Abstract
Background. Poststroke dysphagia can persist, leading to many complications. Objective. We investigated whether noninvasive brain stimulation to the pharyngeal motor cortex combined with intensive swallowing therapy can improve dysphagia. Methods. A total of 20 patients who had dysphagia for at least 1 month after stroke were randomly assigned to receive 10 sessions lasting 20 minutes each of either 1-mA anodal transcranial direct current stimulation (tDCS) or a sham procedure to the ipsilesional pharyngeal motor cortex, along with simultaneous conventional swallowing therapies. We evaluated swallowing function with the Dysphagia Outcome and Severity Scale (DOSS) before, immediately after, and 1 month after the last session. Results. Anodal tDCS resulted in an improvement of 1.4 points in DOSS (P = .006) immediately after the last session and 2.8 points (P = .004) 1 month after the last session. The sham tDCS group improved 0.5 points (P = .059) after the last session and 1.2 points (P = .026) 1 month after the final session. The improvements in the anodal tDCS group were significantly greater than those in the sham tDCS group (P = .029 after the last session, and P = .007 1 month after the last session). Conclusions. Anodal tDCS to the ipsilesional hemisphere and simultaneous peripheral sensorimotor activities significantly improved swallowing function as assessed by the DOSS.
Keywords
Introduction
In addition to stroke treatment, it is important to address poststroke complications. Dysphagia is prevalent in acute stroke patients, and most (more than 50%) regain swallowing abilities within 1 month after stroke. 1 In chronic stroke patients, the prevalence of dysphagia is less than 10%. 2 Prolonged swallowing problems in stroke patients are associated with many complications, including dehydration, malnutrition, aspiration, respiratory infection, and death. 3 Ipsilesional hemisphere brain function is believed to be compensated for with increasing activity of the area responsible for swallowing in the contralesional hemisphere. 4 The aim of managing acute stroke dysphagia is to achieve natural recovery of swallowing function and to prevent pneumonia, dehydration, and malnutrition, which hamper patient prognosis. In subacute and chronic patients, various therapies can restore swallowing function, and efforts have been made to develop new treatment methods; however, only a small number of these therapies are effective. Furthermore, the pathology of dysphagia is diverse. In general, compensatory methods, such as position adjustment and diet modification are used to treat poststroke dysphagia.
Swallowing movements are produced by a central pattern generator located in the rostral brainstem, 5 and neural control centers for swallowing are located in the bilateral frontal cortex anterior to the sensorimotor cortex. Sensory feedback from peripheral structures and nerves plays an important role in swallowing function, which is closely associated with both cortical and subcortical regions, the brainstem, and peripheral structures. Although many conventional dysphagia treatment strategies typically involve peripheral nerve stimulation or peripheral sensorimotor activities, there have been few attempts to facilitate cortical region stimulation to improve swallowing function.
Recent studies suggest that brain stimulation can enhance the beneficial effects of motor function in the rehabilitation process. 6 Repetitive transcranial magnetic stimulation (rTMS) and transcranial direct current stimulation (tDCS) have been investigated as potential tools for facilitating recovery of deficits, such as those related to motor function, 7 aphasia, 8 and cognitive functions. 9 tDCS is applied through 2 surface electrodes placed on the skull. Anodal and cathodal tDCS increase and decrease cortical excitability, respectively. In the present study, we investigated whether the application of tDCS to the cortical motor and sensory pharyngeal areas can improve swallowing function in poststroke patients.
Patients and Methods
This was a prospective, single-center, single-blind trial approved by the Ethics Committee of Hamamatsu City Rehabilitation Hospital. Inclusion criteria were occurrence of stroke at least 4 weeks prior to enrollment; admitted to our hospital for rehabilitation after stroke; chronic, severe poststroke dysphagia; and need for tube feeding therapy. Exclusion criteria consisted of subarachnoid hemorrhage, history of epileptic seizures, severe consciousness disturbance, organic neck disease, or history of surgery, except for tracheotomy. We included 23 patients with poststroke dysphagia and no other muscular and neurological disorders who had undergone treatment and rehabilitation. All were able to understand the purpose of the study and provided written informed consent. The site, type, and size of stroke were identified on the basis of patient history and computed tomography findings on admission. Motor function and overall neurological disability were assessed with the motor function items of the Functional Independence Measure (motor FIM), 10 which consists of 13 items to evaluate functional dependence; the score ranges from 13 for total disability/dependence to 91 for full function/independence.
Clinical Swallowing Assessments
Swallowing function was clinically evaluated by rehabilitation doctors and speech-language-hearing therapists specializing in dysphagia who were blinded to study allocation. All patients were rated for swallowing impairments with the validated Dysphagia Outcome and Severity Scale (DOSS). 11 The DOSS score ranges from 1 to 7, with 1 representing severe dysphagia, and 7 representing normal swallowing. The DOSS is a very useful scale for measuring the outcomes of recommended diet, independence level, and type of nutrition. We compared the DOSS scores before the first stimulation session (pre-DOSS), immediately after the last session (post-DOSS), and 1 month after the last session (1M-DOSS).
Swallowing Examinations and Therapies
Patient swallowing function was examined using both videofluoroscopy (VF) and videoendoscopy (VE). For VF, we used barium-containing foods, including jelly (semisolid), honey liquid, nectar liquid, puree solid, thin liquid, and cookies (hard solid). We evaluated the presence or absence of pharyngeal residue and Penetration Aspiration Scale 12 with a VF study. For VE, we assessed anatomic abnormalities of the pharynx, vocal cord, and pharyngeal muscle movement and pharyngeal residue of saliva and foods. Patients received intensive swallowing therapies with simultaneous brain stimulation. Training content was determined according to the VF and VE findings rather than lesion location. Patients who were at risk of aspiration and could not meet the safety conditions required for oral intake were tube fed and underwent nonoral intake rehabilitation (indirect therapy). On the other hand, patients who could eat and drink safely with compensatory conditions received both oral intake rehabilitation (direct therapy) and indirect therapy. 13 Indirect therapy consisted of blowing, ice massage, 14 pushing exercises, 15 supraglottic swallowing, 16 Shaker exercise, 17 effortful swallow, 18 and K-point stimulation 19 (stimulating the trigger point for opening the mouth and facilitating the swallowing reflex in pseudobulbar palsy patients). Patients with swallow delay received training to induce the swallow reflex, such as ice massage and K-point stimulation. Patients with pharyngeal residue received muscle-strengthening exercise, such as the Shaker exercise. Intensive swallowing therapies were continued after the final brain stimulation session.
tDCS Protocol
tDCS was delivered with a DC stimulator (neuroConn GmbH, Granzhammer, Germany) through 2 saline-soaked electrodes. The machine was programmed according to the anodal/sham stimulation mode. Patients were randomly issued a code number by one of the coauthors without knowledge of patient background to receive 20 minutes of either 1-mA anodal tDCS or sham tDCS for 10 days (once a day and 2 blocks of 5 days). The anodal electrodes were placed over the ipsilesional hemisphere, 15 cm from Cz to A1 and 2 cm in the front direction on the right and from Cz to A2 in the front direction on the left according to the international 10-20 EEG electrode system 20 ; the cathode was placed over the contralesional supraorbital region. This anodal site was considered the pharyngeal motor cortex. The same electrode positions were used in the sham stimulation. The current was ramped up to 1 mA and slowly decreased over 40 s to ensure an initial tingling sensation. 21 The patients received both stimulation and conventional swallowing therapy at the same time.
Statistical Analysis
To analyze the effects of tDCS on swallowing function improvement, a rehabilitation doctor and speech-language-hearing therapists who did not know the patient’s group assessed the 3 DOSS scores. A responder value was defined as at least a >1 point improvement on the DOSS. Although we needed 14 patients in each group, we studied 10 cases each. All results are expressed as mean ± standard deviation (SD) values. Because DOSS score changes were not normally distributed, the improvements in each group were compared using Wilcoxon signed-rank test, and improvement in both groups was compared using the Mann-Whitney U test. Differences were considered significant when the probability (P) of a type I error was .05 or less. All statistical analyses were performed using SPSS, version 20 (IBM Corp, Chicago, Illinois IL).
Results
Participation
Figure 1 shows participant flow through the study. In all, 23 patients agreed to participate in this study, but 3 patients were excluded before starting the stimulation treatment because of respiratory complications. We randomly divided 20 patients (13 men, 7 women; mean ± SD, 65.8 ± 7.8 years) into 2 groups: anodal tDCS and sham tDCS. Patient demographic and clinical characteristics are summarized in Table 1. Radiologic examination revealed that 13 patients had supratentorial stroke, and 7 had infratentorial stroke. The duration from onset to treatment was between 5 and 38 weeks (mean ± SD = 12.5 ± 8.4). The motor FIM score ranged from 13 to 66 (mean ± SD = 34.1 ± 16.8). All patients completed all sessions, and no patient was excluded because of pain or other reasons. In addition, there were no complications during the entire study period, except a mild tingling sensation at the site of electrode placement at the beginning of the stimulation, which is a common finding across different studies. 21 Before brain stimulation, 3 patients in the anodal tDCS group (A3, A6, and A7) and 2 patients in the sham tDCS group (S8 and S9) only received nasogastric tube nutrition. Two and 3 patients in the anodal tDCS and sham tDCS groups received totally oral intake nutrition, respectively. A4 received total parenteral nutrition, and S8 received tube feeding from gastrostomy in combination with oral intake rehabilitation. After brain stimulation and 1-month swallowing rehabilitation, all patients in the anodal tDCS group received oral intake nutrition exclusively. In contrast, 2 patients in the sham tDCS group required nasogastric tube feeding.
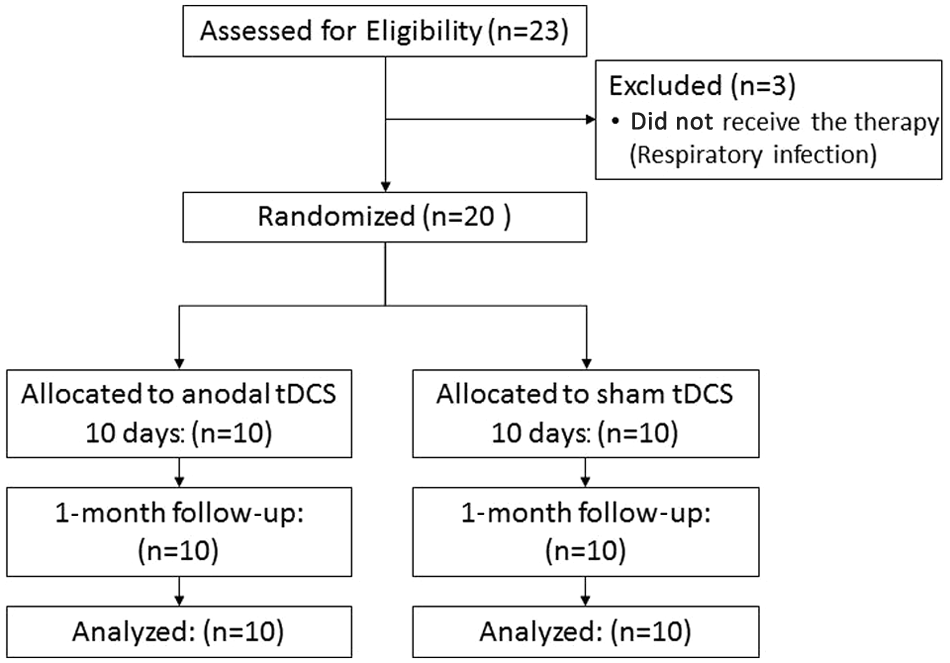
Participant flow. In all, 23 patients were assessed for eligibility for this study. Among these, 3 patients were excluded because they could not receive brain stimulation and swallowing therapies because of respiratory infection. The remaining 20 patients were randomly allocated to 2 groups: anodal tDCS and sham tDCS. All patients received 10 sessions of stimulation and were followed up for 1 month. There were no complications during the whole period.
Patient Characteristics a
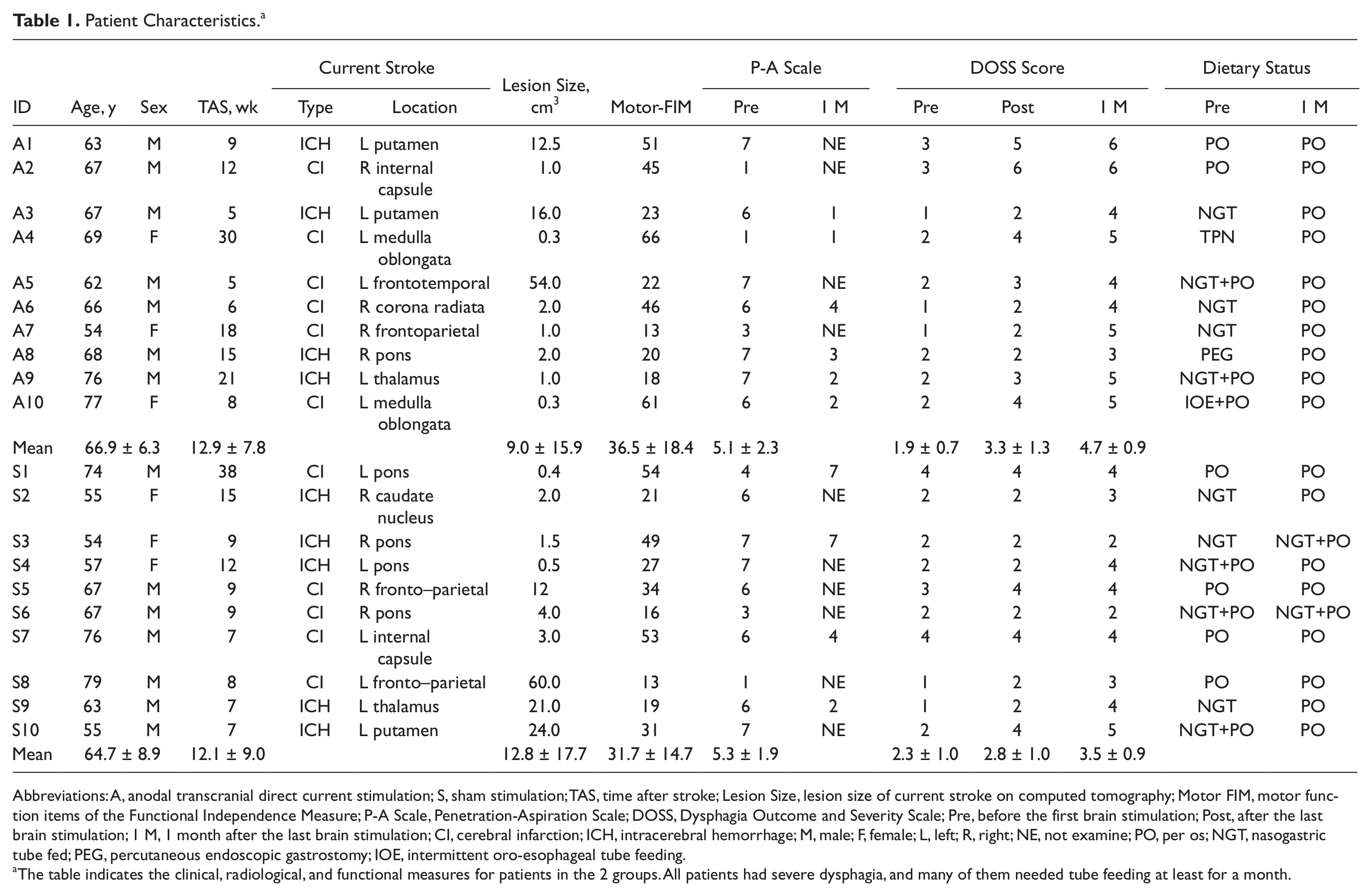
Abbreviations: A, anodal transcranial direct current stimulation; S, sham stimulation; TAS, time after stroke; Lesion Size, lesion size of current stroke on computed tomography; Motor FIM, motor function items of the Functional Independence Measure; P-A Scale, Penetration-Aspiration Scale; DOSS, Dysphagia Outcome and Severity Scale; Pre, before the first brain stimulation; Post, after the last brain stimulation; 1 M, 1 month after the last brain stimulation; CI, cerebral infarction; ICH, intracerebral hemorrhage; M, male; F, female; L, left; R, right; NE, not examine; PO, per os; NGT, nasogastric tube fed; PEG, percutaneous endoscopic gastrostomy; IOE, intermittent oro-esophageal tube feeding.
The table indicates the clinical, radiological, and functional measures for patients in the 2 groups. All patients had severe dysphagia, and many of them needed tube feeding at least for a month.
Brain Stimulation and DOSS Score Improvement
As shown in Table 1, there was no significant difference between the 2 groups in regard to age, sex, stroke type, lesion site, duration from onset to stimulation, Penetration-Aspiration Scale scores, and DOSS scores before stimulation sessions (pre-DOSS). As shown in Figure 2, we observed increased DOSS scores in the anodal tDCS group from 1.9 ± 0.7 to 3.3 ± 1.3 (post-DOSS) and 4.7 ± 0.9 (1M-DOSS). Both post-DOSS and 1M-DOSS were significantly increased from baseline (P = .006 and P = .004, respectively). In the sham tDCS group, DOSS score increased from 2.3 ± 1.0 to 2.8 ± 1.0 (post-DOSS) and 3.5 ± 0.9 (1M-DOSS). The sham-tDCS exhibited a significant increase in the 1M-DOSS but not the post-DOSS (P = .059, post-DOSS; P = .026, 1M-DOSS).
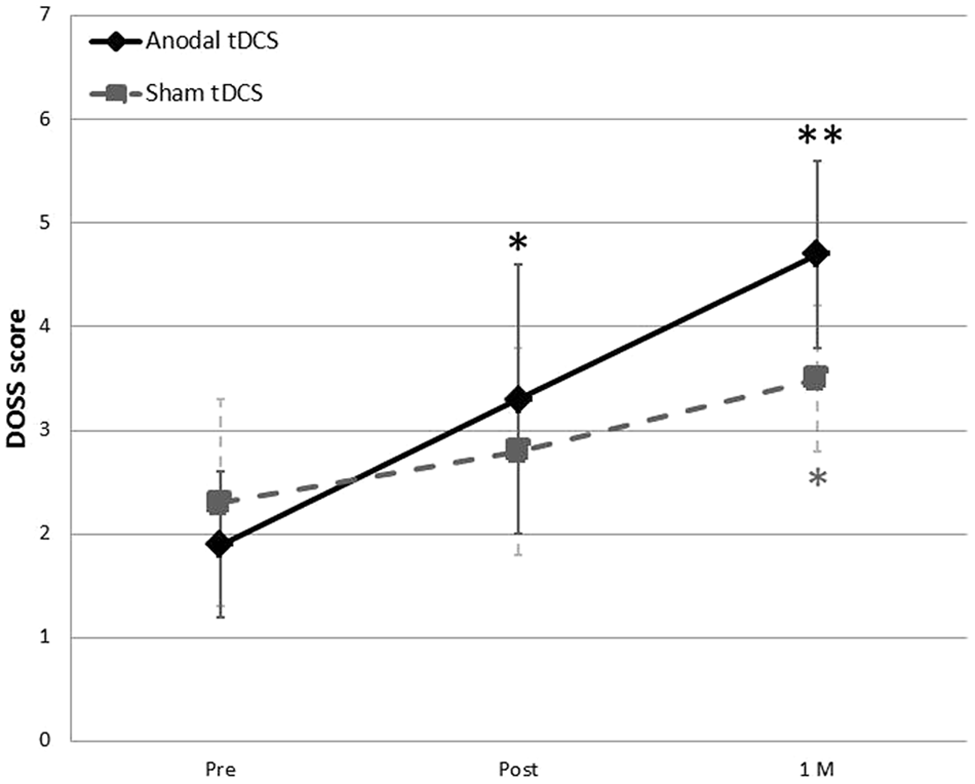
Change in DOSS score (anodal tDCS and sham tDCS). *P < .05; **P < .01. In anodal tDCS, the DOSS score was significantly improved at both post (P = .006) and 1 M (P = .004), but in sham tDCS, only the 1 M (P < .026) score improved.
The DOSS score improved significantly with anodal tDCS as compared with sham tDCS (Mann-Whitney U test, P = .029 for pre vs post, and P = .007 for pre vs 1 M).
Discussion
The present study showed that anodal tDCS combined with conventional swallowing therapies effectively improved poststroke dysphagia as compared with swallowing training alone.
Recently, noninvasive cortical stimulation has been used to enhance neural plasticity and treat hemiplegia 7 and aphasia. 8 However, little is known about the possible effects of tDCS on swallowing function. Only Kumar et al 22 have reported that anodal tDCS improves swallowing function associated with acute stroke dysphagia. However, much poststroke dysphagia is transient, and it is likely that tDCS does not improve dysphagia itself in the acute stage; rather, it improves consciousness disturbance, and dysphagia recovers spontaneously. In addition, they did not examine the long-term effects of tDCS treatment. In the present study, we only included patients with prolonged dysphagia for more than 4 weeks to exclude those with transient dysphagia. Furthermore, we selected patients who were tube fed after the onset of stroke to reduce the effects of malnutrition and dehydration. We also examined swallowing function after 1 month and found that the effects of tDCS were sustained.
We focused on the importance of brain stimulation in combination with conventional swallowing exercises. Studies in rat models have shown that combining rehabilitative training with cortical electrical stimulation resulted in improved functional outcome and greater cortical neuronal structural plasticity. 23 In a study of chronic stroke patients, the combination of tDCS and peripheral sensorimotor activities 24 or peripheral nerve stimulation 25 improved motor functions. Therefore, we hypothesized that cortical stimulation in combination with peripheral sensorimotor activities would produce greater swallowing function improvement. Swallowing movements occur in association with the cortex and subcortical regions, brainstem, and peripheral structures, such as nerves and muscles, and sensory feedback from these structures plays an important role in swallowing. Many traditional types of swallowing training are designed to enhance sensory input from peripheral nerves or strengthen the swallowing muscles and are effective in improving swallowing function. On the other hand, noninvasive brain stimulation is clinically applied to treat various disabilities. tDCS is one noninvasive brain stimulation technique that utilizes weak direct current to shift neural excitability.26,27 In previous reports, tDCS at intensities of 1 to 2 mA was applied through 2 surface electrodes placed on the skull. Depending on stimulation duration and polarity, tDCS can increase or depress excitability in the stimulated region from a few minutes to 1 to 2 hours. 9 Jefferson et al 28 reported that anodal tDCS could alter pharyngeal motor cortex excitability, which might affect the brainstem swallowing center. The present study showed that the combination of anodal tDCS and intensive swallowing therapies improved swallowing function to a greater extent than swallowing therapies only.
We made several interesting observations during the course of this study. First, we applied anodal tDCS to the ipsilesional hemisphere. Although previous studies of anodal tDCS for limb function or aphasia have stimulated the ipsilesional hemisphere, in the case of dysphagia, Kumar et al 22 reported that anodal tDCS to the contralesional hemisphere in acute stroke dysphagia could improve swallowing function. In general, pharyngeal muscles are represented asymmetrically between the 2 cortical hemispheres, and anodal tDCS to both hemispheres is probably effective in improving swallowing function. However, recent reports have described some mechanisms underlying the beneficial effects of rehabilitative interventions and functional recovery after stroke, including increased blood flow to different cortical areas, 29 cortical plastic reorganization,30,31 and recovery of diaschisis. 32 Some functional neuroimaging studies suggested that ipsilesional motor regions could be targeted because the reactivation of intact portions of the ipsilesional motor cortex is associated with better outcome after stroke.33,34 Second, with regard to comparing the 2 noninvasive brain stimulation techniques, both rTMS and tDCS are noninvasive and appear to be safe when used according to established safety guidelines. Compared with tDCS, rTMS equipment is more expensive but allows focal stimulation and better temporal resolution; on the other hand, tDCS is inexpensive and is easier to simultaneously apply with other training protocols compared with rTMS. 9 In addition, sham tDCS is easier to perform than sham rTMS for double-blind experimental designs. 21
The strengths of the present study were the success in starting oral intake without tube feeding for many postacute stroke patients with severe dysphagia. All patients suffered from some dysphagia symptoms for at least 1 month. Furthermore, many patients required tube feeding because of problems associated with dysphagia, such as aspiration or pharyngeal residue. Many previous studies have targeted acute dysphagia patients; however, few studies have assessed postacute stroke patients.
We selected the stimulation site according to a previous Japanese article and our previous study in healthy volunteers, without the use of Neuro Navigation, such as near-infrared spectroscopy (NIRS) or TMS. tDCS electrodes were wide enough to cover the area we wanted to stimulate. We applied 1 mA for 20 minutes, which is sufficient to safely excite the pharyngeal motor cortex. However, additional studies are necessary to investigate optimal tDCS parameters.
The present study has several limitations. First, we could not collect the sample number of cases expected, and the variation in time from onset to stimulation was rather wide. We included patients with swallowing problems for more than 4 weeks to exclude patients who might improve spontaneously. Second, the DOSS score is not suitable to evaluate improvement in short-term swallowing. Therefore, it might be desirable to evaluate the swallowing function using VF and VE. Third, we examined all patients by VF before the intervention but could not perform VF for all for reevaluation. Fourth, we included both supratentorial and infratentorial lesions. Although almost all supratentorial patients showed pseudobulbar dysphagia, infratentorial patients showed both pseudobulbar and bulbar dysphagia. It is unknown whether tDCS might be effective for bulbar palsy. Khedr and Abo-Elfetoh 35 reported that rTMS could be a useful adjuvant strategy for neurorehabilitation of dysphagia caused by lateral medullary syndrome or other brainstem stoke.
In conclusion, our results suggest that anodal tDCS to the ipsilesional hemisphere of poststroke patients effectively improves swallowing function when combined with intensive swallowing therapy. Further studies are needed to establish the optimal dose for stimulation, including the stimulating hemisphere, frequency, and parameters.
Footnotes
Acknowledgements
The authors thank Dr Tomohisa Ohno for help with the statistical analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
