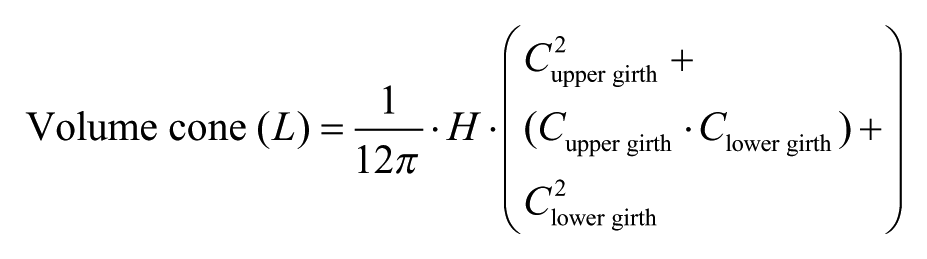Abstract
Background. While upper body training has been effective for improving aerobic fitness and muscle strength after spinal cord injury (SCI), activity-based therapies intended to activate the paralyzed extremities have been reported to promote neurological improvement. Objective. To compare the effectiveness of intensive whole-body exercise compared with upper body exercise for people with chronic SCI. Methods. A parallel-group randomized controlled trial was conducted. Participants with a range of SCI levels and severity were randomized to either full-body exercise (FBE) or upper body exercise (UBE) groups (3 sessions per week over 12 weeks). FBE participants underwent locomotor training, functional electrical stimulation-assisted leg cycling, and trunk and lower extremity exercises, while UBE participants undertook upper body strength and aerobic fitness training only. The primary outcome measure was the American Spinal Injury Association (ASIA) motor score for upper and lower extremities. Adverse events were systematically recorded. Results. A total of 116 participants were enrolled and included in the primary analysis. The adjusted mean between-group difference was −0.04 (95% CI −1.12 to 1.04) for upper extremity motor scores, and 0.90 (95% CI −0.48 to 2.27) for lower extremity motor scores. There were 15 serious adverse events in UBE and 16 in FBE, but only one of these was definitely related to the experimental intervention (bilateral femoral condyle and tibial plateau subchondral fractures). No significant between-group difference was found for adverse events, or functional or behavioral variables. Conclusions. Full-body training did not lead to improved ASIA motor scores compared with upper body training in people with chronic SCI.
Keywords
Introduction
The prediction of neurological recovery after spinal cord injury (SCI) has been based on physical examination of the acute patient using the International Standards for the Neurological Classification of Spinal Cord Injury (ISNCSCI). 1 The degree of spared volitional control is an important predictor of functional outcomes, such as walking ability. 2 Rehabilitation programs after SCI have traditionally focused on achieving optimal independence expected for a specific injury level, given the identified activity limitations and participation restrictions, and may rely on compensatory strategies. 3 While this approach may result in improvements in independent function, it does not promote recovery of motor control in the paralyzed limbs.
Loss of sensorimotor function after SCI induces musculoskeletal and metabolic adaptations which are associated with a higher risk of cardiovascular disease compared with the general population. 4 Regular physical activity can improve fitness and psychosocial well-being in the SCI population; however, a systematic review of combined muscle strength and aerobic training (upper body training) has reported that there is a limited number of low-quality studies with inconsistent findings. 5 Additionally, recent research provides new understanding of the benefits of exercise after SCI, including its positive impacts on the nervous system. 6
The concept of activity-based therapies arose from research in animals and humans showing that recovery of function could be enhanced through activity-dependent plasticity driven by neuromuscular activation below the level of injury. 7 These therapies include locomotor training and functional electrical stimulation (FES)–assisted exercise. While most research attention has focused on the benefits of these interventions for people with incomplete SCI,8-10 those with complete lesions may also benefit, since it is recognized that many are electrophysiologically incomplete (discomplete), that is, have subclinical evidence of descending cortical influence below the level of injury. 11 To date, interventions that exercised the paralyzed limbs have mostly been investigated as single entities using a range of research designs, and with various endpoints. The most recent systematic review of activity-based therapies for the lower limbs included a meta-analysis showing that, compared with no intervention or conventional physical interventions, activity-based therapy was not more effective for improving independence, lower limb mobility, or quality of life. 12 The effects of locomotor training in people with motor incomplete SCI (American Spinal Injury Association Impairment Scale—AIS C and D) on balance and ambulation have been investigated in a multisite observational study, 13 whereas a randomized controlled trial (RCT) investigated the effect on walking ability of treadmill versus overground walking training. 9 An RCT in people with motor incomplete SCI 10 and a nonrandomized, nonblinded study 14 have reported improvements in neurological recovery (ASIA motor score) and walking performance after combinations of activity-based interventions. There is a need for high-quality evidence to address this question.
The primary objective of this project was to investigate the effectiveness—on neurological recovery—of an intensive “whole-body” exercise program compared with an upper body only training program in people with complete and incomplete SCI. The activity-based program included locomotor training, FES-assisted leg cycling, and trunk and lower extremity exercises. In the case of complete SCI, locomotor training was used to explore the potential for stimulating neurological improvement, rather than as a means of attaining functional ambulation. Secondary objectives were to examine functional and psychological changes and quality of life.
Methods
Design
A multicenter, randomized controlled, assessor-blinded trial was undertaken in community-dwelling participants at 6 SCI units in Australia and New Zealand. The trial was registered with the Australian and New Zealand Clinical Trials Registry (ACTRN12610000498099) and ClinicalTrials.gov (NCT01236976). The trial was managed by a professional clinical trial management company (Neuroscience Trials Australia) and overseen by an independent Data Safety Monitoring Board. Ethical approval was obtained from the Human Research Ethics Committees at each site.
Participants
Participants were included if they were more than 18 years of age, had sustained a motor complete or incomplete traumatic SCI above the level of T12 at least 6 months prior to consent, and had medical clearance to participate. Participants were excluded if they had a pressure injury, history of long bone fracture, or were postmenopausal. Full inclusion and exclusion criteria are detailed in the published protocol. 15
Randomization
Participants were randomized to groups using a computer-generated randomization schedule, stratified by site and and injury status (AIS A/B or AIS C/D) using permuted blocks of random sizes. Randomization occurred after completion of baseline assessments and was controlled independently by Neuroscience Trials Australia. Assessors and statisticians performing the analyses were blinded to group allocation.
Usual Care
Participants had previously completed their primary rehabilitation but were required to cease any other therapeutic intervention (eg, physiotherapy, occupational therapy, FES, or complementary therapies) during the 12-week intervention period.
Full-Body Exercise Program
Participants in this group received a triad of interventions comprising locomotor training, FES-assisted cycling, and trunk and upper and lower extremity exercise. These interventions were provided at the participating spinal units 3 times per week over 12 weeks. Training in the delivery of the interventions was provided to all staff prior to trial commencement.
Locomotor training was deployed using a Therastride system (Innoventor Inc, St Louis, MO). The intervention was conducted in accordance with published protocols. 16 Training sessions initially comprised a series of 1- to 2-minute bouts of stepping training, interspersed with rests, for up to 30 minutes of stepping. As the participants improved their stepping ability, the duration of stepping intervals was increased to maintain progressive overload in longer intervals of up to 7 to 10 minutes and the amount of body weight support was gradually reduced as the participants improved their ability to bear weight on the lower limbs.
FES-assisted cycling was provided using a RT300 cycle (Restorative Therapies, Baltimore, MD) according to published protocols.15,17 Amplitude and pulse width were varied according to individual tolerance and capability. Participants exercised at the maximal power output possible, commencing with 10 minutes of training and gradually building up to an exercise duration maximum of 60 minutes. Warm up and cool down comprised 3 minutes of passive cycling.
Trunk and upper and lower limb exercises comprised assisted and/or resisted movements in different positions (eg, lying or weightbearing positions) aimed at facilitating and strengthening voluntary muscle activity and improving movement quality. Participants also undertook practice of functional tasks, involving moving the upper body over and outside the base of support, and exercises in motor imagery.
If participants missed any treatments during the intervention period, additional sessions were offered at another time during the week or during an optional additional week at the end of the intervention period.
Upper Body Exercise Program
Participants in this group received a circuit-based exercise program for the upper body, incorporating resistance and aerobic training (eg, chest press, boxing, resisted exercises, biceps/triceps curls, arm cranking). The 10-point Borg Rating of Perceived Exertion was used to describe aerobic exercise intensity, with participants expected to exercise between levels 4 and 8 on this scale. This program was provided 3 times weekly over 12 weeks and supervised by a therapist and/or clinical exercise instructor. Guidelines for the content, delivery and progression of exercises were outlined in a handbook to ensure standardization across sites.
Follow-up
At the end of the intervention period, all participants were free to continue with an exercise program if they wished. All therapy received by participants during the follow-up period was recorded in a patient diary.
Outcome Measures
All measurements were taken at baseline, 12 weeks (ie, at the end of the intervention period), and 24 weeks after randomization by therapists blinded to group allocation. Although the original protocol included a 12-month follow-up assessment, this assessment was removed half-way through the trial to reduce participant burden and encourage recruitment.
Primary Outcome
The primary outcome was neurological function at 12 weeks, measured by the ASIA motor score. 1
Secondary Outcomes
All secondary outcome measures are described in detail in the published protocol. 15 They included
ASIA motor score at 24 weeks and ASIA sensory scores at 12 and 24 weeks 1
Leg exercise capacity, a graded exercise test using FES cycling
Anthropometric measurements: Lower limb girths, waist circumference and skinfold thickness. Leg volumes were calculated by measuring girths at 7 levels along the leg and the height between these levels according to the protocol of Heesterbeek et al. 18 Using this method, the leg is divided into 6 imaginary cones (3 in the upper leg and 3 in the lower leg) and the volume calculated for each cone. The volume of each cone is calculated using the formula:
Spinal Cord Independence Measure (SCIM)
Measures of trunk function: Maximal balance range, and the Spinal Cord Injury–Falls Concern Scale (SCI-FCS).
Walking tests (for participants capable of walking): 6-Minute Walk Test (6MWT) and 10-Meter Walk Test (10MWT).
Spasticity over the previous week was measured via self-report using the Penn Spasm Frequency Score (PSFS). Any change in the dosage of antispasticity medication during the intervention period was also monitored.
Pain was measured using the SCI version of the Multidimensional Pain Inventory (SCI-MPI).
Psychological Measures included: Perceived Stress Scale, Hospital Anxiety and Depression Scale (HADS), Multidimensional Health Locus of Control, Moorong Self-Efficacy Scale and Rosenberg Self-Esteem Scale.
Quality of Life measures included the Assessment of Quality of Life–8 (AQoL-8), the Health Utilities Index Mark 3 (HUI3), and the WHO QoL-BREF.
Adverse Events
All adverse events (AEs) and serious adverse events (SAEs), whether related or unrelated to the interventions, were monitored and recorded. Clinical investigators or site coordinators used clinical judgement to determine severity, causality, and expectedness of all adverse events.
Data Analyses
Sample Size Calculation
We powered this study to detect a between-group minimum worthwhile treatment effect on the ASIA motor score at 12 weeks of 4.0 at the postintervention time-point (SD 1) based on available published data. 14 A sample size of 188 participants (94 per group) was estimated to provide 80% power to detect a significant intervention effect (2-sided, P = .05) using an analysis of covariance model that included baseline ASIA motor score as a covariate, a correlation between baseline and post-intervention ASIA motor score of at least 0.8, and an adjustment to allow for a dropout rate of 20%.
Data Integrity and Management
Data were stored electronically on a database with secured and restricted access and an audit trail in line with ICH GCP (International Conference on Harmonisation of technical requirements for registration of pharmaceuticals for human use Good Clinical Practice) guidelines.
Monitoring
The trial was overseen and monitored by a Program Manager and Data Safety Monitoring Board. The Program Manager ensured data quality and monitored compliance with the trial protocol during visits to each site. Interim safety analyses were planned to take place when 60 and 120 participants had completed their postintervention assessment.
Statistical Analysis
All endpoints and analyses were prospectively categorized as either primary or secondary. The primary and secondary endpoints analyses were conducted according to a predetermined analysis plan on an intention-to-treat basis and using the full dataset comprising all randomized participants. In addition, a per protocol analysis of the primary outcome for participants who adhered to all aspects of the protocol and received at least 80% of the training sessions was undertaken. Differences in both primary and secondary endpoints between the two arms of the study were tested independently at the .05 level of significance, using a linear regression model with robust sandwich estimator that also included the baseline value of the given outcome in question as a covariate. No formal adjustments were undertaken to constrain the overall type I error associated with the secondary analyses, as their purpose was to supplement evidence from the confirmatory primary analysis to help more fully characterize the treatment effect. Results from the secondary analyses were interpreted in this context.
The intention-to-treat strategy was based on an assumption that data were missing at random. The sensitivity of the results to plausible departures from the missing-at-random assumption as part of the intention-to-treat analysis were explored by using both a selection model (modeling of the missing data mechanism) and a pattern mixture model (modeling of the differences between missing and observed data). Assumptions about the missing data were expressed via a parameter that measures the degree of departure from the missing-at-random assumption. The results were graphed over a range of assumptions using the rctmiss suite of commands in Stata (version 13, StataCorp, College Station, TX).
Results
Recruitment
A total of 748 potential participants with SCI were screened for inclusion. Of these, 116 were eligible, agreed to participate and were subsequently randomized to the FBE or UBE groups. Figure 1 shows the reasons for exclusion and the flow of participants through the trial. Despite extensive screening through each spinal unit, recruitment took longer than expected and because of the limited funding period, the trial was terminated after randomization of 116 participants. This protocol change was made prior to data analysis.
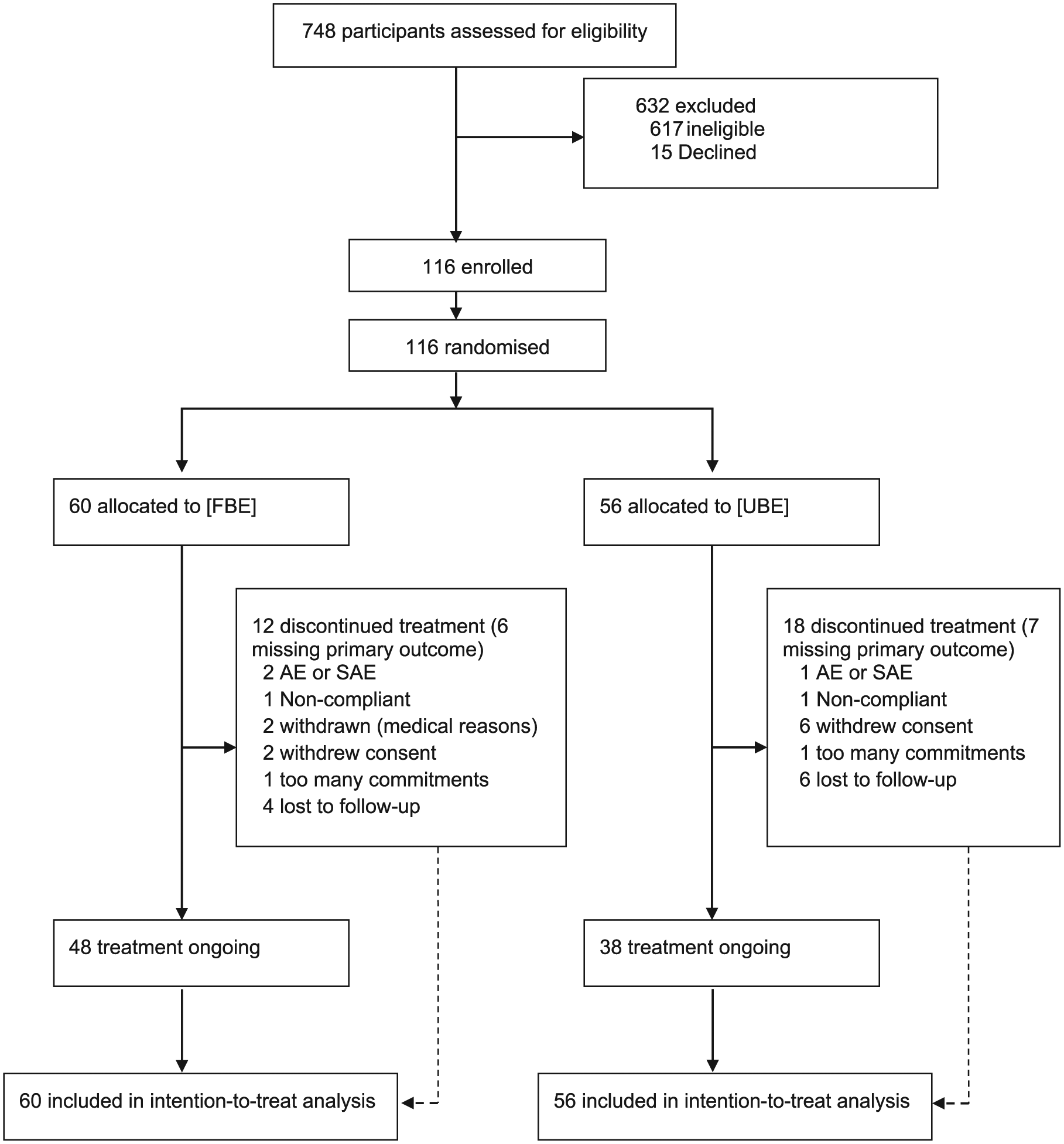
CONSORT (Consolidated Standards of Reporting Trials) flowchart for intention-to-treat analysis.
Participants
Table 1 presents demographic data and baseline characteristics for each group. The groups were similar with respect to age, time since injury, and AIS classification.
Demographics and Baseline Characteristics.
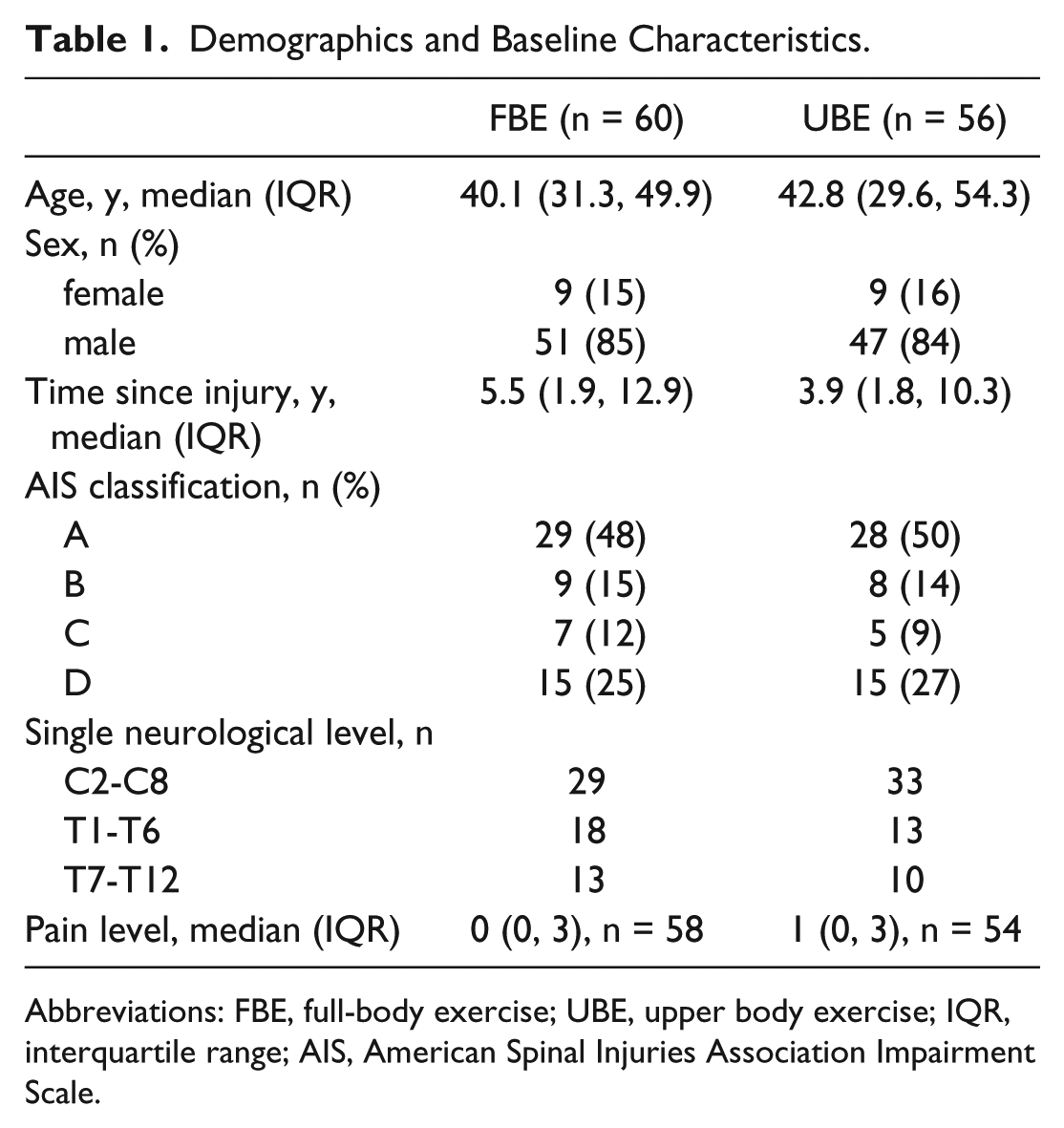
Abbreviations: FBE, full-body exercise; UBE, upper body exercise; IQR, interquartile range; AIS, American Spinal Injuries Association Impairment Scale.
Primary Outcome
No statistically significant between-group differences were found for the primary outcome measure (Table 2). The adjusted between-group difference for the upper extremity motor score (UEMS) was −0.04 (95%CI −1.12 to 1.04) and for the lower extremity motor score (LEMS) 0.90 (95% CI −0.48 to 2.27). The results of the per protocol analysis were similar (UEMS, difference = −0.002, 95% CI −1.11 to 1.1; LEMS difference = 0.81, 95% CI −0.64 to 2.26).
Summary of Treatment Effects.
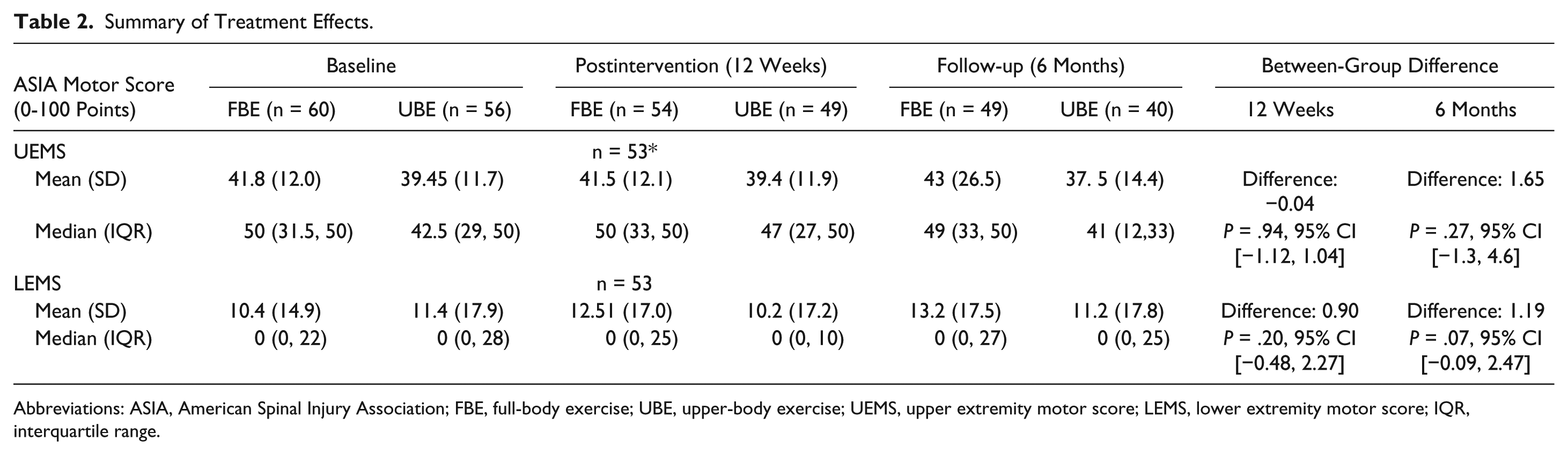
Abbreviations: ASIA, American Spinal Injury Association; FBE, full-body exercise; UBE, upper-body exercise; UEMS, upper extremity motor score; LEMS, lower extremity motor score; IQR, interquartile range.
Fifteen participants recorded a change in their AIS classification over the period of the trial (Table 3), though there appeared to be no consistent pattern within or between groups.
Changes in American Spinal Injuries Association Impairment Scale (AIS) Classification. a
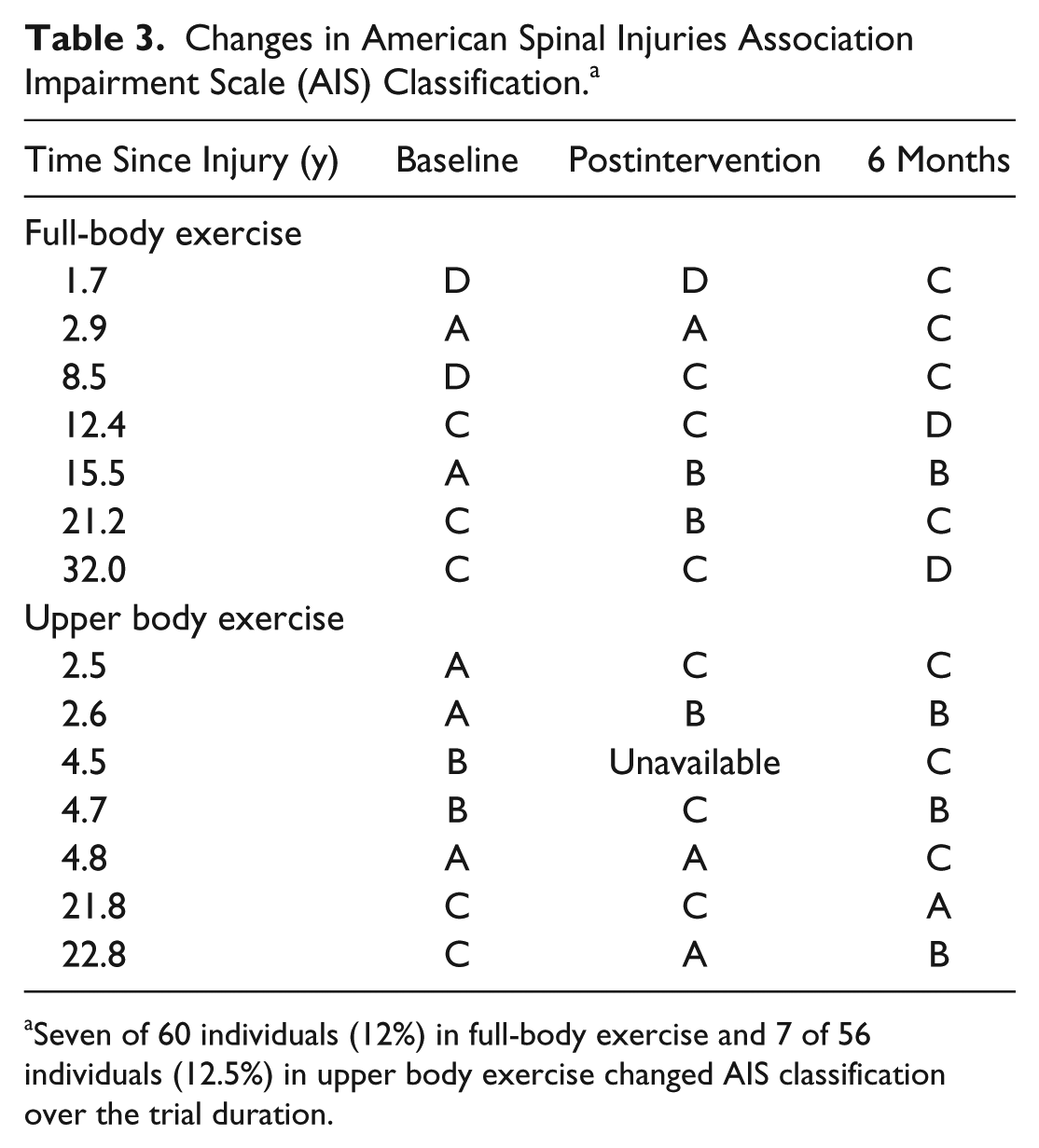
Seven of 60 individuals (12%) in full-body exercise and 7 of 56 individuals (12.5%) in upper body exercise changed AIS classification over the trial duration.
Secondary Outcomes
There were no statistically significant between-group differences for any of the secondary outcome measures (Table 2, Supplementary Tables 1 and 2). There was neither consistent trend toward improvement nor deterioration of walking performance on the 10-m walk test or the 6-minute walk test; the number of participants in each group who could walk (FBE, n = 14; UBE, n = 10) was low. Both groups showed a small reduction in waist circumference.
Adverse Events
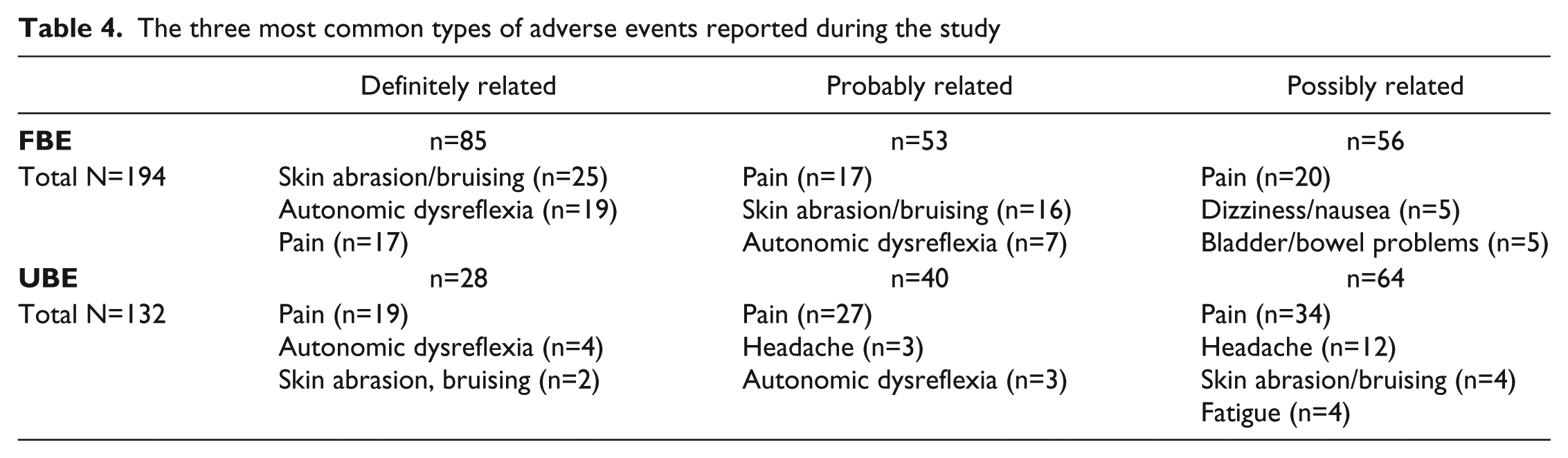
Thirty-one SAEs (16 FBE, 15 UBE) and 719 AEs (404 FBE, 309 UBE) were recorded over the 6-month trial period. One SAE in FBE (bilateral medial femoral condyle and tibial plateau subchondral insufficiency fractures) was considered to be definitely related to the intervention; this participant was withdrawn from the intervention and all sites were notified. The Therapeutic Goods Administration (TGA), Data Safety Monitoring Board and Human Research Ethics Committees from all SCIPA sites provided additional guidance for screening participants but permitted the study to continue. No other changes were made to the study as a result of this event. Another SAE in FBE (severe worsening back pain) was considered probably related to the intervention and the participant was withdrawn. Other SAEs were considered unrelated to the intervention and included severe autonomic dysreflexia (n = 3), chest infection (n = 3), and bladder/catheter-related problems (n = 3). In UBE, 1 SAE (feeling off balance, pain and loss of strength in upper limbs) was considered possibly related to the intervention. All other SAEs in UBE were unrelated to the intervention: 1 participant died of heart failure, but more than half the SAEs in this group were related to bladder problems (n = 6). Table 4 shows the most frequently recorded AEs considered definitely, probably, or possibly related to the intervention in both groups.
The three most common types of adverse events reported during the study
Retention and Adherence
All participants had preplanned 36 training sessions over a period of 12 weeks. Eight participants withdrew consent after randomization (6 UBE), 3 withdrew because of an SAE or AE (2 FBE), 2 in FBE were withdrawn from the study for medical reasons. Thirteen participants in UBE received fewer than 80% (28/36) of the required interventions. In FBE, not all components of the triad of interventions were completed at each session; 15 participants received fewer than 28 trunk exercise sessions, 18 received fewer than 28 FES-cycling bouts, and 21 participants received fewer than 28 treadmill training sessions. There were multiple reasons for this, including illness, study or work commitments and equipment failure.
Blinding of assessors was well maintained, with assessors being unblinded on only 8 occasions overall, mainly from participant disclosure. In these cases, a different assessor completed the assessments.
Discussion
The findings of our study do not support the hypothesis that an intensive FBE program might lead to neurological recovery, as assessed by the ASIA motor score. This was in contrast to the observations of Harness and colleagues, 14 which is the only other study that sought to directly compare an upper body exercise program with a multimodal program that involved the trunk and lower limbs, and which also included participants with motor complete and incomplete SCI. However, the study by Harness et al 14 had a number of design flaws, including nonrandomization, nonblinding of assessors, and disparity between groups in time postinjury. Similarly, we found no significant between group differences in ASIA sensory scores in our study.
Other studies involving people with motor incomplete injuries have reported changes in neurological function as a result of a similar intensive FBE program. Jones et al, 10 in a randomized controlled trial with wait-list control, showed significant improvements in the experimental group (total motor score and LEMS) compared with the control group. No significant difference in LEMS was observed in an RCT comparing locomotor training with overground walking training in people with incomplete SCI, although participants were recruited a mean of 4.5 weeks postinjury. 9 We did not require participants in our study to cease using prescribed oral baclofen for spasticity management. However, this agent, when used chronically, may lead to inhibition of neuromuscular activity. 19
The mechanisms underlying changes in ASIA motor score or function as a result of exercise have not yet been fully elucidated. These may include modulation of spinal neuronal excitability, 20 reorganization of spinal neuronal networks, 21 and sprouting of uninjured corticospinal axons to make contact with long propriospinal neurons which increased their terminal arborization onto motor neurons. 22 In the case of humans with neurophysiologically discomplete SCI, exercise training may strengthen the connectivity of extant descending pathways and lead to recovery of function. 23 Further electrophysiological studies are needed to understand these processes in humans with SCI.
The proportion of participants in our study with AIS C and D injuries was too low (30% of the sample) to discern any consistent difference in walking outcomes between groups. A systematic review 24 of studies of locomotor interventions (including body weight–supported treadmill training and robotic-assisted training) included 8 RCTs of variable quality examining a range of locomotor outcomes. Overall, there is limited effectiveness of locomotor interventions in people with incomplete SCI, although walking outcomes were superior in people with subacute injury. With 2 exceptions,9,25 the total sample sizes have been low. Although the originally calculated sample size in our study was not achieved, participant numbers were reasonable considering the relatively low incidence and prevalence of SCI in Australia and New Zealand. The length of our interventions (3 times per week for 12 weeks) was consistent with that in the majority of published trials. This dosage may be insufficient. In an observational study of locomotor training 13 participants received a median of 47 training sessions (range 20-251), with the number of intervention sessions determined by participant progress. However, the feasibility of regular attendance for intensive training needs to be examined in light of the barriers to exercise participation in people with SCI, especially socioeconomic factors. A significant proportion of participants in our study were unable or unwilling to attend all the required sessions because of work or study commitments, or ill health.
We observed changes in AIS classification in 14 participants (FBE n = 7; UBE n = 7; Table 3), with both improvements and deterioration. Although spontaneous AIS conversion has been reported within the first 12 months postinjury, it is noteworthy that several participants in both groups with AIS A classification converted to AIS B or C, which can be interpreted as neurological improvement, even though they were between 2.5 and 32 years postinjury. Of possible concern is that 2 participants with long-term (21.8 and 22.8 years) injuries in UBE converted from AIS C to A or B, indicating deterioration of function; however, conversely, 2 participants with long-term (21.2 and 32 years) injuries in FBE converted from AIS A to B or C.
To our knowledge, this is the first study of exercise interventions after spinal cord injury that has fully reported on adverse events whether related to the trial interventions or not. There was only 1 SAE that was definitely related to the study intervention (bilateral subchondral tibial plateau and medial femoral condyle fractures) in a participant classified as AIS A. A proximal tibial fracture in a participant classified as AIS C was reported following robotic treadmill training. 26 Clearly, it is important to be vigilant about the possibility of fracture given the higher risk in people with SCI. Dual energy X-ray absorptiometry (DEXA) is the most widely used method of assessing bone mineral content and bone mineral density, but has limited utility for the prediction of fracture threshold. Moreover, there are no bone mineral density reference values for the sites at which insufficiency fractures typically occur in SCI. Peripheral quantitative computed tomography (pQCT) is more precise in people with SCI 27 and can provide separate values for cortical and trabecular compartments; this is important because bone mass is lost through reduction of bone mineral density in the long bone metaphyses, whereas in the shaft, bone mass is lost through a reduction in thickness and an increase in the porosity of the cortical wall. 27 Guidelines for screening for fracture risk prior to a person with motor complete SCI undertaking load-bearing exercise such as locomotor training would be useful. However, since there was only one instance of fractures related to the intervention in our study, on balance the intervention can be considered safe under clinical trial conditions.
As shown in Table 4 among the most common AEs considered to be definitely related to the interventions were skin abrasion/bruising, autonomic dysreflexia, and pain, well-recognized risks for exercise interventions. Skin abrasion or bruising was mostly caused by rubbing of straps or knocking of limbs against apparatus. The incidence of this and autonomic dysreflexia was more common in FBE. Five instances of dizziness/nausea were reported as possibly related to the experimental intervention; this may be related to postural hypotension. Fifteen instances of headache in UBE participants were considered possibly or probably related to the intervention; these may be symptoms of autonomic dysfunction. Pain was commonly reported in both groups, and although the site was frequently not recorded, there were instances of upper limb pain resulting from the use of gym apparatus. Continual vigilance by treatment staff is therefore required to minimize injury and compromise of health for participants in intensive exercise programs.
Since intensive exercise programs can feasibly only be provided for a limited period, mechanisms to facilitate ongoing participation in physical activity need to be considered. There is increasing evidence that participation in exercise and sport promotes physical and psychological well-being in people with spinal cord injury. 28 Among the benefits of such exercise is a reduction in cardiovascular risk factors. In spinal cord–injured people who are sedentary, the levels of visceral fat are in the “at-risk” category. 29 Obesity significantly increases the compressive forces on the ischial tuberosities in people with SCI, thus increasing the likelihood of deep tissue injury. 30 Therefore, a reduction in waist circumference in participants in both exercise groups suggests a beneficial reduction in comorbidity.
We did not observe changes in quality of life, or on psychological measures. However, a small qualitative study conducted at one of the trial sites in parallel with this study highlighted the benefits associated with study participation, which increased motivation and interest in continuation of physical activity after the trial; these included access to an appropriately equipped gym in a supportive environment and being able to safely challenge one’s physical and psychological boundaries. 31 Many barriers exist to participation in leisure time physical activity by people with SCI, including motivation, finances, and accessibility to specialized programs or equipment. 32 Programs to overcome some of these barriers are now being developed 28 and are similar to the exercise program delivered to UBE in this study.
Ultimately however, while exercising the paralyzed limbs is a necessary condition for neural activation, exercise on its own may be insufficient to effect neurological change, and the addition of neuromodulation may be needed to do so. Recent studies of spinal cord neuromodulation using epidural stimulation in people with motor complete injury 33 have demonstrated latent voluntary control below the level of injury; these individuals had received 80 locomotor training sessions prior to electrode implantation. Repetitive training such as locomotor training engages central pattern generators which are sensitive to tactile and proprioceptive input from the lower limbs and serve to control stepping. 34 Raising the level of excitability of the spinal cord circuitry using epidural stimulation facilitates activity-dependent reorganization of the spinal neuronal network via the propriospinal network. The advent of transcutaneous spinal cord stimulators 35 brings this technology closer to wider clinical translation and underlies the importance of further investigation of the delivery and dosage of exercise and neural stimulation programs.
Conclusion
Our study is the first RCT comparing whole-body exercise with an upper body exercise program in people with spinal cord injury. We did not demonstrate greater neurological improvements as assessed by ASIA motor scores after a whole-body exercise program compared with an upper body exercise program of 12 weeks duration. The heterogeneity of our sample, which included participants with a range of injury levels and severity, may have contributed to this result. On the whole, both experimental and control interventions were safe. Exercise is important for physical conditioning and well-being after spinal cord injury and has positive effects on nervous system function. However, exercise may need to be combined with neuromodulation to achieve neurological recovery.
Supplementary Material
Supplementary Material, Supplementary_Table_1_rev – SCIPA Full-On: A Randomized Controlled Trial Comparing Intensive Whole-Body Exercise and Upper Body Exercise After Spinal Cord Injury
Supplementary Material, Supplementary_Table_1_rev for SCIPA Full-On: A Randomized Controlled Trial Comparing Intensive Whole-Body Exercise and Upper Body Exercise After Spinal Cord Injury by Mary P. Galea, Sarah A. Dunlop, Timothy Geraghty, Glen M. Davis, Andrew Nunn, Liudmyla Olenko, and SCIPA Switch-On Trial Collaborators in Neurorehabilitation and Neural Repair
Supplementary Material
Supplementary Material, Supplementary_Table_2_rev – SCIPA Full-On: A Randomized Controlled Trial Comparing Intensive Whole-Body Exercise and Upper Body Exercise After Spinal Cord Injury
Supplementary Material, Supplementary_Table_2_rev for SCIPA Full-On: A Randomized Controlled Trial Comparing Intensive Whole-Body Exercise and Upper Body Exercise After Spinal Cord Injury by Mary P. Galea, Sarah A. Dunlop, Timothy Geraghty, Glen M. Davis, Andrew Nunn, Liudmyla Olenko, and SCIPA Switch-On Trial Collaborators in Neurorehabilitation and Neural Repair
Footnotes
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Collaborators
Melanie Hurley (The University of Melbourne, Parkville, Victoria, 3010, Australia) - Trial coordinator Janette Alexander (Austin Health, Heidelberg, Victoria, Australia) - Site coordinator Sarah Fereday (Royal Rehab, Ryde, NSW) - Principal investigator Clare Goodman (Royal Rehab, Ryde, NSW) - Site coordinator Julia Batty (Prince of Wales Hospital, Randwick,NSW) - Principal investigator Trent Li (Prince of Wales Hospital, Randwick, NSW) - Site coordinatorProf John Buchanan (Royal Perth Hospital, Perth, Western Australia) - Principal investigator Julie Bullick (Royal Perth Hospital, Perth, Western Australia) - Site coordinator A/Prof Ruth Marshall (Hampstead Rehabilitation Centre, Lightsview, South Australia) - Principal investigator A/Prof Jillian Clark (Hampstead Rehabilitation Centre, Lightsview, South Australia) - Site coordinator Dr Rick Acland (Burwood Spinal Unit, Christchurch, New Zealand) - Principal investigator Dr Jo Nunnerley (Burwood Academy of Independent Living, Christchurch, New Zealand) - Site coordinator
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Transport Accident Commission (Victorian Neurotrauma Initiative), the Lifetime Care and Support Authority NSW, the University of Melbourne and The University of Western Australia.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.