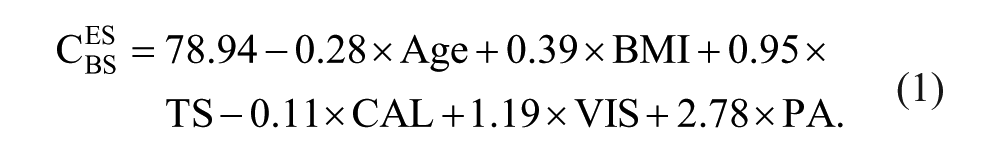Abstract
Background. Balance compensation after vestibular schwannoma (VS) surgery is under the influence of specific preoperative patient and tumor characteristics. Objective. To prospectively identify potential prognostic factors for balance recovery, we compared the respective influence of these preoperative characteristics on balance compensation after VS surgery. Methods. In 50 patients scheduled for VS surgical ablation, we measured postural control before surgery (BS), 8 (AS8) days after, and 90 (AS90) days after surgery. Based on factors found previously in the literature, we evaluated age, body mass index and preoperative physical activity (PA), tumor grade, vestibular status, and preference for visual cues to control balance as potential prognostic factors using stepwise multiple regression models. Results. An asymmetric vestibular function was the sole significant explanatory factor for impaired balance performance BS, whereas the preoperative PA alone significantly contributed to higher performance at AS8. An evaluation of patients’ balance recovery over time showed that PA and vestibular status were the 2 significant predictive factors for short-term postural compensation (BS to AS8), whereas none of these preoperative factors was significantly predictive for medium-term postoperative postural recovery (AS8 to AS90). Conclusions. We identified specific preoperative patient and vestibular function characteristics that may predict postoperative balance recovery after VS surgery. Better preoperative characterization of these factors in each patient could inform more personalized presurgical and postsurgical management, leading to a better, more rapid balance recovery, earlier return to normal daily activities and work, improved quality of life, and reduced medical and societal costs.
Keywords
Introduction
Among vestibular pathologies known to adversely affect balance control, a vestibular schwannoma (VS) is a benign tumor that develops from the Schwann cells surrounding the vestibular nerve, which slowly grows within the internal auditory canal and then into the cerebellopontine angle. The slow, progressive alteration of vestibular function from the tumoral growth allows the gradual implementation of central adaptive mechanisms, called vestibular compensation. This minimizes VS-related symptoms, such as perceptual syndrome with vertigo or dizziness, clinical signs like body and limb deviation, and nystagmus seen in acute vestibular lesions. 1 The surgical tumor removal, which corresponds to a total unilateral vestibular deafferentation (uVD), leads to a decompensation of this previously compensated situation. This causes perturbations in postural control; indeed, most patients report severe vertigo immediately after surgery. Then, a progressive implementation of central adaptive mechanisms leads to vestibular and balance compensations. These are associated with the development of more appropriate sensorimotor and/or behavioral strategies, efference copy/reafference copy reconciliation, and better resolution of sensory conflicts.2,3
Some preoperative individual and VS-related characteristics are known to modify the classical time course of postural compensation, such as age, tumor stage, preoperative preference for visual cues to control balance, preoperative vestibular pattern, and physical activity (PA).4-9 Among these, some play a positive or negative role on balance performance at a given evaluation stage, whereas others modify the time course of balance performance. Although these characteristics that influence postural control after VS surgery have been clearly identified, the weight of each, not in isolation but in the general mapping of the patient, remains unknown. The understanding of the respective contributions of these intrinsic characteristics in postsurgery balance compensation could establish a better prognosis for balance recovery and be helpful for clinicians in determining the best therapeutic follow-up for patients. In this respect, our study aimed to determine the relative weight of certain preoperative individual and VS-related characteristics on the patient’s balance control to predict the balance compensation at short- and medium-term recovery time points (relative to the preoperative state) and the overall postoperative balance recovery. To quantify which preoperative characteristics most influence balance control and its changes (ie, balance compensation) in VS patients who underwent surgical uVD, we performed a series of 6 multiple linear regression analyses. The first 3 regression models (ie, explanatory models) used these preoperative characteristics to estimate the balance control assessed 3 days before surgery (BS), 8 days after surgery (AS8), and 90 days after surgery (AS90). The last 3 regression models (ie, predictive models) used the same preoperative characteristics to estimate the changes in balance control over time between BS and AS8 (ie, short-term balance compensation), BS and AS90 (ie, middle-term balance compensation), and AS8 and AS90 (ie, postoperative balance recovery).
Methods
Participants
The period of recruitment ran from April 2002 to April 2011. Patients meeting eligibility criteria were enrolled in the study. Patients included were those scheduled for a surgical ablation of a unilateral VS without brainstem/cerebellum compression, between 18 and 75 years old, and affiliated to the French social welfare. Patients not included were those with some disorders from the motor and/or somesthetic systems (especially the lower limbs), those with contraindications to the scheduled functional assessments (ear pathology different from VS such as cholesteatoma of the middle ear, tympanic membrane perforation), and those who refused the surgical procedure. During this period, 63 patients gave their written informed consent to participate voluntarily in the study and were followed up during 90 days after surgery. Among these 63 patients, 7 did not perform the tests 8 days after surgery because of postoperative complications (cerebrospinal fluid leak [n = 5], meningitis [n = 1], and neurological disorders [n = 1]) and were excluded from the analyses. Five other participants decided to stop their participation in the study and did not perform the tests 90 days after surgery. Therefore, the final sample was composed of 50 patients (31 women and 19 men; mean age = 50.8 ± 9.8 years; mean height = 1.68 ± 0.09 m; mean weight = 74.9 ± 13.2 kg; and mean body mass index [BMI] = 26.5 ± 4.3 kg/m2).
Our study was approved by an appropriate Human Ethics Committee (Comité de Protection des Personnes de Lorraine).The tests were carried out in the Laboratory for the Analysis of Posture, Equilibrium and Movement at the University Hospital of Nancy (Ministry of Health approval for research). All the patients were submitted to vestibular and posturographic evaluations 3 days BS and twice after surgery, at a short-term point (AS8) and a medium-term point (AS90).
Posturographic Evaluation
Postural regulation, with or without sensory conflicting situations, was analyzed before and after surgery with a sensory organization test (SOT), which was performed on a computerized dynamic posturography platform (EquiTest, Neurocom; Clackamas, OR). During the SOT, the patient’s task was to maintain an upright stance, as stable as possible, during three 20-s trials in 6 conditions that combined 3 visual conditions with 2 platform conditions. During the first 3 conditions, the support was fixed, and the patient’s eyes were open (condition 1 = C1), closed (C2), and open within a sway-referenced visual surround (C3). For conditions 4 to 6, somatosensory information was disrupted by a sway-referenced support, and the patient’s eyes were open (C4), closed (C5), and open within a sway-referenced visual surround (C6). An equilibrium score (ES) was calculated for each condition (C1ES to C6ES), and a composite ES (CES), representing the global balance performance, provided the weighted average of the ES from all the SOT trials.2,10
To quantify the changes in CES values over time, 3 indexes were calculated: (1) the balance compensation at short-term time points (ΔCESsh = [AS8/BS] × 100 − 100), (2) the balance compensation at middle-term time points (ΔCESmid = [AS90/BS] × 100 − 100), and (3) the postoperative balance recovery (ΔCESpost = [AS90/AS8] × 100 − 100). Further methodological details, as supplemental material, are available online.
Predictive Factors of Balance Performance and Balance Compensation
Six potential factors (ie, independent variables) were integrated into the regressive models to predict the balance performances and the balance compensations determined from the CES of the SOT: age, BMI, tumor size (TS), vestibular status (CAL), visual status (VIS), and PAs. The CES value was selected to represent the measure of balance control because it remains a sensitive score to identify the dynamics of the postural control of VS patients over time. 2
TS was measured according to the Koos classification in 4 stages 11 (the higher the stage, the higher the TS or the brainstem damage) and led to the following distribution: stage I (n = 9), stage II (n = 18), and stage III (n = 23). No patient had stage IV tumors. If a patient had had this stage, he/she would not have been included in our study because of any potential brainstem contributions to the postural dysfunction.
Bithermal caloric vestibular testing (CAL) was performed using videonystagmography (Synapsys; Marseille, France), which evaluated the sensitivity of the vestibular reflectivity. 12 BS, 3 types of patterns were detected: 13 patients with an absent response on the side of the affected labyrinth (vestibular areflexy), 27 patients with a decreased response on the side of the affected labyrinth (vestibular hyporeflexy), and 10 patients with a normal symmetrical response (vestibular normoreflexy).
Presurgery visual contribution to balance control (VIS) was determined using static posturography, which was performed with a vertical force platform (Toennies GmbH; Freiburg, Germany). Center of foot pressure displacements were recorded for 20 s, first in an eyes open (EO) and then in an eyes closed (EC) condition. During these conditions, the patient’s task was to remain as stable as possible. The VIS importance was determined using the Romberg’s quotient (Romberg’s quotient = EC/EO). This split the patients into 2 groups: those relying to a lower degree (n = 26 patients) and those relying to a higher degree (n = 24 patients) on visual cues to manage balance control. 7
Each patient provided information about their PA, when the PA occurred, and the frequency and intensity of the exercise.5,13,14 In all, 20 patients were considered physically active. The remaining 30 patients were considered sedentary, neither practicing leisure sports, except for walking, nor working in physically trying conditions.
Further methodological details, as supplemental material, are available online.
Statistical Analysis
The effect of time (BS, AS8, and AS90) on the ES of each condition of the SOT plus the composite ES was assessed using a 1-way analysis of variance (ANOVA) with repeated measures. A probability level of P ≤ .05 was used as an indicator of statistically significant results for overall heterogeneity, and the Bonferroni procedure was applied to pairwise comparisons by adjusting “familywise α” to a significance level of P ≤ .05/3 = .017 in accordance with the 3 possible intragroup comparisons (BS, AS8, and AS90). Explanatory and predictive models were established through multiple linear regression analysis. Postural performance before (CESBS), 8 days (CESAS8), and 3 months (CESAS90) after surgery were the dependent variables for each of the 3 explanatory models. Postural compensation at short (ΔCESsh) and middle (ΔCESmid) term and postoperative balance recovery (ΔCESpost) were the dependent variables for each of the 3 predictive models. For all the models (explanatory and predictive), the 6 predictive factors (age, BMI, TS, CAL, VIS, and PA) were used as independent variables. The quality of all the models was assessed by the coefficient of determination (R2), which expressed the variance explained by all the independent variables and by the F value and the P value (P ≤ .05) computed from an ANOVA. In addition to the regression coefficients that composed the equation of each model, the relative weight of each predictive factor was given by its standardized regression coefficient (β). The adequacy between the dependent variable and each independent variable was assessed by a t test, which tested the null hypothesis of no linear relationship between the dependent variable and each predictive factor. The Bonferroni procedure was applied to adjust the significance level of P ≤ .05/6 = .008 in accordance with the number of independent variables. Finally, we assessed the quality of the significant explanatory/predictive models by a comparison of the measured postural performances and the associated estimated postural performance by means of an independent Student t test (measures vs estimates of 1 model) or 1-way ANOVA (factor = methods: measures, estimates of the explanatory model, estimates of the predictive model).
Results
Postural Performances
The results from the SOT analysis showed significant heterogeneity among the 3 time periods of measurement (BS, AS8, and AS90) for C3ES (P = .019), C4ES (P < .001), C5ES (P < .001), C6ES (P < .001), and CES (P < .001). Pairwise comparisons revealed that C4ES, C5ES, C6ES, and CES were significantly lower at AS8 than at BS and AS90 (Figure 1). For C3ES, there was solely 1 significant difference (P = .011) between AS8 and AS90. At the middle-term time point (AS90), the results for C4ES (P = .005), C5ES (P < .001), C6ES (P < .001), and CES (P < .001) were significantly higher than at the BS time point. The variability (expressed by the standard deviation) of the postural performances was highest for C6ES (24.1%, 30.8%, and 15.8% for BS, AS8, and AS90, respectively), C5ES (19.9%, 26.5%, and 14.8% for BS, AS8, and AS90, respectively), and CES (10.1%, 12.8%, and 7.8% for BS, AS8, and AS90, respectively). Overall, these results demonstrated that the CES was mainly under the influence of the vestibular-related conditions of the SOT (ie, C5 and C6) for the average scores as well as the variability. Therefore, the global score of postural performance (CES) remains a sensitive value to identify the dynamics of the postural control of VS patients over time.
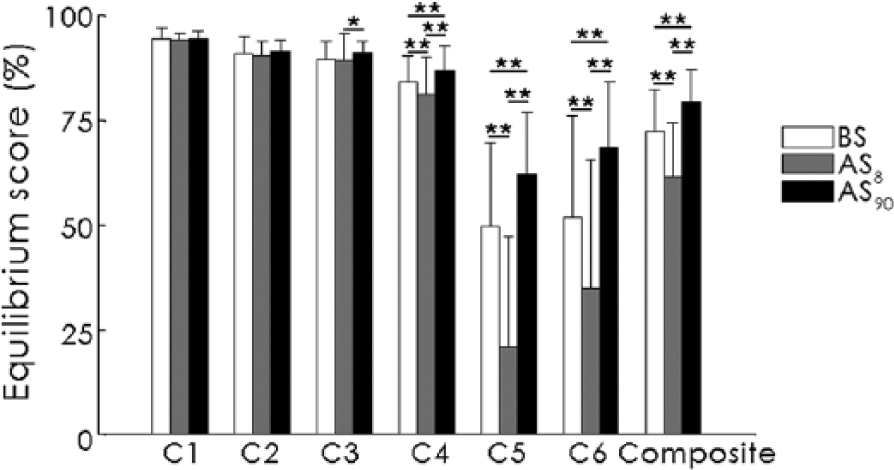
Mean values of the equilibrium scores (%) of the 6 conditions of the sensory organization test and composite score observed before (BS), 8 days after (AS8), and 90 days after (AS90) surgery. *P < .017; **P < .01.
Explanatory Models of Postural Performance
The CESBS from the 6 preoperative factors based on the multiple linear regression model significantly (P = .021) provided 28% of the variance (R2 = 0.28), as follows:
Among the 6 preoperative factors, CAL alone (P = .006) had a significant relative weight (Table 1) and accounted for approximately 59% of the R2 of the model.
Relative Weight (Expressed by the Standardized Coefficient β) of the 6 Preoperative Factors of the 3 Models to Explain the Postural Performance Before (CESBS), 8 Days After (CESAS8), and 3 Months After (CESAS90) Surgery.
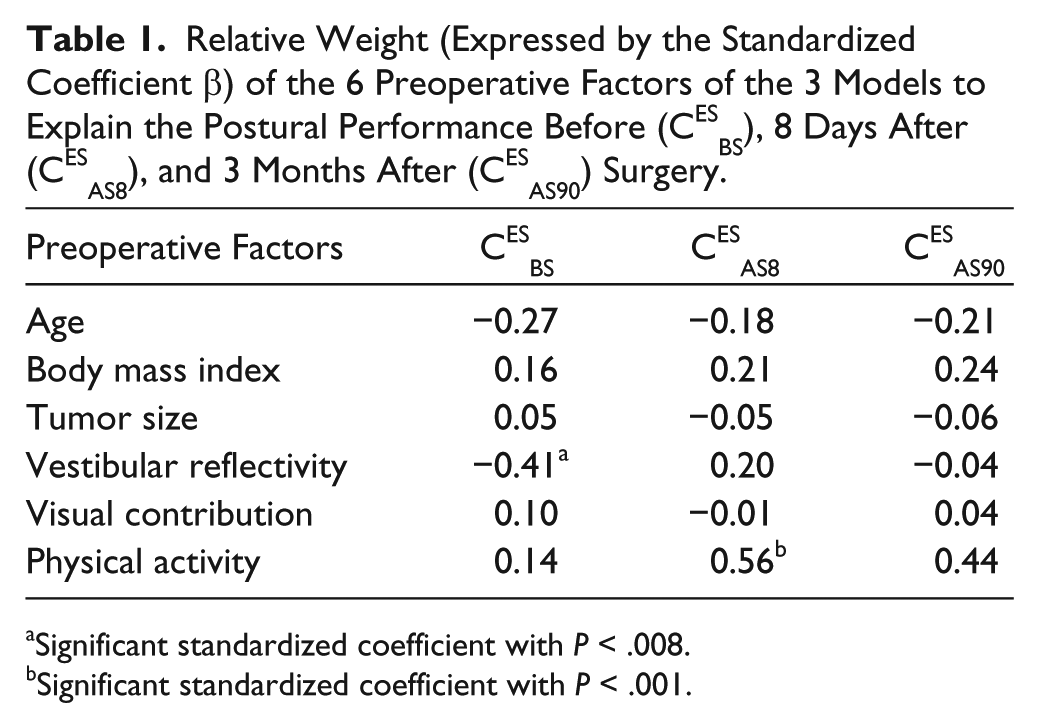
Significant standardized coefficient with P < .008.
Significant standardized coefficient with P < .001.
The CESAS8 from the 6 preoperative factors based on the multiple linear regression model significantly (P < .001) contributed to 48% of the variance, as follows:
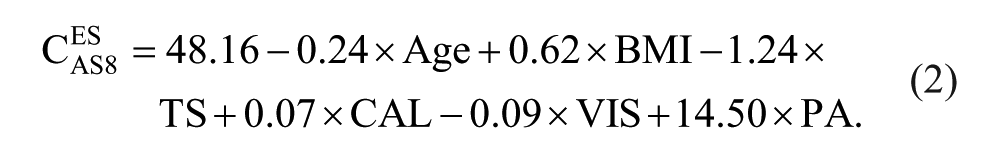
PA alone had a significant relative weight (P < .001; Table 1) and accounted for approximately 80% of the R2 of the model.
The CESAS90 from the 6 preoperative factors based on the multiple linear regression model significantly (P = .021) contributed to 28% of the variance, as follows:
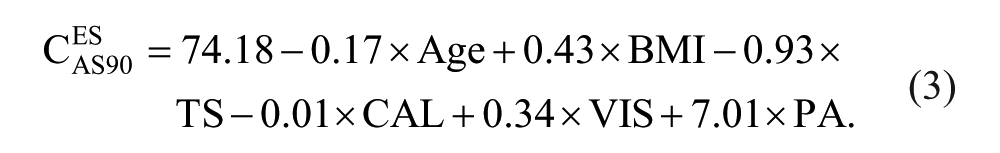
None of the 6 preoperative factors had a significant relative weight.
Predictive Models of Balance Compensation and Postoperative Postural Recovery
The prediction of the ΔCESsh from the multiple linear regression model based on the 6 preoperative factors significantly (P < .001) provided 55% of the variance (Figure 2), as follows:
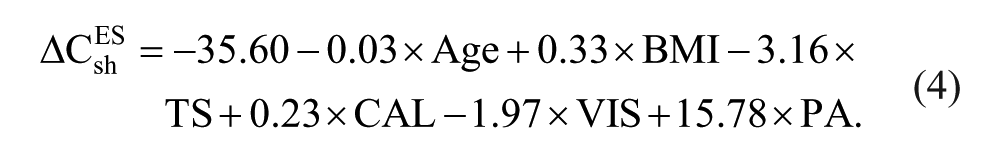
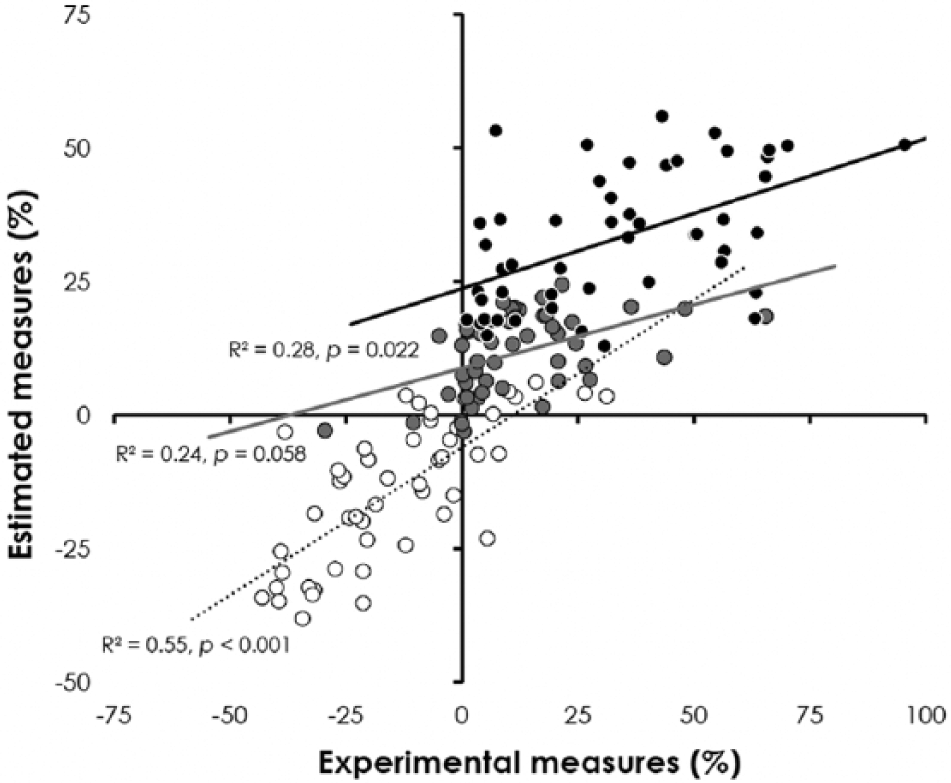
Scatter plots of the estimated measures from the predictive models as a function of the experimental measures. White circles correspond to short-term postural compensation (ΔCESsh), gray circles to middle-term postural compensation (ΔCESmid), and black circles to postoperative balance recovery (ΔCESpost). The quality of the estimation and its significance are expressed by the R2 and P value, respectively.
PA (P < .001) and CAL (P = .003) had significant relative weights (Table 2). Indeed, these were the 2 most important variables for predicting the ΔCESsh, comprising approximately 59% and 36% of the R2 of the predictive model, respectively.
Relative Weight (Expressed by the Standardized Coefficient β) of the 6 preoperative factors of the 3 Predictive Models of the Postural Compensation at Short- (ΔCESsh) and Medium- (ΔCESmid) Term Time Points and the Postoperative Postural Recovery (ΔCESpost).
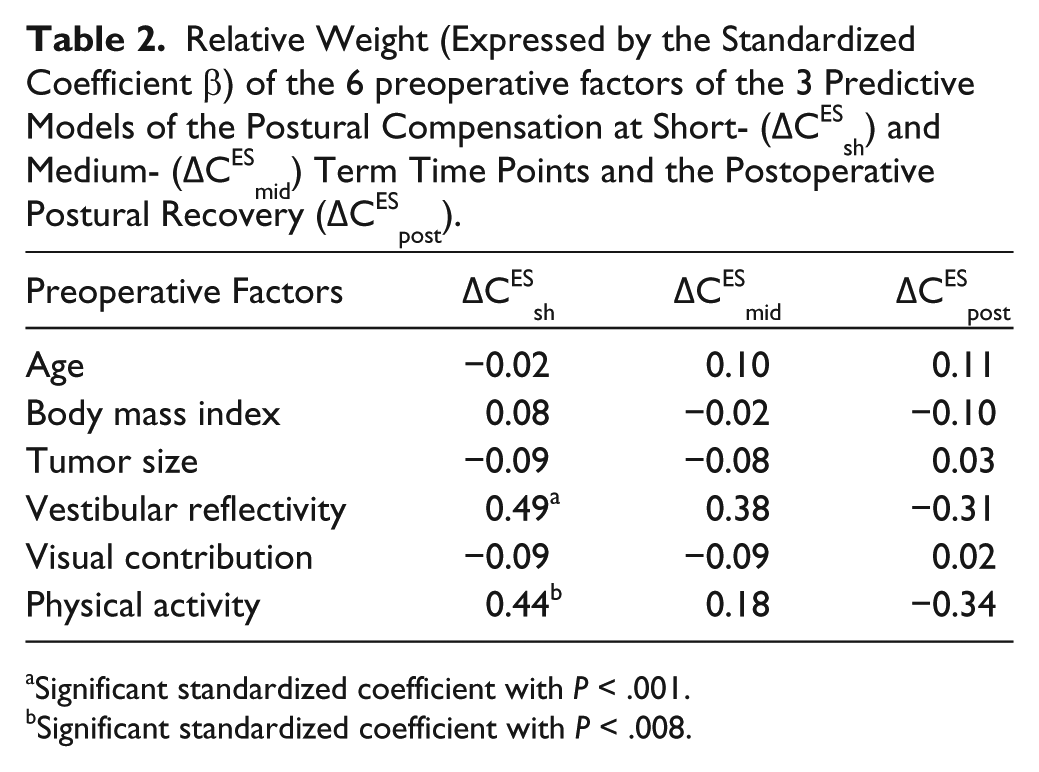
Significant standardized coefficient with P < .001.
Significant standardized coefficient with P < .008.
The predictive model of the ΔCESmid failed to achieve any significant (P = .058) estimation, with a low explained variance (R2 = 0.24, Figure 2), as follows:
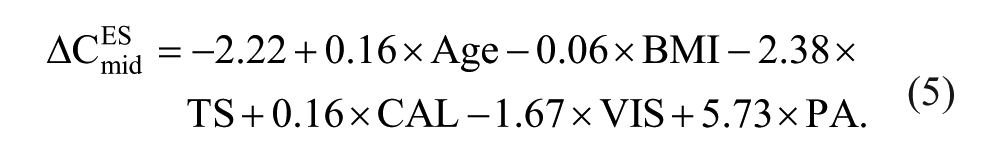
Therefore, none of the 6 preoperative factors had a significant relative weight.
The prediction of the ΔCESpost from the multiple linear regression model based on the 6 preoperative factors significantly (P = .022) contributed to 28% of the variance (Figure 2), as follows:
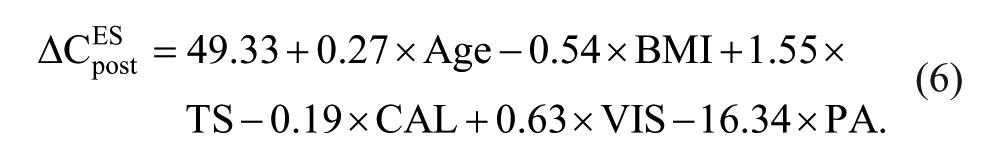
However, none of the 6 preoperative factors had a significant relative weight.
Measured Versus Estimated Postural Performances
There was no significant (P = .986) difference between the measured CESBS (72.15% ± 5.39%) and the estimated CESBS (72.18% ± 10.13%) from the explanatory model (Equation 1). Furthermore, there was no significant main effect of the method on the CESAS8 and CESAS90 (P = .981 and P = .837, respectively). The measured CESAS8 was 62.00% ± 11.68%; the estimate from the explanatory model of CESAS8 (Equation 2) was 61.63% ± 8.80%; and the estimate from the predictive model of the short-term postural compensation (Equation 4) was 61.63% ± 12.77%. The measured CESAS90 was 80.88% ± 12.91%; the estimate from the explanatory model of CESAS90 (Equation 3) was 79.45% ± 4.17%; and the estimate from the predictive model of the postoperative recovery (Equation 6) was 79.45% ± 7.84%.
Discussion
Our study demonstrated that preoperative intrinsic characteristics, such as those related to the individual or vestibular status, were predictive factors of balance control before and early after surgery. The postural performances that we examined were accounted for by 2 main factors: vestibular status and PA, with a differential contribution according to the evaluation stage. Whereas vestibular status was the main predictive factor of global balance performance BS, PA was the main factor at short- (AS8) term during the recovery process. These 2 factors significantly contributed to the short-term balance compensation (ie, the variation in the postural performances: ΔCESsh).
Major Role of Vestibular Status and PA
Our study revealed that the greater the preoperative vestibular asymmetry, the worse the preoperative postural performances and the higher the short-term balance compensation. This was in line with a previous study of Parietti-Winkler et al, 8 which showed that the preoperative vestibular status could modify the postsurgery postural compensation. A high vestibular asymmetry (eg, preoperative vestibular areflexy determined from the videonystagmographic caloric test) led to an implementation of central vestibular compensatory mechanisms, which developed even BS with the growth of the tumor. These compensatory mechanisms were modified by surgery-related decompensation after tumor removal, but the neural networks remained present and could serve as a neuroanatomical support to balance compensation. 8 In contrast, the normoreflexy status did not lead to the preoperative implementation of new neural networks because of a lack of sufficient asymmetry to induce the compensation-related mechanisms. In this respect, uVD induced a sudden and huge asymmetry that led to high balance disturbances.
Our study has also shown a strong association of PA with both the postoperative short-term balance performances and the time course of its short-term compensation. The PA led to training of the postural control system BS. PA allowed management of more difficult tasks, both in terms of pure biodynamics and feed-forward models and in training of an individual’s feedback during the disease. 5 Therefore, PA can enhance the motor learning necessary for the neural remodeling generated by vestibular compensation. The development of new neural networks with training 15 would favor the implementation of new, more efficient sensorimotor and behavioral strategies, providing better and faster postural compensation. By contrast, physical inactivity made the consequences of surgery more difficult to manage in the acute stage, even though it did not prevent compensation at later time points. We propose that preoperative PA promotes the neuroplasticity of neural networks involved in motor learning. Therefore, preoperative PA could not only enhance the postoperative quality of life and speed up the recovery of postural performance, but also reduce medical management and societal cost, with a faster and easier return to daily-life activities and work.
Keeping in mind these 2 important factors, an emerging treatment plan can be proposed and would occur prior to VS surgery. Indeed, it is interesting to note that our 2 significant predictive factors of short-term balance compensation are perfectly in line with the paradigm proposed in previous studies.16-19 They showed that a concomitant preoperative vestibular rehabilitation (PREHAB) with a preoperative uVD induced by aminoside intratympanic injection in patients with remaining vestibular function improved postural performances, both at short- and medium-term time points after surgery. A preoperative vestibular suppression before VS surgery and/or a vestibular training should enhance postoperative recovery and quality of life for patients. According to the rationale of a vestibular PREHAB of these previous studies,16-19 the current results support the hypothesis that learning processes might be initiated and promoted by means of challenging sensory mismatch exercises (eg, visual fixation during slow rotations or visual target changes, optokinetic stimulation), which could improve the recalibration of the vestibular reflexes 18 and might, thus, limit the early postoperative decrease in postural performance as already identified in a long-term perspective. 19 In addition, the galvanic vestibular stimulation might be a promising rehabilitation technique to enhance perceptual deficits 20 and improve vestibular rehabilitation 21 and balance recovery 22 by centrally stimulating the vestibular system. 23 Concomitant with a vestibular PREHAB, repeated galvanic vestibular stimulations would be an appealing framework to reduce the short-term nasty consequences of the surgical uVD and to speed up balance recovery.
Limited Role of Age and Visual Status
In a previous study using univariate analyses, patients >60 years old had better postural compensation compared with patients <60 years old. 4 The age-related decline in postural control and cognitive processes did not impair the postural compensation mechanisms. Indeed, the surgery-related neuroplasticity allowed not only recovery but an improvement in balance after surgical VS removal in the elderly. 4 Nevertheless, our present study using multivariate analyses revealed that age was not a critical factor in the postural outcome of VS surgery. In agreement with our result, Samii et al 24 did not observe an age effect on the surgery outcome or on the postoperative quality of life. Therefore, we do not consider age as a risk factor for disabilities after VS surgery.
Similarly, high visual dependency BS has previously been shown to alter balance control early after uVD and to delay short-term balance compensation. 7 However, our present study with multivariate analysis showed that visual dependence was not a critical factor conditioning postural performances or its compensation after VS surgery. One can hypothesize that the visual clues contributed less than the neural plasticity related to the vestibular and balance compensations or those promoted by PA, in the balance recovery after a uVD.
Limitations and Perspectives
The first limitation of our study is the quality of our models, which explained from 24% to 55% of the variance, suggesting that other preoperative factors have yet to be determined. Nonetheless, the comparison between the measured postural performances and the estimated ones showed that the models are adequate in their current state. However, elucidation of further preoperative factors will improve these predictive models. For instance, emotional and psychological factors are known to influence balance regulation and, in this way, could also play a role in the quality of postoperative recovery. 25 Another limitation of our study is the lack of inclusion of other neuro-otological tests. The caloric test focuses on the low-frequency range of the vestibulo-ocular reflex and the semicircular canal solely, but the otolith function is not tested. Head impulse test and vibration-induced nystagmus assess dynamic semicircular canal function, and vestibular evoked myogenic potentials assess the otolith function.26,27 Collectively, they provide critical information on vestibular function before and after uVD, and future prospective studies are needed to increase our ability to predict balance control and vestibular compensation in VS patients after uVD.
Conclusions/Implications
Our study demonstrated that preoperative PA and vestibular status are prognostic for short-term balance compensation after VS surgical removal. A better preoperative characterization of the patient and their vestibular function will allow a better prediction of postural control recovery after surgery. The PA appears to have a protective effect against impairment of postoperative postural performances. Also, preoperative asymmetric vestibular function seems to be a suitable factor because it allows a faster short-term postural recovery after surgery. A systematic preoperative characterization of these factors in each patient could lead to higher and faster balance recovery, earlier return to normal daily activities and work, improvement in quality of life, and decrease in medical and societal costs.
Footnotes
Acknowledgements
The authors are sincerely grateful to Dr Benoîte Lassalle-Kinic and Dr Hadrien Ceyte for the meaningful advice and discussions on the drafts of this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.