Abstract
Background. Studies demonstrate that most arm motor recovery occurs within three months after stroke, when measured with standard clinical scales. Improvements on these measures, however, reflect a combination of recovery in motor control, increases in strength, and acquisition of compensatory strategies. Objective. To isolate and characterize the time course of recovery of arm motor control over the first year poststroke. Methods. Longitudinal study of 18 participants with acute ischemic stroke. Motor control was evaluated using a global kinematic measure derived from a 2-dimensional reaching task designed to minimize the need for antigravity strength and prevent compensation. Arm impairment was evaluated with the Fugl-Meyer Assessment of the upper extremity (FMA-UE), activity limitation with the Action Research Arm Test (ARAT), and strength with biceps dynamometry. Assessments were conducted at: 1.5, 5, 14, 27, and 54 weeks poststroke. Results. Motor control in the paretic arm improved up to week 5, with no further improvement beyond this time point. In contrast, improvements in the FMA-UE, ARAT, and biceps dynamometry continued beyond 5 weeks, with a similar magnitude of improvement between weeks 5 and 54 as the one observed between weeks 1.5 and 5. Conclusions. Recovery after stroke plateaued much earlier for arm motor control, isolated with a global kinematic measure, compared to motor function assessed with clinical scales. This dissociation between the time courses of kinematic and clinical measures of recovery may be due to the contribution of strength improvement to the latter. Novel interventions, focused on the first month poststroke, will be required to exploit the narrower window of spontaneous recovery for motor control.
Introduction
Clinical studies have shown that arm paresis is present in 50-80% of stroke survivors, and that recovery from this impairment is complete by six months after stroke.1-7 The precise time course of this recovery, however, depends on the outcome measure chosen. Activity limitation assessments, such as the Action Research Arm Test (ARAT), cannot reliably distinguish restitution from compensation. 8 Restitution entails a return toward premorbid patterns of motor control and normal levels of strength, whereas compensation refers to the use of alternative strategies to accomplish a task.8-11 Poststroke paresis comprises deficits in both strength and motor control, the latter being defined as the ability to make coordinated, accurate, goal-directed movements. 12 Motor control, and not just strength, is essential for skilled use of the limb and may determine how much it is used in everyday activities. The Fugl-Meyer Assessment of the upper extremity (FMA-UE), an impairment scale, is largely immune to compensation but, like the ARAT, has significant antigravity strength requirements.13-15 Consequently, its score blends strength with motor control, two components that have been shown to have dissociable recovery time courses after stroke.16-18 Recently, 3-dimensional (3D) kinematic tasks have been used to assess the recovery of motor control poststroke; however, 3D tasks also require significant antigravity strength.19-22
Here we sought to isolate and characterize the time course of recovery of arm motor control over the first year poststroke. Motor control was assayed with a global kinematic measure derived from a 2-dimensional (2D) reaching task, which we have previously used to dissociate motor control from antigravity strength and compensation.23,24 This task allows precise measurement of arm kinematics, thereby increasing specificity and sensitivity to motor control changes poststroke.23-26 The time course of recovery of arm motor control was compared with recovery measured with clinical scales of arm motor impairment, activity limitation, and strength.
Methods
Study Design
The Study of Motor Acute Recovery Time course after Stroke (SMARTS) was a multicenter, longitudinal investigation of motor recovery of the upper extremity. The participating centers were Johns Hopkins University, Columbia University, and University of Zurich. Here we report the arm kinematics substudy, for which the latter center did not participate.
Participants were scheduled to undergo testing at five poststroke intervals: Visit-1 within the first 2 weeks (±5 days), Visit-2 at 4 weeks (±7 days), Visit-3 at 12 weeks (±14 days), Visit-4 at 24 weeks (±14 days), and Visit-5 at 52 weeks (±14 days). Participants had to be enrolled by Visit-2. All local ethics boards approved this study. Informed consent was obtained for all study procedures.
Study Participants
Eligible participants had magnetic resonance imaging–confirmed first-time ischemic stroke with arm paresis. Exclusion criteria were the following: FMA-UE >63, hemorrhagic stroke, traumatic brain injury, visual field deficit greater than quadrantanopsia (assessed with item 3 in National Institutes of Health Stroke Scale), preexisting condition affecting arm function, additional neurological/psychiatric illness affecting motor performance or recovery, and inability to give informed consent.
We recruited participants from Johns Hopkins Hospital, New York Presbyterian Hospital, and affiliated institutions, between March 2012 and January 2014. Participants who completed at least two visits were included; the total sample size was 18. Demographics and clinical characteristics are summarized in Table 1.
Participant Demographic and Clinical Characteristics at Enrollment.
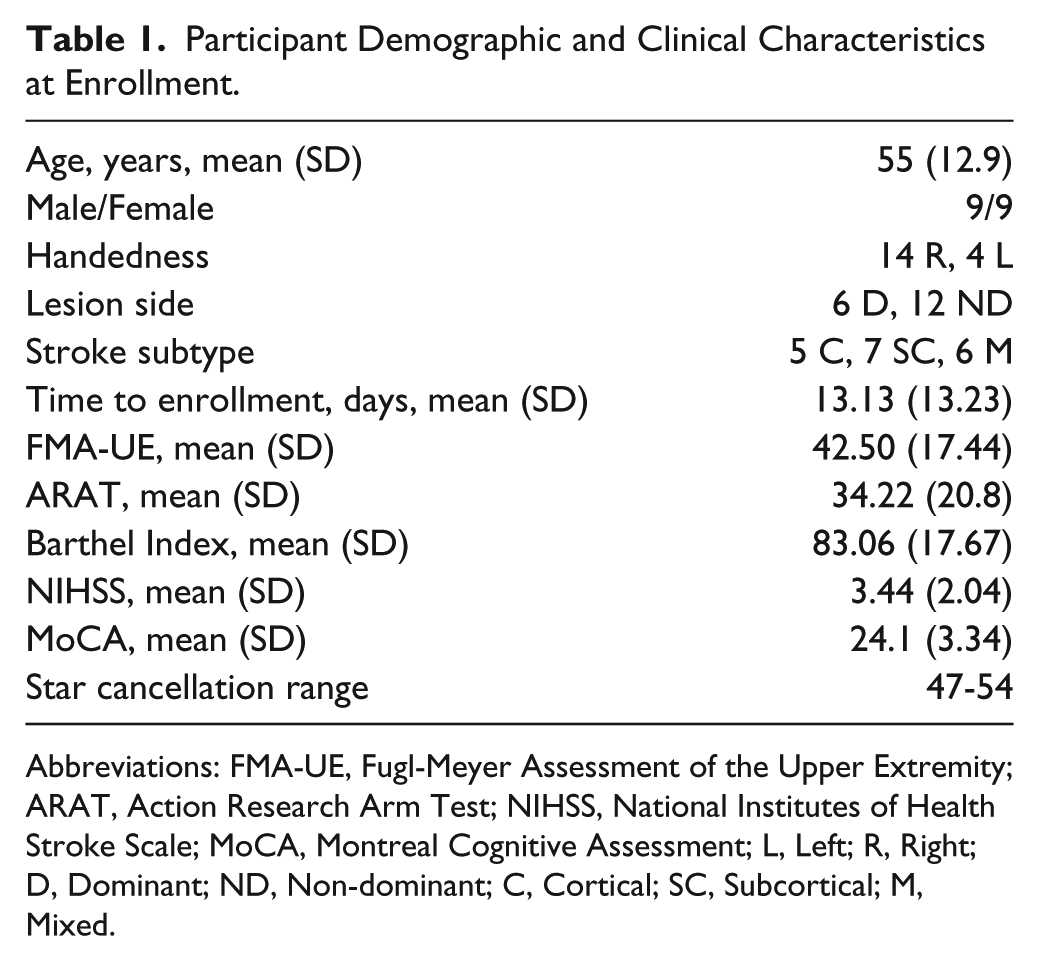
Abbreviations: FMA-UE, Fugl-Meyer Assessment of the Upper Extremity; ARAT, Action Research Arm Test; NIHSS, National Institutes of Health Stroke Scale; MoCA, Montreal Cognitive Assessment; L, Left; R, Right; D, Dominant; ND, Non-dominant; C, Cortical; SC, Subcortical; M, Mixed.
Twelve neurologically healthy volunteers with a similar age distribution as the stroke participants (mean age = 58.4 years) underwent arm kinematic testing at a single visit. This control group was used as the reference population for our trajectory analysis.
Clinical Measures
FMA-UE and ARAT were used to assess arm motor impairment and activity limitation, respectively.13,27,28 Both scales are ordinal, with a maximum score of 66 for FMA-UE, and of 57 for ARAT. Both measures have shown good reliability, validity, and sensitivity to poststroke motor changes.15,29,30 We tracked strength recovery of biceps brachii using dynamometry (MicroFET2TM, Pro Med Products, Atlanta, GA). The biceps brachii was chosen as it is a muscle used in our kinematic task. Maximum voluntary elbow flexion was measured while holding the dynamometer stationary against the participant’s wrist. 31 The average of 3 trials was transformed into a Z-score using a normative dataset. 32
Kinematic Task
Arm motor control was assayed with a 2D reaching task using the Kinereach® apparatus, which is designed to decrease strength requirements by providing antigravity support and reducing friction (Figure 1A). 33
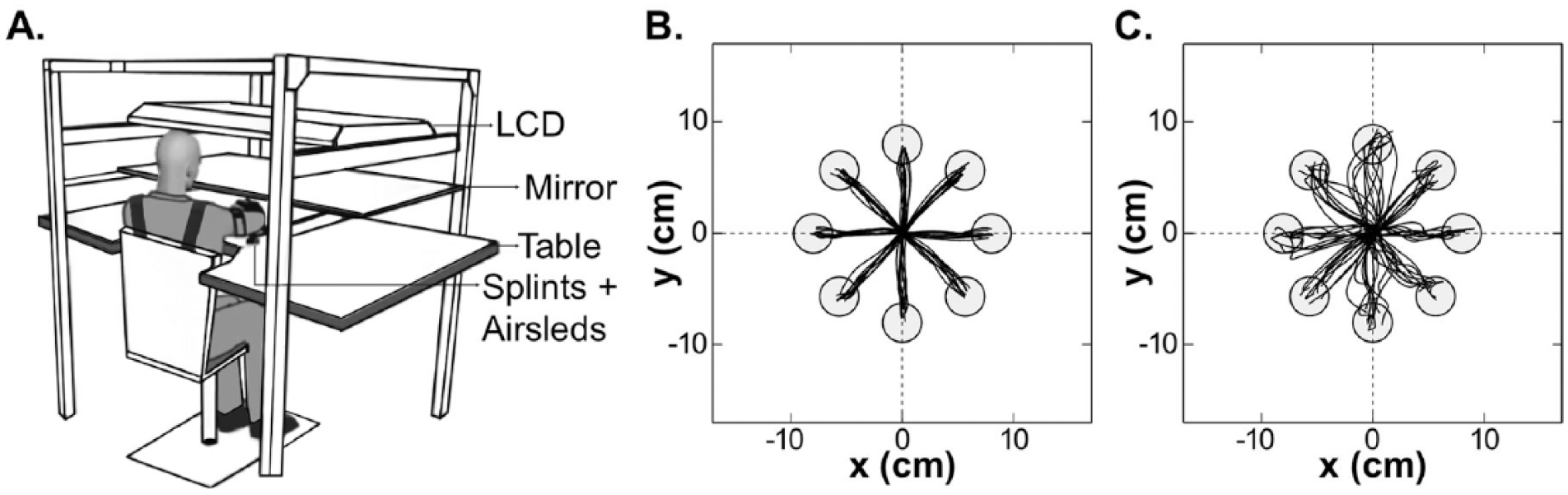
(A) Experimental kinematic apparatus (modified from Przybyla et al 33 ), and sample trajectories for (B) the dominant right arm of a healthy control and (C) the paretic dominant right arm of a stroke participant (Fugl-Meyer Assessment of the upper extremity = 58).
Participants sat at a glass-surface table with their trunk secured to a high-backed chair, thereby minimizing truncal compensation. Seat height was adjusted to bring the shoulder, elbow, and wrist close to the same horizontal plane. The hand and forearm were splinted, only allowing shoulder and elbow movements. Frictionless movements were attained via an air-sled system. To prevent direct visualization of the limb, a mirror reflecting an LCD lay above the forearm. Hand position was represented by a cursor on the display.
Participants were instructed to make 80 straight movements to eight circular targets with 1 cm radius, arrayed radially at 8 cm from a central start position. After holding the cursor at the start position for 0.5 seconds, individual targets appeared in a balanced pseudo-randomized order. Once a target appeared, participants had 3 seconds to complete the movement. Targets turned green making a pleasant ding for movements that ended inside them and had a peak speed between 10 and 40 cm/s. Hand position was tracked at 130 Hz with the Flock of Birds® system (Ascension Technology, USA). Both arms were tested in a pseudo-randomized order at each visit.
Kinematic Analysis
Hand position data were analyzed using IGOR Pro (Wavemetrics, USA). Position time-series were low-pass filtered (8 Hz Butterworth), and differentiated to yield speed and acceleration. Left hand data were horizontally flipped, thus allowing grouping of movements directed toward targets that required similar joint configurations. For each movement, we identified the peak speed at the first zero-crossing of acceleration above a threshold of 10 cm/s. The movement-start was defined as the time, before peak speed, when speed surpassed 2 cm/s. The movement-end was defined as the time, after peak speed, when speed remained less than 2 cm/s for more than 0.1 seconds. As we have done previously,23,24,34 in order to decrease contamination from target guesses, incomplete movements due to late reaction to target appearance, and involuntary movements due to failure to fully stabilize the arm in the starting position, we applied the following exclusion criteria: direction at peak speed ≥90° away from target direction, or ending ≤30% of target distance. However, for sake of completeness, we also conducted analyses with the full dataset.
Reaching kinematics were characterized using functional principal components analysis (FPCA), a generalization of traditional PCA to time-series data. This technique dramatically reduces the dimensionality of the analysis problem while retaining the major patterns that differ across movements. 35 This analysis strategy precludes a priori choice of specific kinematic variables, such as directional error, smoothness, or endpoint accuracy, which bias the analysis toward specific components of motor control. Instead, FPCA compares distributions of movements at a global level and is sensitive to changes in overall movement quality. We computed the squared Mahalanobis distance (MD2) with regard to the reaching trajectories from the dominant arm of the reference population. Participant-specific average squared Mahalanobis distances (AMD2) were computed for each participant at each target for each time-point. Details of this analysis have been reported previously and are provided in Supplemental Materials.24,34
Statistical Analysis
For each outcome variable (FMA-UE, ARAT, biceps dynamometry Z-score, and AMD2), linear mixed models were used to examine changes over time; this analytic framework extends repeated measures ANOVA to allow more general tests of association. Participant-specific random intercepts were used to account for within-participant correlations for all outcome variables. Because AMD2 was measured for each target, additional Participant/Visit-specific intercepts were used to account for correlations across targets at each visit. Visit was treated as a categorical predictor with five levels. This mixed-model analysis uses all available data to estimate Visit means so that missing visits do not cause the remainder of a participant’s data to be omitted. Nonconstant residual variance across visits was addressed by using weighted least squares; alternative methods to account for nonconstant variance, including square root and log transformations, yielded similar results. In this model framework, we made comparisons of interest (eg, Visit-1 vs. Visit-2, Visit-2 vs. Visit-5) using Wald tests for contrasts in coefficients.
Based on our recent work, the sample size was deemed to be powered to detect changes in motor control similar to those observed in chronic stroke patients undergoing therapy. 24
Results
From the cohort of 18 stroke participants, 13 were enrolled at Visit-1 and five at Visit-2. Nine participants completed all five visits. Four dropped out of the study (one after Visit-2, one after Visit-3, and two after Visit-4) due to lack of interest. One participant was too paretic to perform the kinematic task at Visit-1. Combining late enrollment, dropouts, and inability to perform the task, 13% of the data were missing. We conducted a sensitivity analysis on missing data by removing an additional 13% of the data at random and repeating our model fitting and testing steps, and obtained significant results for the same comparisons as in the analysis presented here (see Supplemental Materials). Additionally, applying our prespecified kinematic criteria we excluded 4.56% of movements in Visit-1, 0.58% in Visit-2, 0.61% in Visit-3, 0.78% in Visit-4, and 0.34% in Visit-5. Analysis of the whole dataset (without excluding any movements) did not change the main finding (see Supplemental Materials).
Median time-after-stroke, in weeks, for each visit was as follows: 1.6 for Visit-1 (SD = 2.7 days), 5.3 for Visit-2 (SD = 6.2 days), 13.9 for Visit-3 (SD = 5.6 days), 27 for Visit-4 (SD = 12.1 days), and 53.7 for Visit-5 (SD = 10 days). Henceforth, all results are reported using the rounded median time-after-stroke for each visit: Visit-1 = week 1.5, Visit-2 = week 5, Visit-3 = week 14, Visit-4 = week 27, and Visit-5 = week 54.
The primary analysis focused on changes in the paretic arm during early recovery (between weeks 1.5 and 5) and late recovery (between weeks 5 and 54). For each of the outcome variables, FMA-UE, ARAT, biceps dynamometry Z-score, and AMD2, we computed the difference from the regression model values between two given visits (delta), and the significance level for each comparison (adjusted P values reflecting multiple comparisons for primary outcomes appear in Supplementary Table 3, and agree with the conclusions presented here). AMD2 is a measure of the statistical distance between entire reaching trajectories made by each stroke participant and healthy controls; hence it is a global kinematic measure.
Recovery of Motor Control of the Arm Reached a Plateau at Week 5 Poststroke
Stroke participants had abnormal reaching trajectories (Figure 1B and C). Most notably, we found that recovery of arm motor control plateaued after five weeks (Figure 2). During early recovery, AMD2 for the paretic arm showed a significant decrease, that is, movements became more like those made by healthy controls (delta = −18.39, P < .001). During late recovery, AMD2 in the paretic arm showed no significant change (delta = 1.65, P = .50). Comparison of the paretic arm AMD2 with healthy controls showed significant differences across all visits (largest P < .01), thereby excluding the possibility of a ceiling effect for the kinematic task. Analyses for the nonparetic arm also showed a significant decrease in AMD2 in early recovery (delta = −3.54, P < .001), with no significant difference in late recovery (delta = 0.68, P = .08).
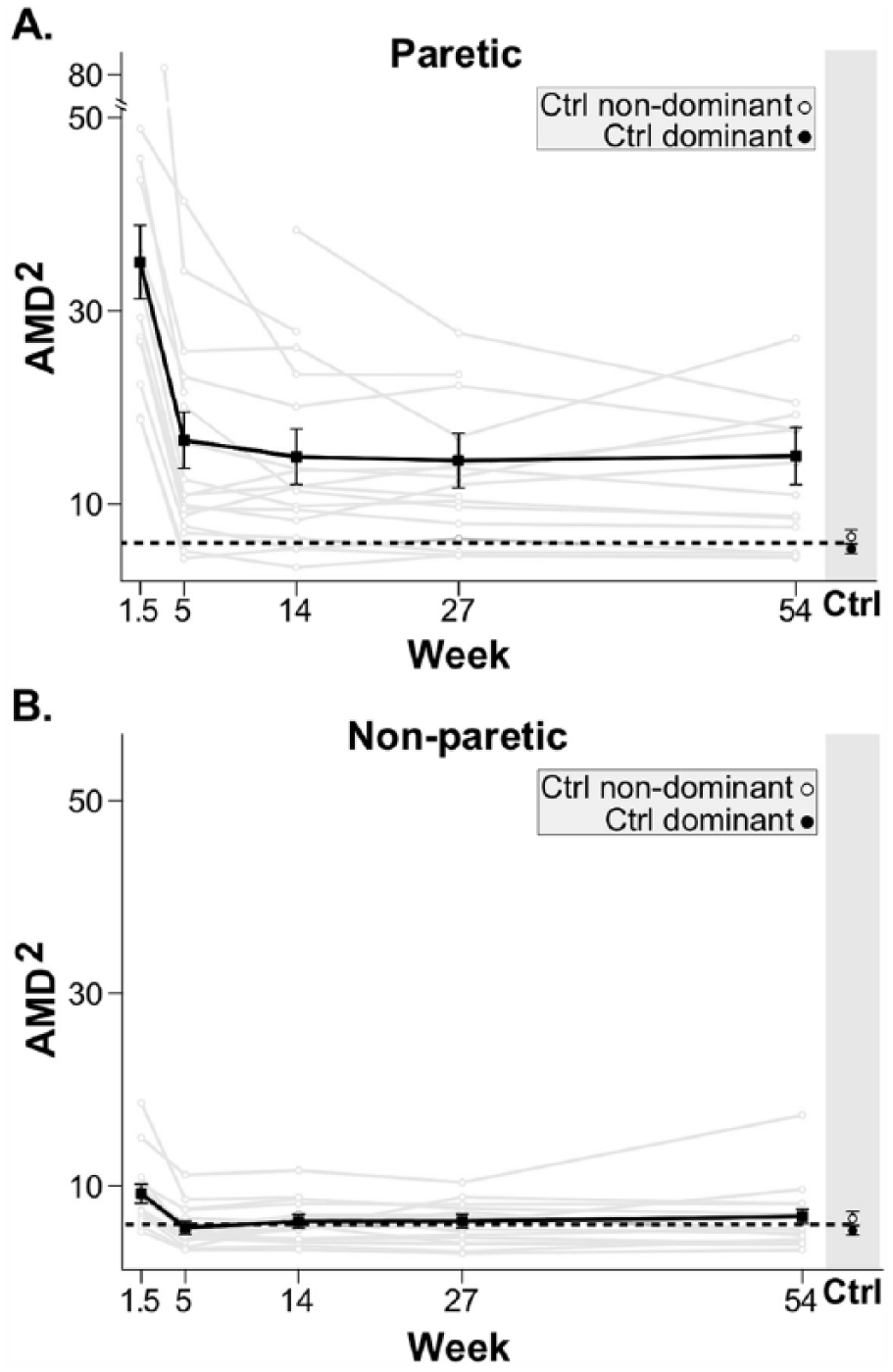
Time course of motor control (AMD2) for paretic (A) and nonparetic (B) arms for individual stroke participants (gray) and mean (black) with standard error bars. The dotted lines indicate the average AMD2 of both dominant and nondominant arms in healthy controls. For both arms, there was clear improvement in motor control between week 1.5 and 5. No further improvement in motor control was seen beyond week 5.
Comparisons between contiguous visits in the late recovery period showed no significant changes in AMD2 in either arm. Paretic arm deltas were as follows: week 5 to 14 = −1.72 (P = .463), week 14 to 27 = −0.37 (P = .87), and week 27 to 54 = 0.44 (P = .85). Nonparetic deltas were as follows: week 5 to 14 = 0.6 (P = .28), week 14 to 27 = 0.02 (P = .97), and week 27 to 54 = 0.49 (P = .46).
Finally, we conducted a secondary analysis motivated by the hypothesis that a subset of stroke participants, specifically those with severe motor control impairment at week 5, may continue to improve to week 14 or beyond. For this purpose, participants were split into “high” and “low” AMD2 using their week 5 value in the paretic arm; the median was used as the threshold to define the two groups. Within each group, we then repeated the primary comparisons (Figure 3). For the “low” group, we found a significant decrease in AMD2 during early recovery (delta = −7.66, P < .001) and a smaller nonsignificant increase during late recovery (delta = 1.35, P = .23). For the “high” group, the early recovery decrease in AMD2 was large and significant (delta = −31.59, P < .001), while the decrease during late recovery was smaller and nonsignificant (delta = −6.89, P = .08).
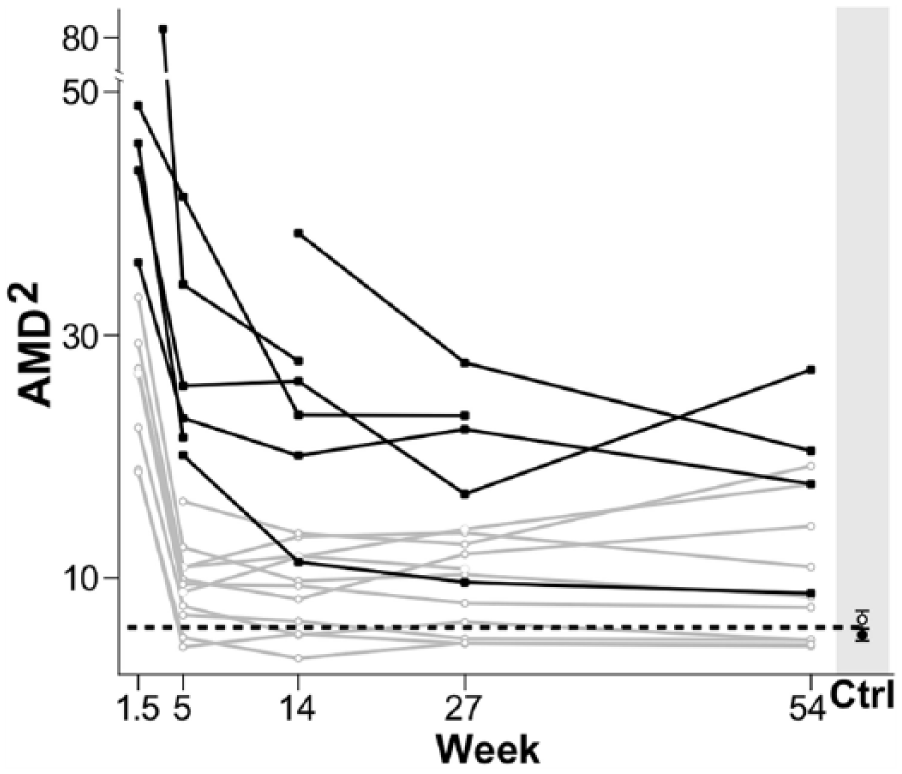
Severity of impairment and time course of motor control (AMD2) for the paretic arm of all stroke participants. Participants were divided in 2 groups based on their motor control impairment severity (AMD2 at 5 weeks): in black, participants above the AMD2 median value (representing the more severely impaired), and in gray, participants below the AMD2 median value (representing the less impaired.) The dotted line indicates the average AMD2 for both dominant and nondominant arms in healthy controls. Although not significant, we observed some recovery of motor control beyond week 5 for participants in the more severely impaired group.
In conclusion, motor control improved in the paretic and nonparetic arms during early recovery (up to week 5 poststroke), with no significant further improvement during late recovery (beyond week 5). For the paretic arm, an early plateau in recovery of motor control was observed regardless of initial impairment severity, although it should be emphasized that our sample size could have been underpowered to detect a possible interaction between initial impairment severity and time to plateau. Still, any changes beyond week 5 would only represent a small fraction of those seen before that time point.
Clinical Motor Scores Continued to Improve Beyond Week 5 Poststroke
FMA-UE, ARAT, and biceps dynamometry Z-scores were used to track arm motor impairment, activity limitation, and arm strength, respectively (Figure 4). All three measures showed significant improvements during early recovery, FMA-UE delta = 8.2, P = .010; ARAT delta = 9.7, P = .019; biceps dynamometry Z-score delta = 0.57, P = .008. During late recovery, all these clinical measures also showed significant improvements, FMA-UE delta = 10.1, P < .001; ARAT delta = 10.0, P < .001; biceps dynamometry Z-score delta = 0.94, P = .003. Analysis of the correlation between these clinical measures is included in Supplementary Materials (see Supplementary Figure 1).
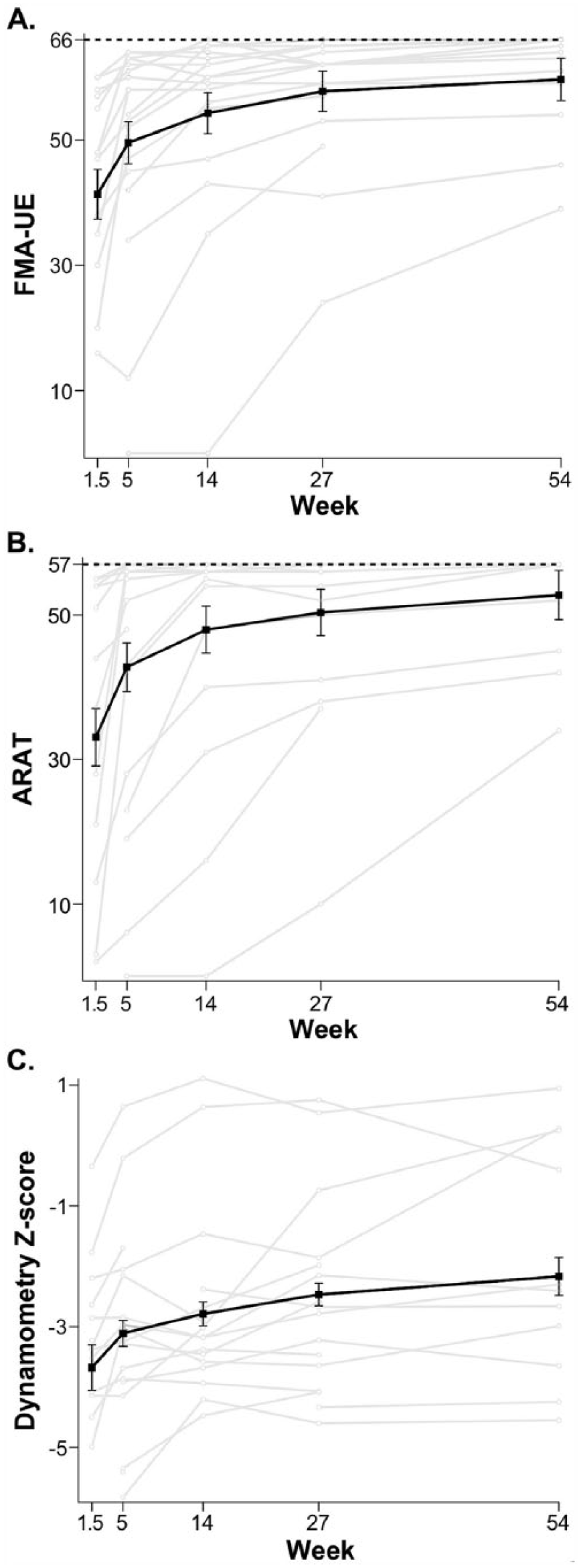
Time course of clinical scales: Fugl-Meyer Assessment of the upper extremity (FMA-UE) (A), Action Research Arm Test (ARAT) (B), and biceps dynamometry Z-score (C) for individual stroke participants (gray), and mean (black) with standard error bars. The dotted lines in A and B indicate the maximum possible scores for both measures (FMA-UE = 66, ARAT = 57). For all clinical measures, improvement was seen beyond week 5.
Figures 2 and 4 suggest distinct time courses for motor recovery depending on the measure used. To emphasize these differences in the recovery curves, the normalized values of FMA-UE, ARAT, biceps dynamometry Z-scores, and AMD2 are plotted on a single axis (Figure 5). Normalization was accomplished by subtracting the mean value for each measure at week 1.5 and dividing by the difference in means at week 5 and at week 1.5. This normalization results in an average of 0 at week 1.5 and an average of 1 at week 5; normalized values are scaled by the average delta between weeks 1.5 and 5. Qualitative assessment of Figure 5, in addition to the statistical analyses above, support the hypothesis that recovery of motor control, measured using AMD2, plateaued much earlier than improvements in FMA-UE, ARAT, and biceps dynamometry Z-score. Repeating all analyses using log and square root transformations of clinical and kinematic measures did not qualitatively alter Figure 5 or its interpretation.
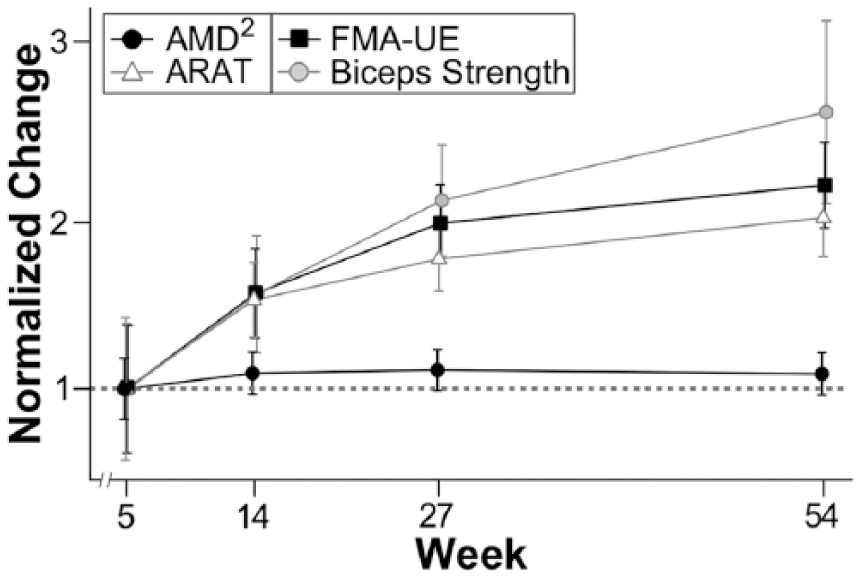
Normalized time course for all outcome variables: AMD2, Action Research Arm Test (ARAT), Fugl-Meyer Assessment of the upper extremity (FMA-UE), and biceps dynamometry (strength) Z-score. The dotted line indicates the normalized value of the recovery achieved between the first and the second visit for each measure, which is 1. AMD2 plateaued at week 5, while all clinical measures continued to improve through the first year after stroke.
Figure 5 emphasizes that for each clinical measure the delta from week 5 to week 54 is roughly 100% of the delta from week 1.5 to week 5. This is not the case for the motor control measure (AMD2) in the paretic arm, where the delta from week 5 to week 54 is roughly 10% of the delta from week 1.5 to week 5. We conducted an analysis to determine the power to detect a delta between weeks 5 and 54 in AMD2 analogous to the one found in the clinical measures, the results indicated >99% power to detect such a delta. Qualitative comparison of the normalized time courses of the outcome measures also suggest that FMA-UE, ARAT, and biceps dynamometry Z-score improved as much during late recovery (between weeks 5 and 54) as they did during early recovery (between weeks 1.5 and 5), whereas AMD2 did not improve beyond week 5.
Discussion
Here we sought to determine the time course of recovery of motor control after stroke using a 2D reaching task that largely isolates motor control from the contaminating effects of weakness and compensation. The global kinematic measure of motor control was derived from a comparison between stroke participants’ reaching trajectories and trajectories from the dominant arm of healthy controls. The main finding was that improvement in motor control for the paretic arm was almost all over by five weeks, whereas FMA-UE and ARAT scores continued to show robust improvements up to 14 weeks poststroke. Furthermore, these two clinical measures, along with arm strength, continued to show improvements through the first year post-stroke. The results presented here not only suggest that there is a limited time-period for recovery of motor control, but also that this recovery process is distinct from what is captured by clinical measures of motor impairment, activity limitation, and strength.
The critical question raised by our results is why, at the group level, did recovery of motor control plateau at five weeks, while FMA-UE and ARAT continued to show improvement beyond this point. Although this result is consistent with our a priori hypotheses about the potential for dissociation between control, strength, and compensation, it is important to first address the concern that perhaps the kinematic measure, being new and less validated, failed to detect the improvements seen with the other three clinical measures. There are several reasons that make this unlikely. In a recent study in more than 200 stroke participants, a composite measure of arm kinematics was obtained using a robot. This kinematic measure was found to be more sensitive than FMA-UE in detecting motor recovery over the first three months post-stroke. 36 Furthermore, we have recently shown that our global kinematic measure can detect changes after robotic therapy in chronic stroke that were undetectable with either FMA-UE or ARAT. 24 Thus continuous 2D kinematic data are in fact more sensitive to small differences in motor behavior than standard clinical measures.24,34 Moreover, it is also highly unlikely that the AMD2 improved in parallel with the clinical measures but went undetected. Analyses indicate >99% power to detect a change in AMD2 of the same percent magnitude as was seen for the clinical measures beyond week 5.
We speculate that the dissociation between the time courses of recovery of motor control and the clinical measures is mainly due to the improvements in strength beyond week 5. This is based on the observation that FMA-UE, ARAT, and biceps dynamometry followed similar time courses of recovery during the later recovery period (between weeks 5 and 54). Previous work in humans and nonhuman primates37-40 has shown that there are descending pathways that can explain return of strength without parallel returns in motor control. That fractionated control of the proximal upper extremity can be affected by stroke is consistent with a study that demonstrated short-latency excitation in deltoid muscles in healthy humans, which strongly suggests that monosynaptic corticomotoneuronal projections exist for proximal muscles of the contralateral arm. 41 The authors of this study commented that bilateral organization of additional medium-latency projections to these same proximal arm muscles might explain why strength is relatively spared.
Functional imaging has also shown that humans have extensive representational maps across all of M1 even when they are only moving individual fingers. 42 Loss of a piece of this motor cortical neural representation or of its output due to stroke would lead to a concomitant decrease in signal-to-noise, compromising motor control. Supporting this view, a recent study in mice showed that blocking intra-cortical synaptic transmission in motor cortex abolished complex movement trajectories elicited by long duration stimulation (500 ms), whereas map topography for initial movement direction elicited with 10-ms pulses remained intact, presumably because corticofugal output was spared. 43 This result suggests that even when considering motor cortex without invoking alternative descending pathways, a mechanism for dissociation between control and strength can be envisaged.
It is of interest that there was a small but transient worsening in motor control in the ipsilateral arm. Previous studies have reported motor abnormalities in the poststroke ipsilateral upper extremity.17,44,45 The underlying mechanism seems to be mainly driven by the disruption of bihemispheric circuitry, which has been shown to be involved during the planning and execution of complex motor tasks.46-48 The alternative explanation of a disruption in nondecussating corticospinal fibers is not supported by recent evidence. 40
Our study has some limitations. Given the strict inclusion criteria, our cohort represents a specific subset of the general stroke population. Another limitation is the sample size of 18 participants. Larger studies would be needed to definitively address the effect of initial severity and other variables of interest, such as lesion location, on the time course of recovery of motor control. Our comparison between “high” (more severely impaired) versus “low” motor control impairment groups, was motivated in part by a recent study by Semrau and colleagues, in which they showed that all their kinematic measures returned to normal within six weeks in mildly affected patients but some of the measures continued to show improvement beyond six weeks in severely affected patients. 49 Several points should be made about the comparison between our results and the results by Semrau and colleagues. First, they found an interaction between severity and time to maximum recovery using kinematic measures, which is consistent with previous natural history studies using clinical measures.4-6,11,50,51 Due to our limited sample size, we were neither able to confirm nor refute the presence of this interaction: the decline in AMD2 after five weeks among the “high” group was not statistically significant, but visual inspection suggests modest recovery beyond week 5 for participants in this group. Thus, it is very likely that a small but nevertheless significant fraction of recovery of motor control is prolonged beyond five weeks in patients with more severe hemiparesis. Second, our global measure represents a mathematical distance from normal trajectories, which makes it more robust to compensatory contamination than any individual kinematic measure. That is to say, we constrained recovery of motor control to mean similarity to normal movements, whereas it is conceivable that single variables like initial directional error and speed could reflect recovery via compensation and strength. Third, the main point of our study was not to determine the absolute time at which recovery of motor control occurs but to contrast its time course with motor recovery measured with standard clinical scales.
Another potential concern is that any continued improvements of motor control in the hand and wrist, which were not directly measured, could contribute to the prolonged time course of recovery seen with clinical scales. We think this is unlikely, however, since this would explain neither the prolonged improvement of biceps strength nor the fact that after removing the hand and wrist components from the FMA-UE, its time course remained unchanged (see Supplementary Figure 2).
In conclusion, motor control shows maximal improvement in the first five weeks poststroke, whereas clinical measures of motor impairment, activity limitation, and strength continued to improve beyond this point. We postulate that improvements in strength may be the underlying basis for the continued improvement in clinical measures, based both on their similar time courses of recovery and the correlations between them. In a recent study that measured kinematics in a 3D reaching task, which necessarily requires anti-gravity strength, trajectory smoothness improved substantially beyond five weeks up to eight weeks poststroke. 21 This result is consistent with the idea that strength improvements may give the appearance of a more extended time course of recovery for motor control. The distinct time course for motor control recovery suggests that more precise kinematic measures could better isolate the underlying process of spontaneous biological recovery. Strength training and learning of compensatory strategies can happen at any time, whereas spontaneous biological recovery only occurs early after stroke. Tracking motor control could enable us to identify and exploit the limited time window for spontaneous biological recovery in stroke survivors, as it may also be a period of heightened behavioral responsiveness to training.52-54,55
Our results highlight the urgency of implementing novel interventions to improve arm motor control very early on during the acute poststroke period. It may be asked, why should rehabilitation target motor control specifically? The answer is that motor control, and not just strength, is essential to use the upper limb in everyday complex tasks, for example typing and writing. One potential candidate treatment for improving motor control is robotics, which allows extended practice. In a recent study in chronic stroke patients, we showed that robotic arm therapy improved motor control on the same 2D reaching task as was used in the current study. 24 The changes were small but robust. It is possible that administering high-dose robotic therapy in the first month post-stroke, coinciding with the period of spontaneous biological recovery, might lead to larger improvements in motor control than those seen in chronic stroke.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grant from James S. McDonnell Foundation, grant no. 220020220. Also, support was provided from NIH R01HL123407, R01NS097423, and an unrestricted gift from John K. Castle
Supplementary material is available for this article online on the Neurorehabilitation & Neural Repair website.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
