Abstract
Background and objective. Proportional recovery of upper-extremity motor function and aphasia after stroke may suggest common mechanisms for spontaneous neurobiological recovery. This study aimed to investigate if the proportional recovery rule also applies to visuospatial neglect (VSN) in right-hemispheric first-ever ischemic stroke patients and explored the possible common underlying mechanisms. Methods. Patients with upper-limb paresis and VSN were included. Recovery defined as the change in Letter Cancellation Test (LCT) score at ~8 days and 6 months poststroke. Potential recovery defined as LCTmax-LCTinitial = 20 − LCTinitial. Hierarchical clustering separated fitters and nonfitters of the prediction rule. A cutoff value on LCTmax-LCTinitial was determined. The change in LCT and Fugl-Meyer Assessment Upper Extremity was expressed as a percentage of the total possible score to investigate the communality of proportional recovery. Results. Out of 90 patients, 80 displayed proportional recovery of VSN (ie, “fitters,” 0.97; 95% CI = 0.82-1.12). All patients who did not follow the prediction rule for VSN (ie, ”nonfitters”) had ≥15 missing O’s at baseline and failed to show proportional recovery of the upper limb. Conclusions. This study shows that the proportional recovery rule also applies to patients with VSN poststroke. Patients who fail to show proportional recovery of VSN are the same patients who fail to show proportional recovery of the upper limb. These findings support the idea of common intrahemispheric mechanisms underlying spontaneous neurobiological recovery in the first months poststroke. Future studies should investigate the prognostic clinical and neurobiological markers of these subgroups.
Introduction
Early prediction of outcome after stroke is a major challenge in neurorehabilitation. Depending on stroke severity, there is strong evidence that the logistic pattern of improvement during the first 10 weeks after stroke is mainly driven by the progress of time. 1 In 2008, Prabhakaran et al 2 suggested that the amount of motor recovery in the first 6 months after stroke is proportionally fixed at 70% of the patients’ maximum possible improvement on the Upper Extremity motor section of the Fugl-Meyer Assessment (FMA-UE). 2 This 70% prediction rule of UE motor recovery was confirmed in a number of prognostic studies.3-5 Recently, studies showed that functional intactness of the corticospinal pathway is essential to recover to a level of 70% of the maximum possible change.4-7
The amount of spontaneous neurobiological recovery early poststroke and, with that, the maximum proportional recovery rule seems not to be related to age, gender, and intensity of exercise therapy.1-4,8,9 This latter finding suggests that the improvement in motor impairment is mainly spontaneous by nature,2,8 whereas evidence that interventions are able to influence this spontaneous return is still lacking.9,10 Krakauer and coworkers showed that the maximum proportional recovery rule was not unique for motor recovery2,11 but also applies to aphasia 12 in patients with a first-ever left-hemispheric stroke.
In line with these findings one may question if this maximum proportional recovery rule of spontaneous neurobiological recovery is also applicable to other neurological (cognitive) impairments such as visuospatial neglect (VSN) in patients with a right-hemispheric stroke. To investigate the commonality of the underlying mechanisms of spontaneous neurobiological recovery, one may ask indeed whether patients who fail to follow the maximum proportional recovery rule in VSN also fail to show spontaneous motor recovery early poststroke.
The first aim of the present study was to investigate whether the maximum proportional recovery rule also applies to VSN in patients with a first-ever ischemic right-hemispheric stroke. Subsequently, we sought to identify the clinical characteristics of these hemiplegic patients with VSN who failed to show proportional recovery. We hypothesized that the proportional recovery rule is generalizable to VSN—that is, that the majority of patients recover to a level of 70% of the maximum possible improvement. 2 Furthermore, we anticipated that those patients who did not show the expected recovery of VSN would clinically present with more neurological deficits and more severe strokes according to the Bamford classification early poststroke. 13 To unravel whether the proportional prediction rule of spontaneous recovery reflects common underlying mechanisms, we explored if patients who fail to show the expected amount of proportional recovery for VSN would also fail to show proportional motor recovery of the upper paretic limb poststroke.
Methods
Participants
The present study included 2 samples of patients from the Stroke Intensity trial1,14,15 and the EXPLICIT-stroke trial.9,16 The ethics committees of the participating hospitals approved both trials. Patients were included within 14 days after stroke onset.
Within the Stroke Intensity trial, patients were tested weekly during the first 10 weeks poststroke, biweekly until 20 weeks, and at the 26-week follow-up assessment. For the first 20 weeks after stroke onset, all patients received 15 minutes of leg rehabilitation, 15 minutes of arm rehabilitation, and 90 minutes of activities of daily living training per day. In addition, the different treatment groups received the following interventions for the first 20 weeks after stroke: (1) control treatment: immobilization of the paretic arm and leg using an inflatable pressure splint (30 minute on weekdays); (2) arm training: functional exercises with the arm and hand including grasping, moving objects, and punching a ball (30 minutes on weekdays); and (3) leg training: sitting, standing, and weight-bearing exercises, with emphasis on stability and improving walking velocity (30 minutes on weekdays). After the 20-week intervention period, the treatment was not controlled for; on average, patients received treatment on 3 days, 30 minutes per week. 14
Patients participating in the EXPLICIT-stroke trial were assessed weekly during the first 5 weeks and at 8, 12, and 26 weeks after stroke. 16 Patients were stratified based on the presence or absence of voluntary finger extension prior to the randomization procedure. Patients who presented with voluntary finger extension were placed in the modified Constraint Induced Movement Therapy (mCIMT) trial and the other patients in the ElectroMyoGraphy-triggered NeuroMuscular Stimulation (EMG-NMS) trial. The interventions were provided on weekdays for 3 consecutive weeks starting within 2 weeks after stroke onset. The interventions received by the different groups were as follows: (1) mCIMT experimental group: 60 minutes of UE task-oriented training with increasing task difficulty and 3 hours of wearing a padded safety mitt on the nonparetic arm 17 ; (2) mCIMT control group: 30 minutes of conventional care based principles of the Neuro Development Treatment, including facilitation movements and training of mobility, force, and coordination; (3) EMG-NMS experimental group: 60 minutes of stimulation of the finger extensors, requiring active participation of the patient; and (4) EMG-NMS control group: 30 minutes of passive range-of-motion exercise and facilitation of voluntary movements. After the 3-week intervention period, all patients received a maximum of 30 minutes of upper-limb exercise therapy. 9,16 In both trials, no specific intervention was provided for VSN.
For the present study, patients were included when they satisfied the following criteria: (1) had a first-ever ischemic stroke in the territory of the anterior or middle cerebral artery; (2) were adults up to 80 years of age; (3) did not have severe comorbidities, such as other neurological disorders; (4) did not have severe deficits in communication, memory, or understanding (Mini Mental State Examination of at least 23 points); (5) had upper-limb paresis (FMA-UE <66 points); (6) gave written informed consent; (7) had a lesion in the right hemisphere; and (8) presented with VSN.
Outcome Measures
A single-target Letter Cancellation Test (LCT) served as the main outcome measure to assess VSN.18,19 Patients were requested to cross all O’s on an A4 paper taped on the table and aligned to their sagittal midline. Each paper contained 40 O’s (20 in the contralesional and 20 in the ipsilesional visual field, respectively left and right) and 425 distractor letters. We defined VSN as an asymmetry between the LCT omissions in the contralesional (left) and ipsilesional (right) visual field of at least 2. 20
Baseline characteristics of the patients with a first-ever, ischemic, right-hemispheric stroke included the following: time between stroke onset and baseline assessment, gender, age, clinical severity of stroke using the Bamford classification, 13 and hemianopia and conjugate deviation of the eyes measured with the National Institutes of Health Stroke Scale. 21 UE motor function was measured with the FMA-UE 22 and the Motricity Index. 23 The Motricity Index was also used to assess lower-extremity motor function. At 6 months, the LCT and the FMA-UE were again determined. Three trained researchers performed all assessments.
Analysis
The effect of the type of intervention on the recovery of VSN (ie, LCT score contralesional visual field) was tested with generalized estimating equations for both trials separately. 24 All the available longitudinal data were used for this analysis. The model included an exchangeable working correlation matrix, LCT baseline score, intervention group, time (continuous), and the interaction between intervention group and time.
Maximum potential recovery (LCTmax-LCTinitial) was calculated by subtracting the baseline LCT score from the maximum possible score of 20 O’s in the contralesional (left) visual field. The observed change in LCT score was calculated as follows: ΔLCT = LCT6-months − LCTinitial. To investigate the relationship between ΔLCT and LCTmax-LCTinitial, we applied linear regression analysis to all available data. Hierarchical clustering based on average pairwise Mahalanobis distance (Matlab’s Statistics toolbox, version 8.1, Matlab version 2012a, Mathworks Inc, Natwick, MA) was used to identify 2 groups (ie, fitters and nonfitters). 25 This analysis was based on the ΔLCT versus LCTmax-LCTinitial scatterplot. We determined the robustness of that clustering by computing the cophenetic correlation coefficient 26 and the Spearman correlation coefficient between the Mahalanobis and cophenetic distances obtained from the dendrogram. In the group of fitters, we further investigated the relationship between ΔLCT and LCTmax-LCTinitial and the impact of possible confounding factors. Thereafter, we determined a cutoff value on LCTmax-LCTinitial to separate fitters from nonfitters of the VSN prediction rule.
Baseline variables were compared between fitters and nonfitters, and tested for normality by visual inspection of histograms and Q-Q plots. The Levene’s test served to evaluate the assumption of homogeneity of variance. Group comparisons for categorical variables were realized using Fisher’s exact test, for normally distributed data with equal variances using the independent t-test, and for nonnormally distributed data using the Mann-Whitney U-test.
Observed and predicted maximum potential change in UE motor function were determined similar to LCT using the FMA-UE with a maximum score of 66 points (ΔFMA-UE = FMA-UE6-months − FMA-UEinitial, and FMA-UEmax-FMA-UEinitial = 66 − FMA-UEinitial). In addition, ΔLCT, LCTmax-LCTinitial, ΔFMA-UE, and FMA-UEmax-FMA-UEinitial were expressed as percentage of the total possible score (20 and 66 points, respectively) to visually compare the distribution of observed recovery and maximum potential recovery on both measures. Analyses were performed with SPSS version 20 using a 2-tailed significance level of .05, unless indicated otherwise.
Results
A total of 90 patients with an ischemic first-ever right-hemispheric stroke were eligible for analysis (Figure 1) and assessed at 7.6 ± 2.9 and 189.6 ± 2.9 days after stroke onset. Patients’ baseline characteristics are shown in Table 1. For both trials, the type of intervention did not significantly affect the recovery of VSN in terms of LCT score for the contralesional (left) visual field. No significant interaction effects between type of intervention and time were found (p = .473 and p = .978, respectively, for the Intensity Stroke trial and EXPLICIT-stroke trial).
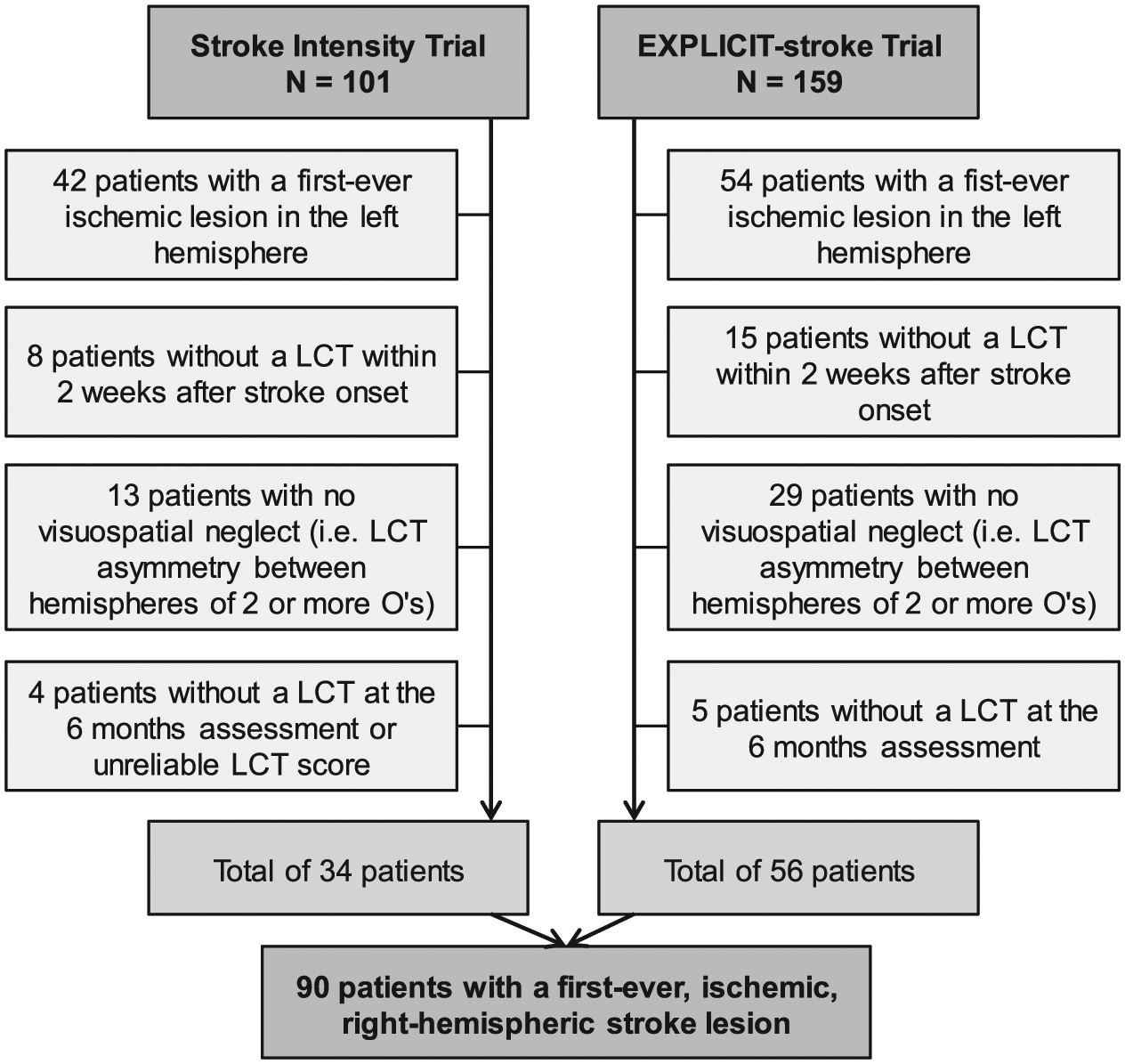
Patient exclusion flowchart to select patients with ischemic, first-ever, right-hemispheric stroke lesions.
Characteristics of the Patients with First-Ever, Ischemic, Right-Hemispheric Strokes.

Abbreviations: FMA-UE, Upper Extremity motor section of the Fugl-Meyer Assessment; LACI, lacunar anterior cerebral infarction; LCT, Letter Cancellation Test; MI, Motricity Index; NIHSS, National Institutes of Health Stroke Scale; PACI, partial anterior cerebral infarction; TACI, total anterior cerebral infarction.
Mean ± SD.
Number of patients.
Median (interquartile range).
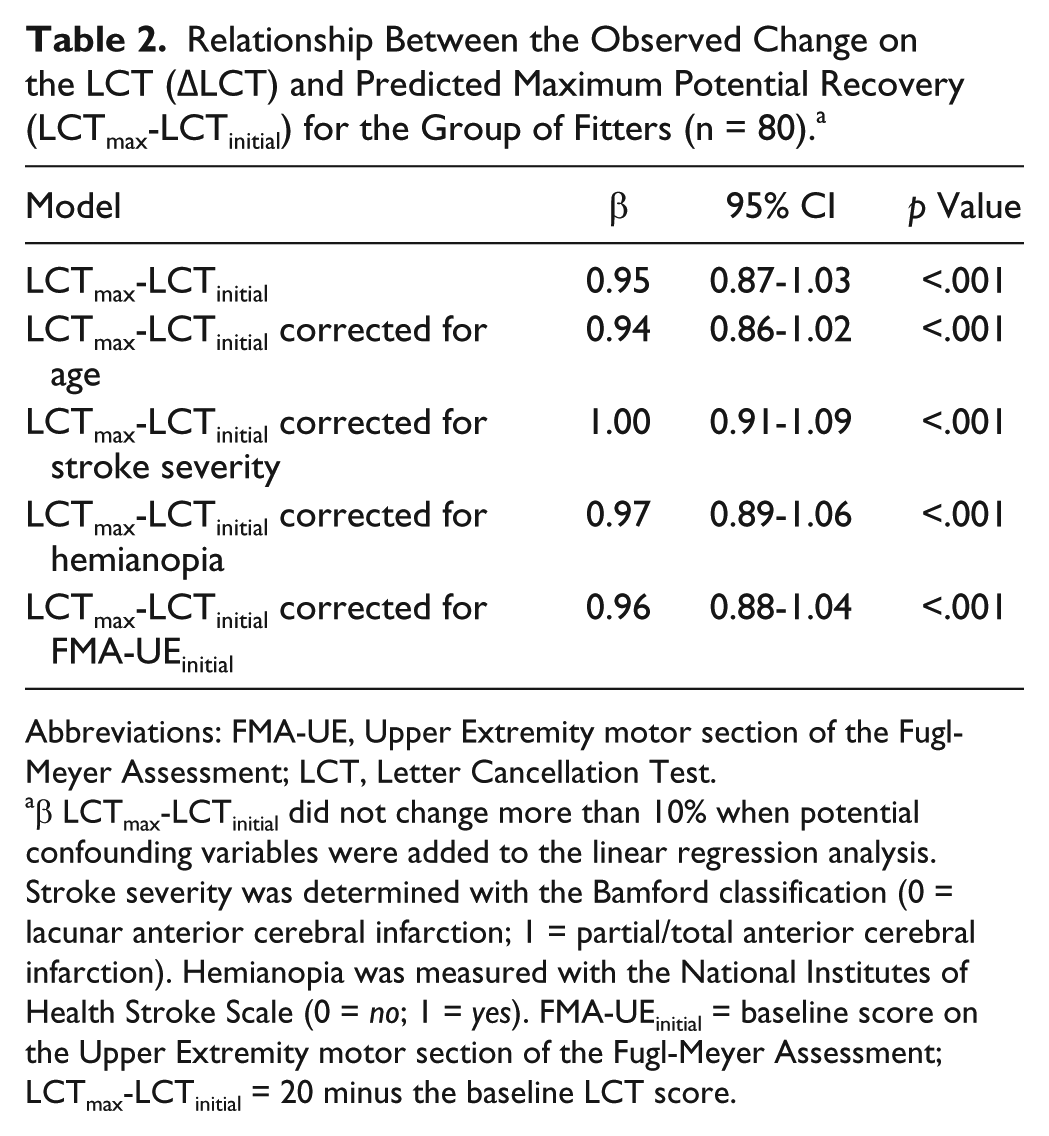
Figure 2 shows the ΔLCT versus the LCTmax-LCTinitial scatterplot for all 90 patients. The relationship between LCTmax-LCTinitial and ΔLCT for the whole group was ΔLCT = 0.73 (95% CI = 0.57-0.89) LCTmax-LCTinitial + 0.21 (95% CI = −2.24 to 2.67), with R2 = 0.48. Using the hierarchical cluster analysis, we identified 2 groups: N1 = 80 and N2 = 10 (clustering cutoff = 2.07). The cluster analysis had a goodness-of-fit of 0.86 and 0.87, for the cophenetic and Spearman correlation coefficients, respectively. For the fitters (N = 80), the relationship between LCTmax-LCTinitial and ΔLCT was ΔLCT = 0.97 (95% CI = 0.82-1.12) LCTmax-LCTinitial − 1.41 (95% CI = −2.85 to 0.03), with R2 = 0.78. This relationship was not influenced by age, stroke severity, hemianopia, or upper-limb motor impairment (Table 2).
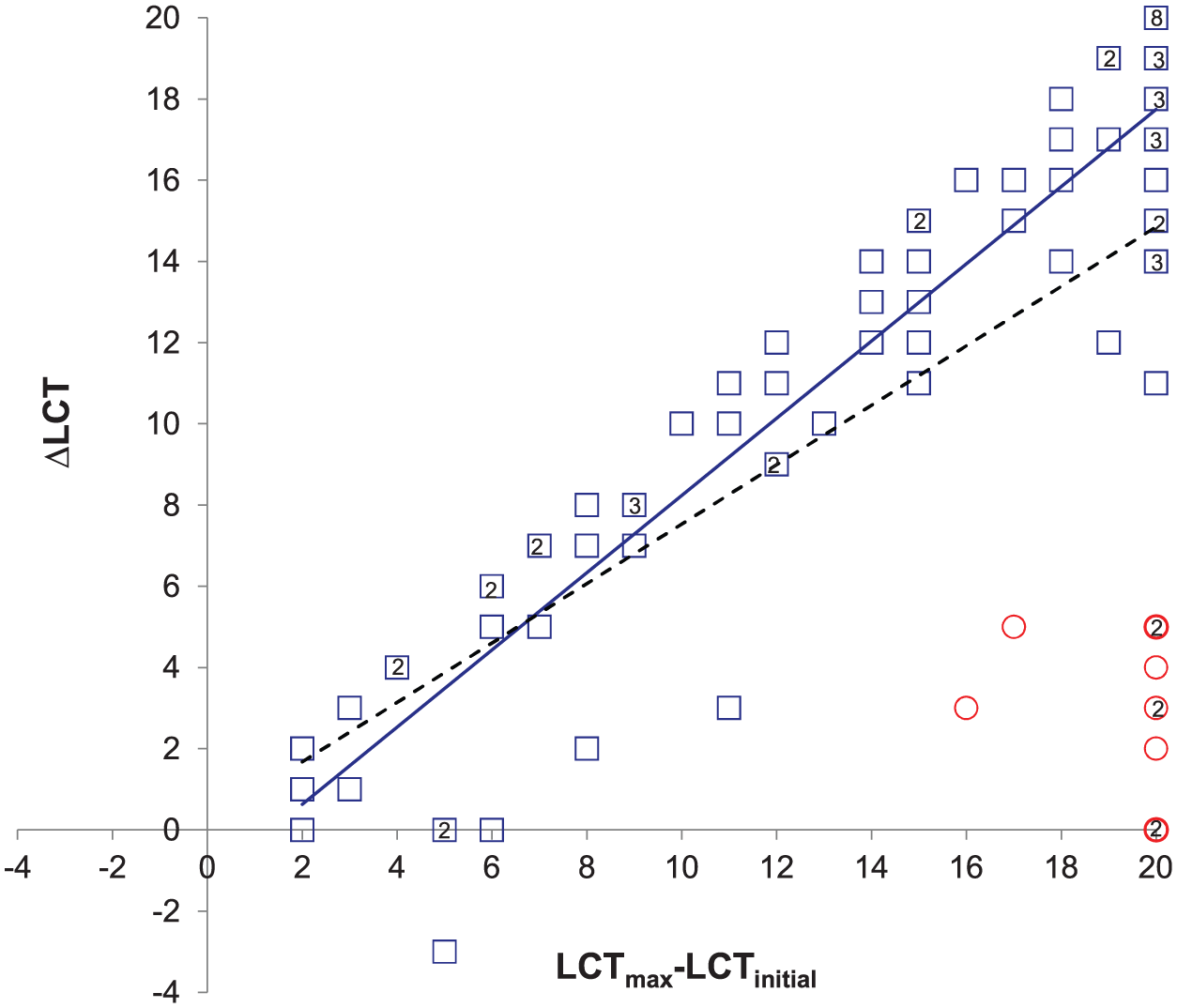
Proportional recovery of visuospatial neglect: observed change on the LCT (ΔLCT) in the contralesional (left) visual field versus predicted maximum potential recovery (LCTmax-LCTinitial). The circles and squares represent, respectively, the nonfitters and fitters according to the hierarchical cluster analysis. The dashed line represents the regression line of the whole group (N = 90): ΔLCT = 0.73 (95% CI = 0.57-0.89) LCTmax-LCTinitial + 0.21 (95% CI = −2.24 to 2.67); R2 = 0.48. The solid line represents the regression line for the fitters (N = 80): ΔLCT = 0.97 (95% CI = 0.82 to 1.12) LCTmax-LCTinitial − 1.41 (95% CI = −2.85 to 0.03); R2 = 0.78. LCT contralesional (left) visual field score, score ranging from 0 to 20 O’s. Not all data points are visible because of overlap. The numbers within the bold symbols indicate the number of participants having the same score.
Relationship Between the Observed Change on the LCT (ΔLCT) and Predicted Maximum Potential Recovery (LCTmax-LCTinitial) for the Group of Fitters (n = 80). a
Abbreviations: FMA-UE, Upper Extremity motor section of the Fugl-Meyer Assessment; LCT, Letter Cancellation Test.
β LCTmax-LCTinitial did not change more than 10% when potential confounding variables were added to the linear regression analysis. Stroke severity was determined with the Bamford classification (0 = lacunar anterior cerebral infarction; 1 = partial/total anterior cerebral infarction). Hemianopia was measured with the National Institutes of Health Stroke Scale (0 = no; 1 = yes). FMA-UEinitial = baseline score on the Upper Extremity motor section of the Fugl-Meyer Assessment; LCTmax-LCTinitial = 20 minus the baseline LCT score.
The patients in the nonfitter group (N = 10) all had 15 or more missing O’s (80%) at baseline assessment (Figure 2). This cutoff was used to separate patients into group 1 (LCTmax-LCTinitial ≤15 O’s; N = 45) with solely fitters and group 2 (LCTmax-LCTinitial >15 O’s; N = 45) with both fitters and nonfitters. Table 3 shows the comparison between a subgroup of fitters (N = 35) and nonfitters (N = 10) with a LCTmax-LCTinitial of more than 15 O’s. At baseline assessment, fitters were 11 years younger than the nonfitters (p = .007) and presented with significantly higher LCT scores for the ipsilesional (right) visual field (fitters: 11.3 ± 5.5; nonfitters: 7.2 ± 5.6; p = .044). Gender, severity of stroke according to the Bamford classification, hemianopia, déviation conjugée, UE and lower-extremity motor function, and time between stroke and baseline assessment, were not significantly different between these 2 subgroups, with 15 or more missing O’s at baseline.
Comparison of Baseline Variables of Fitters and Nonfitters With a Predicted Maximum Potential Recovery Score (LCTmax − LCTinitial) of More Than 15 O’s.
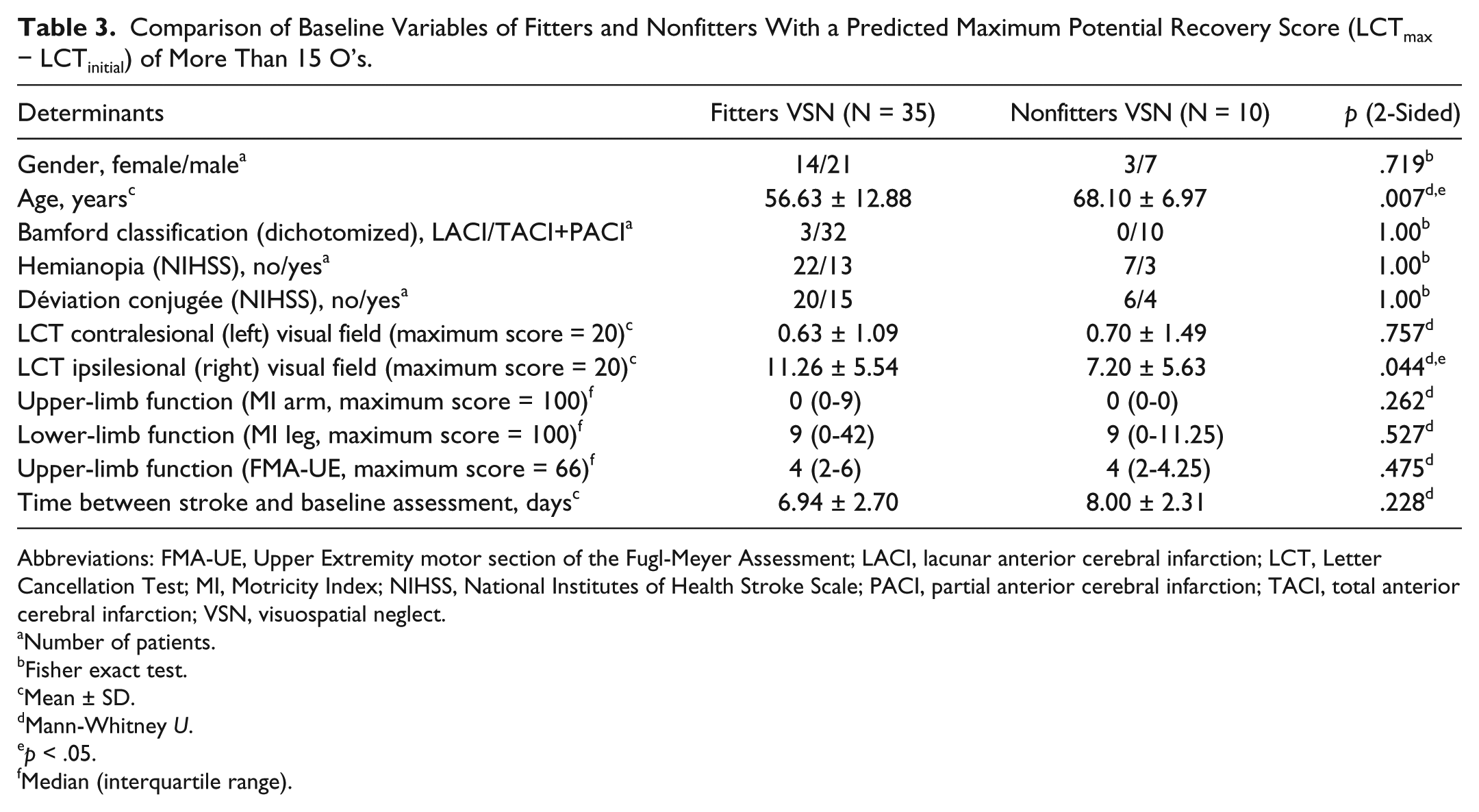
Abbreviations: FMA-UE, Upper Extremity motor section of the Fugl-Meyer Assessment; LACI, lacunar anterior cerebral infarction; LCT, Letter Cancellation Test; MI, Motricity Index; NIHSS, National Institutes of Health Stroke Scale; PACI, partial anterior cerebral infarction; TACI, total anterior cerebral infarction; VSN, visuospatial neglect.
Number of patients.
Fisher exact test.
Mean ± SD.
Mann-Whitney U.
p < .05.
Median (interquartile range).
Observed change and predicted maximum potential recovery on the LCT and FMA-UE showed a similar pattern when expressed as percentage of the total possible score on each test, although more patients showed proportional recovery in VSN than in FMA-UE (Figure 3). All VSN nonfitters (N = 10) lacked proportional recovery for upper-limb motor function. Cluster analysis on the FMA-UE scores yielded 2 groups: N1 = 66 and N2 = 24. The relationship between ΔFMA-UE and FMA-UEmax-FMA-UEinitial for the fitters (N = 24) was: ΔFMA-UE = 0.81 (95% CI = 0.66-0.96) FMA-UEmax-FMA-UEinitial − 2.29 (95% CI = −8.74 to 4.17) with R2 = 0.85. This relationship was not influenced by age, stroke severity according to the Bamford classification, homonymous hemianopia, or LCTinitial.
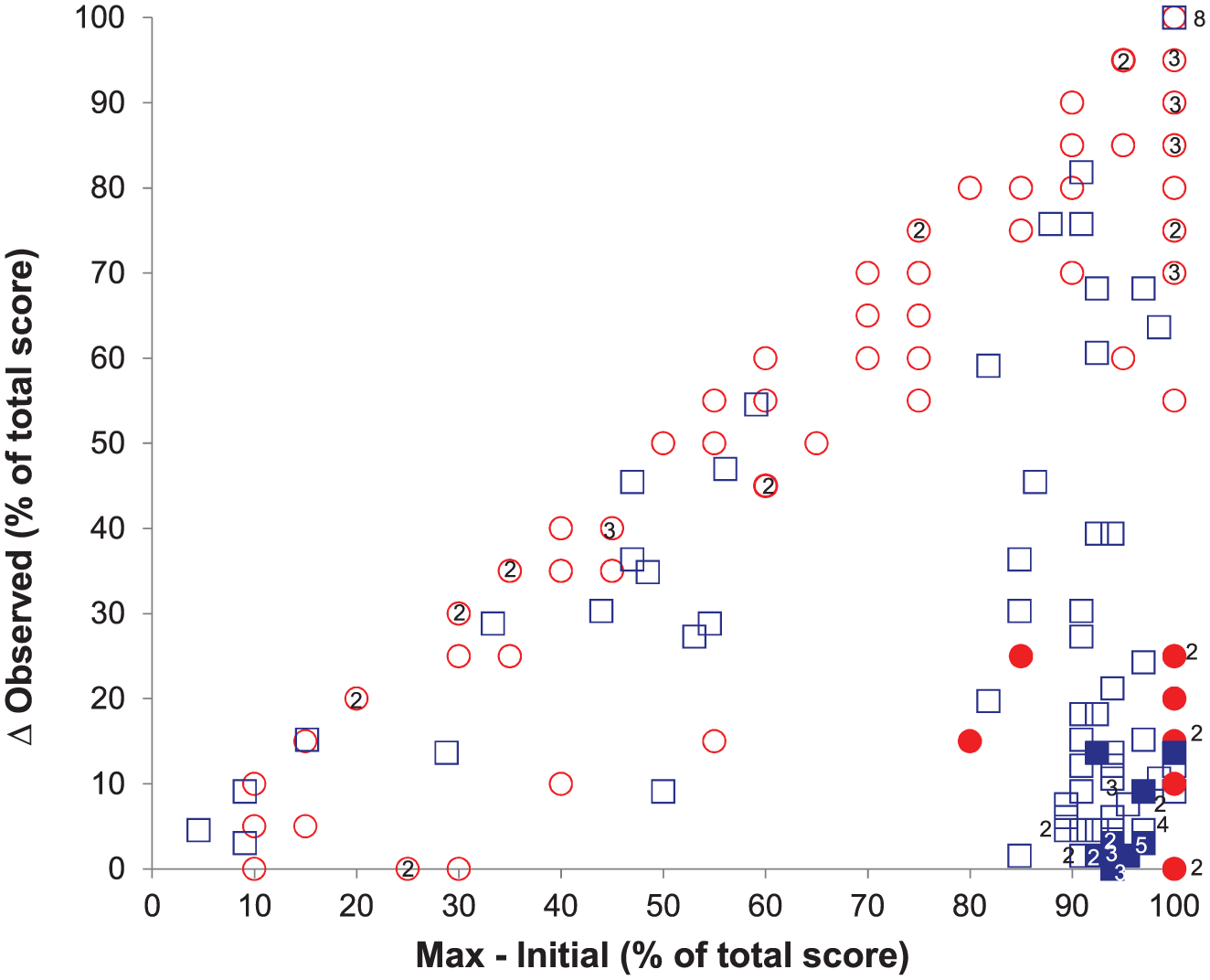
Proportional recovery of upper-extremity motor function and visuospatial neglect in the contralesional (left) visual field. The circles and squares represent, respectively, the results for the Letter Cancellation Test (LCT; contralesional visual field, score: 0-20) and Upper Extremity motor section of the Fugl-Meyer Assessment (score: 0-66). The closed circles and squares represent the group of visuospatial neglect nonfitters (see text for details). Δ Observed = observed change on the LCT; Max-Initial = Predicted maximum potential recovery. Not all data points are visible as a result of overlap. The numbers within or beside the bold symbols indicate the number of individuals having the same score.
Discussion
The first aim of the present study was to probe the generalizability of the fixed proportional recovery rule—that is, to determine if the 70% rule for UE motor recovery also applies to VSN in first-ever ischemic stroke patients with a lesion in the right hemisphere. We also sought to identify clinical characteristics of patients who failed to follow the proposed maximum proportional recovery rule. Subsequently, we investigated whether there are common biological mechanisms underlying this prediction rule by identifying VSN patients who lacked both the predicted amount of proportional recovery for VSN as well as recovery of UE motor function following the FMA-UE score.
To the best of our knowledge, this is the first study showing that the majority of first-ever ischemic right-hemispheric stroke patients (89%) follow a proportionally fixed amount of recovery of approximately 97% in VSN. The amount of proportional recovery seen in this cohort is higher when compared with the percentages previously found for UE motor function (means ranging from 64% to 83%)2-5,11 and aphasia (means ranging from 68% to 78%). 12 Patients who had 15 or fewer missing O’s on the LCT at baseline assessment showed an improvement on the LCT that was similar to their maximum potential recovery score at baseline, whereas nonfitters were only found at thresholds above 15 missing O’s in the first days poststroke. Most (78%) patients who had severe VSN impairment (more than 15 missing O’s at baseline) did follow the proportional recovery rule.
Our results suggest that in the subgroup of patients with severe VSN, older patients with seemingly bilateral VSN (represented by lower LCT scores for the ipsilesional visual field) were more likely to not show proportional recovery. We did not expect age to be a potential predictor for proportional recovery of VSN because it did not appear to be a strong predictor in previous studies investigating proportional recovery.2-5,12 Neglect, however, was suggested to be more common and severe in older stroke survivors, which may be related to the larger amount of prestroke brain atrophy.27,28 The seemingly more bilateral impairment in the group of nonfitters in comparison to the fitters with severe VSN (15 or more missing O’s) may reflect a process of remote suppression of the nonaffected hemisphere by transhemispheric diaschisis, as hypothesized by Von Monakow more than a century ago.29-31
More important, patients who fail to show the proportional amount of spontaneous neurobiological recovery in VSN also fail to follow the proportional recovery rule in UE motor function. The latter lends further support to the idea that the processes that drive spontaneous neurobiological recovery are part of common biological mechanisms, which is irrespective of the type of neurological impairment involved. As a consequence, a minority of all first-ever, ischemic stroke patients fail to show the expected amount of spontaneous neurobiological recovery of impairments. 3
Our findings are in line with previous prospective cohort studies showing that the time course of VSN parallels spontaneous neurobiological recovery in motor function such as synergistic movements measured with the FMA-UE and strength of the upper and lower paretic limbs.1,20,32 A number of prospective cohort studies demonstrated that VSN may have a suppressive effect on the recovery patterns of UE function via dysfunction of cortical networks.32,33 We believe that one needs to be careful in using strict cutoff points below which patients are expected to lack proportional recovery. Even very severely affected patients with scores below the threshold of 4 points (6% of maximum score) on the FMA-UE and 18 missing O’s (10% of maximum score) on the LCT within the first week may still follow the maximum proportional recovery rule.
Unfortunately, we cannot explain why 10% to 30% of the first-ever ischemic stroke patients fail to follow the proportional recovery rule. Previous studies showed that intactness of the corticomotor pathways, measured with transcranial magnetic stimulation and diffusion tensor imaging, is an important predictor for discriminating between patients with or without proportional recovery of upper limb motor function.4,5,7 Other key white matter pathways may also be essential for spontaneous neurobiological recovery in the context of VSN. For example, Corbetta et al 33 showed that attention deficits in VSN are mediated by dysfunction of the frontoparietal attention networks.
Some limitations should be considered when interpreting the present results. First, only patients with a right-hemispheric, first-ever ischemic stroke were investigated. Our findings suggest, however, that there are unknown mechanisms driving spontaneous neurobiological recovery, irrespective of the type of neurological impairment involved. Hence, it appears obvious to investigate whether left-hemispheric stroke patients who lack proportional recovery of speech 34 also fail to show proportional improvement of sensory-motor impairments. Second, we did not present direct measures for lesion size and location. However, the large array of clinical outcome measures suggest that the nonfitters had not only larger but also more cortical involvement of their strokes when compared with the fitters who presented with impairments related to smaller subcortical strokes in the white matter. Third, because of the relatively late initial assessment of on average 8 days (compared to within 72 hours after onset in other studies), we might have missed some cases of very early recovery of VSN. Fourth, we assessed VSN with a paper-and-pen Single-Letter Cancellation Test and used the asymmetry between the left and right side as a measure of neglect. 32 We used an asymmetry cutoff point of 2 to include a range of patients with mild to severe neglect. Future studies may use a variety of tests to assess the different aspects of neglect and use other measurements and/or cutoff scores to determine neglect severity. 35 Fifth, we only reported the LCT score for the contralesional (left) visual field. However, the recovery of VSN in terms of LCT score for the ipsilesional (right) visual field also followed the proportional recovery rule. Beyond a critical threshold of 15 missing O’s at baseline, the nonfitters did show more bilateral impairment in comparison to those severely affected patients who did follow the recovery rule. Sixth, although the present study reflects one of the largest cohorts suffering from neglect poststroke, the relatively small sample size and lack of neurobiological markers did not allow further investigation of the clinical differences between fitters and nonfitters using multivariate regression analysis.
In conclusion, the current findings provide further support for the conceptual notion that the amount of recovery is proportional to the initial impairment. There may be common biological mechanisms underlying spontaneous neurobiological recovery in the first months after stroke onset. To substantiate this premise, future studies should investigate the proportional recovery for other neurological impairments in larger stroke populations. These studies should combine early clinical and neurophysiological biomarkers, 36 primarily to identify fitters and nonfitters of the maximum proportional recovery rule in order to optimize early stroke triage, to better inform patients and caregivers, and most important, to stratify patients for neurorehabilitation in early-started intervention trials. 37
Footnotes
Acknowledgements
The authors thank the Stroke Intensity trial and EXPLICIT-stroke trial physicians, therapist, and nurses at the stroke units of the participating university centers, local hospitals, and in the associated rehabilitation centers and nursing homes as well as the patients who participated in the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results has received funding from the Royal Dutch Society of Physical Therapy, supported by the EXPLICIT-stroke grant from the Netherlands Organization for Health Research and Development (ZonMw; Grant No. 89000001) and the European Research Council under the European Union’s Seventh Framework Programme (FP/2007-2013)/ERC Grant Agreement n. 291339-4D-EEG. The funding organization had no role in the design or conduct of the study; the collection, management, analysis, or interpretation of the data; or the preparation, review, or approval of the manuscript.
