Abstract
Introduction
Stroke is a leading cause of severe disability in adults.1-3 Only 5% to 20% of all patients demonstrate full functional recovery of the paretic upper limb, whereas the arm and hand remain without function in 30% to 66% of all stroke survivors. 4 In patients who do show some improvements of upper limb function within the first 6 months, the greatest improvements occur typically in the first 5 weeks and taper off over 6 months poststroke. 5 However, the mechanisms underlying the spontaneous improvements of upper limb function early poststroke are still poorly understood.5,6
Several longitudinal studies revealed that motor recovery profiles are characterized by an improved ability to dissociate from pathological upper limb synergies, as reflected by increasing scores on the Fugl-Meyer Motor Assessment (FMA).7-9 Clinically, these improvements in FMA scores are often interpreted as a reflection of true neurological repair by which patients regain their ability to selectively control the different joints in the paretic upper limb. 10 In addition, prospective cohort studies have shown that functional outcome after stroke can be optimally predicted by increases in FMA scores within the first 4 to 5 weeks poststroke.7,10,11 This suggests that early improvements in selectively controlling the paretic upper limb are indicative if not determinant for motor recovery after stroke.
It has been suggested that, to enhance motor control by true neurological recovery, interventions should be applied within the first 4 to 5 weeks poststroke and should preferably be targeted at distinctively controlling selected joints in the paretic upper limb.
12
Unfortunately, there is still a lack of evidence that therapeutic rehabilitation interventions such as modified forms of constraint-induced movement therapy (mCIMT) in the first 10 weeks poststroke
13
yield true neurological repair and hence restore the quality of motor control.
5
A recent systematic review of clinical trials of the upper paretic limb suggests that therapeutic effects are mainly driven by improvements in proximal motor control, whereas improvements for hand recovery are poor.
14
Most clinical trials, however, do not incorporate longitudinal 3-dimensional (3D) kinematics, which certainly limits our understanding of
Specifically, studies are needed to establish whether motor recovery early poststroke during functional upper limb tasks, such as reaching, is mediated by restitution of motor function, which is reflected by learning to dissociate from pathological synergies. 18 Alternatively, functional recovery might rather be a process of adaptive motor learning by which patients learn to compensate for the inability to control degrees of freedom (DOFs) in the paretic upper limb by substitution of motor function such as using compensatory trunk movements.16,18 In a recent cross-sectional study, we were able to show that principal component analysis (PCA) is a valid tool to unravel the interaction between the pathological flexion synergy, represented by shoulder abduction and elbow flexion, and compensation strategies. 18 Capitalizing on this result, with the present study we aimed to assess longitudinal improvements in dissociated upper limb movements during a standardized reach-to-grasp task in patients with a first-ever ischemic stroke. Data obtained from healthy age-matched subjects during the same reach-to-grasp paradigm were considered “normal” and served as control. Based on the improvements in FMA in the first 5 weeks poststroke,7,8,10 we hypothesized that improvements in the control over DOFs in the paretic upper limb during reach-to-grasp, as revealed by PCA, parallel the hierarchical stages of FMA and occur mainly in the critical time window of the first 5 weeks poststroke.
Methods
Participants
The present study was part of the EXPLICIT-stroke program.
19
Stroke was defined according to the World Health Organization criteria.
20
Type and localization of stroke were determined using computed tomography or magnetic resonance imaging. Thirty-one patients with stroke (13 females and 18 males) were included with a mean age (±SD) of 60.0 (±11.2) years. All patients met the following criteria: (
In addition, 12 healthy participants (5 females/7 males) with no reported history of neurological and/or orthopedic disorders were included. Mean age (±SD) was 52.8 (±5.9) years. The EXPLICIT-stroke protocol (registered with the Netherlands National Trial Register as trial number NTR1424) was approved by the local ethics committee. 19 All patients underwent a clinical and 3D kinematic assessment of the paretic upper limb in 6 consecutive sessions (week 2, 3, 5, 8, 12, and 26 after stroke) whereas the healthy participants’ 3D kinematics was assessed once.
Clinical Evaluation
During the first assessment, the severity of the lesion was assessed with the National Institutes of Health Stroke Scale. 23 The functional ability of the paretic upper limb was measured using the Action Research Arm Test. 24 Physical disability with regard to activities of daily living was determined with the Barthel index. 25 All repeated measurements involved clinical assessment of motor function with the upper limb section of the FMA (FMA-UE). 26 The presence of pathological upper limb synergies between the shoulder and elbow was assessed with the shoulder and elbow subsections of the FMA (FMA-SE). 26
Kinematic Data Collection
Kinematic data were recorded with a portable 6 degrees of freedom electromagnetic tracking device (Polhemus Liberty, Polhemus, Colchester, VT). All movements were measured relative to a global reference frame with its origin at the center of the magnetic source,
Double-sided adhesive tape was used to attach the motion sensors to the thorax and to four segments of the paretic arm of each participant: scapula, upper arm, forearm, and hand. In the healthy participants, the nondominant side was used for 3D kinematic measurements. Using a pointer device (ST8 stylus, Polhemus), we conducted an anatomical calibration before each measurement to locate the position of 17 predefined anatomical landmarks (Appendix A) relative to their associated sensors. These locations were used to transform the rotations of each sensor into those of the trunk, upper arm, forearm, and hand. A detailed description of these transforms has been published previously. 18
Procedure
While seated at a table with a height of 76 cm, patients with stroke performed a functional reaching task with the paretic arm. In healthy individuals, motor control of the dominant upper limb is superior to that of the nondominant arm.
27
Therefore, we used the nondominant upper limb in healthy participants to exclude the possibility that potential differences between patients with stroke and healthy controls were caused by the superiority of dominant upper limb control in healthy subjects. The task consisted of 2 parts: (
The reach-to-grasp movement ended successfully as soon as the block had been grasped and lost contact with the table. Directly after this block lift, the reach-to-grasp movement proceeded into the second part of the movement, during which the block had to be displaced toward a target position located at the contralateral side at a distance equal to MRD.
Patients were specifically instructed to transport the block through the air, without touching the table, and to grasp the block between the thumb and index finger. Furthermore, participants were allowed to move their trunk away from the back of the chair, after the “GO” signal, but they were specifically instructed to remain seated and not to slide or twist over the seat of the chair throughout all motion recordings. If participants did not adhere to these instructions, the experimenter marked the recording as unsuccessful and a new trial was recorded. Four successful recordings were included in each measurement.
Data Analysis
In the present study, we focused on the first part of the experimental paradigm: the reach-to-grasp movement. The start of reach-to-grasp was defined as the moment at which the forearm sensor exceeded 5% of the maximum speed during the forward reach. 28 The end of reach-to-grasp was given as the moment at which the block lost contact with the table and the displacement of the block started. The end of reach-to-grasp was thus defined to be the moment at which the forearm sensor exceeded 5% of the maximum speed during the displacement of the block. 28 Movement duration was given as the time between the start and end of reach-to-grasp.
Trunk, shoulder, elbow, and wrist rotations between the start and end of reach-to-grasp were calculated according to the recommendations of the International Society of Biomechanics.
29
A detailed description of the 3D kinematic analysis has been published previously.
18
This procedure yielded 10 time series for trunk and joint rotations (positive/negative direction): (
To eliminate systematic error per measurement, the mean of each trunk and joint rotation within a measurement was subtracted from the time series.
Principal Component Analysis
For the patient group, the time series of the trunk, shoulder, elbow, and wrist rotations during the 4 trials of each of the 6 consecutive assessment days were concatenated. We concatenated all the 31 patient data, which yielded a combined multivariate set of 10 time series (trunk and joint rotations) containing (31 × 6 × 4 × number of samples per trial) data points each. Similarly, for the 12 healthy controls, we constructed 10 time series with (12 × 1 × 4 × number of samples per trial) data points each. Both of these 10-dimensional data sets underwent a PCA. 30 A PCA’s outcome can in general be interpreted as follows.
A principal component covers a certain amount of variance given by the corresponding eigenvalue of the data’s covariance matrix. 30 That variance (or eigenvalue) is considered the component’s degree of importance within the entire data set. Components are ranked by that variance in descending order, that is, by degree of importance. Projecting the data onto a component yields a time series that reveals the component’s contribution to the data as a function of time. This projection under the constraint of maximum variance is in fact equivalent to a linear regression. For example, if 2 joint rotations are fully synchronized (with zero phase-lag), all the accompanying variance can be described by a single linear regression equation, with the 2 joint rotations as independent variables. By contrast, if two joint rotations are executed linearly independent of one another, 2 regression equations will be required. Since the data set in the present study comprised 10 time series of joint rotations, a maximum of 10 regression equations or principal components could be identified. Because of sorting of eigenvalues in descending order, the first principal component represents a linear regression equation explaining the largest portion of variance of the set. We note that in the current study principal components were only considered relevant if the corresponding eigenvalue exceeded the broken stick distribution. 31 Furthermore, we interpreted the sum of the remaining components as overall deviation from the main coordination pattern given by the (sum of) relevant principal components mentioned above. Finally, the (absolute value of the) coefficients of each principal component’s eigenvector served to indicate the individual contributions of trunk and joint rotations to the component in question.
To establish the contribution of the principal components (ie, the main pattern) and residual components (deviations from the main pattern) for each patient at each time point poststroke, we adopted an approach introduced by Lamoth et al. 32 In brief, the original time series of each measurement (ie, for each patient at each time point after stroke) were first projected onto the principal and residual components. By this projection, the original time series of each measurement was divided into a global and a residual time series. Subsequently, the variance of the principal (Varprincipal) and residual (Varresidual) time series in each measurement was determined. The square root of the variance was used to obtain the standard deviation (SD) of the principal (SDprincipal) and of the residual (SDresidual) time series. See Appendix B for details.
Statistics
We investigated the changes of SDprincipal and SDresidual as a function of time poststroke using a repeated-measures analysis of variance with time of recording as a within-subject factor. Significance level was set at
Results
Table 1 shows the characteristics of the 31 patients with stroke and 12 healthy participants who were included in the present study. Nineteen patients had a lacunar anterior cerebral infarction (LACI) according to the Bamford classification, indicating that a majority of the patients had pure motor deficits without cognitive and/or visual impairments. Baseline motor impairments were present in the paretic upper limb and hand, as indicated by a mean score of 46 on the FMA-UE. In addition, disabilities were indicated by mean scores of 32 and 16 on the Action Research Arm Test and Barthel index, respectively.
Participant Characteristics.

Abbreviations: ARAT, Action Research Arm Test; BI, Barthel Index; FMA, Fugl-Meyer Motor Assessment; IQR, interquartile range; LACI, lacunar anterior cerebral infarction; NIHSS, National Institutes of Health Stroke Scale; PACI, partial anterior cerebral infarction; SD, standard deviation; TACI, total anterior cerebral infarction.
The median FMA-SE score increased to 35 (out of 36) points within the first 57 days poststroke and was then constant until 189 days poststroke. In addition, the mean movement duration of the reach-to-grasp task decreased mainly in the first 57 days poststroke and saturated to an asymptotic level at 189 days poststroke (Figure 1).
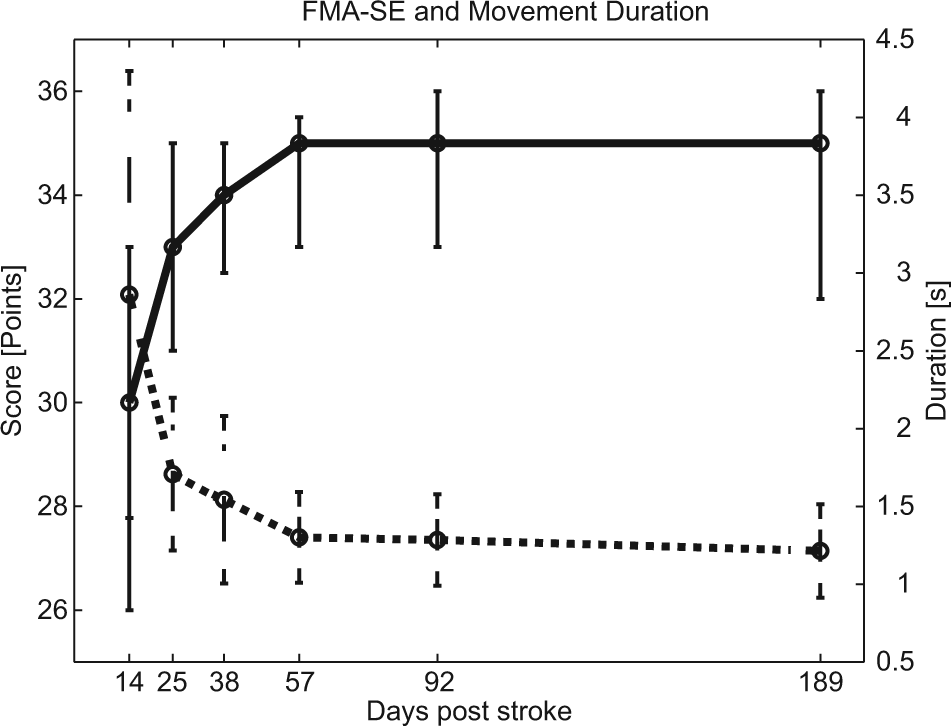
Median scores on the arm section of the Fugl-Meyer Motor assessment (FMA; solid line) and mean movement duration of the reach-to-grasp task (dashed line) at the different time points poststroke. Maximum score on the FMA-SE (SE, shoulder and elbow) is 36 points. Error bars represent interquartile ranges and standard deviations for the FMA-SE and movement duration, respectively.
Principal component analysis revealed that one principal component could be extracted on the basis of the broken stick distribution (Figure 2). This first principal component (PC1) explained 82% and 87% of the variance in trunk and joint rotations in patients with stroke and healthy subjects, respectively. The eigenvectors of PC1 in patients with stroke and healthy subjects are depicted in Figure 3. Visual inspection revealed that these vectors largely agreed for both groups. Horizontal shoulder adduction and elbow extension were dominant contributors to this component, whereas trunk and wrist rotations had little impact.
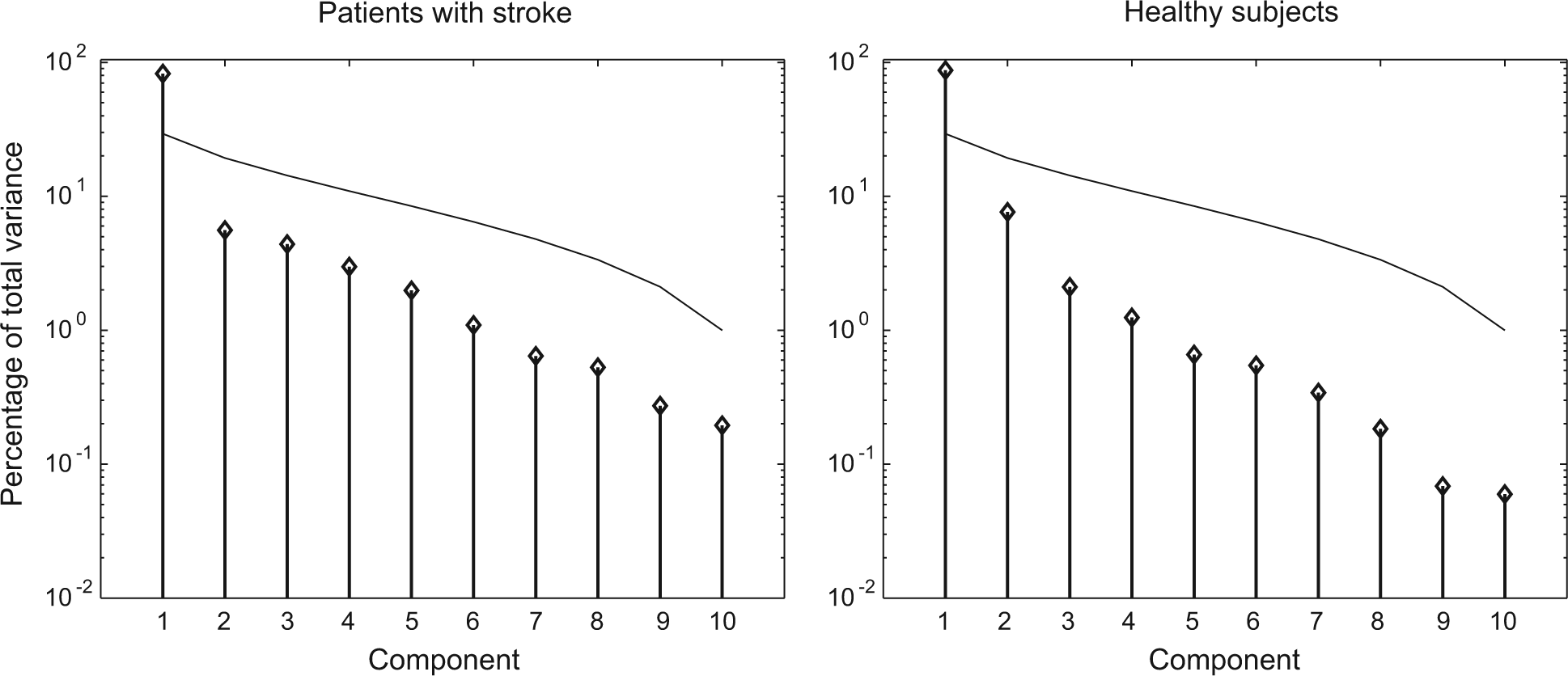
Percentage of variance explained by each component in patients with stroke (left panel) and healthy subjects. For both groups, only the first component could be identified as principal component (PC1), since it exceeded the broken stick distribution (thin line). PC1 explained 82% and 87% of the total variance in degrees of freedom (DOF) in patients and healthy subjects, respectively.
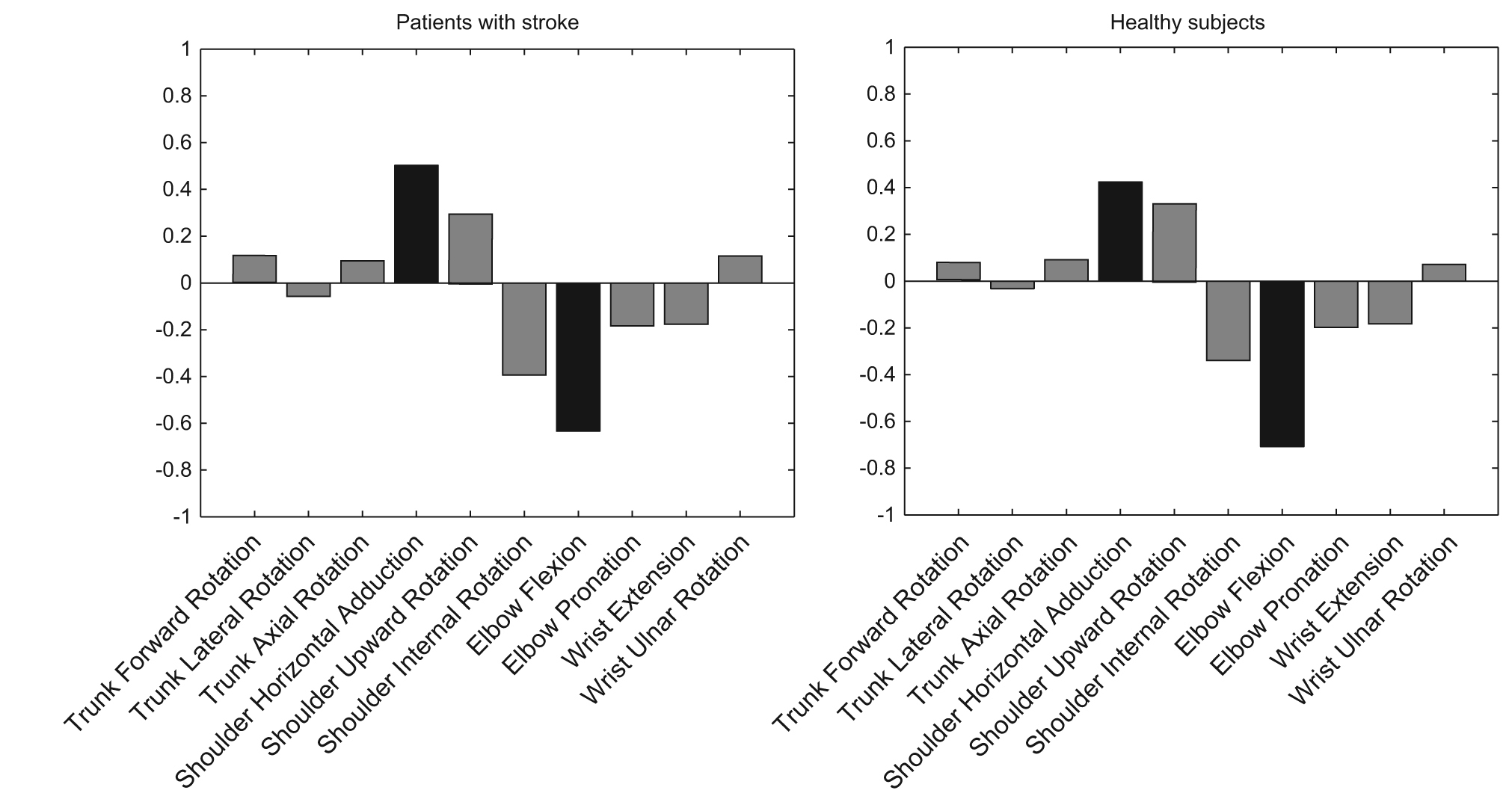
Eigenvectors of the first principal component (PC1) in patients with stroke and healthy subjects. The eigenvector is very similar for both groups. Shoulder horizontal adduction and elbow extension are the degrees of freedom (DOF) contributing most to reach-to-grasp in both groups.
The mean standard deviation of the main coordination pattern in the patients with stroke, as specified by the mean SDprincipal, increased with 19.3° between 14 and 189 days poststroke and this increase was significant (
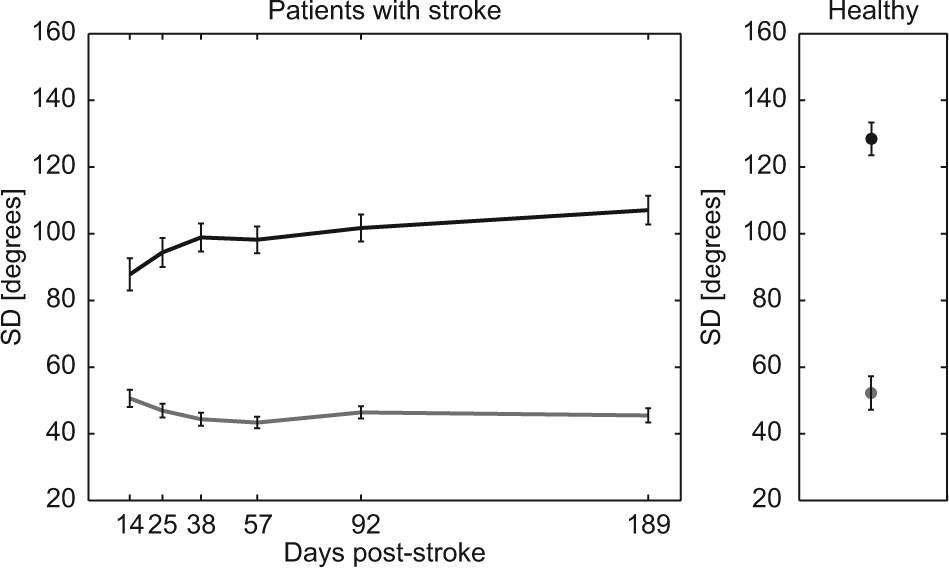
Mean SDprincipal (black) and SDresidual (gray) as a function of time poststroke. Error bars represent standard error (SD = standard deviation).
Discussion
In the present study, we tested the hypothesis that improvements in the ability to make dissociated upper limb movements, as revealed by the FMA, in the first 5 weeks poststroke, are paralleled by improvements in the control over DOFs in the paretic upper limb during a reach-to-grasp task. The FMA score increased until 8 weeks poststroke and, by applying PCA, we found a considerable increase in the variance that was explained by the main coordination pattern during the first 6 months poststroke (19.3°). Moreover, the most prominent increase occurred within the first 5 weeks poststroke (0.46° per day) and tapered off until 189 weeks poststroke (0.05° per day). The main coordination pattern consisted largely of horizontal shoulder adduction and elbow extension. The considerable increase in the standard deviation of this pattern early poststroke suggests that patients were increasingly able to dissociate from the pathological flexion synergy, which is often defined as a pathological coupling between shoulder abduction and elbow flexion.9,18,33
To the best of our knowledge, our study is the first to apply PCA for identifying longitudinal improvements in dissociated upper limb movements during reach-to-grasp in the first weeks poststroke. Our results are in line with previous longitudinal studies that used intensively repeated measurements of the FMA early poststroke.7,8,11 These studies showed that dissociations from the pathological flexion synergy as a reflection of spontaneous neurological recovery plateaued from 5 to 8 weeks onward.7,8,11 Hence, our findings do support our hypothesis that restitution of motor control, as quantified by improved control over the kinematic degrees of freedom in the paretic upper limb during functional reaching tasks, is primarily achieved in the first 5 weeks poststroke.6,8,11 whereas functional improvements in the chronic phase are primarily achieved by motor compensations. 34
It remains unclear, however, whether the detected changes in coordination over time poststroke are clinically meaningful, since minimally clinically important differences are still unknown for 3D kinematic analyses in stroke research. 35 In addition, it remains unclear which neurobiological processes are responsible for changes in adaptive motor control in the first 5 weeks poststroke. One may hypothesize that spontaneous neurological processes in the brain early poststroke, such as salvation of penumbral tissue and elevation of diaschisis, can explain early restitution of selective control over the joints in the paretic upper limb after stroke.6,36 Animal studies have shown that there is a critical time window early poststroke, which involves an upregulation of nerve growth factors, that may enhance neuronal recovery and hence improve motor control.12,37 The incomplete dissociated upper limb movements during reach-to-grasp at 6 months poststroke, as compared with those in healthy subjects, might reflect the residual neurological damage.
The main coordination pattern explained, relatively, an equal amount of the total variance in patients with stroke and healthy individuals, that is, 82% and 87%, respectively. However, in an absolute sense, the SD of the main coordination pattern remained lower throughout the first 6 months poststroke compared with healthy individuals. These lower absolute standard deviations suggest that fully dissociated upper limb movements were not achieved in the present sample of patients. Such incomplete dissociations in patients with stroke are consistent with findings in previous cross-sectional studies among patients with chronic stroke.38,39 Furthermore, our results suggest that upper limb motor control remains adaptive during reach-to-grasp.
Notwithstanding these differences between the patients with stroke and the healthy subjects, the median FMA-SE score was almost maximal (35 out of 36 points) at 6 months and the mean movement duration in patients with stroke was similar to that healthy subjects. This finding further supports the view that 3D kinematic measurements are valuable, next to routine clinical assessments, to increase our understanding of adaptive motor control after stroke.6,16,39,40
The promising results with regard to
Footnotes
Appendix A
Anatomical Landmarks
| Anatomical Landmarks (Defined in Anatomical Position) | Description |
|---|---|
| Thorax | |
| IJ: incissura jugularis | Deepest point (suprasternal notch) |
| PX: processus xiphoideus | Most caudal point on the sternum |
| C7: processus spinosus 7th cervical vertebra | Most dorsal point |
| T8: processus spinosus 8th thoracic vertebra | Most dorsal point |
| Scapula | |
| TS: trigonum spinae | Midpoint of the triangular surface on the medial border of the scapula in line with the scapular spine |
| AI: angulus inferior | Most caudal point of the scapula |
| AA: angulus acromialis | Most laterodorsal point of the scapula |
| PC: processus coracoideus | Most ventral point of the scapula |
| AC: acromioclavicular joint | Most dorsal point of the acromioclavicular joint |
| Humerus | |
| GH: glenohumeral rotation center a | Rotation center of the glenohumeral joint |
| EL: lateral epicondyle | Most caudal point on the EL |
| EM: medial epicondyle | Most caudal point on the EM |
| Lower arm | |
| US: ulnar styloid | Most caudal and medial point on the US |
| RS: radial styloid | Most caudal and lateral point on the RS |
| Hand | |
| MC3: processus styloideus os metacarpal 3 | Most dorsal point on dorsal side of the hand |
| MCP2: metacarpophalangeal 2 | Distal head of MCP 2 |
| MCP3: metacarpophalangeal 3 | Distal head of MCP 3 |
| MCP5: metacarpophalangeal 5 | Distal head of MCP 5 |
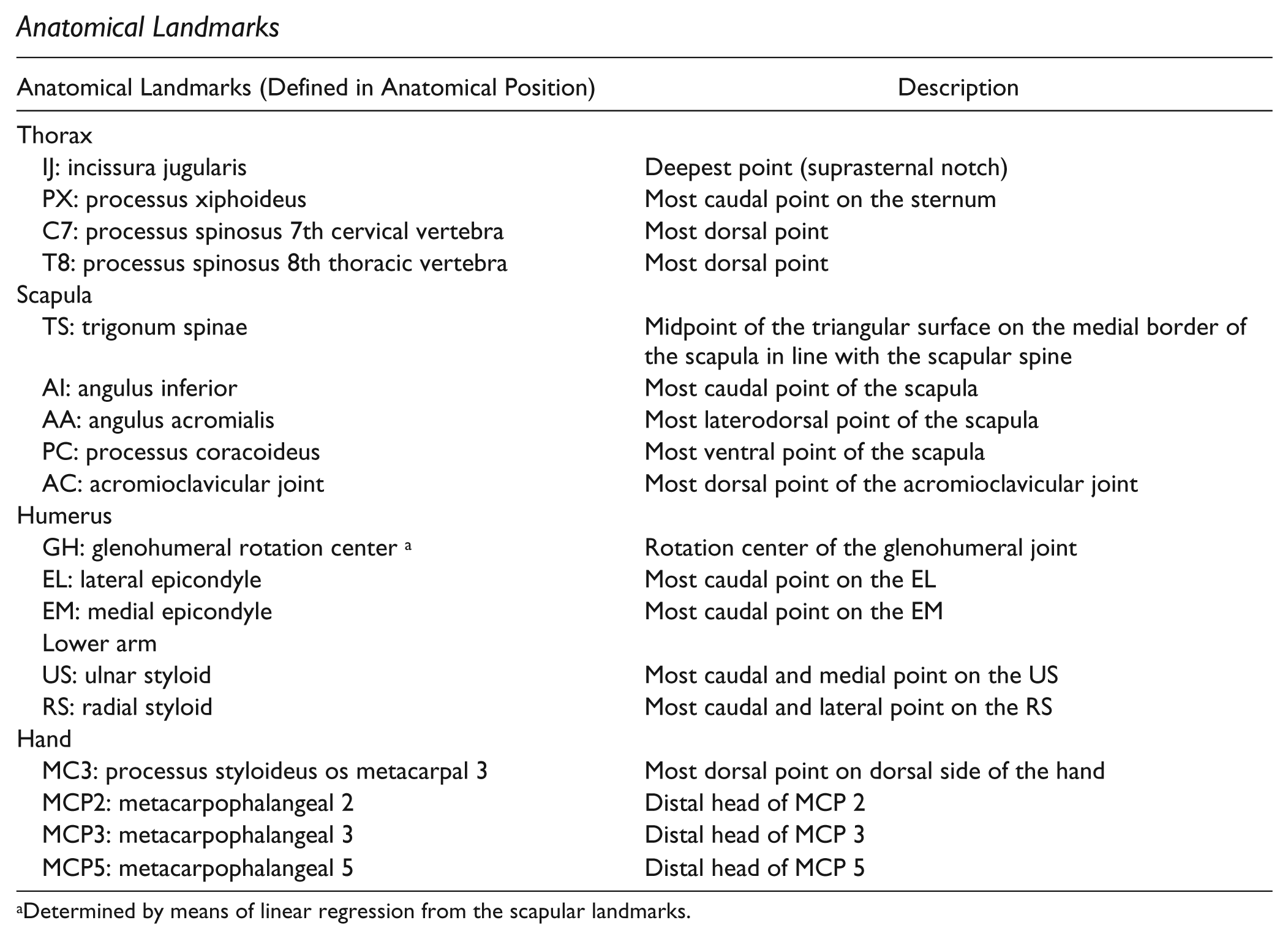
Determined by means of linear regression from the scapular landmarks.
Appendix B
The PCA resulted in 10 time evolutions
where
A measure of the variance that was explained by the principal and residual components was determined for each individual joint rotation in each individual subject and measurement:
where
To improve readability,
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was part of the EXPLICIT-stroke program (![]() ), which is registered with the Netherlands National Trial Register (NTR1424); it was supported by Het Revalidatiefonds, Revalidatie Nederland, and the VRA and was financed by ZonMw (Grant #89000001). AD received financial support from the Netherlands Organisation for Scientific Research (NWO Grant #400-08-127). GK received financial support from the European Research Council for the 4D-EEG program (ERC Advanced Grant #291339).
), which is registered with the Netherlands National Trial Register (NTR1424); it was supported by Het Revalidatiefonds, Revalidatie Nederland, and the VRA and was financed by ZonMw (Grant #89000001). AD received financial support from the Netherlands Organisation for Scientific Research (NWO Grant #400-08-127). GK received financial support from the European Research Council for the 4D-EEG program (ERC Advanced Grant #291339).
