Abstract
Background and Objective. Tinnitus is the perception of a phantom sound. The aim of this study was to compare current intensity (center anode 1 mA and 2 mA), duration (10 minutes and 20 minutes), and location (left temporoparietal area [LTA] and dorsolateral prefrontal cortex [DLPFC]) using 4 × 1 high-definition transcranial direct current stimulation (HD-tDCS) for tinnitus reduction. Methods. Twenty-seven participants with chronic tinnitus (>2 years) and mean age of 53.5 years underwent 2 sessions of HD-tDCS of the LTA and DLPFC in a randomized order with a 1 week gap between site of stimulation. During each session, a combination of 4 different settings were used in increasing dose (1 mA, 10 minutes; 1 mA, 20 minutes; 2 mA, 10 minutes; and 2 mA, 20 minutes). The impact of different settings on tinnitus loudness and annoyance was documented. Results. Twenty-one participants (77.78%) reported a minimum of 1 point reduction on tinnitus loudness or annoyance scales. There were significant changes in loudness and annoyance for duration of stimulation, F(1, 26) = 10.08, P < .005, and current intensity, F(1, 26) = 14.24, P = .001. There was no interaction between the location, intensity, and duration of stimulation. Higher intensity (2 mA) and longer duration (20 minutes) of stimulation were more effective. Conclusions. A current intensity of 2 mA for 20-minute duration was the most effective setting used for tinnitus relief. The stimulation of the LTA and DLPFC were equally effective for suppressing tinnitus loudness and annoyance.
Keywords
Introduction
Tinnitus is the perception of a phantom sound. 1 Tinnitus perception is thought to be the by-product of altered neuronal activity in the central nervous system,2-5 and modification of this pathological neuronal activity for tinnitus management is referred to as neuromodulation.6,7 Neuromodulation techniques are hypothesized to work by inducing neural plasticity and disturbing the pathological neural networks responsible for tinnitus. 6 Several noninvasive neuromodulation techniques have been developed, including transcranial magnetic stimulation (TMS), transcranial direct current stimulation (tDCS), and most recently, random noise stimulation and high-definition transcranial direct current stimulation (HD-tDCS).8-13
Transcranial direct current stimulation results in transient (seconds to hours) reduction in tinnitus loudness or distress, depending on the site of cortical stimulation. 14 tDCS has been applied safely without any significant adverse effects in more than 3000 individuals. 15 Depending on the method of the stimulation, tDCS can increase or decrease the cortical excitability in the brain region to which it is applied. The 2 most common targets of stimulation investigated for transient tinnitus suppression are the left temporoparietal area (LTA) and the dorsolateral prefrontal cortex (DLPFC). The LTA and the DLPFC are part of a neural network that appears to play a significant role in tinnitus perception: Brodmann areas (BA) 41, 42 (primary auditory cortex), BA areas 21, 22 (auditory association areas), and parts of the limbic system (amygdala and hippocampus).3,16,17 Anodal tDCS over the LTA (with cathode over contralateral frontal scale) led to transient suppression of tinnitus in 35% to 40%18,19 of participants. Shekhawat et al 20 explored tDCS dose (current intensity and duration) effects for tinnitus suppression with LTA stimulation. This demonstrated that anodal tDCS of LTA using a 2 mA current intensity delivered for 20 minutes was the most effective, of the settings tested, for tinnitus relief.
Bifrontal tDCS over the DLPFC (anodal stimulation with anode over right DLPFC and cathode over left DLPFC; cathodal stimulation with anode over left DLPFC and cathode over right DLPFC) also results in significant suppression of tinnitus perception (42%) and distress (43%). 21 Although structures stimulated during DLPFC or LTA tDCS appear to contribute to tinnitus, their exact role in tinnitus perception is still relatively unknown. Conclusions are further complicated by the comparatively broad stimulation pattern resulting from conventional tDCS. Parazzini et al 22 investigated the distribution of electric field and current density during LTA and DLPFC stimulation and found that during LTA stimulation the distribution is widespread due to the large distance between anode and cathode. The electric field and current density is relatively more localized during the DLPFC stimulation due to the short distance between the anode and cathode. 22 But in both cases, broad regions of the brain between and underneath both electrodes are stimulated, making interpretation based on anatomical specificity difficult. There is some preliminary evidence that tDCS of DLPFC and LTA may modulate tinnitus loudness and annoyance differently. 23 However, this issue has not been investigated independently, the present study will be the first attempt to explore this aspect of the possibility of differential impact of site of stimulation and modulation of tinnitus loudness and annoyance.
One potential solution to these broad excitation patterns is the use of a new technique called HD-tDCS. In HD-tDCS the conventional large sponges electrodes of tDCS are replaced with smaller gel electrodes. tDCS uses 2 electrodes placed across the head this is believed to produce deep brain current flow, since current is not lost as it crosses gray matter. tDCS stimulates large tracts of white matter, deep and mid-brain structures. 24 In contrast, 4 × 1 HD-tDCS produces current flow that is restricted to areas below the 4 electrodes and with limited depth of penetration. 25 A further advantage of the 4 × 1 HD-tDCS montage is that the diffusion of return current along the 4 electrode forming the ring results in a more unidirectional modulation such that the polarity of the center electrode (anode or cathode) determines the primary change in excitation; this is compared with conventional tDCS where both anodal and cathodal effects must always be considered.9,26
We compared the distribution of current density during the conventional tDCS and HD-tDCS using the finite element models and found HD-tDCS to be more focal than tDCS (Figure 1). For example, the 4 × 1-montage of HD-tDCS is proposed to produce brain current flow in the region circumscribed by the ring of 4 electrodes, with a polarity set by the center electrode.9,25 Current is applied to selected HD electrodes to optimize current flow to target. Kuo et al 10 compared the cortical excitability induced by conventional tDCS and the 4 × 1 HD-tDCS montage and found the after effects of HD-tDCS to be more lasting and focal compared with tDCS. No negative side effects have been reported to date in the eight research studies published using HD-tDCS; 75 patients with stroke, 8 pain,26,27 and normal healthy volunteers.9,10,12
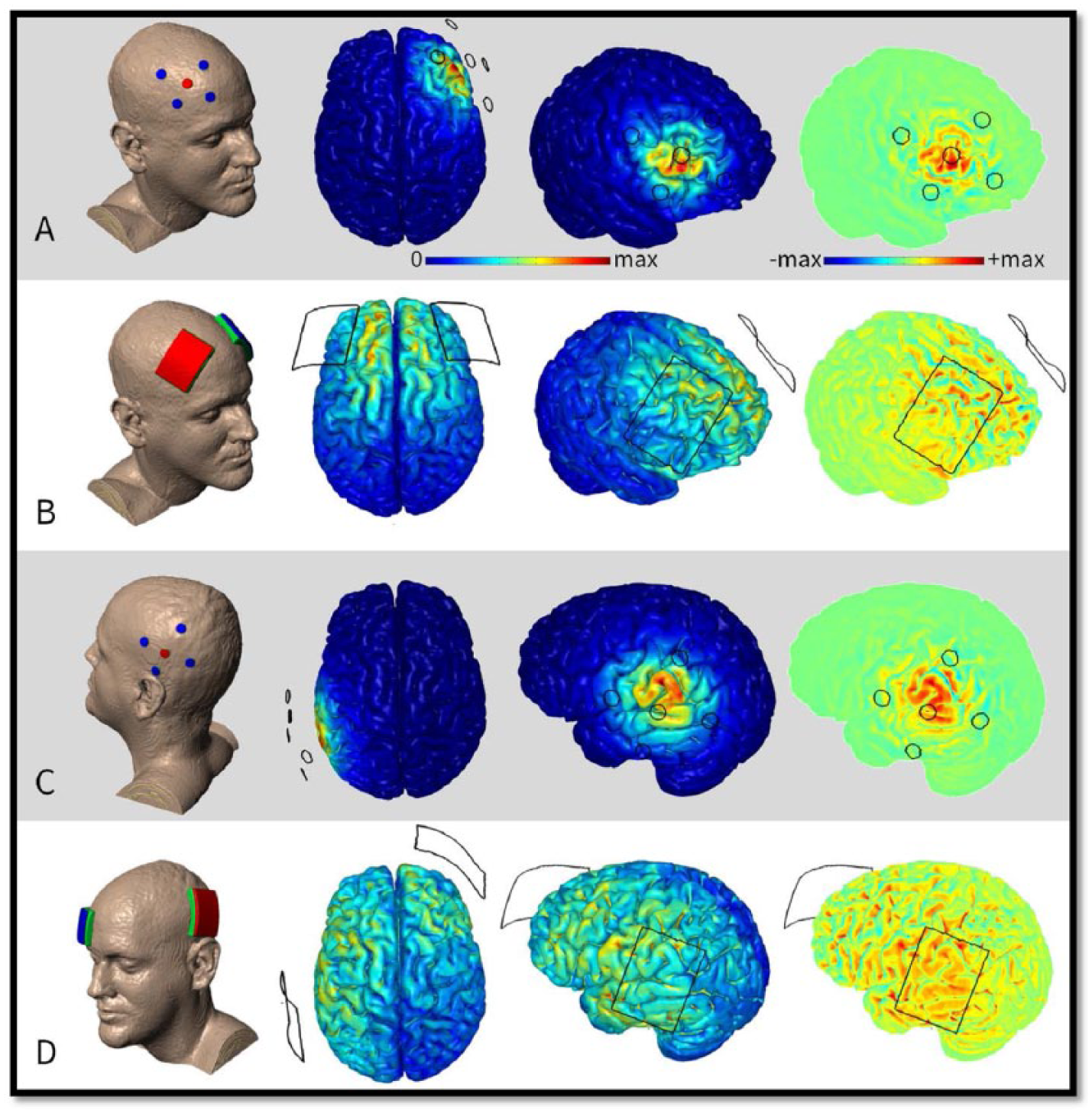
Finite element models of transcranial direct current stimulation (tDCS) montages nominally targeting and dorsolateral prefrontal cortex (DLPFC) (A, B) and left temporoparietal area (LTA) (C, D). The high-definition (HD) 4 × 1 montages (A, C) used by this study are contrasted with the conventional 5 × 7 cm sponge montages (B, D) previously used. Electric field magnitude from 0 to maximum and radial electric field from maximum cathodal (blue) to maximum anodal (red) were predicted. HD montages have greater focality and accuracy under the anode. Intensity is however lower. In the case of radial electric field, a concentrating effect can be observed under the center of the 4 × 1 ring—anodal/inward electric field being relatively more intense than the cathodal/outward electric field at the perimeter of the ring. Maximum scale values for montages A, B, C, and D, are 0.3, 0.64, 0.28, and 0.7 in V/m, respectively. Predictions are based on 2-mA stimulation. Methodology of the data—high-resolution (1 mm3) T1-weighted magnetic resonance imaging (MRI) scans were segmented into tissues of varying conductivities using the “New Segment” algorithm 1 included in SPM8 (Wellcome Trust Centre for Neuroimaging, London, UK). Updated tissue probability maps with an extended field of view were combined with postprocessing filters to improve continuity in skull, cerebrospinal fluid (CSF), and gray matter. 2 Additional manual segmentation corrected remaining artifacts and resolved finer detail throughout the image volume. A multipart adaptive volume mesh was generated using the voxel-based meshing algorithm available in ScanIP (Simpleware, Exeter, UK). Meshes were imported into a finite element package (COMSOL Multiphysics 4.3, COMSOL, Burlington, MA, USA) where volume conductor physics were applied. Tissues and materials were given the following conductivies (S/m): skin (0.465), fat (0.025), bone (0.01), CSF (1.6), gray matter (0.276), white matter (0.126), air (10−15), electrode (5.99 × 107), and conductive gel (1.4).3,4 The Laplace equation (∇⋅(σ∇V) = 0) was solved with the following boundary conditions: inward current density on the anode, ground on the cathode, insulated on all other surfaces. Electric field magnitude was calculated on the cortical surface. The component of the electric field perpendicular to the cortical surface was calculated as radial electric field. Both of which are believed to modulate firing rate and in turn represent stimulation.5,6
High-definition tDCS is yet to be used in tinnitus research. This study is the first attempt to use HD-tDCS to modulate tinnitus; the aim was to optimize 4 × 1 HD-tDCS current intensity (1 vs 2 mA), duration (10 vs 20 minutes), and location (LTA vs DLPFC) for tinnitus relief.
We hypothesized the following:
2 mA HD-tDCS current intensity would be more effective in suppressing tinnitus loudness and annoyance than 1 mA.
20-minute HD-tDCS duration would be more effective in suppressing tinnitus loudness and annoyance than 10 minutes.
HD-tDCS of LTA would result in more tinnitus loudness suppression than DLPFC stimulation.
HD-tDCS of DLPFC would result in more tinnitus annoyance suppression than LTA stimulation.
Material and Methods
This open-label within-subject treatment optimization design study was approved by the University of Auckland Human Participant’s Ethics Committee. Each participant provided written informed consent in accordance with the Declaration of Helsinki.
Participants
Participants were recruited through the University of Auckland Hearing and Tinnitus Research Database. For inclusion in this study, participants had to be at least 18 years old and have experienced chronic tinnitus for a minimum of 2 years. Participants were screened by a psychiatrist for their candidacy to undergo HD-tDCS and were excluded if they had any contraindications such as previous brain surgery, metal or electronic implants, pregnancy, cardiovascular disease, psychotropic medication, and a history of seizures.
Figure 2 shows the CONSORT 2010 patient flow diagram for this study. Forty participants were screened for the eligibility to undergo HD-tDCS. Twenty-seven participants completed the study. Demographic details of the participants included in this study are provided in Table 1.
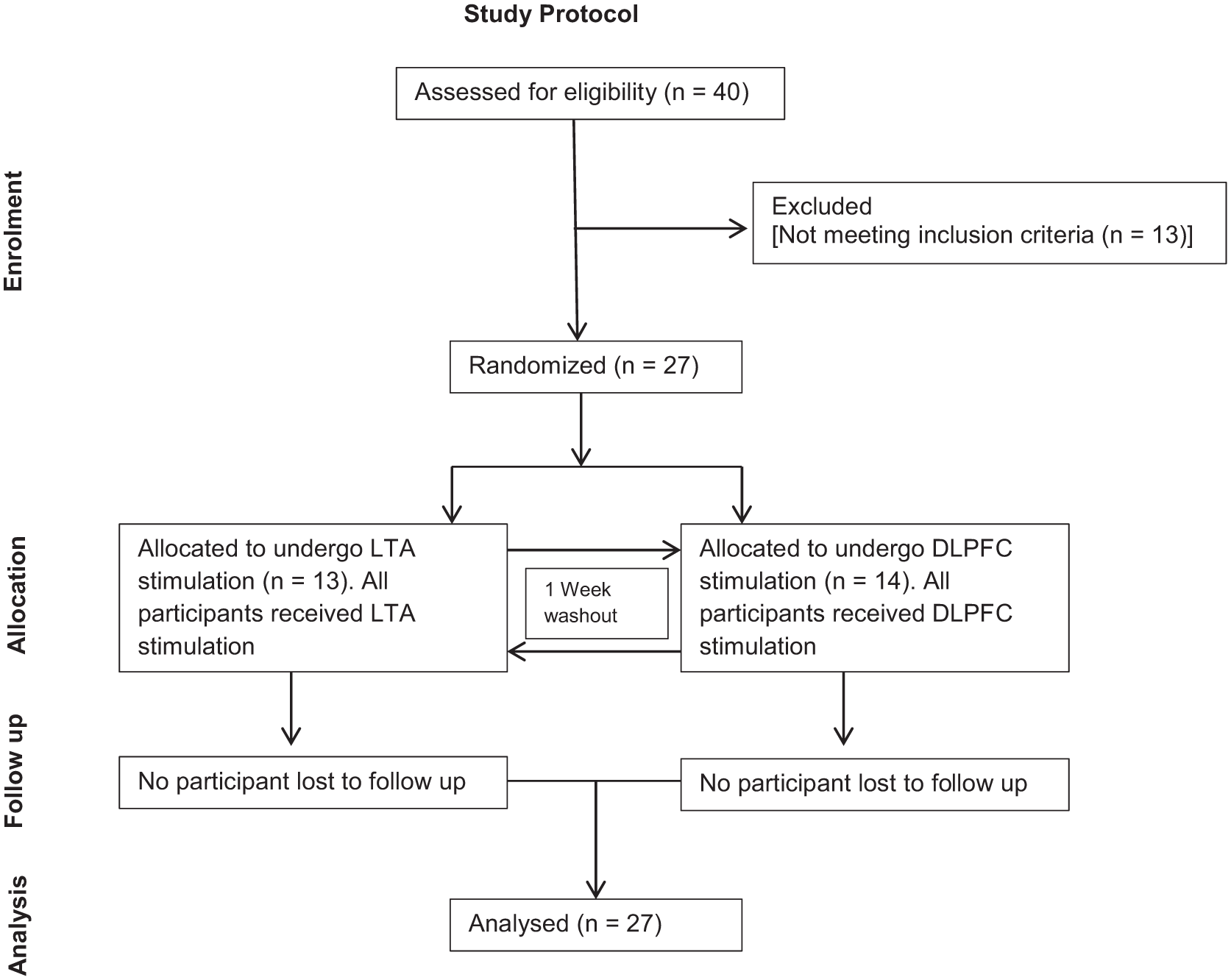
CONSORT 2010 patient flow diagram of the progress through the phases (Enrolment, Allocation, Follow-up, and Analysis) of the study of high-definition transcranial direct current stimulation (HD-tDCS) of left temporoparietal area (LTA) and dorsolateral prefrontal cortex (DLPFC).
Demographic Details of Participants Included in the Study.
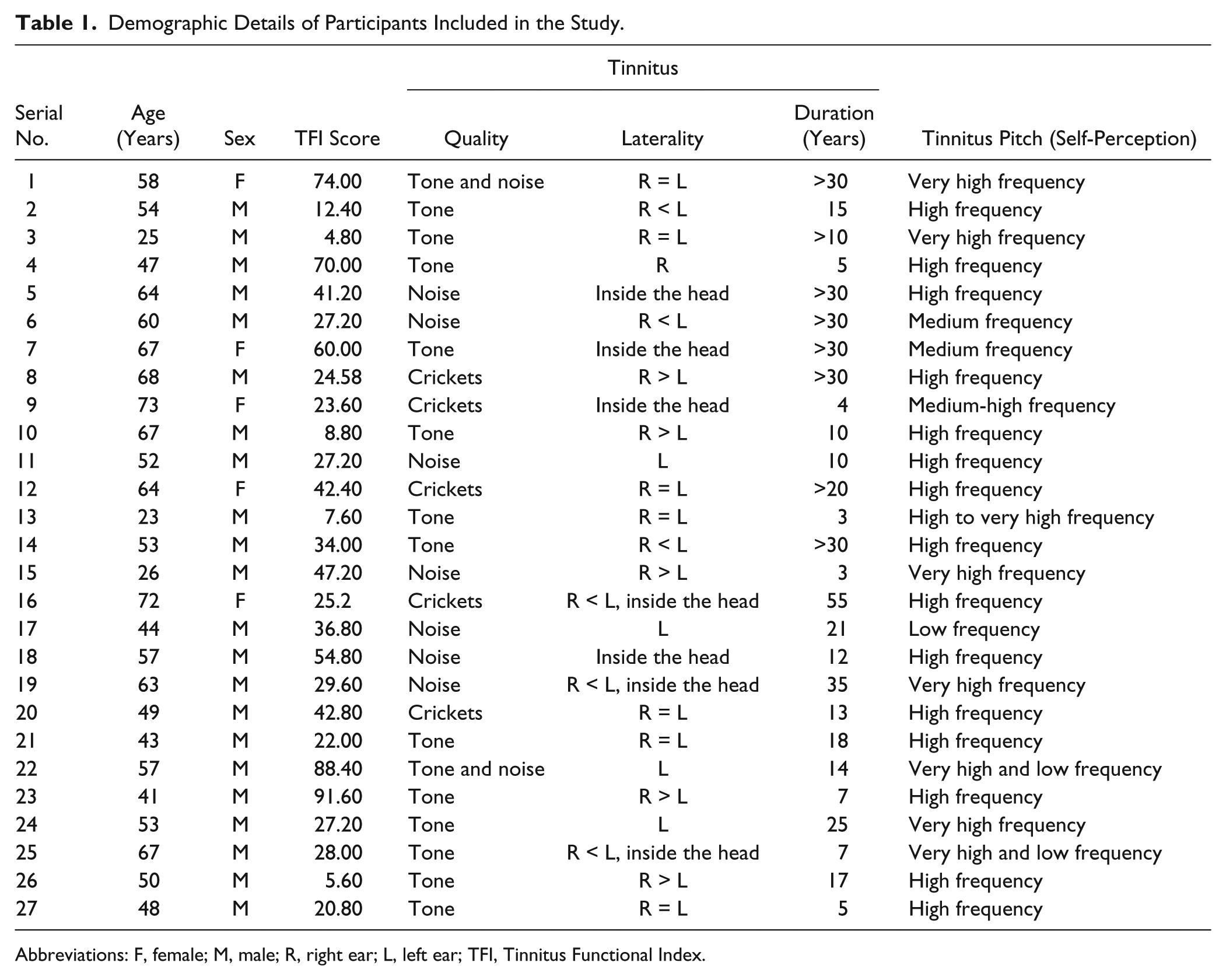
Abbreviations: F, female; M, male; R, right ear; L, left ear; TFI, Tinnitus Functional Index.
Procedure
This was a dose-response study to investigate optimal HD-tDCS current intensity (1 vs 2 mA), duration (10 vs 20 minutes), and location (LTA vs DLPFC) for tinnitus relief based on Shekhawat et al. 20 HD-tDCS sessions were conducted in a sound treated room (ISO 8253-1:2010). Four combinations of stimulus intensity and duration were used in the following incremental order: 1 mA for 10 minutes, and 20 minutes followed by 2 mA for 10 minutes, and 20 minutes for both sites of stimulation (LTA and DLPFC). Participants were not informed about the intensity and duration of the stimulation and were told that the 4 settings would be presented randomly and these could suppress, elevate, or have no effect on their tinnitus.
Participants were randomly allocated to receive either LTA or DLPFC stimulation first (13 participants received LTA stimulation first, 14 participants received DLPFC stimulation first). The washout period between the 2 sessions was 1 week. In total, each participant underwent 2 sessions of 4 HD-tDCS stimulation settings in increasing dose. The sessions were 1 week apart.
Hearing Assessment
All the recruited participants underwent hearing assessment. Pure tone audiometry (0.125-16 kHz) was undertaken using a 2-channel audiometer (Madson Itera II) with supra-aural (Telephonics, TDH–50P, frequencies 0.125-8 kHz) and high-frequency circumaural headphones (Sennheiser HDA 200, frequencies 9-16 kHz). Audiometry was performed using the modified Hughson-Westlake procedure, 28 in a sound proof room (ISO 82531-2009).
Clinical Evaluation
A Tinnitus Case History Questionnaire 29 and the Tinnitus Functional Index (TFI) 30 questionnaire was completed by participants. Tinnitus loudness and annoyance were rated using a 9-point rating scale where 1 was not audible/not annoying and 9 was as loud/annoying as imaginable. Participants rated their tinnitus loudness and annoyance twice before the HD-tDCS stimulation as baseline (the second rating after arrival was used as baseline for evaluation, to accommodate the influence of change in environment [arrival in sound treated room]) and after the completion of each stimulation setting. Postevaluation ratings were used as baseline for later stimulations.
The criterion for tinnitus loudness and annoyance suppression used in this study was a minimum of 1-point reduction in the ratings following stimulation.20,31 If total tinnitus suppression was obtained (defined as not being able to hear tinnitus at all), participants returned for the next stimulation a minimum of 24 hours following the previous stimulation (to allow for an extinction of effect) otherwise participants received the next stimulation (up to a maximum of all 4 stimulations in 1 session). Participants rated their perception and sensations experienced while undergoing HD-tDCS after every stimulation setting. 32
High-Definition Transcranial Direct Current Stimulation
High-definition tDCS was conducted using Neuroconn DC-Stimulator PLUS version (Ilmenau Germany) and 4 × 1 multichannel stimulation adaptor from Soterix Medical Inc, New York, NY. Sintered silver/silver chloride (Ag/AgCl) ring electrodes were used with inner radius of 6 mm and outer radius of 12 mm. Ring electrodes were placed using the international 10/20 electroencephalographic (EEG) system guidelines. The ring electrodes were stabilized using plastic holders hooked in the EEG recording cap (EASYCAP, Herrsching, Germany) and filled with EEG conduction gel (Signa gel, Parker Laboratories, Inc, Fairfield, NJ) according to the guidelines provided by Villamar et al 32 for 4 × 1 HD-tDCS stimulation. The gel-skin contact area was approximately 25 ± 2.5 mm2. For the LTA stimulation, the anode was placed halfway between C3 and P5; 4 adjoining cathodes were placed at C5, TP7, CP3, and P5, respectively. During the right DLPFC stimulation, the anode was placed at F4 and 4 adjoining cathodes were placed at F2, FC4, F6, and AF4, respectively. Cathodes were approximately 3.5 cm away from the anode in both stimulation montages (LTA and DLPFC). The participants did not wear any ear plugs during the HD-tDCS testing; however, the examiner was sitting in a second room (of a 2-room sound booth [ISO 8253-1:2010] with a glass window) entering for administering rating scales and changing the HD-tDCS settings.
Data Analysis
The changes in tinnitus loudness and annoyance ratings were analyzed using SPSS software (version 19; IBM, Armonk, NY). Tinnitus loudness and annoyance ratings were analyzed using 2 separate 3-way repeated-measures analyses of variance with current intensity (1 mA, 2 mA), duration (10 minutes, 20 minutes), and site of stimulation (LTA, DLPFC) as factors. Mauchly’s test for sphericity indicated that the sphericity assumption of the repeated-measures analysis of variance was met. A criterion for statistical significance of .05 was chosen. T-tests were performed to explore if the responders and nonresponders to HD-tDCS differed in their hearing loss and tinnitus severity (TFI).
Results
Twenty-seven participants (mean age 53.5 years, age range 23-73 years, 5 female, 22 male) with chronic tinnitus (>2 years) completed this study. Participants rated the sensations experienced while undergoing HD-tDCS and their responses are summarized in Tables 2 and 3. Before the actual stimulation, participants sat for few minutes with the electrodes cap and no stimulation; however, they were not aware of the absence of stimulation, their ratings, and self-reported sensations they experienced were recorded. Overall, HD-tDCS was well tolerated by all the participants. Any reported sensations of headache, neck pain, scalp pain, sensation of scalp burn, tingling, sleepiness, trouble concentrating, mood change, were brief and at the onset of stimulation. A tingling sensation, sleepiness, sensation of scalp burn, and scalp pain were the most commonly encountered sensations, and these were experienced mainly for the 2-mA, 10-minute stimulation.
Sensations Experienced During HD-tDCS of DLPFC (n = 27 Participants).
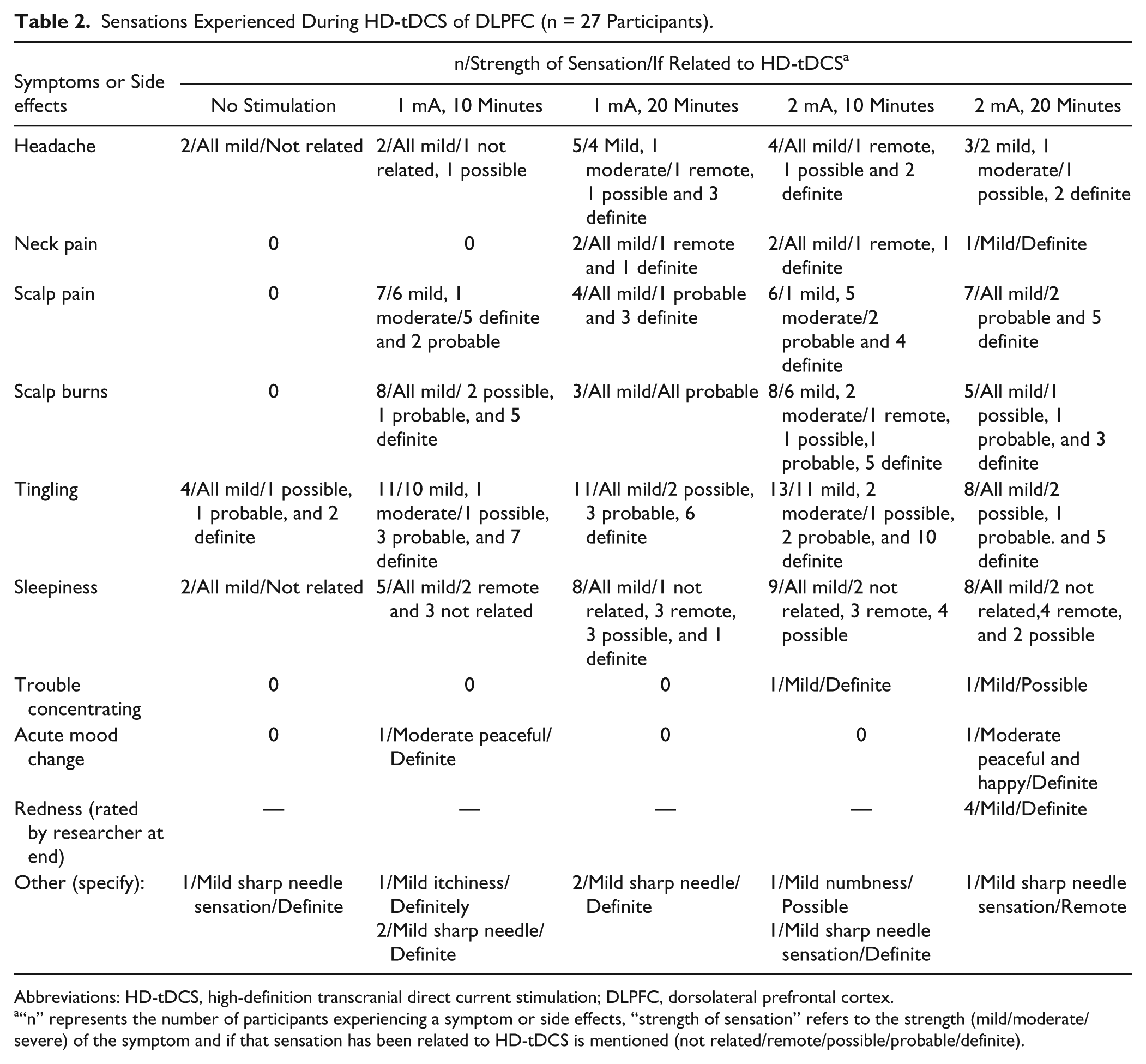
Abbreviations: HD-tDCS, high-definition transcranial direct current stimulation; DLPFC, dorsolateral prefrontal cortex.
“n” represents the number of participants experiencing a symptom or side effects, “strength of sensation” refers to the strength (mild/moderate/severe) of the symptom and if that sensation has been related to HD-tDCS is mentioned (not related/remote/possible/probable/definite).
Sensations Experienced During HD-tDCS of LTA (n = 27 Participants).
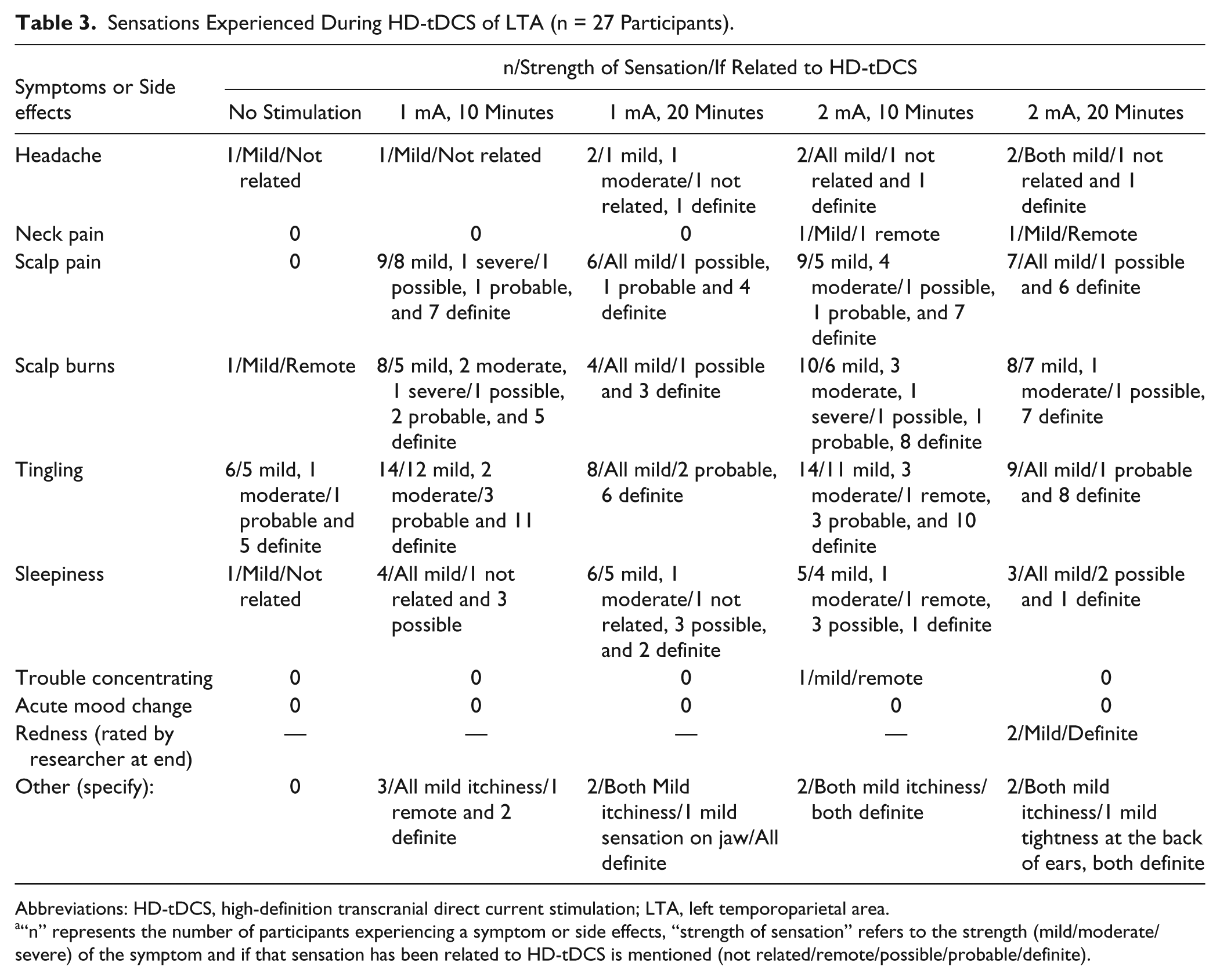
Abbreviations: HD-tDCS, high-definition transcranial direct current stimulation; LTA, left temporoparietal area.
“n” represents the number of participants experiencing a symptom or side effects, “strength of sensation” refers to the strength (mild/moderate/severe) of the symptom and if that sensation has been related to HD-tDCS is mentioned (not related/remote/possible/probable/definite).
The first hypothesis proposed was that 2 mA HD-tDCS current intensity will be more effective in suppression of tinnitus loudness and annoyance than 1 mA. The stimulus with 2 mA current intensity caused more reduction in both tinnitus loudness, F(1, 26) = 16.84, P < .001, and annoyance, F(1, 26) = 14.24, P = .001 (Figure 3).
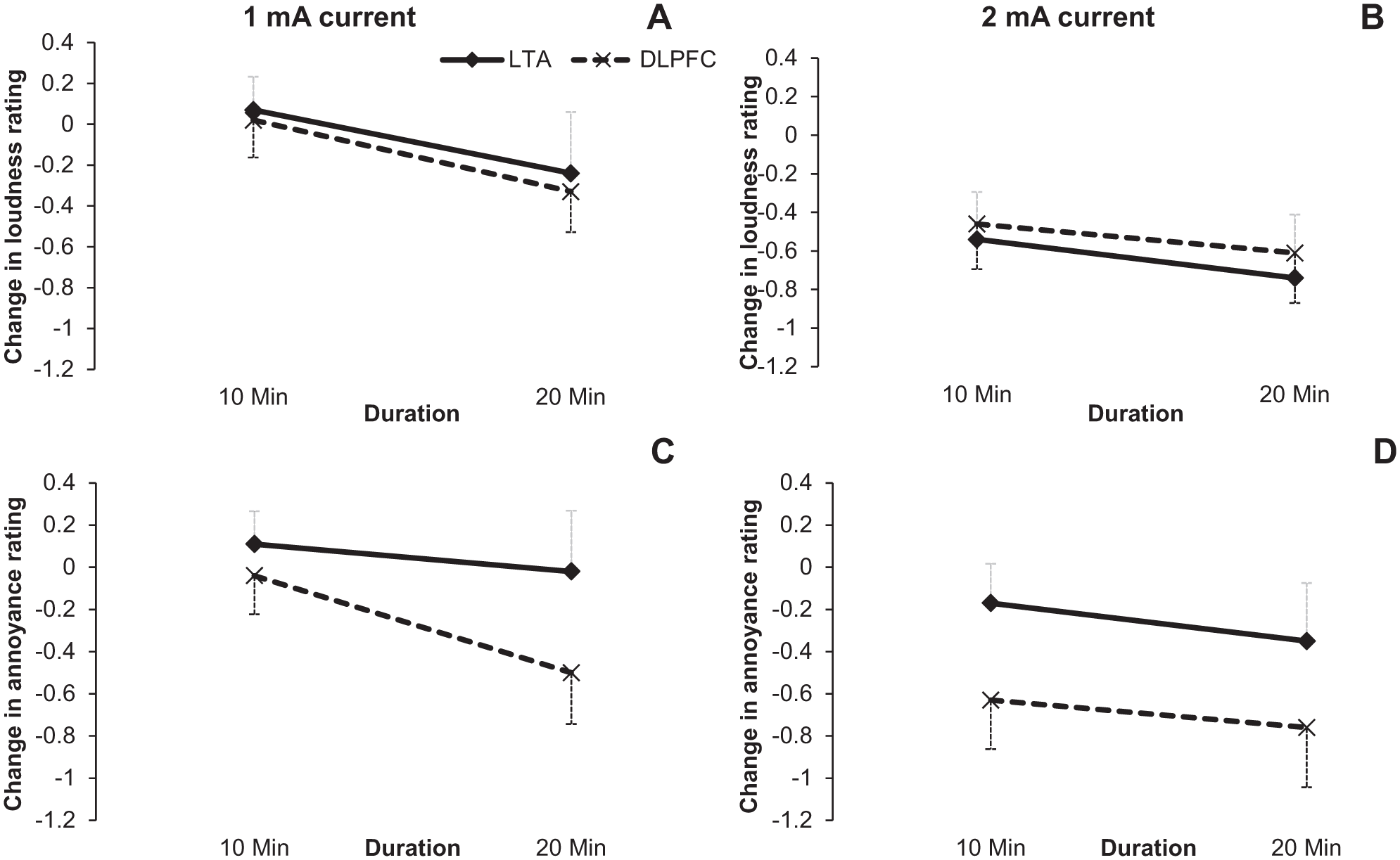
Loudness change (A, B) and annoyance change (C, D) during 1 mA and 2 mA HD-tDCS stimulation. X-axis represents the stimulation duration in min and Y –axis is change in the loudness/annoyance rating. Error bars represent ± 1 standard deviation of the mean.
The second hypothesis proposed was that 20-minute HD-tDCS duration will be more effective in suppressing tinnitus loudness and annoyance than 10 minutes. The 20-minute stimulus duration resulted in a greater reduction than the shorter in tinnitus loudness, F(1, 26) = 10.08, P = .004, and annoyance, F(1, 26) = 6.88, P = .01 (Figure 4).
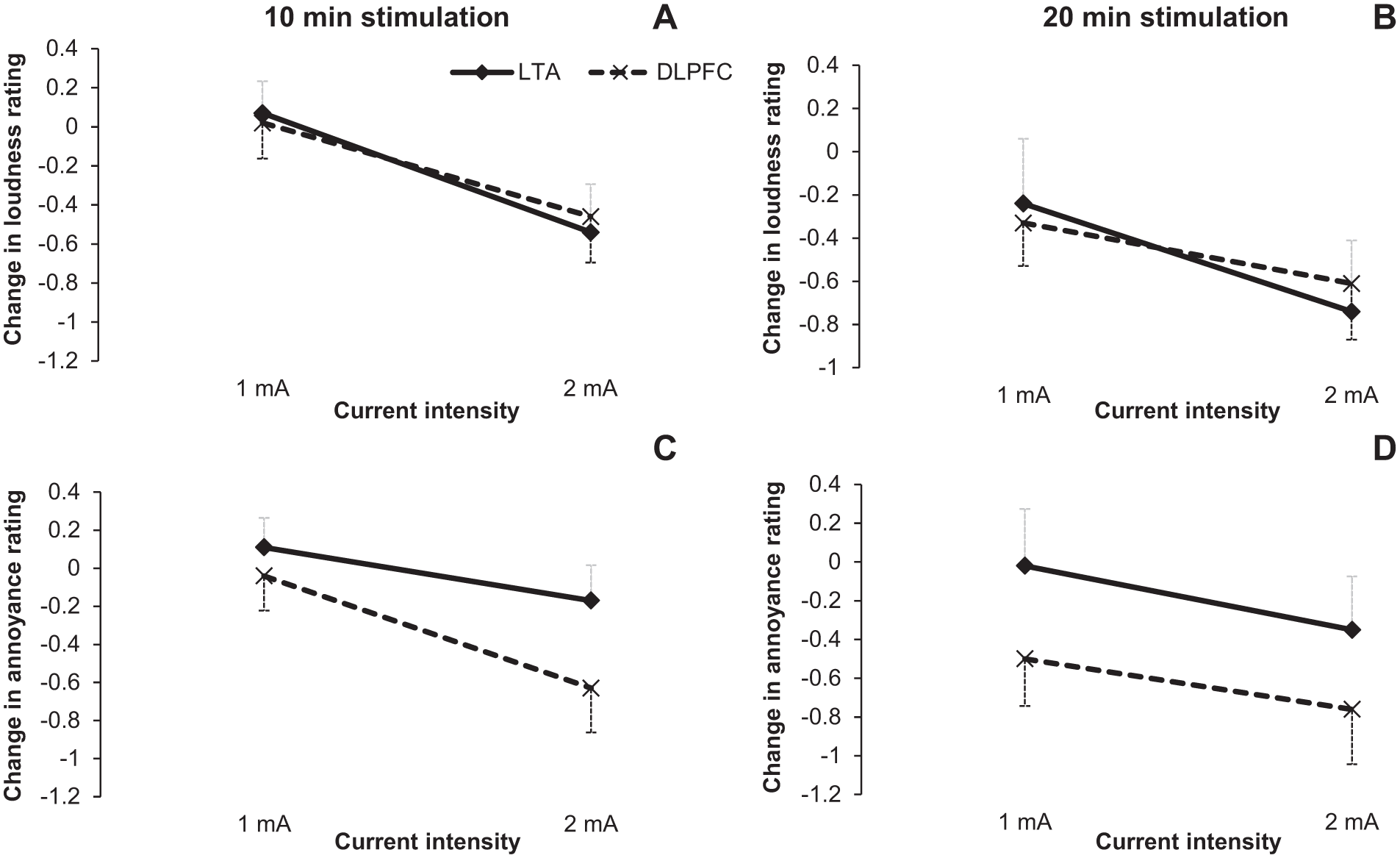
Loudness change (A, B) and annoyance change (C, D) during 10- and 20-min high-definition transcranial direct current stimulation (HD-tDCS) stimulation. X-axis represents the stimulation intensity in milliamperes (mA) and Y-axis is change in the loudness/annoyance rating. Error bars represent ± 1 standard deviation of the mean.
These results supported the hypotheses that higher current intensity (2 mA) and longer duration (20 minutes) were more effective in suppressing tinnitus.
The third hypothesis was that LTA stimulation would result in greater tinnitus loudness suppression than DLPFC stimulation. We found slightly more reduction in tinnitus loudness with LTA stimulation than DLPFC stimulation (−0.74 vs −0.61), though this was not statistically significant, F(1, 26) = 0.003, P = .960.
The fourth hypothesis was that tinnitus annoyance would be suppressed more by DLPFC stimulation than LTA stimulation. Tinnitus annoyance was suppressed slightly more with DLPFC stimulation than LTA stimulation (−0.76 vs −0.35) but this again did not reach the criterion for statistical significance, F(1, 26) = 1.646, P = .211). There was no significant 2- or 3-way interaction between site of stimulation, stimulus duration, and current intensity for tinnitus loudness or annoyance change (all P values >.1).
To explore the data more fully, we compared the groups who reported the most and the least change in tinnitus with HD-tDCS stimulation. The criterion used for tinnitus loudness and annoyance suppression in this study was a minimum of 1-point reduction in the rating scales. Out of the 27 participants tested in this study, 21 reported some benefit, 22% (n = 6) reported both tinnitus loudness and annoyance suppression with both LTA and DLPFC stimulation, 26% (n = 7) reported reduction in either loudness or annoyance with both LTA and DLPFC stimulation, 22% (n = 6) experienced a worsening or no change in tinnitus loudness and annoyance with both LTA and DLPFC stimulation, and 30% (n = 8) reported improvement in either tinnitus loudness or annoyance with either LTA or DLPFC stimulation.
We conducted an exploratory analysis to investigate if the degree of hearing loss, severity of tinnitus, tinnitus onset, laterality, and tinnitus pitch were linked to responsiveness toward HD-tDCS. Those participants who reported tinnitus loudness and/or annoyance suppression with both LTA and DLPFC stimulation were grouped as “responders” (n = 13) and the others as “nonresponders” (n = 14). Responders had slightly better hearing thresholds (30.6 dB) than nonresponders (37.5 dB); however, this difference was not statistically significant, t(25) = −1.031; P = .312; Cohen’s d = 0.32. Responders had higher overall TFI scores (42.95) than nonresponders (29.95), this difference did not reach statistical significance, t(25) = 1.445; P = .161; Cohen’s d = 0.56. Tinnitus onset, t(22) = −0.47, P = .63; laterality, t(4, n = 27) = 2.78, P = .59; and tinnitus pitch, t(2, n = 27) = 0.79, P = .67, were not related to responsiveness toward HD-tDCS.
Discussion
High-definition tDCS appears to be safe and well tolerated; none of the participants experienced a significant adverse event or reason to stop testing. Some physiological indication that stimulation was being provided most common being mild tingling was experienced by majority of participants. Twenty-one of the 27 participants (77.78%) experienced transient tinnitus loudness and/or annoyance suppression following HD-tDCS. This response rate is higher than previous tDCS trials 20 ; we speculate this could potentially be due to the more focal nature of HD-tDCS compared to conventional tDCS. 10
Dorsolateral prefrontal cortex stimulation resulted in slightly more annoyance suppression (compared with LTA stimulation) and LTA stimulation led to more loudness suppression (compared with DLPFC stimulation); however, the difference in the loudness and annoyance suppression between the two sites of stimulation was not statistically significant. This supports previous research, which has independently reported loudness and annoyance suppression with both LTA18-20,31 and DLPFC33-35 stimulation. LTA and DLPFC stimulation were equally effective in suppressing tinnitus loudness and annoyance. There is some suggestion that tDCS of DLPFC modulates annoyance and not loudness 35 and LTA might modulate loudness but not tinnitus annoyance. 36 However, this view has been challenged by Vanneste et al,33,37-40 and we found no strong evidence for this here.
During the application of HD-tDCS, the distance between the anode and adjoining cathode was approximately 3.5 cm, which makes it more focal than conventional tDCS 10 ; however, current flow will still result in stimulation of adjoining areas above the LTA and DLPFC, making it difficult to rule out the stimulation of various cortical and subcortical areas (primary auditory cortex, auditory association area, amygdala, hippocampus).3,16,17 Stimulation of LTA and DLPFC could lead to modulation of tinnitus loudness and annoyance via competition or inhibitory control of DLPFC on the auditory cortex. 19 In future studies, it would be useful to explore the impact of a larger distance (7 cm radius) between anode and cathodes. Considering tinnitus perception is believed to be due to a network of various cortical and subcortical areas, 41 we anticipate that large radius (7 cm) stimulation may modulate tinnitus loudness and annoyance more than stimulation using the small radius (3.5 cm). We are currently conducting a study exploring the impact of large (7 cm) and small (3.5 cm) radius of HD-tDCS on tinnitus perception.
The difference between the hearing thresholds and TFI scores between the responders and nonresponders did not research statistical significance in our study. Participants who responded to HD-tDCS tended to have better hearing and higher mean TFI scores compared with nonresponders, suggesting a possibility that people with less severe hearing loss and higher TFI score may respond better to HD-tDCS. Other possible explanations are random fluctuation in the data and/or regression to the mean. Therefore, further research may confirm these hypothesis.
Positive response rate toward tDCS in previous studies (defined as a minimum of 1 point reduction18-20,31 or any change21,33,37-40,42 in tinnitus loudness and annoyance rating) have ranged between 29.9% and 56%. This wide range in responsiveness toward tDCS could be due to differences in a variety of factors, including the response criteria, research design, protocol, washout period, stimulation parameters, and potentially in many studies as placebo effect. We used the criterion of a minimum 1 point reduction as defined in previous studies18-20,31 and found a total of 77.78% (n = 21) participants responded positively to HD-tDCS and reported tinnitus suppression. This is a very high positive response rate compared with past tDCS studies,18-21,31,33-40,42,43 which could be due to the relatively focal and lasting effect of HD-tDCS compared with tDCS; however, a head to head comparison of tDCS and HD-tDCS is required. We plan to conduct such comparison in our laboratory in future work.
Previous studies have used LTA, DLPFC, and auditory cortex as sites of stimulation for tinnitus relief. This is one of the first studies comparing LTA and DLPFC for tinnitus modulation. Our study revealed modulation of tinnitus loudness as well as annoyance with both sites of stimulation; it would be beneficial to explore ways of stimulating multiple target sites simultaneously and also to explore other possible sites of stimulation that may modulate tinnitus.
Limitations
This study was not an efficacy trial; it was a phase II, open-label within-subject treatment optimization design, dose-response study exploring the immediate impact of HD-tDCS current intensity, duration, and location on modulation of tinnitus loudness and annoyance. It was difficult to rule out the cumulative impact of the 4 HD-tDCS settings used. Additionally, this study was not a sham controlled trial. However, participants were told that 4 different HD-tDCS settings will be randomly used which could suppress, elevate, or have no effect on their tinnitus loudness and annoyance. However, our results support ongoing trials on the efficacy and mechanisms of HD-tDCS treatment of tinnitus with carefully matched sham protocols. 44
Conclusions
High-definition tDCS appears to be a safe and well-tolerated neuromodulation technique for transient tinnitus suppression. A high (77.78 %) positive response rate to HD-tDCS was observed in this study. The stimulation of the LTA and DLPFC were equally effective for suppressing tinnitus loudness and annoyance. A current intensity of 2 mA delivered for 20 minutes was the winning setting for tinnitus suppression. There may be a possibility of differential impact of LTA and DLPFC stimulation on loudness and annoyance suppression; however, further research is required to test this hypothesis.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this project was provided by the Auckland Medical Research Foundation Jean Cathie project grant.
