluded because they did not meet inclusion criteria (respiratory infection, epileptic seizures). The remaining 40 patients were randomly allocated to 2 groups: dual transcranial direct current stimulation (tDCS) and sham tDCS. All patients received 10 sessions of treatment and were followed up for 2 months. There were no severe complications during the whole period.
The stroke involved the left hemisphere in 31 patients and the right hemisphere in the other 9 patients. In all, 34 patients were receiving one or more medications acting on the nervous system, 22 had a medical device such as nasogastric or tracheostomy tube, and 32 patients had other, coexistent neurological symptoms that could influence dysphagia. All patients were right-handed. Overall, at baseline, the patients had medium-severe neurological impairment, as determined by the NIHSS score, and severe functional dependence, measured with the FIM scale, but there were no significant differences between the 2 groups (tDCS/sham) with regard to the distribution of the scores of these scales, use of drugs, site and side of the stroke, or other neurological symptoms. No serious side effects were observed, and all patients completed the 10 stimulation/sham sessions. None of the patients in the overall cohort had adverse events or changes in diet in the 2 months following the treatment. Table 1 reports the distribution of the analyzed variables measured at baseline in the whole cohort and in the tDCS and sham treatment groups.
Baseline Characteristics of Patients in the Overall Cohort and in the tDCS and Sham Treatment Groups. a
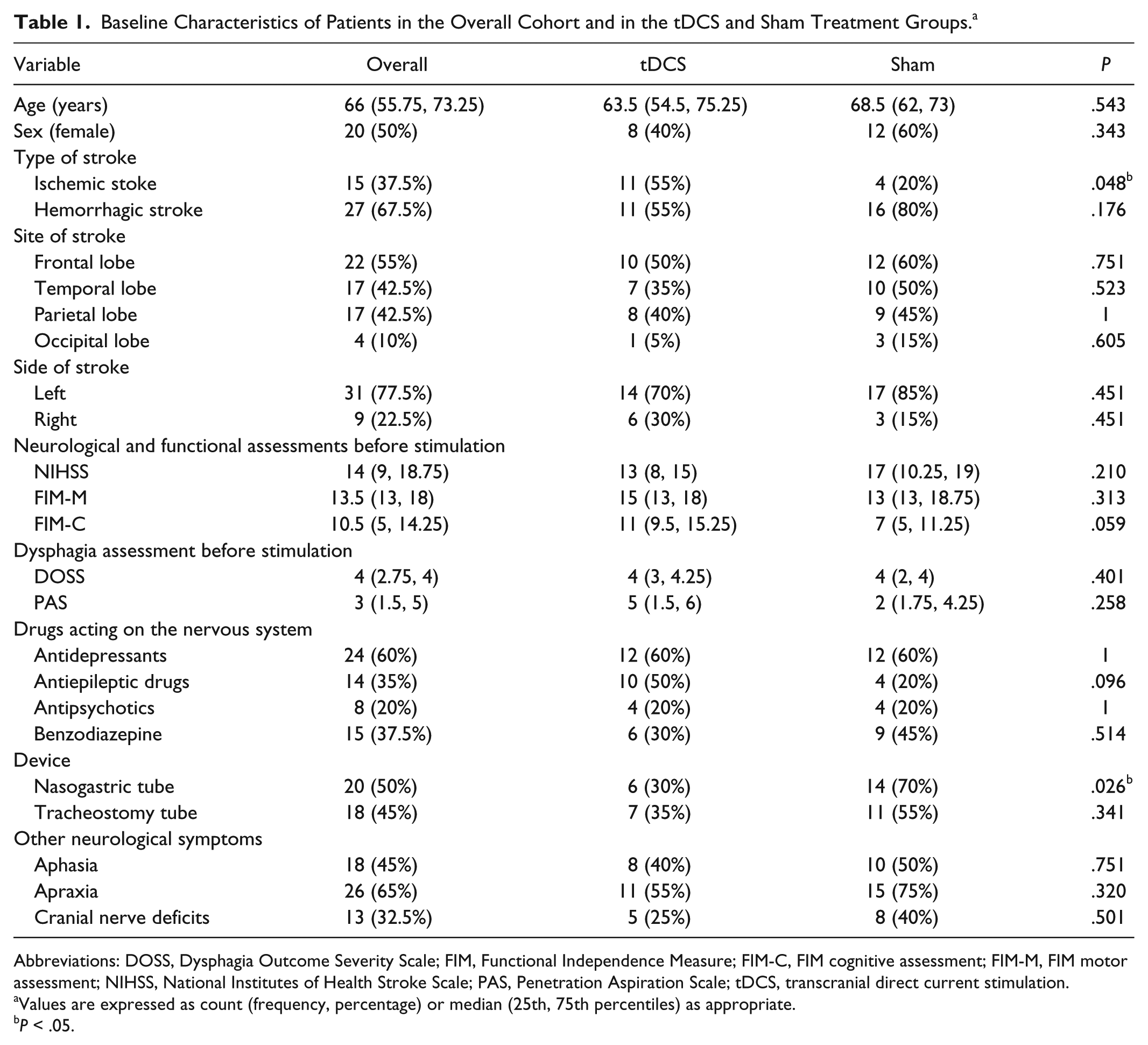
Abbreviations: DOSS, Dysphagia Outcome Severity Scale; FIM, Functional Independence Measure; FIM-C, FIM cognitive assessment; FIM-M, FIM motor assessment; NIHSS, National Institutes of Health Stroke Scale; PAS, Penetration Aspiration Scale; tDCS, transcranial direct current stimulation.
Values are expressed as count (frequency, percentage) or median (25th, 75th percentiles) as appropriate.
P < .05.
First, we estimated the absolute variation from baseline (Δ) in DOSS and PAS scores (the primary outcome of the study) in the whole sample and tDCS and sham treatment groups. DOSS scores increased significantly from baseline in the whole sample (P = .021) but not in the tDCS or sham treatment groups (P > .05), whereas PAS scores decreased significantly both in the whole sample and in the 2 treatment groups (P < .01; Table 2). Thus, we compared DOSS Δ and PAS Δ between the tDCS and sham treatment groups. Results reported in Table 2 and in Figure 2 show no significant differences between the 2 groups (P > .05). Similarly, we analyzed the percentage variations from baseline in DOSS score (DOSS Δ%) and PAS score (PAS Δ%)—the secondary outcomes of the study—by treatment group (Table 2, Figure 2) and found no significant differences in score distributions between groups (P > .05).
Absolute Variation From Baseline (Δ) and Percentage Variation From Baseline (Δ%) of DOSS and PAS Scores in the Whole Sample and in tDCS and Sham Treatment Groups. a
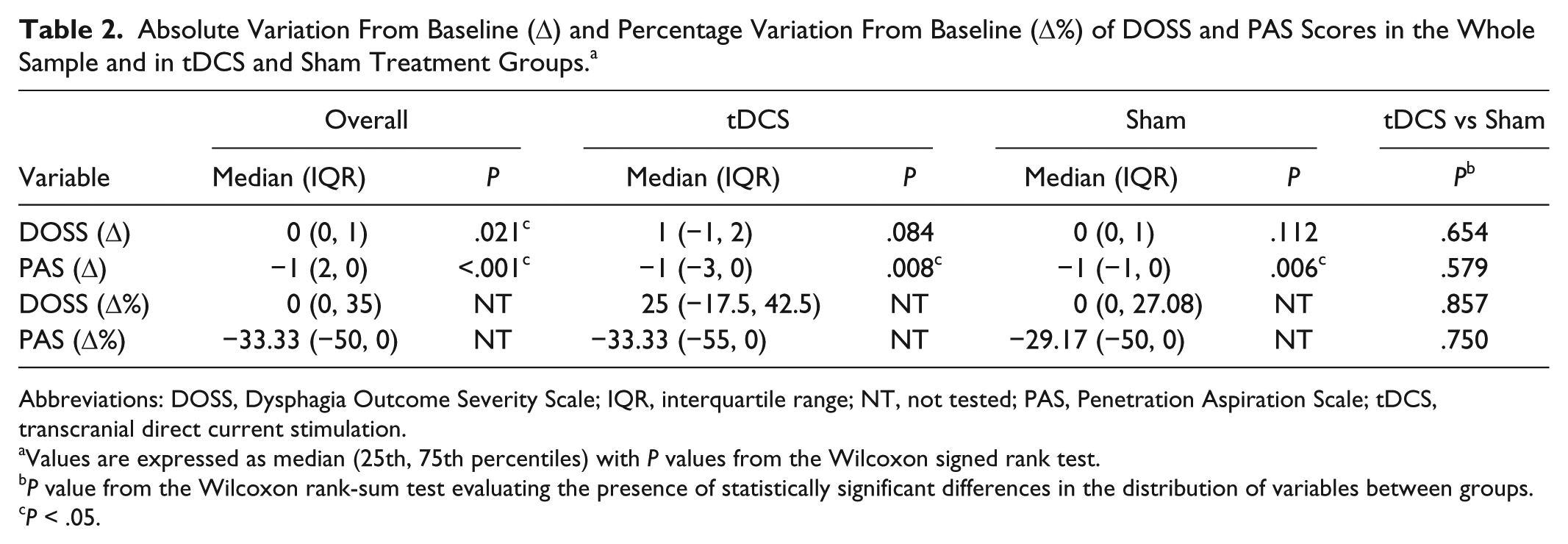
Abbreviations: DOSS, Dysphagia Outcome Severity Scale; IQR, interquartile range; NT, not tested; PAS, Penetration Aspiration Scale; tDCS, transcranial direct current stimulation.
Values are expressed as median (25th, 75th percentiles) with P values from the Wilcoxon signed rank test.
P value from the Wilcoxon rank-sum test evaluating the presence of statistically significant differences in the distribution of variables between groups.
P < .05.
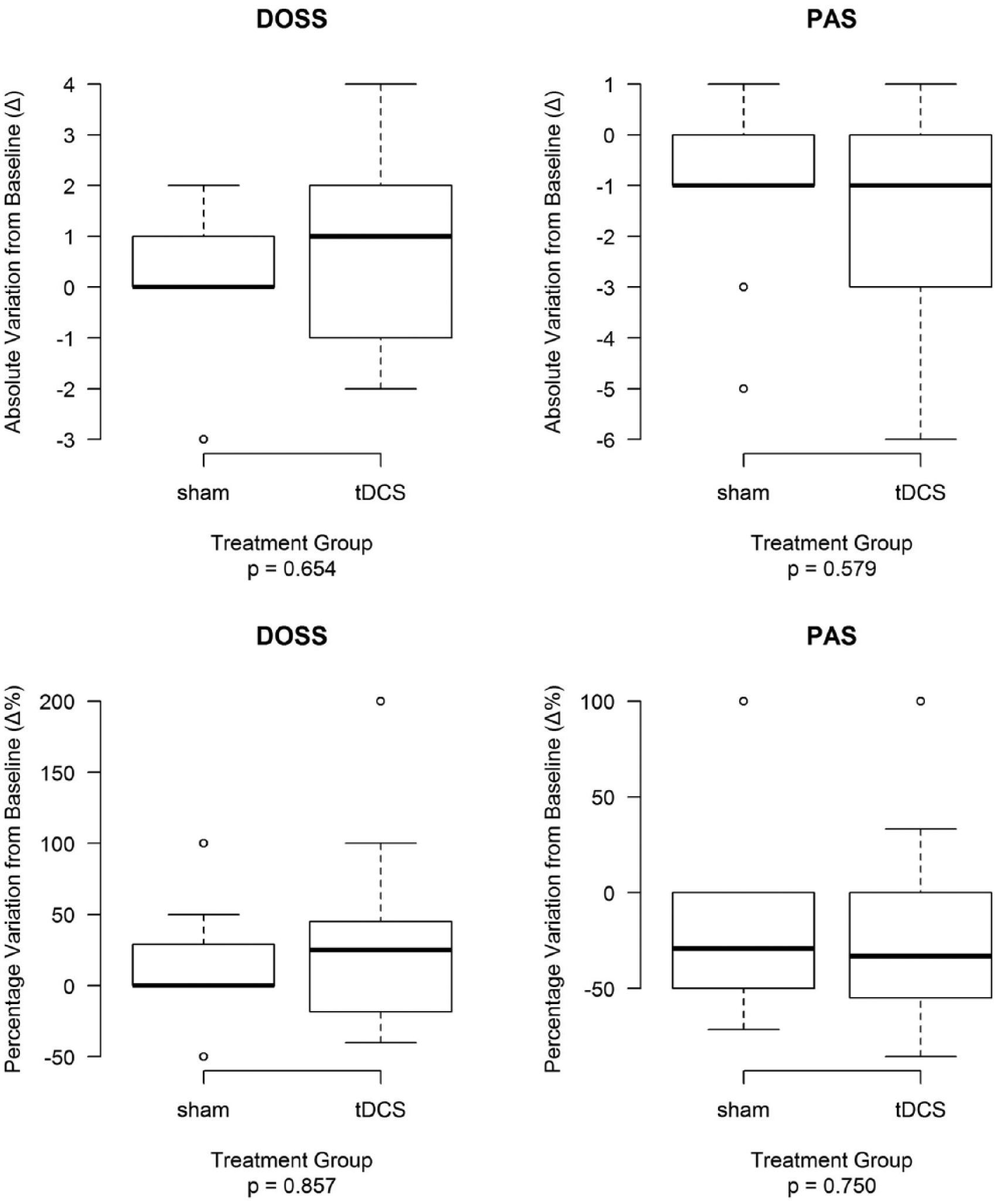
Absolute variations from baseline (Δ) and percentage variations from baseline (Δ%) in DOSS and PAS scores in tDCS and sham treatment groups.
We considered at least 2 points of improvement in the DOSS score as clinically relevant. Of 20 patients, 6 (30%) in the tDCS group and only 2 of 20 (10%) in the sham treatment group had ≥2 points of improvement in their DOSS score. Although the frequency of individuals achieving an improvement was higher in the tDCS group than in the sham group, the difference between groups was not significant (P = .235). Similarly, comparing the percentage improvement from baseline in DOSS score (positive Δ% variations) and PAS score (negative Δ% variations) between the 2 groups according to increasing thresholds (ie, from ≥10% to ≥90%), we found that the probability of individuals treated with tDCS achieving an improvement was higher for each threshold than that of patients given the sham treatment (Table 3). For example, 55% of patients treated by tDCS had a ≥20% improvement in DOSS score from baseline compared with 40% of patients treated with sham stimulation (P = .53), but in no case was the difference between groups significant.
Probability of Improvement in the DOSS and PAS Scores in the tDCS and Sham Treatment Groups. a
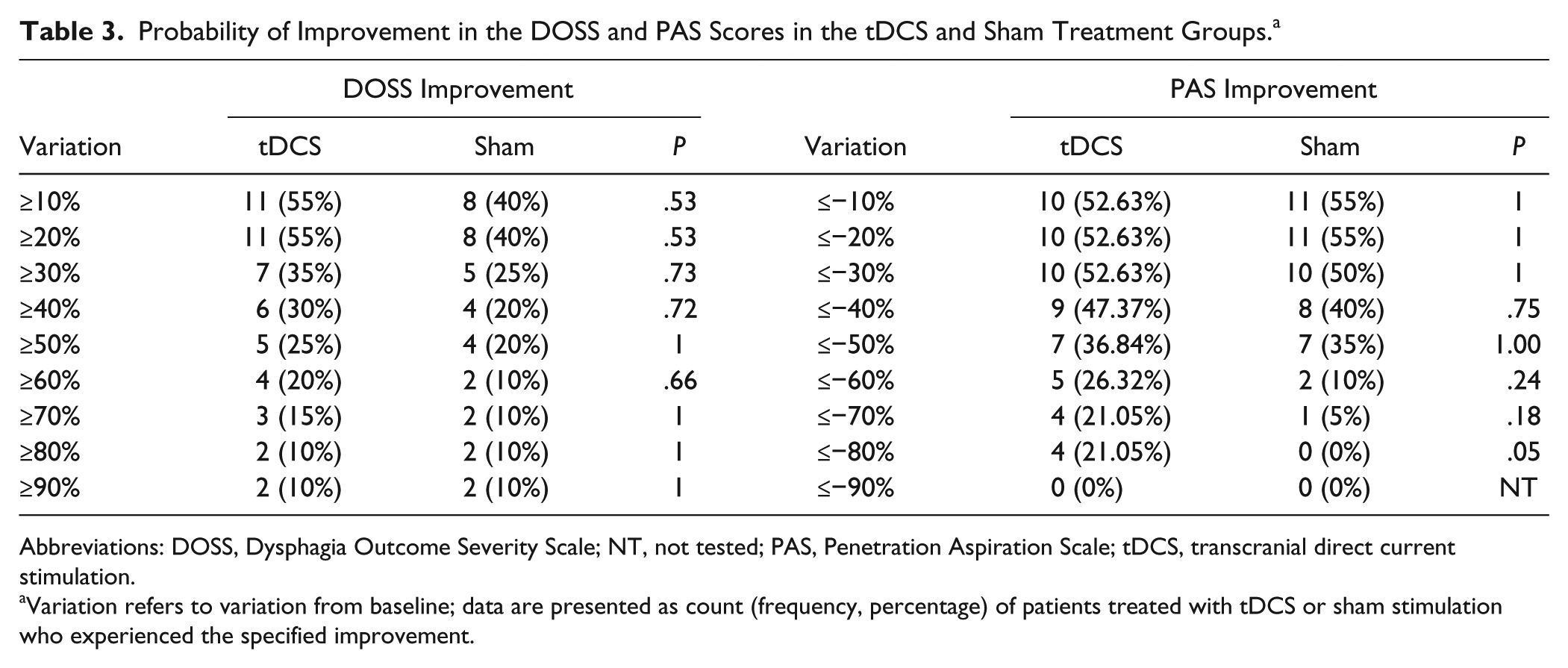
Abbreviations: DOSS, Dysphagia Outcome Severity Scale; NT, not tested; PAS, Penetration Aspiration Scale; tDCS, transcranial direct current stimulation.
Variation refers to variation from baseline; data are presented as count (frequency, percentage) of patients treated with tDCS or sham stimulation who experienced the specified improvement.
Univariate tests were carried out to identify characteristics that were significantly unbalanced between the 2 groups of individuals given tDCS or sham treatment considering all variables recorded at baseline (age, sex, type/site of stroke, use of any drugs acting on the nervous system, presence of devices). The results, reported in Table 1, identified 2 potential confounders: the proportion of patients with a nasogastric tube was significantly lower among individuals treated with tDCS compared to those given the sham treatment (30% vs 70%, P = .026) and the proportion of individuals with ischemic stroke was significantly higher in the group treated with tDCS than in the sham group (55% vs 20%, P = .048; Table 1). We, therefore, compared the percentage improvement from baseline in DOSS score and PAS score between the 2 groups according to increasing thresholds within the subgroups of individuals with/without a nasogastric tube (Table 4) and with/without an ischemic lesion. Among the subgroup of individuals with no nasogastric tube (Table 4), patients treated with tDCS (14 patients) had a significantly higher probability of ≥10% and ≥20% improvement from baseline in the DOSS score than those (6 patients) receiving the sham treatment (64.29% vs 0%, P = .01). Similarly, 6/14 patients (42.85%) in the tDCS group had ≥2 points of improvement in their DOSS score versus 0/6 (0%) in the sham group (P = .11).
Probability of Improvement in DOSS and PAS Scores in Patients With Versus Without Nasogastric Tube. a
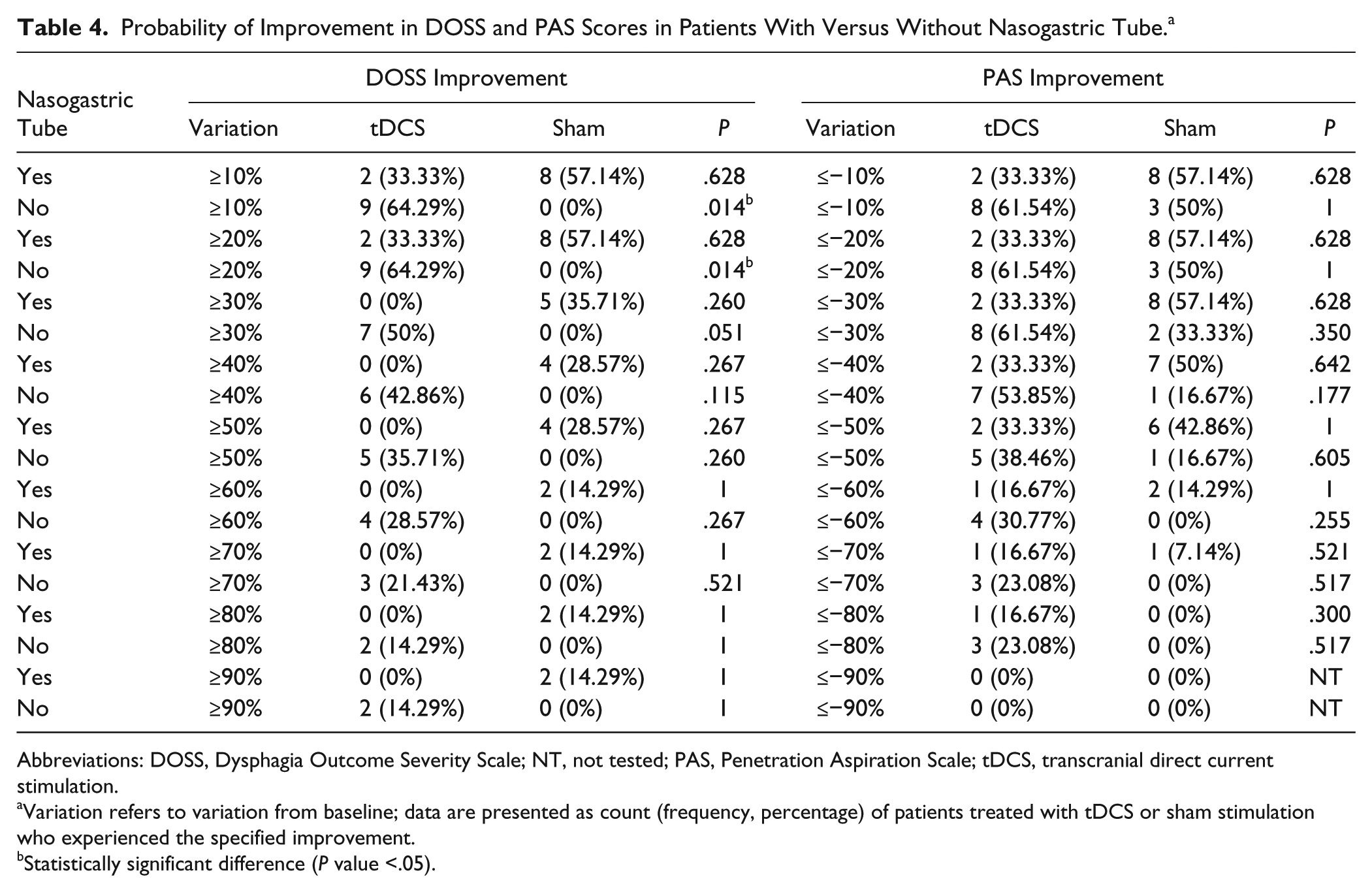
Abbreviations: DOSS, Dysphagia Outcome Severity Scale; NT, not tested; PAS, Penetration Aspiration Scale; tDCS, transcranial direct current stimulation.
Variation refers to variation from baseline; data are presented as count (frequency, percentage) of patients treated with tDCS or sham stimulation who experienced the specified improvement.
Statistically significant difference (P value <.05).
No significant differences were observed between tDCS and sham treatment in the subgroup of patients with a nasogastric tube or in the 2 subgroups with/without ischemic stroke.
Discussion
The main finding of this study is that treatment with dual tDCS of patients with poststroke dysphagia in the early phase of intensive rehabilitation seems to have no significant effect; no significant difference was observed between patients treated with dual-tDCS and those treated with sham stimulation. Stimulating the lesioned or nonlesioned hemisphere remains a controversial topic in poststroke recovery of dysphagia. Recently, investigators have shown that stimulating the unaffected hemisphere could be more effective.9,17 Some studies found that anodal stimulation of the affected hemisphere and cathodal stimulation of the contralesional one both led to improved outcomes in terms of motor recovery after stroke.22,29 The aim of this approach was to restore output from the lesioned side and counteract a suppressive effect from the contralesional hemisphere, which is believed to be hyperactive after a stroke.10,11 We decided to apply the same approach to patients with poststroke dysphagia admitted to our intensive neurorehabilitation unit.
Unlike previous studies, we decided to use dual tDCS and we recruited a large number of patients and analyzed possible associations between the eventual efficacy of tDCS versus sham treatment and some clinical conditions typical of patients referred to an intensive neurorehabilitation unit that can influence normal recovery from dysphagia or treatment with tDCS, such as the presence of devices (including tracheostomy and nasogastric tubes) and pharmacological medications acting on the nervous system.
We used internationally validated scales to assess the neurological and functional impairment at baseline and, like the majority of previous studies, we measured recovery of swallowing with the PAS30,31 and the DOSS.16,17 We chose a stimulation protocol based on previous studies showing that the application of 2 mA is effective and well tolerated.17,32,33 Our intent was to evaluate the recovery of poststroke dysphagia in patients undergoing intensive rehabilitation in the early stage of stroke rehabilitation when neuroplastic mechanisms can be best exploited. Second, we aimed to better understand how brain stimulation can be integrated into the clinical activity of a neurorehabilitation unit.
We found that improvements in DOSS scores were higher in the tDCS group than in the sham treatment group, but the difference between groups was not significant. No serious side effects were observed throughout the duration of the study. The rehabilitation outcome appeared to be maintained at 2-month follow-up in both groups.
The present study has several limitations. First, the follow-up was conducted by recording changes in diet or occurrence of lung infections reported during a telephonic interview, and although we enrolled a large number of patients, the study population was not homogeneous in terms of severity of stroke or other clinical parameters. However, at baseline, there were no differences between the tDCS and sham groups as regards scores on all scales used to assess neurological and functional impairment and dysphagia. Another important limitation was that we included patients receiving pharmacological treatment that can modulate the effects of tDCS on the nervous system. This could have contributed to a nonlinear response of stimulation, like other individual physiological and pathological conditions characterizing patients admitted to neurorehabilitation units. However, there were no significant differences between the 2 groups (tDCS/sham treatment) concerning the use of drugs at baseline (Table 1), and we did not observe any particular changes regarding drug use during the 10 days of treatment.
The only significantly unbalanced characteristics were the proportions of patients with a nasogastric tube and those with ischemic stroke in the 2 groups (Table 1). In the subgroup of patients without a nasogastric tube, those who were treated with tDCS (n = 14) had a significantly higher probability of up to 20% improvement in DOSS score from baseline compared with those who were treated with sham stimulation (n = 6). This suggests that dual tDCS stimulation might help recovery in patients whose dysphagia is less severe, although the strong asymmetry in the numbers in this small sample does not allow generalizations to be drawn (Table 1). Furthermore, the intragroup comparison of outcomes represents another clear limitation of this study because the subgroup effects observed could have been heavily influenced by confounders (eg, unbalanced sample size between groups, secondary characteristics of patients). Future studies with a more balanced study design to minimize confounders are required to evaluate the efficacy of tDCS in patients with as compared with those without nasogastric tube.
As shown in previous studies,9,17 it is possible that cortical stimulation of the contralesional hemisphere could play a more effective substantial role in swallowing recovery by improving natural compensatory plasticity. It is still necessary to understand whether stimulation can be effective in patients with severe dysphagia whose clinical conditions are usually complicated and who are most in need of recovery. Swallowing is a complex motor function, and the effect of stimulation may depend on potential activity of brain circuits engaged in the execution of a given task. The final outcome of stimulation, in particular with regard to complex functions, depends not only on the integrity of peripheral structures, but also on the individual’s level of interaction and, therefore, on the patient’s physiological and pathological conditions. It is, of course, difficult to generalize. It is clear that the real effect of stimulation is not based only on a specific protocol, but also depends on the excitability of the target area, the clinical characteristics, and the task during the stimulation. 34 This study sheds some light on in which patients and in what clinical circumstances better results can be obtained from stimulation.
In conclusion, from a practical point of view, the main finding of this study is that anodal stimulation over the lesioned hemisphere and cathodal inhibition of the contralesional one associated with traditional swallowing therapy in the early phase of intensive rehabilitation does not increase the probability of recovery compared with sham stimulation.
Only in the subgroup of patients without nasogastric tube did we find a significantly higher improvement with tDCS treatment as compared with sham stimulation, even though the strong asymmetry in sample numbers between the subgroups and the possible influence of confounders does not allow conclusions to be drawn. Further studies are needed to explore not only the parameters of stimulation, but the patients’ profiles and the clinical conditions that respond better to stimulation in poststroke dysphagia.
Footnotes
Acknowledgements
The authors thank Dr Rachel Stenner for her help with English.
Authors’ Note
AP is the founder and a stakeholder of Newronika srl.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
