Abstract
Introduction
The majority of patients after stroke are left with deficits in upper limb function.1,2 Improvements in functional reaching can occur either by regaining the ability to make movements that were lost completely after the stroke 3 or by increasing the accuracy and/or speed of preserved movements.4,5
In the chronic phase after stroke, multiple studies have shown that training can produce task-specific improvements even many years after stroke, although the speed of recovery slows.3,6 However, there are few detailed investigations of biomechanical changes induced by training in chronic stroke patients.3-5 Some authors have argued that in the chronic phase all improvement is compensatory,4,7 in that the goal is achieved by replacing lost abilities using other joints. This results in solutions that are not optimal for the task. 8 Thus, patients’ movements may become more accurate with training but this may be achieved by increased trunk flexion during reaching.8-10 However, improvement may occur through 2 other mechanisms. Even if patients do not recover lost function, they may recover better control of their movements, resulting in movements that are less variable from trial-to-trial, and hence on average more accurate.3,11,12 Another possibility is that patients relearn to produce combinations of muscle activity lost due to stroke. Improvements in performance in this case would be detected as reduced endpoint bias and/or straighter trajectories.3,13 Additionally, an important issue in motor learning is the speed-dependency of improvements. In a previous study, 13 we found that if healthy adults practiced reaching at one speed they improved performance at that, but not at untrained speeds. After a neurological insult, individuals tend to move slowly, 14 possibly due to greater difficulties of generating activity, 15 increases in stretch-reflexes, 16 avoidance of increased interaction torques with higher velocities, 17 or to compensate for decreases in accuracy.11,18,19 However, many movements such as catching a falling object, driving a car, or stabilizing yourself while on a bus rely on the ability to generate accurate, fast bursts of muscle activity. 15 Current clinical guidelines do not emphasize the need to train patients at a variety of movement speeds, 20 and there are limited studies investigating how movement speed during training effects learning after stroke. Continual exposure to slow movements in daily behavior and rehabilitation training may prevent regaining the ability to move accurately at fast speeds, or they may even reinforce the slowness of movement through use-dependent learning.13,21
We therefore investigated whether improvements in reaching are possible when practicing an arm-reaching task for 4 days when compensatory movements are minimized. We measured changes in endpoint accuracy in terms of endpoint bias and variability when patients trained either at fast or slow movement speed and analyzed the effect of the training on the speed-accuracy trade-off function.18,22,23 We hypothesized that, as for healthy individuals, some of the movement improvements would be specific to the trained speed. More specifically, we predicted that improvements during fast reaching would be achieved only after training at the fast movement speed. 5 We further investigated how improvements in fast movements matter to clinical motor impairment measures, hypothesizing that improved ability to generate fast movements may have clinical relevance. Finally, we studied how different factors of impairment (sensory loss, spasticity, weakness) influence the ability to profit from training.
Materials and Methods
Subjects
This parallel-randomized (1:1 allocation) study was approved by the Joint Ethics Committee of University College London and the National Hospital for Neurology and Neurosurgery (NHNN). Patients were recruited from NHNN and charity stroke clubs and websites. (For clinical details, see Supplementary data, Table I). Prior to participation, informed consent was obtained from each participant according to the Declaration of Helsinki. All patients met the following inclusion criteria: (1) chronic stroke survivors (≥1 year history) with (2) persistent upper limb weakness (≤4 Medical Research Council [MRC]) of either triceps or anterior deltoid muscles. (3) Participants had to be able to perform the training task of ≥15 cm reach with the weight of the arm supported in a robotic manipulandum (Figure 1A). We excluded individuals with (1) history of previous stroke or other concomitant neurological or musculoskeletal disease, (2) cerebellar stroke, (3) proximal upper limb hypertonus ≥3 on Modified Ashworth scale (MAS), (4) severe sensory impairment (light-touch <50% accuracy on 1 g Bailey monofilament sensory testing on dorsum and palm of hand), (5) shoulder pain ≥3/10 on self-rated continuous visual analogue scale, (6) uncorrected visual impairment, (7) hemi-spatial neglect established by the Star Cancellation Task, 24 and (8) cognitive and language impairment impeding co-operation in study protocol.
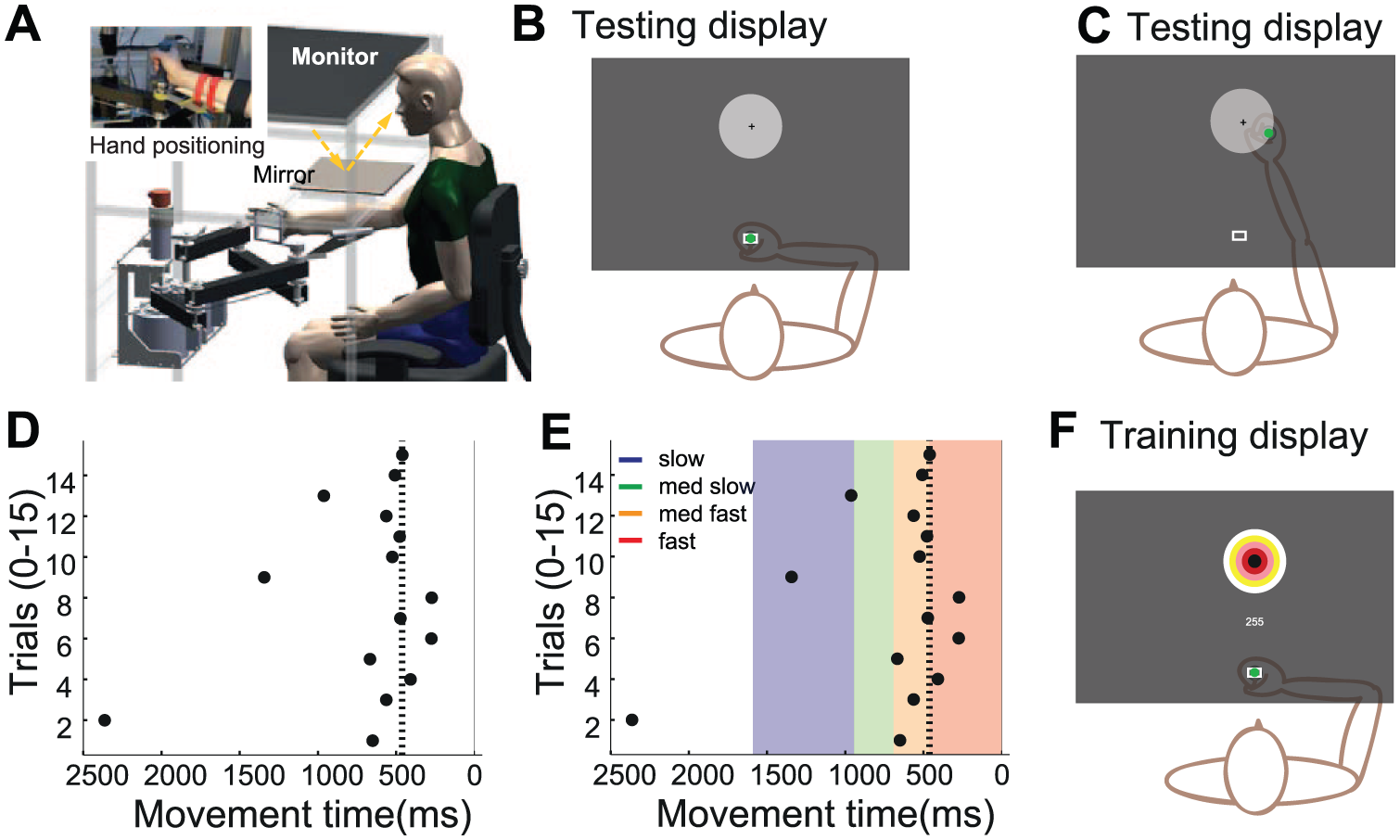
Reaching protocol. (A) Experimental set-up. (B-C) Experimental display during accuracy testing. Target (5 cm radius) with centre cross, positioned at 20 cm distance. Hand position is displayed to participant as a green dot at the start (B) and at the end (C) but not during the reaching movement. (D-E) Method of determining individual movement speed limits. (D) Example data of movement times for 15 trials when attempting fast reaching. indicating) The 80th percentile is indicated by a dotted line (E). Therefore the fast movement limit is less than 460ms (dark grey) with incremental increase of 200ms for medium fast (460-660ms med dark grey), medium slow (660-880 medium light grey) and slow (880-1600ms light grey). (F) Bullseye display of target during training days with points as feedback of endpoint accuracy.
Clinical assessments were performed before and on the last day of the testing week by a neurologist (DH) blinded to training group allocation. Testing consisted of the Fugl-Meyer upper limb subset (/66), muscle strength (MRC grading), 25 sensory impairment (1 g monofilament), and elbow flexor hypertonus (MAS). 26 MAS scores were converted to a 6-point scale (0-5) prior to nonparametric analysis and are depicted as such throughout. 27
Reaching Paradigm
Hand position was measured using a custom-built 2D manipulandum (Figure 1A), 28 with an incremental quadrature encoder at each of the 2 joints (65 500 steps/revolution). This resulted in accuracy at the handle of ~0.03 mm. Movement speed was calculated by differentiation of the position signal. All kinematic data were sampled at 200 Hz. Participants were seated with forehead support, a shoulder strap, and backrest support preventing compensatory movement in the sagittal and frontal plane while limiting shoulder girdle movement. Subjects held a handle (inset Figure 1A) or if required the hand was strapped onto the handle by a custom-made glove. 13
A forearm support eliminated gravity, and vision of the hand was occluded by a mirror displaying visual feedback (Figure 1B). Feedback comprised of a 2 cm diameter starting box, a green cursor (0.5 cm diameter) representing manipulandum position, and a circular 10 cm diameter target with a small black cross at its center, which was located 20 cm from the start box at an angle of zero degrees. A change of the target from an outline to a solid white color indicated the start of a trial. Individuals were instructed to reach and terminate movement as close as possible to the center of the target (center cross) in their own time. When movement was initiated, the green cursor disappeared and only reappeared, displaying feedback of the end position (Figure 1C) for 1 second when movement stopped. Feedback was removed to prevent corrections during the movement because with corrections the relationship between speed and accuracy is complicated, as slower movements allow for more complete corrections. Visual feedback at the endpoint (knowledge of results 29 ) is essential to prevent complete dis-calibration without knowledge of hand position, of the reaching movements, and to motivate participants to move accurately. The robot was used primarily to measure movement however; assistance was provided to move the handle back to the starting position after the completion of each trial.
Initial assessment (pre) was performed on a Thursday and the final assessment on the following Friday (posttraining). In these sessions, reaching accuracy was established at 4 different speeds 13 depending on each individual’s fastest movement ability. After task familiarization (15 repetitions with and 15 without visual feedback of hand position), participants were encouraged to reach as quickly as possible in the third block (Figure 1D). The 80th percentile or fourth shortest movement time was used to set the limit for the individual’s fast movement time (Figure 1E dotted line, ie, 460 ms). Movements during fast reaching conditions had to be terminated faster than this limit (red shaded area), which we found to be challenging but achievable in pilot testing. For the other 3 movement speeds, the lower movement time limit was incrementally increased by 200 ms resulting in this example, in limits of 460 ms to 660 ms for medium fast (yellow) reaches, 660 ms to 860 ms for medium slow (green), and slow (blue) between 860 ms and 1600 ms while allowing some redundancy at the slow movement speed to increase ease of task performance. This incremental increase allowed us to test individuals reaching accuracy at similar intervals along their speed-accuracy trade-off function. The order of testing movement accuracy at the 4 movement speeds was randomized across patients. At every speed, reaching movements were repeated until 20 successful trials or a maximum of 60 trials were performed.
Training Paradigm
Blocked, stratified randomization to the fast or slow training group was performed after completion of the initial assessment. Sequentially numbered sealed envelopes contained group allocation stratified for functional impairment (Fugl-Meyer ≤50 or ≥51). Training sessions were always performed on the consecutive Monday to Thursday between the assessment sessions. All movements during the 4 training days were performed at the individually determined fast or slow movement time limit as described in the reaching paradigm. The trainer (UH) was not blinded to group allocation as the speed of movement was visually apparent and patients required prompting to perform movements at the correct speed. Patients were instructed to perform reaching movements in the robotic manipulandum, to a bulls-eye target for 420 reaches per day (7 blocks of 60 repeats; Figure 1F). This protocol was established in pilot testing to achieve ≥400 movement repetitions in training.30,31 Movements had to be performed at the movement speed of the allocated group and were rewarded for endpoint accuracy to a maximum of 300 points (60 × 5 points) per block (Figure 1F). Five points were awarded for terminating in the bulls-eye (<1 cm error) with incremental reduction to 1 point in the outer ring (4-5 cm error). Accumulative points were displayed on the screen for each block, and a beep indicated that the trial was successful within the speed limit and in the target area receiving at least 1 point. Movements that ended outside the target area and/or did not fall within the required movement limit were awarded zero points. Visual feedback of endpoint location was provided after each trial for 1 second. Participants were encouraged to increase their points per block and were reminded of their performance on the previous block and the previous day(s). Each training session lasted between 1 and 1½ hours.
Outcome Measures
The primary outcome measure was spatial accuracy at movement end. We studied how accuracy changed due to training and how these reductions generalized to untrained speeds. As an overall measure of accuracy, we used average distance from the center of the target (cm). This error could be further subdivided into the average deviation from the target (constant error) and the standard deviation around the mean endpoint (variable error). 32 For some analyses, the error was further subdivided into parallel (ie, movement direction) and perpendicular movement error (ie, orthogonal to movement). To allow comparisons across individuals, movements of individuals with left hemiparesis were mirrored along the sagittal plane and data are presented as right arm movements for all participants.
For each trial, the maximum tangential movement speed of the hand was determined and averaged per individual for each tested target speed (maximum speed). 13 The standard deviation around the mean was taken as a measure of variability of movement speed (movement speed variability).
Data Analysis
IBM SPSS software and custom written Matlab (Mathworks, Nattick, MA) routines were used for data analysis (
Repeated-measures ANOVAs (Greenhouse-Geisser corrected) were used to analyze performance during training BLOCK(7)*DAY(4)*GROUP(2) and change (day 1 compared to day 6) after training TIME(2)*MOVEMENT SPEED(4)*GROUP(2) and assessed by post hoc Student’s
The regression slope of performance change due to training was depicted in both training groups (intercept fixed to residual RMS error of 0.93 cm; ±0.06 observed in healthy individuals; see supplementary information Figure I). Regression coefficients were compared by
Results
Thirty-six stroke survivors (57.5 years, SD ± 11.5; 10 females) successfully trained at their target speeds (n = 17 slow at average movement speed 32.2 ± 0.3 cm/s and n = 19 fast at 77.9 ± 0.45 cm/s) with no adverse events. The study participants comprised 27 individuals with an infarct and 9 hemorrhagic stroke survivors. The lesion site was cortical in 13 individuals, subcortical in 6, and 9 patients presented with a combination (see supplementary information Table 1). Lesion location was not known in the remaining 10 individuals. Intergroup comparison for lesion type, side, or site did not demonstrate any group effect in this small sample. Over 4 days (days 2-5), reaching accuracy improved (Figure 2A; effect of DAY
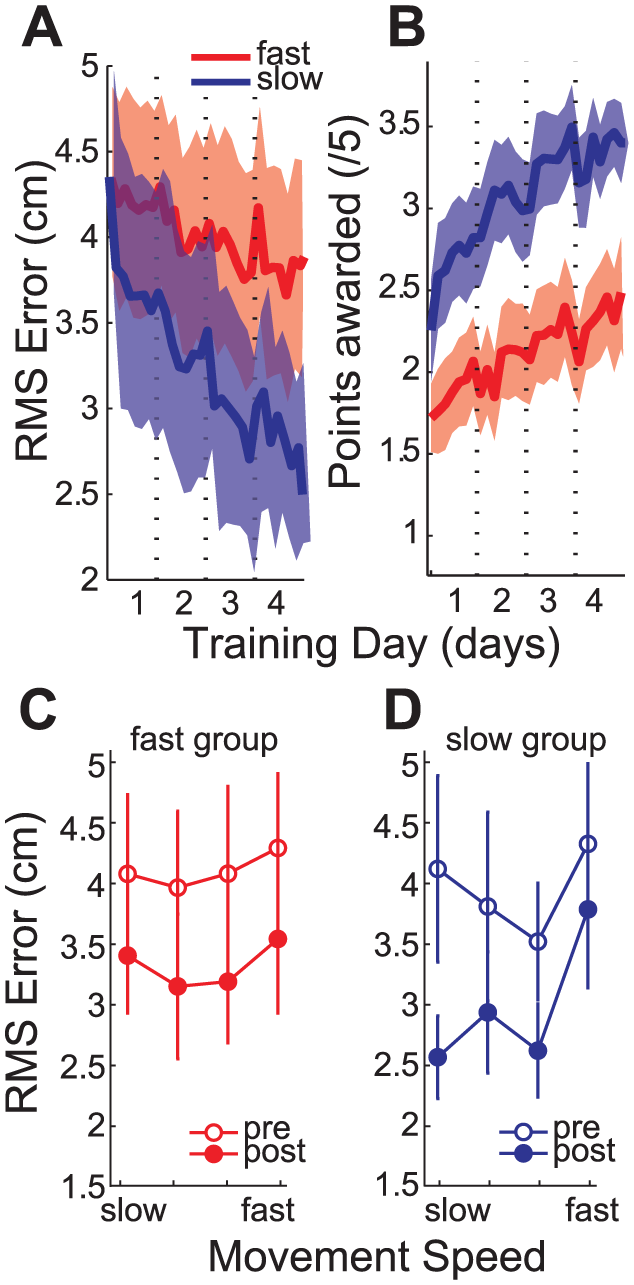
Change in amount of endpoint error. (A) The mean endpoint error (RMS ± SEM) for fast (dark grey) and slow (light grey) group reduced during the training days. (B) The mean points (± SEM) per training block reduced for both training groups over the training days. (C) RMS (± SEM) error at the four individually set target speeds before (unfilled) and after(filled) training for the fast and (D) slow training group.
Accuracy Improvements at Trained and Nontrained Movement Speeds
Before training, stroke survivors had poor endpoint accuracy at all 4 tested movement speeds without a difference in baseline performance for participants randomized to slow and fast training (Figure 2C and D). In a retention test, a day after the last training session (day 6), both groups improved their endpoint accuracy in comparison to performance on day 1 but the pattern of improvement differed for the 2 training groups (GROUP(2) × MOVEMENT SPEED(2) interaction,
We next established to which extent this improvement was achieved by a reduction in endpoint bias and/or a reduction in endpoint variability by investigating the combined data of the 2 training groups.
Before training individuals demonstrated a bias to undershoot and terminate in the opposite workspace as indicated by the groups mean endpoint location and standard error of the mean (Figure 3A-D), generally indicative of an elbow and shoulder flexion bias (supplementary information Figure IIIA). There was no interaction or significant change in the bias (repeated-measures ANOVA: no effect of TIME) for both parallel (
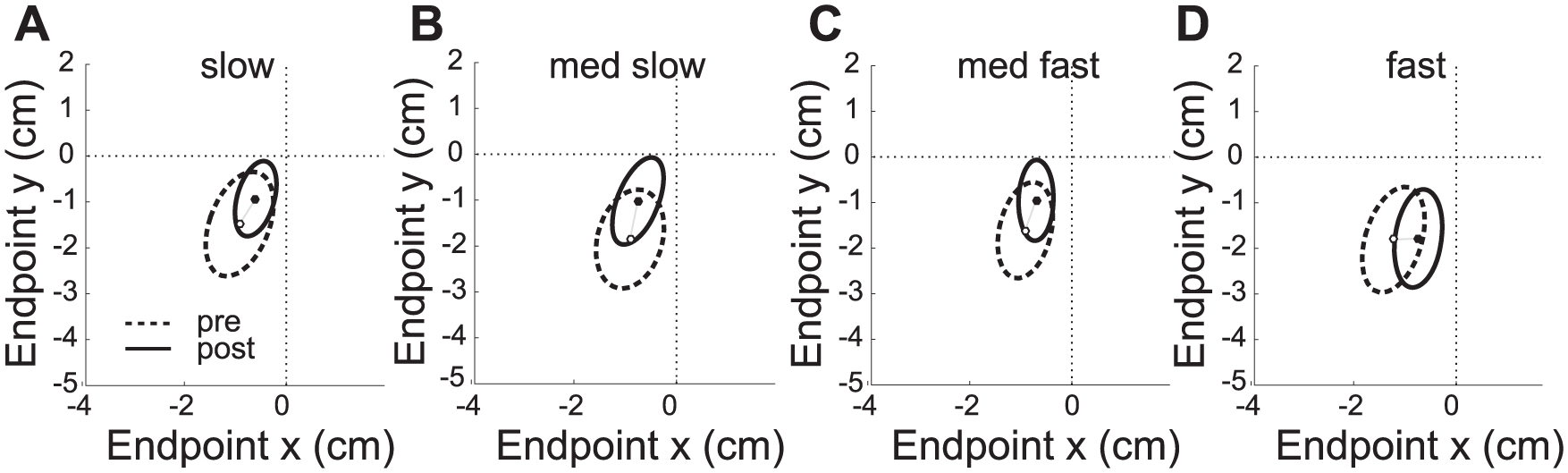
Endpoint variability and bias. Mean endpoint bias and variability (SD) in relationship to the target center (0, 0) at the 4 movement times (A slow, B medium slow, C medium fast, D fast) before (dashed) and after training (solid). The change in endpoint bias was not significant; however, the reduction in endpoint variability was significant at all movement speed. Participants tended to undershoot end movement in the contralateral workspace (flexor bias). Data of individuals with left hemiplegia are mirrored along the sagittal plane and data are presented as right arm movements for all participants.
Movement Speed Variability
Although patients were required to move at specific speeds (supplementary information Figure IV), their actual speed varied slightly from trial-to-trial (Figure 4). The variability of the peak speed was the same in both groups before training (no interaction
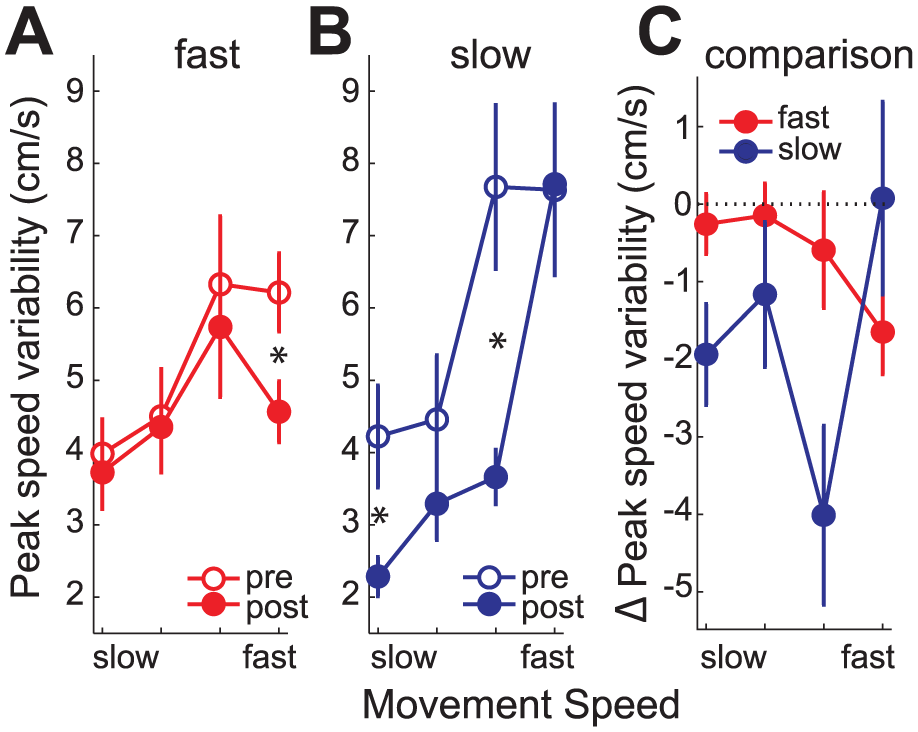
Change in movement speed variability. Mean peak speed variability (± SEM) for the slow, medium slow, medium fast and fast movement speed before (unfilled) and after (filled) training for the (A) fast (dark grey) and (B) slow (light grey) training group. (C) Mean change in movement speed variability at the 4 tested movement speed for the fast (dark grey) and slow (light grey) training group. A significant change in maximum speed variability was detected at the training speed for both groups as well as at the medium fast speed for the slow training group.
The Influence of Baseline Impairment and Clinical Measures on Behavioral Change
The RMS error of individuals with good baseline performance improved less than those with poor performance (Figure 5A), probably because of a floor effect, as movement error is never completely eliminated
33
(supplementary information Figure I). This meant that the improvement in endpoint error was roughly proportional to the initial deficit.
34
The regression slopes of error reduction indicated a 20% to 30% improvement in performance (fast:
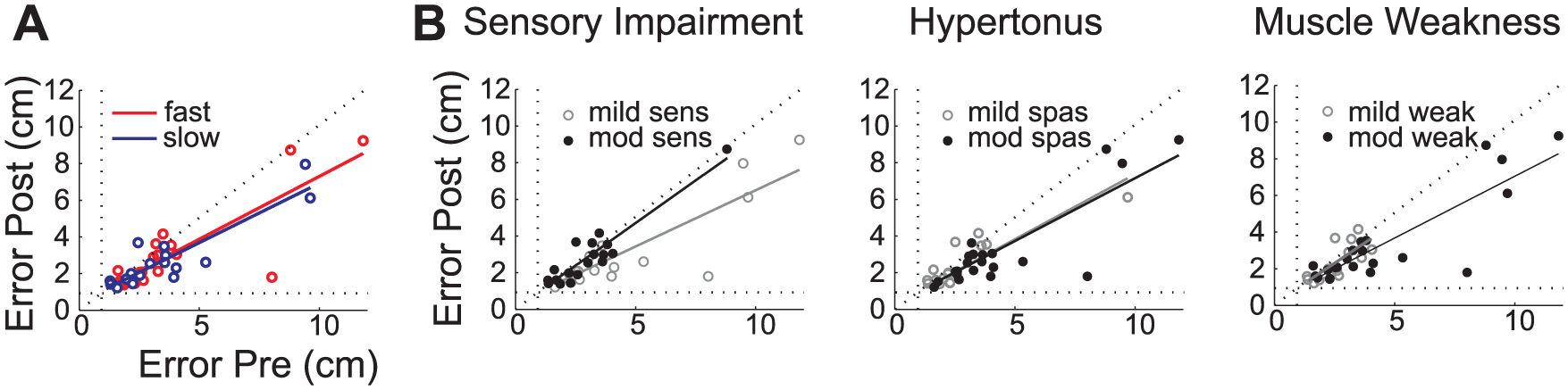
Effect of baseline ability and impairment on learning. (A) Correlation of baseline RMS error with the post training performance on an individual basis for the fast (dark grey) and slow (light grey) training group. The performance floor of 0.928cm is depicted by a dotted line. (B) Correlation of pre and post training measures of all individuals divided into groups of mild (grey) and moderate (black) sensory impairment, hypertonus and muscle weakness.
We asked whether the benefit of training varied between different subgroups of patients characterized by specific deficits. Severity of sensory impairment was the only factor that influenced learning (Figure 5B) as detected by the difference of the slope (Independent
The Influence of Training on Clinical Measures of Impairment
Elbow flexor hypertonus (MAS; Figure 6A) reduced in the group training at fast movement speed (related samples, Wilcoxon signed rank test,
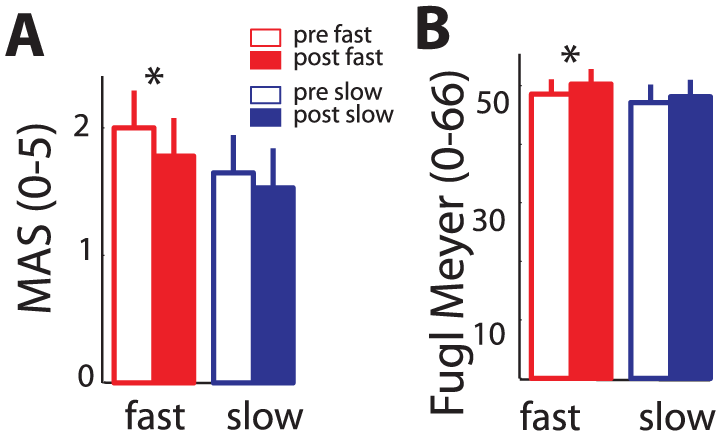
Functional outcome measures. (A) Mean elbow flexor hypertonus (MAS) and (B) Fugl–Meyer score for the fast (dark grey) and slow (light grey) training groups before (unfilled) and after (filled) training.
Discussion
Our experiment showed that with 4 days’ training chronic stroke survivors could improve reaching accuracy but correction for endpoint flexor bias was more difficult. Improvements in accuracy were achieved by reducing endpoint variability and were greatest at the trained speed but generalized to reaches made at untrained speeds. We recommend that training should be executed at a variety of speeds to maximize the breadth of generalization of improvements after training.
Reducing Movement Variability
Limiting compensatory trunk movement, while performing reaching movement, has been shown to be effective in improving movement quality in stroke survivors.35,36 Our setup prevented trunk flexion and rotation and minimized shoulder girdle movement, permitting only elbow and shoulder movement for the performance of the reaching movement. The change in the speed-accuracy relationship11,19,22 meant that at a retention test 1 day after training, patients could perform movements of a given speed more accurately than on the testing session before training. These improvements were not due to patients employing a different (ie, “compensatory”) strategy to achieve the same outcome. Instead, improved performance was the result of an established core characteristic of skill learning, namely, reduced trial-to-trial variation of movement extent and peak velocity.11,12 A similar conclusion was reached recently by Kitago and colleagues. 3 The neural mechanisms underlying these changes are still unknown, but it seems likely that they are similar to those underlying reduction in variability in healthy adults who learn comparable tasks. 11 These improvements are possibly mediated by the recruitment of more neurons for the execution of the task, 37 which effectively increases the neural signal-to-noise ratio 11 and improves performance.
Acquiring New Movement Patterns
Improvement in the speed-accuracy relationship is only one type of learning required after stroke. 38 Another component is reacquiring movements that were lost and are not within the present movement repertoire. In our protocol, the reaching movement required a range of active elbow extension, which was not initially possible for all patients. It produced an endpoint bias, which often involved undershooting the target with a bias toward flexion. However, training produced very little change in endpoint bias so that we have no evidence for this type of learning in the present data. The implication is that within the confines of their damaged motor system, chronic patients can still learn to control variability but find it more difficult to regain new movement patterns. Whether the latter would be possible in subacute stroke or with more extensive training is an important question.
Influence of Movement Speed During Training on Performance Changes
A recent article demonstrated that chronic stroke survivors demonstrated long-standing improvements in movement velocity and movement smoothness after performing only 2 training sessions consisting of 600 fast reaching movements. 5 However, limited evidence is available about the importance of performing training at different movement speeds in stroke rehabilitation2,14 nor are recommendations to incorporate different movement speeds during training included in clinical guidelines. 20 While it is difficult to compare accuracy improvements across different movement speeds directly, as the task difficulty is different between speeds, 18 our data clearly show that improvements for faster movement speeds cannot be effectively achieved by training at slow speeds. Fast training also resulted in a small improvement in clinical scores, which could indicate that performing fast movements is important for recovery after stroke. While our data suggest that fast movements speed improve slightly different aspects of motor control than training at slower speeds, we can only speculate about the underlying mechanisms. One possibility is that generation of larger agonist bursts necessary for fast movements led to more neuronal recruitment and therefore better improvements in functions.37,39 Alternatively, it could be that the increased necessity to account for interaction torques (eg, by stabilizing the shoulder) led to better learning outcome. 17
We suggest that training regimes for the upper limb should include a proportion of training with an emphasis on increasing movement speed, thereby also counteracting the general slowing of movements after stroke. 14 Our data show that training at fast speed did not increase hypertonus. However, at the current training intensity we found that training benefits were too small to be clinically relevant and did not lead to a change in the flexor bias. This can possibly be attributed to the fact that the short training period was insufficient to alter longer standing movement patterns.
The Impact of Impairment on Learning and Vice Versa
It is well established that muscle weakness, sensory loss, and increased muscle tone influence motor control after stroke.40,41 Less is known of the effect of these impairments on learning. In the present study, we found that sensory impairment reduced learning, consistent with previous studies.41-43 In contrast, we found no effect of increased tone or weakness. It is possible that removal of visual feedback during movement increased reliance on somatosensory feedback. If so, other types of training, using continuous visual feedback, might be less affected by sensory impairment.
Limitations
As this was a pilot study, there was no calculation of the number of subjects performed a priori to ensure study power and therefore a definitive trial would be required to validate these findings.
We investigated training at different movement speeds and therefore adjusted task difficulty according to each individual’s maximum movement speed. The target location and size remained constant for all individuals irrespective of their arm length or reaching distance. Therefore, task difficulty was slightly different depending on each individual’s initial ability but as we only included individuals who could end their movement within the 5 cm target, we believe that similar strategies were still required throughout our sample. Although arm dominance has been found to influence the performance of reaching in stroke survivors movements, 44 this study was not designed or powered to explore these aspects of motor learning.
The training period in this trial was too brief to allow for clinically meaningful changes in outcome measures, and the long-term retention of the altered behavior in our study was not explored; however, the small improvements in impairment are encouraging and might indicate the potential utility of more intensive training.
Conclusion
A greater understanding of recovery mechanisms is required in order to tailor individualized rehabilitation protocols.2-4,13 This repetitive training protocol improved performance in line with previous findings,1,2 despite training not being varied. 45 Our results show that performance improvement can be achieved without the use of compensatory strategies.4,7 Chronic stroke survivors improve reaching accuracy most notably at the trained movement speed by a reduction in movement variability. However, movement bias was not significantly changed. We can therefore conclude that in chronic stroke, improvements to the quality of existing movements is possible; however, the ability to learn new movements or muscle synergies may take longer periods of training or need to be achieved by alternative training strategies. Over the short training period, we did not observe clinically relevant group differences in clinical outcomes. However, these may emerge over longer training periods, and if so a variety of movement speeds should be included during training as accuracy improvements achieved after slow movement training do not generalize to fast movements.
Footnotes
Acknowledgements
We thank Nick Ward, Jonathan Marsden, and Sarah Tyson for helpful comments during the preparation of this article and Julius Klein for the provision of graphics.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Stroke Association (TSA 2010/06) to JCR and UH.
Supplementary material is available for this article online on the
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
