Abstract
Introduction
Vestibular schwannomas are benign tumors that arise from the vestibular division of the eighth cranial nerve. These tumors will almost always result in some vestibular hypofunction arising from damage to the affected nerve. Surgical resection of the tumor leads to complete unilateral vestibular loss because the nerve is by necessity transected to remove the tumor. Individuals who have undergone resection of a vestibular schwannoma experience a loss of vestibular ocular reflex (VOR) function primarily during head movements toward the affected side. Such VOR deficits result in a substantial loss of visual acuity during dynamic movements of the head. 1 These functional VOR deficits may result in individuals limiting their overall mobility and/or altering their head and trunk motion in attempts to stabilize their gaze.
Consistent exposure to head movements is a critical factor for recovery of gaze and postural stability following acute unilateral vestibular hypofunction (UVH), 2 with demonstrated efficacy of gaze and postural stability interventions over control interventions on varied domains of disability 3 following unilateral vestibular loss (neuritis or vestibular schwannoma).4-7 Nonetheless, postsurgical management of individuals who have undergone vestibular schwannoma resection surgery is variable and may include patient education (about the importance of head movement and regular physical activity) and/or early or delayed referral to rehabilitation. 4 Education without referral to vestibular rehabilitation is predicated on the assumption that individuals will perform head movements and head-trunk decoupling with a sufficient dosage (frequency, amplitude, and velocity) of head movements to induce vestibular adaptation and optimization of gaze and postural stability mechanisms. Unfortunately, recent research suggests that when examined during standardized, unconstrained gait activities of short duration, those with UVH constrain their head and trunk movements.8-11
Despite the fact that the most disabling aspects of UVH occur during active movements, the majority of the clinical tests utilized for diagnosis and quantification of these deficits involve no head movement (calorics, vestibular-evoked myogenic potentials), passive movements of the head (videographic head impulse testing), or active movements of the head without body movement (dynamic visual acuity). 1 Although these measures provide a clear picture of the physiological deficits, they fail to provide information about how these deficits influence functional movement during daily living activities, particularly community ambulation.
The main limitations of previous research are that (1) studies are constrained to laboratory testing and, therefore, do not examine individuals during community ambulation and (2) the duration of measurement is generally quite limited (<1 minute). To address these gaps in the literature, we examined how individuals with UVH coordinated their head and trunk movement during community ambulation at their first outpatient follow-up after resection of vestibular schwannoma—that is, approximately 6 weeks after surgery. We used commercially available wearable sensors to quantify head kinematics and head-trunk coordination. Our primary hypothesis was that individuals with UVH 6 weeks after surgical resection of a vestibular schwannoma would demonstrate alterations in the frequency, amplitude, and velocity of head movements; differences in head-trunk coupling; and reduced gait speed during community ambulation compared with individuals with normal, symmetrical vestibular function. Our secondary hypothesis was that individuals with UVH would demonstrate differences in the kinematic characteristics of small- and large-amplitude head turns during community ambulation compared with individuals with normal, symmetrical vestibular function.
Methods
Individuals who had undergone surgical resection of a vestibular schwannoma and healthy individuals with normal, symmetrical vestibular function were recruited for this cross-sectional study between April and November 2016. Individuals aged 18 to 70 years, able to walk unaided, and without a history of lower-extremity surgery or injury within the past 12 months were eligible to participate. Healthy individuals were also eligible if they had no history of central or peripheral nervous system or vestibular pathology. Individuals with UVH were eligible if they had undergone resection of a vestibular schwannoma within the past 2 months but were excluded if they were taking vestibular suppressant medications. Individuals who were unable to safely comply with the study procedures were also excluded. This study was approved by the University of Utah Institutional Review Board. All participants gave written informed consent prior to data collection. This study conforms to STROBE reporting guidelines. 12
Demographic data, including age, body mass index, angular VOR gain (values <0.8 indicate vestibular hypofunction), and the Dizziness Handicap Inventory (higher values indicate a greater degree of dizziness handicap) 13 were gathered for all participants. The Vestibular Activities and Participation Measure (higher values indicate greater activity limitation and participation restriction) was utilized for individuals with UVH. 14
For the community ambulation task, participants walked at their self-determined pace along 1 of 2 routes while wearing 3 small, synchronized wireless inertial sensors (Opal sensors, APDM Inc, Portland, OR), on their forehead, sternum, and waist. One walking route involved walking around a university building and on university grounds, whereas the other route involved walking around public areas of a university hospital. Once the sensors were affixed to participants via fitted elastic straps, they sat stationary for 30 s to ensure a quiet period prior to the start of data collection. Each walking route took approximately 10 minutes to complete and required participants to navigate through crowded hallways, ascend and descend stairs, and scan for pedestrian and/or vehicular traffic. On completion of the route, participants again sat stationary for 30 s; then, the sensors were removed from participants and docked to download the data.
The primary outcomes of interest to characterize head and trunk coordination during community ambulation included frequency of yaw plane head turns (number), head rotation amplitude (degrees), head rotation velocity (degrees/second) and amount of head-trunk coupling (percentage; small values indicate that the head moved independently of the trunk, whereas values close to 100% indicate en bloc turns). The time taken by each participant to complete the walking route was used as a proxy of gait speed. Secondary outcomes of interest were the frequency of head turns (number), head rotation velocity (degrees/second), and amount of head-trunk coupling (%) for small and large head turns. All outcomes were derived from sensor data using previously documented algorithms with demonstrated reliability and responsiveness. 15 In brief, the algorithms determined head turns by numerical integration of gyroscopic velocity data between the period when rotational speed exceeded 3 times the expected noise value of the signal and when the rotational speed dropped below the relevant expected noise value or the rotational velocity changed sign. For each valid head turn, head rotation amplitude was determined by the area under the curve (AUC) of the signal obtained from the head sensor, whereas head rotation velocity was determined as the maximum velocity value obtained from the head sensor. An equivalent AUC from the trunk sensor was also calculated for the time period for each head turn; thus, head-trunk coupling was determined by dividing the AUC from the trunk sensor by the AUC from the head sensor. The unconstrained nature of the community ambulation routes resulted in a relatively large amount of white noise; therefore, only head turn amplitudes exceeding the threshold value of 8.37° were counted as valid turns.
Data Reduction and Statistical Analysis
Analysis was constrained to include head turns up to 90° in order to examine purposeful head turns and exclude turns dictated by the environment, such as walking around corners and pivot turns. 9 To answer our secondary objective, outcomes of head rotation frequency, head rotation velocity and head-trunk coupling were subdivided based on the size of head turns, using 45° as a cutoff (small turns: 8.37° to 45° vs large turns: >45° to 90°). Outcomes from all valid turns were also plotted to descriptively examine how head rotation frequency, head rotation velocity, and head-trunk coupling varied as head rotation amplitude increased in each group.
Because the community ambulation task involved multiple turns and each individual contributed multiple data points for 3 of the 4 outcomes (ie, head rotation amplitude, head rotation velocity, and head-trunk coupling), raw data were plotted to visually check the distribution of these outcomes within individuals. All outcomes except for frequency of turns had right-skewed distributions within individuals, so the median value for each participant was derived for head rotation amplitude, head rotation velocity, and amount of head-trunk coupling, for the overall analysis, for analyzing the subsets of small versus large head turns, and for analyzing differences in head turns according to lesion side in the UVH group. Differences in outcomes based on lesion side for participants with UVH were compared descriptively. Differences in outcomes between participants with and without UVH were compared using separate independent samples
Results
We consented 16 participants with UVH and 10 healthy participants. One participant with UVH developed postoperative complications and withdrew prior to data collection, and 1 healthy participant experienced sensor malfunction during the community ambulation task that resulted in no data collected. Therefore, data for 15 participants with UVH and 9 healthy participants were analyzed (Table 1). Participants with UVH were a mean 44 days (SD = 5.6) postresection of vestibular schwannoma. Nine participants (60%) had a left-sided resection, and 6 (40%) had a right-sided resection.
Participant Characteristics by Groups. a
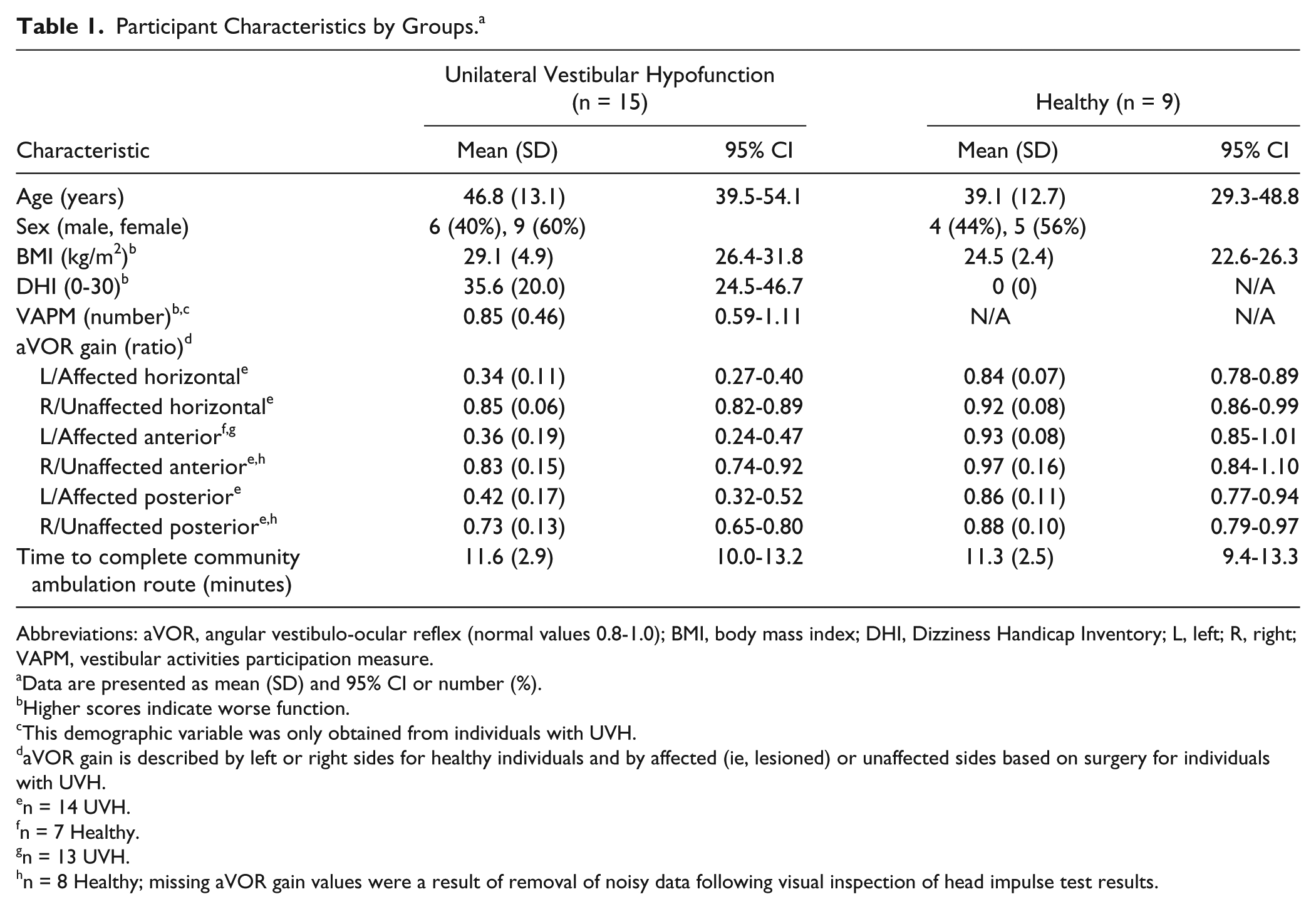
Abbreviations: aVOR, angular vestibulo-ocular reflex (normal values 0.8-1.0); BMI, body mass index; DHI, Dizziness Handicap Inventory; L, left; R, right; VAPM, vestibular activities participation measure.
Data are presented as mean (SD) and 95% CI or number (%).
Higher scores indicate worse function.
This demographic variable was only obtained from individuals with UVH.
aVOR gain is described by left or right sides for healthy individuals and by affected (ie, lesioned) or unaffected sides based on surgery for individuals with UVH.
n = 14 UVH.
n = 7 Healthy.
n = 13 UVH.
n = 8 Healthy; missing aVOR gain values were a result of removal of noisy data following visual inspection of head impulse test results.
For participants with UVH, there was no effect of lesion side on any outcome as demonstrated by the overlapping 95% CIs (Figure 1). For this reason, all turns regardless of direction were collapsed together for subsequent statistical analyses. Participants with and without UVH completed the community ambulation circuits in a similar amount of time (
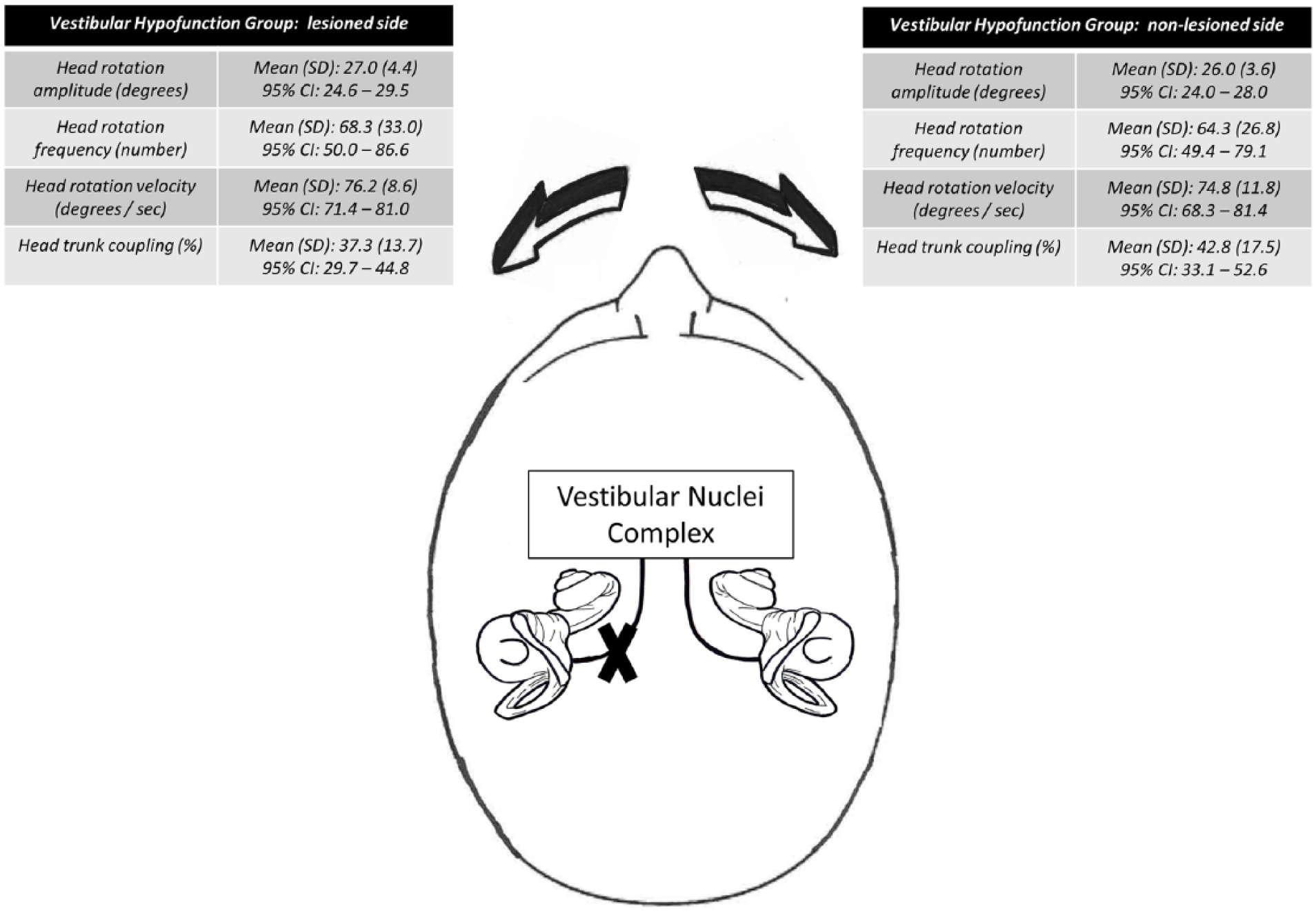
Head kinematics and head-trunk coordination for the unilateral vestibular hypofunction (UVH) group, separated by lesion side. Data are presented as mean (SD) and 95% CI for head turn frequency, and as the mean (SD) and 95% CI of medians from each UVH participant for all other outcomes.
Head Kinematics and Head-Trunk Coordination for All Head Turns and for Subsets of Small (8.37°-45°) and Large (>45°-90°) Amplitude Head Turns, by Groups. a
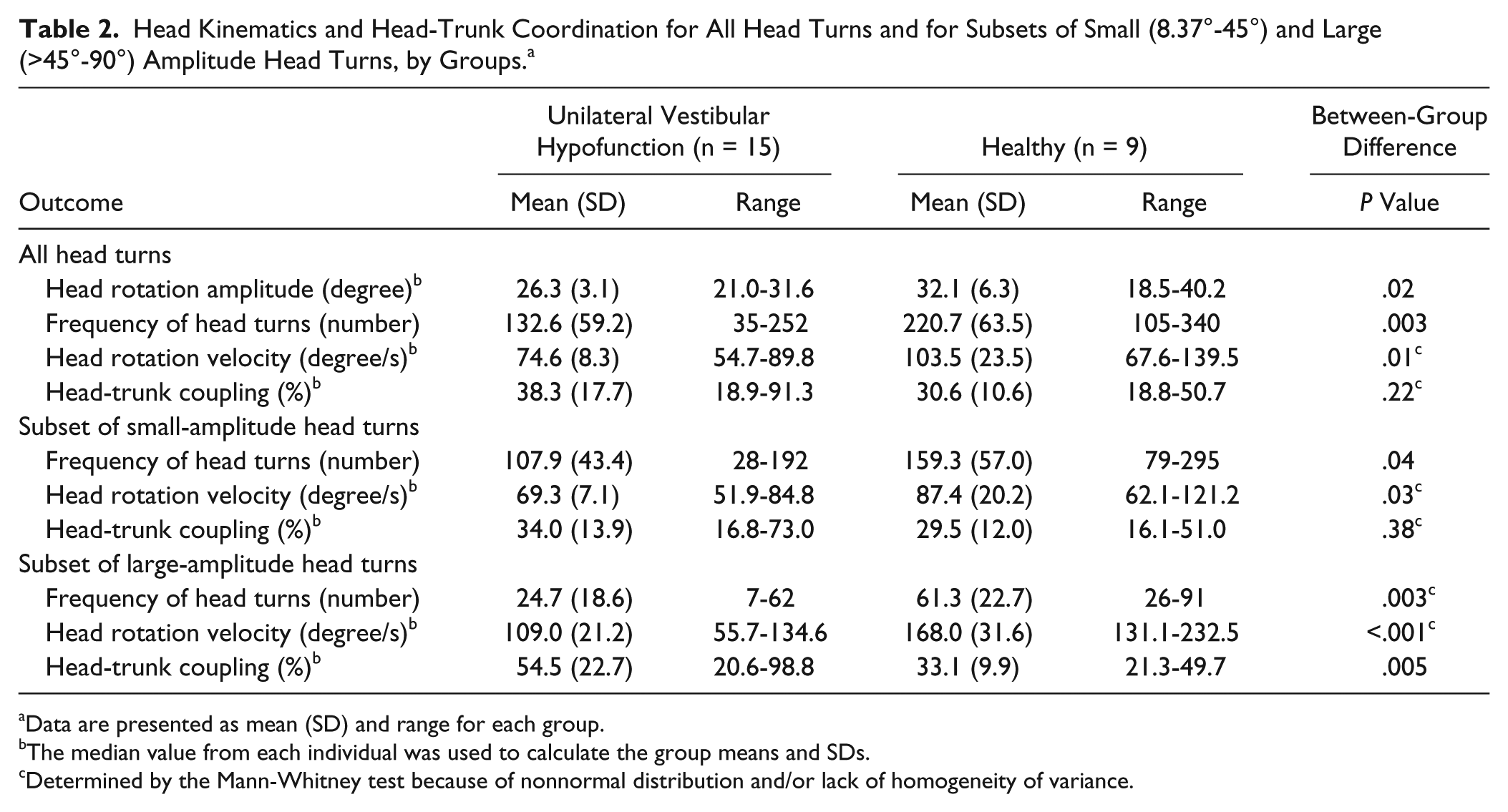
Data are presented as mean (SD) and range for each group.
The median value from each individual was used to calculate the group means and SDs.
Determined by the Mann-Whitney test because of nonnormal distribution and/or lack of homogeneity of variance.
When subsets of head turns were considered (Table 2), the pattern of results for small head turns was similar to the main findings; that is, participants with UVH had significantly reduced head turn frequency (
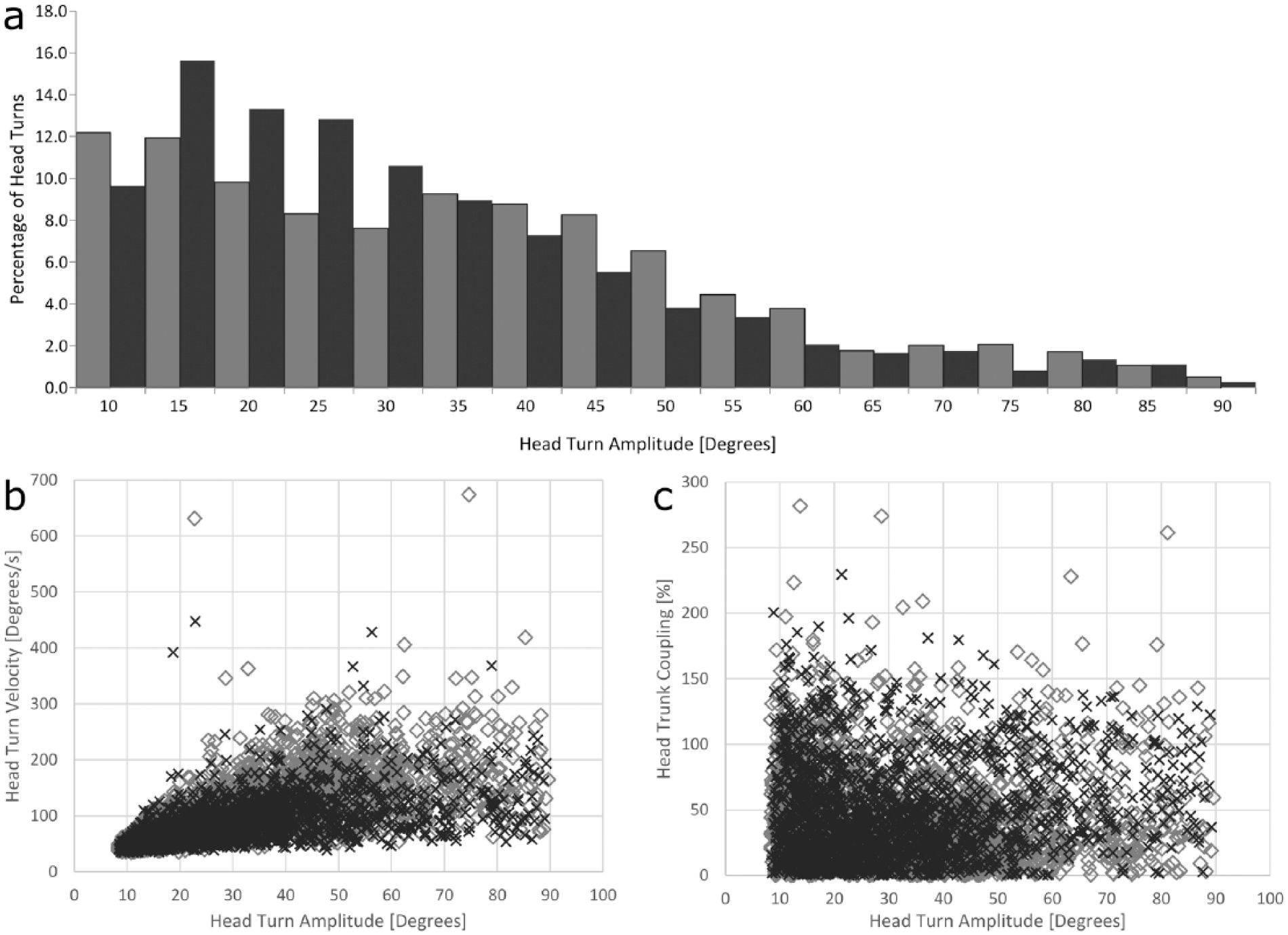
(A) The percentage of head turns (ie, head turn frequency), (B) head turn velocity, and (C) head-trunk coordination, as a function of head turn amplitude, in individuals with and without unilateral vestibular hypofunction (UVH). Data for all turns by all individuals are presented, with black columns or crosses denoting participants with UVH (15 individuals made 1989 head turns), and gray columns or diamonds denoting healthy individuals (9 individuals made 1986 head turns).
Discussion
We utilized wearable sensors to examine head and trunk kinematics during a 10-minute bout of unconstrained ambulation in a small sample of people with and without UVH. In support of our a priori hypothesis, we demonstrated that individuals 6 weeks after resection of vestibular schwannoma had reduced amplitude, frequency, and velocity of head movements compared with individuals with normal, symmetrical vestibular function. Additionally, individuals with UVH showed increased head trunk coupling for large head turns and a greater frequency of small-amplitude head turns. Unlike prior findings, gait speed during community ambulation appeared preserved, with both groups completing the community ambulation routes in a similar amount of time.8,16 Ideally, following resection of a vestibular schwannoma, individuals will independently engage in head and body movements that provoke gaze and postural instability. Exposing the brain to such error signals appears to promote neural adaptation that restores gaze and postural stability function toward premorbid levels. Unfortunately, our results along with those of prior studies, 8 suggest that individuals constrain their head movements after surgery, though there was no differential effect of lesion side on head turn behavior following UVH. Such findings call into question whether or not the dosage (frequency, amplitude, and intensity [velocity]) of head movements is sufficient to optimize recovery from such a peripheral vestibular lesion.
Alterations in Purposeful Head Movements Represent a Novel Example of Persistent Disability Following Vestibular Schwannoma Surgery
Our participants were tested at 6 weeks after surgery during what is considered the “subacute” period. At this time point, we hypothesized and confirmed that disability was present, using measures of gaze stability, dizziness handicap, and self-report of participation in daily activities requiring vestibular input. 14 Such findings are consistent with previous cross-sectional and prospective experimental research examining dizziness handicap, dynamic gait stability, dynamic visual acuity, and posturography outcomes, 17 which show that deficits do not plateau until at least 6 months after surgery, and in some instances, disability may persist for up to 10 years following surgery.18,19 Although improvements in the above-mentioned measures of disability appear to improve during controlled intervention studies,5,6 the use of head kinematics and head-trunk coordination during uncontrolled ambulation is a novel outcome.
Our participants demonstrated variable functional recovery by 6 weeks post–UVH surgery. 20 They appeared to achieve adequate gait speed during a short bout of community ambulation, unlike prior findings examining standardized tests of gait and balance 9 or performance of daily living activities. 8 Nonetheless, there were pronounced behavioral adaptations in head and trunk coordination during community ambulation, with people with UVH constraining their head movements relatively symmetrically and in comparison to those without UVH. Reductions in head-trunk kinematics regardless of direction may represent coordination of movement to maintain symmetry, yet reflect the capabilities of the more impaired side. Head movement coordination and head-trunk coupling alterations appear dependent on head movement demands, given that the magnitude of differences between those with and without UVH increased when large-amplitude head turns were analyzed individually. The greater disparity in head rotation velocity and head-trunk decoupling between those with UVH and healthy individuals during large compared with small head turns along with the higher number of small head turns made by people with UVH suggests that those with UVH constrain movements that are more likely to provoke oscillopsia and postural instability associated with vestibular deficiency. 21
If Vestibular Rehabilitation Is Prescribed, What Dosage Is Needed to Optimize Recovery?
Prior research has characterized several key aspects of efficacious gaze stability training in individuals with acute vestibular hypofunction. One key aspect of gaze stability training is that eye and head movements must be initiated with adequate acceleration and sustained with sufficient velocity to reproduce those movements seen in typical daily activities.22-25 Such data suggest that head movements should be of a sufficient velocity to elicit position or velocity error signals to drive adaptation and/or substitution with saccadic corrections.26,27 Another key aspect of gaze stability training is that the frequency of head movements must occur often enough to drive vestibulo-ocular functional recovery through cerebellum-mediated neuroplastic changes.2,5,28 Finally, the daily volume of training-related head turns necessary to drive gaze stability in persons with UVH may be as high as 2700 repetitions per day (conservatively calculated as 1 Hz movement [at least 1 head turn per second] × 15 minutes × 5 sets per day = 2700 repetitions). 4 Our data clearly show that individuals with UVH who have not received formal rehabilitation reduced the frequency of head turns by an average of 40% compared with persons with symmetric vestibular function. Additionally, those with UVH appear to prefer small-amplitude yaw plane rotations compared with their healthy counterparts (81% vs 72%), where high-velocity movement can be minimized. These observations most likely represent compensatory mechanisms to limit symptom provocation acutely but may have the longer-term consequence of failing to stimulate physiological adaptation of gaze and postural stabilization mechanisms.
Limitations and Directions for Future Research
Although our use of wearable sensors to document persistent head kinematic alterations at 6 weeks after surgery is novel, the findings from this study should be interpreted with caution. Because this was the first study using wearable sensors to quantify head-trunk coordination of people with surgically induced UVH relative to those with normal, symmetrical vestibular function during community ambulation, we chose to only characterize yaw plane rotations of the head. Given the ubiquity of inertial measurement units available today, future research should seek to characterize multiplanar movement in free-living environments across a larger spectrum of individuals with vestibular hypofunction as well as describe the dose-response relationship between head movements in these environments and clinical outcomes. Future studies should also investigate whether improvements in head movements are associated with improvements in dizziness (measured with the Dizziness Handicap Inventory) and activities of daily living (measured with the Vestibular Activities and Participation Measure) and how the time course of symptom reduction affects purposeful head movements during community ambulation.
Another limitation to this study is that we limited our outcome measures to spatial components of head and trunk behaviors. Future investigations should include temporal components of these behaviors to better ascertain the severity of disability associated with these disorders. A further limitation was the lack of constraint on participant behaviors during the 10-minute bout of community ambulation, potentially affecting internal validity. However, given that the primary objective of this study was to assess participants in an ecologically valid manner, we felt that it was of paramount importance to not attempt to control the variability of performance between individuals. There was heterogeneity in performance19,29 within our small sample, although both groups were of similar age. This heterogeneity is likely a result of various factors that can influence recovery following vestibular schwannoma resection, including but not limited to preoperative physical activity and function, tumor size, and extent and rapidity of vestibular compensation following surgery. 30
An unexplored use of wearable sensors would be to measure the amplitude and velocity of head movements during prescribed gaze stabilization exercises in vestibular rehabilitation. Such data would provide insights into the quality of the exercises as well as document patient adherence to prescribed dosages. Head kinematic outcomes obtained using wearable sensors may also be used in vestibular rehabilitation studies to investigate the integrity of interventions and strengthen the internal validity of such studies.
Conclusion
This study demonstrated that despite walking at an adequate speed, individuals with UVH made fewer, smaller, and slower head movements than healthy individuals and did not decouple their head movement relative to their trunk when required to make larger purposeful head turns during community ambulation. If this change in strategy results in increased handicap, this is worthy of future research as are the potential resultant needs for post–tumor resection vestibular rehabilitation. 4
Footnotes
Acknowledgements
Sherry Hogge assisted in recruiting individuals with vestibular schwannoma. Ethan Beseris, Alicia Dibble, Kirsten Gorski, Jaclyn Hill, and Jane Saviers-Steiger assisted with data collection.
Authors’ Note
Contributions of authors: Substantial contributions to conception or design of the work, or the acquisition, analysis, or interpretation of data for the work (SSP, LED, RGW, MEL); drafting of the work or revising it critically for important intellectual content (SSP, LED, CS, RKG, MEL); and final approval of the version to be published (SSP, LED, RGW, CS, RKG, MEL).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a US Army Advanced Medical Technology Initiative Grant (2015-2016; principal investigators Drs Dibble and Lester). LED also received personal fees from the National Multiple Sclerosis Society.
