Abstract
Introduction
Spinal cord injury (SCI) generally leads to a partial or complete loss of motor and sensory functions below injury level. Various types of locomotor training enhanced by pharmacological aids (eg, clonidine, cyproheptadine, and quipazine), body weight assistance, tail pinching, and other sensory stimuli have been shown to induce hindlimb stepping induction in spinal cord–transected (Tx) animal models.1-3 Some of these findings led to innovative rehabilitation strategies (eg, body weight–supported treadmill training or BWSTT) for the partial recovery of voluntary ambulation, mainly in incomplete SCI patients.4-6 Recently, novel strategies for locomotor training using electrical, engineering, and pharmacological approaches have also been explored as promising therapeutic avenues specifically for individuals with complete SCI.7-9
We identified a combined pharmacological therapy that activates the central pattern generator (CPG), eliciting powerful episodes of locomotor movements in nonassisted Tx mice.10,11 Buspirone, a 5-HT1A receptor agonist; carbidopa, a decarboxylase inhibitor; and L-DOPA, a noradrenergic/dopaminergic precursor when administered together (BCD) induce, within 15 minutes, robust episodes of bilaterally alternating hindlimb movements (locomotor-like movements [LMs]) accompanied by weight bearing and plantar foot placement capabilities in untrained and nonassisted Tx mice. 10 Here, we test the extent to which regular training and treatment with CPG-activating drugs will further improve locomotor function and diminish complications commonly found in chronic motor-complete SCI patients. We also tested whether clenbuterol, a β2-adrenergic agonist with effects similar to anabolic steroids, in combination with BCD treatment and training will improve function still further.
Body weight, skeletal muscle, bone, and adipose tissue losses occur within days to a few weeks following transection in both SCI patients and animal models. 12 A relation between levels of physical activity, muscle adipose tissue, and bone metabolism is well established. 13 Anabolic substances, such as testosterone, estrogen, and other steroids, and growth factors, including insulin-like growth factor (IGF-1), have protective properties on the motor system. We have shown promising results with BCD-induced training without anabolic aids, although only partial effects on muscle tissue loss were found in Tx mice. 11 Consequently, we chose to characterize and determine the potential benefits of using pharmacologically induced muscular activity and regular training on muscle, bone, and adipose tissue preservation in Tx mice. Clenbuterol was chosen for this study based on promising muscle-building effects already shown in models of muscle wasting as well as in paraplegic mice.14-18
Materials and Methods
Animal Model
All experimental procedures were conducted in accordance with the Canadian Council for Animal Care guidelines and were accepted by the Laval University Animal Care and Use Committee. Male CD1 mice (Charles River Canada, St-Constant, Québec, Canada) initially weighing 30 to 35 g were used for this study. All surgical procedures were performed under aseptic conditions in anesthetized mice (2.5% isoflurane). The spinal cord was completely Tx intervertebrally between the 9th and 10th thoracic vertebrae (Th9/10), as described in previous studies.19-21 Complete low-thoracic Tx was confirmed by (1) initial full paralysis of the hindlimbs, (2) postmortem visual and microscopic examination of the spinal cord lesion, and (3) histological assessment of coronal or midsagittal sections stained with luxol fast blue/cresyl violet.
Drug Treatment and Experimental Design
Animals were randomly divided into groups: control groups included (1) unoperated control mice (n = 11) and (2) operated control Tx mice (n = 11); experimental treatment groups included (1) BCD-treated Tx mice (n = 11) and (2) BCD + clenbuterol–treated Tx mice (n = 10). The unoperated and operated control mice received saline injections before testing but did not receive training. Both BCD and BCD + clenbuterol groups received treadmill training as well as drug combinations. The triple drug combination included buspirone hydrochloride (3 mg/kg, intraperitoneally [i.p.]), L-DOPA (50 mg/kg, i.p.), and carbidopa (12.5 mg/kg, i.p., coadministered with L-DOPA to increase bioavailability of L-DOPA centrally 22 ; Sigma, St Louis, Missouri). BCD was administered 3/wk at the time of locomotor testing. Clenbuterol (1 mg/kg) was administered subcutaneously daily. All drugs were dissolved in sterile saline.
Drug treatments and training sessions began 1 week post-Tx to allow recovery from surgery. BCD and BCD + clenbuterol mice were trained 3 times/wk for 8 weeks on a motor-driven treadmill at 8 to 10 cm/s. Training sessions lasted 15 minutes, after which mice were returned to their cage. No sensory stimulation or body weight support assistance was provided during training. However, a harness placed on the torso was needed to maintain the animals perpendicular to the camera used for recording movements.
Body Weight, Muscle Mass, and Adipose Tissue Mass
Body weight was monitored daily during the first week and once a week subsequently. After being killed humanely, forelimb (biceps brachii and triceps brachii) and hindlimb (extensor digitorum longus [EDL], soleus, biceps femoris, and quadriceps) muscles were dissected and weighed. Adipose tissues from the abdominal subcutaneous, inguinal, visceral, retroperitoneal, and interscapular areas were collected and weighed after 8 weeks.
Bone Densitometry
At sacrifice, femoral bones were dissected and cleaned of soft tissue. Bones were wrapped in saline-soaked gauze and frozen at −20°C in sealed vials until analyses. Dual-energy X-ray absortiometry measurements (PIXImus 2, Lunar Corp, Madison, Wisconsin) were used to determine bone mineral density (BMD) and bone mineral content (BMC) within a predetermined region of interest near the metaphyseal area. For additional details, see references Guertin et al 11 and Picard et al. 23
Assessment of Locomotor Movement
Once a week, mice were placed on a motor-driven treadmill for 2 minutes to assess spontaneously occurring locomotor movements in unoperated and operated controls and movements induced by drugs in the treatment groups. Each session of locomotor movement assessment lasted 2 minutes. One 2-minute session was conducted immediately prior to drug or saline administration, and another 2-minute session was conducted 15 minutes after drug or saline injection.
Two tests, the Average Combined Score (ACOS)24-26 and the Antri, Orsal, and Barthe (AOB) scale, 27 assessed locomotor movements. Kinematic analyses assessed step length, height, and angular excursion of hindlimb joints. The ACOS scoring method uses an arithmetic formula to characterize and quantify hindlimb movement frequency (movements per minute) for both LMs and non–locomotor-like movements (NLMs). A NLM was defined as a nonbilaterally alternating movement, including jerks, fast paw shaking, and twitches. A LM was defined as an alternating flexion–extension movement occurring bilaterally. For the AOB method, hindlimb locomotor movement recovery was evaluated using a 22-level scoring scale. Scores above the 10th level include weight-bearing stepping. For kinematic analyses, movements were filmed using a digital video camera (JVC GZ-MG330; shutter speed, 1/4000; and acquisition, 60 frames/s) placed perpendicularly to the animal. MaxTRAQ and MaxMATE softwares (Innovision System, Columbiaville, Michigan) were used for these analyses. Additional details regarding ACOS, AOB, or kinematic analysis are given in other references.24-27,11
Muscle Fiber Characterization by Immunofluorescence
After death, the left soleus and EDL muscles were dissected, frozen in melting isopentane, and stored at −80°C until further use. Serial cross sections (12-µm thick) prepared from the muscle midportion were cut with a cryostat maintained at −20°C (2800E Frigocut, Leica Instruments, Germany) and mounted on SuperFrost Plus glass slides (VWR Canlab, Mississauga, ON, Canada). In brief, for the labeling of individual fibers, myosin heavy chain primary antibodies were used for “slow” or “fast” fiber types (MHCs or MHCf, specific for MHC isoform type I and type II, respectively; Vector Laboratories, Burlingame, California). Control sections were processed without primary antibodies. Immunofluorescence was visualized with a 10× water-immersion objective placed on an Olympus BX61WI confocal microscope. Images were captured using FluoView 300 (Olympus Canada Inc, Markham, ON, Canada) and analyzed with ImageJ (ImageJ 1.40, Research Services Branch, NIH, Bethesda, Maryland). Cross-sectional area (CSA) values were measured for each fiber type (50 fibers per type/muscle were averaged).11,18
Data Analyses
Locomotor movement (ACOS and AOB scores) and body weight were analyzed using a 2-way analysis of variance (ANOVA) followed by least significant difference (LSD) post hoc tests. Differences between groups for muscle mass, soleus and EDL muscle CSA, fiber-type distribution, single fiber-type CSA, adipose tissue, and femoral BMD and BMC values were analyzed using a 1-way ANOVA followed by LSD post hoc tests. Statistical significance was set at
Results
Induced Locomotor Movements
Prior to injection of BCD or saline, relatively low and generally nonsignificant levels (eg, AOB scores ≤ 3) of LM were found in all groups of Tx mice. These levels were found not to be significantly different between untreated (saline), BCD-treated, and BCD + clenbuterol–treated Tx animals (data not shown). Motor scores remained low in untreated Tx mice, although a modest recovery was seen over time (
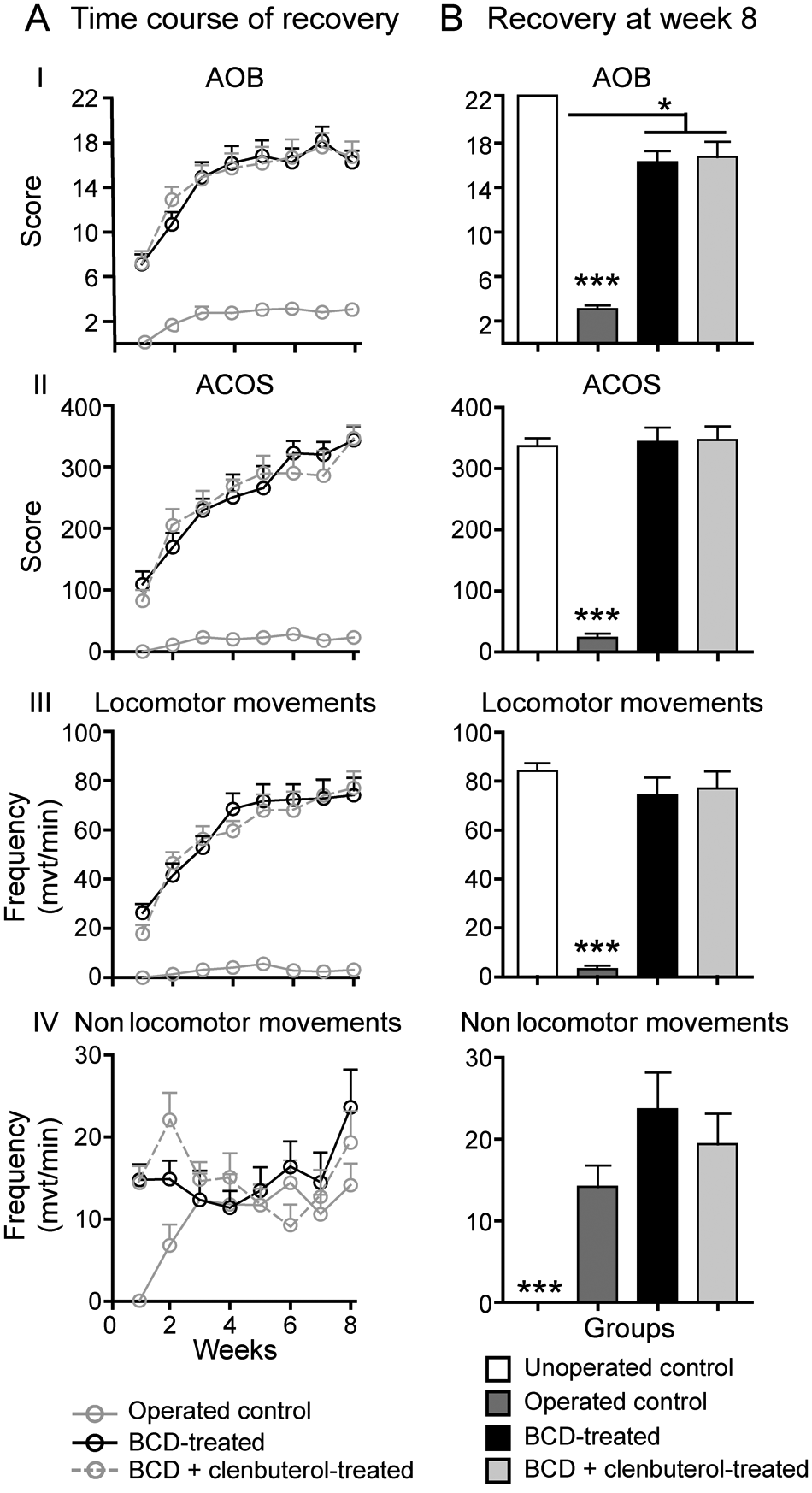
Locomotor recovery assessed using 2 locomotor rating scales: Antri, Osral, and Barthe (AOB) and Average Combined Score (ACOS) locomotor scales. The ACOS is a combination of nonlocomotor movements (NLMs), locomotor movements (LMs), and amplitude. The AOB is a qualitative locomotor rating scale developed by Antri, Orsal, and Barthe. 27 In postdrugs results, a 2-way ANOVA revealed significant differences between both tritherapy-treated groups compared with untreated Tx mice. At the 8th week, comparison between unoperated control mice and both BCD-treated mice showed no difference in ACOS score and LM frequency.
Differences between groups were examined at 8 weeks post-Tx (Figure 1B). At this time, locomotor movements were comparable between unoperated control mice and Tx mice treated with drugs (Figure 1B, II-III), with ACOS scores of 336.9 ± 11.5 for unoperated controls, and LM averaging between 74.1 ± 7.2, 77.0 ± 6.8, and 84.2 ± 11.1 per minute (BCD, BCD + clenbuterol treated, and unoperated controls, respectively). Whereas AOB scores were comparable at 8 weeks, but slightly lower (
To further characterize drug-induced movements, we used kinematics to measure angular excursion at the hip, knee, and ankle and movement amplitude. Typical examples of hindlimb movements are shown in Figure 2. Hip, knee, and ankle angular displacement values showed similar patterns in unoperated controls and both drug-treated groups (Figures 2A, 2C, and 2D, top panels; hip, 29°-117°; knee, 47°-123°; ankle, 41°-132°). Operated control Tx animals displayed occasional small-amplitude movements involving mainly rotations at the knee and ankle joints (knee, 30°-47°; ankle, 28°-125°). Stepping amplitude measured by toe displacement values in X and Y directions (step “length” and “height”; Figures 2A-2D, bottom panels, and Table 1) revealed that unoperated control mice displayed greater (
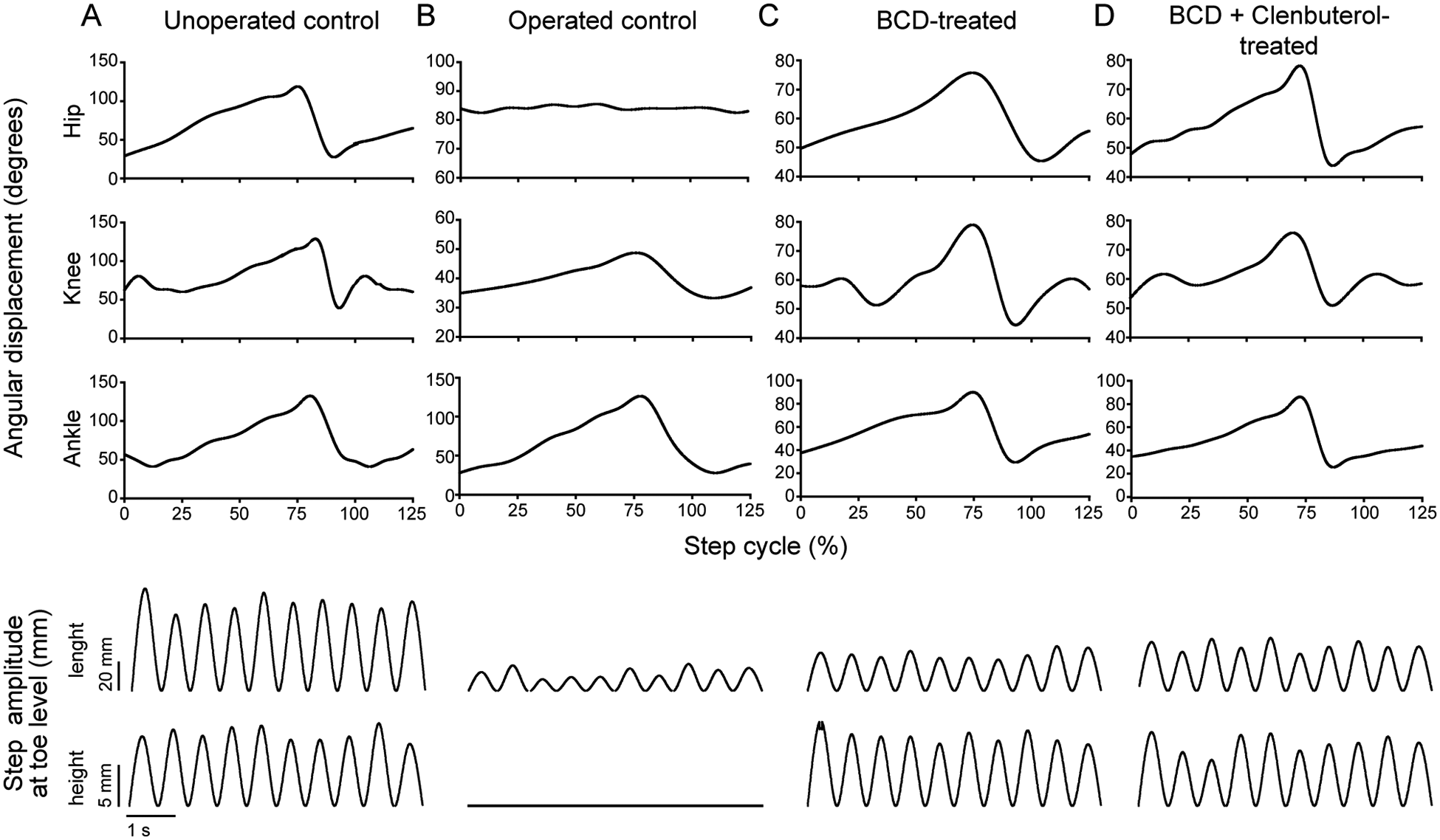
Representative hindlimb kinematics: For unoperated controls (A), operated controls (B), BCD-treated mice (C), and BCD + clenbuterol–treated mice (D), joint angle displacements were averaged in 10 consecutive steps. For operated controls, extension–flexion was considered as a step, without toe lift. Hip, knee, and ankle joint angular displacement are shown in the top 3 panels. Step amplitude (length and height at toe level) is shown in the bottom 2 panels.
Step Amplitude a
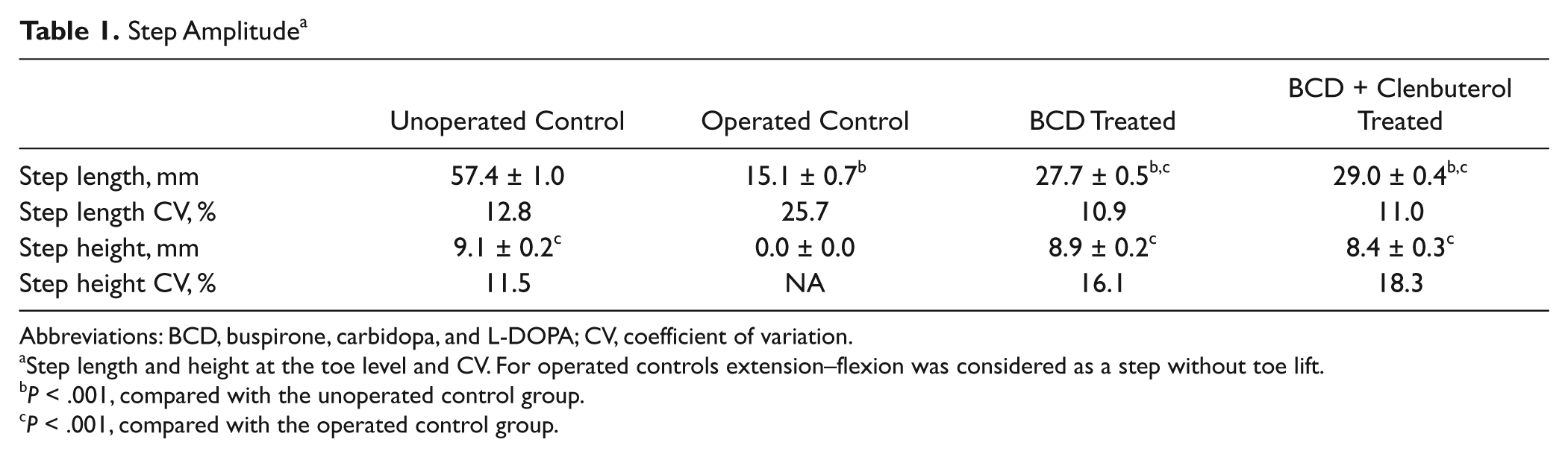
Abbreviations: BCD, buspirone, carbidopa, and L-DOPA; CV, coefficient of variation.
Step length and height at the toe level and CV. For operated controls extension–flexion was considered as a step without toe lift.
Body Composition
Body weight was monitored on a weekly basis (Figure 3A). All groups of Tx mice lost on average 15.8% ± 0.8% of body weight within the first week post-Tx. Body weight increased subsequently, although initial weight values were not reached after 8 weeks in any of the Tx groups. By weeks 4 to 5, both groups of drug-treated Tx mice had significantly higher weight gains than operated control mice.
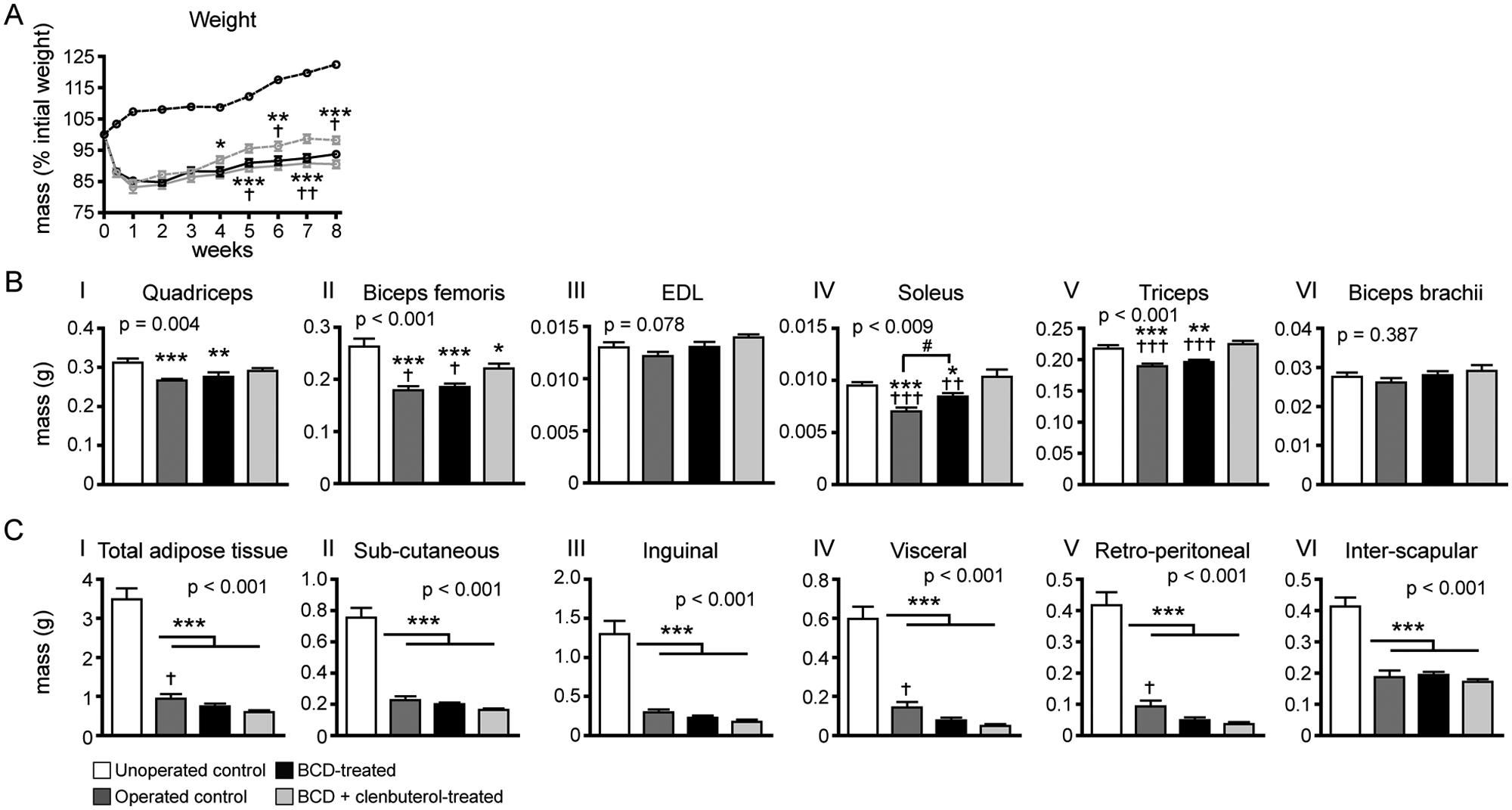
Body weight, muscle mass, adipose tissue mass: (A) average body weight increased over 8 weeks in all groups, but weights of operated groups were less than that of unoperated controls and did not reach preoperative weights. Both muscle atrophy and loss of adipose tissue contributed to decreased body weight. (B) At 8 weeks, muscle mass was similar for the quadriceps, soleus, and triceps in unoperated controls and in the BCD + clenbuterol group but decreased in the BCD groups. Biceps femoris mass was also decreased in BCD groups and to a lesser extent in BCD + clenbuterol groups. No difference among groups was found in the EDL and biceps brachii. (C) At 8 weeks, adipose weights were diminished in all operated animals compared with unoperated controls.
The body weight loss found in Tx mice could be partially attributed to fat tissue loss. Indeed, all groups of Tx mice showed at least a 3.5-fold decrease in total adipose tissue amount compared with unoperated control animals (Figure 3B). Body weight loss could also be attributed partially to bone tissue loss because all groups of Tx mice displayed comparable losses in relation to non-Tx animals (Figure 4). Thus, neither adipose tissue nor bone loss is prevented by drug therapy or training. Body weight loss can also be partially attributed to muscular atrophy found in operated control mice (Figures 3B, I-VI). It is important to note that muscular atrophy was completely prevented in all muscles examined in the BCD + clenbuterol group, which did not differ from what was observed in unoperated control mice.
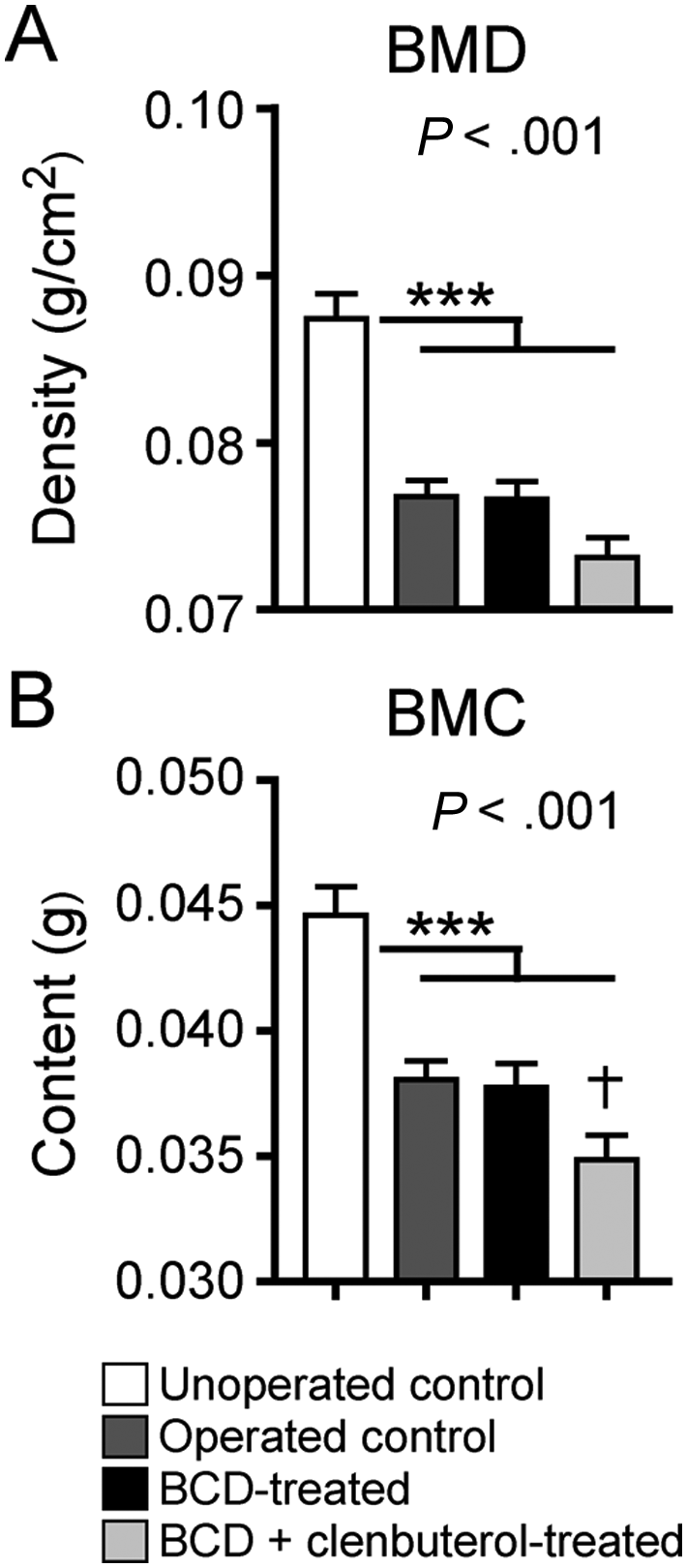
Bone mineral density (BMD) and bone mineral content (BMC) quantification: measurements were performed by dual X-ray absorptiometry (PIXImus). All Tx mice showed lower BMD and BMC values than non-Tx mice
Muscle and Fiber-Type Morphometry
Morphometric analyses of the soleus and EDL were performed to further characterize specific changes in muscle properties. Muscle CSA, fiber type–specific CSA, and relative distribution values were analyzed. Operated control and BCD-treated animals had significantly lower muscle CSA values for the soleus compared with unoperated control and BCD + clenbuterol–treated Tx groups. For the EDL, CSA values were greater (
Soleus and EDL Muscle Cross-Sectional Area a
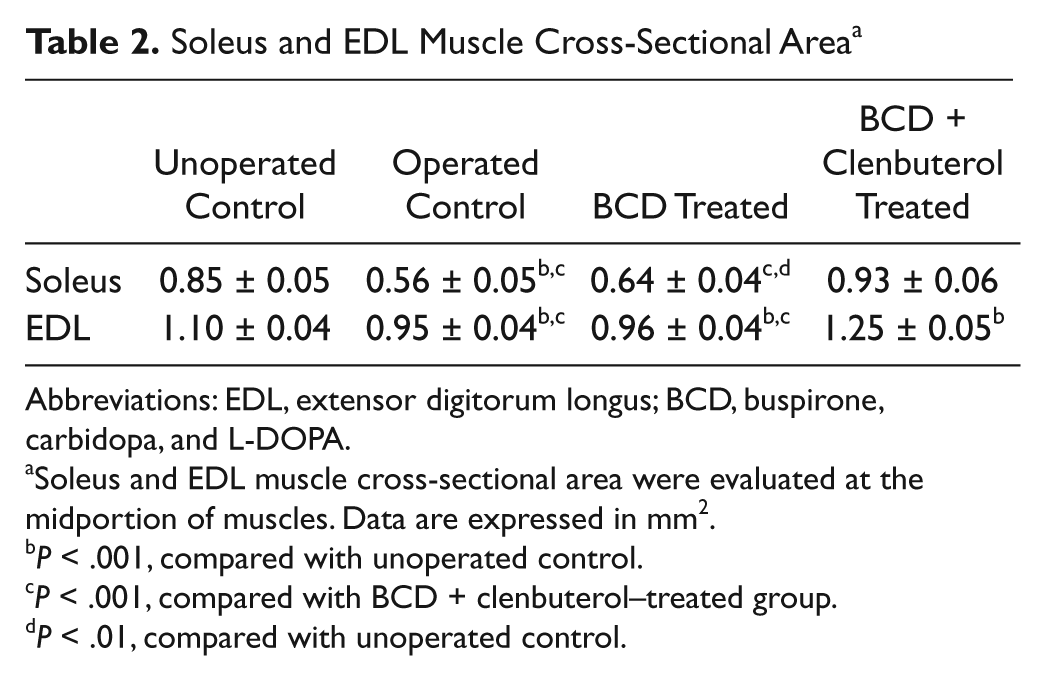
Abbreviations: EDL, extensor digitorum longus; BCD, buspirone, carbidopa, and L-DOPA.
Soleus and EDL muscle cross-sectional area were evaluated at the midportion of muscles. Data are expressed in mm2.
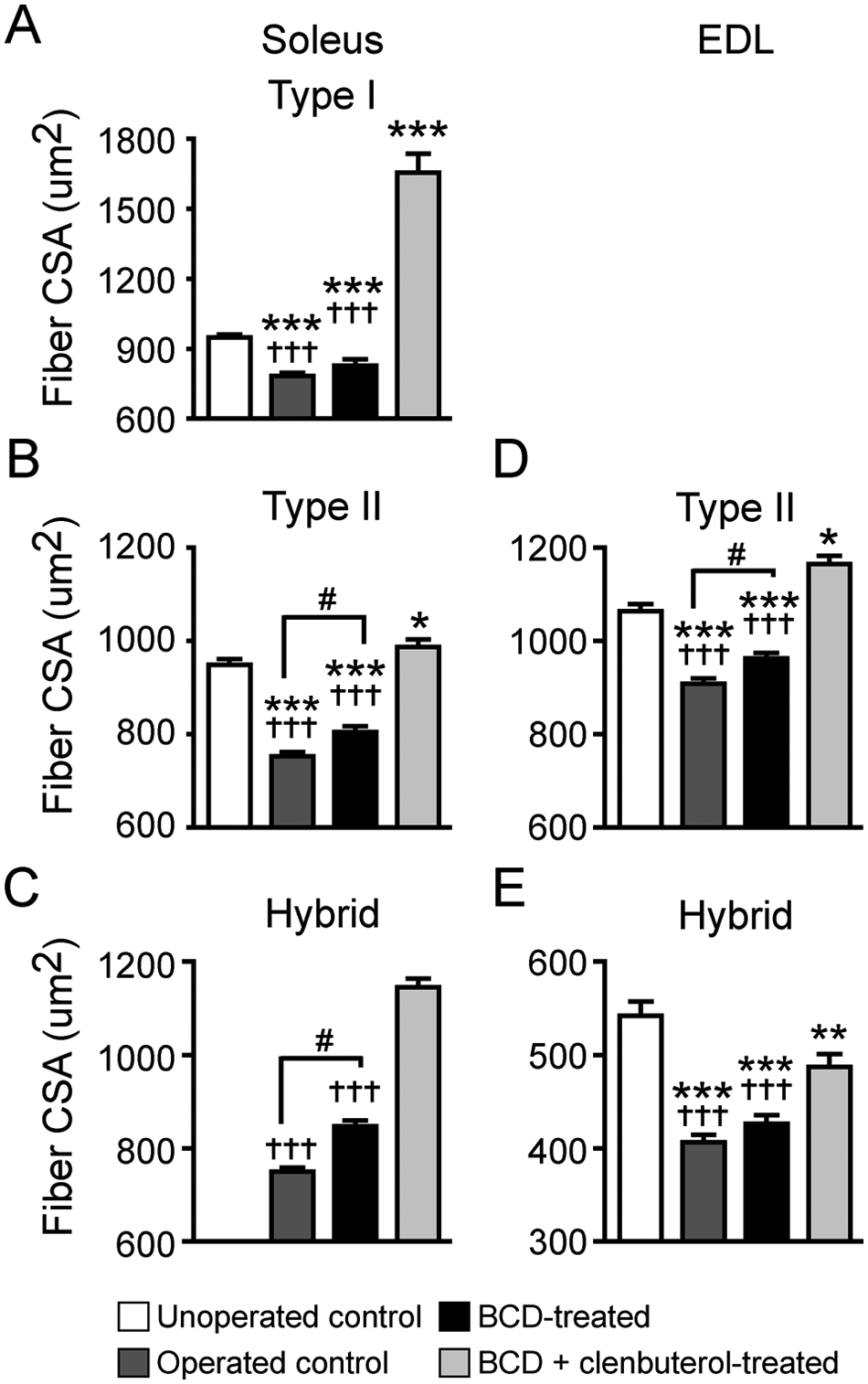
Muscle fiber type cross-sectional area (CSA): comparisons between groups for soleus (A, B, C) and EDL (D, E) type I, II, and hybrid fiber CSAs. No EDL type I fibers were found
Generally, after SCI, slow fibers tend to switch to a faster phenotype.28,29 In this study, after Tx, 50% to 55% of the slow types showed such fiber type conversion, shifting to a hybrid isoform. None of the unoperated mice expressed hybrid fibers. Fiber type conversion was not observed in the EDL muscle (Table 3).
Fiber Type Relative Distribution a
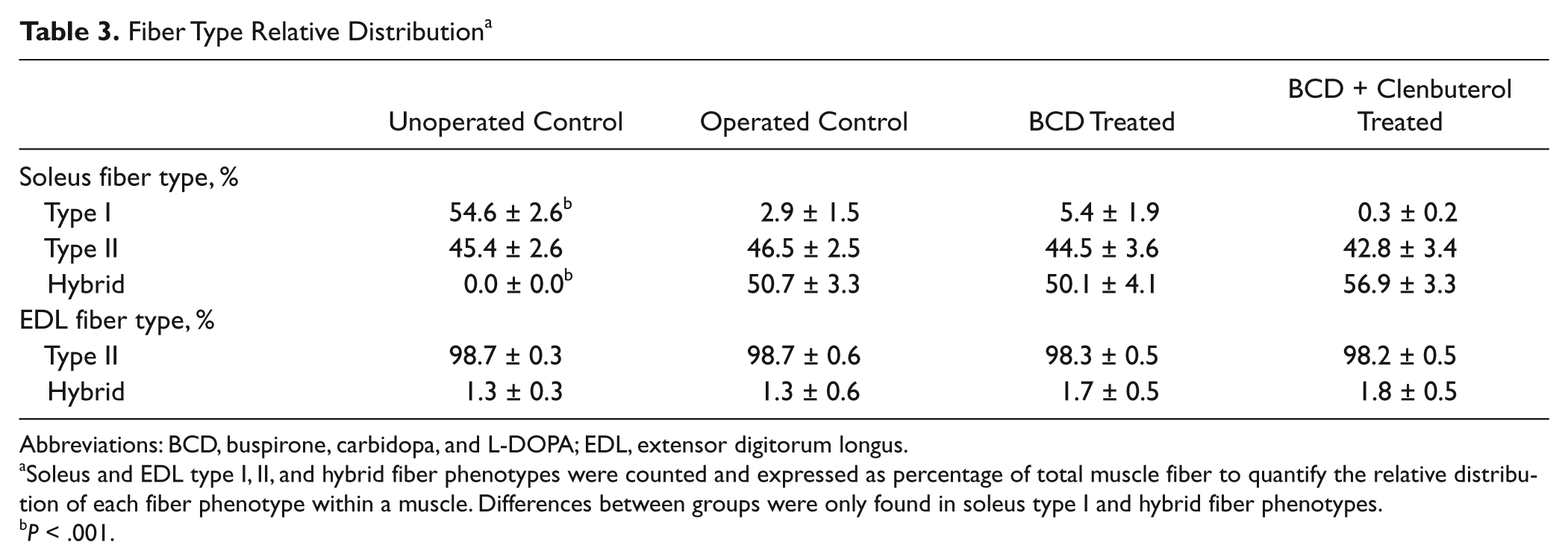
Abbreviations: BCD, buspirone, carbidopa, and L-DOPA; EDL, extensor digitorum longus.
Soleus and EDL type I, II, and hybrid fiber phenotypes were counted and expressed as percentage of total muscle fiber to quantify the relative distribution of each fiber phenotype within a muscle. Differences between groups were only found in soleus type I and hybrid fiber phenotypes.
Discussion
This study demonstrates that spinalized mice receiving clenbuterol daily and a CPG-activating BCD therapy and treadmill training 3 times/wk can recover near-normal induced-locomotor movements, muscle mass, and muscle fiber size (CSA) by 8 weeks. Thus, this combination therapy can entirely prevent the substantial muscular atrophy usually found within a few days to a few weeks after transection in adult mice. Spinalized mice that received the BCD without clenbuterol also achieved comparably high levels of induced-locomotor movement improvements over time in weight-bearing stepping, bilateral alternation (LM), and plantar foot placement. Body weight, bone, and adipose tissue losses, however, were not prevented by training plus pharmacotherapy.
Pharmacological Activation of the CPG in Nonassisted Paraplegic Mice
In adult rodents, low levels of spontaneously reoccurring hindlimb movements have been reported within a few weeks post-Tx. These movements are largely nonlocomotor movements without weight-bearing stepping or plantar foot placement.24-26 The extent of locomotor movements that is restorable in Tx mice can improve with a variety of pharmacological aids. We have shown that administration of combinations of drugs injected by several routes (i.p., subcutaneously, and by oral gavage) will activate the CPG.11,19-21,30,31 In the present study, we specifically used a pharmacology comprising buspirone, L-DOPA, and carbidopa (BCD). 10 We have also shown that BCD + training increased soleus muscle mass, soleus fiber type II and hybrid fiber CSA values, and EDL type II CSA values compared with operated, untreated Tx animals. This complements previous findings showing that training itself can increase, to some extent, soleus muscle mass and fiber CSA.32,33 However, the proportion of muscle increase that was attributable to treadmill training or BCD treatment could not be determined. But an interaction between both is more likely to explain the results in this study. Indeed, treadmill training alone was not beneficial for muscle atrophy. 34 Furthermore, although BCD treatment alone can induce some weight-bearing steps (unpublished data), sensory cues provided by the moving treadmill are essential to improve locomotor recovery in BCD-treated animals, thus increasing the number of weight-bearing steps, enabling training and increasing hindlimb muscle mass. Nonetheless, as shown in this study, BCD-induced training alone (ie, without clenbuterol) can only partially prevent muscular atrophy in the lower extremities. We have previously shown that clenbuterol alone can partially increase muscle mass and size in Tx mice. 18 We now show that clenbuterol treatment combined with BCD-induced training increases muscle mass, whole muscle CSA, and single-fiber CSA values. In fact, those values were similar to those in non-Tx mice. However, not all muscles were equally affected by this treatment (Figure 3B). Other tests are needed to investigate further the mechanisms underlying this muscle type–specific effect and also to determine whether this increase in muscle mass and fiber CSA was indeed translated into higher levels of contraction, force production, or resistance to fatigue.
Our study also showed that there was important adipose tissue alteration after SCI. Obesity in the spinal cord–injured population is often reported. 35 It is well known that excess intra-abdominal fat is strongly correlated with serious health complications, such as cardiovascular disease, type 2 diabetes, and insulin resistance. Treadmill training induced by BCD + clenbuterol could potentially counter obesity problems in this population because, as we have shown, intra-abdominal adipose tissue accumulations (visceral and retroperitoneal area) were lower in this group, probably because of training and clenbuterol lipolytic action. 36
The use of clenbuterol in SCI patients has not been explored. In animal models of disuse and atrophy, clenbuterol has been shown to stimulate muscle growth and anabolism.14-17,37 Adverse effects may also be found. For example, clenbuterol induced a slow to fast muscle phenotype transition, especially in the soleus.38,39 We showed that BCD + clenbuterol–treated Tx mice had the lowest proportion of type I fiber in the soleus, which also corroborates our previous finding. 18 Clenbuterol may also induce or accelerate bone tissue loss, as suggested in this study (Figure 4) and elsewhere.40,41 It remains unclear if such deleterious effects on bone properties could be prevented with administration of bisphosphonates typically used for age-related osteoporosis.42,43
Although, physical activity itself is generally known to possess osteogenic potential, 44 2 months of treadmill training induced pharmacologically did not prevent femoral bone tissue losses with or without clenbuterol. 11 A single case report also showed lack of effects of exercise only on SCI-related bone loss in humans. 45 Because severe bone loss is rapidly induced after immobilization, increased fracture incidence is generally found in patients with early, chronic SCI.46,47 Other strategies will be needed to prevent osteoporosis and osteopenia.
Clinical Implications
This study has provided strong evidence that a combination of CPG-activating drugs, training, and clenbuterol may largely prevent muscular atrophy. All the drugs that we used to activate the CPG (buspirone, carbidopa, and L-DOPA) are already approved by the Food and Drug Administration as treatments for other neurological problems (Parkinson disease and anxiety), which may accelerate their development for SCI.
Footnotes
Acknowledgements
This study was supported by the Canadian Institutes of Health Research (CIHR), the Fonds de Recherche en Santé du Québec (FRSQ), and Nordic Life Science Pipeline.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
