Abstract
Introduction
Parkinson’s disease (PD) is one of the most common neurodegenerative disorders. Although pharmacological treatment has changed its evolution, PD still leads to major motor disabilities that impair the quality of life.
1
In the past 10 years, a variety of rehabilitation treatments have been used with success to target one or more motor deficits that are typical of PD.2-6 More interestingly, a careful review of the results of randomized studies with rehabilitative approaches in PD suggests that: first, exercise improves several aspects of motor performance,7-10 and second, exercise might have a neuroplastic action that might slow down the disease progression.11-13 Nevertheless, the evidence of a possible neuroprotective action of exercise in PD has been obscured by the fact that all the studies were performed in patients in different stages of disease and under a variety of pharmacological treatments. In fact, because of the diversity of mechanisms and targets, the various anti-Parkinsonian drugs (levodopa [
To fill this gap, in a group of “de novo” Parkinsonian patients in monotherapy with rasagiline, we investigated over the course of 2 years the effects of a 4-week multidisciplinary intensive rehabilitation treatment (MIRT), repeated once a year. As a control group, we also followed, for the same period of time, a group of patients with the same clinical characteristics and pharmacological treatment that did not perform the 2 MIRTs. Then we compared the progression of the 2 groups in terms of motor performance, autonomy in daily living activities and drug requirements over the 2-year period.
Methods
Study Population
This was a random assignment, parallel-group, pilot study with a 1:1 allocation ratio. For allocation of the participants, a list of random numbers was computer generated, using Matlab random numbers generator (MATLAB version 7.14.0.739, MathWorks, Inc, Natick, MA). The sequence was concealed, so that the investigators enrolling patients in the study were blind to this aspect until group assignment. The study was approved by the local institutional review board and by the Central Ethics Committee (approval number 584 CEC, September, 21, 2009), and all subjects gave their informed written consent. Eligibility criteria were the following: diagnosis of “clinically probable” idiopathic PD according to Gelb et al, 14 Hoehn–Yahr stage 1 to 1.5, ability to walk without physical assistance, Mini-Mental State Examination score ≥26, no serious comorbidity, and no vestibular/visual dysfunction limiting locomotion or balance.
“De novo” patients with PD were diagnosed and screened by a neurologist specialized in movement disorders, who was blind to group assignment. All eligible patients began treatment with rasagiline. Four weeks from the onset of rasagiline treatment, following the randomization list, a group of patients (Control Group) continued with the pharmacological treatment only, while the other (MIRT Group) took part in a 4-week treatment at the Maugeri Rehabilitation Institute of Montescano. The same MIRT treatment was repeated 1 year later.
Assessment of Outcomes
The primary outcome measures were the following: the Unified Parkinson’s Disease Rating Scale, sections II and III (UPDRS II and III), 15 the 6-minute walking test (6MWT), the Timed Up-and-Go Test (TUG), 16 and the self-assessment Parkinson’s Disease Disability Scale (PDDS). 17 Secondary outcome measures were the dosage of levodopa equivalent and the number of patients in monotherapy with rasagiline.
All patients were evaluated at baseline (T0), 6 months (T1), 1 year (T2), 18 months (T3), and at 24 months (T4) (see Figure 1).
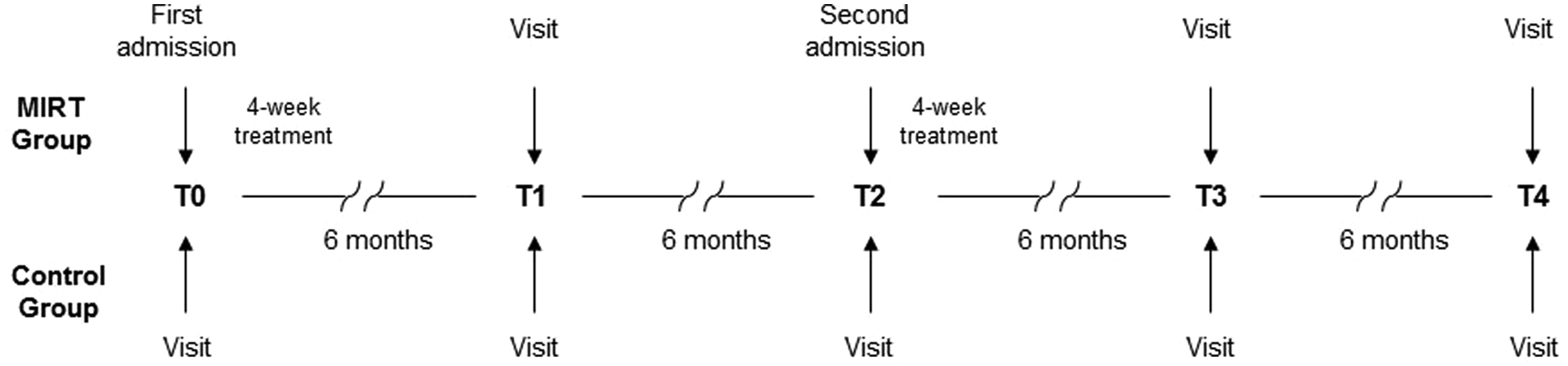
Study design.
Patients in the MIRT group underwent the first 4-week MIRT just after T0 and the second MIRT after T2.
All examinations were performed at about 9
Patients in the MIRT group were also evaluated immediately after each 4-week treatment. This assessment was carried out by a neurologist who was not involved in the study and thus, who was not blind to patient allocation.
Sample Size Computation
The sample size was determined on the basis of the outcome variables with standard error of measurement (SEM) available from published studies. The published SEMs that we used for UPDRS II, UPDRS III, 6MWT, and TUG were 1.4, 4, 30 m, and 0.59 seconds, respectively.18,19 We expected an effect size around 2, 5.2, 35 m, and 1.2 seconds for the same variables (clinically moderate difference). 20 Hence, to detect a change with a 2-tailed type I error of 0.05 and a power of 80%, the largest estimated sample size was 20 patients per group.
Statistical Analysis
Descriptive statistics are reported as mean ± standard deviation (SD). The normality of the distribution of all variables was assessed by the Shapiro–Wilk statistic. To investigate the primary endpoint, we tested the differences of outcome variables in the MIRT Group, compared with the Control Group over the 2-year follow-up period. Thus, we used a mixed model analysis of variance, with treatment as between factor (MIRT vs Control) and time (T0, T1, T2, T3, T4) as within factor (repeated measure).
If a significant interaction effect for time and treatment was found, post hoc analyses were performed (Tukey’s honestly significant difference test) to compare follow-up and baseline measures in both groups of patients.
For within-group comparisons, we used paired
Finally, we assessed the change of levodopa equivalent dosage in the 2 groups across testing points with a 2-factor analysis of variance, with treatment (MIRT v Control) as between factor and time (T0, T1, T2, T3, T4) as within factor (repeated measures).
A
Intervention
Patients in the MIRT Group were admitted to the Rehabilitation Institute of Montescano, Pavia, Italy, to undergo MIRT, a treatment which involved the participation of neurologists, physiatrists, psychologists, nurses, physiotherapists, and occupational therapists. 12 Briefly, MIRT consisted of a 4-week treatment with three 1-hour daily sessions (2 in the morning and 1 in the afternoon), 5 days a week. The first session consisted of cardiovascular warm-up activities, relaxation exercises, muscle stretching (scapular, hip flexor, hamstring, and gastrocnemius muscles), exercises to improve the range of motion of spinal, pelvic and scapular joints, and postural changes in the supine position. The second session consisted of exercises to improve balance and gait, using stabilometric platforms with visual cue and treadmill training associated with auditory and visual cues (treadmill plus). In the exercises with the stabilometric platform, patients were asked to follow a circular pathway on the screen with a cursor that represented the movements of their feet on the platform. For the treadmill training, the visual cue was a target displayed on a screen that the patients had to reach within a stride; the auditory cue consisted of musical beats, synchronized with the visual cues, with a frequency of 0.5 cycles/s. 5 All the exercises on the treadmill were aerobic with a heart rate reserve 60% to 70% and a maximum speed of treadmill scrolling of 3.5 km/h. Patients trained for 30 minutes every day for 4 weeks, for a total of 20 sessions. A physiotherapist monitored the patients’ activities during the entire treadmill training. Before the training session, we determined the maximum tolerated walking speed. This speed was reduced (−40%) and used for a 2-day warm-up. The belt speed was then increased every 3 days by 0.05 stride cycles/s. We selected to adjust the speed in terms of stride cycles/s in order to normalize the data in terms of patients’ height.
The last session was an occupational therapy session designed to improve autonomy in daily living activities. It included transfers from sitting to standing, rolling from supine to sitting and from sitting to supine, dressing, use of tools, and exercises to improve hand functionality and skills (eg, using screws and bolts). At discharge, patients were instructed to continue a set of learned exercises, in order to maintain functionality of the spine, scapular and pelvic joints, along with the instruction to walk at least 30 minutes a day or to use the treadmill for 20 min/d. Patients were also asked to keep a diary of these activities.
Patients in the Control Group underwent only pharmacological treatment.
Results
The recruitment started on January 2010 and closed 14 months later.
In the MIRT Group, a total of 16 patients completed the study, with 1 dropout for cerebrovascular disease, 2 for orthopedic problems, and 1 was lost at follow-up for unknown reasons. In the Control Group, 15 patients completed the study: 2 patients were lost at follow-up for unknown reasons, 1 for cerebrovascular disease, 1 for cardiovascular problems, and 1 for developing diabetic polyneuropathy (Figure 2).
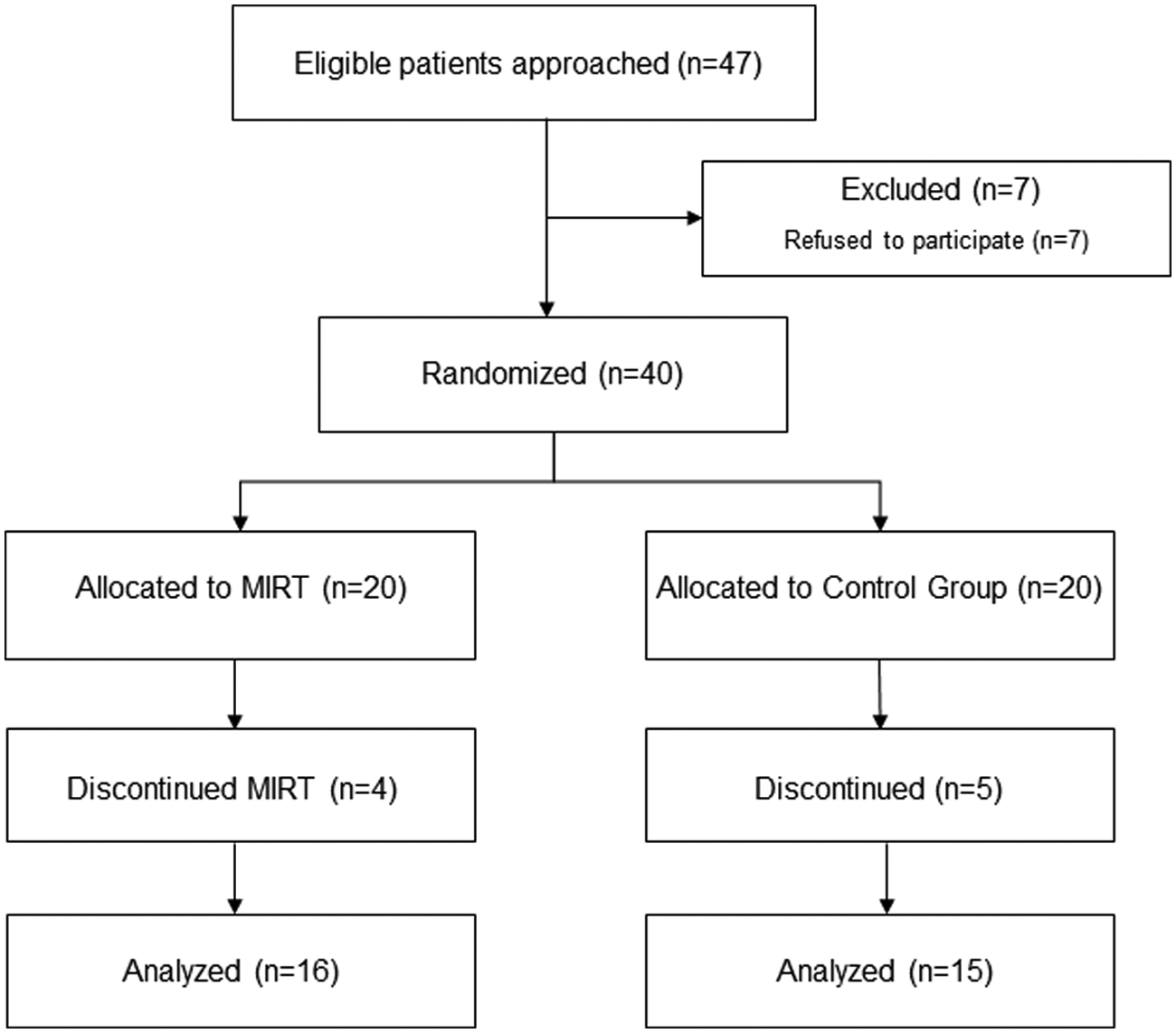
Flowchart. MIRT, multidisciplinary intensive rehabilitation treatment.
No differences were observed in the 2 groups of patients in terms of age, Hoehn–Yahr stage, and baseline pharmacological treatment (Table 1).
Demographic Characteristics of Patients (Mean ± SD) at Baseline (T0) for the 2 Groups.
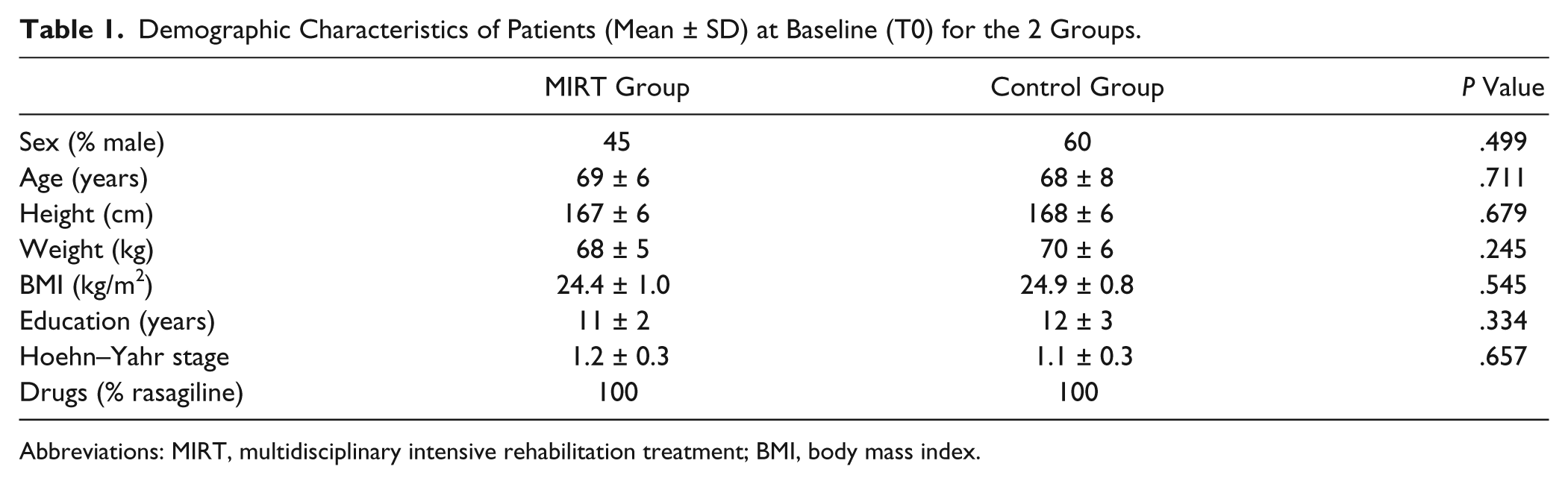
Abbreviations: MIRT, multidisciplinary intensive rehabilitation treatment; BMI, body mass index.
On average, patients in MIRT Group carried out 95% of the scheduled MIRT sessions, for a total of more than 55 hours of treatment. No adverse events were observed during the entire MIRT treatment.
In the MIRT-treated patients, all outcome variables improved acutely immediately after both MIRT treatments (Table 2).
Acute Changes (Values at Discharge − Values at Admission) in Outcome Measurements After First and Second MIRT (Values are Reported as Mean ± SD).
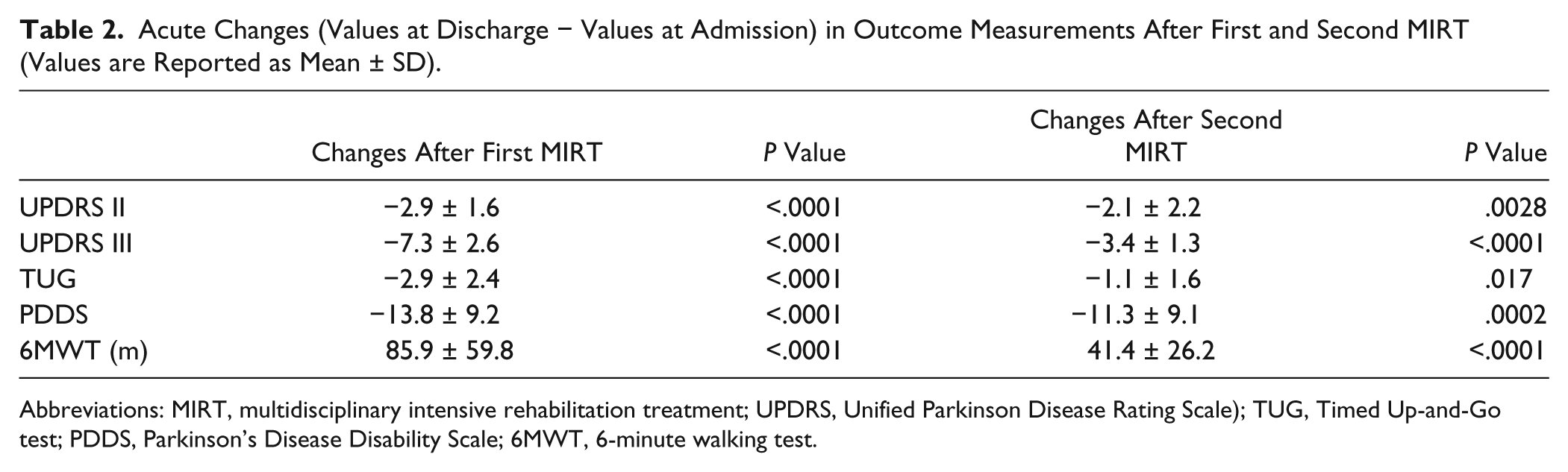
Abbreviations: MIRT, multidisciplinary intensive rehabilitation treatment; UPDRS, Unified Parkinson Disease Rating Scale); TUG, Timed Up-and-Go test; PDDS, Parkinson’s Disease Disability Scale; 6MWT, 6-minute walking test.
Patients’ compliance regarding home activities was on average good. Analysis of diaries revealed that exercises were performed on average 3 d/wk and treadmill 2 d/wk. Patients’ compliance was always better immediately after MIRT and gradually decreased with time.
Descriptive statistics for the outcome measurements of the 2 groups at enrolment (T0), at 6 months (T1), 12 months (T2), 18 months (T3), and 24 months (T4) are reported in Table 3.
Outcome Measurements (mean ± SD) of the 2 Groups for the 5 Observation Times Considered.
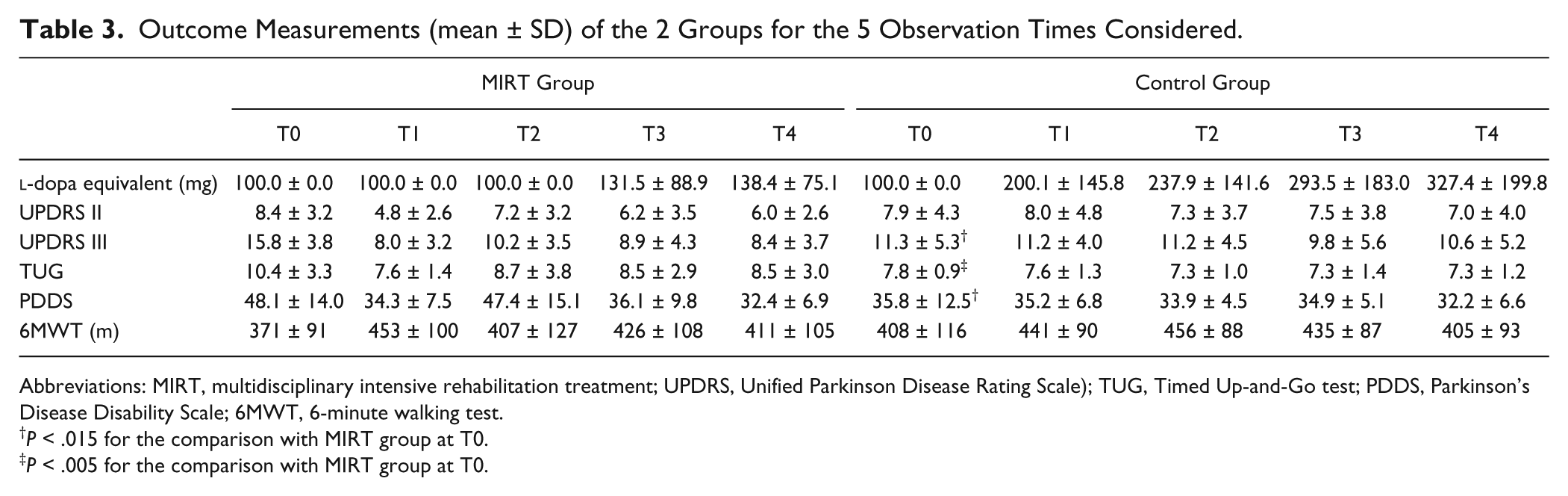
Abbreviations: MIRT, multidisciplinary intensive rehabilitation treatment; UPDRS, Unified Parkinson Disease Rating Scale); TUG, Timed Up-and-Go test; PDDS, Parkinson’s Disease Disability Scale; 6MWT, 6-minute walking test.
Some outcome variables at baseline were better in the Control Group (
Results of analysis of variance (ANOVA) for all variables are summarized in Table 4.
Analysis of Variance Results for Primary Outcome Variables.
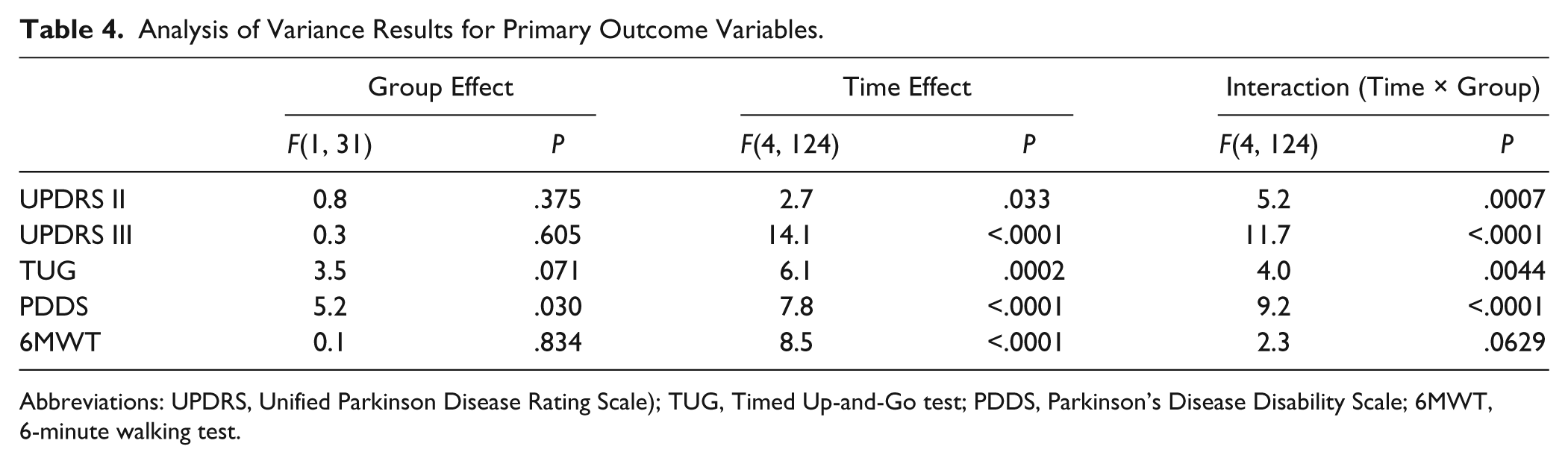
Abbreviations: UPDRS, Unified Parkinson Disease Rating Scale); TUG, Timed Up-and-Go test; PDDS, Parkinson’s Disease Disability Scale; 6MWT, 6-minute walking test.
The main finding of this study was that the time course of UPDRS II, UPDRS III, TUG, and PDDS was different in the MIRT and Control groups, as revealed by the significant time × treatment interaction in the repeated-measures ANOVA (Table 4, last column). While no significant changes were found over time in the control group, all variables significantly improved over time in the MIRT Group. Post hoc analysis in MIRT Group showed that, compared with baseline, UPDRS II, UPDRS III, TUG, and PDDS significantly improved at T1 (
In addition, with a subject-by-subject analysis for each scores, we computed the percentage of patients that showed improvements greater than 10% at the end of the follow-up compared with baseline. The results are reported in Table 5.
Percentage of Patients Who Showed a Score Improvement Greater Than 10% at the 6-Month Point (T1) and at the End of the Follow-up period (T4) Compared With the Baseline (T0).
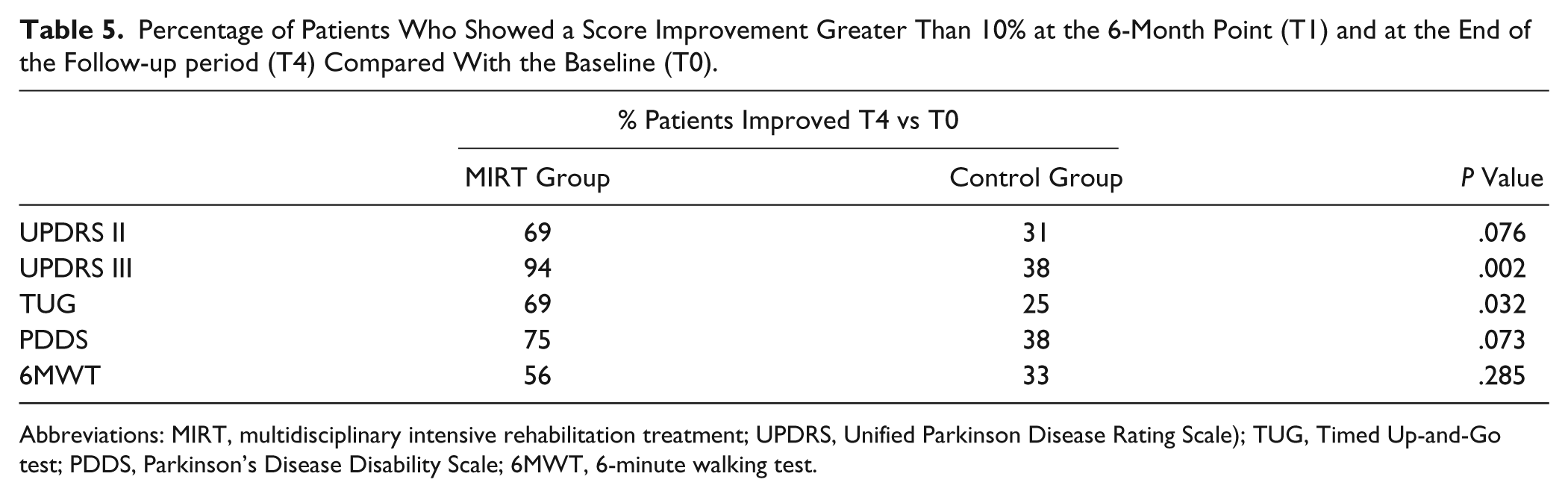
Abbreviations: MIRT, multidisciplinary intensive rehabilitation treatment; UPDRS, Unified Parkinson Disease Rating Scale); TUG, Timed Up-and-Go test; PDDS, Parkinson’s Disease Disability Scale; 6MWT, 6-minute walking test.
Finally, we verified the effect of rehabilitation on the drug treatment regimen. Figure 3 shows the average of
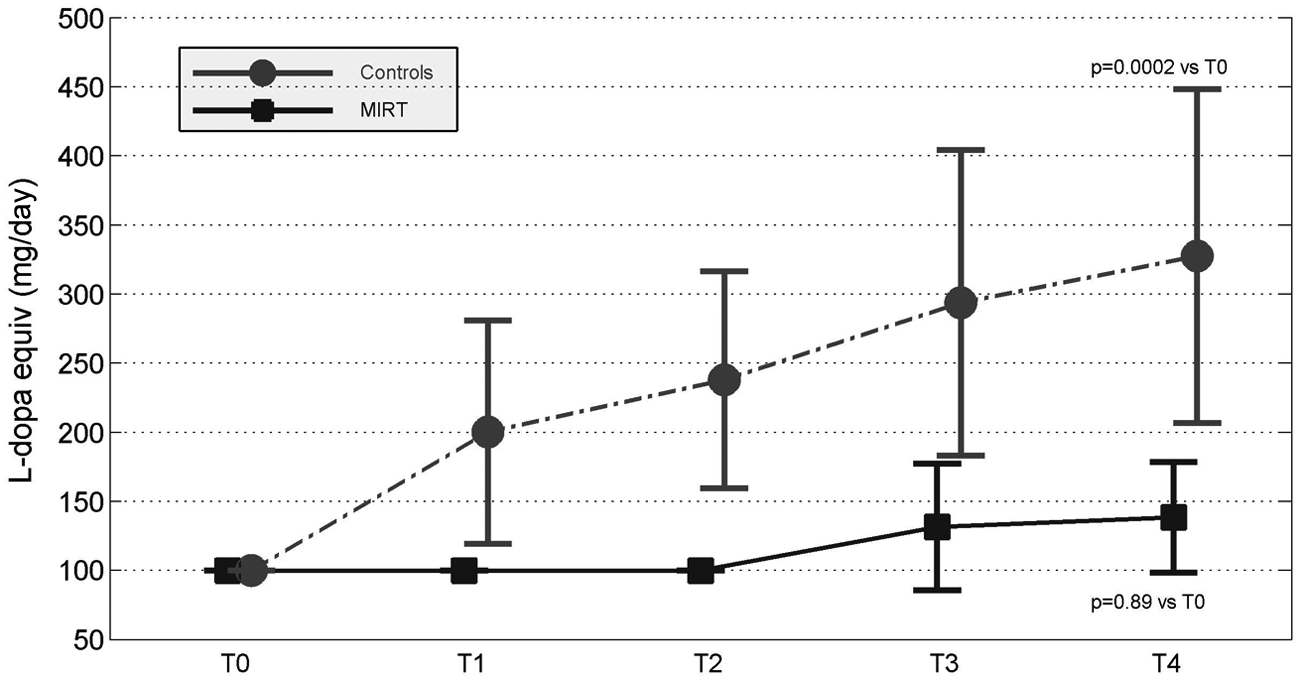
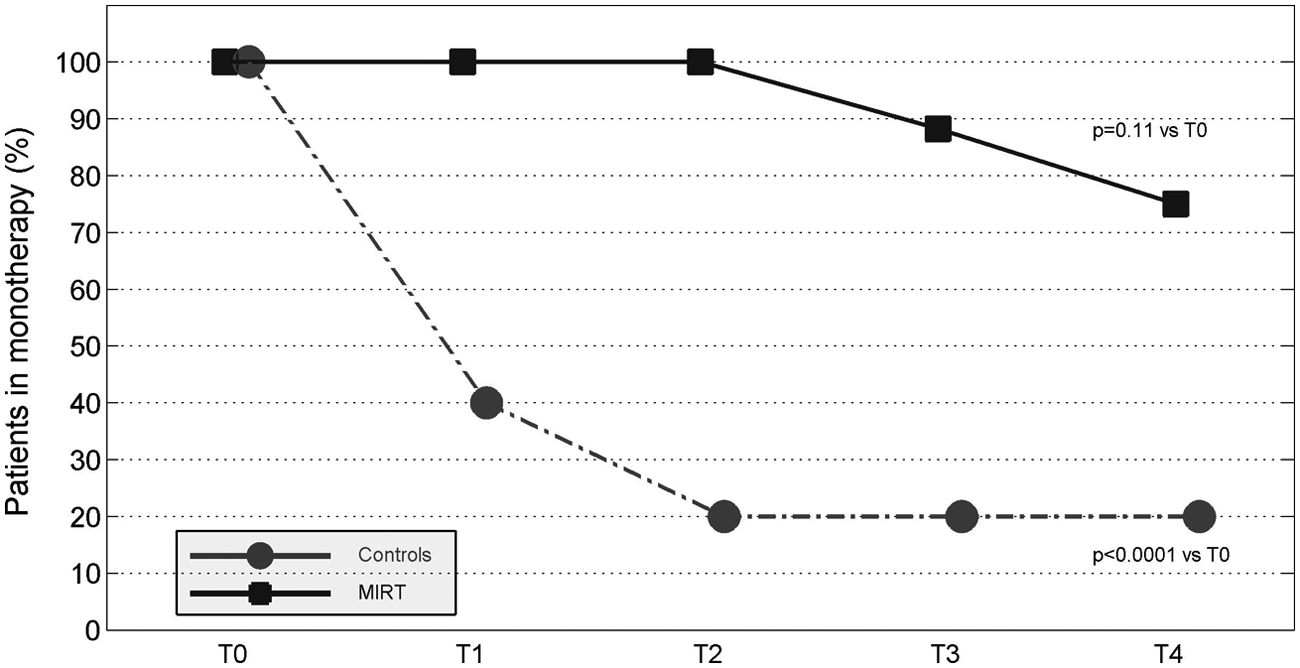
Patients in monotherapy with resagiline. MIRT, multidisciplinary intensive rehabilitation treatment.
Discussion
This is the first randomized, controlled, follow-up study that was designed to determine whether 2 MIRTs have some positive effects on the progression of PD. Importantly, this study was performed in patients in the early stage of PD, with the same pharmacological treatment (rasagiline) at enrolment. The main result of this pilot study is that, already in early stage of disease, MIRT seems to slow down the progression of the motor decay, possibly modifying the course of disease and MIRT seems to delay the need for increasing drug treatment. On the whole, these results suggest a possible neuroprotective or neurorestorative aspect of exercise in PD.
Immediately after the first and the second treatment, the scores of the scales, used to evaluate several aspects of motor performance and autonomy in daily living activities, showed a significant improvement compared with their baseline values. These results, obtained in the early stage of disease, are similar to the ones reported in our previous study, with patients in more advanced stages. 12 Usually, the presence of some motor symptoms is the reason why patients consult a neurologist and, at that time, the diagnosis is made. 21 Although symptoms are usually mild at this stage, they almost invariably reduce the patients’ motor activity with a progressive worsening of motor performance. Based on the present results, we speculate that early intervention with intensive rehabilitation programs at the initial stage might act as a “motor shock,” by breaking this vicious cycle, resetting and restoring motor performance to values close to premorbid state and, plausibly, slowing down the rate of motor decay.
The second important result of this study is that the progression of the motor decay was slower (and in some cases, even reversed) in the MIRT Group, compared with the Control Group: In fact, at the end of the 2-year observation period, motor performance and daily living activities scores were better than at baseline in the MIRT Group, while they remained stable in the control group. Importantly, this difference in progression was present despite the fact that most MIRT patients were still in rasagiline monotherapy and their levodopa equivalent dosages were significantly lower than in the control group. Thus, it is possible that early MIRT interventions might modify the course and the rate of progression of disease, by decreasing both the symptoms severity and the need for increasing drug regimen.
An important aspect of this study is that both groups of patients started at enrolment with the same pharmacological treatment, that is, a monotherapy with the same dosage of rasagiline. We chose this treatment because the inhibition of MAO-B prolongs the availability and activity of endogenous dopamine in the striatum, without alteration of the normal synaptic function.22,23 The study by Lew et al, 24 in which the long-term efficacy, safety, and tolerability of rasagiline was tested in a large population of patients with PD in the early stages, indicated that, after 12 months, 70% of patients were still in rasagiline monotherapy. However, 24 months later, only 46% used only rasagiline. The results of our study suggest that MIRT might improve this outcome.
The main goal of pharmacological therapy in PD is to help patients retain functional independence as long as possible. Levodopa remains the most effective drug in reducing symptoms, but in general, there are several reasons to postpone its use for the treatment of early PD, such as the development of “wearing-off” effects and of levodopa-induced dyskinesias. MAO-B inhibitors, which have only a mild symptomatic effect, might slow the progression rate in de novo patients,25,26 although the American Academy of Neurology Quality Standard Subcommittee did not recognize them as neuroprotective agents.27,28 Likewise, the hypothesis about a neuroprotective action of dopamine agonists has not been confirmed. 28 Interestingly, recent studies have shown that moderate exercise habits in midlife significantly reduce the risk of developing PD,29,30 leading to the speculation that exercise might play a role in slowing down the progression of disease. 28
The mechanism underlying the beneficial effect of exercise in PD is still a matter of speculation. Several studies with Parkinsonian animal models have shown that physical exercise enhances brain neuroplasticity31-34 and increases plasmatic levels of different neurotrophic factors.35,36 In normal human subjects, exercise increases serum levels of different growth factors, 37 which can cross the blood–brain barrier in both directions. 38 Thus, it is possible that, following exercise, the increase of circulating growth factors may serve as a relevant reserve for brain cells, promoting neuroplasticity in PD, at least in the early stages of disease. In addition, studies in animal models have shown that intensive treadmill exercises play a neurorestorative action, improving postlesion dopamine neurotransmission.34,39
In conclusion, we speculate that the association of exercise with MAO-B inhibitor therapy could amplify the beneficial effects of both treatments through different mechanisms, possibly achieving a certain degree of neuroprotection.
Beyond the speculation about a possible protective effect of exercise, our results suggest that MIRT, in the early stage of disease, can not only slow down disease progression, but it can also lead to a better motor performance. This conclusion is also in agreement with the current guidelines, which encourage patients with PD to begin exercise training programs with a high training intensity, “beyond what they may self-select,” 40 with a multidisciplinary approach. 41 In fact, with the disease progression the sedentary lifestyle imposed by the motor symptoms, worsens balance and gait disturbances, which do not respond well to dopaminergic therapy. 42 In addition, other nonmotor symptoms such as depression, fatigue, apathy, and constipation, further reduce physical activity in these patients. That is why this vicious cycle should be stopped as soon as possible, as its onset produces early deterioration of physical fitness, which in turn leads to a more rapid disease progression. 43
Study Limitations
The major limitation of the study is that the number of patients for each group was rather small. However, the sample size was dimensioned a priori, based on the outcome indices used to assess the effect of exercise. Another limitation is related to exercise at home: We did not strictly monitor this aspect, but we encouraged the patients in the MIRT Group to perform the learnt exercises at home. During follow-up patients, reported that they had carried out regularly the recommended exercises. Further investigations are also needed to define the neural mechanisms underlying these effects.
The neurologists in charge of assessing patients were blind to treatment allocation, but we cannot exclude that some information could have been sporadically provided by the patients. However, being the neurologists unaware of the aims of the study, this possible lack of blinding should not have biased the evaluations.
Finally, in this pilot study, we did not collect data to adequately assess the cost-effectiveness of MIRT. Ad hoc studies should be planned to evaluate this important issue.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
