Abstract
Keywords
Introduction
Considering that recovery of upper-arm function after stroke is challenging, one promising technique in stroke rehabilitation is bimanual arm training (BAT), 1 using interlimb coordination to facilitate motor recovery. 2 When performing a bimanual task, synchronous activation of both hemispheres with equal balance of mutual inhibitory control is observed that does not occur during unimanual movements in healthy populations. 3 After stroke, the unilateral lesion leads to more or less important destruction of ipsilateral motor areas, worsened by a decoupling of interhemispheric inhibitions, inducing a loss of motor function of the paretic hand. 4 Bimanual therapy, by rebalancing the shift of activity toward the affected hemisphere, is assumed to increase recovery. This rehabilitation approach relies on both behavioral and neurophysiological concepts, considering that, in a bimanual movement, both upper limbs influence and are dependent on each other to perform the required task. This process entails that the nonparetic limb involves the paretic limb in the task execution and improves its output 1 and, conversely, that the paretic limb compels the nonparetic limb to match its own capabilities.5,6 Moreover, usefulness of BAT seems obvious, considering the large involvement of bilateral tasks in activities of daily living.7,8 The development of a detailed timely diagnosis is required to use BAT to its full potential and let it contribute to rebalancing interhemispheric inhibition. 9
A recent meta-analysis provided strong evidence supporting BAT, 1 especially when combined with rhythmic training or functional electric stimulation. In contrast, Pollock and colleagues 10 concluded that there was insufficient good-quality evidence that BAT would be more effective than usual care or other upper-limb interventions. Also, bimanual therapeutic benefit has not been demonstrated in all stroke patients, inducing some controversies. 9 Whitall et al 11 also concluded that BAT protocols were not superior to standard unimanual rehabilitation programs. However, it seems premature to draw firm conclusions because many studies do not account for various phases of recovery (acute, subacute, and chronic) or localization of the lesion. Therefore, current results cannot be considered as clinical recommendations. Especially given the heterogeneous nature of patients with hemiparesis, the question remains if one single rehabilitative approach will ever emerge as the best. Complementary works have to be prioritized to gain more insight into recovery after stroke.
Developing specific rehabilitation protocols and optimally adjusting them to the right patient at the right moment 12 is the next step to consider. From this point of view, information about natural recovery of bimanual coordination in the early weeks after stroke is still lacking. Detailed kinematic analysis of reach-to-grasp movements is a relevant approach to provide such information. 13 Kinematic assessments are strongly suspected to provide more accurate and more sensitive measures than clinical outcomes.14,15 Clinical tests only provide a global score and do not clarify whether fluctuations depend on motor recovery or compensation and how the movement has been constructed. Kinematic assessments have shown that in stroke patients, movements are characterized by a decrease in velocity, an increase in the number of submovements, and abnormal muscular activation patterns. 15 Therefore, kinematics may be used to assess movement characteristics in a more precise and informative way.16,17
We investigated how the relationship between the nonparetic limb and the paretic limb evolves over the first 3 months of standard rehabilitation treatment. We focused on exploring whether motion kinematics could help distinguish the recovery factors of bimanual coordination. We made the assumption that (1) the reaching properties of both limbs would be deteriorated in the bimanual condition, (2) the kinematics of the 2 limbs would improve over sessions (in both unimanual and bimanual conditions), and (3) the between-hands time delays among bimanual tasks would shorten over sessions, to reach synchronization.
Methods
Experimental Design
In all, 8 kinematic assessments of reach-to-grasp movements were realized once a week for 6 weeks (week 0 to week 6) and at a follow-up assessment 3 months after inclusion (week 12) in a group of stroke patients. Assessments took place during regularly scheduled routine sessions (eg, occupational therapy, physical therapy, and speech therapy). Rehabilitation consisted of standard occupational therapy and physiotherapy, including upper-extremity training following neurodevelopmental protocols, fine-motor-tasks practice, and training of compensatory strategies for daily activities. No specific BAT was executed. All participants and rehabilitation therapists were blinded to the study hypotheses.
Participants Involved
A total of 12 hemiparetic patients (9 men, age 65.6 ± 9.7 years) were included at around 3 weeks poststroke (20.6 ± 7.1 days). Participants’ characteristics are summarized in Table 1. The study was executed at the department of Physical and Rehabilitation Medicine of the CHU Nîmes in Le Grau du Roi, France. Participants were included within 30 days after a primary unique supratentorial ischemic or hemorrhagic stroke from May 2009 to April 2010. Inclusion criteria were as follows: (1) clinical and radiological diagnosis of first unilateral stroke, (2) no serious cognitive deficits (Mini-Mental State Examination score > 25 18 ), (3) no sensitive aphasia, (4) no right-hemispheric syndrome (Bergego Scale < 15/30 19 ). All participants signed informed consent, approved by the institutional review board of the Hospital of Nîmes. The South Mediterranean III Ethical Committee approved the study protocol.
General Characteristics of Participants a
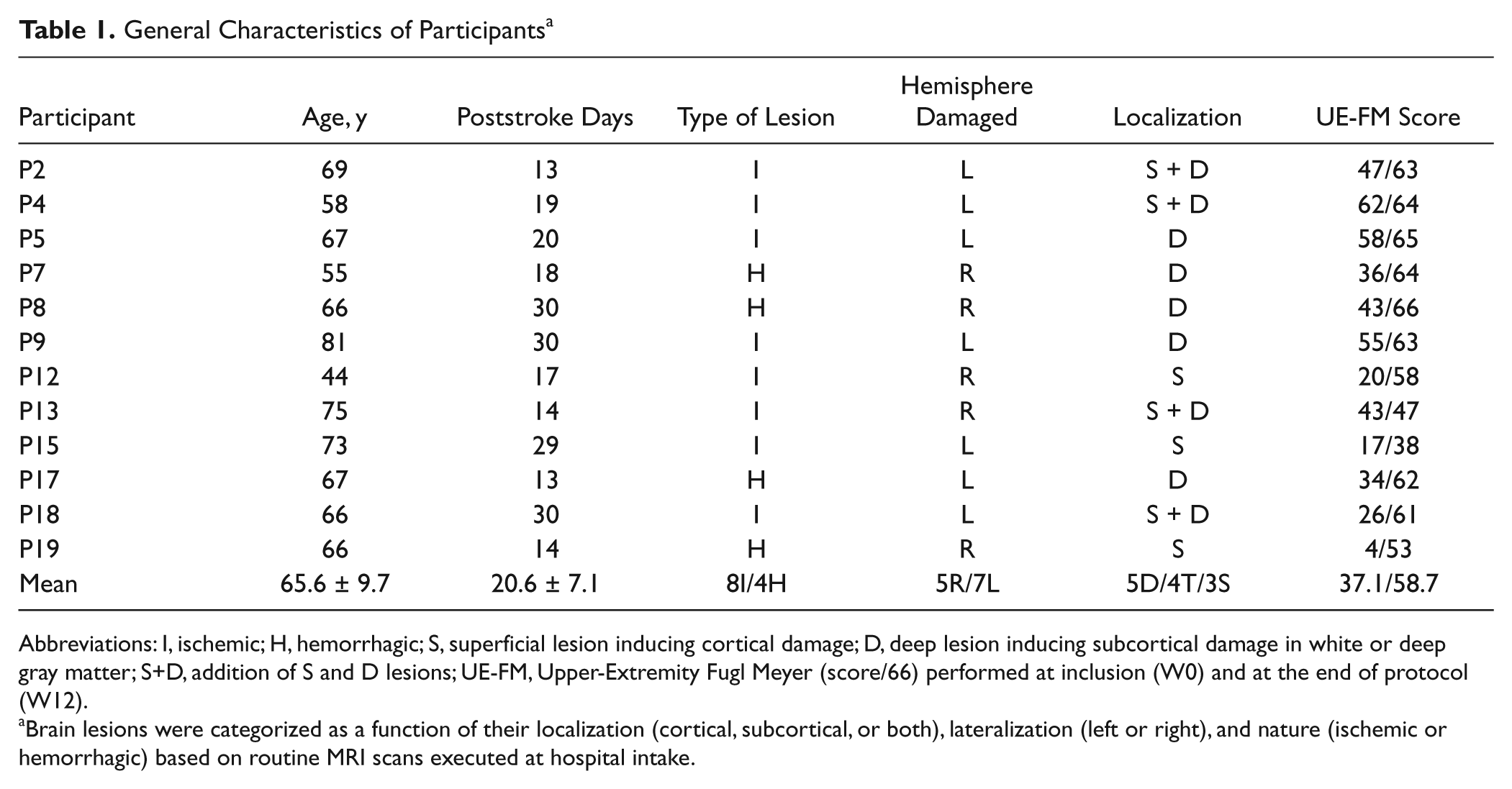
Abbreviations: I, ischemic; H, hemorrhagic; S, superficial lesion inducing cortical damage; D, deep lesion inducing subcortical damage in white or deep gray matter; S+D, addition of S and D lesions; UE-FM, Upper-Extremity Fugl Meyer (score/66) performed at inclusion (W0) and at the end of protocol (W12).
Brain lesions were categorized as a function of their localization (cortical, subcortical, or both), lateralization (left or right), and nature (ischemic or hemorrhagic) based on routine MRI scans executed at hospital intake.
Task and Procedure
Participants were asked to grasp a polystyrene ball (5 cm in diameter) lying on the table 25 cm away from the starting point of the hand. This starting point was standardized for all participants. The reach-to-grasp task consisted of 3 different conditions, with fixed order of execution: unimanual with the nonparetic limb (UN), unimanual with the paretic limb (UP), and bimanual movement (BN/BP). Each condition consisted of 5 trials. After 2 practice trials (one with each hand), acquisition was started. Participants were asked to grasp the ball at a self-selected comfortable speed and to move it to the target point materialized by a red dot located along the participant’s midsagittal plane, 20 cm away from the initial location of the ball. An illustration of the experimental task is provided in Figure 1. During bimanual reaching, participants were asked to simultaneously initiate the movement of both upper limbs and to hold the ball with both hands. A belt prevented compensatory trunk movements. Kinematic assessments lasted 15 minutes per session.
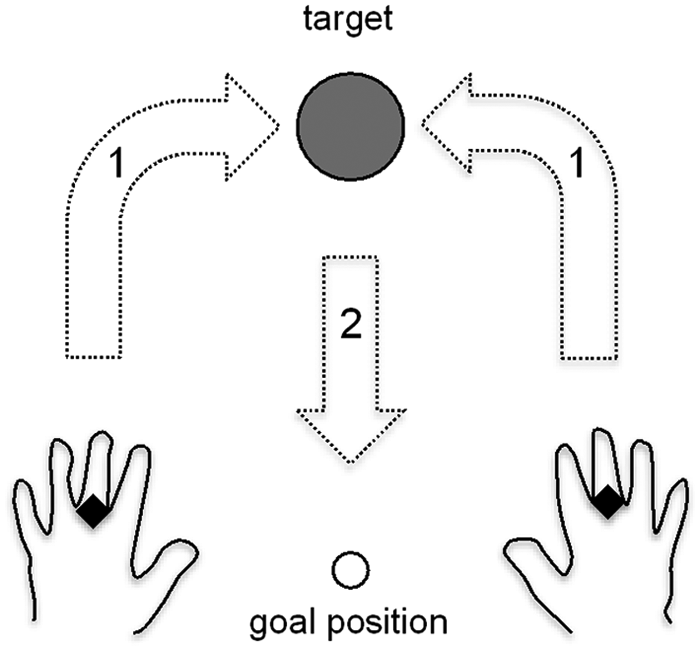
Experimental setup: This upper view shows the location of the starting points of the hands and of the object to grasp. The dark squares represent the markers, one on each hand. The task was to reach the ball (arrow 1), grasp it, and bring it to the goal position (arrow 2). The task was performed with the paretic hand (UP), the nonparetic hand (UN) and bimanually (BN/BP). Only the reaching part of the movement (arrow 1) was analyzed.
The participant was seated facing the table plan; the height of the table could be adjusted, so that the participant’s elbow could be placed above the table top at rest. Before each trial, the participant placed her/his hands in pronation at their starting point. Then, the experimenter placed the ball, followed by a verbal warning to prepare the participant for the “go” signal a few seconds later.
Outcome Measures
Kinematic information was recorded with a 3D motion recording system (Polhemus Fastrak, Colchester, Vermont). The system used a magnetic field and 2 electromagnetic sensors recording 3D positions (
We analyzed the reaching part of the whole reach-to-grasp movements (Figure 1). First, we addressed the kinematics of the movements of each hand individually. Second, we analyzed the synchronization between the 2 hands in the bimanual condition.
We selected kinematics to describe the movements in accordance with a previous study. 13 For each hand, we focused on movement timing and movement fluency, which seem to be the most sensitive variables to quantify upper-limb motor performance.20,21 The following measures were retained: the movement time (MT), the number of velocity peaks (NVPs), the maximal reaching velocity (Vmax), and the directness of the trajectory (DT) calculated as the trajectory/distance ratio. 21
For the bimanual reaching condition, the previous measures were complemented with an analysis of the synchronization of the 2 hands. We measured the between-hands delay at movement beginning (ΔBEG), at movement end (ΔEND), and at the highest velocity peak (ΔPeakVel). Illustration of how these variables are computed is given in Figure 2.
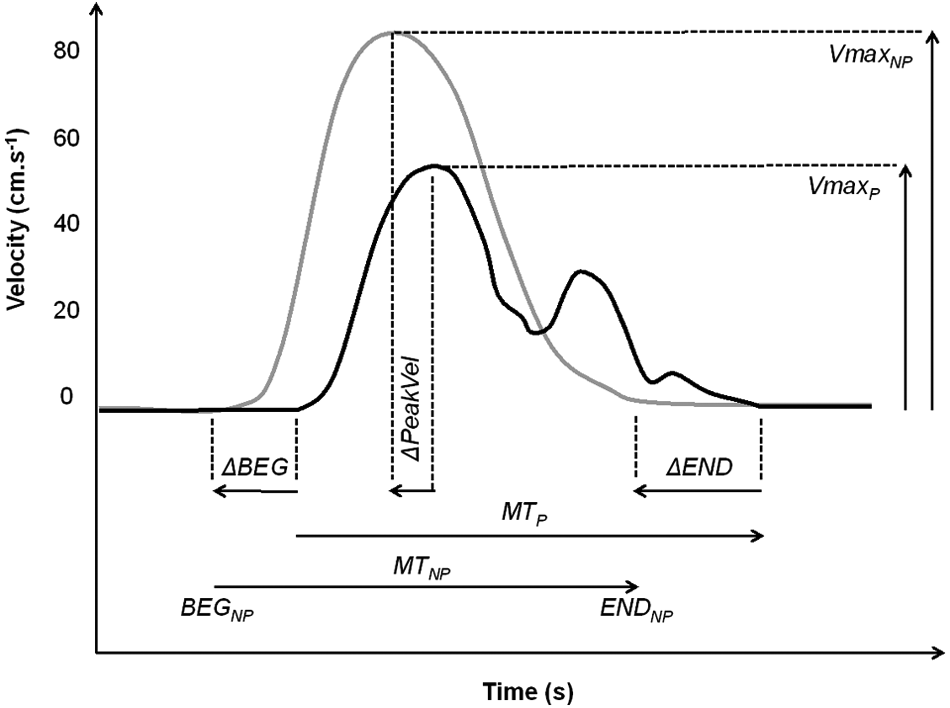
Illustration of the kinematic variables: The gray curve represents the nonparetic limb, and the black curve represents the paretic limb. The beginning (BEG) and the end (END) of each pointing movement were determined using a velocity threshold of 0.025 m/s and visually checked. Movement time (MT) is computed for each hand from the beginning to the end of the movement. Maximal velocity (Vmax) is the maximal amplitude of the velocity peak. To assess bimanual synchronization, we measured between-hand delays. ΔBEG and ΔEND represent the between-hand delays at the beginning and at the end of the movement, respectively. ΔPeakVel indicates the between-hand delay recorded at velocity peak.
Statistical Analysis
For each measure, the mean and standard deviation of the 5 trials per condition were calculated. For various reasons, all participants but 2 randomly missed at least 1 evaluation session, rendering a total of 19.2% of missing data in the repeated measures. The missing data were corrected using linear interpolation between adjacent sessions, participant by participant. 22
The significance of the effects of the session (W0, W1, W2, W3, W4, W5, W6, and W12), of the reaching mode (unimanual vs bimanual), and of the reaching limb (nonparetic vs paretic) on the outcome measures were assessed using an analysis of variance with repeated measures (rANOVA).
All
Results
Unimanual Versus Bimanual Conditions
Below we focus on the comparison of the unimanual versus bimanual conditions by averaging over the reaching limb and over sessions. The 3-way rANOVA showed a significantly lower performance in the bimanual condition, both for the paretic and the nonparetic hand. The higher NVP (
This decreased performance in the bimanual condition was also partly visible in the intertrial variability: the NVP (
Evolution of Kinematic Variables Over Recovery
In this section, we focus on the session effect and detail how the session effect depended on reaching mode and reaching limb. The 3-way rANOVA showed a principal effect of session on NPV (
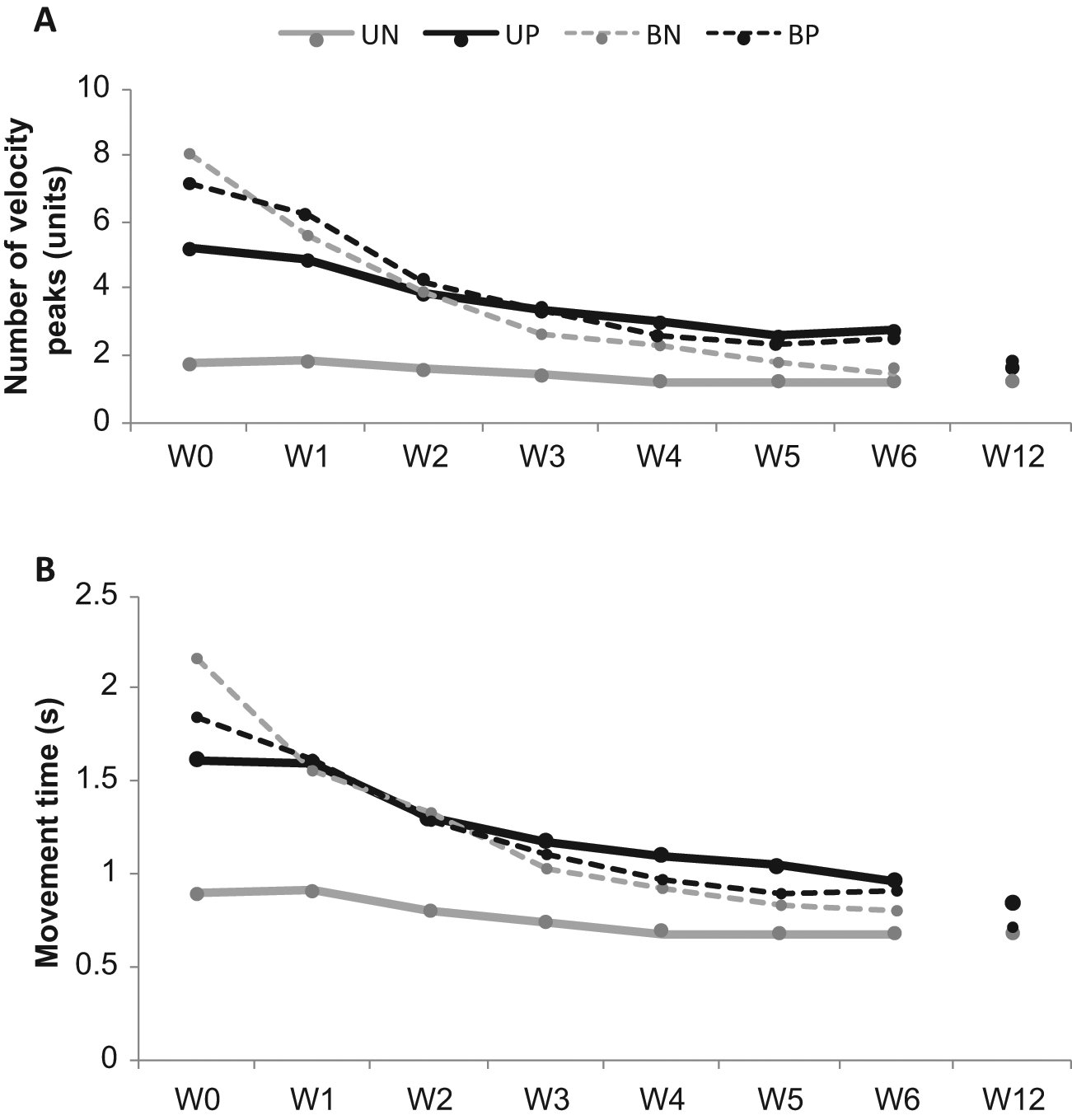
Mean unimanual and bimanual performances as function of rehabilitation duration: The nonparetic limb is represented by gray curves and the paretic limb by black curves. Unimanual conditions are illustrated by thick solid lines and bimanual conditions by thin dotted lines. The number of velocity peaks (panel A) and the movement time (panel B) returned to normal values after 3 weeks of standard rehabilitation for the paretic and nonparetic hand as well. Two different phases were observed: a decrease phase during the first 3 weeks of standard rehabilitation, followed by a plateau phase.
We also found a decrease in the intertrial standard deviation over sessions for NPV (
Post hoc analysis provided insight into the distinct evolution of each separate variable over recovery. Regarding NPV and MT, a plateau was reached at W3, which significantly differed from the values at W0 and W1 (Figure 3). This indicated that during bimanual reaching, the paretic and nonparetic limbs operated asymmetrically during the first weeks of rehabilitation. After 3 weeks of rehabilitation treatment, however, the kinematics of the 2 hands became more similar.
Synchronization During Bimanual Reaching
Finally, we detailed the time order of the movement of the paretic hand relative to the nonparetic one. We analyzed the evolution of synchronization between hands over sessions using a 1-way rANOVA on the between-hands delays at movement onset (ΔBEG), at velocity peak (ΔPeakVel), and at movement end (ΔEND). Synchronization did not significantly change over sessions (ΔBEG:
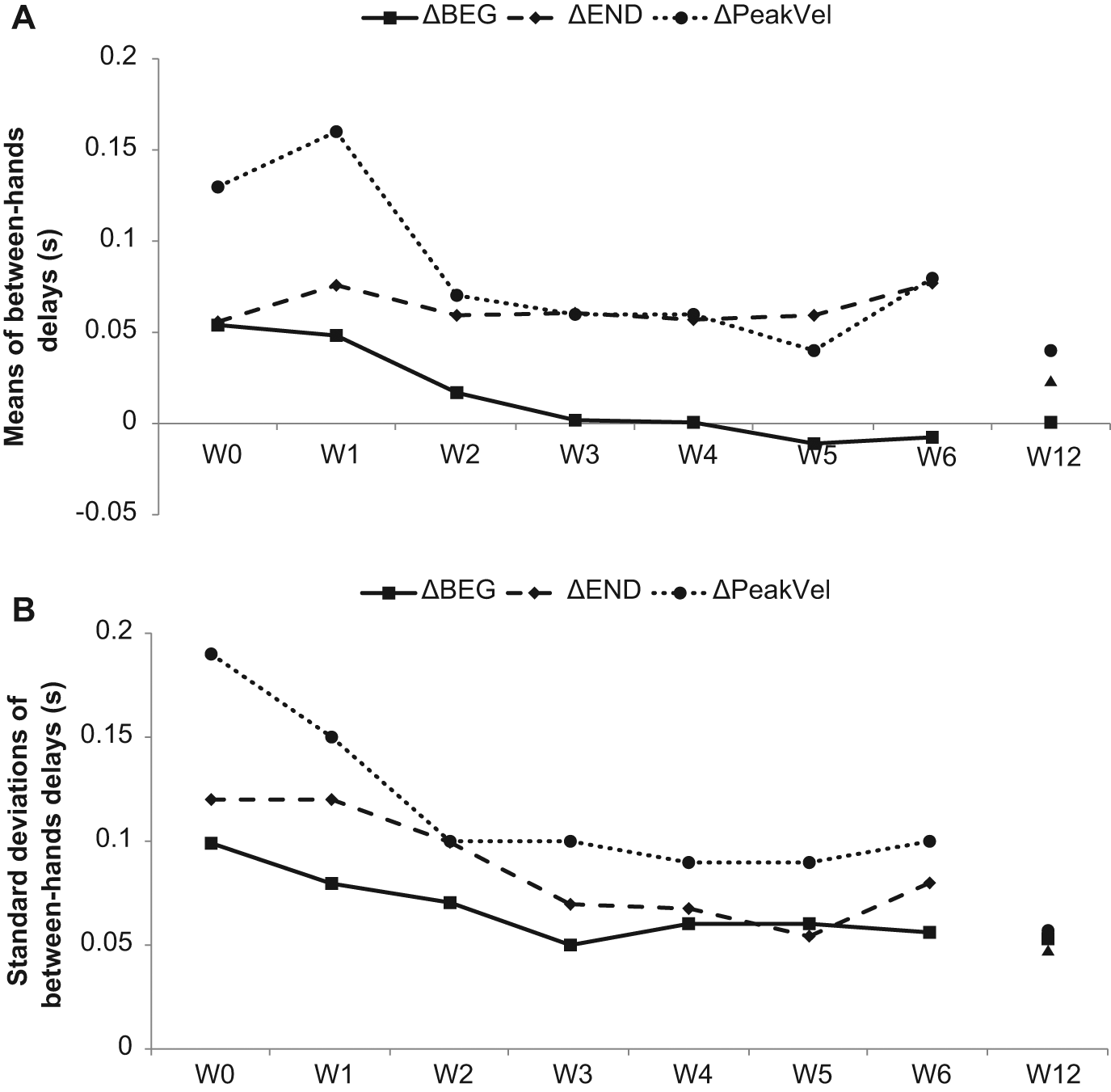
Mean values (panel A) and within-participant intertrial standard deviation of between-hands delays (panel B). The between-hands delay is computed at movement onset (ΔBEG), at velocity peak (ΔPeakVel), and at the end of the movement (ΔEND). Although the mean between-hands delays did not evolve significantly over sessions (panel A), participants became less variable with recovery (panel B): the variability of the synchronization between the hands reached a plateau after 3 weeks of treatment.
Discussion
The kinematic features of unimanual and bimanual reaching were described over the first months of a standard rehabilitation program in a group of 12 patients with moderate poststroke impairment. 2 It was found that (1) the kinematics of both limbs were less smooth in bimanual reaching compared with unimanual reaching, (2) recovery patterns of both limbs in the bimanual condition followed a similar profile with a plateau phase after 3 weeks of rehabilitation, and (3) between-hands synchronization became less variable in time after 3 weeks of rehabilitation, whereby the mean between-hands delays remained the same.
Because bimanual movements require the preservation of the cooperation of the 2 hands, the nonparetic hand had to adapt to the paretic hand’s behavior. In such an asymmetric bimanual cooperation, the paretic limb compels the nonparetic limb, hence superposing the deteriorated movement pattern on the unaffected arm to match the movement patterns of the affected arm. 24 Our results support previous studies, 25 which also observed a deterioration of the nonparetic limb performances in the bimanual condition.
Our experiment offers more fine-grained evaluation of motor control over time because it relies on kinematics.13,26 We found a decrease in movement smoothness of the paretic limb in the bimanual condition. This reveals that patients needed more submovement units in the bimanual case, which suggests a higher workload when operating the 2 hands together. Healthy people usually plan and perform reaching movements with 1 single and smooth motion unit. When neural noise is increased as a result of fatigue in healthy people 27 or poststroke, 28 prediction of the effective motor output from a planned command is more difficult. As a consequence, reaching is organized with iterative corrections, with a higher workload that results in longer MTs. 28 In our study, the higher dependence of the paretic limb on visual feedbacks was added to the burden of operating the 2 hands synchronously in the bimanual case.29,30 This increase in workload can explain the increase in submovement number during bimanual reaching.
In the present experiment, the session factor provided the greatest effect size: all participants improved movement fluency in unimanual and bimanual reaching during the rehabilitation program. The kinematics of the nonparetic limb matched the kinematics of the paretic limb in bimanual reaching, following a similar recovery profile over time: participants improved movement kinematics for the first 3 weeks of standard rehabilitation before reaching a plateau phase around 6 weeks postinjury (Figure 3). Such a pattern of change over recovery is not surprising because greater gains are usually noticed early after stroke and then progressively level off.31,32 The observed improvements in reaching kinematics for the first 3 weeks can be the consequence of spontaneous recovery because it has been described as maximal in the first 4 weeks poststroke 33 and likely explaining 16% to 42% of the functional improvements in the first 6 to 10 weeks after stroke onset. 34 The observed gains can also be the consequence of learning-dependent plasticity: a plateau effect is also expected according to motor (re)learning principles 33 : in patients poststroke, as in healthy humans, the learning curve gets less and less steep with time and expertise, up to a point where learning becomes no longer visible. Little is known about the most optimal time window for initiation of learning-dependent plasticity. 35 Neuronal plasticity is often experience dependent, time sensitive, and strongly influenced by features of the environment or by the status of the patient, and attention and engagement can be critical modulators of plasticity. 36
The coordination of the 2 hands during bimanual movements was therefore considered to be efficient at around 3 weeks after inclusion (ie, 6 weeks after onset of stroke), potentially highlighting a key period in rehabilitation programs. Whitall 37 previously proposed that mildly affected patients would benefit the most from unilateral training, whereas moderately affected patients would respond better to bimanual rehabilitation programs. According to this assumption, rehabilitation might follow a continuum, starting from unimanual to bimanual approaches, when the recovery phase allows greater motor capabilities. Considering that the plateau reflects the onset of functional interhand coordination, it may be the best moment to start bimanual-oriented training in hemiparetic patients. Moreover, bimanual coordination may be used as a kinematic indicator of recovery. This predictive value might inform about usefulness of bimanual therapy and likely contribute to the implementation of an algorithm to find the critical time window for each patient to start bimanual-oriented rehabilitation.
Though the average between-hands delays remained unchanged across sessions, we found that the between-hand synchronization became less variable with recovery. Again, a plateau was reached after 3 weeks of standard rehabilitation (Figure 4), indicating that participants had less variable bimanual movements around this period. Punt et al 38 studied hand synchronization in healthy individuals using a pointing task, where the hands moved to targets of variable distance and direction. They demonstrated a strong coupling between the hands, reflected by an almost synchronous movement onset, time to peak velocity, and movement offset. Despite some similarities, in our subacute group, the coupling between the hands remained impaired.
To enable simultaneous arrival, the nonparetic limb was forced to adapt to the impaired hand. The between-hands delays at the end of the reaching revealed the existence of a hover phase that represents the time allowing the participant to transport 1 hand (mostly nonparetic here) and wait for the other. 29 Actually, healthy people visually focused more on targets and only a little on the hands during the whole movement. 39 Conversely, stroke patients are assumed to focus more on their paretic hand than on targets, explaining the emergence of a hover phase during bimanual movements. 40 As a consequence, the coupling asymmetries manifest as lead-lag relationships between the limbs. We and others propose that this bimanual coordination should be addressed during stroke rehabilitation. 41 Yet because bimanual synergies are more than the performance of each limb separately, we suggest that they should be investigated as impairments of the linkage between the 2 limbs 42 and trained as specific synergies.43,44
Some limitations emerged from our methodology. Our sample size (n = 12) was reduced, and the measured performances during trials showed strong intrapersonal and interpersonal differences, revealing the heterogeneity of our population. Intraparticipant variability among trials played an important part in the dispersion of the results, the high rate of variability being a well-known index of dysfunction after stroke. 45 The Fugl-Meyer score clinically illustrated the between-participant variability at inclusion, and impairments were categorized as 3 severe, 6 moderate, and 3 mild poststroke impairments 2 with good recovery (except for 1 participant; see Table 1). Though the interparticipant differences were large at inclusion, the majority of our participants had a mild-moderate level of impairment and were much improved after 6 weeks of rehabilitation. Participant variability could have been related to our inclusion of patients with hemorrhagic and ischemic strokes. 46 In the present experiment, we did not find a statistical difference in recovery between the subgroup of 4 hemorrhagic versus 8 ischemic stroke patients. Therefore, we reported all analyses on the overall group of participants, although we realize that a larger sample size might reveal different recovery profiles for the 2 etiologies.
To restrain additional biases concerning the heterogeneity of our population, we decided to include participants at admission to the rehabilitation center and to report treatment duration in weeks of rehabilitation. This choice ensured that the group of patients was homogeneous in terms of rehabilitation amount and duration but with the consequence that patients differed in terms of poststroke time delay (Table 1). Because participants were not included in the protocol at the same poststroke delay, comparison of equivalent standard rehabilitation workloads between patients was required. Obviously, this choice emphasized the effects of the rehabilitation program and tended to cancel the effects of time-dependent poststroke recovery processes.
Because of the heterogeneity of distal capabilities between patients, we partially failed to avoid bias related to standardization of the movement performed. Participants had to reach to grasp a ball, but only the reaching phase was studied. Grasping and reaching for an object involve different kinematics. 47 Different temporospatial coupling modes for the reach and the grasp components of bimanual grasping movements have been reported. 48 Furthermore, reaching components could be less damaged than grasping components after a cerebral lesion. 38 As prehension of a 5-cm ball did not constrain hand configuration, participants could catch the ball either with the volar region of the hand (from the top) or with fingers used like pliers. These various strategies in the elaboration of the movement could alter kinematics. For these reasons, generalization of our results has to be considered with caution.
Disorders following stroke are unique for each patient, and the idea of 1 rehabilitation method fitting all patients has limited plausibility. 49 In the present study, we defined a baseline recovery profile of moderate-to-mild subacute stroke patients within a standard rehabilitation program. Even if we were unable to sort out different recovery profiles, we can hypothesize that some patients would benefit more from bimanual-oriented rehabilitation. According to our results, bimanual-oriented rehabilitation may be started at the end of the second month postinjury, as soon as the plateau in kinematic features of both hands occurred. Further research will analyze recovery profiles depending on the localization of the lesion. For example, Carson et al 50 found that modulation of the excitability of the corticospinal pathways mediated the stability of bimanual coordination, likely provoking a failure of inhibitory mechanisms.
Motor recovery is modulated by lesion location, specifically by involvement of cortical or subcortical structures. 51 In our study, lesion location was used as a 2-level factor that constituted 2 subgroups (left-hemispheric damaged patients vs right-hemispheric damaged patients). We found no significant results, but the kinematics of left-hemispheric patients tended to be more deteriorated than those of right-hemispheric patients in bimanual reaching, and cortical lesions tended to have a more negative effect on bimanual reaching than subcortical lesions, for instance. Larger sample sizes are necessary to evaluate the consistency of these exploratory results, but it confirms once more that no technique is equally efficient for all patients and for all severity levels. 52 Developing complete patient profiles will be the next logical step to be able to propose optimal bimanual training to the right patient at the right moment after stroke onset.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the University Hospital of Nîmes (2009-A00872-55) by the MARGAUT Project (2010- A00596-33), and by the SKILLS Project of the European Commission (FP6 IST 035005).
