Abstract
Background. Substantial skeletal muscle atrophy after spinal cord injury (SCI) carries significant repercussions for functional recovery and longer-term health. Objective. To compare the efficacy, safety, and feasibility of functional electrical stimulation–assisted cycling (FESC) and passive cycling (PC) to attenuate muscle atrophy after acute SCI. Methods. This multicenter, assessor-blinded phase I/II trial randomized participants at 4 weeks post-SCI to FESC or PC (4 sessions per week, 1 hour maximum per session, over 12 weeks). The primary outcome measure was mean maximum cross-sectional area (CSA) of thigh and calf muscles (magnetic resonance imaging), and secondary outcome measures comprised body composition (dual energy X-ray absorptiometry), anthropometry, quality of life, and adverse events (AEs). Results. Of 24 participants, 19 completed the 12-week trial (10 FESC, 9 PC, 18 male). Those participants completed >80% of training sessions (FESC, 83.5%; PC, 85.9%). No significant between-group difference in postintervention muscle CSA was found. No significant between-group difference was found for any other tissue, anthropometric parameter, or behavioral variable or AEs. Six participants experienced thigh hypertrophy (FESC = 3; PC = 3). Atrophy was attenuated (<30%) in 15 cases (FESC = 7; PC = 8). Conclusions. Both cycle ergometry regimens examined were safe, feasible, and well tolerated early after SCI. No conclusions regarding efficacy can be drawn from our data. Further investigation of both modalities early after SCI is required.
Keywords
Introduction
Acute physiological responses to traumatic spinal cord injury (SCI) involve a period of spinal shock, which may take weeks to resolve.1-3 The combination of flaccid paralysis and disuse causes rapid musculoskeletal atrophy and deterioration,4-6 with detrimental consequences for functional recovery and longer-term health.
After the resolution of spinal shock, spontaneous recovery of neurological function or reflex activity (hypertonicity), or both may occur. A majority of spontaneous motor recovery occurs within 3 to 9 months postinjury,7,8 well after substantial atrophy has occurred. Advancements in emergency and surgical management have seen increased proportions of incomplete injuries, such cases retaining some neuroanatomical continuity and neurophysiological function across the lesion site. 9 To maximize rehabilitation potential, it is critical to maintain the health of musculoskeletal target tissues in anticipation of the resolution of spinal shock and the subsequent occurrence of neurological recovery.
Considerable cardiometabolic health benefits are conferred through skeletal muscle tissue metabolism, even in the absence of substantial functional recovery (eg, complete SCI). A major cause of death of SCI survivors is cardiovascular disease.10,11 After SCI, intramuscular adiposity is observed as early as 6 weeks postinjury. 12 Accrual of adipose tissue and its associated inflammatory burden over time contributes significantly to cardiometabolic risk.5,11,13 High prevalence of associated cardiometabolic health risks (eg, hyperinsulinemia, diabetes mellitus, and dyslipidemia) has been documented.14,15 Maintaining lean body mass and skeletal muscle metabolism through physical exercise interventions could modify these risk factors.16-18
Whereas evidence supports the health benefits of neuromuscular stimulation in chronic SCI,17,19,20 animal studies suggest that initiating exercise during a critical early period may optimize latent potential for functional recovery. 21 Notably, combining cellular or pharmacological therapies with exercise early after experimental injury shows synergistic benefits.22,23 Recent phase I/IIa clinical studies, which administered therapeutic agents within 48 hours, 24 emphasize the importance of understanding the effects of early interventions.
Given the rapid onset of musculoskeletal deterioration, identifying safe, effective, and well-tolerated exercise interventions to attenuate early muscle atrophy is crucial. Early mobilization of critically ill patients improves patient outcomes and decreases length of intensive care stay.25,26 Specifically, supine functional electrical stimulation (FES) cycling was safe and effective as early as 5 days after intensive care admission.27-29
A recent systematic review concluded that various forms of exercise for the paralyzed lower limbs have been utilized within the first several weeks postinjury, without associated adverse events (AEs). 30 However, the studies were small and few, and subject to bias. Conclusions regarding efficacy were further limited by diverse and suboptimal outcome measures.
The present investigation sought to address these gaps in the literature by utilizing rigorous research methods to examine the effects of early exercise, with maximum thigh cross-sectional area (CSA) as the primary outcome. Magnetic resonance imaging (MRI) has been validated against cadaver dissection for the measurement of muscle mass and determination of maximum thigh CSA, and is considered the gold standard. 31
The aim of this study was to examine the efficacy, safety, feasibility, and tolerability (ie, compliance) of 2 interventions—FES-assisted cycling (FESC) and passive cycling (PC)—initiated within 4 weeks of sustaining traumatic SCI. Our a priori hypotheses were that FESC would better attenuate the occurrence of muscle atrophy in the lower limbs than PC and that both interventions would be safe, feasible, and well tolerated. 32 Secondary objectives were to compare differences in body composition, neurological function, depression, and quality of life between the interventions.
Methods
Design
The Switch-On study was a multisite, assessor-blinded, phase I/II randomized controlled trial. The trial was registered on the Australian and New Zealand Clinical Trials Registry (ACTRN12611001079932) on October 18, 2011. The first participant was randomized on November 27, 2012, and the final participant on March 31, 2014.
Setting
Four specialized spinal units in Australia and New Zealand were involved. Ethical approval was obtained from the Human Research Ethics Committees at Austin Health (H2012/04476), Royal Perth Hospital (EC 2012/080), Princess Alexandra Hospital (HREC/12/QPAH/085), the Multi-region Ethics Committee in New Zealand (MEC/12/02/020), and the University of Melbourne (Trial Sponsor, HREC 1237599).
Participants
Participants were recruited from a consecutive sample of admissions at each site. Participants were included if they were ≥18 years and had sustained a motor complete or incomplete SCI (AIS [American Spinal Injuries Association (ASIA) Impairment Scale] A-C) above the neurological level of T12 no more than four weeks post-injury, and had medical and surgical clearance to participate. Exclusion criteria were selected to maximize safety and experimental rigor. Participants were excluded if they had a non-traumatic SCI, a long bone or pelvic fracture, or pressure injury. Full inclusion and exclusion criteria are detailed in the published protocol. 32
Randomization
Eligible participants were provided with written information and informed consent was obtained prior to baseline assessment. Participants then were randomly assigned to treatment group with a 1:1 allocation using a computer-generated randomization schedule, stratified by site and injury status (AIS A, B or C). Randomization was controlled independently by Neuroscience Trials Australia. In this single-blind design, assessors were blinded to group allocation.
Usual Care
All participants continued to receive usual care, including personalized physiotherapy provided by registered physiotherapists, as appropriate for the stage and according to the participants’ impairments and goals; however, clinical lower-limb electrical stimulation was not permitted during the 12-week intervention period.
Interventions
The supine cycling protocol utilized a stationary leg cycle ergometer (MOTOmed Letto, Hasomed GmbH, Magdeburg, Germany). Another upright ergometer, operated from a seated position (RT300, Restorative Therapies, Baltimore, MD) was used once participants were mobilized. All ergometers could provide either FESC or PC. Sessions were scheduled 4 times per week for 12 weeks. The maximum duration of each session was 60 minutes.
For FESC, a multichannel stimulator coordinated the neuromuscular stimulation of the quadriceps, hamstrings, and gluteal muscles in a pattern synchronized to the pedal position, such that the activated muscles would drive the ergometer at a low cadence (30-50 rpm), which has been shown to evoke greater hypertrophy than higher cadences. 33 The FESC protocol included 5 minutes of PC as a warm-up prior to stimulation. The electrodes used were pairs of 5 cm × 9 cm reusable self-adhesive bipolar surface electrodes applied to quadriceps, hamstrings, and gluteal muscles proximal and distal to the motor points, spaced approximately 4 cm apart. The quadriceps electrodes were placed approximately 5 cm from the proximal part of the patella, and the hamstrings electrodes were placed approximately 5 cm above the knee crease. The distal gluteal electrode was placed along the gluteal fold. Stimulation parameters for the FESC group were the following: stimulation intensity gradually increased to a maximum of 140 mA, pulse width between 0.3 and 0.5 ms, frequency of 35 Hz, pedal cadence of 30 to 40 rev min-1. Amplitude and pulse width were varied according to individual comfort and capability, depending on muscle size and thickness of subcutaneous fat. The stimulation parameters were consistent for each participant.
When muscles failed to produce enough power to maintain 30 rpm with the stimulation amplitude at a tolerable intensity, or at the onset of signs of muscle fibrillation (palpable or visual), stimulation ceased and 2 minutes of PC concluded the session. The procedure for PC was the same, without stimulation or resistance. PC participants were instructed to relax and not try to push the pedals.
Outcome Measures
Blinded assessors from all sites were trained in the procedures described below. Assessments were conducted after obtaining consent and prior to randomization (baseline) and within 1 week after completion of the intervention (postintervention). Data collected by clinicians included all AEs, body weight, and physiological responses, including pain, heart rate, and blood pressure.
Muscle Atrophy: MRI Acquisition
The primary outcome measure was maximum CSA measurement of the thigh and calf (mean of left and right), as determined from MRI T1-weighted images. Participants wore antiembolism stockings for ≥30 minutes prior to imaging to minimize fluid shifts. Patients were positioned on the scanner platform supine, with knee and hip joints extended and ankles braced at 90°. A 1.5-T whole-body coil acquired transaxial images of the thigh and leg. A T1-weighted, high-resolution, gradient echo profile maximized anatomical visualization (matrix 256 × 256 pixels, slice thickness 10 mm, and interslice gap 5 mm).
Muscle Atrophy: Image Analysis
Dicom files were analyzed off-line by a single-blinded assessor using open-source software (OsiriX, version 6.5.2 32-bit, Pixmeo, Bernex, Switzerland). Thigh muscles were traced separately. Calf muscles were traced in groups (anterior and posterior). Because the quadriceps comprises 45% to 50% of the thigh muscle volume,34,35 maximum thigh CSA was defined as the summed CSA of all muscles at the level of maximum quadriceps CSA. Both limbs were analyzed and values averaged.
Neurological Function
Trained physiotherapists and physicians assessed neurological impairment using the International Standards for the Neurological Classification of Spinal Cord Injury instrument (revised 2011). 36 Pain was recorded using a Visual Analogue Scale (VAS; 0-10).
Anthropometric Measurements
Girth measurements of the chest and waist were taken using a cloth measuring tape. Lower-limb girths and skinfold thickness were also recorded according to the International Society for the Advancement of Kinanthropometry standards. 37
Body Composition
Dual energy X-ray absorptiometry (DEXA) whole body photon beam scans were performed at each site according to the standardized procedure. Instruments included 2 GE-Lunar Prodigy scanners (GE Healthcare, Cleveland, OH; enCORE software versions 10.5 and v13.60, Madison, WI), a GE-Lunar iDXA (GE Healthcare, Cleveland OH; enCORE software version 13.40, Madison, WI), and a Hologic Discovery A (Hologic, Bedford, MA; APEX software version 3.5.2.1). The average absolute difference between measurements on these scanners was reported to be 1.4% for percentage fat mass, and <700 g for appendicular lean mass. 38 Inherent limitations include the following: (1) data from different DEXA scanners cannot be readily aggregated; (2) total body water assumptions contribute to variability in lean body mass estimates; and (3) fat mass influences photon beam dispersion. Participants wore light clothing with no metal components, and all metal jewelry was removed. Patients were positioned supine with the limbs abducted slightly. If pillows were required for positioning, this was noted and replicated for the postintervention scan.
Quality of Life
Depression and quality of life were assessed using validated instruments: Assessment of Quality of Life 8, Health Utilities Index 3, Hospital Anxiety and Depression Scale, and World Health Organisation Quality of Life—Australian Version 2000. 32
Adverse Events
All AEs and serious AEs (SAEs) were monitored and recorded. The clinical investigator or site coordinator used clinical judgment to determine severity, causality, and expectedness of all AEs.
Statistical Analyses
Sample Size Calculation
A power analysis was based on available published data 4 from which we assumed that participants in the PC group would lose approximately 33% of baseline CSA over a 12-week period. Assuming a clinically relevant FESC effect would limit atrophy to 10%, and α = .05, a total sample size of 42 patients would yield a power of 0.8 to reject the null hypothesis using analysis of covariance (ANCOVA). Allowing for a 15% drop-out rate, the required sample size was calculated to be 50 patients, equally distributed between groups.
Data Integrity and Management
Data were stored electronically on a database with secured and restricted access and an audit trail in line with (International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use [ICH] -Good Clinical Practice [GCP]) guidelines.
Monitoring
The trial was overseen and monitored by a Trial Manager and Data Safety Monitoring Board. The Trial Manager ensured data quality and monitored compliance with the trial protocol during visits to each site. An interim safety analysis was planned to take place when 25 participants had completed their postintervention assessment.
Hypothesis Testing
The primary outcome was maximum CSA measurement of the thigh and calf (mean of left and right). Secondary outcomes were AEs, body composition (total percentage fat, lower-limb lean and fat mass, anterior thigh skinfold), quality of life, and depression.
The blinded analyses were conducted according to a predetermined analysis plan on an intention-to-treat basis. Between-group differences were tested independently at the 0.05 level of significance, using a 1-way ANCOVA model, with robust standard error estimates that included the baseline value of the outcome as a covariate. This analysis was then further cross-validated using median regression. Median regression was also used to analyze between-group differences in scores on the questionnaires for Depression and Quality of Life. The differences in AEs between groups were investigated using negative binomial regression and the effects reported as incidence rate ratios (IRRs) with corresponding 95% CIs. No formal multiplicity adjustments were undertaken to constrain the overall type I error associated with the secondary analyses. Their purpose was to supplement evidence from the confirmatory primary analysis to more fully characterize the treatment effect. Results from the secondary analyses should be interpreted in this context.
The intention-to-treat strategy was based on an assumption that data were missing at random. 39 The sensitivity of the results to plausible departures from the missing-at-random assumption as part of the intention-to-treat analysis were explored using both a selection model (modeling of the missing data mechanism) and a pattern mixture model (modeling of the differences between missing and observed data). Assumptions about the missing data were expressed via a parameter that measures the degree of departure from the missing-at-random assumption. The results were graphed over a range of assumptions using the rctmiss suite of commands in Stata (version 13, StataCorp, College Station, TX).
Results
Participants
Recruitment
Of 187 acute SCI admissions screened, 61% were excluded according to the criteria. Figure 1 shows the reasons for exclusion and the flow of participants through the study. In all, 8 patients were ineligible based on inability to consent because of psychological (n = 5) or language barriers (n = 3). Transfers to the spinal unit after 4 weeks postinjury occurred at 2 sites (n = 10). Planned discharges resulting in short lengths of stay prohibited enrolment in 14 cases; 7 eligible participants did not consent. Recruitment was slower than expected; the trial was terminated after randomization of 24 participants because of time limitations associated with funding. Sufficient sample size, according to an a priori power calculation, was not achieved.
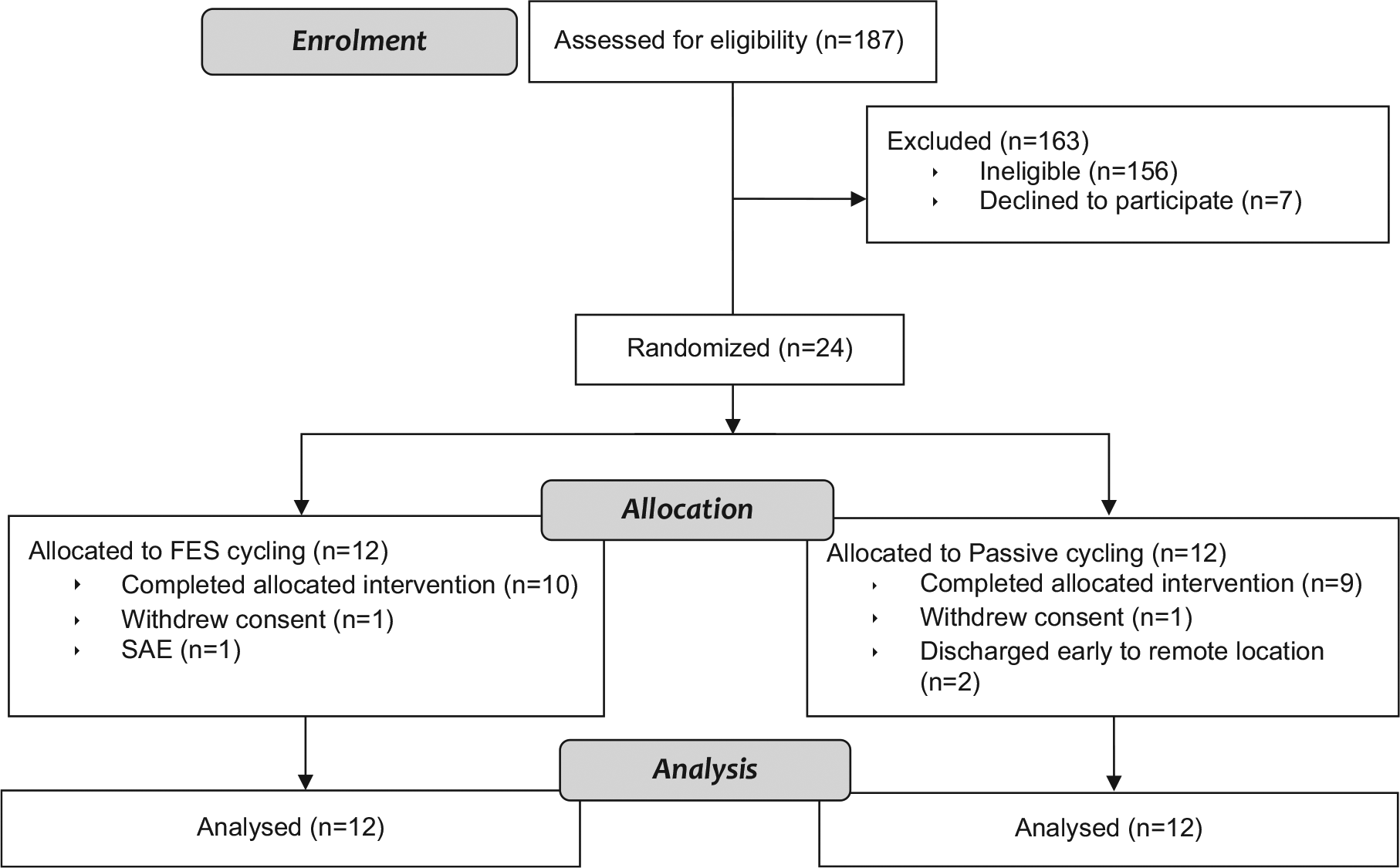
CONSORT flowchart for intention-to-treat analysis.
Baseline Characteristics
Table 1 presents demographic data and baseline characteristics for each group. The groups were similar with respect to age, body mass index, days postinjury, and AIS classification with the following exceptions. In the FESC group, 7 participants had normal upper-limb function, compared with 3 in the PC group. Notably, 4 participants in the FESC group were classified AIS C, with some lower-limb function (lower-extremity motor score [LEMS] range = 4-19), compared with 2 in the PC group (LEMS range = 2-9).
Baseline Demographic, and Injury Characteristics.
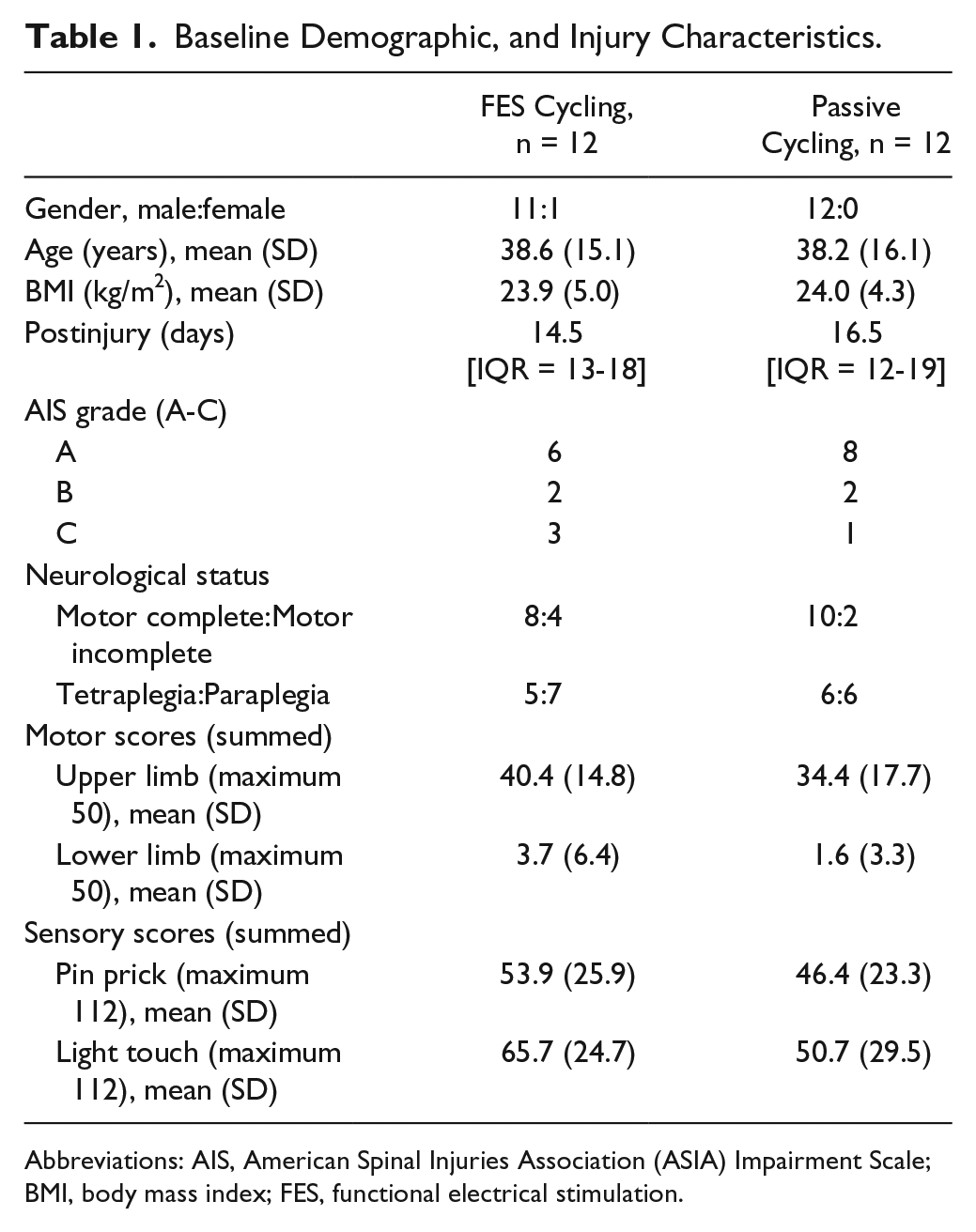
Abbreviations: AIS, American Spinal Injuries Association (ASIA) Impairment Scale; BMI, body mass index; FES, functional electrical stimulation.
Hypothesis Testing
No statistically significant between-group differences were found for the primary outcome measures (mean maximum CSA of thigh and calf), nor for any other tissue, anthropometric, or behavioral parameter or AEs. Between-group results are presented in Table 2 (primary outcomes) and Table 3 (secondary outcomes). Figure 2 demonstrates considerable within-group variation in percentage change for CSA thigh, specific to the FESC group.
Intention-to-Treat Analysis for Primary Study Outcomes (Mean Thigh CSA, Mean Calf CSA).
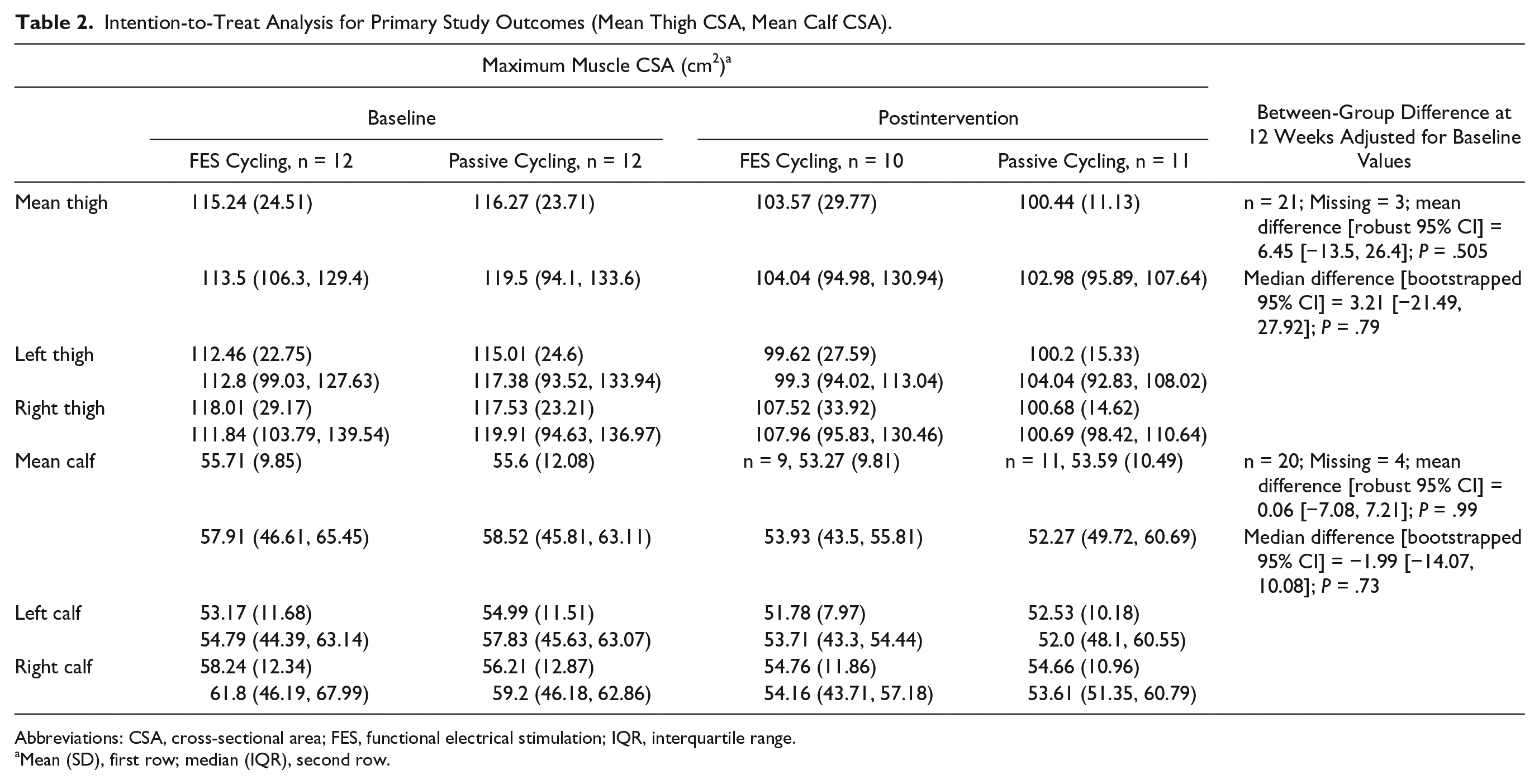
Abbreviations: CSA, cross-sectional area; FES, functional electrical stimulation; IQR, interquartile range.
Mean (SD), first row; median (IQR), second row.
Intention-to-Treat Analysis for Secondary Study Outcomes.
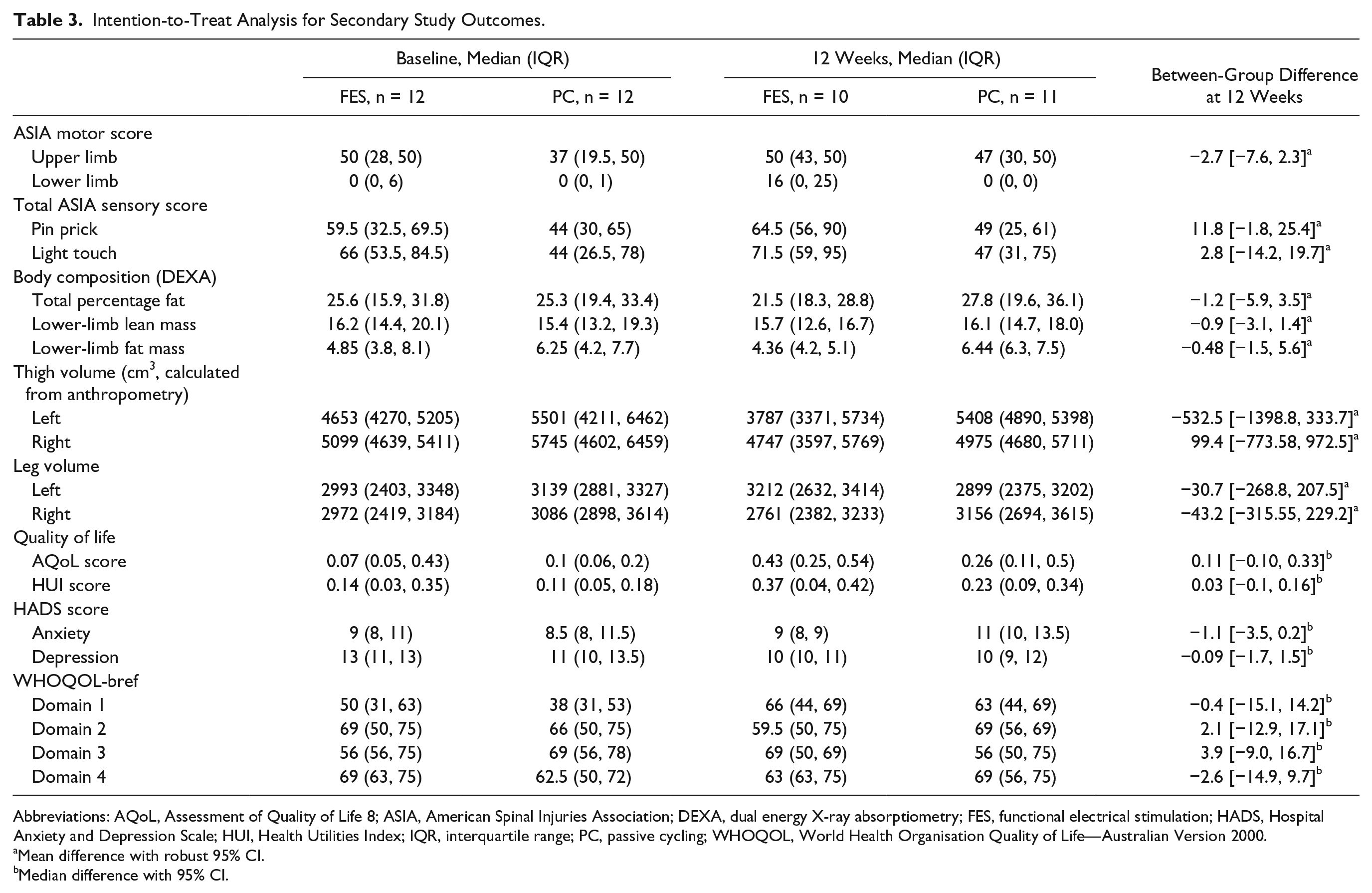
Abbreviations: AQoL, Assessment of Quality of Life 8; ASIA, American Spinal Injuries Association; DEXA, dual energy X-ray absorptiometry; FES, functional electrical stimulation; HADS, Hospital Anxiety and Depression Scale; HUI, Health Utilities Index; IQR, interquartile range; PC, passive cycling; WHOQOL, World Health Organisation Quality of Life—Australian Version 2000.
Mean difference with robust 95% CI.
Median difference with 95% CI.
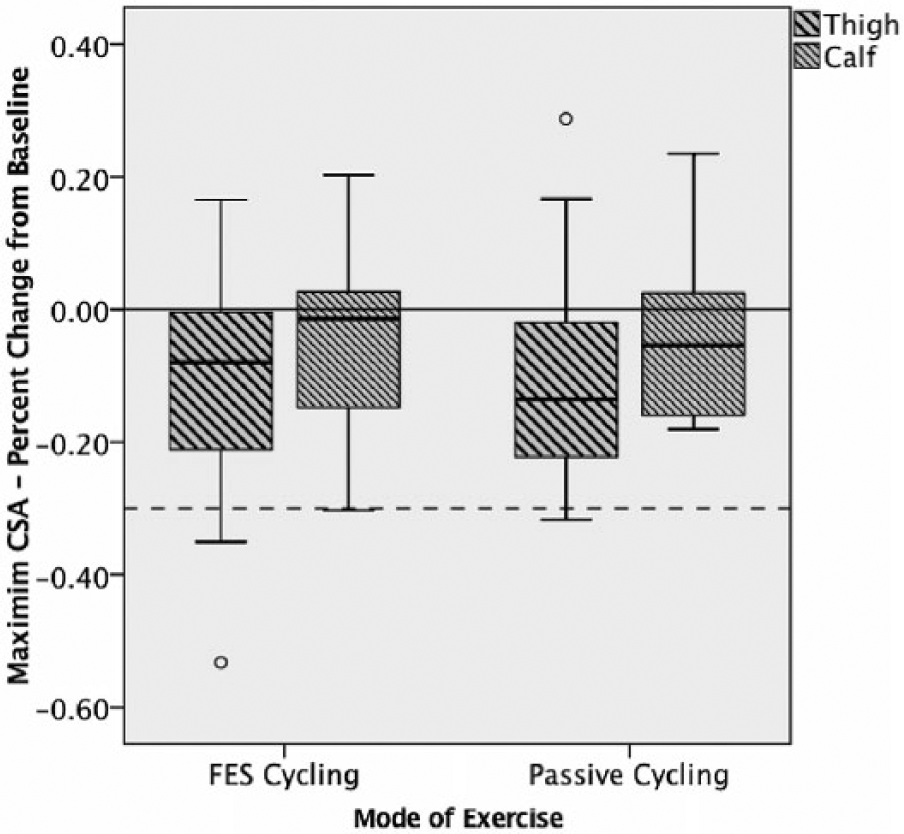
Maximum CSA of thigh and calf (percentage change from baseline) by group.
Retention and Intervention Compliance
Participants completed 12 to 13 weeks of intervention with 5 exceptions. In the FESC group, 1 participant withdrew consent after 6 weeks, and 1 was withdrawn as a result of a SAE. One person from the PC group withdrew consent after 4 sessions, and 2 were discharged early (to remote locations). Those who completed the intervention attended a mean of 3.3 (±0.4) sessions per week. The mean cumulative total exercise time was slightly longer for the PC group (32.6 hours, SD = 10.5 hours) compared with the FESC group (30 hours, SD = 11 hours), but this difference was not statistically significant. Median maximum heart rate for the PC group (78 bpm, interquartile range [IQR] = 71-89) was significantly lower than that for the FESC group (98 bpm, IQR = 87-105, P = .013). The median stimulation amplitude of all FES channels was 90 mA (IQR = 47-130 mA). The power output for FESC and PC groups is presented in Table 4.
Individual Participant Characteristics, Intervention Parameters, and Outcomes.
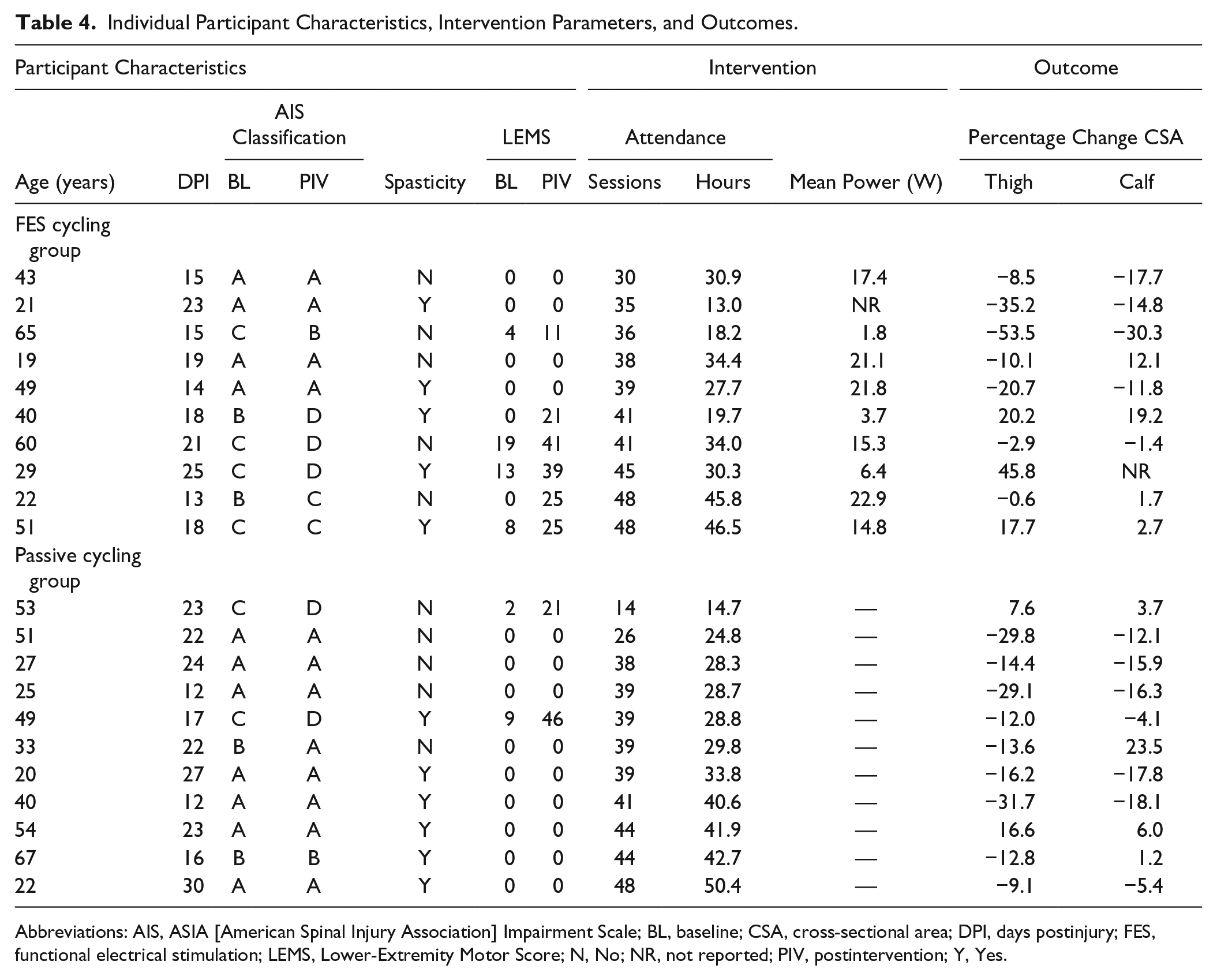
Abbreviations: AIS, ASIA [American Spinal Injury Association] Impairment Scale; BL, baseline; CSA, cross-sectional area; DPI, days postinjury; FES, functional electrical stimulation; LEMS, Lower-Extremity Motor Score; N, No; NR, not reported; PIV, postintervention; Y, Yes.
Adverse Events and Pain
A total of 10 SAEs (6 FESC; 4 PC) and 281 AEs were recorded (159 FESC; 122 PC). No significant between-group difference in SAEs (IRR = 1.25 [95% CI = 0.24, 6.65]; P = .79), or AEs (IRR = 1.38 [95% CI = 0.72, 2.65]; P = .34) was observed. One SAE (hematoma over the ischium) was recorded that was considered probably causally related to the FESC intervention, and the participant was withdrawn. Although the participant was at increased risk as a result of anticoagulation therapy, such treatments are routine, and care should be taken to monitor for hematoma. One AE (participant grazed leg against bike) was considered definitely related to the FESC intervention; no treatment was required. Another AE (increase in blood pressure during an intervention session) was considered probably related to the FESC intervention, and that intervention session was stopped. However, the participant subsequently resumed intervention. No difference in mean reported pain level was detected between groups: VAS FES 1.33/10 (IQR = 0.08-4.34), VAS PC 2.23/10 (IQR = 0.37-3.82); P = .53.
AIS Conversion and Ambulation (Table 4)
Two participants were classified as AIS B at baseline (FESC, n = 2). At study exit, 1 participant had converted to AIS C and another to AIS D. The AIS C participant was able to walk short distances with assistive devices, whereas the AIS D participant remained wheelchair dependent.
Six participants were classified as AIS C at baseline (PC, n = 2; FESC, n = 4). Of these, 4 participants were able to walk short distances with therapist assistance or assistive devices (PC, n = 1; FESC, n = 3) at 12 weeks. The remainder did not recover ambulatory functions (PC, n = 1; FESC, n = 1).
Discussion
The results of this randomized controlled trial indicate that cycle ergometry is safe, and feasible early post-SCI, when administered under controlled conditions. One SAE was potentially causally related and 2 of 281 AEs were deemed to be definitely and probably causally related to the FESC intervention, respectively. In contrast, no SAE or AE was deemed to be definitely causally related to the PC intervention. Both the FESC and PC interventions were initiated prior to 4.5 weeks postinjury. Sessions were scheduled to occur 4 times per week; the actual mean attendance exceeded expectations, and high overall attendance (80%) further demonstrates the feasibility and tolerability of these early exercise interventions.
Our hypothesis, that FES cycling would be more efficacious than PC in preventing skeletal muscle atrophy, was not borne out; we were unable to detect a significant benefit of one intervention over the other. According to an a priori power calculation based on published data of thigh atrophy post-SCI, the study was underpowered to detect the hypothesized between-group differences. Despite the groups being comparable across most factors, the variability in baseline muscle CSA was substantial, and although unexpected, divergent responses to PC were a significant contributor to postintervention heterogeneity, This diluting effect further challenged the ability to detect within- or between-group differences.
The wide variability observed in percentage change in muscle CSA in the FESC group could reflect differences in the implementation, or fidelity, of the intervention. For example, explanations may include differences in cumulative exercise times or resistance achieved, or postintervention lower-limb motor scores. It was neither feasible, nor ethical, to control for effects of standard care physical interventions other than electrical stimulation, and in cases where substantial neurological recovery occurred, weight-bearing or over-ground gait training may have been a confounding factor. A larger sample size and longitudinal follow-up would be required to determine any causal relationships between exercise, muscle anabolism, neurological recovery, and functional gains. Despite these limitations, these results were positive, in that the mean percentage reduction in thigh and calf muscle CSA for both groups was lower than would have been expected 40 if the intervention had had no anabolic (trophic) or anticatabolic effect (Table 4). In all, 75% of all participants (8 FESC, 10 PC) experienced a <30% reduction in thigh muscle CSA, signifying an attenuation of muscle atrophy against previous reports.4,5,11 This anabolic or anticatabolic effect in the PC group further contributed to the dilution of any between-group effects.
We examined the hypothesis that FESC would better attenuate the occurrence of muscle atrophy in the lower limbs than PC, in part as a result of the interplay between resistance and muscle fiber–type properties and between muscle contraction and muscle-secreted trophic factors in the former. Whereas hypertrophy was hypothesized to occur in the FESC group, an increase in thigh CSA was observed in only 3 cases and was accompanied by concomitant locomotor recovery in this subset. Previously, 2 small randomized controlled trials had reported statistically significant FESC-inducible increases in vastus lateralis fiber CSA 41 and DEXA lower-limb lean mass estimates. 42 The latter study, which compared 12 to 13 weeks of FESC in acute complete SCI with standard care, also detected differences in lean mass with low-load cycling versus unloaded static electrical stimulation after 6 months of intervention but not after 3 months. It is not possible to compare these results with those of the present study because the present study did not use a standard care control group, and the intervention duration was limited to 12 weeks. Nonetheless, the extant data argue for a relationship between time postinjury and the ability to mount an anabolic response to neuromuscular stimulation. Another possible explanation for the lack of observable hypertrophy in more participants who underwent FESC could be related to the sensitivity of the primary outcome measure—maximum thigh muscle CSA measured at the axial location of maximum quadriceps CSA. Maximum CSA of the hamstrings is typically found more distally than the quadriceps and may have been undetected by our primary outcome measure. 43
Intriguingly, some participants in the PC group experienced hypertrophy of the thigh muscles, despite a lower-limb motor score of zero. Although explanations could involve accrual of intramuscular fat, tissue fibrosis, or spontaneous hypertrophy related to the resolution of spinal shock and subsequent emergence of hypertonicity, this finding warrants closer examination. Given the apparent attenuation of atrophy (<30%) in 6 other PC participants with motor complete injuries, these results point toward a treatment effect. There is scant evidence regarding the trophic effects of PC in humans. One observational study of 5 intensive care unit (ICU) patients reported that 1 week of single-leg PC resulted in 11% less muscle fiber atrophy than the control limb (which decreased by 35%). 44 In a rat model of ICU, attenuation of muscle fiber atrophy with PC was accompanied by reduced oxidative stress, decreased myosin losses, and spared force-generating capacity. 45 Attenuation of atrophy has also been demonstrated with PC in rat models of acute SCI.46,47 Early studies in “electrically silent” rat muscles demonstrated mechanical effects of PC on satellite cell activation, the precursor cell of muscle fibers, signifying muscle hypertrophy. 48 Gene expression of brain-derived neurotrophic factor (BDNF) and glial cell line–derived neurotrophic factor, growth factors which help regulate neuronal survival and synaptic strength, was upregulated in paralyzed muscles of SCI rats after only 1 PC session. 49 Similarly, BDNF, neurotrophin-3, and neurotrophin-4 were upregulated in the lumbar spinal cord enlargement within 4 weeks of PC, implying potentially bidirectional signaling via a neuromuscular retrograde/anterograde transport pathway. 50 Given these plausible mechanisms of action, the potential for PC, initiated early after SCI to attenuate atrophy in humans, together with the safety data presented here, merits further clinical investigation.
This clinical trial is important because it investigated exercise in paralyzed lower limbs very early after injury and used a rigorous research methodology to do so. Provision of early exercise interventions of this kind may well promote neurological recovery at a critical period postinjury when the extent of functional loss is not fully known. Indeed, the promising improvements reported following training of ambulation using neuromuscular stimulation 51 or a brain-computer interface 52 in people with chronic SCI support this notion. The main limitation of our study was the small sample size, which occurred despite recruitment at multiple sites over 28 months.
Conclusions
Our study demonstrates the safety, feasibility, and tolerability of cycle ergometry provided to the paralyzed lower limbs within 4 weeks post-SCI. Despite being underpowered to detect whether one intervention was more efficacious in attenuating skeletal muscle atrophy than the other, we can conclude that the interventions are not harmful and are potentially beneficial. Indeed, it appears that both interventions may be able to attenuate muscle atrophy compared with published data of thigh muscle atrophy without intervention.
Attenuation of skeletal muscle atrophy early post-SCI may be crucial to maximize rehabilitation potential. Furthermore, because early exercise is increasingly used as an adjunct in trials of therapeutic agents,7,53 its singular effects should be documented. Given the known difficulties of achieving sufficient sample size in SCI trials, further investigation of such interventions should be undertaken in a rigorous and standardized manner to facilitate future meta-analysis.
Footnotes
Acknowledgements
The authors gratefully acknowledge the participants and their families.
Authors’ Note
The trial was registered on the Australian and New Zealand Clinical Trials Registry (ACTRN12611001079932) on October 18, 2011.
Collaborators
Melanie Hurley (The University of Melbourne, Victoria, Australia) - Trial Coordinator
Dr Andrew Nunn (Austin Health, Heidelberg, Victoria, Australia) - Principal Investigator
Janette Alexander (Austin Health, Heidelberg, Victoria, Australia) - Site Coordinator
Prof John Buchanan (Royal Perth Hospital, Perth, Western Australia) - Principal Investigator
Sherilyn Nolan (Royal Perth Hospital, Perth, Western Australia) - Site Coordinator
Dr Sridhar Atresh (Princess Alexandra Hospital, Woolloongabba, Queensland, Australia) - Principal Investigator
Valerie Pick (Princess Alexandra Hospital, Woolloongabba, Queensland, Australia) - Site Coordinator
Dr Rick Acland (Burwood Spinal Unit, Christchurch, New Zealand) - Principal Investigator
Jo Nunnerley (Burwood Spinal Rehabilitation Unit, Christchurch, New Zealand) - Site Coordinator
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Transport Accident Commission (Victorian Neurotrauma Initiative) and the National Health and Medical Research Council of Australia (APP1042653).
