Abstract
Background
The potential for aerobic exercise (AE) to enhance neuroplasticity post-stroke has been theorized but not systematically investigated. Our aim was to determine the effects of forced-rate AE (FE) paired with upper extremity (UE) repetitive task practice (FE + RTP) compared to time-matched UE RTP (RTP only) on motor recovery.
Methods
A single center randomized clinical trial was conducted from April 2019 to December 2022. Sixty individuals ≥6 months post-stroke with UE hemiparesis were randomized to FE + RTP (N = 30) or RTP only (N = 30), completing 90-minute sessions, 3×/week for 8 weeks. The FE + RTP group underwent 45-minute of FE (5-minute warm-up, 35-minute main set, and 5-minute cool down) followed by 45-minute of UE RTP. The RTP only group completed 90-minute of RTP. Primary outcomes were the Fugl-Meyer Assessment (FMA) and Action Research Arm Test (ARAT). The 6-minute Walk Test (6MWT, secondary outcome) assessed walking capacity.
Results
Sixty individuals enrolled and 56 completed the study. The RTP only group completed more RTP in terms of repetitions (411.8 ± 44.4 vs 222.8 ± 28.4, P < .001) and time (72.7 ± 6.7 vs 37.8 ± 2.4 minutes, P < .001) versus FE + RTP. There was no significant difference between groups on the FMA (FE + RTP, 36.2 ± 10.1-44.0 ± 11.8 and RTP only, 34.4 ± 11.0-41.2 ± 13.4, P = .43) or ARAT (FE + RTP, 32.5 ± 16.6-37.7 ± 17.9 and RTP only, 32.8 ± 18.6-36.4 ± 18.5, P = .88). The FE + RTP group demonstrated greater improvements on the 6MWT (274.9 ± 122.0-327.1 ± 141.2 m) versus RTP only (285.5 ± 160.3-316.9 ± 170.0, P = .003).
Conclusions
There was no significant difference between groups in the primary outcomes. The FE + RTP improved more on the 6MWT, a secondary outcome.
Trial Registration:
ClinicalTrials.gov: NCT03819764.
Introduction
Advances in the medical management of stroke have improved survival rates by nearly one-third over the past 20 years; however, disability rates post-stroke have not changed substantially. 1 Despite extensive resources dedicated to rehabilitation, nearly two-thirds of survivors do not regain full use of their hemiparetic upper and lower extremities resulting in the inability to use the more affected hand, deficits in locomotion and mobility, and decreased community reintegration.2,3 Neuroplasticity, the ability of the central nervous system (CNS) to change in response to stimuli by strengthening existing and forming new neural connections, remains the tenet of motor recovery post-stroke.1,2,4,5 Motor learning-based therapies to drive the recovery of upper extremity (UE) function are considered the standard of care in post-stroke rehabilitation and include constraint-induced movement therapy and repetitive task practice (RTP).1,2,5-10 Clinical trials in the past 2 decades have focused on variations to these approaches of motor retraining therapies7,10,11 and titrating the optimal dosage to drive neural recovery.8,9 However, the optimal method to facilitate UE motor recovery has not been determined and the causal relationship between dose and recovery remains unclear.3,9,10
Principles of motor recovery indicate that neuroplasticity is experience-dependent, requiring motor task practice to improve function. Unfortunately, the dose of therapy most individuals post-stroke receive does not meet a level of practice necessary to realize the full recovery potential.6,8 There is emerging evidence to indicate that aerobic exercise (AE) improves brain function and promotes neuroplasticity.4,12-17 Studies in animals and humans indicate that AE can increase levels of brain-derived neurotrophic factor and insulin-like growth factor-1, neurotrophins thought to be key facilitators of neuroplasticity, implicated on a molecular level to encourage axonal and dendritic growth and remodeling, synaptogenesis, and synaptic transmission efficacy.2,4,13,14,17,18 Numerous systematic reviews of animal and human literature have found that exercise intensity is an important factor in upregulating neurotrophins, with higher intensities eliciting greater responses compared to low-intensity AE approaches.13,14,16,17,19-22
To harness the potential neuroplastic priming effects of AE, studies conducted in animals and neurologically healthy humans indicate that AE administered immediately prior to motor task practice enhances motor skill acquisition. 18 Our underlying hypothesis is that AE primes the CNS to facilitate neuroplasticity and its use in combination with motor task practice will optimize motor recovery.4,13,15 To evaluate this hypothesis, we conducted successful preliminary studies suggesting AE training can enhance the motor learning benefits associated with UE RTP in individuals with stroke.23,24 However, a barrier is that persons post-stroke often present with considerable declines in cardiovascular fitness and endurance, which necessarily limits the duration and intensity of AE. To overcome this deconditioning, we utilized a forced-rate exercise (FE) paradigm, an approach in which the voluntary efforts of participants are supplemented to facilitate exercise of greater intensity and duration to trigger potential neuroplastic benefits. During FE, pedaling cadence on a stationary cycle is supplemented by a motor to assist, but not replace, the voluntary efforts of the individual. We previously reported that when comparing FE to voluntary-rate exercise (VE), FE elicited greater improvements in motor recovery associated with UE RTP.23,24
While our pilot studies demonstrated superiority of FE, the dose–response relationship with UE RTP was not evaluated. The aim of this project was to determine the effects of FE paired with an abbreviated session of UE RTP (FE + RTP) compared to time-matched UE RTP (RTP only) on the recovery of motor function in individuals with chronic stroke. We hypothesized that FE + RTP would yield greater improvements in UE motor recovery compared to time-matched RTP only.
Materials and Methods
A single-center, parallel group, rater blinded randomized clinical trial with a 1:1 allocation ratio was conducted at the Cleveland Clinic between April 2019 and December 2022. The trial was funded by the National Institutes of Health (K01HD092556), registered prospectively at ClinicalTrials.gov (NCT03819764) and conducted in accordance with the Consolidated Standards of Reporting Trials (CONSORT) guidelines.
Participants and Screening
Sixty individuals were recruited from the community via a stroke research participant registry, clinician outreach, and support group education sessions. Inclusion criteria were: (1) ≥6 months since a single ischemic or hemorrhagic stroke; (2) UE Fugl-Meyer motor score 19 to 55; and (3) ambulatory ≥20 m with no more than contact guard assistance; and (4) 18 to 85 years of age. Exclusion criteria were: (1) hospitalization for myocardial infarction, heart failure, or heart surgery within 3 months, (2) cardiac arrhythmia, (3) hypertrophic cardiomyopathy, (4) severe aortic stenosis, (5) pulmonary embolus, (6) significant contractures, (7) anti-spasticity injection to paretic UE within 3 months of enrollment, and (8) other contraindications to exercise. The study was approved by the Cleveland Clinic Institutional Review Board and the informed consent process was completed with all participants.
After meeting initial screening criteria, 25 participants underwent cardiopulmonary exercise (CPX) testing administered by an exercise physiologist blinded to group allocation at baseline and end of treatment (EOT) to determine baseline function and cardiopulmonary response to maximum exertion. Participants completed testing while on all prescribed medications including anti-hypertensives to obtain an accurate measure of heart rate (HR) and blood pressure response. A 12-lead electrocardiogram was assessed prior to exercise and monitored continuously throughout exercise and recovery. An individualized ramp protocol was employed on an electronically controlled Lode cycle ergometer (Lode, Groningen, The Netherlands) using MedGraphics CardioO2/CP system (MCG Diagnostics, St. Paul, MN) with the ramp set to elicit a test duration of approximately 10 minutes. Participants were encouraged to exercise until volitional fatigue or standard test termination criteria according to the American College of Sports Medicine (ACSM) Guidelines for Exercise Testing and Prescription. 26 To capture gas analyses during steady state, breath by breath samples were collected and data were averaged during the last 30 seconds of each stage. Peak oxygen consumption (Peak VO2) was defined as the highest average collected. Results were interpreted by the study cardiologist blinded to group allocation to determine participant safety.
Outcomes
Clinical outcomes evaluating UE motor recovery and walking capacity were obtained at baseline, EOT, and 4 weeks following EOT (EOT + 4) by the same trained physical therapist blinded to group allocation. The primary impairment-based motor outcome was the Fugl-Meyer Assessment (FMA), 27 which evaluates the ability of the individual to move out of synergistic patterns toward isolated movement control. 28 The primary functional-based motor outcome was the Action Research Arm Test (ARAT) total score. 29 The ARAT includes 19 tests of fine and gross motor function, grouped in subscales of grasp, grip, pinch, and gross movement. The ARAT subscale scores were secondary outcomes. The Wolf Motor Function Test served as secondary motor outcome. 30
The 6-minute walk test (6MWT) was a secondary outcome administered along a continuous 100-m oval lap as a measure of walking capacity according to methods described by Eng et al. 31 Specifically, participants were instructed to: “walk as far as possible around the oval path within 6 minutes and do not stop unless you need to.” 31 Assistive devices and orthoses as prescribed for community ambulation were used, with equipment remaining consistent across testing time points.
Interventions
Following baseline testing, participants (N = 60) underwent concealed randomization stratified by baseline FMA level (≤33 or >33) using a nonreplenished sealed opaque envelope pull method to 1 of 2 time-matched groups: (1) forced-rate AE and RTP (FE + RTP, N = 30) or (2) RTP only (N = 30). Randomization was conducted by the research therapist who was not involved in the collection of outcomes. Participants in both groups completed 24 visits, 3× per week for 8 weeks, and were permitted up to 12 weeks to complete the 24 visits to allow for vacations, illness, or holidays. Study visits for both groups were matched at 90 minutes in length. Participants were asked to refrain from formal therapy or enrolling in other trials until after the 4-week follow up. All were taught specific strategies and advised to incorporate their more affected UE into daily tasks. Those in the FE group did not have access to a FE bike following the 8-week intervention.
Forced Exercise + Repetitive Task Practice
Participants in the FE + RTP group completed a 45-minute session of supervised AE on a custom-engineered motor-assisted stationary semi-recumbent cycle ergometer, followed by 45-minutes of UE RTP. Participants were instructed to exercise within their target HR zone during the 35-minute main exercise set, which occurred between a 5-minute warm-up and 5-minute cool-down. Target HR range was individualized using the Karvonen formula at 60% to 80% of peak HR based on CPX testing. 32 Heart rate was monitored and recorded continuously using a Wahoo chest strap (Wahoo Fitness, Atlanta, GA), synced and displayed continuously on an Apple iPad (Apple, Inc, Cupertino, CA), allowing the therapist and participant to monitor aerobic intensity, facilitating adherence to prescribed values. Participants were instructed to actively pedal with the motor to achieve target HR response. Cycling cadence was programmed by the therapist based on the individual’s ability to contribute to the workload with a target cadence of ≥75 revolutions per minute (RPM), based on results from previous studies indicating better outcomes at higher cycling cadence. 23 Exercise intensity was progressed by encouraging a higher aerobic response, increased power output, or increased cycling cadence. Given that cycling cadence was programmed and driven by the motor, the therapist monitored the individual’s ability to contribute to the workload when progressing cycling cadence, ensuring that the motor was not passively spinning the participant’s legs. Average HR during the warm-up, main set, and cool down were recorded. Blood pressure (BP) and rating of perceived exertion were recorded every 10 minutes. Training was conducted under the supervision of a physical therapist or physical therapist assistant certified in Basic Life Support. Participants completed a 45-minute session of UE RTP immediately following their FE session.
Repetitive Task Practice
RTP emphasizes highly-repetitious UE tasks that are functional, goal-oriented, and relevant to the individual, administered using blocked practice. 33 The RTP approach was identical to our previous studies,23,24 originally modeled after Birkenmeier and colleagues.6,9,34 Tasks were individualized to each participant’s goals and abilities and included gross and fine motor components involving proximal and distal motor control; for example, reaching to a shelf positioned at knee height, grasping a plastic cup, and placing it onto a shelf positioned at shoulder height. Task difficulty was progressed according to motor control principles and guided by the manual of Upper Extremity Task Specific Training by Birkenemeier and Lang. 35 Specific strategies included increasing the range of movement of the task, adding weight to the object or limb, modifying the size, shape, or pliability of the object, and increasing the complexity of fine motor tasks from gross motor to include object manipulation. 35 The RTP activities were administered by a licensed neurologic physical therapist or physical therapist assistant who tailored each task to ensure appropriate difficulty. During a 45-minute RTP session, participants completed 3 to 5 tasks, targeting 60 to 90 repetitions of each task. Time spent completing tasks and number of repetitions were recorded. The RTP intervention was administered in the same manner for both groups; however, to ensure a time-matched intervention, the FE + RTP group participated in a 45-minute session immediately after 45-minute of FE, while the RTP only group completed 2 sequential 45-minute RTP sessions.
Statistical Analysis
The power and sample size calculation was based on FMA scores from our pilot study. 24 Assuming a minimal clinically important difference (MCID) value for the FMA of 4.25 points 36 and an increase of 2 × MCID for the FE + RTP group and 1 × MCID for the RTP only group, an n = 30 per group provided a 0.87 power at the .05 significance level to detect pairwise group differences equivalent to an effect size of 0.4.
Intention to treat analysis was conducted. Participant demographics, clinical characteristics, intervention parameters, and outcomes were summarized by mean with standard deviation or median with interquartile range for continuous variables, and count with percentage for categorical variables.
Mixed-effects models were used to model outcomes over time. The primary outcome of FMA was measured at baseline, mid-point, EOT, and EOT + 4. All other outcomes were measured at baseline, EOT, and EOT + 4, which were dependent variables. Independent variables included time point (baseline, mid-point as applicable, EOT, and EOT + 4), group, and interaction between time points and group. Subject random effect was included in the model. Interaction term was excluded if it was non-significant (P > .05). There were 5 primary hypotheses of interest which were evaluated in contrast statements in the mixed effects models: group comparisons of change from baseline for FMA at mid-point, EOT, and EOT + 4 and for ARAT Total Score at EOT and EOT + 4. For these 5 primary hypotheses, a Bonferroni correction was applied with significance set at P < .01.
A responder analysis was conducted by determining the number of participants with change scores that exceed the MCID from baseline to EOT, and baseline to EOT + 4, for FMA, ARAT, and 6MWT. The entire distribution of responses for FE + RTP and RTP only groups were graphed using a cumulative distribution function. 37
Statistical analyses were conducted using SAS version 9.4 (SAS Institute Inc, Cary, NC) with statistical significance established throughout at P < .05 for secondary analyses.
Results
Sixty participants met participation criteria, were consented, and enrolled. Participants were 60.5 (±10.6) years of age and 58.3% male. Racial distribution included 78.3% White, 20.0% Black/African American, and 1.7% Asian. Median time since stroke was 26.0 [13.0, 69.0] months. Detailed demographics, which were similar across groups, are presented in Table 1. Both interventions were well-tolerated by participants, with an overall retention of 93.3% and no serious adverse events related to study interventions. Twenty-nine participants in the FE + RTP group and 27 in the RTP only group completed the intervention and EOT testing. Two FE + RTP participants impacted by the mandatory COVID-19 shutdown completed 20 and 21 sessions rather than the planned 24 sessions. The study team decided to conduct EOT testing immediately prior to the shutdown to preserve outcomes from their participation. Two additional participants randomized to FE + RTP completed only 2 and 6 sessions prior to the shutdown. Both resumed the intervention 10 and 12 weeks later, restarting with session 1. The CONSORT flow diagram is shown in Figure 1.
Demographics, Clinical Characteristics, and Intervention Parameters of the Study Sample, Stratified by Groups.
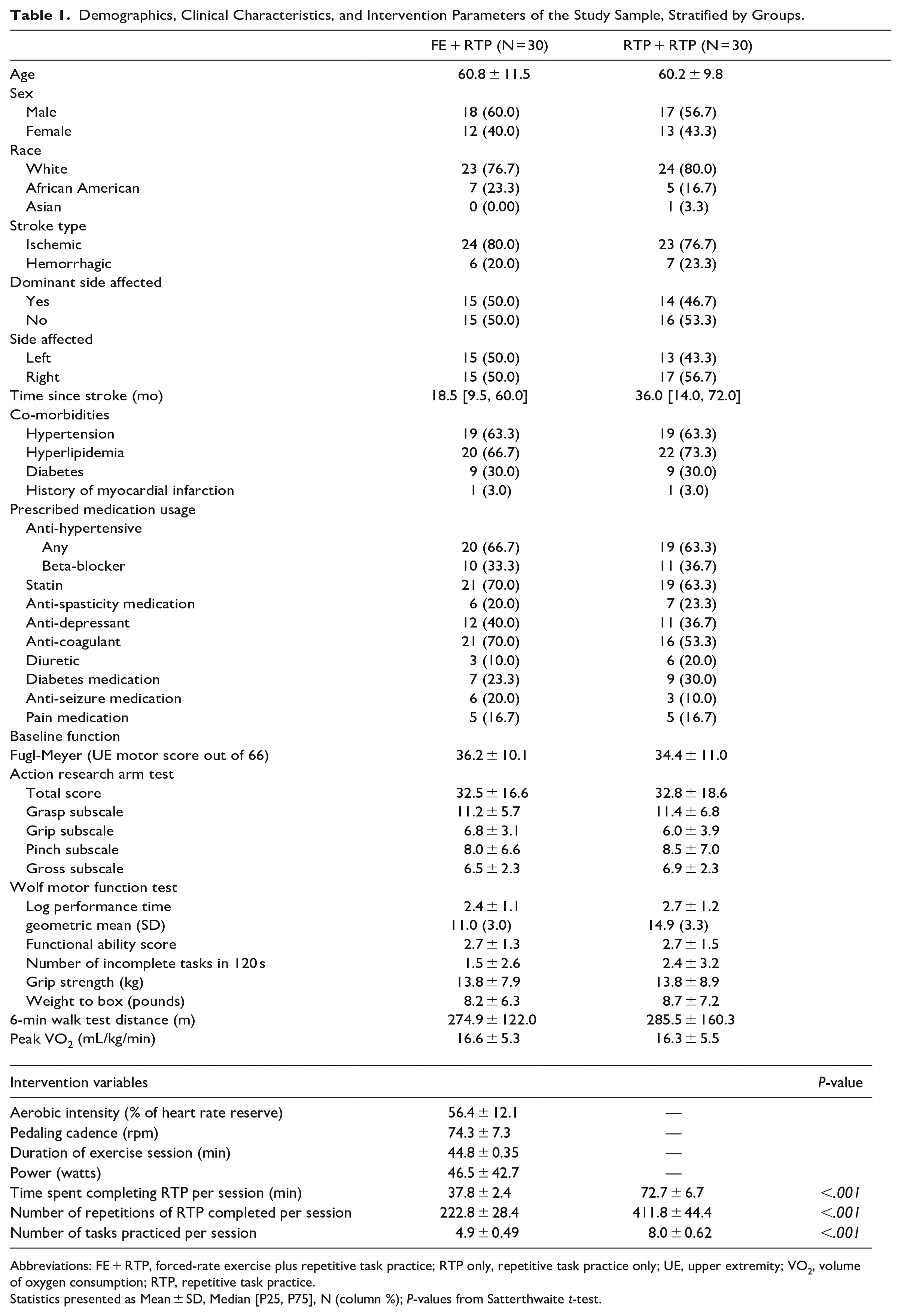
Abbreviations: FE + RTP, forced-rate exercise plus repetitive task practice; RTP only, repetitive task practice only; UE, upper extremity; VO2, volume of oxygen consumption; RTP, repetitive task practice.
Statistics presented as Mean ± SD, Median [P25, P75], N (column %); P-values from Satterthwaite t-test.
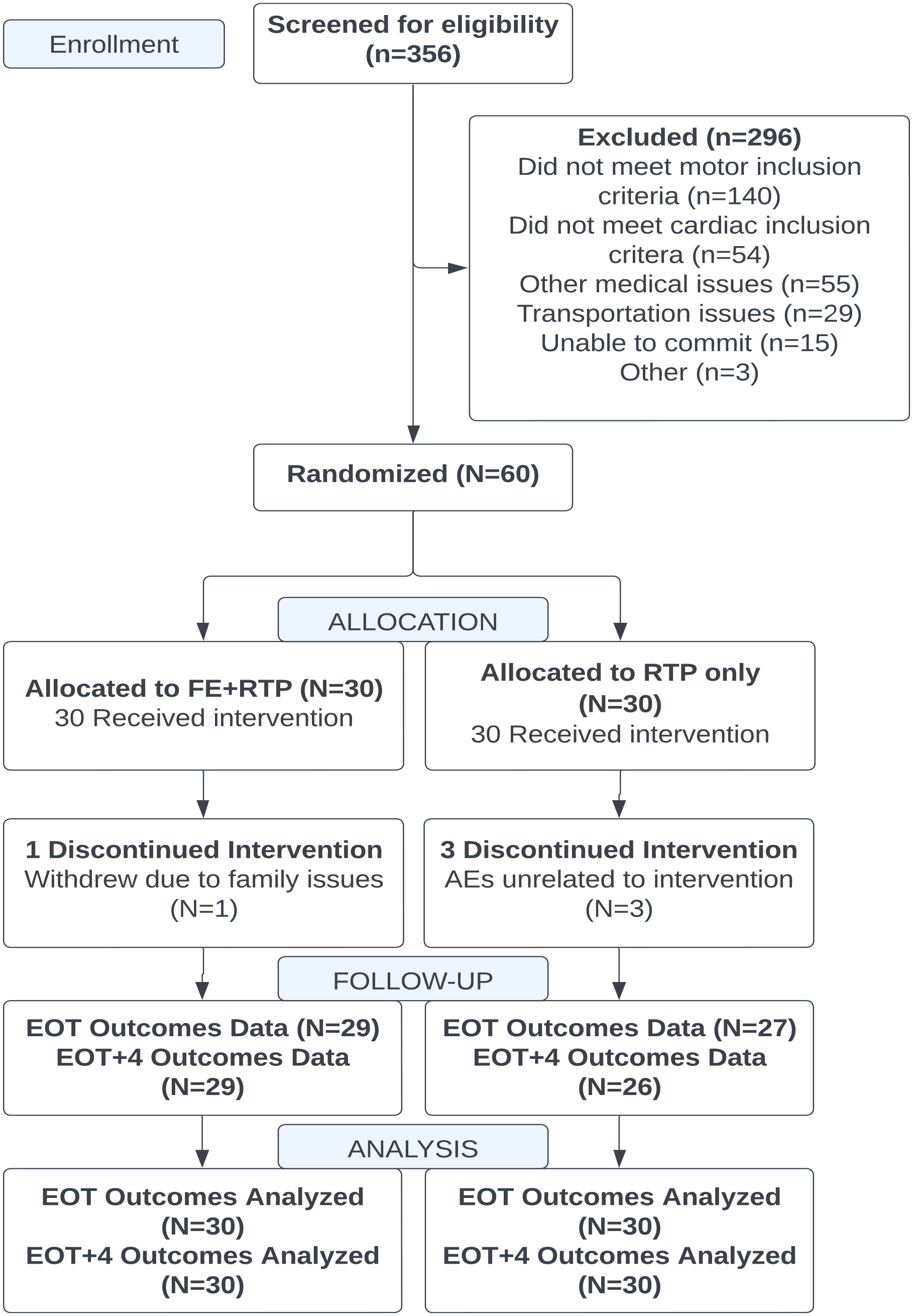
CONSORT flow diagram.
Intervention Variables
Average FE session duration was 44.8 (±0.35) minutes, aerobic intensity was 56.4 (±12.1) percent of heart rate reserve (HRR), power output was 46.5 (±42.7) watts, and cycling cadence was 74.3 (±7.3) RPM (Table 1). Mean rating of perceived exertion for the FE group using Borg’s category ratio 10-point scale 38 was 3.6 at 10 minutes, 4.1 at 20 minutes, 4.5 at 30 minutes, and 4.7 at 40 minutes, indicative of “moderate” (3) to “somewhat hard” (4) or “hard” (5) perceived exertion. As expected, those in the RTP only group, compared to the FE + RTP group, completed nearly twice the repetitions of RTP (411.8 ± 44.4 vs 222.8 ± 28.4, P < .001) and spent more time completing RTP (72.7 ± 6.7 minutes vs 37.8 ± 2.4 minutes, P < .001).
Given the paucity of data on FE in persons post-stroke, BP was monitored to ensure that exercise was occurring within safe cardiovascular parameters. Systolic and diastolic BP measurements of 200 and 100 mmHg, respectively, were established thresholds for which a FE session was paused. Sessions resumed after BP dropped to below these pre-determined thresholds. There were incidences in 4 participants when BP rose to above threshold values and a brief rest break was provided prior to resuming and completing the exercise session. This was often related to the individual forgetting to take their BP medication, running out of medication, or ceasing to take it against medical advice. These episodes allowed for study staff to monitor and educate patients on the importance of medication adherence, particularly to anti-hypertensives in individuals with a history of stroke.
Upper Extremity Motor Outcomes
Group effects were not significant for the FMA or ARAT in contrast statements for any time-point comparison (Figure 2). Significant improvements in UE motor recovery (Table 2) were measured for both groups from baseline to mid-point in the FMA (FE + RTP, 36.2 ± 10.1-40.4 ± 11.3 and RTP only, 34.4 ± 11.0-37.8 ± 11.7, P < .001), baseline to EOT in the FMA (FE + RTP, 36.2 ± 10.1-44.0 ± 11.8 and RTP only, 34.4 ± 11.0-41.2 ± 13.4, P < .001) and baseline to EOT in the ARAT (FE + RTP, 32.5 ± 16.6-37.7 ± 17.9 and RTP only, 32.8 ± 18.6-36.4 ± 18.5, P < .001).
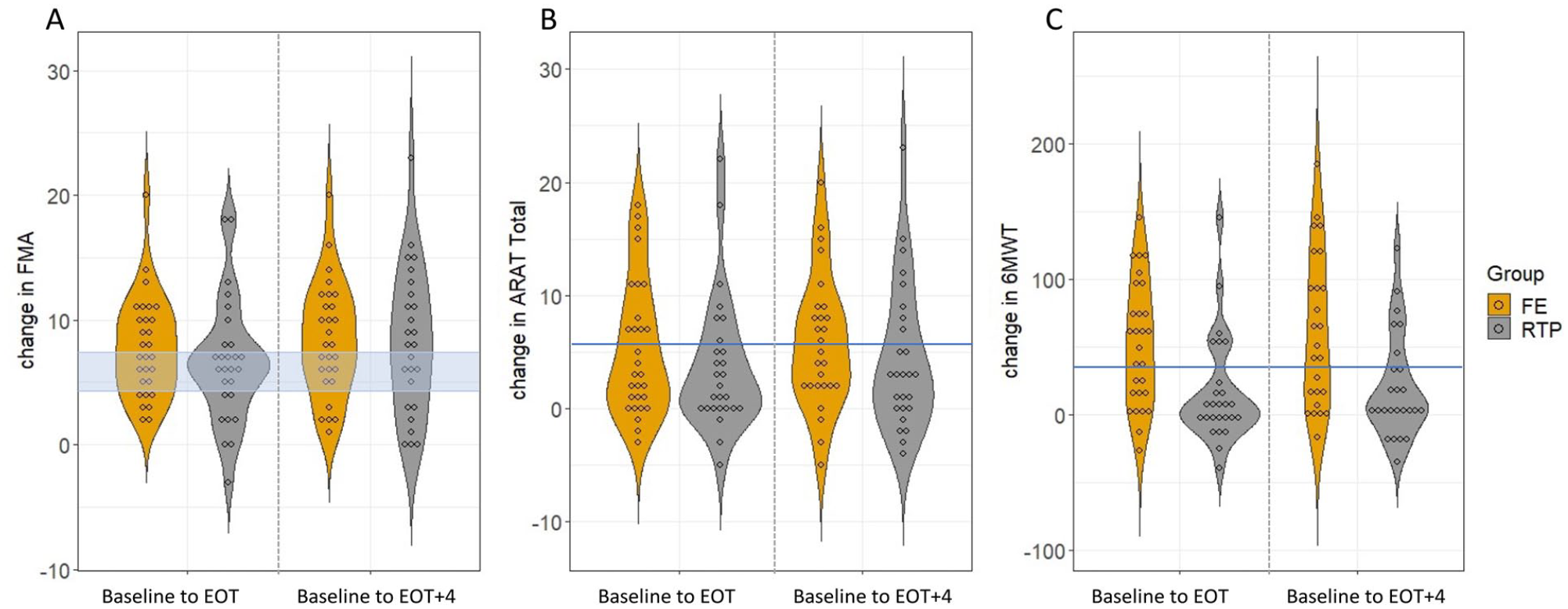
Violin dot plots depicting change scores from baseline to end of treatment (EOT, left panel) and baseline to EOT+4 weeks (right panel) with each dot representing a participant. Change scores for the Upper Extremity Fugl-Meyer Motor score (FMA, 3a), Action Research Arm Test (ARAT, 3b), and Six-minute walk test (6MWT, 3c) are shown with the minimal clinically important difference (MCID) range or value depicted by the blue horizontal shading or line, respectively. While both groups made significant improvement on the FMA and ARAT, more participants in the FE+RTP group exceeded MCID thresholds at all time points with the exception of FMA ≥7.25 at EOT+4. With respect to change in walking capacity measured by the 6MWT, only the FE+RTP group made significant improvements at both time points with group means exceeding MCID.
Changes From Baseline to EOT and EOT + 4 by Group.
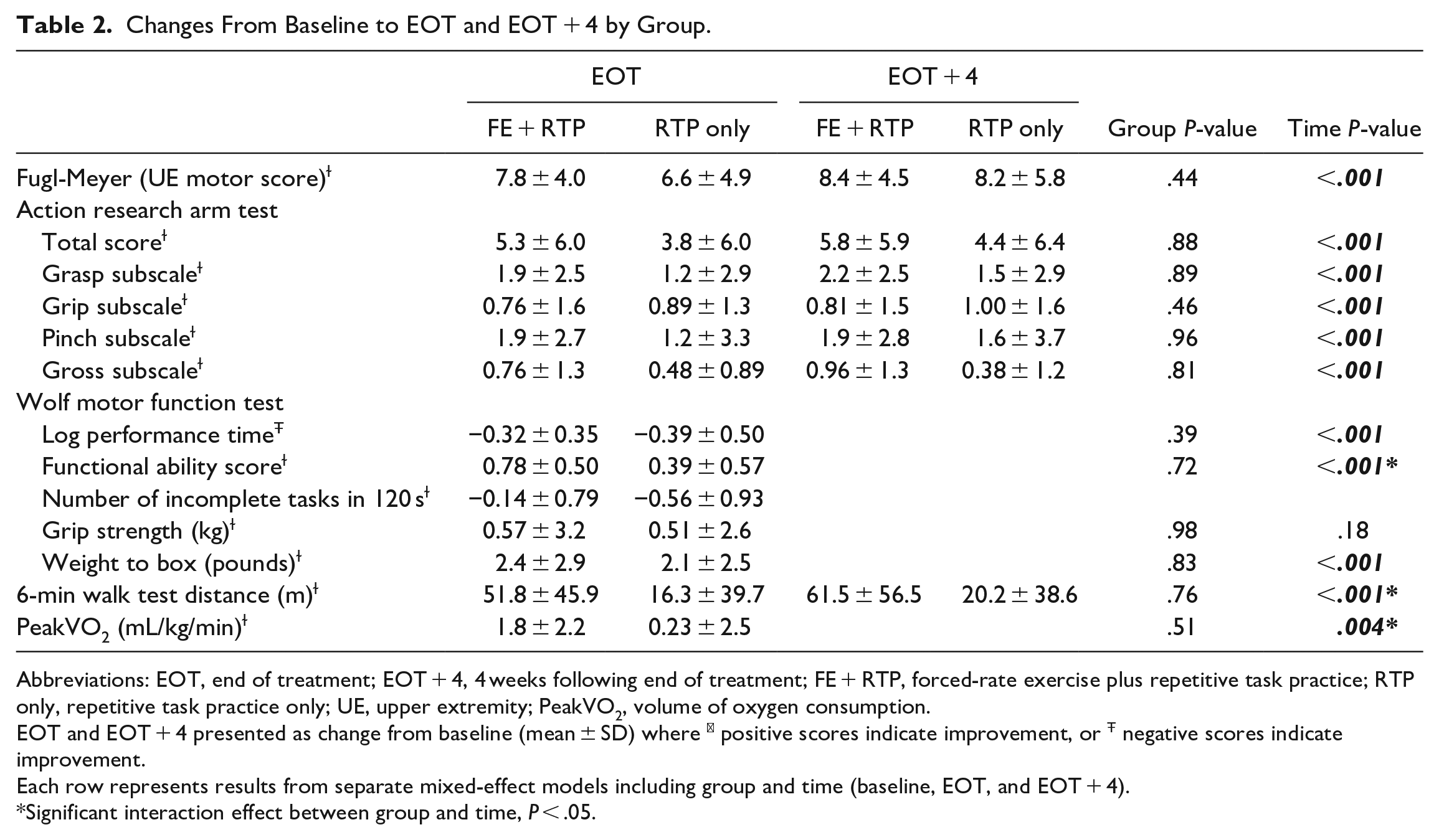
Abbreviations: EOT, end of treatment; EOT + 4, 4 weeks following end of treatment; FE + RTP, forced-rate exercise plus repetitive task practice; RTP only, repetitive task practice only; UE, upper extremity; PeakVO2, volume of oxygen consumption.
EOT and EOT + 4 presented as change from baseline (mean ± SD) where ɫ positive scores indicate improvement, or Ŧ negative scores indicate improvement.
Each row represents results from separate mixed-effect models including group and time (baseline, EOT, and EOT + 4).
Significant interaction effect between group and time, P < .05.
Walking Capacity and Cardiovascular Fitness
The FE + RTP group demonstrated significantly greater improvements in the 6MWT (274.9 ± 122.0-327.1 ± 141.2 m) compared to the RTP only group (285.5 ± 160.3-316.9 ± 170.0, P = .003, Table 2, Figure 2) and aerobic capacity measured by peak oxygen consumption (FE + RTP, 16.6 ± 5.3-18.3 ± 6.4 vs RTP only, 16.3 ± 5.5-17.2 ± 5.6 mL/kg/minute, P = .024, Table 2).
Responder Analysis
Change scores for the FMA, ARAT, and 6MWT that exceeded the respective MCIDs are shown in Table 3 and Figure 3. More participants in the FE + RTP group exceeded MCID thresholds at EOT and EOT + 4 for all outcomes, with the exception of FMA ≥7.25 at EOT + 4. Significantly more FE + RTP participants exceeded MCID on the 6MWT at EOT and EOT + 4 than the RTP only group.
Percentage of Patients With Change Scores Exceeding Minimal Clinically Important Difference.
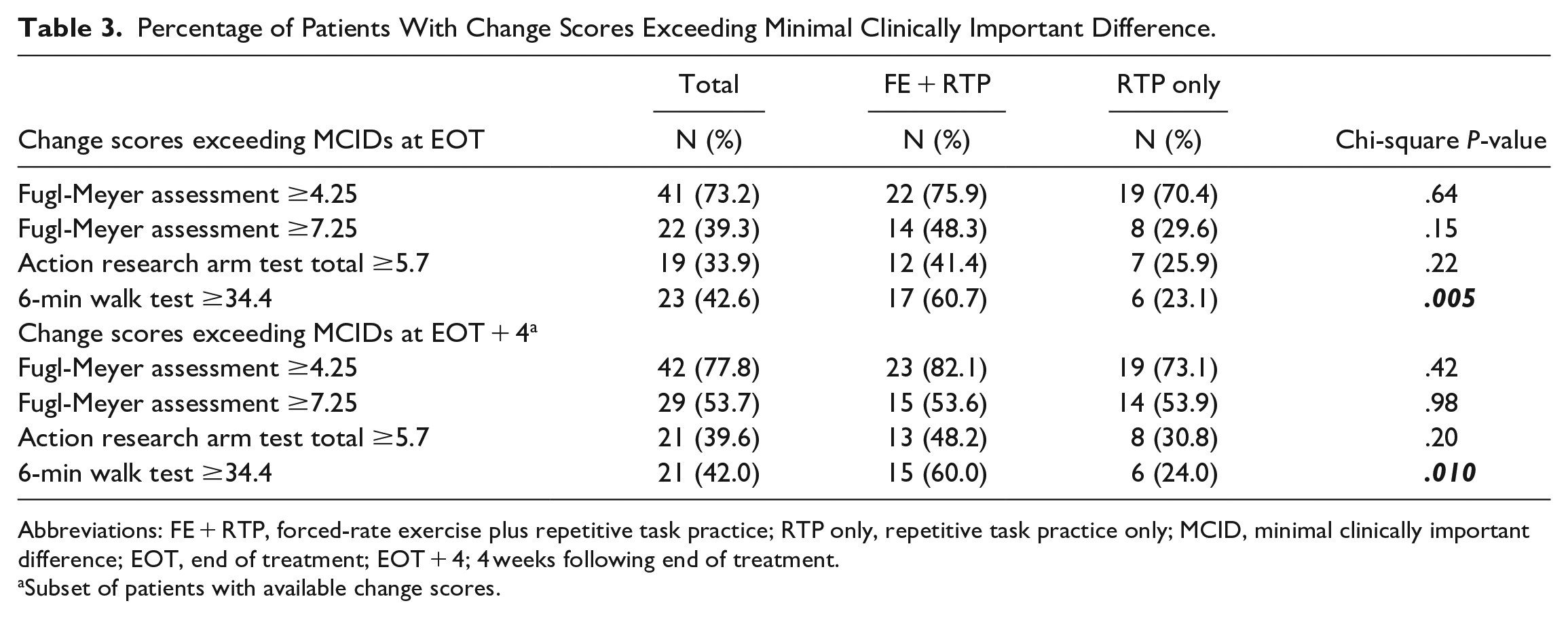
Abbreviations: FE + RTP, forced-rate exercise plus repetitive task practice; RTP only, repetitive task practice only; MCID, minimal clinically important difference; EOT, end of treatment; EOT + 4; 4 weeks following end of treatment.
Subset of patients with available change scores.
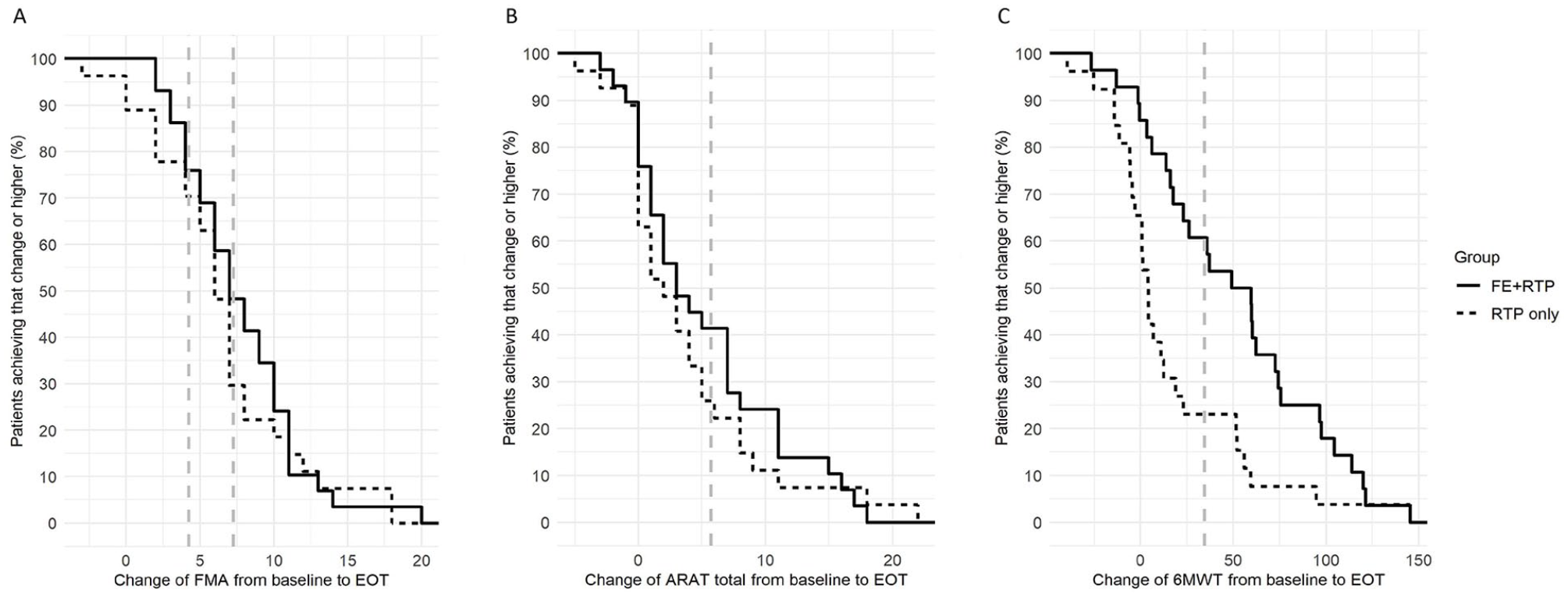
Change scores for the Upper Extremity Fugl-Meyer Motor score (FMA, 2a), Action Research Arm Test (ARAT, 2b), and Six-minute walk test (6MWT, 2c) are shown with the minimal clinically important difference (MCID) range or value depicted by the blue horizontal shading or line, respectively. The y-axis shows the proportion of patients at each point along the outcome scale (x-axis) who experience change at that point or lower. While both groups made statistically significant improvement on the FMA and ARAT, more participants in the FE+RTP group exceeded MCID thresholds at EOT.
Discussion
An intervention comprised of AE and RTP produced similar improvements in UE motor performance as an RTP intervention that was twice as long. Similar motor response with half the RTP dose suggests that AE may be priming the CNS to optimize neuroplasticity in persons with stroke and should be considered as an adjunct to traditional RTP. While we hypothesized that FE + RTP would yield superior improvements in UE motor recovery, the null hypothesis was not rejected. However, unique to the FE + RTP group, significant and clinically meaningful improvements in locomotion and cardiovascular fitness were elicited that were not observed in the RTP only group.
Recommendations for individuals post-stroke to engage in AE are not new; however, the benefits for CNS function have historically not been emphasized. 39 Sustained and intensive AE has been shown to elicit vascular and neurochemical responses within the CNS, hypothesized as the mechanism by which AE facilitates neuroplasticity.4,15,16,40-46 Based on these data, our scientific premise was that sustained moderate- to high-intensity AE administered through FE would prime the CNS and optimize recovery associated with motor task practice. We used FE to supplement the voluntary efforts of participants to overcome barriers to completing high intensity AE that individuals post-stroke experience, including hemiparesis, fatigue, spasticity, impaired balance, and reduced cardiovascular fitness. 47 Participants in the FE + RTP group exercised for 45 minutes at an aerobic intensity (56.4% of HRR) that is nearly equivalent to ACSM exercise guidelines for the general population. 48 Therefore, supplementing the individuals’ voluntary efforts using FE facilitated a relatively intensive exercise duration (ie, 45 minutes/session) without compromising aerobic intensity.
Because FE involved lower limb cycling, it was unlikely to elicit a transfer of training effect to impact UE function; thus, we hypothesize that a global priming effect was induced, enhancing UE recovery. The MCID for the FMA ranges from 4.25 to 7.25 points. 36 Despite half the RTP dosage, mean change scores for the FE + RTP group exceeded the upper value of MCID range for the FMA at both time points. Additionally, responder analyses (Table 3) revealed that more participants in the FE + RTP group exceeded the MCID for the ARAT at both time points, although this was not statistically significant between groups. In a previous study investigating RTP dosage, Lang et al 9 reported change in ARAT scores of 5.8 and 5.1 for participants receiving 13.6 and 20 hours of UE RTP, respectively, while our FE + RTP group demonstrated a 5.3 point change after 18 hours of RTP. By comparison, the RTP only group receiving a total of 36 hours of RTP improved by 3.8 points. While in part our study was designed to investigate the effects of 2 different doses of RTP, a sham non-AE intervention would be an interesting comparator group to evaluate the differential effects of AE on motor recovery. Additionally, investigating different intensities of aerobic training would further elucidate characteristics of AE that prime the brain for neuroplasticity. We acknowledge that a gap is the investigation of mechanisms that may be responsible for the improvements observed. Future studies have been proposed that include neural and biochemical mechanisms that underlie exercise-induced plasticity.
While not designed to improve walking, a significant and meaningful by-product of FE + RTP was improvements in walking capacity, with a change in 6MWT distance of 52.2 m, exceeding the MCID of 34.4 m. 49 These results approach the robust improvements in 6MWT values recently reported by Boyne et al 50 after 8 weeks of high-intensity interval treadmill-based gait training administered 3×/week and suggest that FE improves locomotor function by potentially eliciting a transfer of training to walking. 51 An interim analysis of data from the FE + RTP group revealed normalization of gait kinematics and kinetics associated with increased gait velocity following FE, indicating participants improved locomotor control and did not exaggerate existing compensatory strategies to walk faster. 52 Task-specificity, a fundamental concept in motor learning, has long been considered critical for neuroplasticity; yet our FE + RTP intervention did not involve task-specific gait training. While cycling and walking are different tasks, improvements in LE kinematics and kinetics from cycling may transfer to gait, as both require the rapid reciprocal activation and relaxation of lower extremity muscles synergistically, and some have proposed similar controlling neural networks.53-60 Pedaling data from our pilot study revealed increased hemiparetic limb torque production, improved symmetry of force production, and diminished roughness index (a measure of motion smoothness) as a result of cycling.61,62 It is plausible that increased somatosensory input, muscle power, lower limb motor control, and/or endurance may have elicited these gains. 63
AE has been recommended for decades to improve physical and psychosocial function in persons post-stroke.39,47 While lab-based studies have repeatedly demonstrated the value of AE, feasibility, and reimbursement remain as barriers to implementation in clinical settings. The cardiac rehabilitation model has been previously proposed; however, stroke is not a diagnosis for which cardiac rehabilitation is covered in the United States. Additionally, in isolation, AE administered in a cardiac rehab setting would undoubtedly have numerous benefits but is not designed to enhance neuroplasticity. Novel approaches to include AE in stroke rehabilitation settings are warranted to improve clinical and value-based outcomes.
Conclusions, Limitations, and Future Directions
Our findings that an aerobic cycling intervention administered immediately prior to UE RTP can elicit robust improvements in UE function and locomotion comparable to task-specific training approaches can have significant implications on clinical practice. The FE + RTP intervention was tolerated by all participants without serious adverse events and its summative value in improving UE, lower extremity, and cardiovascular outcomes was demonstrated. The 60% to 80% target HR range was calculated based on CPX results; however, FE sessions were not completed at the same time of day as the CPX test. Therefore, participants on beta blockers may have had slight alterations in their target HR range and actual aerobic intensity, based on precise medication half-life and potency. Future studies employing mechanistic and clinical outcomes to elucidate the role of AE in eliciting a global response within the CNS and facilitating neuroplasticity are planned.
Footnotes
Acknowledgements
The authors wish to acknowledge Anson Rosenfeldt, DPT, MBA and Cindy Clark, OT for their contributions in administering study-related interventions.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Alberts has authored intellectual property protecting the algorithm associated with the forced-rate exercise cycle.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The trial was funded by the National Institutes of Health K01HD092556 to Susan Linder.
